A Review of Dietary (Phyto)Nutrients for Glutathione Support
Abstract
1. Introduction
2. The Role of Gene Deletions and Single Nucleotide Polymorphisms (SNPs)
3. Optimizing Glutathione Production with Nutrients
4. Preformed Glutathione
5. N-Acetylcysteine (NAC)
6. Dietary Protein Considerations
7. Omega-3 Fatty Acids
8. Vitamins
8.1. B Vitamins
8.2. Vitamin C
8.3. Vitamin E
9. Other Nutrients
9.1. Alpha-Lipoic Acid
9.2. Selenium
9.3. Phytonutrients
9.4. Brassica Vegetables
9.5. Green Tea
9.6. Juice Studies
9.7. Herbs and Roots
9.8. Plant Foods that Contain Glutathione
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pizzorno, J. Glutathione! Integr. Med. 2014, 13, 8–12. [Google Scholar]
- Franco, R.; Schoneveld, O.J.; Pappa, A.; Panayiotidis, M.I. The central role of glutathione in the pathophysiology of human diseases. Arch. Physiol. Biochem. 2007, 113, 234–258. [Google Scholar] [CrossRef] [PubMed]
- Ballatori, N.; Krance, S.M.; Notenboom, S.; Shi, S.; Tieu, K.; Hammond, C.L. Glutathione dysregulation and the etiology and progression of human diseases. Biol. Chem. 2009, 390, 191–214. [Google Scholar] [CrossRef] [PubMed]
- Pompella, A.; Emdin, M.; Franzini, M.; Paolicchi, A. Serum gamma-glutamyltransferase: linking together environmental pollution, redox equilibria and progression of atherosclerosis? Clin. Chem. Lab. Med. 2009, 47, 1583–1584. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Jacobs, D.R., Jr. Hormesis and public health: can glutathione depletion and mitochondrial dysfunction due to very low-dose chronic exposure to persistent organic pollutants be mitigated? J. Epidemiol. Commun. Health 2015, 69, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. The antioxidant paradox: less paradoxical now? Br. J. Clin. Pharmacol. 2013, 75, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Feoli, A.M.; Siqueira, I.; Almeida, L.M.; Tramontina, A.C.; Battu, C.; Wofchuk, S.T.; Gottfried, C.; Perry, M.L.; Gonçalves, C.A. Brain glutathione content and glutamate uptake are reduced in rats exposed to pre- and postnatal protein malnutrition. J. Nutr. 2006, 136, 2357–2361. [Google Scholar] [CrossRef] [PubMed]
- López-López, A.L.; Jaime, H.B.; Escobar Villanueva, M.D.C.; Padilla, M.B.; Palacios, G.V.; Aguilar, F.J.A. Chronic unpredictable mild stress generates oxidative stress and systemic inflammation in rats. Physiol. Behav. 2016, 161, 15–23. [Google Scholar] [CrossRef]
- Lee, D.H.; Jacobs, D.R., Jr. Serum gamma-glutamyltransferase: new insights about an old enzyme. J Epidemiol. Commun. Health 2009, 63, 884–886. [Google Scholar] [CrossRef]
- Hall, M.N.; Niedzwiecki, M.; Liu, X.; Harper, K.N.; Alam, S.; Slavkovich, V.; Gamble, M.V. Chronic arsenic exposure and blood glutathione and glutathione disulfide concentrations in Bangladeshi adults. Environ. Health Perspect. 2013, 121, 1068–1074. [Google Scholar] [CrossRef]
- Nebraska Redox Biology Center Educational Portal. Available online: http://genomics.unl.edu/RBC_EDU/gp.html (accessed on 7 July 2019).
