The Effect of Athletes’ Probiotic Intake May Depend on Protein and Dietary Fiber Intake
Abstract
1. Introduction
2. Materials and Methods
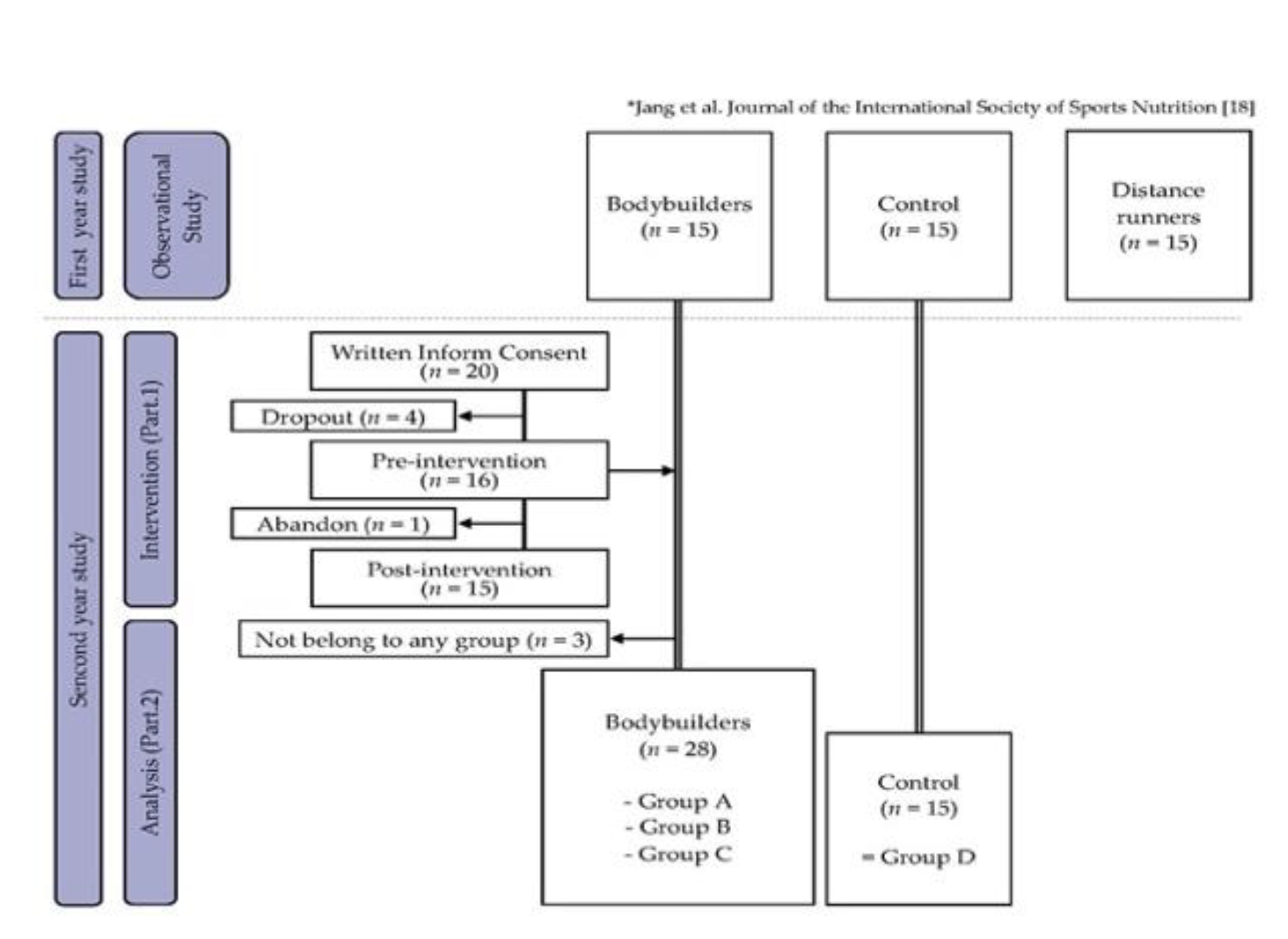
2.1. Study Design
2.2. Recruitment of Participants and Supplementary Information
- -
- Probiotic group: Capsule consisting of 1012 CFU of each of the following species: L. acidophilus, L. casei, L. helveticus, and Bifidobacterium bifidum;
- -
- Placebo group: Capsule consisting of corn starch.
2.3. Gut Microbiota
2.4. SCFA
2.5. Nutrient Intake Analysis
- Group 1. Bodybuilder–high protein and meager dietary fiber (n = 12);
- Group 2. Bodybuilder–high protein and proper dietary fiber (n = 10);
- Group 3. Bodybuilder–proper protein and meager dietary fiber (n = 6);
- Group 4. Sedentary (n = 15).
