Could Omega 3 Fatty Acids Preserve Muscle Health in Rheumatoid Arthritis?
Abstract
1. Introduction
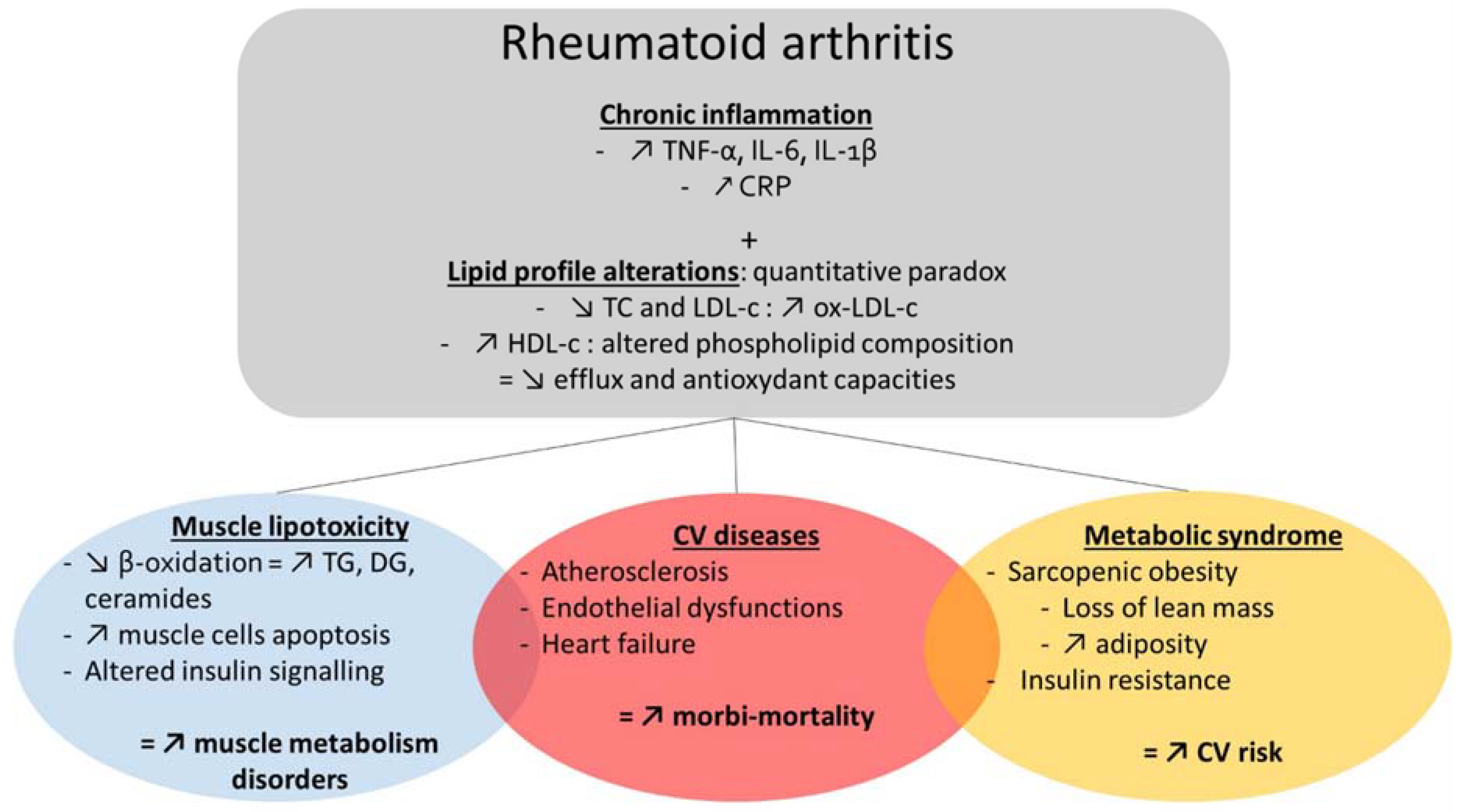
2. Cardiometabolic Comorbidities in Rheumatic Diseases
2.1. Cardiometabolic Comorbidities in RA
2.2. Cardiometabolic Comorbidities in Other Rheumatic Diseases
3. Alterations of Lipid Profile: A Mutual Denominator of Comorbidities in RA
3.1. Altered Lipidome in RA
3.2. Metabolic Disorders and Sarcopenia in RA
3.2.1. Inflammation
3.2.2. Insulin Resistance
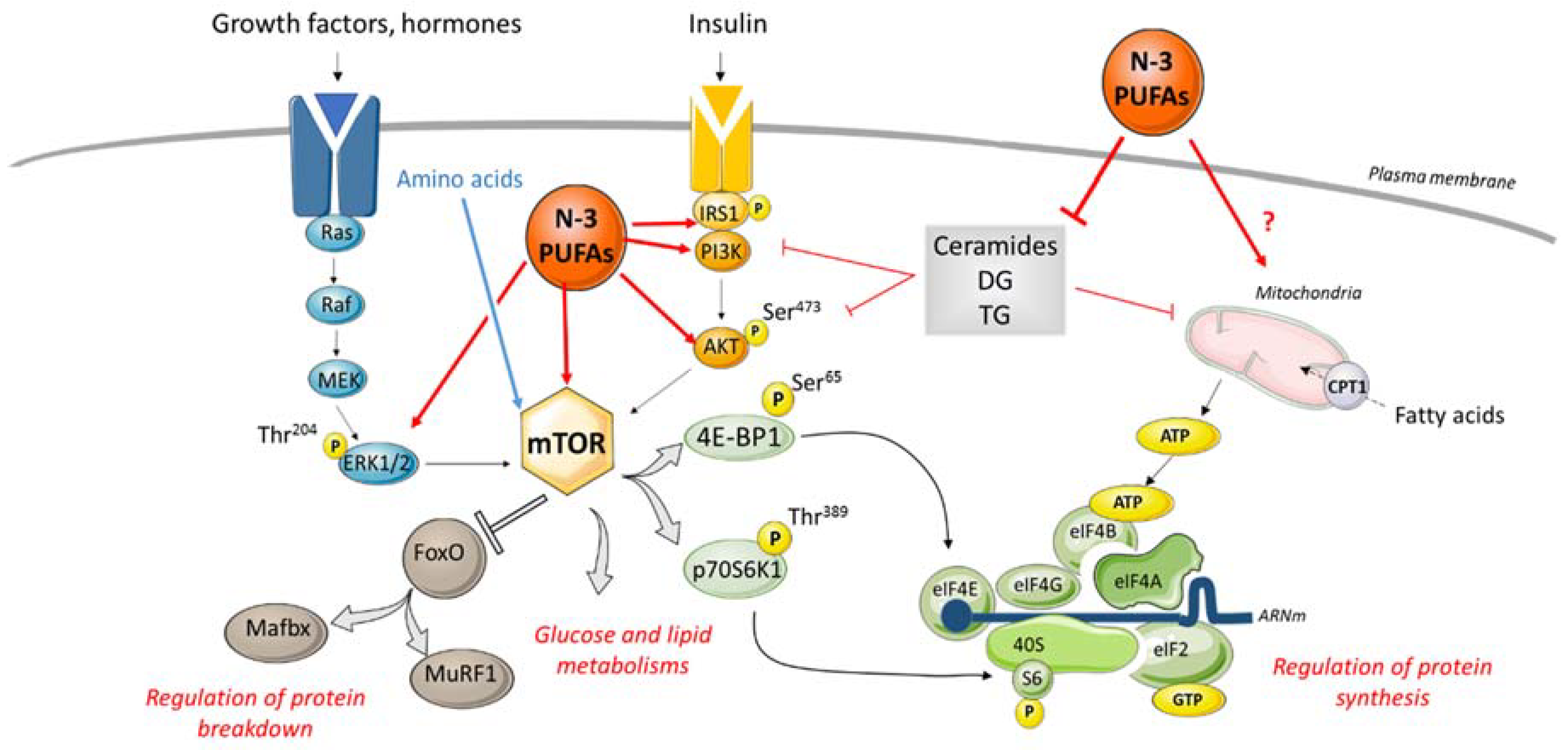
3.2.3. Lipotoxicity
3.2.4. Mitochondrial Dysfunction
4. Consuming Omega 3 to Improve Muscle Health in RA?
4.1. Omega 3 and Inflammation in RA
4.2. Omega 3 and CV Risk
4.2.1. Effects on Lipid Profile
4.2.2. Effects on CVD
4.3. Omega 3 and Muscle Metabolism
5. Conclusions
6. Perspectives: Multimodal Approaches
Author Contributions
Funding
Conflicts of Interest
References
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038. [Google Scholar] [CrossRef]
- Abhishek, A.; Nakafero, G.; Kuo, C.-F.; Mallen, C.; Zhang, W.; Grainge, M.J.; Doherty, M. Rheumatoid arthritis and excess mortality: Down but not out. A primary care cohort study using data from Clinical Practice Research Datalink. Rheumatology 2018, 57, 977–981. [Google Scholar] [CrossRef]
- Paul, B.J.; Kandy, H.I.; Krishnan, V. Pre-rheumatoid arthritis and its prevention. Eur. J. Rheumatol. 2017, 4, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.F.; Mostoufi-Moab, S.; Long, J.; Zemel, B.; Ibrahim, S.; Taratuta, E.; Leonard, M.B. Intramuscular Fat Accumulation and Associations with Body Composition, Strength and Physical Functioning in Rheumatoid Arthritis. Arthritis Care Res. 2018, 70, 1727–1734. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 Fatty Acids and Inflammatory Processes. Nutrients 2010, 2, 355–374. [Google Scholar] [CrossRef]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Functional Roles of Fatty Acids and Their Effects on Human Health. J. Parenter. Enter. Nutr. 2015, 39, 18S–32S. [Google Scholar] [CrossRef]
- James, M.; Proudman, S.; Cleland, L. Fish oil and rheumatoid arthritis: Past, present and future. Proc. Nutr. Soc. 2010, 69, 316–323. [Google Scholar] [CrossRef]
- Pinel, A.; Rigaudière, J.-P.; Laillet, B.; Pouyet, C.; Malpuech-Brugère, C.; Prip-Buus, C.; Morio, B.; Capel, F. N—3PUFA differentially modulate palmitate-induced lipotoxicity through alterations of its metabolism in C2C12 muscle cells. Biochim. Biophys. Acta 2016, 1861, 12–20. [Google Scholar] [CrossRef]
- Jeromson, S.; Gallagher, I.; Galloway, S.; Hamilton, D. Omega-3 Fatty Acids and Skeletal Muscle Health. Mar. Drugs 2015, 13, 6977–7004. [Google Scholar] [CrossRef]
- Tournadre, A.; Mathieu, S.; Soubrier, M. Managing cardiovascular risk in patients with inflammatory arthritis: Practical considerations. Ther. Adv. Musculoskelet. Dis. 2016, 8, 180–191. [Google Scholar] [CrossRef] [PubMed]
- Aviña-Zubieta, J.A.; Choi, H.K.; Sadatsafavi, M.; Etminan, M.; Esdaile, J.M.; Lacaille, D. Risk of cardiovascular mortality in patients with rheumatoid arthritis: A meta-analysis of observational studies. Arthritis Care Res. 2008, 59, 1690–1697. [Google Scholar] [CrossRef]
- Chung, C.P.; Oeser, A.; Solus, J.F.; Avalos, I.; Gebretsadik, T.; Shintani, A.; Raggi, P.; Sokka, T.; Pincus, T.; Stein, C.M. Prevalence of the metabolic syndrome is increased in rheumatoid arthritis and is associated with coronary atherosclerosis. Atherosclerosis 2008, 196, 756–763. [Google Scholar] [CrossRef]
- Di Minno, M.N.D.; Ambrosino, P.; Lupoli, R.; Di Minno, A.; Tasso, M.; Peluso, R.; Tremoli, E. Clinical assessment of endothelial function in patients with rheumatoid arthritis: A meta-analysis of literature studies. Eur. J. Intern. Med. 2015, 26, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.J.L.; van Halm, V.P.; Voskuyl, A.E.; Smulders, Y.M.; Boers, M.; Lems, W.F.; Visser, M.; Stehouwer, C.D.A.; Dekker, J.M.; Nijpels, G.; et al. Does rheumatoid arthritis equal diabetes mellitus as an independent risk factor for cardiovascular disease? A prospective study. Arthritis Rheum. 2009, 61, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Giraud, C.; Tournadre, A.; Pereira, B.; Dutheil, F.; Soubrier, M.; Lhomme, M.; Kontush, A.; Sébédio, J.-L.; Capel, F. Alterations of HDL particle phospholipid composition and role of inflammation in rheumatoid arthritis. J. Physiol. Biochem. 2019, 75, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Nurmohamed, M.T.; Heslinga, M.; Kitas, G.D. Cardiovascular comorbidity in rheumatic diseases. Nat. Rev. Rheumatol. 2015, 11, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, L.; Delzell, E.; Muntner, P.; Hillegass, W.B.; Safford, M.M.; Millan, I.Y.N.; Crowson, C.S.; Curtis, J.R. The association between inflammatory markers, serum lipids and the risk of cardiovascular events in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2014, 73, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Davidson, L.E.; Hunt, S.C.; Adams, T.D. Fitness versus adiposity in cardiovascular disease risk. Eur. J. Clin. Nutr. 2019, 73, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Giles, J.T.; Danielides, S.; Szklo, M.; Post, W.S.; Blumenthal, R.S.; Petri, M.; Schreiner, P.J.; Budoff, M.; Detrano, R.; Bathon, J.M. Insulin Resistance in Rheumatoid Arthritis: Disease-Related Indicators and Associations with the Presence and Progression of Subclinical Atherosclerosis: Insulin Resistance in RA. Arthritis Rheumatol. 2015, 67, 626–636. [Google Scholar] [CrossRef]