- Allocati, N.; Masulli, M.; Di Ilio, C.; & Federici, L. Glutathione transferases: substrates, inihibitors and pro-drugs in cancer and neurodegenerative diseases. Oncogenesis 2018, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.C. Regulation of hepatic glutathione synthesis: current concepts and controversies. FASEB J. 1999, 13, 1169–1183. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Fang, Y.Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione metabolism and its implications for health. J. Nutr. 2004, 134, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Lang, C.A.; Mills, B.J.; Lang, H.L.; Liu, M.C.; Usui, W.M.; Richie, J., Jr.; Mastropaolo, W.; Murrell, S.A. High blood glutathione levels accompany excellent physical and mental health in women ages 60 to 103 years. J Lab. Clin. Med. 2002, 140, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Saharan, S.; Mandal, P.K. The emerging role of glutathione in Alzheimer’s disease. J. Alzheimers Dis. 2014, 40, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Traverso, N.; Ricciarelli, R.; Nitti, M.; Marengo, B.; Furfaro, A.L.; Pronzato, M.A.; Marinari, U.M.; Domenicotti, C. Role of glutathione in cancer progression and chemoresistance. Oxid. Med. Cell Longev. 2013, 2013, 972913. [Google Scholar] [CrossRef] [PubMed]
- Czuczejko, J.; Zachara, B.A.; Staubach-Topczewska, E.; Halota, W.; Kedziora, J. Selenium, glutathione and glutathione peroxidases in blood of patients with chronic liver diseases. Acta Biochim. Pol. 2003, 50, 1147–1154. [Google Scholar] [PubMed]
- Rae, C.D.; Williams, S.R. Glutathione in the human brain: Review of its roles and measurement by magnetic resonance spectroscopy. Anal. Biochem. 2017, 529, 127–143. [Google Scholar] [CrossRef]
- Kettle, A.J.; Turner, R.; Gangell, C.L.; Harwood, D.T.; Khalilova, I.S.; Chapman, A.L.; Winterbourn, C.C.; Sly, P.D.; Arest, C.F. Oxidation contributes to low glutathione in the airways of children with cystic fibrosis. Eur. Respir. J. 2014, 44, 122–129. [Google Scholar] [CrossRef]
- Achari, A.E.; Jain, S.K. l-Cysteine supplementation increases insulin sensitivity mediated by upregulation of GSH and adiponectin in high glucose treated 3T3-L1 adipocytes. Arch. Biochem. Biophys. 2017, 630, 54–65. [Google Scholar] [CrossRef]
- Sekhar, R.V.; McKay, S.V.; Patel, S.G.; Guthikonda, A.P.; Reddy, V.T.; Balasubramanyam, A.; Jahoor, F. Glutathione synthesis is diminished in patients with uncontrolled diabetes and restored by dietary supplementation with cysteine and glycine. Diabetes Care 2011, 34, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.; Hsu, J.W.; Jahoor, F.; Sekhar, R.V. Effect of increasing glutathione with cysteine and glycine supplementation on mitochondrial fuel oxidation, insulin sensitivity, and body composition in older HIV-infected patients. J. Clin. Endocrinol. Metab. 2014, 99, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Robaczewska, J.; Kedziora-Kornatowska, K.; Kozakiewicz, M.; Zary-Sikorska, E.; Pawluk, H.; Pawliszak, W.; Kedziora, J. Role of glutathione metabolism and glutathione-related antioxidant defense systems in hypertension. J. Physiol. Pharmacol. 2016, 67, 331–337. [Google Scholar] [PubMed]
