Infectious Complications in Home Parenteral Nutrition: A Systematic Review and Meta-Analysis Comparing Peripherally-Inserted Central Catheters with Other Central Catheters
Abstract
:1. Introduction
2. Methods
2.1. Data Sources and Searches
2.2. Study Selection
2.3. Data Extraction
2.4. Quality Assessment
2.5. Statistical Analysis
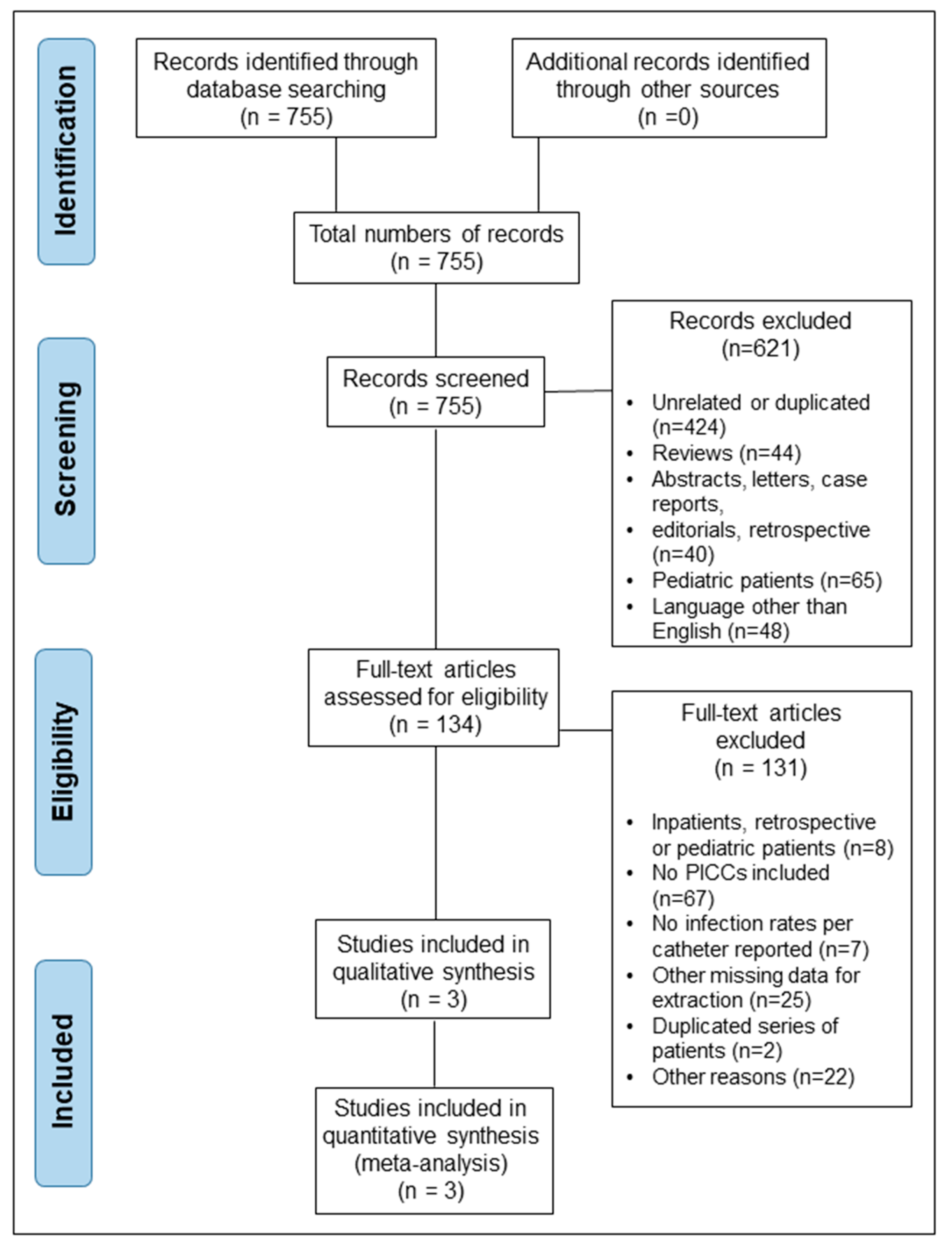
3. Results
3.1. Characteristics of Selected Studies and Patient Demographics
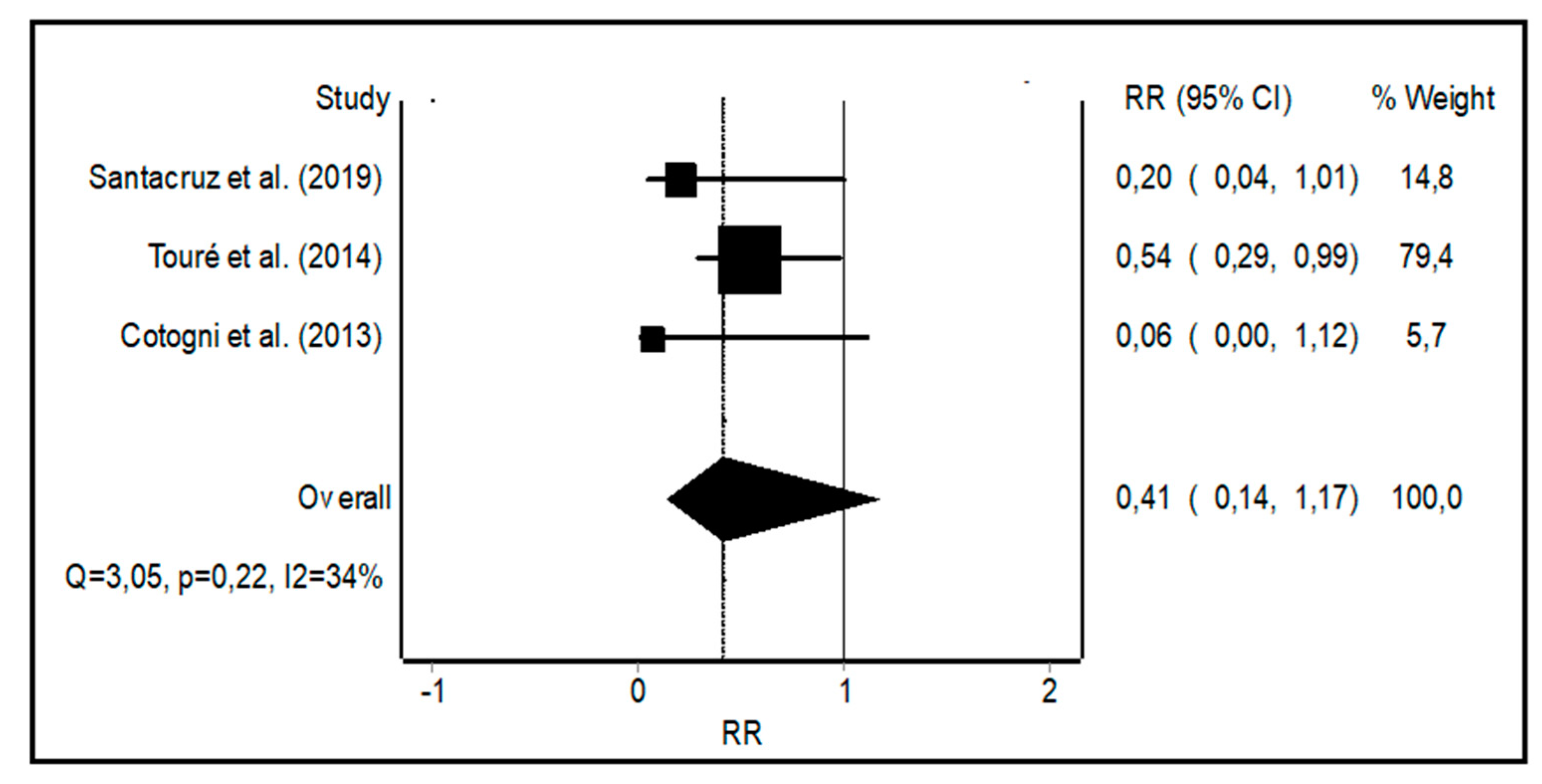
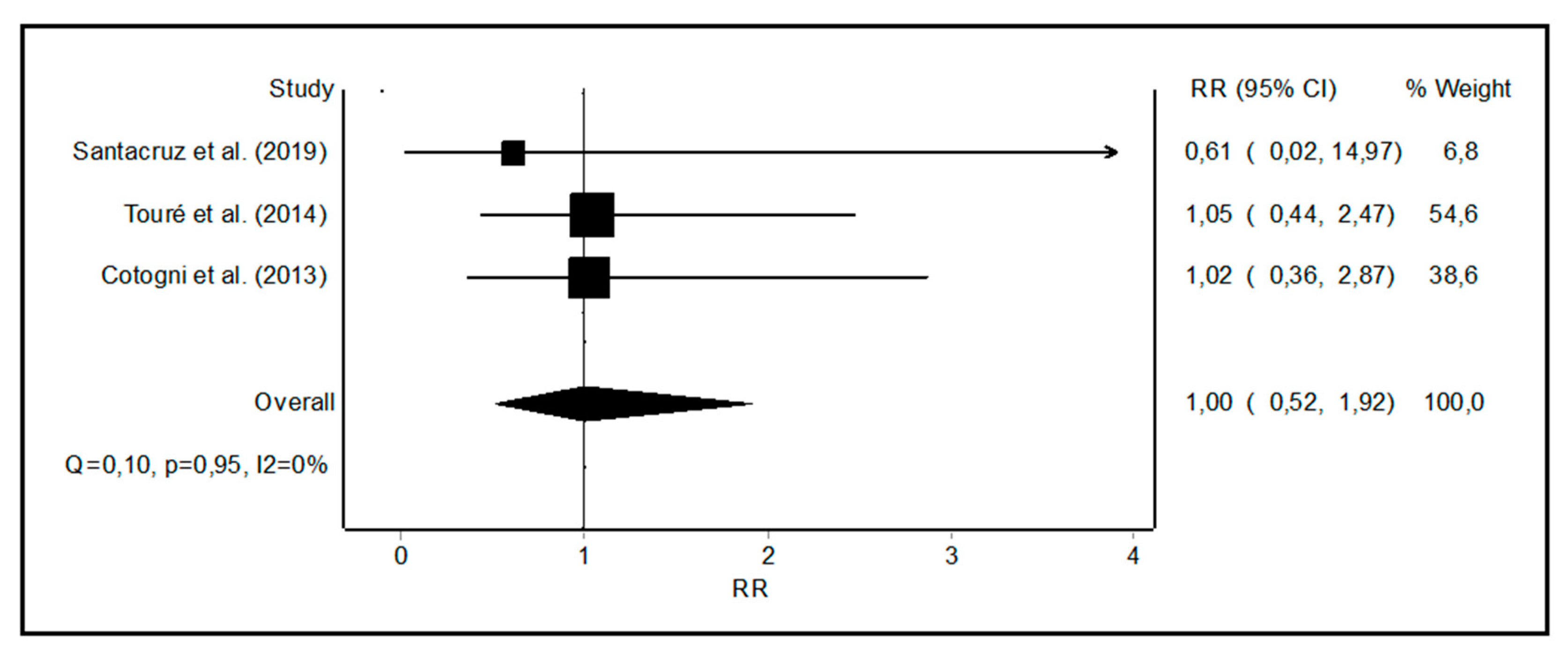
3.2. Meta-Analysis of CRBSI Rates with PICCs vs. Other Central Catheters
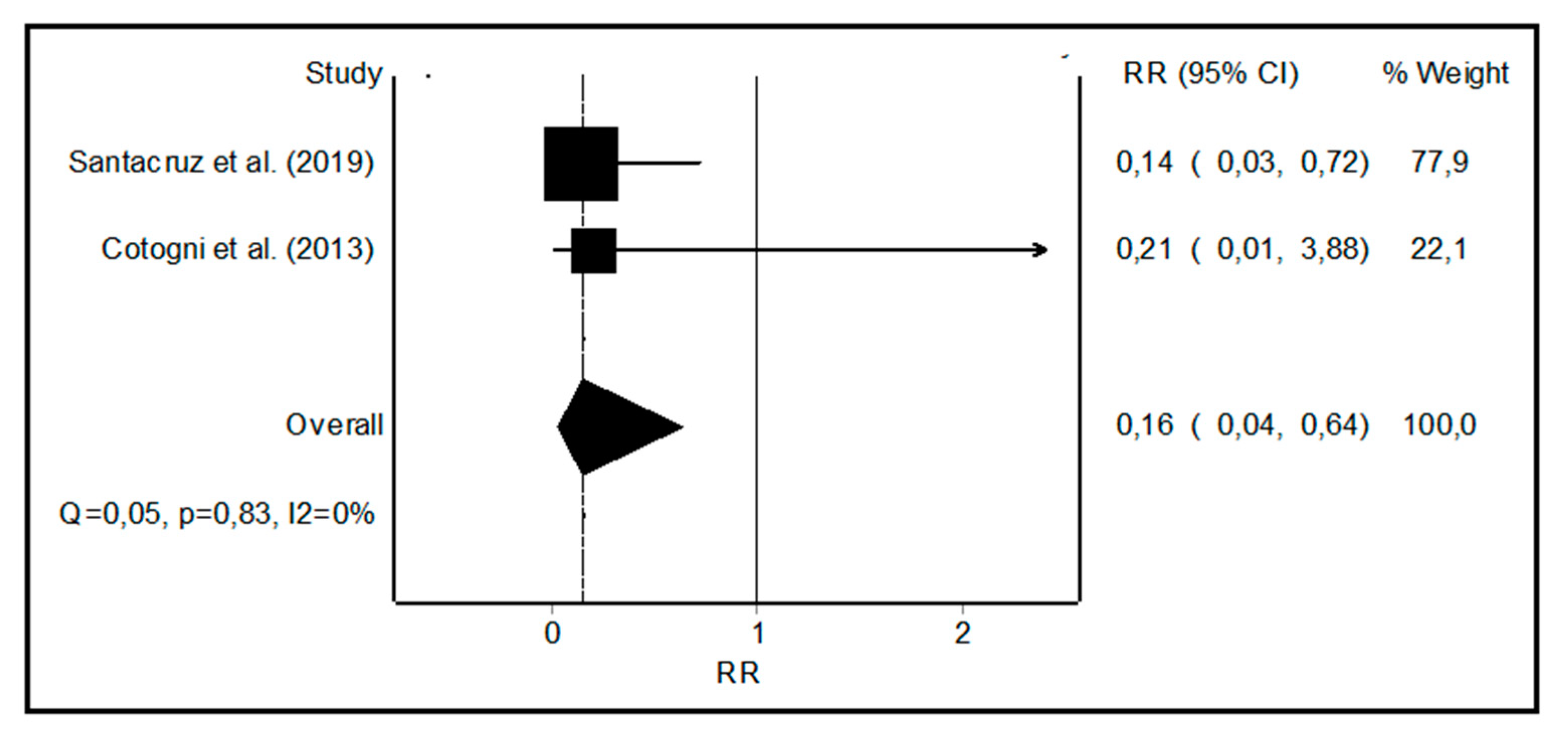
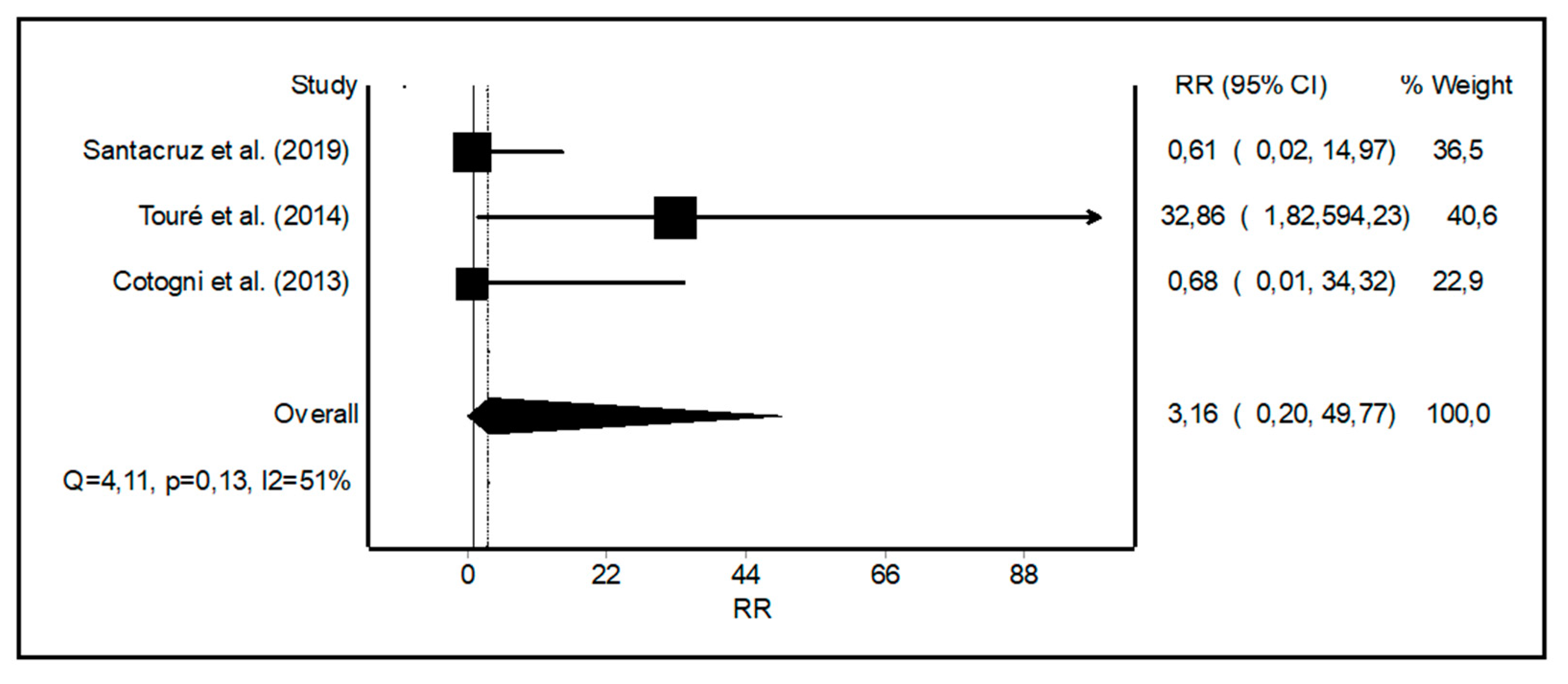
3.3. Meta-Analysis of Non-infectious Catheter Related Complications
4. Discussion
4.1. CRBSI Rates with PICCs vs. Other Central Catheters
4.2. Non-infectious Catheter Related Complications
4.3. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wanden-Berghe Lozano, C.; Cunill, J.L.P.; Compes, C.C.; Boluda, E.R.; Jimenez, M.I.M.; Candela, C.G.; Casas, N.V.; Pelaez, R.B.; de la Cruz, A.P.; Lazaro, M.F.P.; et al. [Home and Ambulatory Artificial Nutrition (Nadya) Report. Home Parenteral Nutrition in Spain, 2017]. Nutr. Hosp. 2018, 35, 1491–1496. [Google Scholar] [CrossRef] [PubMed]
- Hortencio, T.D.; Arendt, B.M.; Teterina, A.; Jeejeebhoy, K.N.; Gramlich, L.M.; Whittaker, J.S.; Armstrong, D.; Raman, M.; Nogueira, R.J.; Allard, J.P. Changes in Home Parenteral Nutrition Practice Based on the Canadian Home Parenteral Nutrition Patient Registry. JPEN J. Parenter. Enteral Nutr. 2017, 41, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Winkler, M.F.; DiMaria-Ghalili, R.A.; Guenter, P.; Resnick, H.E.; Robinson, L.; Lyman, B.; Ireton-Jones, C.; Banchik, L.H.; Steiger, E. Characteristics