Oxidative Stress Parameters in the Liver of Growing Male Rats Receiving Various Alcoholic Beverages
Abstract
1. Introduction
2. Materials and Methods
2.1. Characteristics of the Studied Animals
2.2. Experimental Procedures
2.3. Analytical Methods and Measurements
2.4. Statistical Analysis
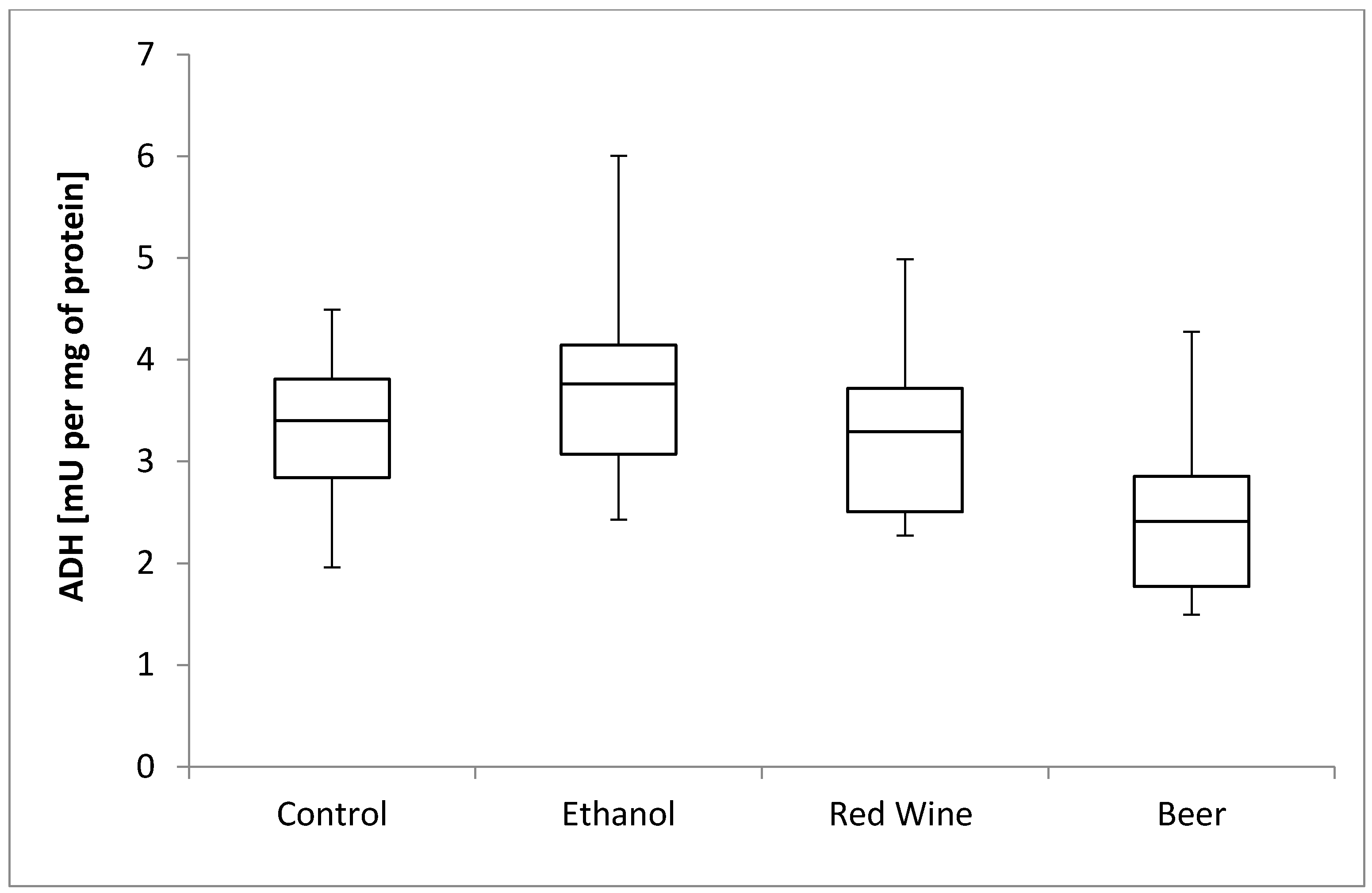
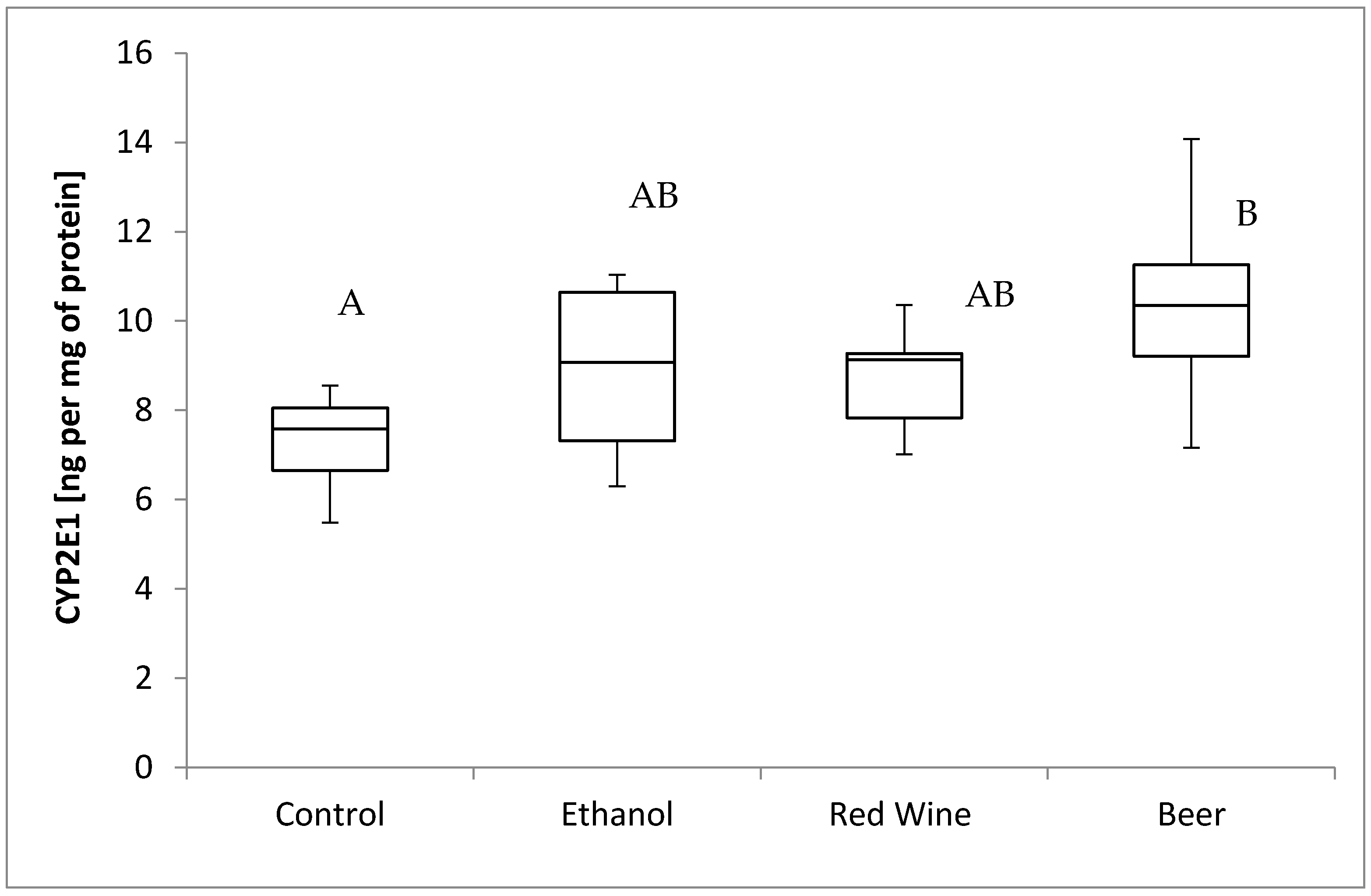
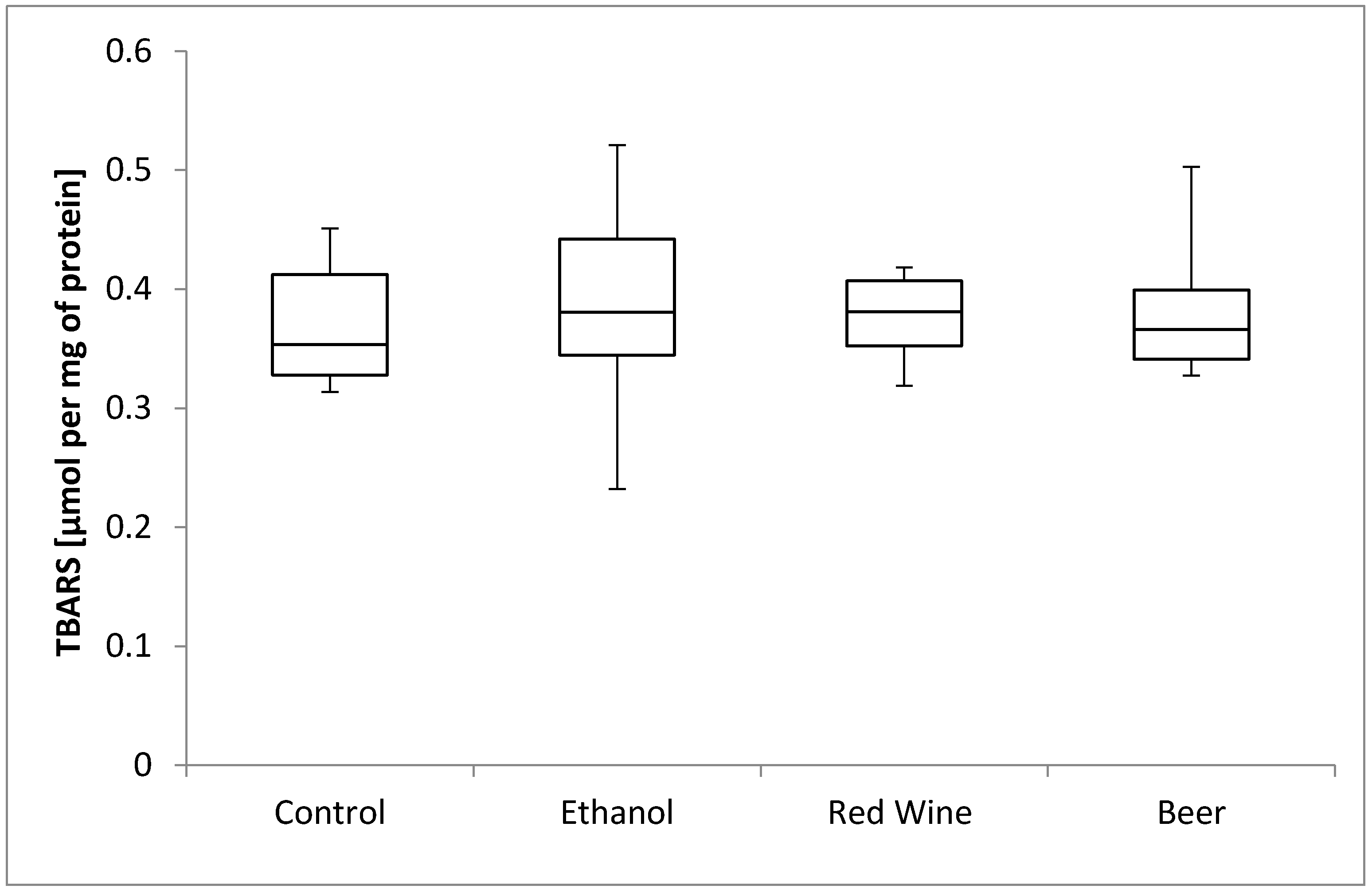
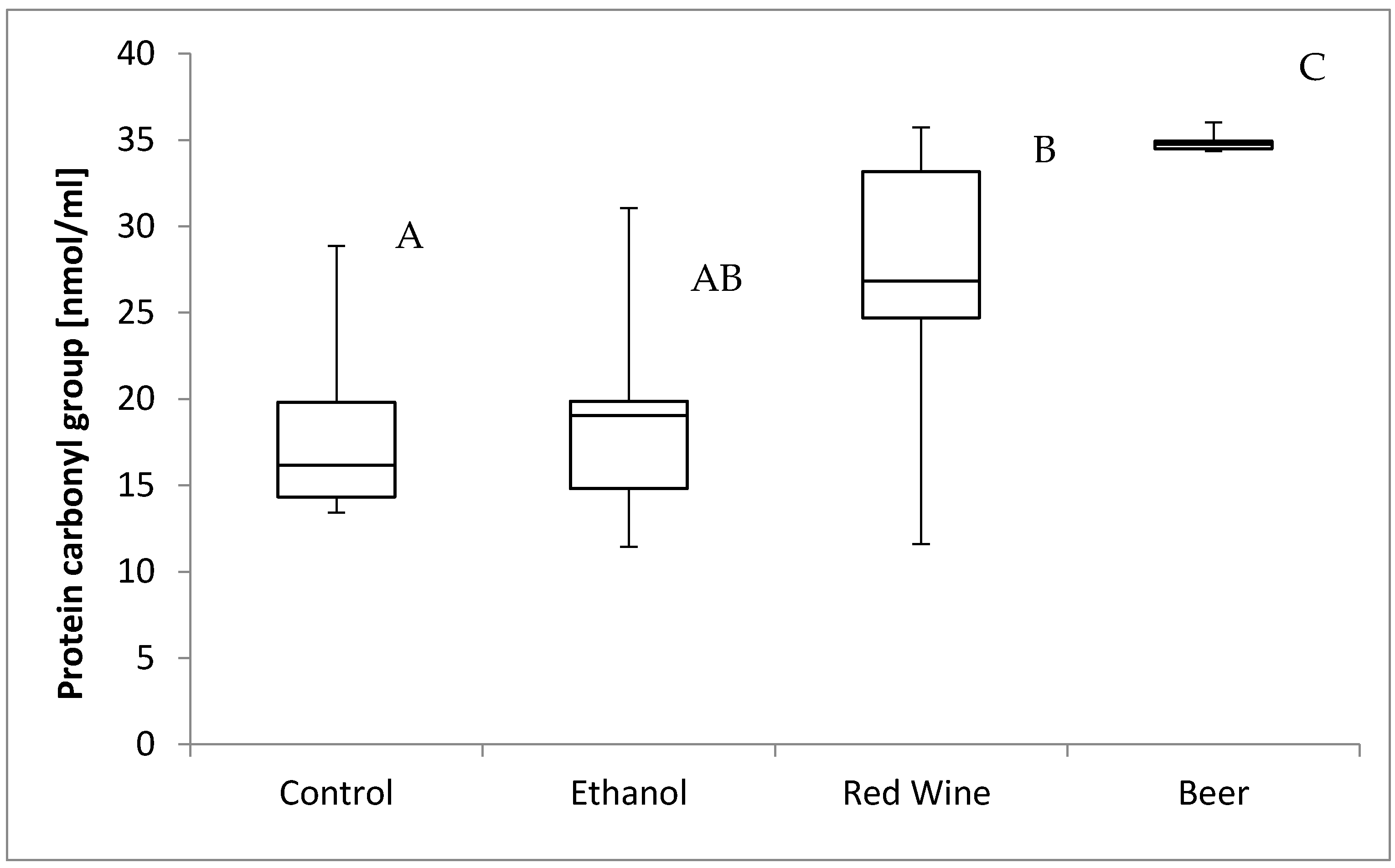
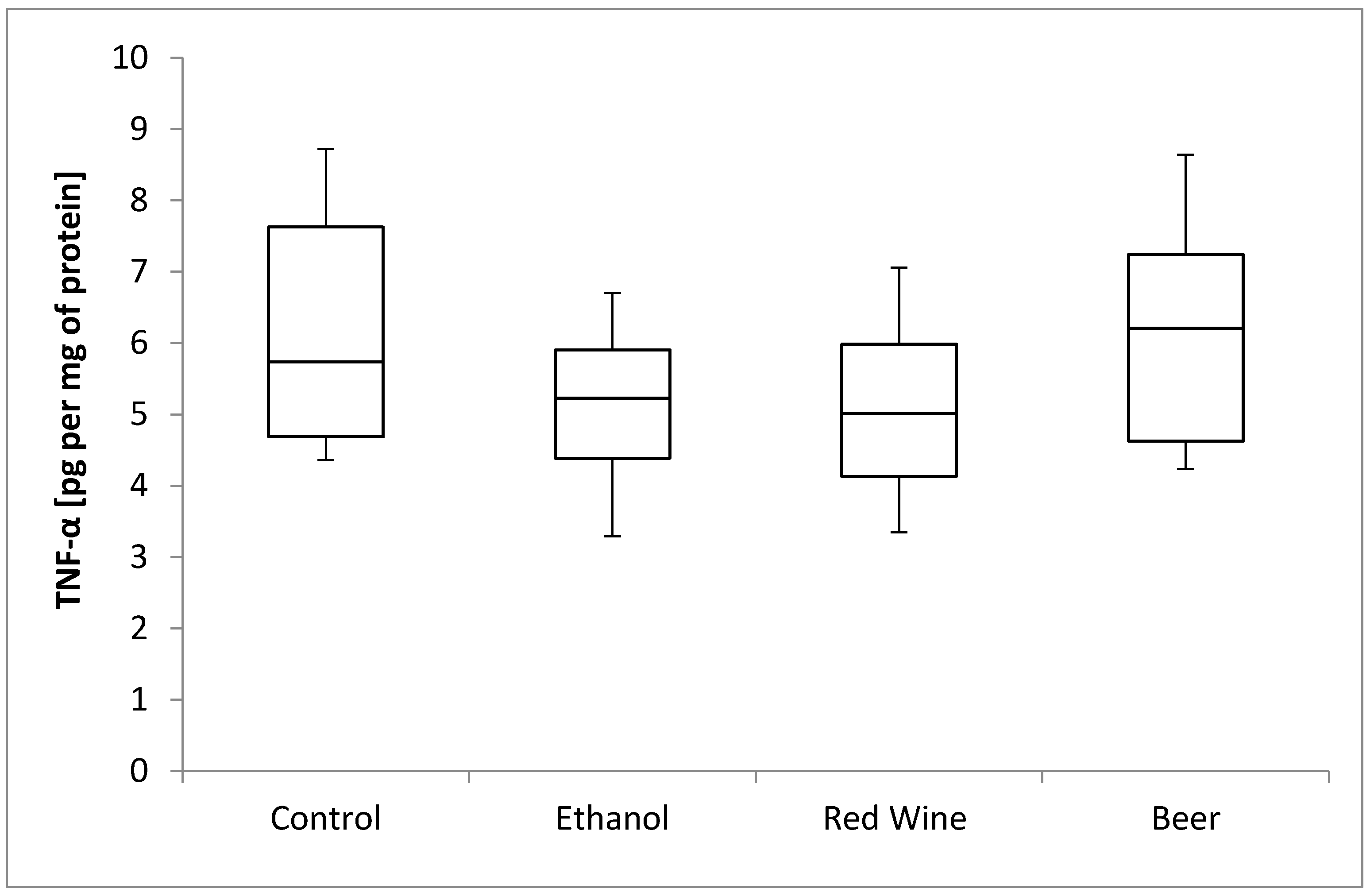
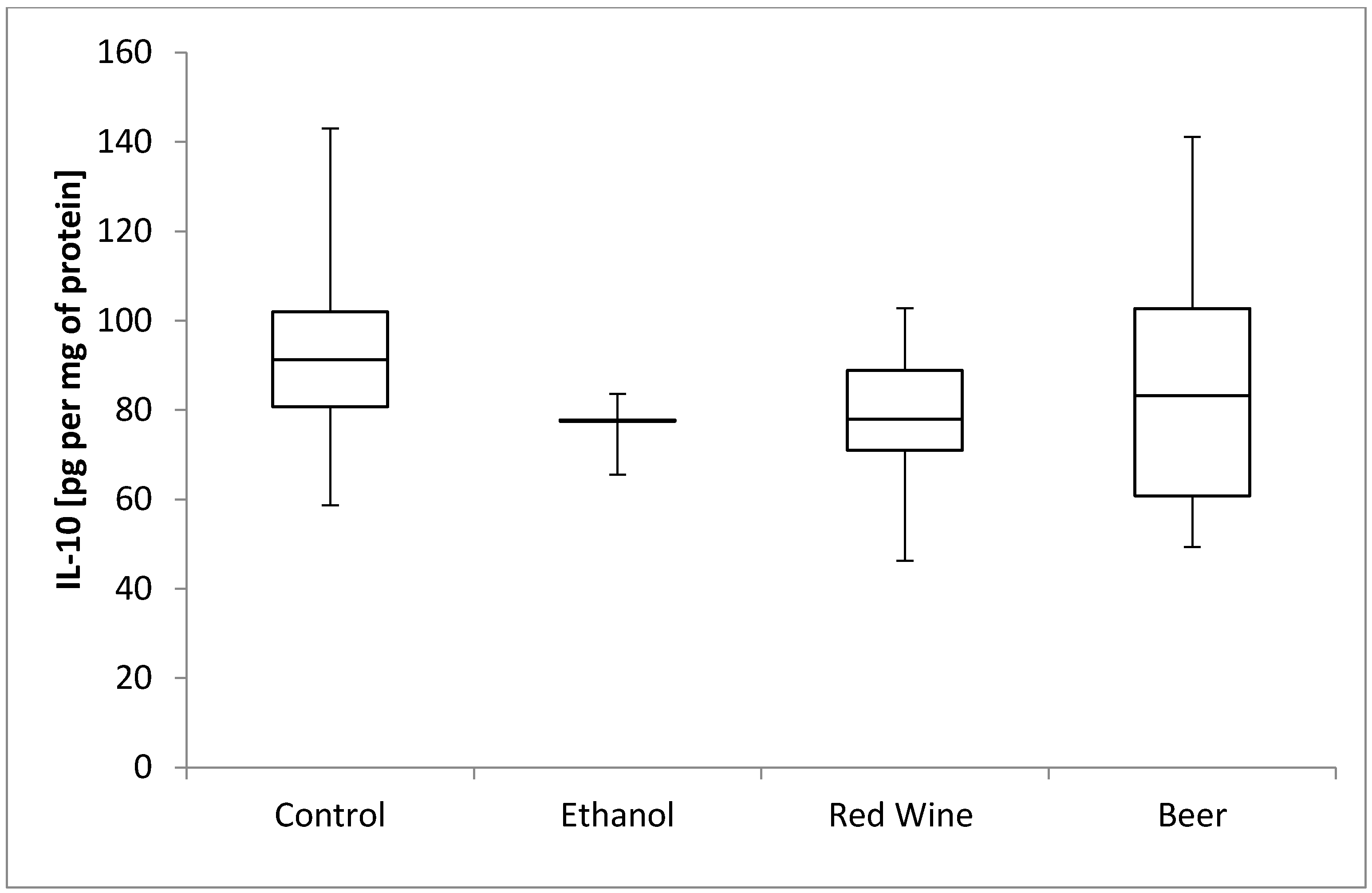
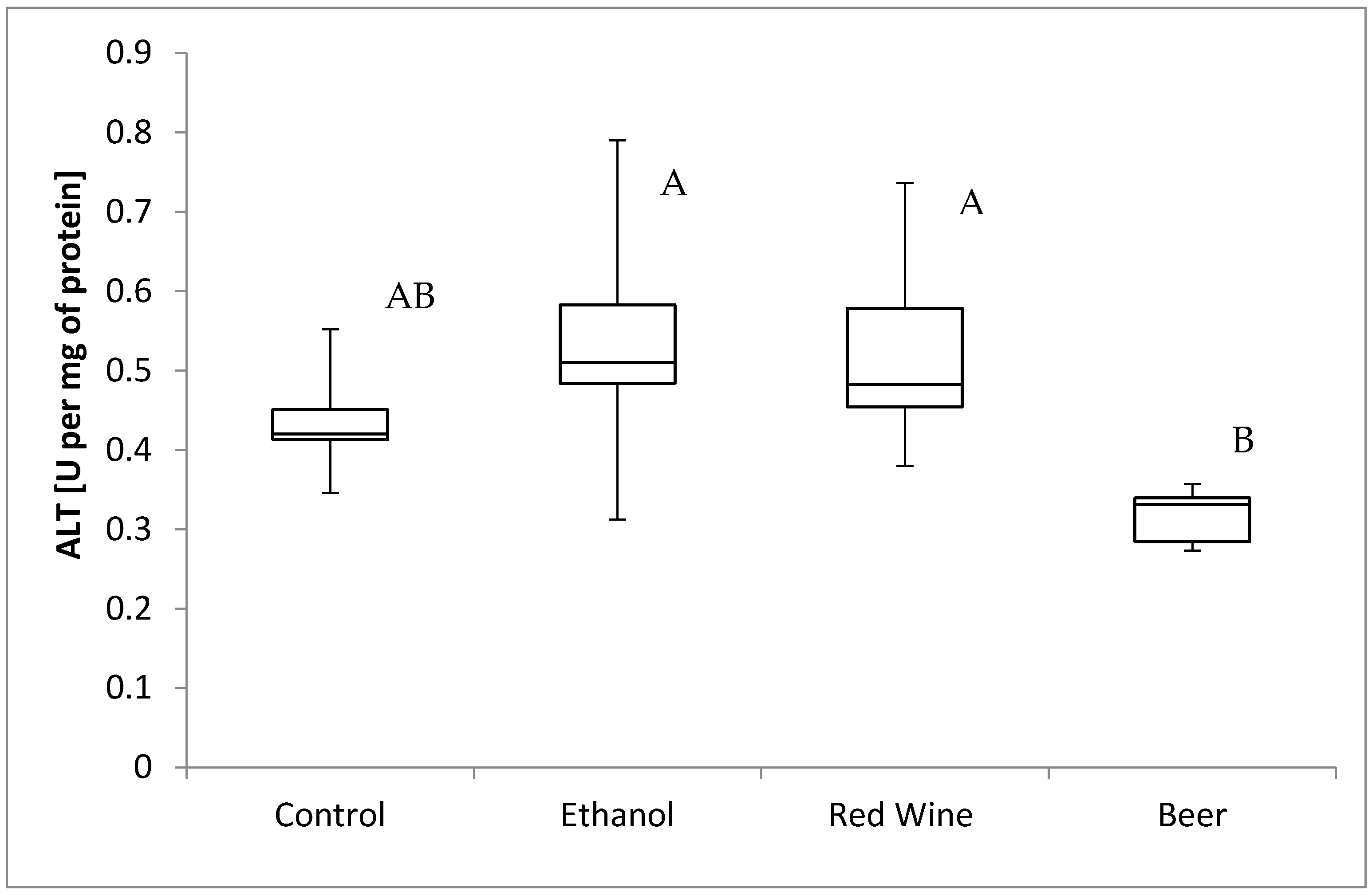
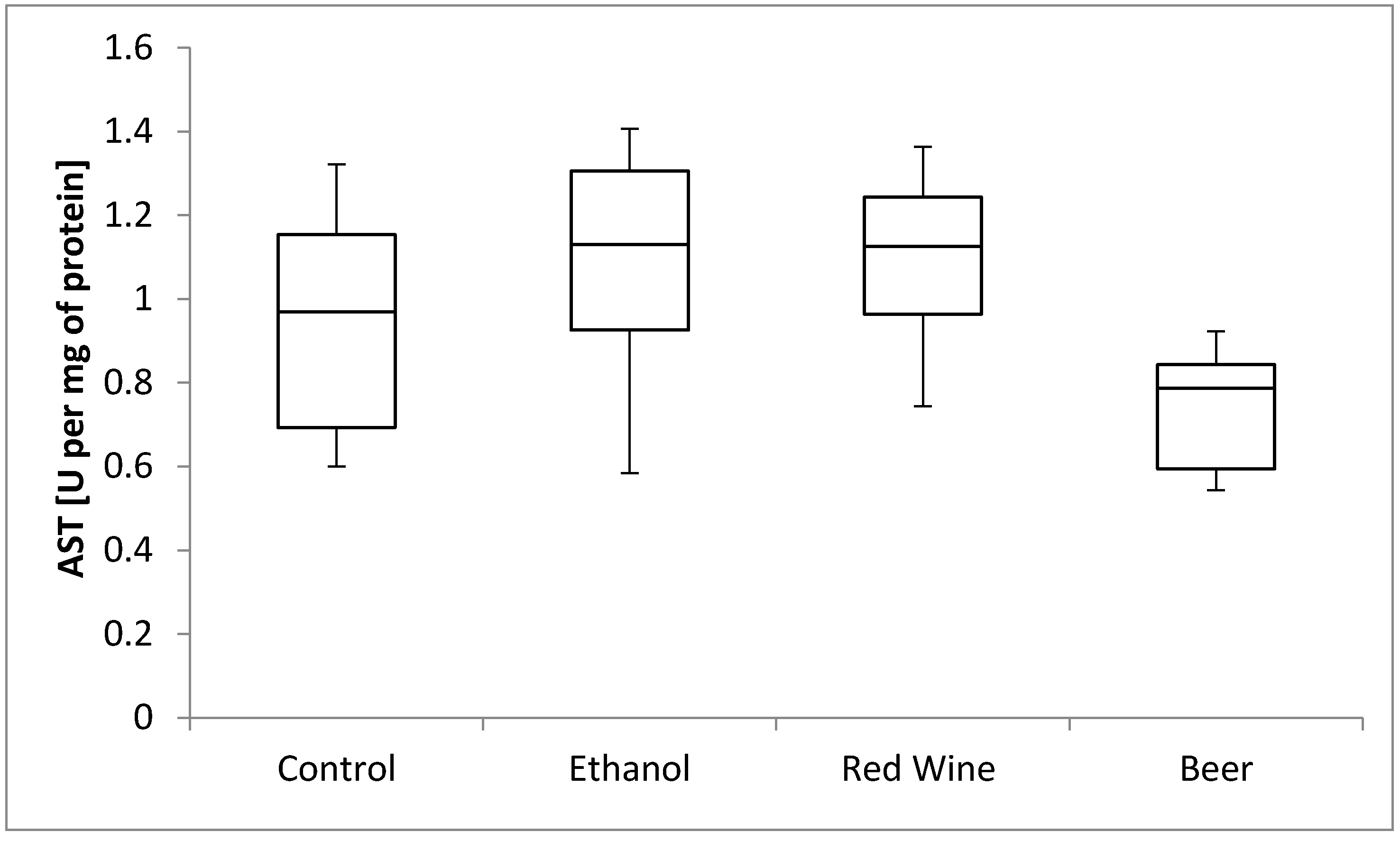
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Global Status Report on Alcohol and Health 2018; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Kuntsche, E.; Gmel, G. Alcohol consumption in late adolescence and early adulthood—Where is the problem? Swiss Med. Wkly. 2013, 143, w13826. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.A.; Miloh, T. Adolescent alcoholic liver disease. Clin. Liver Dis. 2019, 23, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, L.M.P.; Lopes, K.S.; Santana, L.N.S.; Fontes-Júnior, E.A.; Ribeiro, C.H.M.A.; Silva, M.C.F.; de Oliveira Paraense, R.S.; Crespo-López, M.E.; Gomes, A.R.Q.; Lima, R.R.; et al. Repeated cycles of binge-like ethanol intake in adolescent female rats induce motor function impairment and oxidative damage in motor cortex and liver, but not in blood. Oxid. Med. Cell. Longev. 2018, 2018, 3467531. [Google Scholar] [CrossRef]
- Bosetti, C.; Levi, F.; Lucchini, F.; Zatonski, W.A.; Negri, E.; La Vecchia, C. Worldwide mortality from cirrhosis: An update to 2002. J. Hepatol. 2007, 46, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Hagström, H.; Hemmingsson, T.; Discacciati, A.; Andreasson, A. Alcohol consumption in late adolescence is associated with an increased risk of severe liver disease later in life. J. Hepatol. 2018, 68, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Datz, C.; Hampe, J.; Bataller, R. Pathophysiology and management of alcoholic liver disease: Update 2016. Gut Liver 2017, 11, 173–188. [Google Scholar] [CrossRef]