2.6. Statistical Analysis
3. Results
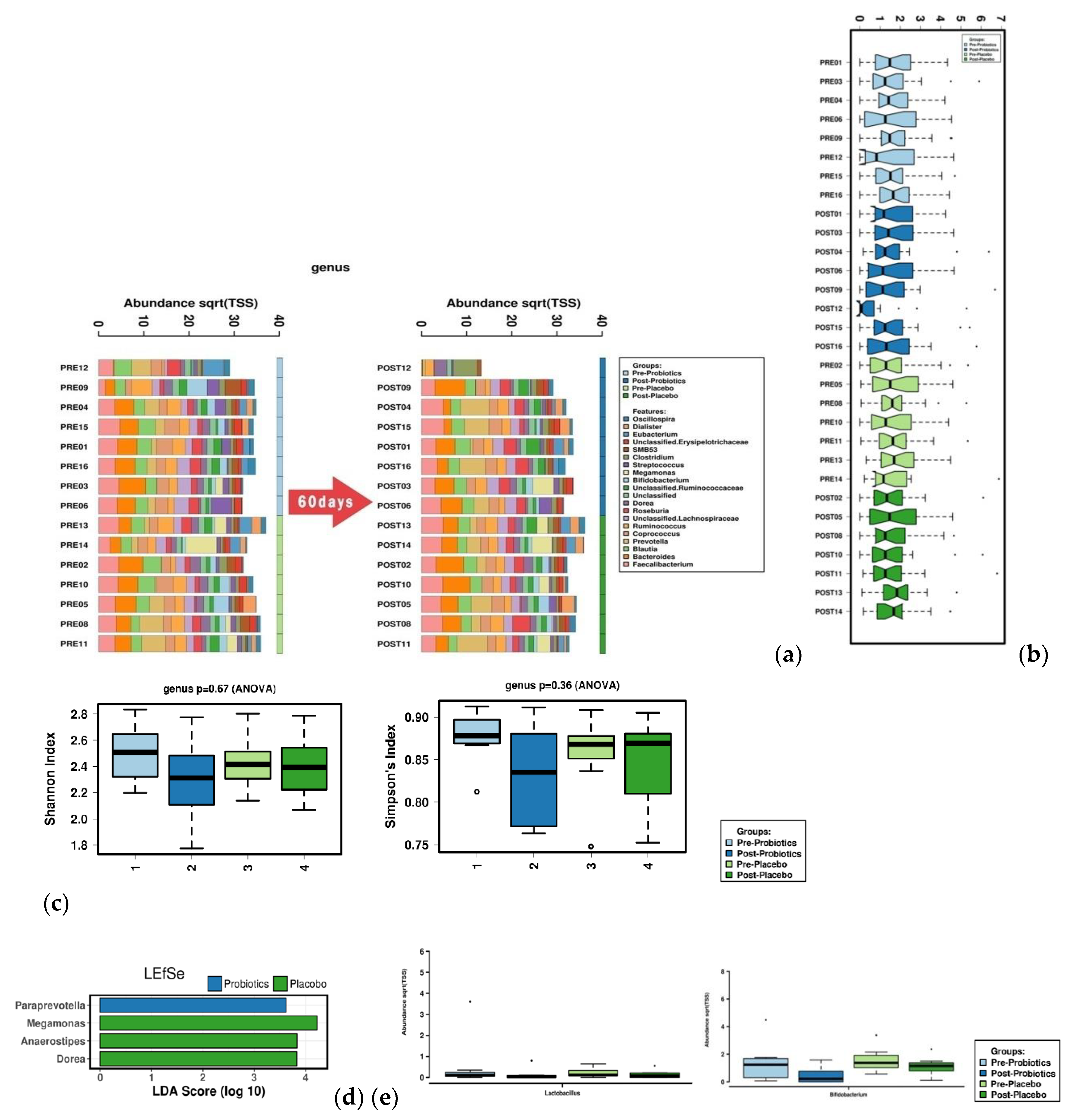
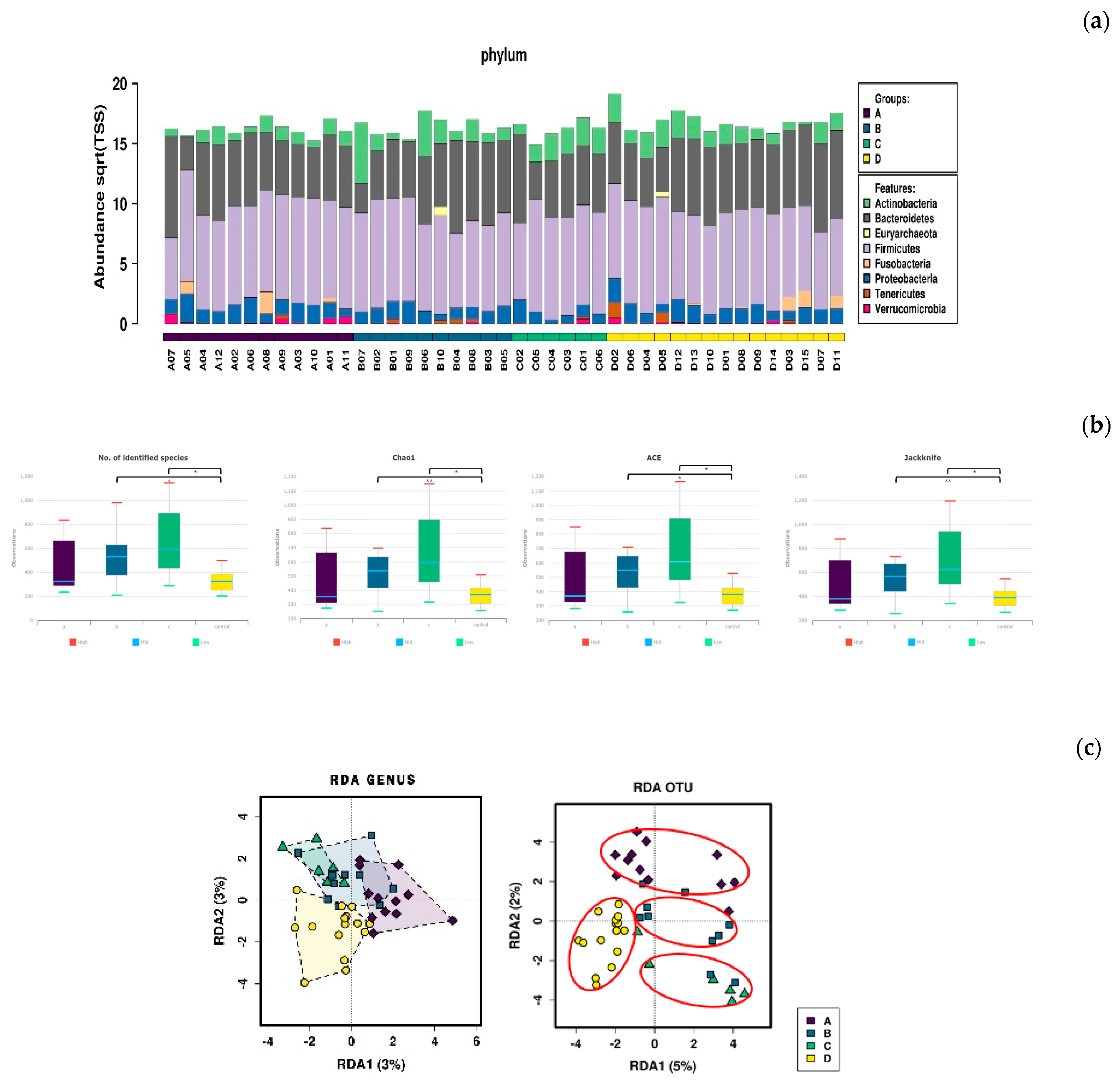
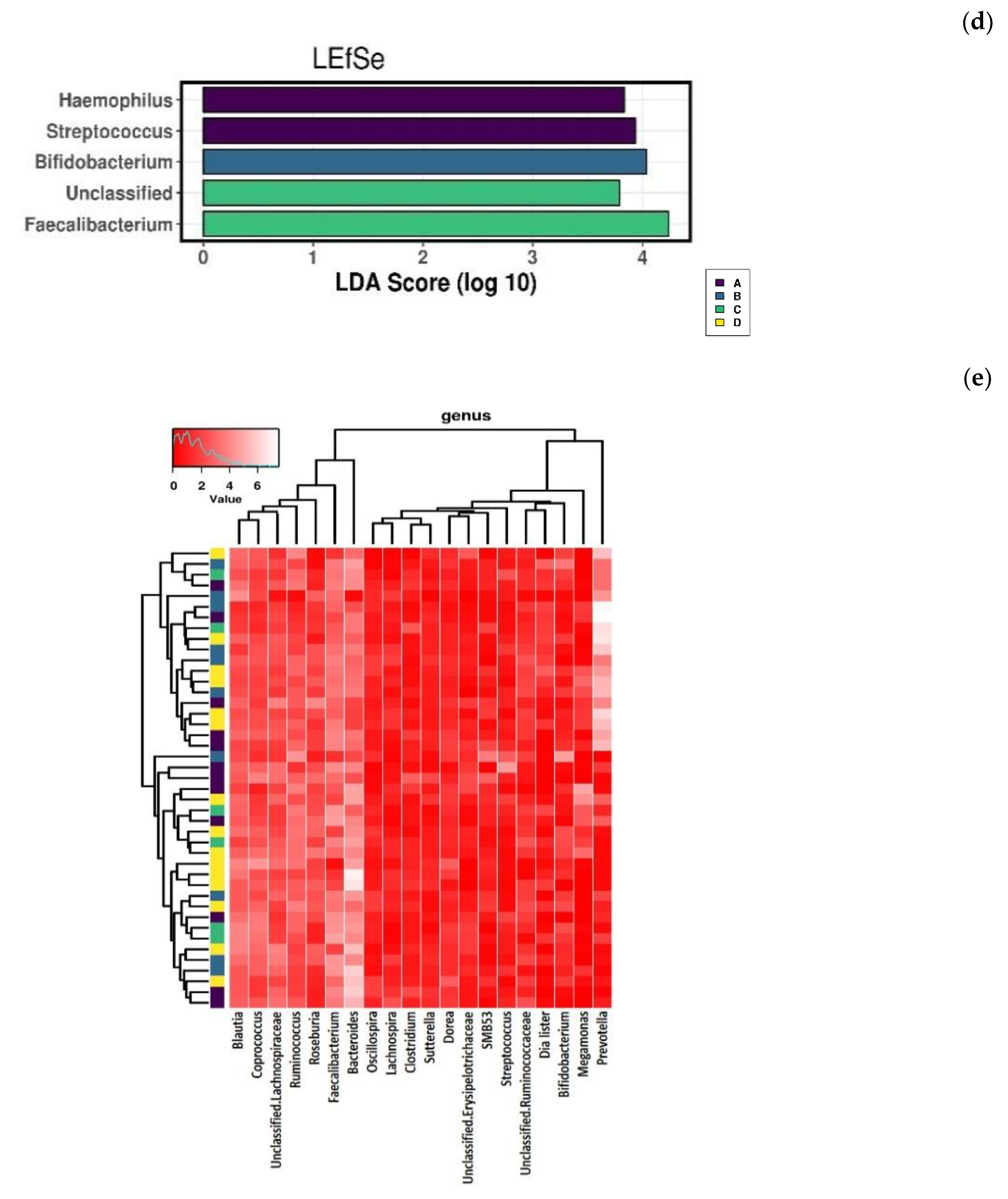
3.1. Microbiome Analysis (Part 1)
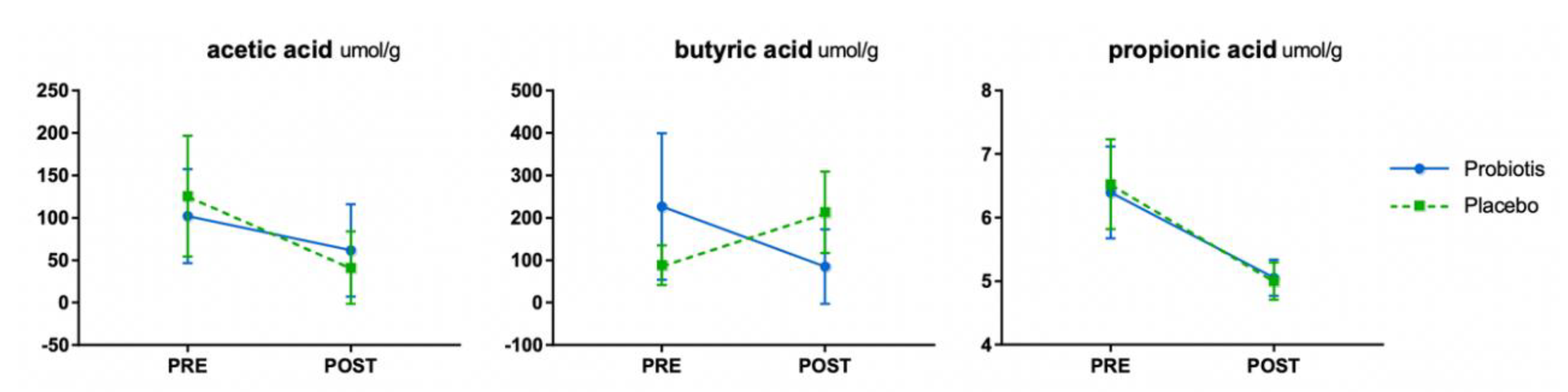
3.2. SCFAs (Part 1)
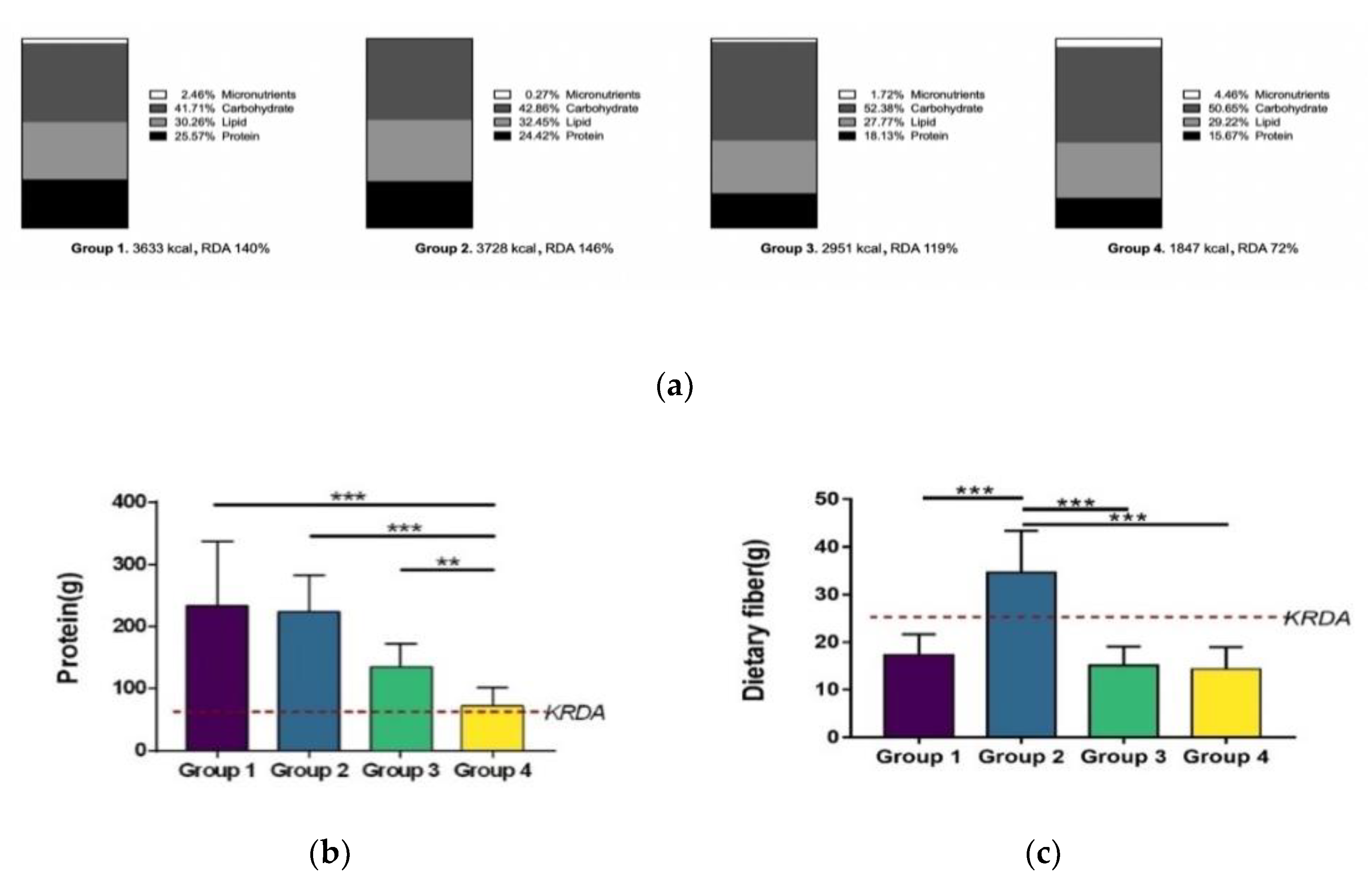
3.3. Nutritional Intake Analysis (Part 2)
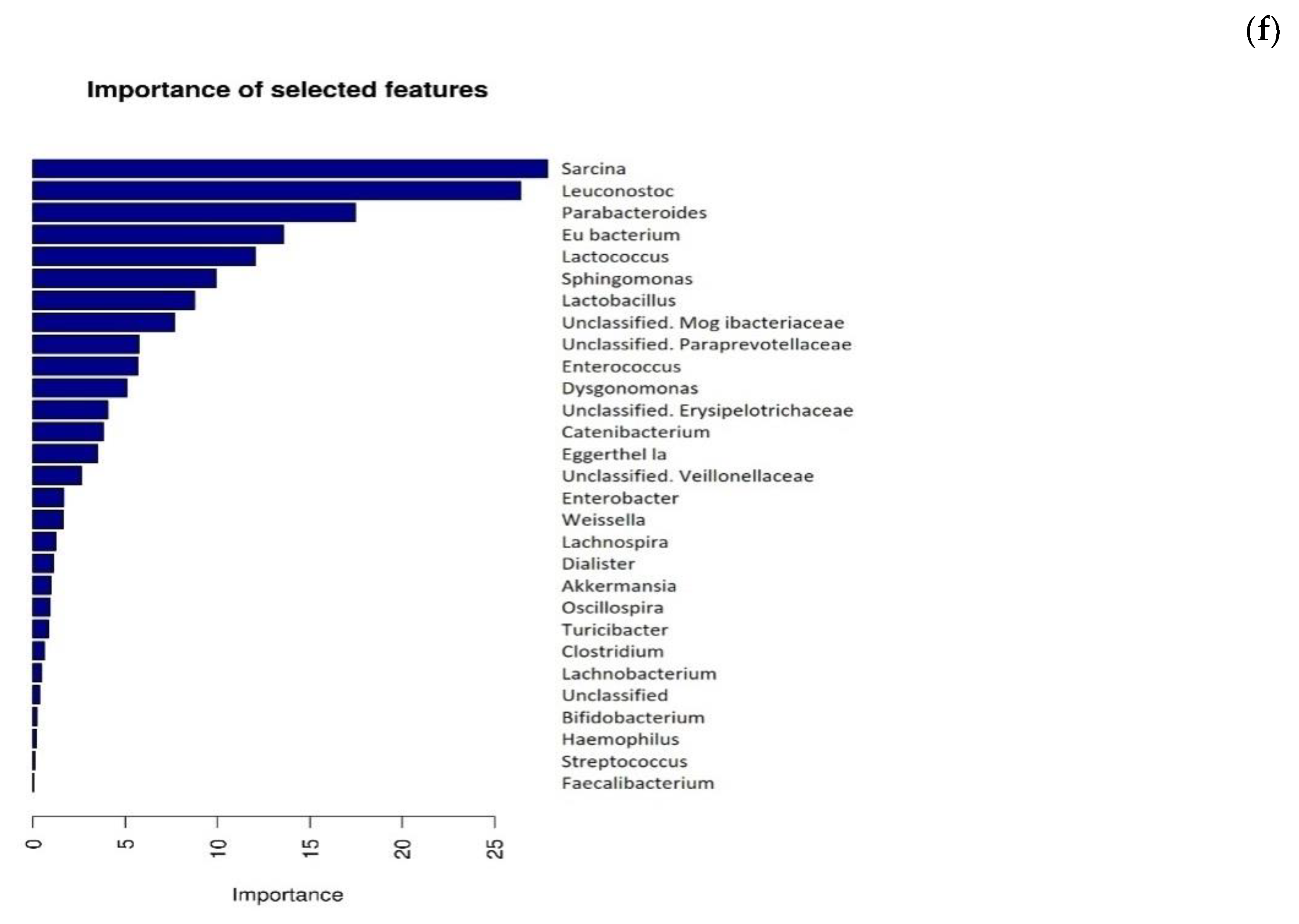
3.4. Microbiome Analysis (Part 2)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Areta, J.L.; Burke, L.M.; Ross, M.L.; Camera, D.M.; West, D.W.; Broad, E.M.; Jeacocke, N.A.; Moore, D.R.; Stellingwerff, T.; Phillips, S.M.; et al. Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J. Physiol. 2013, 591, 2319–2331. [Google Scholar] [CrossRef] [PubMed]
- Hawley, J.A.; Hargreaves, M.; Joyner, M.J.; Zierath, J.R. Integrative biology of exercise. Cell 2014, 159, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R. Nutrition to support recovery from endurance exercise: Optimal carbohydrate and protein replacement. Curr. Sports Med. Rep. 2015, 14, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Neville, B.A.; Forster, S.C.; Lawley, T.D. Commensal Koch’s postulates: Establishing causation in human microbiota research. Curr. Opin. Microbiol. 2018, 42, 47–52. [Google Scholar] [CrossRef]