- Adeoye, O.; Olawumi, J.; Opeyemi, A.; Christiania, O. Review on the role of glutathione on oxidative stress and infertility. JBRA Assist. Reprod. 2018, 22, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.; Sah, S.; Nath, S.K. Interaction between glutathione and apoptosis in systemic lupus erythematosus. Autoimmun. Rev. 2013, 12, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Nucifora, L.G.; Tanaka, T.; Hayes, L.N.; Kim, M.; Lee, B.J.; Matsuda, T.; Nucifora, F.C., Jr.; Sedlak, T.; Mojtabai, R.; Eaton, W.; et al. Reduction of plasma glutathione in psychosis associated with schizophrenia and bipolar disorder in translational psychiatry. Transl. Psychiatry 2017, 7, e1215. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.N.; Lim, J.L.; Nijland, P.G.; Witte, M.E.; Van Horssen, J. Glutathione in multiple sclerosis: More than just an antioxidant? Mult. Scler. 2014, 20, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, K.; Nakaki, T. Impaired glutathione synthesis in neurodegeneration. Int. J. Mol. Sci. 2013, 14, 21021–21044. [Google Scholar] [CrossRef]
- Coles, L.D.; Tuite, P.J.; Öz, G.; Mishra, U.R.; Kartha, R.V.; Sullivan, K.M.; Cloyd, J.C.; Terpstra, M. Repeated-dose oral N-Acetylcysteine in Parkinson’s disease: Pharmacokinetics and effect on brain glutathione and oxidative stress. J. Clin. Pharmacol. 2018, 58, 158–167. [Google Scholar] [CrossRef]
- Lux, O.; Naidoo, D. Biological variability of superoxide dismutase and glutathione peroxidase in blood. Redox Rep. 1995, 1, 331–335. [Google Scholar] [CrossRef]
- Van ‘t Erve, T.J.; Wagner, B.A.; Ryckman, K.K.; Raife, T.J.; Buettner, G.R. The concentration of glutathione in human erythrocytes is a heritable trait. Free Radic. Biol. Med. 2013, 65, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Bulusu, S.; Sharma, M. What does serum γ-glutamyltransferase tell us as a cardiometabolic risk marker? Ann. Clin. Biochem. 2016, 53, 312–332. [Google Scholar] [CrossRef] [PubMed]
- Nebert, D.W.; Vasiliou, V. Analysis of the glutathione S-transferase (GST) gene family. Hum. Genom. 2004, 1, 460–464. [Google Scholar] [CrossRef]
- Sprenger, R.; Schlagenhaufer, R.; Kerb, R.; Bruhn, C.; Brockmöller, J.; Roots, I.; Brinkmann, U. Characterization of the glutathione S-transferase GSTT1 deletion: discrimination of all genotypes by polymerase chain reaction indicates a trimodular genotype-phenotype correlation. Pharmacogenetics 2000, 10, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Hollman, A.L.; Tchounwou, P.B.; Huang, H.C. The association between gene-environment interactions and diseases involving the human GST superfamily with SNP variants. Int. J. Environ. Res. Public Health 2016, 13, 379. [Google Scholar] [CrossRef] [PubMed]
- Meister, A.; Anderson, M.E. Glutathione. Annu. Rev. Biochem. 1983, 52, 711–760. [Google Scholar] [CrossRef] [PubMed]
- Meister, A. Glutathione deficiency produced by inhibition of its synthesis, and its reversal; applications in research and therapy. Pharmacol. Ther. 1991, 51, 155–194. [Google Scholar] [CrossRef]
- Allen, J.; Bradley, R.D. Effects of oral glutathione supplementation on systemic oxidative stress biomarkers in human volunteers. J. Altern. Complement. Med. 2011, 17, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Witschi, A.; Reddy, S.; Stofer, B.; Lauterburg, B.H. The systemic availability of oral glutathione. Eur. J. Clin. Pharmacol. 1992, 43, 667–669. [Google Scholar] [CrossRef] [PubMed]
- Park, E.Y.; Shimura, N.; Konishi, T.; Sauchi, Y.; Wada, S.; Aoi, W.; Nakamura, Y.; Sato, K. Increase in the protein-bound form of glutathione in human blood after the oral administration of glutathione. J. Agric. Food Chem. 2014, 62, 6183–6189. [Google Scholar] [CrossRef] [PubMed]