- Osna, N.A.; Donohue, T.M., Jr.; Kharbanda, K.K. Alcoholic liver disease: Pathogenesis and current management. Alcohol Res. 2017, 38, 147–161. [Google Scholar]
- Doycheva, I.; Watt, K.D.; Rifai, G.; Abou Mrad, R.; Lopez, R.; Zein, N.N.; Carey, W.D.; Alkhouri, N. Increasing burden of chronic liver disease among adolescents and young adults in the USA: A silent epidemic. Dig. Dis. Sci. 2017, 62, 1373–1380. [Google Scholar] [CrossRef]
- Huang, A.; Chang, B.; Sun, Y.; Liu, H.; Li, B.; Teng, G.; Zou, Z.S. Disease spectrum of alcoholic liver disease in Beijing 302 Hospital from 2002 to 2013. Medicine 2017, 96, e6163. [Google Scholar] [CrossRef]
- Sheron, N.; Olsen, N.; Gilmore, I. An evidence-based alcohol policy. Gut 2008, 57, 1341–1344. [Google Scholar] [CrossRef]
- Ceni, E.; Mello, T.; Galli, A. Pathogenesis of alcoholic liver disease: Role of oxidative metabolism. World J. Gastroenterol. 2014, 20, 17756–17772. [Google Scholar] [CrossRef]
- Li, S.; Tan, H.Y.; Wang, N.; Zhang, Z.J.; Lao, L.; Wong, C.W.; Feng, Y. The role of oxidative stress and antioxidants in liver diseases. Int. J. Mol. Sci. 2015, 16, 26087–26124. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R. Alcoholic liver disease: Alcohol metabolism, cascade of molecular mechanisms, cellular targets, and clinical aspects. Biomedicines 2018, 6, 106. [Google Scholar] [CrossRef] [PubMed]
- Meroni, M.; Longo, M.; Dongiovanni, P. Alcohol or gut Microbiota: Who is the guilty? Int. J. Mol. Sci. 2019, 20, 4568. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, F.; Lerma-Cabrera, J.M. Alcohol consumption among adolescents—Implications for public health. In Topics in Public Health; Claborn, D., Ed.; IntechOpen: London, UK, 2015; pp. 51–76. [Google Scholar] [CrossRef]
- Dogan, A.; Anuk, O.O. Investigation of the phytochemical composition and antioxidant properties of chinar (Platanus orientalis L.) leaf infusion against ethanol-induced oxidative stress in rats. Mol. Biol. Rep. 2019, 46, 3049–3061. [Google Scholar] [CrossRef]