- Browne, H.P.; Forster, S.C.; Anonye, B.O.; Kumar, N.; Neville, B.A.; Stares, M.D.; Lawley, T.D. Culturing of ‘unculturable’human microbiota reveals novel taxa and extensive sporulation. Nature 2016, 533, 543–546. [Google Scholar] [CrossRef]
- Bermon, S.; Petriz, B.; Kajeniene, A.; Prestes, J.; Castell, L.; Franco, O.L. The microbiota: An exercise immunology perspective. ExercImmunol. Rev. 2015, 21, 70–79. [Google Scholar]
- O’Sullivan, O.; Cronin, O.; Clarke, S.F.; Murphy, E.F.; Molloy, M.G.; Shanahan, F.; Cotter, P.D. Exercise and the microbiota. Gut Microbes 2015, 6, 131–136. [Google Scholar] [CrossRef]
- Campbell, S.C.; Wisniewski, P.J. Exercise is a novel promoter of intestinal health and microbial diversity. Exerc. Sport Sci. Rev. 2017, 45, 41–47. [Google Scholar] [CrossRef]
- Monda, V.; Villano, I.; Messina, A.; Valenzano, A.; Esposito, T.; Moscatelli, F.; Messina, G.; Viggiano, A.; Cibelli, G.; Chieffi, S.; et al. Exercise modifies the gut microbiota with positive health effects. Oxid. Med. Cell. Longev. 2017, 5, 1–8. [Google Scholar] [CrossRef]
- Stewart, C.J.; Nelson, A.; Campbell, M.D.; Walker, M.; Stevenson, E.J.; Shaw, J.A.; Cummings, S.P.; West, D.J. Gut microbiota of Type 1 diabetes patients with good glycaemic control and high physical fitness is similar to people without diabetes: An observational study. Diabet. Med. 2017, 34, 127–134. [Google Scholar] [CrossRef]