- Nicolau, J.; Lequerré, T.; Bacquet, H.; Vittecoq, O. Rheumatoid arthritis, insulin resistance, and diabetes. Jt. Bone Spine 2017, 84, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Giles, J.T.; Allison, M.; Blumenthal, R.S.; Post, W.; Gelber, A.C.; Petri, M.; Tracy, R.; Szklo, M.; Bathon, J.M. Abdominal adiposity in rheumatoid arthritis: Association with cardiometabolic risk factors and disease characteristics. Arthritis Rheum. 2010, 62, 3173–3182. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Hashimoto, M.; Kawahara, R.; Yamamoto, H.; Tanaka, M.; Ito, H.; Masuda, I.; Hosoda, K.; Yamamoto, W.; Uozumi, R.; et al. Non-obese visceral adiposity is associated with the risk of atherosclerosis in Japanese patients with rheumatoid arthritis: A cross-sectional study. Rheumatol. Int. 2018, 38, 1679–1689. [Google Scholar] [CrossRef] [PubMed]
- Giles, J.T.; Ling, S.M.; Ferrucci, L.; Bartlett, S.J.; Andersen, R.E.; Towns, M.; Muller, D.; Fontaine, K.R.; Bathon, J.M. Abnormal body composition phenotypes in older rheumatoid arthritis patients: Association with disease characteristics and pharmacotherapies. Arthritis Rheum. 2008, 59, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Kremers, H.M.; Nicola, P.J.; Crowson, C.S.; Ballman, K.V.; Gabriel, S.E. Prognostic importance of low body mass index in relation to cardiovascular mortality in rheumatoid arthritis. Arthritis Rheum. 2004, 50, 3450–3457. [Google Scholar] [CrossRef]
- Stavropoulos-Kalinoglou, A.; Metsios, G.S.; Koutedakis, Y.; Nevill, A.M.; Douglas, K.M.; Jamurtas, A.; van Zanten, J.J.C.S.V.; Labib, M.; Kitas, G.D. Redefining overweight and obesity in rheumatoid arthritis patients. Ann. Rheum. Dis. 2007, 66, 1316–1321. [Google Scholar] [CrossRef]
- Stavropoulos-Kalinoglou, A.; Metsios, G.S.; Koutedakis, Y.; Kitas, G.D. Obesity in rheumatoid arthritis. Rheumatology 2011, 50, 450–462. [Google Scholar] [CrossRef]
- Tournadre, A.; Vial, G.; Capel, F.; Soubrier, M.; Boirie, Y. Sarcopenia. Jt. Bone Spine 2019, 86, 309–314. [Google Scholar] [CrossRef]
- Baker, J.F.; Von Feldt, J.; Mostoufi-Moab, S.; Noaiseh, G.; Taratuta, E.; Kim, W.; Leonard, M.B. Deficits in Muscle Mass, Muscle Density, and Modified Associations with Fat in Rheumatoid Arthritis: Muscle Deficits and Modified Association with Fat in RA. Arthritis Care Res. 2014, 66, 1612–1618. [Google Scholar] [CrossRef]
- Kramer, H.R.; Fontaine, K.R.; Bathon, J.M.; Giles, J.T. Muscle density in rheumatoid arthritis: Associations with disease features and functional outcomes. Arthritis Rheum. 2012, 64, 2438–2450. [Google Scholar] [CrossRef]
- Giles, J.T.; Bartlett, S.J.; Andersen, R.E.; Fontaine, K.R.; Bathon, J.M. Association of body composition with disability in rheumatoid arthritis: Impact of appendicular fat and lean tissue mass. Arthritis Rheum. 2008, 59, 1407–1415. [Google Scholar] [CrossRef] [PubMed]
- Lusa, A.L.; Amigues, I.; Kramer, H.R.; Dam, T.-T.; Giles, J.T. Indicators of Walking Speed in Rheumatoid Arthritis: Relative Influence of Articular, Psychosocial, and Body Composition Characteristics: Walking Speed in RA. Arthritis Care Res. 2015, 67, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Lima-Martínez, M.M.; Campo, E.; Salazar, J.; Paoli, M.; Maldonado, I.; Acosta, C.; Rodney, M.; Contreras, M.; Cabrera-Rego, J.O.; Iacobellis, G. Epicardial Fat Thickness as Cardiovascular Risk Factor and Therapeutic Target in Patients with Rheumatoid Arthritis Treated with Biological and Nonbiological Therapies. Arthritis 2014, 2014, 782850. [Google Scholar] [CrossRef] [PubMed]
- Münzel, T.; Gori, T.; Bruno, R.M.; Taddei, S. Is oxidative stress a therapeutic target in cardiovascular disease? Eur. Heart J. 2010, 31, 2741–2748. [Google Scholar] [CrossRef]
- Jeanmaire, C.; Mazières, B.; Verrouil, E.; Bernard, L.; Guillemin, F.; Rat, A.-C. Body composition and clinical symptoms in patients with hip or knee osteoarthritis: Results from the KHOALA cohort. Semin. Arthritis Rheum. 2018, 47, 797–804. [Google Scholar] [CrossRef]
- Vlietstra, L.; Stebbings, S.; Meredith-Jones, K.; Abbott, J.H.; Treharne, G.J.; Waters, D.L. Sarcopenia in osteoarthritis and rheumatoid arthritis: The association with self-reported fatigue, physical function and obesity. PLoS ONE 2019, 14, e0217462. [Google Scholar] [CrossRef]
- Mathieu, S.; Couderc, M.; Tournadre, A.; Soubrier, M. Cardiovascular profile in osteoarthritis: A meta-analysis of cardiovascular events and risk factors. Jt. Bone Spine 2019, 86, 679–684. [Google Scholar] [CrossRef]
