Effects of Capsinoids on Daily Physical Activity, Body Composition and Cold Hypersensitivity in Middle-Aged and Older Adults: A Randomized Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Study
2.1.1. Trial Design
2.1.2. Participants and Ethical Considerations
2.1.3. Investigational Food and Dose Method
2.1.4. Outcomes and Assessments Tools
2.1.5. Measurement Schedule
2.1.6. Randomization and Blinding
2.1.7. Statistical Analysis for the Human Study
2.2. Animal Study
2.2.1. Animals and Diets
2.2.2. Sampling Procedures
2.2.3. Gene Expression Analysis
2.2.4. 8-Hydroxy-2-deoxyguanosine (8-OHdG) Assay
2.2.5. Statistical Analysis for the Animal Study
3. Results
3.1. Human Study
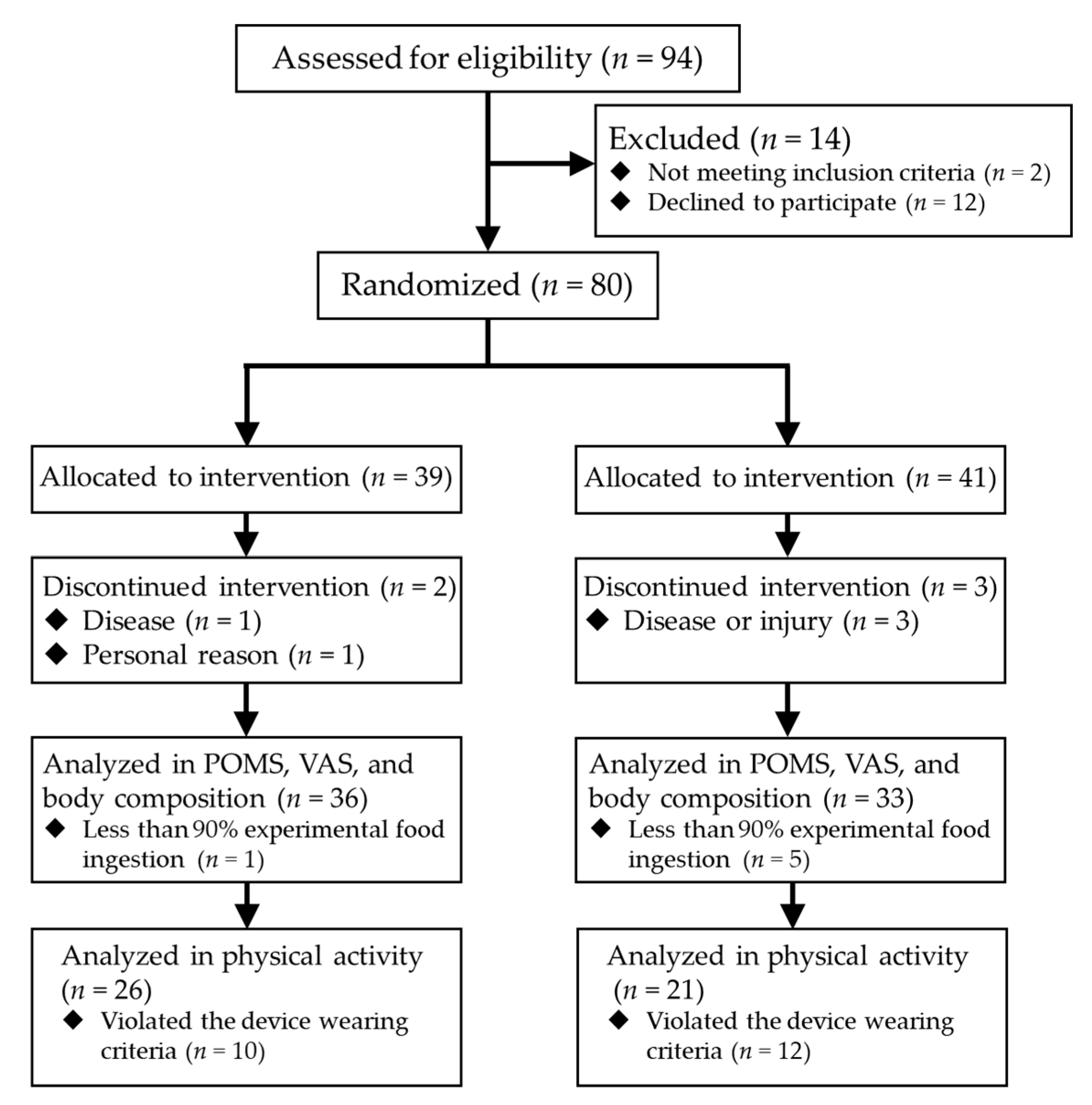
3.1.1. Participant Flow
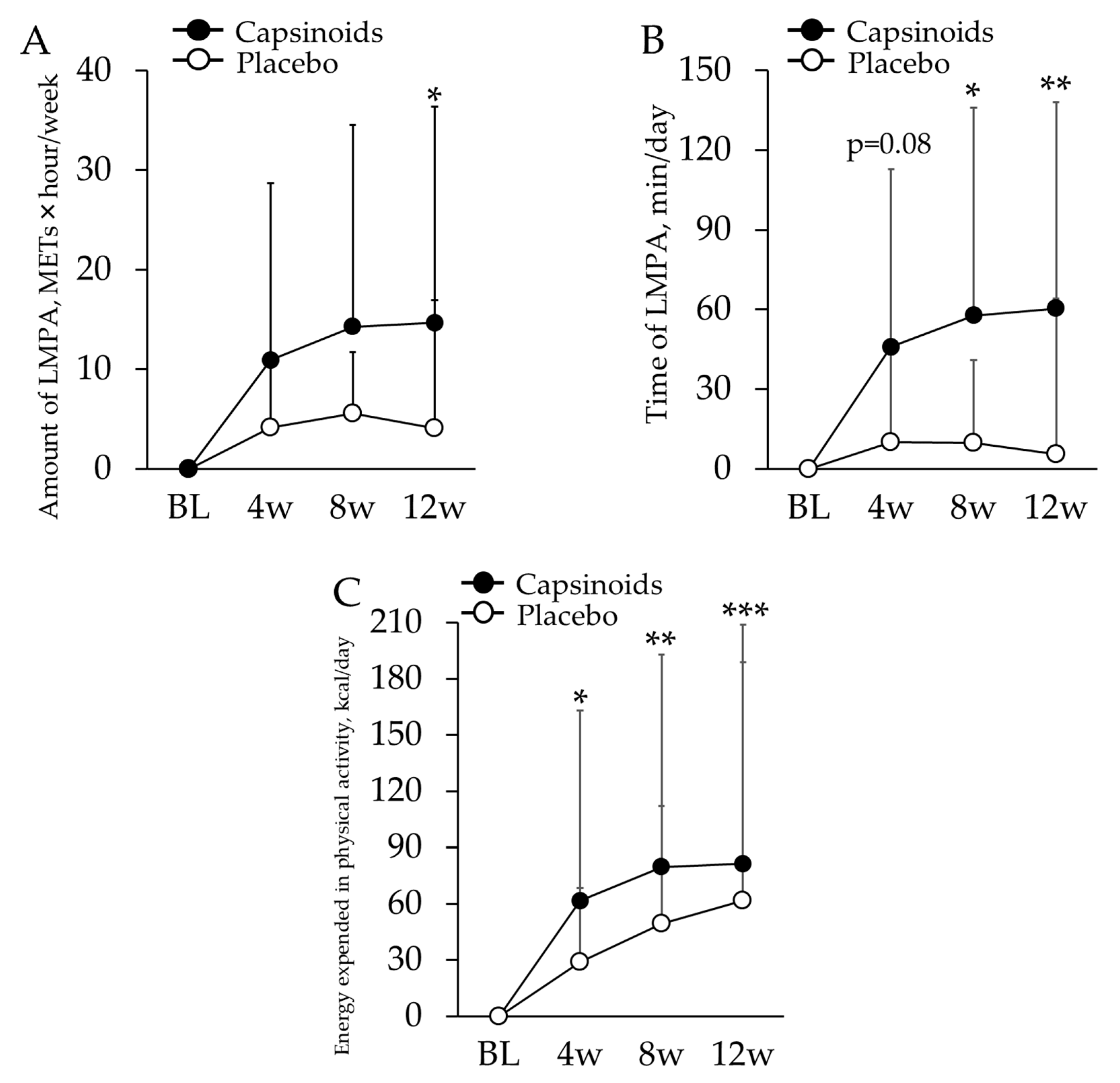
3.1.2. Amount and Time of Physical Activity
3.1.3. Changes in Amount of Physical Activity in Participants Who Have Inactive Lifestyles
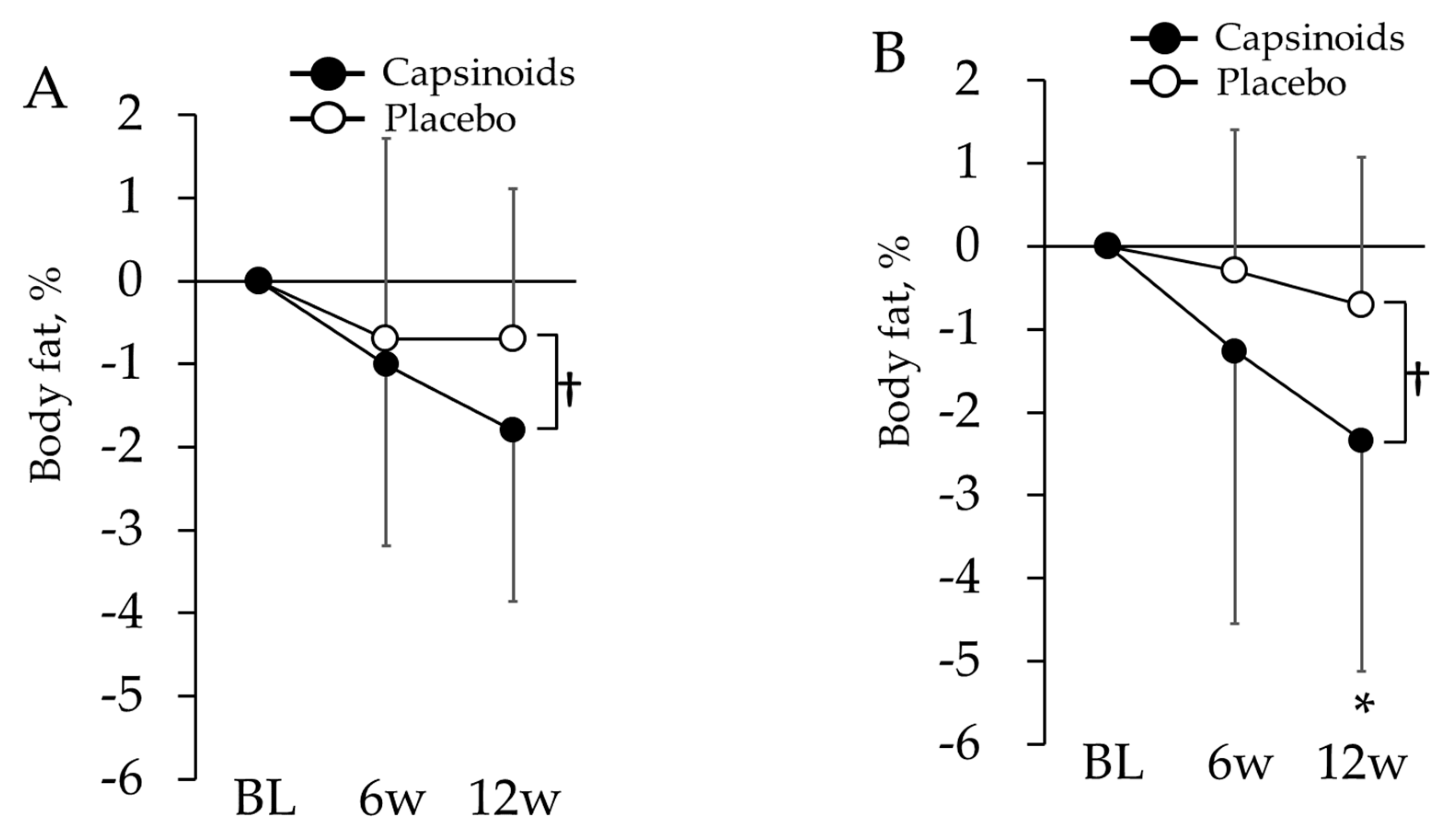
3.1.4. Changes in POMS2 Short Form, VAS and Body Composition
3.1.5. Changes in the Body Composition of Overweight Subjects
3.1.6. Safety Evaluation
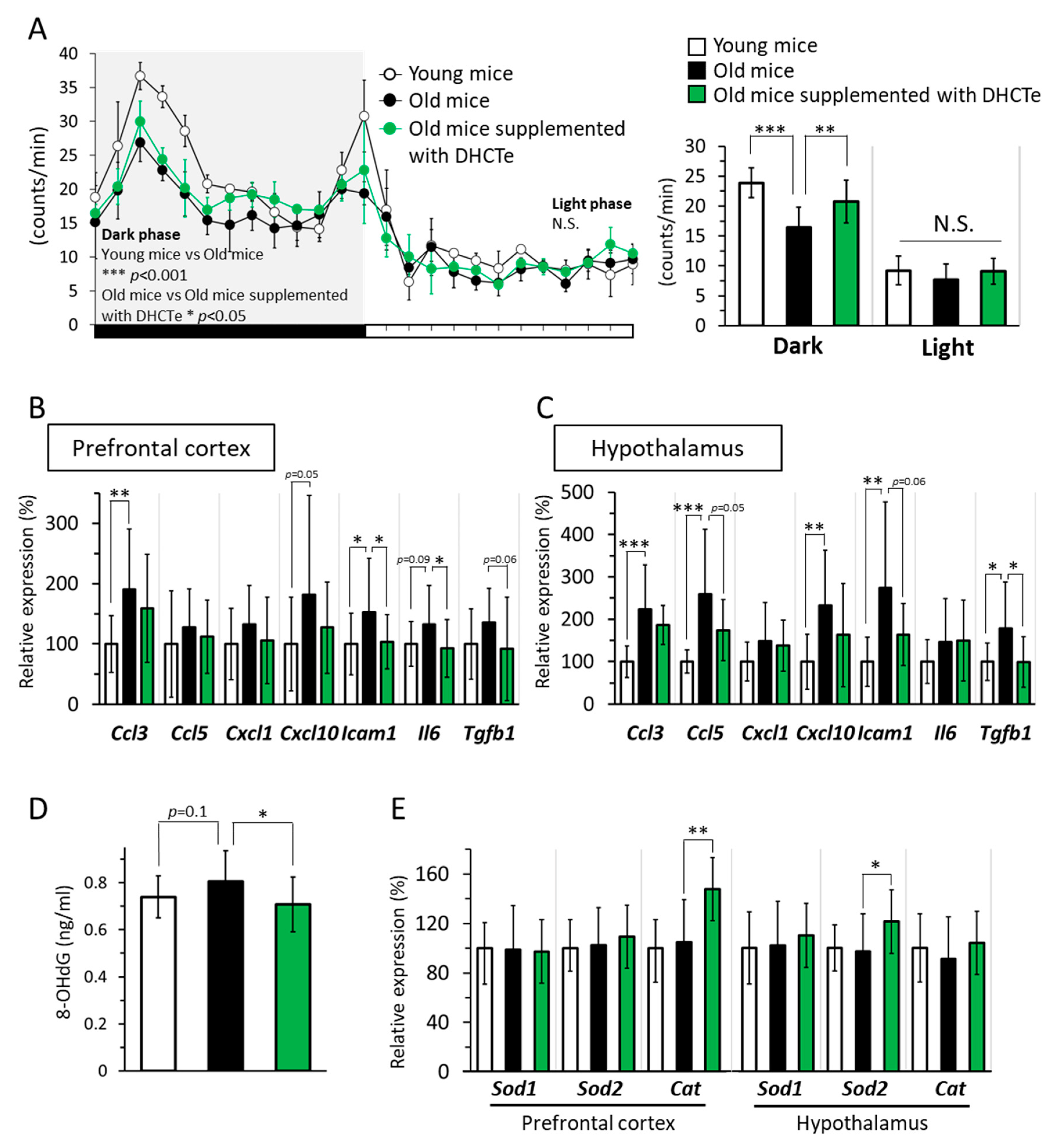
3.2. Animal Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- De Vries, N.M.; Staal, J.B.; van Ravensberg, C.D.; Hobbelen, J.S.; Olde Rikkert, M.G.; Nijhuis-van der Sanden, M.W. Outcome instruments to measure frailty: A systematic review. Ageing Res. Rev. 2011, 10, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, T.; Ohara, T.; Hirakawa, Y.; Yoshida, D.; Doi, Y.; Hata, J.; Kanba, S.; Iwaki, T.; Kiyohara, Y. Midlife and late-life blood pressure and dementia in Japanese elderly: The Hisayama study. Hypertension 2011, 58, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Ohara, T.; Doi, Y.; Ninomiya, T.; Hirakawa, Y.; Hata, J.; Iwaki, T.; Kanba, S.; Kiyohara, Y. Glucose tolerance status and risk of dementia in the community: The Hisayama study. Neurology 2011, 77, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B.; Li, T.Y.; Colditz, G.A.; Willett, W.C.; Manson, J.E. Television watching and other sedentary behaviors in relation to risk of obesity and type 2 diabetes mellitus in women. JAMA 2003, 289, 1785–1791. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.E.; Nicol, C.W.; Bredin, S.S. Health benefits of physical activity: The evidence. Can. Med. Assoc. 2006, 74, 801–809. [Google Scholar] [CrossRef]