of a Cohort of Home Parenteral Nutrition Patients at the Time of Enrollment in the Sustain Registry. JPEN J. Parenter. Enteral Nutr. 2016, 40, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Farmer, D.G.; Venick, R.S.; Colangelo, J.; Esmailian, Y.; Yersiz, H.; Duffy, J.P.; Cortina, G.R.; Artavia, K.; Ngo, K.; McDiarmid, S.V.; et al. Pretransplant Predictors of Survival after Intestinal Transplantation: Analysis of a Single-Center Experience of More Than 100 Transplants. Transplantation 2010, 90, 1574–1580. [Google Scholar] [CrossRef] [PubMed]
- Wanden-Berghe, C.; Nolasco, A.; Planas, M.; Sanz-Valero, J.; Rodriguez, T.; Cuerda, C.; Guardiola, R.; Castello-Botia, I. Health-Related Quality of Life According to the Main Caregiver in Patients with Home Nutritional Support. Med. Clin. (Barc) 2008, 131, 281–284. [Google Scholar] [CrossRef]
- Ross, V.M.; Guenter, P.; Corrigan, M.L.; Kovacevich, D.; Winkler, M.F.; Resnick, H.E.; Norris, T.L.; Robinson, L.; Steiger, E. Central Venous Catheter Infections in Home Parenteral Nutrition Patients: Outcomes from Sustain: American Society for Parenteral and Enteral Nutrition’s National Patient Registry for Nutrition Care. Am. J. Infect. Control 2016, 44, 1462–1468. [Google Scholar] [CrossRef] [PubMed]
- Santacruz, E.; Mateo-Lobo, R.; Riveiro, J.; Nattero, L.; Vega-Pinero, B.; Lomba, G.; Sabido, R.; Carabana, F.; Arrieta, F.J.; Botella-Carretero, J.I. Infectious Complications in Home Parenteral Nutrition: A Long-Term Study with Peripherally Inserted Central Catheters, Tunneled Catheters, and Ports. Nutrition 2019, 58, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Pittiruti, M.; Hamilton, H.; Biffi, R.; MacFie, J.; Pertkiewicz, M. Espen Guidelines on