- Hsu, J.Y.; Lin, H.H.; Hsu, C.C.; Chen, B.C.; Chen, J.H. Aqueous extract of Pepino (Solanum muriactum Ait) leaves ameliorate lipid accumulation and oxidative stress in alcoholic fatty liver disease. Nutrients 2018, 10, 931. [Google Scholar] [CrossRef] [PubMed]
- Kołota, A.; Głąbska, D.; Oczkowski, M.; Gromadzka-Ostrowska, J. Influence of alcohol consumption on body mass gain and liver antioxidant defense in adolescent growing male rats. Int. J. Environ. Res. Public Health 2019, 16, 2320. [Google Scholar] [CrossRef]
- Spear, L.P.; Swartzwelder, H.S. Adolescent alcohol exposure and persistence of adolescent-typical phenotypes into adulthood: A mini-review. Neurosci. Biobehav. Rev. 2014, 45, 1–8. [Google Scholar] [CrossRef]
- Serlin, H.; Torregrossa, M.M. Adolescent rats are resistant to forming ethanol seeking habits. Dev. Cogn. Neurosci. 2015, 16, 183–190. [Google Scholar] [CrossRef]
- Milat, A.M.; Mudnić, I.; Grković, I.; Ključević, N.; Grga, M.; Jerčić, I.; Jurić, D.; Ivanković, D.; Benzon, B.; Boban, M. Effects of white wine consumption on weight in rats: Do polyphenols matter? Oxid. Med. Cell. Longev. 2017, 2017, 8315803. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxidation in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Levine, R.L.; Williams, J.A.; Stadtman, E.R.; Shacter, E. Carbonyl assays for determination of oxidatively modified proteins. Methods Enzymol. 1994, 233, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Reznick, A.Z.; Packer, L. Oxidative damage to proteins: Spectrophotometric method for carbonyl assay. Methods Enzymol. 1994, 233, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Nogales, F.; Rua, R.M.; Ojeda, M.L.; Murillo, M.L.; Carreras, O. Oral or intraperitoneal binge drinking and oxidative balance in adolescent rats. Chem. Res. Toxicol. 2014, 27, 1926–1933. [Google Scholar] [CrossRef]