- Warren, T.Y.; Barry, V.; Hooker, S.P.; Sui, X.; Church, T.S.; Blair, S.N. Sedentary behaviors increase risk of cardiovascular disease mortality in men. Med. Sci. Sports Exerc. 2010, 42, 879–885. [Google Scholar] [CrossRef]
- Karlsson, M.K.; Nordqvist, A.; Karlsson, C. Physical activity, muscle function, falls and fractures. Food Nutr. Res. 2008, 52. [Google Scholar] [CrossRef]
- Warburton, D.E.R.; Bredin, S.S.D. Health benefits of physical activity: A systematic review of current systematic reviews. Curr. Opin. Cardiol. 2017, 32, 541–556. [Google Scholar] [CrossRef]
- Speakman, J.R.; Westerterp, K.R. Associations between energy demands, physical activity, and body composition in adult humans between 18 and 96 y of age. Am. J. Clin. Nutr. 2010, 92, 826–834. [Google Scholar] [CrossRef]
- Wilmot, E.G.; Edwardson, C.L.; Achana, F.A.; Davies, M.J.; Gorely, T.; Gray, L.J.; Khunti, K.; Yates, T.; Biddle, S.J. Sedentary time in adults and the association with diabetes, cardiovascular disease and death: Systematic review and meta-analysis. Diabetologia 2012, 55, 2895–2905. [Google Scholar] [CrossRef]
- Kishimoto, H.; Ohara, T.; Hata, J.; Ninomiya, T.; Yoshida, D.; Mukai, N.; Nagata, M.; Ikeda, F.; Fukuhara, M.; Kumagai, S.; et al. The long-term association between physical activity and risk of dementia in the community: The Hisayama Study. Eur. J. Epidemiol. 2016, 31, 267–274. [Google Scholar] [CrossRef]
- Penedo, F.J.; Dahn, J.R. Exercise and well-being: A review of mental and physical health benefits associated with physical activity. Curr. Opin. Psychiatry 2005, 18, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Yazawa, S.; Suetom, N.; Okamoto, K.; Namiki, T. Content of Capsaicinoids and Capsaicinoid-like Substances in Fruit of Pepper (Capsicum annuum) Hybrids Made with & prime; CH-19 Sweet & prime; as a Parent. J. Jpn. Soc. Hortic. Sci. 1989, 58, 601–607. [Google Scholar] [CrossRef]
- Kobata, K.; Todo, T.; Yazawa, S.; Iwai, K.; Watanabe, T. Novel Capsaicinoid-like Substances, Capsiate and Dihydrocapsiate, from the Fruits of a Nonpungent Cultivar, CH-19 Sweet, of Pepper (Capsicum annuum L.). J. Agric. Food Chem. 1998, 46, 1695–1697. [Google Scholar] [CrossRef]
- Luo, X.J.; Peng, J.; Li, Y.J. Recent advances in the study on capsaicinoids and capsinoids. Eur. J. Pharmacol. 2011, 650, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Masuda, Y.; Haramizu, S.; Oki, K.; Ohnuki, K.; Watanabe, T.; Yazawa, S.; Kawada, T.; Hashizume, S.; Fushiki, T. Upregulation of uncoupling proteins by oral administration of capsiate, a nonpungent capsaicin analog. J. Appl. Physiol. 2003, 95, 2408–2415. [Google Scholar] [CrossRef] [PubMed]