- Shin, N.R.; Lee, J.C.; Lee, H.Y.; Kim, M.S.; Whon, T.W.; Lee, M.S.; Bae, J.W. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut 2014, 63, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Shi, Y.; Wiklund, P.; Tan, X.; Wu, N.; Zhang, X.; Tikkanen, O.; Zhang, C.; Munukka, E.; Cheng, S.; et al. The association between cardiorespiratory fitness and gut microbiota composition in premenopausal women. Nutrients 2017, 9, 792. [Google Scholar] [CrossRef] [PubMed]
- Scheiman, J.; Luber, J.M.; Chavkin, T.A.; MacDonald, T.; Tung, A.; Pham, L.D.; Wibowo, M.C.; Wurth, R.C.; Punthambaker, S.; Tierney, B.T.; et al. Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nat. Med. 2019, 25, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Barton, W.; Penney, N.C.; Cronin, O.; Garcia-Perez, I.; Molloy, M.G.; Holmes, E.; Shanahan, F.; Cotter, P.D.; O’Sullivan, O. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut 2018, 67, 625–633. [Google Scholar] [CrossRef]
- Petriz, B.A.; Castro, A.P.; Almeida, J.A.; Gomes, C.P.; Fernandes, G.R.; Kruger, R.H.; Pereira, R.W.; Franco, O.L. Exercise induction of gut microbiota modifications in obese, non-obese and hypertensive rats. BMC Genom. 2014, 15, 511. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.V.; Leite, G.; Resende, A.; Blachier, F.; AH Jr, L. Exercise, nutrition and gut microbiota: Possible links and consequences. Int. J. Sports Exerc. Med. 2017, 69. [Google Scholar] [CrossRef]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef]