- Courties, A.; Berenbaum, F.; Sellam, J. The Phenotypic Approach to Osteoarthritis: A Look at Metabolic Syndrome-Associated Osteoarthritis. Jt. Bone Spine 2019, 86, 725–730. [Google Scholar] [CrossRef]
- Li, W.; Han, J.; Qureshi, A.A. Obesity and risk of incident psoriatic arthritis in US women. Ann. Rheum. Dis. 2012, 71, 1267–1272. [Google Scholar] [CrossRef]
- Pedreira, P.G.; Pinheiro, M.M.; Szejnfeld, V.L. Bone mineral density and body composition in postmenopausal women with psoriasis and psoriatic arthritis. Arthritis Res. Ther. 2011, 13, R16. [Google Scholar] [CrossRef]
- Ferguson, L.D.; Siebert, S.; McInnes, I.B.; Sattar, N. Cardiometabolic comorbidities in RA and PsA: Lessons learned and future directions. Nat. Rev. Rheumatol. 2019, 15, 461–474. [Google Scholar] [CrossRef] [PubMed]
- Choy, E.; Ganeshalingam, K.; Semb, A.G.; Szekanecz, Z.; Nurmohamed, M. Cardiovascular risk in rheumatoid arthritis: Recent advances in the understanding of the pivotal role of inflammation, risk predictors and the impact of treatment. Rheumatology 2014, 53, 2143–2154. [Google Scholar] [CrossRef] [PubMed]
- Kerekes, G.; Nurmohamed, M.T.; González-Gay, M.A.; Seres, I.; Paragh, G.; Kardos, Z.; Baráth, Z.; Tamási, L.; Soltész, P.; Szekanecz, Z. Rheumatoid arthritis and metabolic syndrome. Nat. Rev. Rheumatol. 2014, 10, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.; Peters, M.J.; McInnes, I.B.; Sattar, N. Changes in lipid levels with inflammation and therapy in RA: A maturing paradigm. Nat. Rev. Rheumatol. 2013, 9, 513–523. [Google Scholar] [CrossRef]
- Nowak, B.; Madej, M.; Łuczak, A.; Małecki, R.; Wiland, P. Disease Activity, Oxidized-LDL Fraction and Anti-Oxidized LDL Antibodies Influence Cardiovascular Risk in Rheumatoid Arthritis. Adv. Clin. Exp. Med. 2016, 25, 43–50. [Google Scholar] [CrossRef]
- Gómez Rosso, L.; Lhomme, M.; Meroño, T.; Sorroche, P.; Catoggio, L.; Soriano, E.; Saucedo, C.; Malah, V.; Dauteuille, C.; Boero, L.; et al. Altered lipidome and antioxidative activity of small, dense HDL in normolipidemic rheumatoid arthritis: Relevance of inflammation. Atherosclerosis 2014, 237, 652–660. [Google Scholar] [CrossRef]
- Lourida, E.S.; Georgiadis, A.N.; Papavasiliou, E.C.; Papathanasiou, A.I.; Drosos, A.A.; Tselepis, A.D. Patients with early rheumatoid arthritis exhibit elevated autoantibody titers against mildly oxidized low-density lipoprotein and exhibit decreased activity of the lipoprotein-associated phospholipase A2. Arthritis Res. Ther. 2007, 9, R19. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Lin, J.; Liang, J.; Ma, J.; Li, Q.; Mo, Y.; Cheng, W.; He, X.; Li, N.; Cao, M.; Xu, D.; et al. Myopenia is associated with joint damage in rheumatoid arthritis: A cross-sectional study. J. Cachexia Sarcopenia Muscle 2019, 10, 355–367. [Google Scholar] [CrossRef]
- Fink, R.I.; Kolterman, O.G.; Griffin, J.; Olefsky, J.M. Mechanisms of Insulin Resistance in Aging. J. Clin. Investig. 1983, 71, 1523–1535. [Google Scholar] [CrossRef]
- Ye, J. Mechanisms of insulin resistance in obesity. Front. Med. 2013, 7, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Drosatos, K. Fatty old hearts: Role of cardiac lipotoxicity in age-related cardiomyopathy. Pathobiol. Aging Age-Relat. Dis. 2016, 6, 32221. [Google Scholar] [CrossRef] [PubMed]
- Slawik, M.; Vidal-Puig, A.J. Lipotoxicity, overnutrition and energy metabolism in aging. Ageing Res. Rev. 2006, 5, 144–164. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Overnutrition, ectopic lipid and the metabolic syndrome. J. Investig. Med. 2016, 64, 1082–1086. [Google Scholar] [CrossRef] [PubMed]
- De Mello, A.H.; Costa, A.B.; Engel, J.D.G.; Rezin, G.T. Mitochondrial dysfunction in obesity. Life Sci. 2018, 192, 26–32. [Google Scholar] [CrossRef]
- Petersen, K.F. Mitochondrial Dysfunction in the Elderly: Possible Role in Insulin Resistance. Science 2003, 300, 1140–1142. [Google Scholar] [CrossRef]
- Liao, K.P.; Playford, M.P.; Frits, M.; Coblyn, J.S.; Iannaccone, C.; Weinblatt, M.E.; Shadick, N.S.; Mehta, N.N. The Association Between Reduction in Inflammation and Changes in Lipoprotein Levels and HDL Cholesterol Efflux Capacity in Rheumatoid Arthritis. J. Am. Heart Assoc. 2015, 4, e001588. [Google Scholar] [CrossRef]
- Tsujinaka, T.; Fujita, J.; Ebisui, C.; Yano, M.; Kominami, E.; Suzuki, K.; Tanaka, K.; Katsume, A.; Ohsugi, Y.; Shiozaki, H.; et al. Interleukin 6 receptor antibody inhibits muscle atrophy and modulates proteolytic systems in interleukin 6 transgenic mice. J. Clin. Investig. 1996, 97, 244–249. [Google Scholar] [CrossRef]
- Schaap, L.A.; Pluijm, S.M.F.; Deeg, D.J.H.; Visser, M. Inflammatory Markers and Loss of Muscle Mass (Sarcopenia) and Strength. Am. J. Med. 2006, 119, 526-e9. [Google Scholar] [CrossRef]
- Tournadre, A.; Pereira, B.; Dutheil, F.; Giraud, C.; Courteix, D.; Sapin, V.; Frayssac, T.; Mathieu, S.; Malochet-Guinamand, S.; Soubrier, M. Changes in body composition and metabolic profile during interleukin 6 inhibition in rheumatoid arthritis: Interleukin 6 inhibition in rheumatoid arthritis. J. Cachexia Sarcopenia Muscle 2017, 8, 639–646. [Google Scholar] [CrossRef]
