The Effect of 12 Weeks of β-Hydroxy-β-Methyl-Butyrate Supplementation after Liver Transplantation: A Pilot Randomized Controlled Study
Abstract
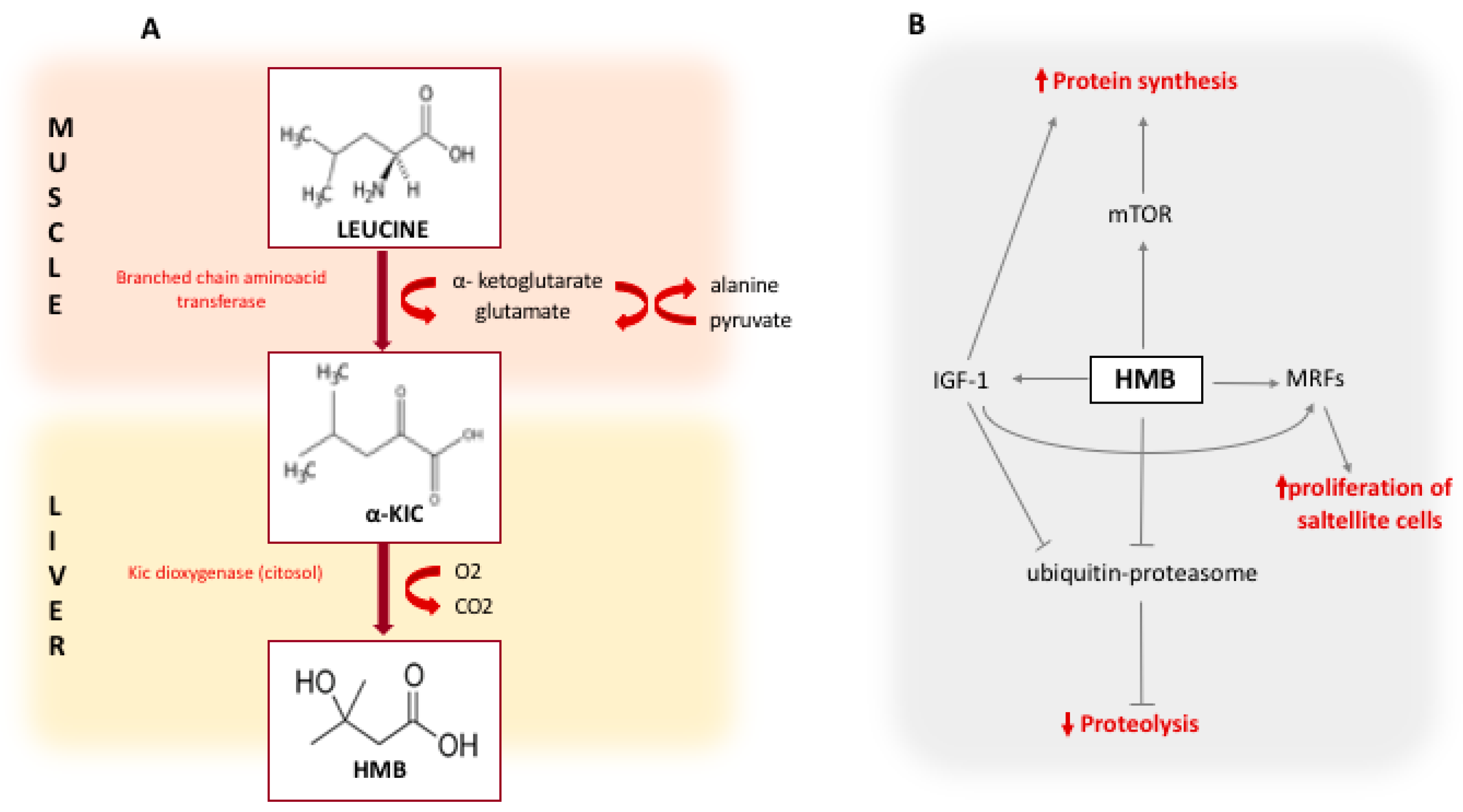
1. Introduction
2. Materials and Methods
3. Statistical Analysis
4. Results
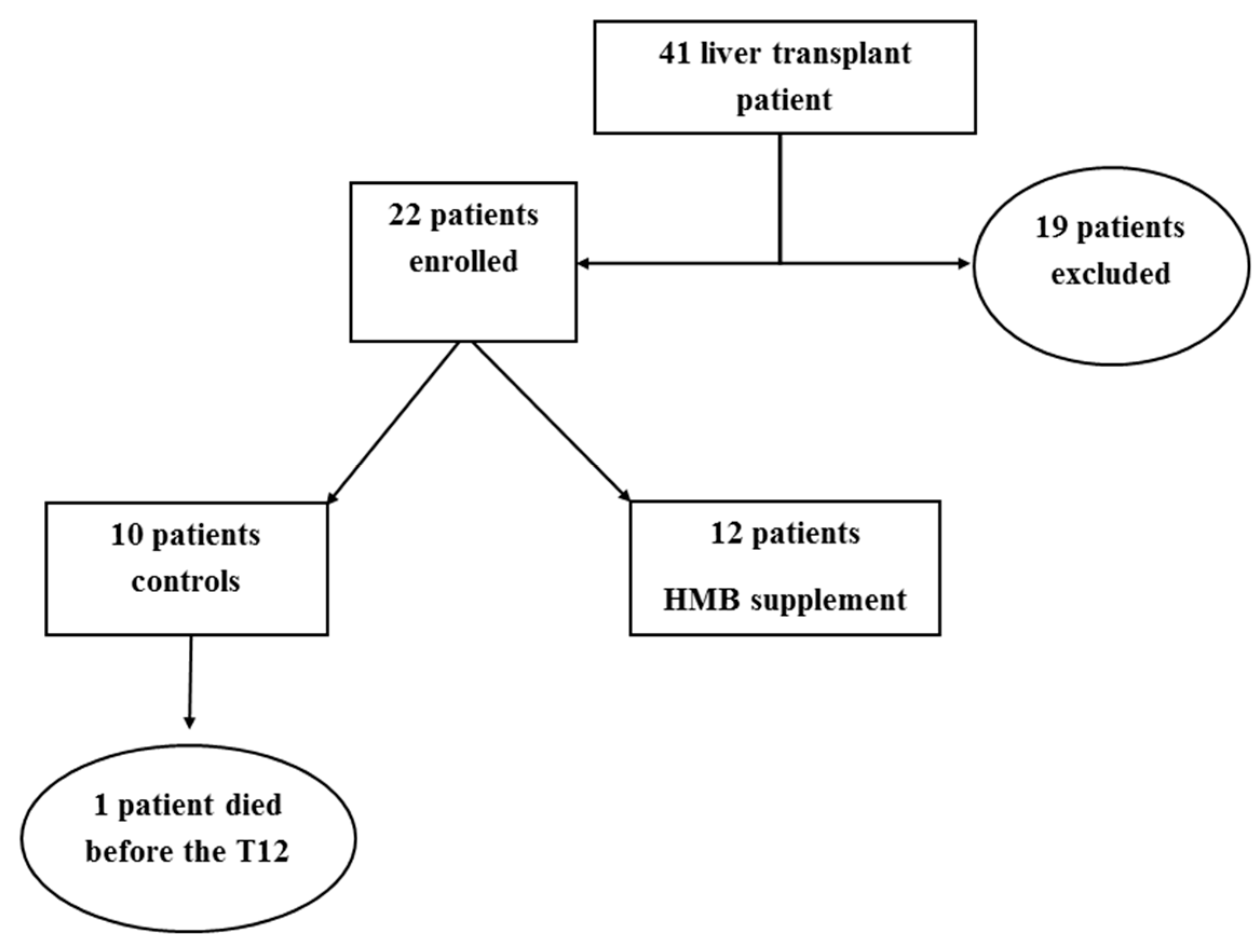
4.1. Study Population and Baseline Characteristic
4.2. Safety and Tolerability
4.3. Modifications of Muscle Mass Evaluated by DEXA
4.4. Modification of Muscle Mass Evaluated by Anthropometry
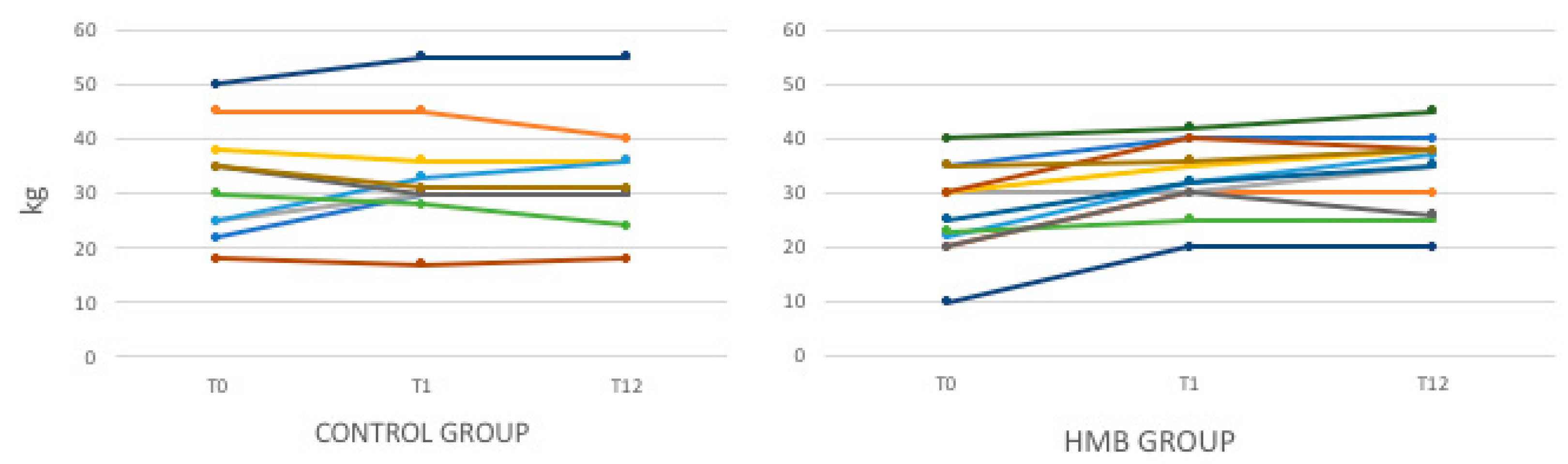
4.5. Muscle Strength and Physical Performance
5. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| β-hydroxy-β-methyl-butyrate | HMB |
| liver transplantation | LT |
| dual energy X-ray absorptiometry | DEXA |
| Appendicular skeletal muscle index | ASMI |
| Mid Arm Muscle Circumference | MAMC |
| Triceps Skinfold-Thickness | TSF |
| fat free mass index | FFMI |
| fat mass index | FMI |
| 6-min walk test | 6MWT |
| Timed Up and Go Test | TUG |
| Hand grip strength | HG |
| branched chain amino acids | BCAA |
References
- Amodio, P.; Bemeur, C.; Butterworth, R.; Cordoba, J.; Kato, A.; Montagnese, S.; Uribe, M.; Vilstrup, H.; Morgan, M.Y. The nutritional management of hepatic encephalopathy in patients with cirrhosis: International Society for Hepatic Encephalopathy and Nitrogen Metabolism Consensus. Hepatology 2013, 58, 325–336. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines on nutrition in chronic liver disease. J. Hepatol. 2019, 70, 172–193. [Google Scholar]
- Plauth, M.; Bernal, W.; Dasarathy, S.; Merli, M.; Plank, L.D.; Schütz, T.; Bischoff, S.C. ESPEN guideline on clinical nutrition in liver disease. Clin. Nutr. 2019, 38, 485–521. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, in press. [Google Scholar] [CrossRef]