Parenteral Nutrition: Central Venous Catheters (Access, Care, Diagnosis and Therapy of Complications). Clin. Nutr. 2009, 28, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Zhao, V.M.; Griffith, D.P.; Blumberg, H.M.; Dave, N.J.; Battey, C.H.; McNally, T.A.; Easley, K.A.; Galloway, J.R.; Ziegler, T.R. Characterization of Post-Hospital Infections in Adults Requiring Home Parenteral Nutrition. Nutrition 2013, 29, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Christensen, L.D.; Holst, M.; Bech, L.F.; Drustrup, L.; Nygaard, L.; Skallerup, A.; Rasmussen, H.H.; Vinter-Jensen, L. Comparison of Complications Associated with Peripherally Inserted Central Catheters and Hickman Catheters in Patients with Intestinal Failure Receiving Home Parenteral Nutrition. Six-Year Follow up Study. Clin. Nutr. 2016, 35, 912–917. [Google Scholar] [CrossRef] [PubMed]
- Hon, K.; Bihari, S.; Holt, A.; Bersten, A.; Kulkarni, H. Rate of Catheter-Related Bloodstream Infections between Tunneled Central Venous Catheters Versus Peripherally Inserted Central Catheters in Adult Home Parenteral Nutrition: A Meta-Analysis. JPEN J. Parenter. Enteral Nutr. 2019, 43, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-Analysis of Observational Studies in Epidemiology: A Proposal for Reporting. Meta-Analysis of Observational Studies in Epidemiology (Moose) Group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The Prisma Statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [PubMed]
- National Heart, Lung, and Blood Institute. Study Quality Assessment Tools. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 30 April 2019).