- Jotty, K.; Ojeda, M.L.; Nogales, F.; Murillo, M.L.; Carreras, O. Selenium dietary supplementation as a mechanism to restore hepatic selenoprotein regulation in rat pups exposed to alcohol. Alcohol 2013, 47, 545–552. [Google Scholar] [CrossRef]
- Yamasaki, K.; Sugamoto, K.; Arakawa, T.; Nishiyama, K.; Yamasaki, M. Chronic intake of high-dose of blueberry leaf extract does not augment the harmful effects of ethanol in rats. PeerJ 2019, 7, e6989. [Google Scholar] [CrossRef]
- Tahir, M.; Sultana, S. Chrysin modulates ethanol metabolism in Wistar rats: A promising role against organ toxicities. Alcohol Alcohol. 2011, 46, 383–392. [Google Scholar] [CrossRef]
- Chandrasekaran, K.; Swaminathan, K.; Mathan Kumar, S.; Clemens, D.L.; Dey, A. In vitro evidence for chronic alcohol and high glucose mediated increased oxidative stress and hepatotoxicity. Alcohol. Clin. Exp. Res. 2012, 36, 1004–1012. [Google Scholar] [CrossRef]
- Hidestrand, M.; Shankar, K.; Ronis, M.J.J.; Badger, M.T. Effects of light and dark beer on hepatic cytochrome P-450 expression in male rats receiving alcoholic beverages as part of total enteral nutrition. Alcohol. Clin. Exp. Res. 2005, 29, 888–895. [Google Scholar] [CrossRef]
- Henderson, M.C.; Miranda, C.L.; Stevens, J.F.; Deinzer, M.L.; Buhler, D.R. In vitro inhibition of human P450 enzymes by prenylated flavonoids from hops, Humulus lupulus. Xenobiotica 2000, 30, 235–251. [Google Scholar] [CrossRef] [PubMed]
- Doody, E.E.; Groebner, J.L.; Walker, J.R.; Frizol, B.M.; Tuma, D.J.; Fernandez, D.J.; Tuma, P.L. Ethanol metabolism by alcohol dehydrogenase or cytochrome P450 2E1 differentially impairs hepatic protein trafficking and growth hormone signaling. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 313, G558–G569. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, R.; Castillo, R.; Carrasco, R.; Huerta, P.; Moreno, M. Diminution of tissue lipid peroxidation in rats is related to the in vitro antioxidant capacity of wine. Life Sci. 2005, 76, 889–900. [Google Scholar] [CrossRef] [PubMed]