- Montano-Loza, A.J.; Meza-Junco, J.; Prado, C.M.; Lieffers, J.R.; Baracos, V.E.; Bain, V.G.; Sawyer, M.B. Muscle wasting is associated with mortality in patients with cirrhosis. Clin. Gastroenterol. Hepatol. 2012, 10, 166–173. [Google Scholar] [CrossRef]
- Englesbe, M.J.; Patel, S.P.; He, K.; Lynch, R.J.; Schaubel, D.E.; Harbaugh, C.; Holcombe, S.A.; Wang, S.C.; Segev, D.L.; Sonnenday, C.J. Sarcopenia and mortality after liver transplantation. J. Am. Coll. Surg. 2010, 211, 271–278. [Google Scholar] [CrossRef]
- Merli, M.; Giusto, M.; Lucidi, C.; Giannelli, V.; Pentassuglio, I.; Di Gregorio, V.; Lattanzi, B.; Riggio, O. Muscle depletion increases the risk of overt and minimal hepatic encephalopathy: Results of a prospective study. Metab. Brain Dis. 2013, 28, 281–284. [Google Scholar] [CrossRef]
- Lucidi, C.; Lattanzi, B.; Di Gregorio, V.; Incicco, S.; D’Ambrosio, D.; Venditti, M.; Riggio, O.; Merli, M. A low muscle mass increases mortality in compensated cirrhotic patients with sepsis. Liver Int. 2018, 38, 851–857. [Google Scholar] [CrossRef]
- Van Vugt, J.L.A.; Alferink, L.J.M.; Buettner, S.; Gaspersz, M.P.; Bot, D.; Darwish Murad, S.; Feshtali, S.; van Ooijen, P.M.A.; Polak, W.J.; Porte, R.J.; et al. A model including sarcopenia surpasses the MELD score in predicting waiting list mortality in cirrhotic liver transplant candidates: A competing risk analysis in a national cohort. J. Hepatol. 2018, 68, 707–714. [Google Scholar] [CrossRef]
- Montano-Loza, A.J. Severe muscle depletion predicts postoperative length of stay but is not associated with survival after liver transplantation. Liver Transpl. 2014, 20, 1424. [Google Scholar] [CrossRef]