- Doi, S.A.; Barendregt, J.J.; Khan, S.; Thalib, L.; Williams, G.M. Advances in the Meta-Analysis of Heterogeneous Clinical Trials Ii: The Quality Effects Model. Contemp. Clin. Trials 2015, 45, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Egger, M. Funnel Plots for Detecting Bias in Meta-Analysis: Guidelines on Choice of Axis. J. Clin. Epidemiol. 2001, 54, 1046–1055. [Google Scholar] [CrossRef]
- Cotogni, P.; Pittiruti, M.; Barbero, C.; Monge, T.; Palmo, A.; Bertinet, D.B. Catheter-Related Complications in Cancer Patients on Home Parenteral Nutrition: A Prospective Study of over 51,000 Catheter Days. JPEN J. Parenter. Enteral Nutr. 2013, 37, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Toure, A.; Duchamp, A.; Peraldi, C.; Barnoud, D.; Lauverjat, M.; Gelas, P.; Chambrier, C. A Comparative Study of Peripherally-Inserted and Broviac Catheter Complications in Home Parenteral Nutrition Patients. Clin. Nutr. 2015, 34, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Elfassy, S.; Kassam, Z.; Amin, F.; Khan, K.J.; Haider, S.; Armstrong, D. Epidemiology and Risk Factors for Bloodstream Infections in a Home Parenteral Nutrition Program. JPEN J. Parenter. Enteral Nutr. 2015, 39, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Sommerfeld, E.; Snyder, G.; Rossi, T.M.; Lebenthal, E. Catheter-Related Complications in 35 Children and Adolescents with Gastrointestinal Disease on Home Parenteral Nutrition. JPEN J. Parenter. Enteral Nutr. 1990, 14, 148–151. [Google Scholar] [CrossRef]
- Vashi, P.G.; Virginkar, N.; Popiel, B.; Edwin, P.; Gupta, D. Incidence of and Factors Associated with Catheter-Related Bloodstream Infection in Patients with Advanced Solid Tumors on Home Parenteral Nutrition Managed Using a Standardized Catheter Care Protocol. BMC Infect. Dis. 2017, 17, 372. [Google Scholar] [CrossRef]