- Bindu, M.P.; Annamalai, P.T. Combined effect of alcohol and cigarette smoke on lipid peroxidation and antioxidant status in rats. Indian J. Biochem. Biophys. 2004, 41, 40–44. [Google Scholar]
- Pushpakiran, G.; Mahalakshmi, K.; Anuradha, C.V. Taurine restores ethanol-induced depletion of antioxidants and attenuates oxidative stress in rat tissues. Amino Acids 2004, 27, 91–96. [Google Scholar] [CrossRef]
- Yao, P.; Li, K.; Jin, Y.; Song, F.; Zhou, S.; Sun, X.; Nüssler, A.K.; Liu, L. Oxidative damage after chronic ethanol intake in rat tissues: Prophylaxis of Ginkgo biloba extract. Food Chem. 2006, 99, 305–314. [Google Scholar] [CrossRef]
- Devipriya, N.; Srinivasan, M.; Sudheer, A.R.; Menon, V.P. Effect of ellagic acid, a natural polyphenol, on alcohol-induced prooxidant and antioxidant imbalance: A drug dose dependent study. Singap. Med. J. 2007, 48, 311–318. [Google Scholar]
- Jayaraman, J.; Veerappan, M.; Namasivayam, N. Potential beneficial effect of naringenin on lipid peroxidation and antioxidant status in rats with ethanol-induced hepatotoxicity. J. Pharm. Pharmacol. 2009, 61, 1383–1390. [Google Scholar] [CrossRef]
- Galligan, J.J.; Smathers, R.L.; Fritz, K.S.; Epperson, L.E.; Hunter, L.E.; Petersen, D.R. Protein carbonylation in a murine model for early alcoholic liver disease. Chem. Res. Toxicol. 2012, 25, 1012–1021. [Google Scholar] [CrossRef]
- Harishekar, M.B.; Kiran, B. Effect of lead, alcohol and vitamin E on protein carbonyl content in rats. J. Appl. Pharm. Sci. 2011, 1, 154–156. [Google Scholar]
- Chienwichai, P.; Reamtong, O.; Boonyuen, U.; Pisitkun, T.; Somparn, P.; Tharnpoophasiam, P.; Worakhunpiset, S.; Topanurak, S. Hepatic protein carbonylation profiles induced by lipid accumulation and oxidative stress for investigating cellular response to non-alcoholic fatty liver disease in vitro. Proteome Sci. 2019, 17, 1. [Google Scholar] [CrossRef]
- Carvalho, D.O.; Oliveira, R.; Johansson, B.; Guido, L.F. Dose-dependent protective and inductive effects of xanthohumol on oxidative DNA damage in Saccharomyces cerevisiae. Food Technol. Biotechnol. 2016, 54, 60–69. [Google Scholar] [CrossRef]