- Jang, L.G.; Choi, G.; Kim, S.W.; Kim, B.Y.; Lee, S.; Park, H. The combination of sport and sport-specific diet is associated with characteristics of gut microbiota: An observational study. J. Int. Soc. Sports Nutr. 2019, 16, 21. [Google Scholar] [CrossRef] [PubMed]
- Wosinska, L.; Cotter, P.D.; O’Sullivan, O.; Guinane, C. The Potential Impact of Probiotics on the Gut Microbiome of Athletes. Nutrients 2019, 11, 2270. [Google Scholar] [CrossRef]
- Yun, B.; Oh, S.; Griffiths, M.W. Lactobacillus acidophilus modulates the virulence of Clostridium difficile. J. Dairy Sci. 2014, 97, 4745–4758. [Google Scholar] [CrossRef]
- Huang, I.F.; Lin, I.C.; Liu, P.F.; Cheng, M.F.; Liu, Y.C.; Hsieh, Y.D.; Chen, J.J.; Chen, C.L.; Chang, H.W.; Shu, C.W. Lactobacillus acidophilus attenuates Salmonella-induced intestinal inflammation via TGF-β signaling. BMC Microbiol. 2015, 15, 203. [Google Scholar] [CrossRef] [PubMed]
- Kapila, S.; Sinha, V.P. Antioxidative and hypocholesterolemic effect of Lactobacillus caseisspcasei (biodefensive properties of lactobacilli). Indian J. Med. Sci. 2006, 60, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Amdekar, S.; Roy, P.; Singh, V.; Kumar, A.; Singh, R.; Sharma, P. Anti-inflammatory activity of lactobacillus on carrageenan-induced paw edema in male wistar rats. Int. J. Inflamm. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Amdekar, S.; Singh, V.; Kumar, A.; Sharma, P.; Singh, R. Lactobacillus casei and Lactobacillus acidophilus regulate inflammatory pathway and improve antioxidant status in collagen-induced arthritic rats. J. Interferon Cytokine Res. 2013, 33, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Schwiertz, A.; Taras, D.; Schäfer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in lean and overweight healthy subjects. Obesity 2010, 18, 190–195. [Google Scholar] [CrossRef]
- Gargari, G.; Taverniti, V.; Balzaretti, S.; Ferrario, C.; Gardana, C.; Simonetti, P.; Guglielmetti, S. Consumption of a Bifidobacterium bifidum strain for 4 weeks modulates dominant intestinal bacterial taxa and fecal butyrate in healthy adults. Appl. Environ. Microbiol. 2016, 82, 5850–5859. [Google Scholar] [CrossRef]
- Evans, C.C.; LePard, K.J.; Kwak, J.W.; Stancukas, M.C.; Laskowski, S.; Dougherty, J.; Moulton, L.; Glawe, A.; Wang, Y.; Leone, V.; et al. Exercise prevents weight gain and alters the gut microbiota in a mouse model of high fat diet-induced obesity. PLoS ONE 2014, 9, e92193. [Google Scholar] [CrossRef]