- Snitker, S.; Fujishima, Y.; Shen, H.; Ott, S.; Pi-Sunyer, X.; Furuhata, Y.; Sato, H.; Takahashi, M. Effects of novel capsinoid treatment on fatness and energy metabolism in humans: Possible pharmacogenetic implications. Am. J. Clin. Nutr. 2009, 89, 45–50. [Google Scholar] [CrossRef]
- Yoneshiro, T.; Aita, S.; Matsushita, M.; Kayahara, T.; Kameya, T.; Kawai, Y.; Iwanaga, T.; Saito, M. Recruited brown adipose tissue as an antiobesity agent in humans. J. Clin. Investig. 2013, 123, 3404–3408. [Google Scholar] [CrossRef]
- Franceschi, C.; Bonafe, M.; Valensin, S.; Olivieri, F.; de Luca, M.; Ottaviani, E.; de Benedictis, G. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef]
- Currais, A.; Fischer, W.; Maher, P.; Schubert, D. Intraneuronal protein aggregation as a trigger for inflammation and neurodegeneration in the aging brain. FASEB J. 2017, 31, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, Y.; Li, J.; Wang, D.; Wu, Y.; Li, Y.; Lu, Z.; Yu, S.C.; Li, R.; Yang, X. Cognitive deficits and decreased locomotor activity induced by single-walled carbon nanotubes and neuroprotective effects of ascorbic acid. Int. J. Nanomed. 2014, 9, 823–839. [Google Scholar] [CrossRef]
- Yamato, M.; Tamura, Y.; Eguchi, A.; Kume, S.; Miyashige, Y.; Nakano, M.; Watanabe, Y.; Kataoka, Y. Brain interleukin-1beta and the intrinsic receptor antagonist control peripheral Toll-like receptor 3-mediated suppression of spontaneous activity in rats. PLoS ONE 2014, 9, e90950. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, K.; Suzuki, K. Dihydrocapsiate improved age-associated impairments in mice by increasing energy expenditure. Am. J. Physiol. Endocrinol. Metab. 2017, 313, E586–E597. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sancho, R.; Lucena, C.; Macho, A.; Calzado, M.A.; Blanco-Molina, M.; Minassi, A.; Appendino, G.; Munoz, E. Immunosuppressive activity of capsaicinoids: Capsiate derived from sweet peppers inhibits NF-kappaB activation and is a potent antiinflammatory compound in vivo. Eur. J. Immunol. 2002, 32, 1753–1763. [Google Scholar] [CrossRef]
- Alexopoulos, G.S.; Morimoto, S.S. The inflammation hypothesis in geriatric depression. Geriatr. Psychiatry 2011, 26, 1109–1118. [Google Scholar] [CrossRef]
- Martínez-Cengotitabengoa, M.; Carrascón, L.; O’Brien, J.T.; Díaz-Gutiérrez, M.J.; Bermúdez-Ampudia, C.; Sanada, K.; Arrasate, M.; González-Pinto, A. Peripheral Inflammatory Parameters in Late-Life Depression: A Systematic Review. Int. J. Mol. Sci. 2016, 17, 2022. [Google Scholar] [CrossRef]