- Jung, F.; Lippmann, T.; Brandt, A.; Jin, C.J.; Engstler, A.J.; Baumann, A. Moderate consumption of fermented alcoholic beverages diminishes diet-induced non-alcoholic fatty liver disease through mechanisms involving hepatic adiponectin signaling in mice. Eur. J. Nutr. 2019. [Google Scholar] [CrossRef]
- Hege, M.; Jung, F.; Sellmann, C.; Jin, C.; Ziegenhardt, D.; Hellerbrand, C.; Bergheim, I. An iso-α-acid-rich extract from hops (Humulus lupulus) attenuates acute alcohol-induced liver steatosis in mice. Nutrition 2018, 45, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Lassaletta, A.D.; Chu, L.M.; Elmadhun, N.Y.; Burgess, T.A.; Feng, J.; Robich, M.P.; Sellke, F.W. Cardioprotective effects of red wine and vodka in a model of endothelial dysfunction. J. Surg. Res. 2012, 178, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.P.; Rizvi, S.I. Resveratrol may protect plasma proteins from oxidation under conditions of oxidative stress in vitro. J. Braz. Chem. Soc. 2010, 21, 909–913. [Google Scholar] [CrossRef]
- de la Lastra, C.A.; Villegas, I. Resveratrol as an antioxidant and pro-oxidant agent: Mechanisms and clinical implications. Biochem. Soc. Trans. 2007, 35, 1156–1160. [Google Scholar] [CrossRef] [PubMed]
- Ozaras, R.; Tahan, V.; Aydin, S.; Uzun, H.; Kaya, S.; Santurk, H. N-acetylcysteine attenuates alcohol-induced oxidative stress in the rat. World J. Gastroenterol. 2003, 9, 125–128. [Google Scholar] [CrossRef]
- Vidhya, A.; Indira, M. Protective effect of quercetin in the regression of ethanol-induced hepatotoxicity. Indian J. Pharm. Sci. 2009, 71, 527–532. [Google Scholar] [CrossRef]
- Das, S.K.; Mukherjee, S.; Gupta, G.; Rao, D.N.; Vasudevan, D.M. Protective effect of resveratrol and vitamin E against ethanol-induced oxidative damage in mice: Biochemical and immunological basis. Indian J. Biochem. Biophys. 2010, 47, 32–37. [Google Scholar]