- Tekeoğlu, İ.; Harman, H.; Sağ, S.; Altındiş, M.; Kamanlı, A.; Nas, K. Levels of serum pentraxin 3, IL-6, fetuin A and insulin in patients with rheumatoid arthritis. Cytokine 2016, 83, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.F.; Von Feldt, J.M.; Mostoufi-Moab, S.; Kim, W.; Taratuta, E.; Leonard, M.B. Insulin-like Growth Factor 1 and Adiponectin and Associations with Muscle Deficits, Disease Characteristics, and Treatments in Rheumatoid Arthritis. J. Rheumatol. 2015, 42, 2038–2045. [Google Scholar] [CrossRef] [PubMed]
- Guillet, C.; Delcourt, I.; Rance, M.; Giraudet, C.; Walrand, S.; Bedu, M.; Duche, P.; Boirie, Y. Changes in Basal and Insulin and Amino Acid Response of Whole Body and Skeletal Muscle Proteins in Obese Men. J. Clin. Endocrinol. Metab. 2009, 94, 3044–3050. [Google Scholar] [CrossRef] [PubMed]
- Tardif, N.; Salles, J.; Guillet, C.; Tordjman, J.; Reggio, S.; Landrier, J.-F.; Giraudet, C.; Patrac, V.; Bertrand-Michel, J.; Migne, C.; et al. Muscle ectopic fat deposition contributes to anabolic resistance in obese sarcopenic old rats through eIF2α activation. Aging Cell 2014, 13, 1001–1011. [Google Scholar] [CrossRef]
- Capel, F.; Acquaviva, C.; Pitois, E.; Laillet, B.; Rigaudière, J.-P.; Jouve, C.; Pouyet, C.; Gladine, C.; Comte, B.; Vianey Saban, C.; et al. DHA at nutritional doses restores insulin sensitivity in skeletal muscle by preventing lipotoxicity and inflammation. J. Nutr. Biochem. 2015, 26, 949–959. [Google Scholar] [CrossRef]
- Chavez, J.A.; Knotts, T.A.; Wang, L.-P.; Li, G.; Dobrowsky, R.T.; Florant, G.L.; Summers, S.A. A Role for Ceramide, but Not Diacylglycerol, in the Antagonism of Insulin Signal Transduction by Saturated Fatty Acids. J. Biol. Chem. 2003, 278, 10297–10303. [Google Scholar] [CrossRef]
- Peterson, J.M.; Wang, Y.; Bryner, R.W.; Williamson, D.L.; Alway, S.E. Bax signaling regulates palmitate-mediated apoptosis in C2 C12 myotubes. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1307–E1314. [Google Scholar] [CrossRef]
- Saini, A.; Sharples, A.P.; Al-Shanti, N.; Stewart, C.E. Omega-3 fatty acid EPA improves regenerative capacity of mouse skeletal muscle cells exposed to saturated fat and inflammation. Biogerontology 2017, 18, 109–129. [Google Scholar] [CrossRef]
- De Furlanetto, A.P.; Nunes, P.R.P.; Michelin, M.A.; Chica, J.E.L.; Murta, E.F.C.; Orsatti, F.L. Acute resistance exercise reduces increased gene expression in muscle atrophy of ovariectomised arthritic rats. Menopausal Rev. 2016, 4, 193–201. [Google Scholar]
- Castillero, E.; Martín, A.I.; López-Menduiña, M.; Granado, M.; Villanúa, M.Á.; López-Calderón, A. IGF-I system, atrogenes and myogenic regulatory factors in arthritis induced muscle wasting. Mol. Cell. Endocrinol. 2009, 309, 8–16. [Google Scholar] [CrossRef]
- Castillero, E.; Martín, A.I.; López-Menduiña, M.; Villanúa, M.A.; López-Calderón, A. Eicosapentaenoic acid attenuates arthritis-induced muscle wasting acting on atrogin-1 and on myogenic regulatory factors. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 297, R1322–R1331. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, A.; Bagchi, A.; De, S.; Mitra, S.; Mukherjee, S.; Ghosh, P.; Ghosh, A.; Chatterjee, M. Role of redox imbalance and cytokines in mediating oxidative damage and disease progression of patients with rheumatoid arthritis. Free Radic. Res. 2019, 53, 768–779. [Google Scholar] [CrossRef] [PubMed]
- Howard, C.; Ferrucci, L.; Sun, K.; Fried, L.P.; Walston, J.; Varadhan, R.; Guralnik, J.M.; Semba, R.D. Oxidative protein damage is associated with poor grip strength among older women living in the community. J. Appl. Physiol. 2007, 103, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Abe, M.; Lee, J.; Tatebayashi, D.; Himori, K.; Kanzaki, K.; Wada, M.; Bruton, J.D.; Westerblad, H.; Lanner, J.T. Muscle dysfunction associated with adjuvant-induced arthritis is prevented by antioxidant treatment. Skelet. Muscle 2015, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.K.; Kwon, J.-E.; Lee, S.-Y.; Lee, E.-J.; Kim, D.S.; Moon, S.-J.; Lee, J.; Kwok, S.-K.; Park, S.-H.; Cho, M.-L. IL-17-mediated mitochondrial dysfunction impairs apoptosis in rheumatoid arthritis synovial fibroblasts through activation of autophagy. Cell Death Dis. 2018, 8, e2565. [Google Scholar] [CrossRef] [PubMed]
- Vial, G.; De Pouilly, A.; Scouppe, L.; Pereira, B.; Daien, C.; Lukas, C.; Ruyssen-Witrand, A.; Vergne-Salle, P.; Richez, C.; Tournadre, A. Factors influencing the choice of biologic therapy following Rituximab in patients with rheumatoid arthritis: A retrospective study using propensity score. Jt. Bone Spine 2019, 87, 43–48. [Google Scholar] [CrossRef]
- Lepretti, M.; Martucciello, S.; Burgos Aceves, M.; Putti, R.; Lionetti, L. Omega-3 Fatty Acids and Insulin Resistance: Focus on the Regulation of Mitochondria and Endoplasmic Reticulum Stress. Nutrients 2018, 10, 350. [Google Scholar] [CrossRef]
- Zlobine, I.; Gopal, K.; Ussher, J.R. Lipotoxicity in obesity and diabetes-related cardiac dysfunction. Biochim. Biophys. Acta 2016, 1861, 1555–1568. [Google Scholar] [CrossRef]