- Mori, H.; Kuge, H.; Sakaguchi, S.; Tanaka, T.H.; Miyazaki, J. Determination of symptoms associated with hiesho among young females using hie rating surveys. J. Integr. Med. 2018, 16, 34–38. [Google Scholar] [CrossRef]
- Unno, T.; Nagano, M.; Yamaguchi, Y.; Oohira, T. Effects of capsinoids-containing dietary supplements on sensation of cold constitution and measure of unidentified complaints in young women. Jpn. Pharmacol. Ther. 2011, 39, 525–531. [Google Scholar]
- Kimura, M.; Mizuta, C.; Yamada, Y.; Okayama, Y.; Nakamura, E. Constructing an index of physical fitness age for Japanese elderly based on 7-year longitudinal data: Sex differences in estimated physical fitness age. Age 2012, 34, 203–214. [Google Scholar] [CrossRef]
- Yamada, Y.; Hashii-Arishima, Y.; Yokoyama, K.; Itoi, A.; Adachi, T.; Kimura, M. Validity of a triaxial accelerometer and simplified physical activity record in older adults aged 64–96 years: A doubly labeled water study. Eur. J. Appl. Physiol. 2018, 118, 2133–2146. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Yokoyama, K.; Noriyasu, R.; Osaki, T.; Adachi, T.; Itoi, A.; Naito, Y.; Morimoto, T.; Kimura, M.; Oda, S. Light-intensity activities are important for estimating physical activity energy expenditure using uniaxial and triaxial accelerometers. Eur. J. Appl. Physiol. 2009, 105, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Konuma, H.; Hirose, H.; Yokoyama, K. Relationship of the Japanese Translation of the Profile of Mood States Second Edition (POMS 2®) to the First Edition (POMS®). Juntendo Med. J. 2015, 61, 517–519. [Google Scholar] [CrossRef]
- Ikenaga, M.; Yamada, Y.; Takeda, N.; Kimura, M.; Higaki, Y.; Tanaka, H.; Kiyonaga, A.; Nakagawa Study, G. Dynapenia, gait speed and daily physical activity measured using triaxial accelerometer in older Japanese men. J. Phys. Fit. Sports Med. 2014, 3, 147–154. [Google Scholar] [CrossRef]
- Ganpule, A.A.; Tanaka, S.; Ishikawa-Takata, K.; Tabata, I. Interindividual variability in sleeping metabolic rate in Japanese subjects. Eur. J. Clin. Nutr. 2007, 61, 1256–1261. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.F.; Schmidt, G.J.; Vandemark, F.L. Determination of epsilon-aminocaproic acid in serum by reversed-phase chromatography with fluorescence detection. Clin. Chem. 1977, 23, 1226–1229. [Google Scholar]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Gando, Y.; Murakami, H.; Kawakami, R.; Tanaka, N.; Sanada, K.; Tabata, I.; Higuchi, M.; Miyachi, M. Light-intensity physical activity is associated with insulin resistance in elderly Japanese women independent of moderate-to vigorous-intensity physical activity. J. Phys. Act. Health 2014, 11, 266–271. [Google Scholar] [CrossRef]