- Apte, U.M.; McRee, R.; Ramaiah, S.K. Hepatocyte proliferation is the possible mechanism for the transient decrease in liver injury during steatosis stage of alcoholic liver disease. Toxicol. Pathol. 2004, 32, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Biondo-Simões, M.L.P.; Matias, J.E.; Montibeller, G.R.; Siqueira, L.C.; Nunes, E.S.; Grassi, C.A. Effect of aging on liver regeneration in rats. Acta Cirúrgica Bras. 2006, 21, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Murali, B.; Korrapati, M.C.; Warbritton, A.; Latendresse, J.R.; Mehendale, H.M. Tolerance of aged Fischer 344 rats against chlordecone-amplified carbon tetrachloride toxicity. Mech. Ageing Dev. 2004, 125, 421–435. [Google Scholar] [CrossRef] [PubMed]
- Schmucker, D.L.; Sanchez, H. Liver regeneration and aging: A current perspective. Curr. Gerontol. Geriatr. Res. 2011, 2011, 526379. [Google Scholar] [CrossRef]
- Sánchez-Hidalgo, J.M.; Naranjo, A.; Ciria, R.; Ranchal, I.; Aguilar-Melero, P.; Ferrín, G.; Valverde, A.; Rufián, S.; López-Cillero, P.; Muntané, J.; et al. Impact of age on liver regeneration response to injury after partial hepatectomy in a rat model. J. Surg. Res. 2012, 175, e1–e9. [Google Scholar] [CrossRef]
- Pérez-Navero, J.L.; Benítez-Sillero, J.D.; Gil-Campos, M.; Guillén-del Castillo, M.; Tasset, I.; Túnez, I. Changes in oxidative stress biomarkers induced by puberty. An. Pediatr. 2009, 70, 424–428. [Google Scholar] [CrossRef]
- Guney, E.; Fatih Ceylan, M.; Tektas, A.; Alisik, M.; Ergin, M.; Goker, Z.; Senses Dinc, G.; Ozturk, O.; Korkmaz, A.; Eker, S.; et al. Oxidative stress in children and adolescents with anxiety disorders. J. Affect Disord. 2014, 156, 62–66. [Google Scholar] [CrossRef]
- Couto, M.; Barbosa, C.; Silva, D.; Rudnitskaya, A.; Delgado, L.; Moreira, A.; Rocha, S.M. Oxidative stress in asthmatic and non-asthmatic adolescent swimmers-A breathomics approach. Pediatr. Allergy Immunol. 2017, 28, 452–457. [Google Scholar] [CrossRef]