- Harika, R.K.; Eilander, A.; Alssema, M.; Osendarp, S.J.M.; Zock, P.L. Intake of Fatty Acids in General Populations Worldwide Does Not Meet Dietary Recommendations to Prevent Coronary Heart Disease: A Systematic Review of Data from 40 Countries. Ann. Nutr. Metab. 2013, 63, 229–238. [Google Scholar] [CrossRef]
- ANSES. Actualisation Des Apports Nutritionnels Conseillés Pour Les Acides Gras. Rapport D’expertise Collective. Available online: https://www.anses.fr/fr/system/files/NUT2006sa0359Ra.pdf (accessed on 12 November 2018).
- Huang, S.; Rutkowsky, J.M.; Snodgrass, R.G.; Ono-Moore, K.D.; Schneider, D.A.; Newman, J.W.; Adams, S.H.; Hwang, D.H. Saturated fatty acids activate TLR-mediated proinflammatory signaling pathways. J. Lipid Res. 2012, 53, 2002–2013. [Google Scholar] [CrossRef]
- FAO; WHO. WHO: Fats and Fatty Acids in Human Nutrition, Report of an Expert Consultation, Food and Nutrition Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010. [Google Scholar]
- Kris-Etherton, P.M.; Innis, S.; Ammerican Dietetic Assocition; Dietitians of Canada. Position of the American Dietetic Association and Dietitians of Canada: Dietary Fatty Acids. J. Am. Diet. Assoc. 2007, 107, 1599–1611. [Google Scholar] [PubMed]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association Between Omega-3 Fatty Acid Supplementation and Risk of Major Cardiovascular Disease Events: A Systematic Review and Meta-analysis. JAMA 2012, 308, 1024. [Google Scholar] [CrossRef] [PubMed]
- Tur, J.A.; Bibiloni, M.M.; Sureda, A.; Pons, A. Dietary sources of omega 3 fatty acids: Public health risks and benefits. Br. J. Nutr. 2012, 107, S23–S52. [Google Scholar] [CrossRef] [PubMed]
- Jacobsson, L.T.H.; Turesson, C.; Gülfe, A.; Kapetanovic, M.C.; Saxne, T.; Geborek, P. Treatment with tumor necrosis factor blockers is associated with a lower incidence of first cardiovascular events in patients with rheumatoid arthritis. J. Rheumatol. 2005, 32, 1213–1218. [Google Scholar] [PubMed]
- Norling, L.V.; Headland, S.E.; Dalli, J.; Arnardottir, H.H.; Haworth, O.; Jones, H.R.; Irimia, D.; Serhan, C.N.; Perretti, M. Proresolving and cartilage-protective actions of resolvin D1 in inflammatory arthritis. JCI Insight 2016, 1, e85922. [Google Scholar] [CrossRef] [PubMed]
- Spite, M.; Clària, J.; Serhan, C.N. Resolvins, Specialized Pro-Resolving Lipid Mediators and their Potential Roles in Metabolic Diseases. Cell Metab. 2014, 19, 21–36. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lim, K.; Kim, K.H.; Kim, J.H.; Choi, J.S.; Shim, S.-C. N-3 polyunsaturated fatty acids restore Th17 and Treg balance in collagen antibody-induced arthritis. PLoS ONE 2018, 13, e0194331. [Google Scholar] [CrossRef]
- Venkatraman, J.T.; Chu, W. Effects of Dietary ω-3 and ω-6 Lipids and Vitamin E on Serum Cytokines, Lipid Mediators and Anti-DNA Antibodies in a Mouse Model for Rheumatoid Arthritis. J. Am. Coll. Nutr. 1999, 18, 602–613. [Google Scholar] [CrossRef]
- Arnardottir, H.H.; Dalli, J.; Norling, L.V.; Colas, R.A.; Perretti, M.; Serhan, C.N. Resolvin D3 is dysregulated in arthritis and reduces arthritic inflammation. J. Immunol. 2016, 197, 2362–2368. [Google Scholar] [CrossRef]
- Zampelas, A.; Panagiotakos, D.B.; Pitsavos, C.; Das, U.N.; Chrysohoou, C.; Skoumas, Y.; Stefanadis, C. Fish Consumption Among Healthy Adults Is Associated with Decreased Levels of Inflammatory Markers Related to Cardiovascular Disease. J. Am. Coll. Cardiol. 2005, 46, 120–124. [Google Scholar] [CrossRef]
- Cleland, L.G.; Caughey, G.E.; James, M.J.; Proudman, S.M. Reduction of cardiovascular risk factors with longterm fish oil treatment in early rheumatoid arthritis. J. Rheumatol. 2006, 33, 1973–1979. [Google Scholar] [PubMed]
- Espersen, G.T.; Grunnet, N.; Lervang, H.H.; Nielsen, G.L.; Thomsen, B.S.; Faarvang, K.L.; Dyerberg, J.; Ernst, E. Decreased Interleukin-1 beta levels in plasma from rheumatoid arthritis patients after dietary supplementation with n-3 polyunsaturated fatty acids. Clin. Rheumatol. 1992, 11, 393–395. [Google Scholar] [CrossRef] [PubMed]
- Kremer, J.M.; Lawrence, D.A.; Jubiz, W.; Digiacomo, R.; Rynes, R.; Bartholomew, L.E.; Sherman, M. Dietary fish oil and olive oil supplementation in patients with Rheumatoid Arthritis clinical and immunologic effects. Arthritis Rheum. 1990, 33, 810–820. [Google Scholar] [CrossRef]
- Di Giuseppe, D.; Wallin, A.; Bottai, M.; Askling, J.; Wolk, A. Long-term intake of dietary long-chain n-3 polyunsaturated fatty acids and risk of rheumatoid arthritis: A prospective cohort study of women. Ann. Rheum. Dis. 2014, 73, 1949–1953. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.W.; Demoruelle, M.K.; Deane, K.D.; Weisman, M.H.; Buckner, J.H.; Gregersen, P.K.; Mikuls, T.R.; O’Dell, J.R.; Keating, R.M.; Fingerlin, T.E.; et al. Omega-3 fatty acids are associated with a lower prevalence of autoantibodies in shared epitope-positive subjects at risk for rheumatoid arthritis. Ann. Rheum. Dis. 2017, 76, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Proudman, S.M.; James, M.J.; Spargo, L.D.; Metcalf, R.G.; Sullivan, T.R.; Rischmueller, M.; Flabouris, K.; Wechalekar, M.D.; Lee, A.T.; Cleland, L.G. Fish oil in recent onset rheumatoid arthritis: A randomised, double-blind controlled trial within algorithm-based drug use. Ann. Rheum. Dis. 2015, 74, 89–95. [Google Scholar] [CrossRef]