- DeLegge, M.H.; Borak, G.; Moore, N. Central Venous Access in the Home Parenteral Nutrition Population-You Picc. JPEN J. Parenter. Enteral Nutr. 2005, 29, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Edakkanambeth Varayil, J.; Whitaker, J.A.; Okano, A.; Carnell, J.J.; Davidson, J.B.; Enzler, M.J.; Kelly, D.G.; Mundi, M.S.; Hurt, R.T. Catheter Salvage after Catheter-Related Bloodstream Infection During Home Parenteral Nutrition. JPEN J. Parenter. Enteral Nutr. 2017, 41, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Durkin, M.J.; Dukes, J.L.; Reeds, D.N.; Mazuski, J.E.; Camins, B.C. A Descriptive Study of the Risk Factors Associated with Catheter-Related Bloodstream Infections in the Home Parenteral Nutrition Population. JPEN J. Parenter. Enteral Nutr. 2016, 40, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Botella-Carretero, J.I.; Carrero, C.; Guerra, E.; Valbuena, B.; Arrieta, F.; Calanas, A.; Zamarron, I.; Balsa, J.A.; Vazquez, C. Role of Peripherally Inserted Central Catheters in Home Parenteral Nutrition: A 5-Year Prospective Study. JPEN J. Parenter. Enteral Nutr. 2013, 37, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Bech, L.F.; Drustrup, L.; Nygaard, L.; Skallerup, A.; Christensen, L.D.; Vinter-Jensen, L.; Rasmussen, H.H.; Holst, M. Environmental Risk Factors for Developing Catheter-Related Bloodstream Infection in Home Parenteral Nutrition Patients: A 6-Year Follow-up Study. JPEN J. Parenter. Enteral Nutr. 2016, 40, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Bond, A.; Teubner, A.; Taylor, M.; Cawley, C.; Varden, J.; Abraham, A.; Chadwick, P.R.; Soop, M.; Carlson, G.L.; Lal, S. Catheter-Related Infections in Patients with Acute Type Ii Intestinal Failure Admitted to a National Centre: Incidence and Outcomes. Clin. Nutr. 2019, 38, 1828–1832. [Google Scholar] [CrossRef]