- Merli, M.; Giusto, M.; Gentili, F.; Novelli, G.; Ferretti, G.; Riggio, O.; Corradini, S.G.; Siciliano, M.; Farcomeni, A.; Attili, A.F.; et al. Nutritional status: Its influence on the outcome of patients undergoing liver transplantation. Liver Int. 2010, 30, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Plank, L.D.; Metzger, D.J.; McCall, J.L.; Barclay, K.L.; Gane, E.J.; Streat, S.J.; Munn, S.R.; Hill, G.L. Sequential changes in the metabolic response to orthotropic liver transplantation during the first year after surgery. Ann. Surg. 2001, 234, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Kaido, T.; Hamaguchi, Y.; Uemoto, S. Sarcopenia plays a crucial role in liver transplantation. Hepatobiliary Surg. Nutr. 2017, 6, 434–436. [Google Scholar] [CrossRef] [PubMed]
- Tsien, C.; Garber, A.; Narayanan, A.; Shah, S.N.; Barnes, D.; Eghtesad, B.; Fung, J.; McCullough, A.J.; Dasarathy, S. Post-liver transplantation sarcopenia in cirrhosis: A prospective evaluation. J. Gastroenterol. Hepatol. 2014, 29, 1250–1257. [Google Scholar] [CrossRef] [PubMed]
- Merli, M.; Giusto, M.; Riggio, O.; Gentili, F.; Molinaro, A.; Attili, A.F.; Corradini, S.G.; Rossi, M. Improvement of nutritional status in malnourished cirrhotic patients one year after liver transplantation. e-SPEN Eur. e-J. Clin. Nutr. Metab. 2011, 6, e142–e147. [Google Scholar] [CrossRef]
- Giusto, M.; Lattanzi, B.; Di Gregorio, V.; Giannelli, V.; Lucidi, C.; Merli, M. Changes in nutritional status after liver transplantation. World J. Gastroenterol. 2014, 20, 10682. [Google Scholar] [CrossRef]