- Izawa, K.P.; Shibata, A.; Ishii, K.; Miyawaki, R.; Oka, K. Associations of low-intensity light physical activity with physical performance in community-dwelling elderly Japanese: A cross-sectional study. PLoS ONE 2017, 12, e0178654. [Google Scholar] [CrossRef]
- Reinholdsson, M.; Palstam, A.; Sunnerhagen, K.S. Prestroke physical activity could influence acute stroke severity (Part of Papsigot). Neurology 2018, 91, e1461–e1467. [Google Scholar] [CrossRef]
- Buman, M.P.; Hekler, E.B.; Haskell, W.L.; Pruitt, L.; Conway, T.L.; Cain, K.L.; Sallis, J.F.; Saelens, B.E.; Frank, L.D.; King, A.C. Objective light-intensity physical activity associations with rated health in older adults. Am. J. Epidemiol. 2010, 172, 1155–1165. [Google Scholar] [CrossRef] [PubMed]
- Yashiro, K.; Tonson, A.; Pecchi, E.; Vilmen, C.; Le Fur, Y.; Bernard, M.; Bendahan, D.; Giannesini, B. Capsiate supplementation reduces oxidative cost of contraction in exercising mouse skeletal muscle in vivo. PLoS ONE 2015, 10, e0128016. [Google Scholar] [CrossRef] [PubMed]
- Kazuya, Y.; Tonson, A.; Pecchi, E.; Dalmasso, C.; Vilmen, C.; Fur, Y.L.; Bernard, M.; Bendahan, D.; Giannesini, B. A single intake of capsiate improves mechanical performance and bioenergetics efficiency in contracting mouse skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E1110–E1119. [Google Scholar] [CrossRef] [PubMed]
- Haramizu, S.; Mizunoya, W.; Masuda, Y.; Ohnuki, K.; Watanabe, T.; Yazawa, S.; Fushiki, T. Capsiate, a nonpungent capsaicin analog, increases endurance swimming capacity of mice by stimulation of vanilloid receptors. Biosci. Biotechnol. Biochem. 2006, 70, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Yoneshiro, T. Capsinoids and related food ingredients activating brown fat thermogenesis and reducing body fat in humans. Curr. Opin. Lipidol. 2013, 24, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Nirengi, S.; Homma, T.; Inoue, N.; Sato, H.; Yoneshiro, T.; Matsushita, M.; Kameya, T.; Sugie, H.; Tsuzaki, K.; Saito, M.; et al. Assessment of human brown adipose tissue density during daily ingestion of thermogenic capsinoids using near-infrared time-resolved spectroscopy. J. Biomed. Opt. 2016, 21, 091305. [Google Scholar] [CrossRef]