- Emery, D.; Pearson, A.; Lopez, R.; Hamilton, C.; Albert, N.M. Voiceover Interactive Powerpoint Catheter Care Education for Home Parenteral Nutrition. Nutr. Clin. Pract. 2015, 30, 714–719. [Google Scholar] [CrossRef]
- Gomez-Candela, C.; Fuentes, M.M.; Vazquez, N.G.; Yanguas, M.C.; Catalan, A.L.; del Portillo, R.C.; Milla, S.P. Twenty-Five Years of Home Parenteral Nutrition Outsourcing: The Experience at Hospital Universitario La Paz, Madrid. Nutr. Hosp. 2014, 30, 1295–1302. [Google Scholar]
- Higuera, I.; Garcia-Peris, P.; Camblor, M.; Breton, I.; Velasco, C.; Romero, R.; Frias, L.; Cuerda, C. Outcomes of a General Hospital-Based Home Parenteral Nutrition (Hpn) Program; Report of Our Experience from a 26-Year Period. Nutr. Hosp. 2014, 30, 359–365. [Google Scholar]
- Obling, S.R.; Wilson, B.V.; Kjeldsen, J. Home Parenteral Support in Patients with Incurable Cancer. Patient Characteristics of Importance for Catheter Related Complications and Overall Survival. Clin. Nutr. ESPEN 2018, 28, 88–95. [Google Scholar] [CrossRef]
- Santarpia, L.; Buonomo, A.; Pagano, M.C.; Alfonsi, L.; Foggia, M.; Mottola, M.; Marinosci, G.Z.; Contaldo, F.; Pasanisi, F. Central Venous Catheter Related Bloodstream Infections in Adult Patients on Home Parenteral Nutrition: Prevalence, Predictive Factors, Therapeutic Outcome. Clin. Nutr. 2016, 35, 1394–1398. [Google Scholar] [CrossRef] [PubMed]
- Maki, D.G.; Kluger, D.M.; Crnich, C.J. The Risk of Bloodstream Infection in Adults with Different Intravascular Devices: A Systematic Review of 200 Published Prospective Studies. Mayo Clin. Proc. 2006, 81, 1159–1171. [Google Scholar] [CrossRef] [PubMed]