- Langer, G.; Großmann, K.; Fleischer, S.; Berg, A.; Grothues, D.; Wienke, A.; Behrens, J.; Fink, A. Nutritional interventions for liver-transplanted patients. Cochrane Database Syst. Rev. 2012, 15, CD007605. [Google Scholar] [CrossRef] [PubMed]
- Reilly, J.; Mehta, R.; Teperman, L.; Cemaj, S.; Tzakis, A.; Yanaga, K.; Ritter, P.; Rezak, A.; Makowka, L. Nutritional support after liver transplantation: A randomized prospective study. Jpen. J. Parenter. Enter. Nutr. 1990, 14, 386. [Google Scholar] [CrossRef] [PubMed]
- Hasse, J.M.; Blue, L.S.; Liepa, G.U.; Goldstein, R.M.; Jennings, L.W.; Mor, E.; Husberg, B.S.; Levy, M.F.; Gonwa, T.A.; Klintmalm, G.B.; et al. Early enteral nutrition support in patients undergoing liver transplantation. Jpen. J. Parenter. Enter. Nutr. 1995, 19, 437–443. [Google Scholar] [CrossRef]
- Moore, F.A.; Feliciano, D.V.; Andrassy, R.J.; McArdle, A.H.; Booth, F.V.; Morgenstein-Wagner, T.B.; Kellum, J.M., Jr.; Welling, R.E.; Moore, E.E. Early enteral feeding, compared with parenteral, reduces postoperative septic complications. The results of a meta-analysis. Ann. Surg. 1992, 216, 172–183. [Google Scholar] [CrossRef]
- Wicks, C.; Somasundaram, S.; Bjarnason, I.; Menzies, I.S.; Routley, D.; Potter, D.; Potter, D.; Tan, C. Comparison of enteral feeding and total parenteral nutrition after liver transplantation. Lancet 1994, 344, 837–840. [Google Scholar] [CrossRef]
- Plank, L.D.; Mathur, S.; Gane, E.J.; Peng, S.L.; Gillanders, L.K.; McIlroy, K.; Chavez, C.P.; Calder, P.C.; McCall, J.L. Perioperative immunonutrition in patients undergoing liver transplantation: A randomized double-blind trial. Hepatology 2015, 61, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Holecek, M. Beta-hydroxy-beta-methylbutyrate supplementation and skeletal muscle in healthy and muscle-wasting conditions. J. Cachexia Sarcopenia Muscle 2017, 8, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Giusto, M.; Lattanzi, B.; Albanese, C.; Galtieri, A.; Farcomeni, A.; Giannelli, V.; Lucidi, C.; Di Martino, M.; Catalano, C.; Merli, M. Sarcopenia in liver cirrhosis: The role of computed tomography scan for the assessment of muscle mass compared with dual-energy X-ray absorptiometry and anthropometry. Eur. J. Gastroenterol. Hepatol. 2015, 27, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Coin, A.; Sarti, S.; Ruggiero, E.; Giannini, S.; Pedrazzoni, M.; Minisola, S.; Rossini, M.; Inelmen, M.; Manzato, E.; Sergi, G.; et al. Prevalence of sarcopenia based on different diagnostic criteria using DEXA and appendicular skeletal muscle mass reference values in an Italian population aged 20 to 80. J. Am. Med. Dir. Assoc. 2013, 14, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Merli, M.; Giusto, M.; Giannelli, V.; Lucidi, C.; Riggio, O. Nutritional status and Liver Transplantation. J. Clin. Exp. Hepatol. 2011, 1, 190–198. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Kiesswetter, E.; Drey, M.; Sieber, C.C. Nutrition, frailty, and sarcopenia. Aging Clin. Exp. Res. 2017, 29, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.J.; Wilson, J.M.; Manninen, A.H. Effects of beta-hydroxy-beta-methylbutyrate (HMB) on exercise performance and body composition across varying levels of age, sex, and training experience: A review. Nutr. Metab. 2008, 5, 1–15. [Google Scholar] [CrossRef]