- Jeffery, L.; Fisk, H.L.; Calder, P.C.; Filer, A.; Raza, K.; Buckley, C.D.; McInnes, I.; Taylor, P.C.; Fisher, B.A. Plasma Levels of Eicosapentaenoic Acid Are Associated with Anti-TNF Responsiveness in Rheumatoid Arthritis and Inhibit the Etanercept-driven Rise in Th17 Cell Differentiation in Vitro. J. Rheumatol. 2017, 44, 748–756. [Google Scholar] [CrossRef]
- Serhan, C.N.; Levy, B.D. Resolvins in inflammation: Emergence of the pro-resolving superfamily of mediators. J. Clin. Investig. 2018, 128, 2657–2669. [Google Scholar] [CrossRef]
- Guichardant, M.; Calzada, C.; Bernoud-Hubac, N.; Lagarde, M.; Véricel, E. Omega-3 polyunsaturated fatty acids and oxygenated metabolism in atherothrombosis. Biochim. Biophys. Acta 2015, 1851, 485–495. [Google Scholar] [CrossRef]
- Mori, T.A. Omega-3 fatty acids and cardiovascular disease: Epidemiology and effects on cardiometabolic risk factors. Food Funct. 2014, 5, 2004–2019. [Google Scholar] [CrossRef]
- Woodman, R.J.; Mori, T.A.; Burke, V.; Puddey, I.B.; Watts, G.F.; Best, J.D.; Beilin, L.J. Docosahexaenoic Acid but Not Eicosapentaenoic Acid Increases LDL Particle Size in Treated Hypertensive Type 2 Diabetic Patients. Diabetes Care 2003, 26, 253. [Google Scholar] [CrossRef] [PubMed]
- Tani, S.; Nagao, K.; Matsumoto, M.; Hirayama, A. Highly Purified Eicosapentaenoic Acid May Increase Low-Density Lipoprotein Particle Size by Improving Triglyceride Metabolism in Patients with Hypertriglyceridemia. Circ. J. 2013, 77, 2349–2357. [Google Scholar] [CrossRef] [PubMed]
- Casanova, M.A.; Medeiros, F.; Trindade, M.; Cohen, C.; Oigman, W.; Neves, M.F. Omega-3 fatty acids supplementation improves endothelial function and arterial stiffness in hypertensive patients with hypertriglyceridemia and high cardiovascular risk. J. Am. Soc. Hypertens. 2017, 11, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Liu, J. Association between circulating oxidized low-density lipoprotein and atherosclerotic cardiovascular disease. Chronic Dis. Transl. Med. 2017, 3, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Zhao, D.; Wang, M.; Zhao, F.; Han, X.; Qi, Y.; Liu, J. Association Between Circulating Oxidized LDL and Atherosclerotic Cardiovascular Disease: A Meta-analysis of Observational Studies. Can. J. Cardiol. 2017, 33, 1624–1632. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The Importance of the Omega-6/Omega-3 Fatty Acid Ratio in Cardiovascular Disease and Other Chronic Diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.-M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Albert, C.M.; Gordon, D.; Copeland, T.; et al. Marine n−3 Fatty Acids and Prevention of Cardiovascular Disease and Cancer. N. Engl. J. Med. 2019, 380, 23–32. [Google Scholar] [CrossRef]
- Alexander, D.D.; Miller, P.E.; Van Elswyk, M.E.; Kuratko, C.N.; Bylsma, L.C. A Meta-Analysis of Randomized Controlled Trials and Prospective Cohort Studies of Eicosapentaenoic and Docosahexaenoic Long-Chain Omega-3 Fatty Acids and Coronary Heart Disease Risk. Mayo Clin. Proc. 2017, 92, 15–29. [Google Scholar] [CrossRef]
- Wang, Q.; Liang, X.; Wang, L.; Lu, X.; Huang, J.; Cao, J.; Li, H.; Gu, D. Effect of omega-3 fatty acids supplementation on endothelial function: A meta-analysis of randomized controlled trials. Atherosclerosis 2012, 221, 536–543. [Google Scholar] [CrossRef]
- Tousoulis, D.; Plastiras, A.; Siasos, G.; Oikonomou, E.; Verveniotis, A.; Kokkou, E.; Maniatis, K.; Gouliopoulos, N.; Miliou, A.; Paraskevopoulos, T.; et al. Omega-3 PUFAs improved endothelial function and arterial stiffness with a parallel antiinflammatory effect in adults with metabolic syndrome. Atherosclerosis 2014, 232, 10–16. [Google Scholar] [CrossRef]
- Nigam, A.; Frasure-Smith, N.; Lespérance, F.; Julien, P. Relationship between n-3 and n-6 plasma fatty acid levels and insulin resistance in coronary patients with and without metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Thorseng, T.; Witte, D.R.; Vistisen, D.; Borch-Johnsen, K.; Bjerregaard, P.; Jørgensen, M.E. The association between n-3 fatty acids in erythrocyte membranes and insulin resistance: The inuit health in transition study. Int. J. Circumpolar Health 2009, 68, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Delarue, J.; Li, C.-H.; Cohen, R.; Corporeau, C.; Simon, B. Interaction of fish oil and a glucocorticoid on metabolic responses to an oral glucose load in healthy human subjects. Br. J. Nutr. 2006, 95, 267. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, M.K.; Turner, N. Mitochondrial dysfunction and insulin resistance: An update. Endocr. Connect. 2015, 4, R1–R15. [Google Scholar] [CrossRef]
- Lovsletten Increased triacylglycerol—Fatty acid substrate cycling in human skeletal muscle cells exposed to eicosapentaenoic acid. PLoS ONE 2018, 13, e0208048.