- Takagi, A.; Yamaguchi, M.; Wakisaka, S.; Sakane, N.; Moritani, T.; Nagai, N. Resting energy expenditure and changes in body temperature and thermal sensation under thermoneutral environment in young women with or without consistently unusual coldness. J. Jpn. Soc. Psychosom. Obstet. Gynecol. 2012, 17, 193–205. [Google Scholar]
- Hachiya, S.; Kawabata, F.; Ohnuki, K.; Inoue, N.; Yoneda, H.; Yazawa, S.; Fushiki, T. Effects of CH-19 Sweet, a non-pungent cultivar of red pepper, on sympathetic nervous activity, body temperature, heart rate, and blood pressure in humans. Biosci. Biotechnol. Biochem. 2007, 71, 671–676. [Google Scholar] [CrossRef]
- Frank, S.M.; Raja, S.N.; Bulcao, C.; Goldstein, D.S. Age-related thermoregulatory differences during core cooling in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 279, R349–R354. [Google Scholar] [CrossRef]
| ND | ND + DHCte (0.3%) | |
|---|---|---|
| Casein | 20 | 20 |
| Sucrose | 10 | 10 |
| Cornstarch | 39.8 | 39.8 |
| α-cornstarch | 13.2 | 13.2 |
| l-cysteine | 0.3 | 0.3 |
| Cellulose | 5 | 5 |
| Lard | 7 | 7 |
| Mineral mix | 3.5 | 3.5 |
| Vitamin mix | 1 | 1 |
| Choline bitartrate | 0.25 | 0.25 |
| Dihydrocapsiate | - | 0.3 |
| Gene | Sense | Antisense | Entrez Gene ID |
|---|---|---|---|
| Cat | AGCGACCAGATGAAGCAGTG | TCCGCTCTCTGTCAAAGTGTG | 12359 |
| Ccl3 | TTCTCTGTACCATGACACTCTGC | CGTGGAATCTTCCGGCTGTAG | 20302 |
| Ccl5 | GCTGCTTTGCCTACCTCTCC | TCGAGTGACAAACACGACTGC | 20304 |
| Cxcl1 | CTGGGATTCACCTCAAGAACATC | CAGGGTCAAGGCAAGCCTC | 14825 |
| Cxcl10 | CCAAGTGCTGCCGTCATTTTC | GGCTCGCAGGGATGATTTCAA | 15945 |
| Icam1 | GTGATGCTCAGGTATCCATCCA | CACAGTTCTCAAAGCACAGCG | 15894 |
| Il6 | TCCAGCCAGTTGCCTTCTTGG | TCTGACAGTGCATCATCGCTG | 16193 |
| Sod1 | AACCAGTTGTGTTGTCAGGAC | CCACCATGTTTCTTAGAGTGAGG | 20655 |
| Sod2 | CAGACCTGCCTTACGACTATGG | CTCGGTGGCGTTGAGATTGTT | 20656 |
| Tgfβ1 | CTCCCGTGGCTTCTAGTGC | GCCTTAGTTTGGACAGGATCTG | 21803 |
| Tbp | ACCCTTCACCAATGACTCCTATG | TGACTGCAGCAAATCGCTTGG | 21374 |
| 18S | GTGGAGCGATTTGTCTGGTT | AACGCCACTTGTCCCTCTAA | 19791 |
| CP (n = 26) | PL (n = 21) | |||||||
|---|---|---|---|---|---|---|---|---|
| BL | 4 w | 8 w | 12 w | BL | 4 w | 8 w | 12 w | |
| Amount of VPA, METs × h/week | 0.7 ± 2.8 | 0.5 ± 1.5 | 0.6 ± 1.7 | 0.9 ± 2.9 | 0.0 ± 0.0 | 0.2 ± 0.3 | 0.8 ± 3.0 | 2.7 ± 9.4 ‡ |
| Time of VPA, min/day | 0.9 ± 3.7 | 0.5 ± 1.8 | 0.7 ± 2.1 | 1.1 ± 3.7 | 0.0 ± 0.1 | 0.2 ± 0.4 | 1.0 ± 3.9 | 3.3 ± 11.8 ‡ |