- Barton-Davis, E.R.; Shoturma, D.I.; Musaro, A.; Rosenthal, N.; Sweeney, H.L. Viral mediated expression of IGF-I blocks the aging-related loss of skeletal muscle function. Proc. Natl. Acad. Sci. USA 1998, 95, 15603–15607. [Google Scholar] [CrossRef]
- Fiorotto, M.L.; Schwartz, R.J.; Delaughter, M.C. Persistent IGF-1 overexpression in skeletal muscle transiently enhances DNA accretion and growth. FASEB J. 2003, 17, 59–60. [Google Scholar] [CrossRef]
- Smith, H.J.; Mukerji, P.; Tisdale, M.J. Attenuation of proteasome-induced proteolysis in skeletal muscle by {beta}-hydroxy-{beta}-methylbutyrate in cancer-induced muscle loss. Cancer Res. 2005, 65, 277–283. [Google Scholar] [PubMed]
- Aversa, Z.; Alamdari, N.; Castillero, E.; Muscaritoli, M.; Rossi Fanelli, F.; Hasselgren, P.O. β-Hydroxy-β-methylbutyrate (HMB) prevents dexamethasone-induced myotube atrophy. Biochem. Biophys. Res. Commun. 2012, 423, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Bear, D.E.; Langan, A.; Dimidi, E.; Wandrag, L.; Harridge, S.D.R.; Hart, N.; Connolly, B.; Whelan, K. beta-hydroxy-beta-methylbutyrate and its impact on skeletal muscle mass and physical function in clinical practice: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2019, 109, 1119–1132. [Google Scholar] [CrossRef] [PubMed]
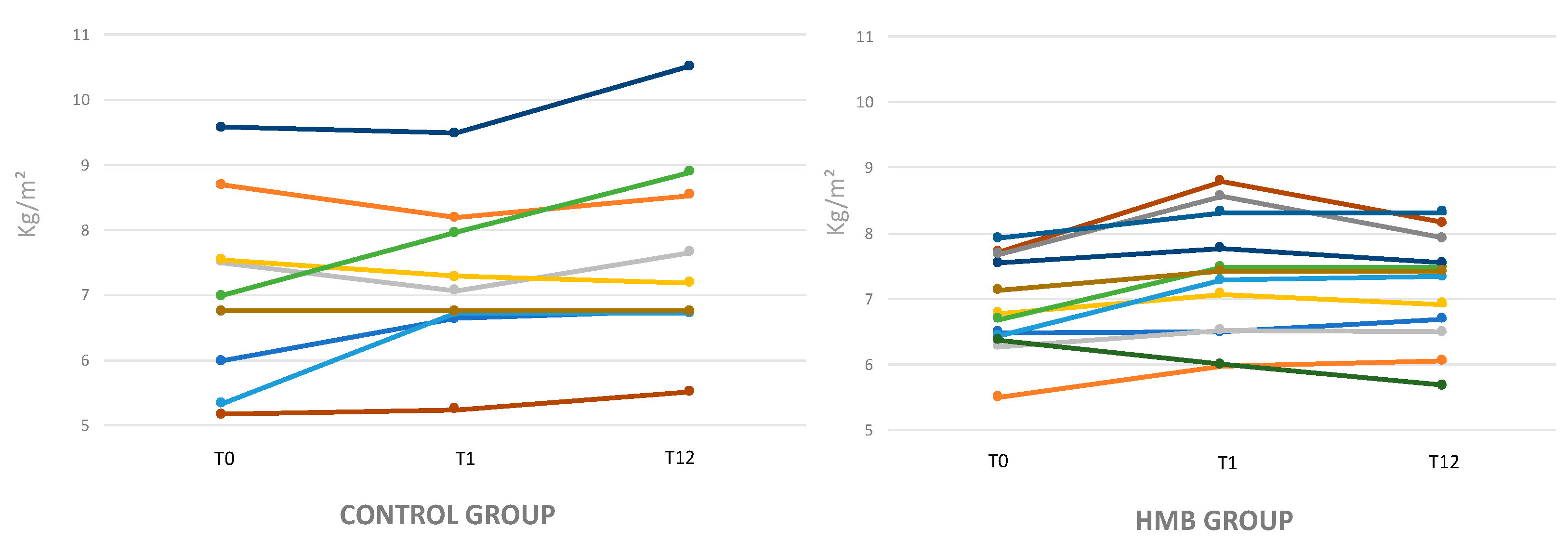
| Variables | Control Group (n = 10) | HMB Group (n = 12) | p-Value |
|---|---|---|---|
| Age (Years) | 59.3 ± 7.3 | 60.4 ± 5.4 | 0.68 |
| MELD score at transplantation | 16.8 ± 8.4 | 15.7 ± 7.1 | 0.70 |
| Hepato Cellular Carcinoma n (%) | 8 (80) | 8 (67) | 0.48 |
| Hospitalization after LT (days) | 10 ± 20.4 | 12 ± 18.25 | 0.57 |
| Aetiology n (%) | |||
| HCV | 4 (40) | 6 (50) | |