- Cavaliere, G.; Trinchese, G.; Bergamo, P.; De Filippo, C.; Mattace Raso, G.; Gifuni, G.; Putti, R.; Moni, B.H.; Canani, R.B.; Meli, R.; et al. Polyunsaturated Fatty Acids Attenuate Diet Induced Obesity and Insulin Resistance, Modulating Mitochondrial Respiratory Uncoupling in Rat Skeletal Muscle. PLoS ONE 2016, 11, e0149033. [Google Scholar] [CrossRef]
- Baillie, R.A.; Takada, R.; Nakamura, M.; Clarke, S.D. Coordinate induction of peroxisomal acyl-CoA oxidase and UCP-3 by dietary fish oil: A mechanism for decreased body fat deposition. Prostaglandins Leukot. Essent. Fatty Acids 1999, 60, 351–356. [Google Scholar] [CrossRef]
- Power, G.W.; Newsholme, E.A. Dietary Fatty Acids Influence the Activity and Metabolic Control of Mitochondrial Carnitine Palmitoyltransferase I in Rat Heart and Skeletal Muscle. J. Nutr. 1997, 127, 2142–2150. [Google Scholar] [CrossRef]
- Figueras, M.; Olivan, M.; Busquets, S.; López-Soriano, F.J.; Argilés, J.M. Effects of Eicosapentaenoic Acid (EPA) Treatment on Insulin Sensitivity in an Animal Model of Diabetes: Improvement of the Inflammatory Status. Obesity 2011, 19, 362–369. [Google Scholar] [CrossRef]
- González-Périz, A.; Horrillo, R.; Ferré, N.; Gronert, K.; Dong, B.; Morán-Salvador, E.; Titos, E.; Martínez-Clemente, M.; López-Parra, M.; Arroyo, V.; et al. Obesity-induced insulin resistance and hepatic steatosis are alleviated by ω-3 fatty acids: A role for resolvins and protectins. FASEB J. 2009, 23, 1946–1957. [Google Scholar] [CrossRef]
- Aas, V.; Rokling-Andersen, M.H.; Kase, E.T.; Thoresen, G.H.; Rustan, A.C. Eicosapentaenoic acid (20:5 n-3) increases fatty acid and glucose uptake in cultured human skeletal muscle cells. J. Lipid Res. 2006, 47, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Mirza, K.A.; Luo, M.; Pereira, S.; Voss, A.; Das, T.; Tisdale, M.J. In vitro assessment of the combined effect of eicosapentaenoic acid, green tea extract and curcumin C3 on protein loss in C2C12 myotubes. In Vitro Cell. Dev. Biol. Anim. 2016, 52, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lin, Q.; Zheng, P.; Zhang, J.; Huang, F. DHA Inhibits Protein Degradation More Efficiently than EPA by Regulating the PPARγ/NFκB Pathway in C2C12 Myotubes. Biol. Med. Res. Int. 2013, 2013, 318981. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.K.; Kim, J.H.; Lee, J.H.; Son, Y.H.; Lee, M.W.; Kim, H.J.; Noh, S.A.; Kim, K.P.; Kim, I.-G.; Lee, M.J. Docosahexaenoic acid-mediated protein aggregates may reduce proteasome activity and delay myotube degradation during muscle atrophy in vitro. Exp. Mol. Med. 2017, 49, e287. [Google Scholar] [CrossRef]
- Deval, C.; Capel, F.; Laillet, B.; Polge, C.; Béchet, D.; Taillandier, D.; Attaix, D.; Combaret, L. Docosahexaenoic acid-supplementation prior to fasting prevents muscle atrophy in mice: Docosahexaenoic acid limits muscle wasting in fasted mice. J. Cachexia Sarcopenia Muscle 2016, 7, 587–603. [Google Scholar] [CrossRef]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 93, 402–412. [Google Scholar] [CrossRef]
- Smith, G.I.; Atherton, P.; Reeds, D.N.; Mohammed, B.S.; Rankin, D.; Rennie, M.J.; Mittendorfer, B. Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperaminoacidemia-hyperinsulinemia in healthy young and middle aged men and women. Clin. Sci. 2011, 121, 267–278. [Google Scholar] [CrossRef]
- AbouAssi, H.; Connelly, M.A.; Bateman, L.A.; Tune, K.N.; Huebner, J.L.; Kraus, V.B.; Winegar, D.A.; Otvos, J.D.; Kraus, W.E.; Huffman, K.M. Does a lack of physical activity explain the rheumatoid arthritis lipid profile? Lipids Health Dis. 2017, 16, 39. [Google Scholar] [CrossRef]
- Daïen, C.I.; Duny, Y.; Barnetche, T.; Daurès, J.-P.; Combe, B.; Morel, J. Effect of TNF inhibitors on lipid profile in rheumatoid arthritis: A systematic review with meta-analysis. Ann. Rheum. Dis. 2012, 71, 862–868. [Google Scholar] [CrossRef]
- Da Silva, R.C.; Diniz, M.D.F.H.S.; Alvim, S.; Vidigal, P.G.; Fedeli, L.M.G.; Barreto, S.M. Physical Activity and Lipid Profile in the ELSA-Brasil Study. Arq. Bras. Cardiol. 2016, 107, 10–19. [Google Scholar] [CrossRef]
- Van Zanten, J.J.C.S.; Sandoo, A.; Metsios, G.S.; Stavropoulos-Kalinoglou, A.; Ntoumanis, N.; Kitas, G.D. Comparison of the effects of exercise and anti-TNF treatment on cardiovascular health in rheumatoid arthritis: Results from two controlled trials. Rheumatol. Int. 2018, 39, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Popa, C.D.; Arts, E.; Fransen, J.; van Riel, P.L.C.M. Atherogenic Index and High-Density Lipoprotein Cholesterol as Cardiovascular Risk Determinants in Rheumatoid Arthritis: The Impact of Therapy with Biologicals. Mediat. Inflamm. 2012, 2012, 785946. [Google Scholar] [CrossRef] [PubMed]
- Zacharewicz, E.; Hesselink, M.K.C.; Schrauwen, P. Exercise counteracts lipotoxicity by improving lipid turnover and lipid droplet quality. J. Intern. Med. 2018, 284, 505–518. [Google Scholar] [CrossRef]
- Lemmey, A.B.; Marcora, S.M.; Chester, K.; Wilson, S.; Casanova, F.; Maddison, P.J. Effects of high-intensity resistance training in patients with rheumatoid arthritis: A randomized controlled trial. Arthritis Rheum. 2009, 61, 1726–1734. [Google Scholar] [CrossRef] [PubMed]
- Khoja, S.S.; Almeida, G.J.; Chester Wasko, M.; Terhorst, L.; Piva, S.R. Association of Light-Intensity Physical Activity with Lower Cardiovascular Disease Risk Burden in Rheumatoid Arthritis: Light-Intensity PA and Lower CVD in RA. Arthritis Care Res. 2016, 68, 424–431. [Google Scholar] [CrossRef]
- Strasser, B.; Leeb, G.; Strehblow, C.; Schobersberger, W.; Haber, P.; Cauza, E. The effects of strength and endurance training in patients with rheumatoid arthritis. Clin. Rheumatol. 2011, 30, 623–632. [Google Scholar] [CrossRef]
- Byram, K.W.; Oeser, A.M.; Linton, M.F.; Fazio, S.; Stein, C.M.; Ormseth, M.J. Exercise is Associated with Increased Small HDL Particle Concentration and Decreased Vascular Stiffness in Rheumatoid Arthritis. JCR 2018, 24, 417–421. [Google Scholar] [CrossRef]
- Francois, M.E.; Durrer, C.; Pistawka, K.J.; Halperin, F.A.; Little, J.P. Resistance-based interval exercise acutely improves endothelial function in type 2 diabetes. Am. J. Physiol. Heart Circ. Physiol. 2016, 311, H1258–H1267. [Google Scholar] [CrossRef]
- Sandoo, A.; van Zanten, J.J.C.S.V.; Toms, T.E.; Carroll, D.; Kitas, G.D. Anti-TNFα therapy transiently improves high density lipoprotein cholesterol levels and microvascular endothelial function in patients with rheumatoid arthritis: A Pilot Study. BMC Musculoskelet. Disord. 2012, 13, 127. [Google Scholar] [CrossRef]