- Campbell, S.C.; Wisniewski, P.J.; Noji, M.; McGuinness, L.R.; Häggblom, M.M.; Lightfoot, S.A.; Joseph, L.B.; Kerkhof, L.J. The effect of diet and exercise on intestinal integrity and microbial diversity in mice. PLoS ONE 2016, 11, e0150502. [Google Scholar] [CrossRef]
- Deehan, E.C.; Yang, C.; Perez-Muñoz, M.E.; Nguyen, N.K.; Cheng, C.C.; Triador, L.; Zhang, Z.; Bakal, J.A.; Walter, J. Precision microbiome modulation with discrete dietary fiber structures directs short-chain fatty acid production. Cell Host Microbe 2020, 27. [Google Scholar] [CrossRef]
- Kostovcikova, K.; Coufal, S.; Galanova, N.; Fajstova, A.; Hudcovic, T.; Kostovcik, M.; Prochazkova, P.; Jiraskova Zakostelska, Z.; Cermakova, M.; Sediva, B.; et al. Diet rich in animal protein promotes pro-inflammatory macrophage response and exacerbates colitis in mice. Front. Immunol. 2019, 10, 919. [Google Scholar] [CrossRef]
| Probiotics (n = 8) | Placebo (n = 7) | |
|---|---|---|
| Age (year) | 26.50 ± 5.01 | 27.14 ± 5.93 |
| Height (m) | 1.75 ± 0.03 | 1.77 ± 0.05 |
| Weigh (kg) | 90.50 ± 9.34 | 83.58 ± 11.64 |
| BMI (%) | 29.71 ± 3.05 | 26.79 ± 3.32 |
| Group 1 (n = 12) | Group 2 (n = 10) | Group 3 (n = 6) | Group 4 (n = 15) | |
|---|---|---|---|---|
| Age (years) | 25.00 ± 3.05 | 25.90 ± 4.09 | 28.67 ± 6.12 | 26.27 ± 2.05 |
| Height (m) | 1.75 ± 0.05 | 1.75 ± 0.06 | 1.75 ± 0.04 | 1.76 ± 0.06 |