| Alcohol | 2 (20) | 3 (25) | |
| Autoimmune | 1 (10) | 0 | |
| Non Alcoholic Steato Hepatitis | 3 (30) | 2 (17) | 0.61 |
| HBV | 0 | 1 (8) | |
| BMI (kg/m2) | 25.7 ± 4.9 | 24.5 ± 3.0 | 0.48 |
| MAC (cm) | 29.8 ± 5.0 | 28.9 ± 2.8 | 0.61 |
| TSF (mm) | 8.75 ± 2.5 | 9.0 ± 2.9 | 0.81 |
| MAMC (cm) | 27.1 ± 4.5 | 26.1 ± 2.3 | 0.51 |
| FMI (kg/m²) | 6.9 ± 4.4 | 7.1 ± 2.1 | 0.9 |
| FFMI (kg/m²) | 17.5 ± 2.4 | 16.6 ± 1.5 | 0.29 |
| ASMI (kg/m²) | 7.2 ± 1.4 | 6.9 ± 0.7 | 0.53 |
| ASMI [<5° percentile] n (%) | 4 (40) | 7 (58) | 0.39 |
| FFMI [<5° percentile] n (%) | 4 (40) | 8 (67) | 0.21 |
| FMI [>95° percentile] n (%) | 3 (30) | 1 (8) | 0.19 |
| HG (kg) | 32 ± 10.2 | 26.7 ± 8.3 | 0.17 |
| 6MWT (m) | 316.5 ± 203.5 | 334.8 ± 129.2 | 0.80 |
| TUG (s) | 8.2 ± 4.9 | 9.7 ± 4.5 | 0.47 |
| Calorie Intake (kcal/kg/die) | 23.8 ± 6.3 | 22.7 ± 8.6 | 0.76 |
| Protein intake (g/kg/die) | 0.7 ± 0.2 | 0.9 ± 0.2 | 0.55 |
| Diabetes pre-LT n (%) | 5 (50) | 6 (50) | 0.99 |
| Arterial Hypertension pre-LT n (%) | 4 (40) | 7 (58) | 0.53 |
| Diabetes post-LT n (%) | 2 (20) | 5 (42) | 0.35 |
| Arterial Hypertension post-LT n (%) | 5 (50) | 7 (58) | 0.53 |
| Control Group (n = 9) | HMB Group (n = 12) | |||||
|---|---|---|---|---|---|---|
| Variable | T0 | T1 | T12 | T0 | T1 | T12 |
| FFMI (kg/m2) | 17.5 ± 2.4 | 17.6 ± 2.1 | 17.9 ± 2.7 | 16.6 ± 1.4 | 17.0 ± 1.6 | 16.8 ± 1.2 |
| ASMI (kg/m2) | 7.17 ± 1.4 | 7.4 ± 1.1 | 6.8 ± 2.87 | 6.8 ± 0.7 | 7.4 ± 0.8 * | 7.2 ± 0.7 * |
| MAMC (cm) | 27.3 ± 5.1 | 28.7 ± 5.1 | 28.7 ±4.6 | 26.1 ± 2.3 | 27.0 ± 2.6 ˠ | 27.7 ± 3.6 ˠ |
| FMI (kg/m2) | 7.3 ± 4.4 | 7.6 ± 5.0 | 8.7 ± 4.7 ˠ | 7.1 ± 2.1 | 7.7 ± 2.5 | 8.0 ± 3.2 |