- Pinelli, F.; Cecero, E.; Degl’Innocenti, D.; Selmi, V.; Giua, R.; Villa, G.; Chelazzi, C.; Romagnoli, S.; Pittiruti, M. Infection of Totally Implantable Venous Access Devices: A Review of the Literature. J. Vasc Access. 2018, 19, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Cuerda, C.; Joly, F.; Corcos, O.; Concejo, J.; Puiggros, C.; Gil, C.; Pironi, L.; Hah-Cif Espen Group. Prospective Study of Catheter-Related Central Vein Thrombosis in Home Parenteral Nutrition Patients with Benign Disease Using Serial Venous Doppler Ultrasound. Clin. Nutr. 2016, 35, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Dibb, M.; Lal, S. Home Parenteral Nutrition: Vascular Access and Related Complications. Nutr. Clin. Pract. 2017, 32, 769–776. [Google Scholar] [CrossRef] [PubMed]
| Author (Year) | Country | N | Mean Age (SD) | F/M | ON/Non-ON | Catheter Type | Catheter n |
|---|---|---|---|---|---|---|---|
| Santacruz et al. (2019) [7] | Spain | 151 | 58 (13) | 95/55 | 125/26 | PICC Tunneled Ports | 116 18 36 |
| Touré et al. (2014) [18] | France | 196 | 56 (17) | 119/77 | 32/164 | PICC Tunneled | 71 133 |
| Cotogni et al. (2013) [17] | Italy | 254 | 67 (29–85) * | 123/131 | 254/0 | PICC Tunneled Ports Non-tunneled | 65 45 72 107 |
| Author (Year) | Catheter Days | CRBSI Rate * | CRBSI Diagnosis | Catheter Care | Catheter Lumens | Quality Index |
|---|---|---|---|---|---|---|
| Santacruz et al. (2019) [7] | PICC: 20495 Tunneled: 4167 Ports: 2970 | 0.15 0.72 2.35 | Cultures ** | Aseptic technique | ML:23 SL:101 | 12 |
| Touré et al. (2014) [18] | PICC: 12322 Tunneled: 36812 | 1.05 1.87 | Cultures ** | NR | NR | 11 |
| Cotogni et al. (2013) [17] | PICC: 11504 Tunneled: 7835 Ports: 21605 Non-tunneled: 10364 | 0 0.64 0.19 0.87 | Cultures ** | Aseptic technique | All SL | 11 |