- Lane-Cordova, A.D.; Phillips, S.A.; Baynard, T.; Woods, J.A.; Motl, R.W.; Fernhall, B. Effects of ageing and physical activity on blood pressure and endothelial function during acute inflammation: Acute inflammation and vascular function. Exp. Physiol. 2016, 101, 962–971. [Google Scholar] [CrossRef]
- Enns, J.E.; Yeganeh, A.; Zarychanski, R.; Abou-Setta, A.M.; Friesen, C.; Zahradka, P.; Taylor, C.G. The impact of omega-3 polyunsaturated fatty acid supplementation on the incidence of cardiovascular events and complications in peripheral arterial disease: A systematic review and meta-analysis. BMC Cardiovasc. Disord. 2014, 14, 70. [Google Scholar] [CrossRef] [PubMed]
- Gross, B.W.; Gillio, M.; Rinehart, C.D.; Lynch, C.A.; Rogers, F.B. Omega-3 Fatty Acid Supplementation and Warfarin: A Lethal Combination in Traumatic Brain Injury. J. Trauma Nurs. 2017, 24, 15–18. [Google Scholar] [CrossRef] [PubMed]
- De La Forest Divonne, M.; Gottenberg, J.E.; Salliot, C. Safety of biologic DMARDs in RA patients in real life: A systematic literature review and meta-analyses of biologic registers. Jt. Bone Spine 2017, 84, 133–140. [Google Scholar] [CrossRef] [PubMed]
| Model | Intervention | Results | References |
|---|---|---|---|
| Lipid Metabolism | |||
| In Vitro | |||
| C2C12 muscle cells | 50 µM EPA or DHA-16 h | Reduction of TG, DG and ceramide content | Pinel et al., 2016 [9] |
| In Vivo | |||
| Rats fed with a HFD (n = 8) | Fish oil supplementation for 6 weeks | Increase of mitochondrial respiratory uncoupling in hind leg muscle | Cavaliere et al., 2016 [118] |
| Wistar rats with a HFD (n = 6) | Fish oil supplementation for 10 weeks | Increase of CPT1 expression and activity | Power et al., 1997 [120] |
| Carbohydrate metabolism | |||
| In Vitro | |||
| C2C12 muscle cells | 500 µM palmitate + 30 µM DHA-16 h | Restoration of insulin response altered by palmitate-treatment | Capel et al., 2015 [65] |
| C2C12 muscle cells | 50 µM EPA treatment-180 min | Increase of 2-DOG uptake | Figueras et al., 2011 [121] |
| In Vivo | |||
| Rat with spontaneous type 2 diabetes (n = 10) | EPA 0.5 g/kg for 28 days | Increase of GLUT4 mRNA in skeletal muscle | Figueras et al., 2011 [121] |
| Male ob/ob mice (n = 16) | 6% of lipid content was provided by omega 3 for 5 weeks | Increase of GLUT4 mRNA and phosphorylation of IRS-1 and Akt in skeletal muscle | González-Périz et al., 2009 [122] |
| Human skeletal muscle cells (vastus lateralis) | 0.6 mM EPA retreatment-24 h | Increase of glucose transport in response to 100 nM insulin-15 min | Aas et al., 2006 [123] |
| Protein metabolism | |||
| In Vitro | |||
| C2C12 muscle cells | 75 mM palmitate + 50 µM EPA pretreatment-1 h | Increase of muscle regeneration capacities | Saini et al., 2017 [68] |
| C2C12 myotubes | 50 µM EPA treatment-24 h | Decrease of 3H-Phe muscle release induced by TNF | Mirza et al., 2016 [124] |
| C2C12 muscle cells | 300–600 µM DHA and EPA-24 h | Inhibition of muscle protein degradation | Wang et al., 2013 [125] |
| C2C12 muscle cells overexpressing aggregation-tau protein | DHA 100 µM-4 h | Reduction of myotube degradation by inhibiting S26 proteasome activity | Shin et al., 2017 [126] |
| In Vivo | |||
| C57BL/6 mice (n = 20) | 8 weeks DHA enriched-diet | Tibialis anterior preserved after a 48 h-fasting | Deval et al., 2016 [127] |
| Wistar collagen-induced arthritis rats (n = 18) | 12 days EPA oral administration | Prevention of TNF-α and atrogin-1 increase induced by arthritisAttenuation of the gastrocnemius atrophy and of the increase of MuRF1 induced by RA | Castillero et al., 2009 [71] |
| Dietary Lipids | Physical Activity | Therapy | |
|---|---|---|---|
| Lipid profile | Omega 3 fatty ↗ HDL-c levels [102] | More exercise is associated with smaller HDL-P, fewer large HDL-P and reduced mean HDL-size [130] | TNF-α inhibitors ↗ TC and HDL-c levels [131] |
| Omega 3 fatty ↘ circulating oxidized LDL-c [105,106,107] | Correlation between the intensity of physical activity and HDL levels [132] | 3 months anti-TNF-α treatment ↘ TC levels [133] | |
| DHA and EPA ↗ LDL particle size in hypertensive type 2 diabetes patients and with a hypertriglyceridemia [103,104] | Inverse correlation between the intensity of physical activity and TG and LDL levels [132] | TNF-α inhibition ↗ anti-inflammatory properties of HDL-c [134] | |
| Muscle lipotoxicity | 50µM EPA or DHA ↘ TG, DG and ceramides content [9] | Endurance training ↗ lipid turnover and improve lipid droplets quality [135] | |
| EPA ↗ muscle regeneration capacity of C2C12 muscle cells exposed to palmitate [68] | |||
| Muscle mass and function | Omega 3 during 8 weeks ↗ protein anabolic response in healthy adults [129] | Acute resistance exercise preserved lean body mass, muscle anabolic response and muscle function [136,137] | |
| Omega 3 supplementation ↗ muscle protein rate and phosphorylation of mTORSer2448 and p70S6KThr389 [128] | Long term training program combining strength and endurance ↗ muscle functions [138] | ||
| DHA ↘ muscle protein degradation in C2C12 [125,126] | |||
| CV diseases | 2g omega 3 (46% EPA-38% DHA) ↗ endothelial function and ↘ arterial stiffness [112] | Exercise is associated with a reduced vascular stiffness in RA [139] | 3 months anti-TNF-α treatment improved blood pressure in RA patients [133] |
| Omega 3 ↗ endothelial function in 16 patients with hypertriglyceridemia [105] | Resistance exercise improves endothelial function in type 2 diabetes subjects [140] | 3 months anti-TNF-α treatment improved endothelial function in RA patients [141] | |
| Omega 3 fatty acids ↗ FMD [111] | Moderated-vigourous physical activity ↘ FMD and blood pressure and not affected vascular function [142] | TNF-α inhibitors ↘ the incidence of CV diseases [86] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lanchais, K.; Capel, F.; Tournadre, A. Could Omega 3 Fatty Acids Preserve Muscle Health in Rheumatoid Arthritis? Nutrients 2020, 12, 223. https://doi.org/10.3390/nu12010223
Lanchais K, Capel F, Tournadre A. Could Omega 3 Fatty Acids Preserve Muscle Health in Rheumatoid Arthritis? Nutrients. 2020; 12(1):223. https://doi.org/10.3390/nu12010223
Chicago/Turabian StyleLanchais, Kassandra, Frederic Capel, and Anne Tournadre. 2020. "Could Omega 3 Fatty Acids Preserve Muscle Health in Rheumatoid Arthritis?" Nutrients 12, no. 1: 223. https://doi.org/10.3390/nu12010223
APA StyleLanchais, K., Capel, F., & Tournadre, A. (2020). Could Omega 3 Fatty Acids Preserve Muscle Health in Rheumatoid Arthritis? Nutrients, 12(1), 223. https://doi.org/10.3390/nu12010223