| 6MWT (m) | 316.5 ± 203.5 | 312.8 ± 186.0 | 431.1 ± 125.3 | 334.8 ± 129.2 | 389.2 ± 143.8 | 413.8 ± 151.1 |
| HG (kg) | 33.2 ± 10.8 | 33.5 ± 10.3 | 33.7 ± 11.1 | 26.6 ± 8.3 | 32.7 ± 6.4 * | 33.7 ± 7.6 * |
| TUG (s) | 8.2 ± 4.9 | 10.7 ± 2.4 | 9.6 ± 2.1 | 9.7 ± 4.5 | 8.7 ± 2.6 | 9.1 ± 3.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lattanzi, B.; Giusto, M.; Albanese, C.; Mennini, G.; D’Ambrosio, D.; Farcomeni, A.; Ginanni Corradini, S.; Rossi, M.; Merli, M. The Effect of 12 Weeks of β-Hydroxy-β-Methyl-Butyrate Supplementation after Liver Transplantation: A Pilot Randomized Controlled Study. Nutrients 2019, 11, 2259. https://doi.org/10.3390/nu11092259
Lattanzi B, Giusto M, Albanese C, Mennini G, D’Ambrosio D, Farcomeni A, Ginanni Corradini S, Rossi M, Merli M. The Effect of 12 Weeks of β-Hydroxy-β-Methyl-Butyrate Supplementation after Liver Transplantation: A Pilot Randomized Controlled Study. Nutrients. 2019; 11(9):2259. https://doi.org/10.3390/nu11092259
Chicago/Turabian StyleLattanzi, Barbara, Michela Giusto, Carlina Albanese, Gianluca Mennini, Daria D’Ambrosio, Alessio Farcomeni, Stefano Ginanni Corradini, Massimo Rossi, and Manuela Merli. 2019. "The Effect of 12 Weeks of β-Hydroxy-β-Methyl-Butyrate Supplementation after Liver Transplantation: A Pilot Randomized Controlled Study" Nutrients 11, no. 9: 2259. https://doi.org/10.3390/nu11092259
APA StyleLattanzi, B., Giusto, M., Albanese, C., Mennini, G., D’Ambrosio, D., Farcomeni, A., Ginanni Corradini, S., Rossi, M., & Merli, M. (2019). The Effect of 12 Weeks of β-Hydroxy-β-Methyl-Butyrate Supplementation after Liver Transplantation: A Pilot Randomized Controlled Study. Nutrients, 11(9), 2259. https://doi.org/10.3390/nu11092259