- Tauman, R.; Shalitin, S.; Lavie, L. Oxidative stress in obese children and adolescents with and without type 2 diabetes mellitus is not associated with obstructive sleep apnea. Sleep Breath. 2019, 23, 117–123. [Google Scholar] [CrossRef]
- Younis, R.M.; Wolstenholme, J.T.; Bagdas, D.; Bettinger, J.C.; Miles, M.F.; Damaj, M.I. Adolescent but not adult ethanol binge drinking modulates ethanol behavioral effects in mice later in life. Pharmacol. Biochem. Behav. 2019, 184, 172740. [Google Scholar] [CrossRef]
| Time | 20th Day | 21th–30th Day | From 31st Day, for Six Weeks | |
|---|---|---|---|---|
| Group | Control | Animals divided into groups | Acclimatization procedure with no alcoholic beverage | Experiment with no alcoholic beverage |
| Experimental (ethanol) | Acclimatization procedure with alcoholic beverage: —21st–22nd day—2% alc. vol. —23rd–24th day—4% alc. vol. —25th–26th day—6% alc. vol. —27th–28th day—8% alc. vol. —29th–30th day—10% alc. vol. | Experiment with alcoholic beverage (10% alc. vol.) | ||
| Experimental (beer) | ||||
| Experimental (red wine) | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kołota, A.; Głąbska, D.; Oczkowski, M.; Gromadzka-Ostrowska, J. Oxidative Stress Parameters in the Liver of Growing Male Rats Receiving Various Alcoholic Beverages. Nutrients 2020, 12, 158. https://doi.org/10.3390/nu12010158
Kołota A, Głąbska D, Oczkowski M, Gromadzka-Ostrowska J. Oxidative Stress Parameters in the Liver of Growing Male Rats Receiving Various Alcoholic Beverages. Nutrients. 2020; 12(1):158. https://doi.org/10.3390/nu12010158
Chicago/Turabian StyleKołota, Aleksandra, Dominika Głąbska, Michał Oczkowski, and Joanna Gromadzka-Ostrowska. 2020. "Oxidative Stress Parameters in the Liver of Growing Male Rats Receiving Various Alcoholic Beverages" Nutrients 12, no. 1: 158. https://doi.org/10.3390/nu12010158
APA StyleKołota, A., Głąbska, D., Oczkowski, M., & Gromadzka-Ostrowska, J. (2020). Oxidative Stress Parameters in the Liver of Growing Male Rats Receiving Various Alcoholic Beverages. Nutrients, 12(1), 158. https://doi.org/10.3390/nu12010158