| Amount of LMPA, METs × h/week | 103.0 ± 28.2 | 108.0 ± 26.2 | 108.3 ± 28.6 | 108.2 ± 28.3 | 104.6 ± 19.8 | 108.8 ± 20.7 | 112.2 ± 20.0 | 115.2 ± 23.6 |
| Time of LMPA, min/day | 395.3 ± 105.2 | 418.5 ± 92.6 | 419.9 ± 101.1 | 420.4 ± 103.2 | 407.0 ± 79.1 | 419.8 ± 83.1 | 428.2 ± 82.7 | 440.6 ± 95.1 |
| Sedentary time, min/day | 477.5 ± 100.5 | 469.4 ± 98.1 | 486.9 ± 118.3 | 496.9 ± 122.2 | 464.1 ± 82.6 | 478.3 ± 92.1 | 477.2 ± 101.4 | 460.0 ± 115.9 |
| Energy expenditure in physical activity, kcal/day | 594.6 ± 167.1 | 619.7 ± 168.8 | 617.5 ± 154.3 | 619.9 ± 155.5 | 565.0 ± 115.6 | 593.3 ± 126.0 | 620.3 ± 126.7 † | 653.3 ± 166.3 ††† |
| CP (n = 13) | PL (n = 11) | |||||||
|---|---|---|---|---|---|---|---|---|
| BL | 4w | 8w | 12w | BL | 4w | 8w | 12w | |
| Amount of VPA, METs×hour/week | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.1 ± 0.2 | 0.0 ± 0.0 | 0.0 ± 0.1 | 0.2 ± 0.3 | 1.3 ± 4.1 | 4.2 ± 12.8 |
| Time of VPA, min/day | 0.0 ± 0.1 | 0.0 ± 0.0 | 0.1 ± 0.2 | 0.0 ± 0.0 | 0.1 ± 0.1 | 0.3 ± 0.4 | 1.8 ± 5.4 | 5.2 ± 16.1 |
| Amount of LMPA, METs×hour/week | 84.5 ± 17.2 | 95.4 ± 21.1 | 98.8 ± 22.8 | 99.2 ± 24.9* | 99.7 ± 23.3 | 103.8 ± 23.5 | 105.2 ± 20.8 | 103.8 ± 21.9 |
| Time of LMPA, min/day | 329.0 ± 63.0 | 374.8 ± 72.5 | 386.6 ± 78.1 * | 389.4 ± 84.2 ** | 385.6 ± 94.2 | 395.6 ± 93.6 | 395.3 ± 84.8 | 391.0 ± 86.3 |
| Sedentary time, min/day | 558.9 ± 62.9 | 532.9 ± 81.8 | 559.2 ± 93.6 | 576.9 ± 90.7 | 524.4 ± 57.7 | 541.3 ± 57.8 | 544.5 ± 75.2 | 533.3 ± 89.0 |
| Energy expenditure in physical activity, kcal/day | 481.2 ± 96.3 | 542.7 ± 127.1 * | 560.9 ± 129.5 ** | 562.5 ± 145.5 *** | 536.8 ± 112.2 | 565.7 ± 109.7 | 586.4 ± 101.7 | 598.6 ± 127.6 |
| CP (n = 36) | PL (n = 33) | |||||
|---|---|---|---|---|---|---|
| BL | 6 w | 12 w | BL | 6 w | 12 w | |
| Body weight, kg | 57.2 ± 8.2 | 57.2 ± 8.4 | 57.0 ± 8.1 | 56.3 ± 7.5 | 56.3 ± 7.3 | 56.3 ± 7.3 |
| BMI, kg/m2 | 23.3 ± 2.3 | 23.3 ± 2.4 | 23.2 ± 2.5 | 23.5 ± 2.8 | 23.5 ± 2.8 | 23.6 ± 2.7 |
| Abdominal circumference, cm | 86.0 ± 7.5 | 85.4 ± 7.0 ** | 86.3 ± 7.1 | 83.3 ± 8.6 | 85.0 ± 8.1 | 84.0 ± 8.3 |
| Percent body fat, % | 35.0 ± 6.5 | 34.0 ± 6.3 | 33.2 ± 6.3 † | 34.9 ± 7.2 | 34.2 ± 8.1 | 34.2 ± 7.3 |
| Visceral fat index | 103.8 ± 31.6 | 102.2 ± 25.4 * | 103.9 ± 24.1 | 98.9 ± 25.2 | 102.7 ± 24.4 | 98.2 ± 24.8 |
| Muscle mass, kg | 14.1 ± 2.6 | 14.2 ± 2.1 | 14.3 ± 2.1 | 14.1 ± 1.9 | 14.3 ± 2.1 | 14.2 ± 2.1 |
| Muscle mass percentage, % | 25.1 ± 2.4 | 25.2 ± 2.3 | 25.4 ± 2.4 | 25.1 ± 2.7 | 25.5 ± 3.0 | 25.3 ± 2.5 |