| Weight (kg) | 87.75 ± 9.42 | 87.05 ± 7.92 | 80.85 ± 11.56 | 79.82 ± 11.04 |
| BMI (%) | 28.52 ± 2.66 | 28.44 ± 2.55 | 26.51 ± 4.27 | 25.86 ± 4.18 |
| Fat (kg) | 11.71 ± 3.81 | 11.42 ± 4.41 | 11.27 ± 4.95 | 19.44 ± 7.90 |
| Lean body mass (kg) | 71.46 ± 9.37 | 72.38 ± 7.24 | 65.32 ± 8.24 | 56.53 ± 4.57 |
| Percentage fat (%) | 13.80 ± 4.68 | 12.92 ± 4.46 | 13.85 ± 4.93 | 23.95 ± 6.93 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Son, J.; Jang, L.-G.; Kim, B.-Y.; Lee, S.; Park, H. The Effect of Athletes’ Probiotic Intake May Depend on Protein and Dietary Fiber Intake. Nutrients 2020, 12, 2947. https://doi.org/10.3390/nu12102947
Son J, Jang L-G, Kim B-Y, Lee S, Park H. The Effect of Athletes’ Probiotic Intake May Depend on Protein and Dietary Fiber Intake. Nutrients. 2020; 12(10):2947. https://doi.org/10.3390/nu12102947
Chicago/Turabian StyleSon, Joy, Lae-Guen Jang, Byung-Yong Kim, Sunghee Lee, and Hyon Park. 2020. "The Effect of Athletes’ Probiotic Intake May Depend on Protein and Dietary Fiber Intake" Nutrients 12, no. 10: 2947. https://doi.org/10.3390/nu12102947
APA StyleSon, J., Jang, L.-G., Kim, B.-Y., Lee, S., & Park, H. (2020). The Effect of Athletes’ Probiotic Intake May Depend on Protein and Dietary Fiber Intake. Nutrients, 12(10), 2947. https://doi.org/10.3390/nu12102947