| Author (Year) | Catheter Days | Catheter Insertion | Catheter Flushing | Thrombosis Rate * | Mechanical Complications * |
|---|---|---|---|---|---|
| Santacruz et al. (2019) [7] | PICC: 20495 Tunneled: 4167 | US RG | Heparin | 0.049 0 | 0.049 0 |
| Touré et al. (2014) [18] | PICC: 12322 Tunneled: 36812 | RG RG | Taurolidine-citrate *** | 0.4 0 | 0.60 0.56 |
| Cotogni et al. (2013) [17] | PICC: 11504 Tunneled: 7835 | US US ** | Saline | 0 0 | 0.78 0.77 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mateo-Lobo, R.; Riveiro, J.; Vega-Piñero, B.; Botella-Carretero, J.I. Infectious Complications in Home Parenteral Nutrition: A Systematic Review and Meta-Analysis Comparing Peripherally-Inserted Central Catheters with Other Central Catheters. Nutrients 2019, 11, 2083. https://doi.org/10.3390/nu11092083
Mateo-Lobo R, Riveiro J, Vega-Piñero B, Botella-Carretero JI. Infectious Complications in Home Parenteral Nutrition: A Systematic Review and Meta-Analysis Comparing Peripherally-Inserted Central Catheters with Other Central Catheters. Nutrients. 2019; 11(9):2083. https://doi.org/10.3390/nu11092083
Chicago/Turabian StyleMateo-Lobo, Raquel, Javier Riveiro, Belén Vega-Piñero, and José I. Botella-Carretero. 2019. "Infectious Complications in Home Parenteral Nutrition: A Systematic Review and Meta-Analysis Comparing Peripherally-Inserted Central Catheters with Other Central Catheters" Nutrients 11, no. 9: 2083. https://doi.org/10.3390/nu11092083
APA StyleMateo-Lobo, R., Riveiro, J., Vega-Piñero, B., & Botella-Carretero, J. I. (2019). Infectious Complications in Home Parenteral Nutrition: A Systematic Review and Meta-Analysis Comparing Peripherally-Inserted Central Catheters with Other Central Catheters. Nutrients, 11(9), 2083. https://doi.org/10.3390/nu11092083