| Fat free mass, kg | 36.1 ± 8.6 | 33.9 ± 12.9 | 38.0 ± 6.1 | 36.5 ± 5.6 | 34.8 ± 10.3 | 36.9 ± 5.7 |
| CP (n = 13) | PL (n = 14) | |||||
|---|---|---|---|---|---|---|
| BL | 6 w | 12 w | BL | 6 w | 12 w | |
| Body weight, kg | 59.2 ± 4.9 | 59.0 ± 5.0 | 58.8 ± 5.0 | 58.4 ± 4.5 | 58.3 ± 4.7 | 58.3 ± 4.7 |
| BMI, kg/m2 | 23.9 ± 0.6 | 23.8 ± 0.8 | 23.8 ± 0.9 | 24.6 ± 1.4 | 24.6 ± 1.5 | 24.7 ± 1.4 |
| Abdominal circumference, cm | 87.6 ± 4.6 | 87.2 ± 5.3 | 88.5 ± 4.3 | 85.1 ± 5.1 | 87.0 ± 4.0 | 86.2 ± 4.9 |
| Percent body fat, % | 37.7 ± 5.4 | 36.5 ± 4.8 | 35.4 ± 4.1 *,† | 37.0 ± 7.0 | 36.7 ± 6.7 | 36.3 ± 7.0 |
| Visceral fat index | 99.6 ± 28.8 | 100.0 ± 19.3 | 102.3 ± 20.6 | 100.4 ± 25.6 | 105.0 ± 23.6 | 100.7 ± 23.5 |
| Muscle mass, kg | 37.0 ± 5.8 | 37.6 ± 5.8 | 38.1 ± 5.3 | 36.9 ± 5.8 | 37.0 ± 5.9 | 37.3 ± 6.1 |
| Muscle mass percentage, % | 14.1 ± 3.4 | 14.8 ± 1.9 | 14.7 ± 1.9 | 14.6 ± 2.3 | 14.7 ± 2.4 | 14.7 ± 2.4 |
| Fat free mass, kg | 24.7 ± 2.2 | 25.0 ± 1.8 | 25.1 ± 2.2 | 24.9 ± 3.0 | 25.2 ± 2.8 | 25.2 ± 2.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yokoyama, K.; Yamada, Y.; Akamatsu, Y.; Yoshinaka, Y.; Yamamoto, A.; Koizumi, T.; Ohyama, K.; Suzuki, K.; Hashimoto, M.; Sato, H.; et al. Effects of Capsinoids on Daily Physical Activity, Body Composition and Cold Hypersensitivity in Middle-Aged and Older Adults: A Randomized Study. Nutrients 2020, 12, 212. https://doi.org/10.3390/nu12010212
Yokoyama K, Yamada Y, Akamatsu Y, Yoshinaka Y, Yamamoto A, Koizumi T, Ohyama K, Suzuki K, Hashimoto M, Sato H, et al. Effects of Capsinoids on Daily Physical Activity, Body Composition and Cold Hypersensitivity in Middle-Aged and Older Adults: A Randomized Study. Nutrients. 2020; 12(1):212. https://doi.org/10.3390/nu12010212
Chicago/Turabian StyleYokoyama, Keiichi, Yosuke Yamada, Yasunori Akamatsu, Yasuko Yoshinaka, Akiko Yamamoto, Tomonori Koizumi, Kana Ohyama, Katsuya Suzuki, Masaki Hashimoto, Hitoshi Sato, and et al. 2020. "Effects of Capsinoids on Daily Physical Activity, Body Composition and Cold Hypersensitivity in Middle-Aged and Older Adults: A Randomized Study" Nutrients 12, no. 1: 212. https://doi.org/10.3390/nu12010212
APA StyleYokoyama, K., Yamada, Y., Akamatsu, Y., Yoshinaka, Y., Yamamoto, A., Koizumi, T., Ohyama, K., Suzuki, K., Hashimoto, M., Sato, H., & Kimura, M. (2020). Effects of Capsinoids on Daily Physical Activity, Body Composition and Cold Hypersensitivity in Middle-Aged and Older Adults: A Randomized Study. Nutrients, 12(1), 212. https://doi.org/10.3390/nu12010212