- Richie, J.P., Jr.; Nichenametla, S.; Neidig, W.; Calcagnotto, A.; Haley, J.S.; Schell, T.D.; Muscat, J.E. Randomized controlled trial of oral glutathione supplementation on body stores of glutathione. Eur. J. Nutr. 2015, 54, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.; Sinha, I.; Calcagnotto, A.; Trushin, N.; Haley, J.S.; Schell, T.D.; Richie, J.P., Jr. Oral supplementation with liposomal glutathione elevates body stores of glutathione and markers of immune function. Eur. J. Clin. Nutr. 2018, 72, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Campolo, J.; Bernardi, S.; Cozzi, L.; Rocchiccioli, S.; Dellanoce, C.; Cecchettini, A.; Tonini, A.; Parolini, M.; De Chiara, B.; Micheloni, G.; et al. Medium-term effect of sublingual l-glutathione supplementation on flow-mediated dilation in subjects with cardiovascular risk factors. Nutrition 2017, 38, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Sechi, G.; Deledda, M.G.; Bua, G.; Satta, W.M.; Deiana, G.A.; Pes, G.M.; Rosati, G. Reduced intravenous glutathione in the treatment of early Parkinson’s disease. Prog. Neuropsychopharmacol. Biol Psychiatry 1996, 20, 1159–1170. [Google Scholar] [CrossRef]
- Otto, M.; Magerus, T.; Langland, J.O. The use of intravenous glutathione for symptom management of Parkinson’s disease: A case report. Altern. Ther. Health Med. 2018, 24, 56–60. [Google Scholar]
- Anderson, M.E.; Meister, A. Glutathione monoesters. Anal. Biochem. 1989, 183, 16–20. [Google Scholar] [CrossRef]
- McCarty, M.F.; O’Keefe, J.H.; DiNicolantonio, J.J. Dietary glycine is rate-limiting for glutathione synthesis and may have broad potential for health protection. Ochsner J. 2018, 18, 81–87. [Google Scholar]
- U.S. National Library of Medicine. Amino Acids. Available online: https://medlineplus.gov/ency/article/002222.htm (accessed on June 26 2019).
- Lu, S.C. Glutathione synthesis. Biochim. Biophys. Acta 2013, 1830, 3143–3153. [Google Scholar] [CrossRef]
- Parcell, S. Sulfur in human nutrition and applications in medicine. Altern. Med. Rev. 2002, 7, 22–44. [Google Scholar]
- Jones, D.P.; Park, Y.; Gletsu-Miller, N.; Liang, Y.; Yu, T.; Accardi, C.J.; Ziegler, T.R. Dietary sulfur amino acid effects on fasting plasma cysteine/cystine redox potential in humans. Nutrition 2011, 27, 199–205. [Google Scholar] [CrossRef]
- Skvarc, D.R.; Dean, O.M.; Byrne, L.K.; Gray, L.; Lane, S.; Lewis, M.; Fernanders, B.S.; Berk, M.; Marriott, A. The effect of N-acetylcysteine (NAC) on human cognition—A systematic review. Neurosci. Biobehav. Rev. 2017, 78, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Holmay, M.J.; Terpstra, M.; Coles, L.D.; Mishra, U.; Ahlskog, M.; Oz, G.; Cloyd, J.C.; Tuite, P.J. N-Acetylcysteine boosts brain and blood glutathione in Gaucher and Parkinson diseases. Clin. Neuropharmacol. 2013, 36, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Wink, L.K.; Adams, R.; Wang, Z.; Klaunig, J.E.; Plawecki, M.H.; Posey, D.J.; McDpugle, C.J.; Erickson, C.A. A randomized placebo-controlled pilot study of N-acetylcysteine in youth with autism spectrum disorder. Mol. Autism. 2016, 7, 26. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Wu, J.L.; Shih, T.S.; Tsai, P.J.; Sun, Y.M.; Ma, M.C.; Guo, Y.L. N-Acetyl-cysteine against noise-induced temporary threshold shift in male workers. Hear Res. 2010, 269, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Jannatifar, R.; Parivar, K.; Roodbari, N.H.; Nasr-Esfahani, M.H. Effects of N-acetyl-cysteine supplementation on sperm quality, chromatin integrity and level of oxidative stress in infertile men. Reprod Biol. Endocrinol. 2019, 17, 24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Ju, Y.; Ma, Y.; Wang, T. N-acetylcysteine improves oxidative stress and inflammatory response in patients with community acquired pneumonia: A randomized controlled trial. Medicine 2018, 97, e13087. [Google Scholar] [CrossRef] [PubMed]
- Rushworth, G.F.; Megson, I.L. Existing and potential therapeutic uses for N-acetylcysteine: the need for conversion to intracellular glutathione for antioxidant benefits. Pharmacol. Ther. 2014, 141, 150–159. [Google Scholar] [CrossRef]
- Ellegaard, P.K.; Licht, R.W.; Nielsen, R.E.; Dean, O.M.; Berk, M.; Poulsen, H.E.; Mohebbi, M.; Nielsen, C.T. The efficacy of adjunctive N-acetylcysteine in acute bipolar depression: A randomized placebo-controlled study. J. Affect. Disord. 2019, 245, 1043–1051. [Google Scholar] [CrossRef]
- Berk, M.; Turner, A.; Malhi, G.S.; Ng, C.H.; Cotton, S.M.; Dodd, S.; Samuni, Y.; Tanious, M.; McAulay, C.; Dowling, N.; et al. A randomised controlled trial of a mitochondrial therapeutic target for bipolar depression: mitochondrial agents, N-acetylcysteine, and placebo. BMC Med. 2019, 17, 18. [Google Scholar] [CrossRef]
- Panizzutti, B.; Bortolasci, C.; Hasebe, K.; Kidnapillai, S.; Gray, L.; Walder, K.; Berk, M.; Mohebbi, M.; Dodd, S.; Gama, C.; et al. Mediator effects of parameters of inflammation and neurogenesis from a N-acetyl cysteine clinical-trial for bipolar depression. Acta Neuropsychiatr. 2018, 30, 334–341. [Google Scholar] [CrossRef]
- Sekhar, R.V.; Patel, S.G.; Guthikonda, A.P.; Reid, M.; Balasubramanyam, A.; Taffet, G.E.; Jahoor, F. Deficient synthesis of glutathione underlies oxidative stress in aging and can be corrected by dietary cysteine and glycine supplementation. Am. J. Clin. Nutr. 2011, 94, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Bhutto, A.; Morley, J.E. The clinical significance of gastrointestinal changes with aging. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Corleto, V.D.; Festa, S.; Di Giulio, E.; Annibale, B. Proton pump inhibitor therapy and potential long-term harm. Curr. Opin. Endocrinol. Diabetes Obes. 2014, 21, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Dvorshchenko, K.O.; Bernyk, O.O.; Dranytsyna, A.S.; Senin, S.A.; Ostapchenko, L.I. Influence of oxidative stress on the level of genes expression Tgfb1 and Hgf in rat liver upon long-term gastric hypochlorhydria and administration of multiprobiotic Symbiter; Article in Ukrainian. Ukrains’ kyi Biokhimichnyi Zhurnal (1999) 2013, 85, 114–123. [Google Scholar]
- Naito, Y.; Yoshikawa, T. Molecular and cellular mechanisms involved in Helicobacter pylori-induced inflammation and oxidative stress. Free Radic. Biol. Med. 2002, 33, 323–336. [Google Scholar] [CrossRef]
- Cavalcoli, F.; Zilli, A.; Conte, D.; Massironi, S. Micronutrient deficiencies in patients with chronic atrophic autoimmune gastritis: A review. World J. Gastroenterol. 2017, 23, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.A.; Gibson, N.R.; Lu, Y.; Jahoor, F. Synthesis of erythrocyte glutathione in healthy adults consuming the safe amount of dietary protein. Am. J. Clin. Nutr. 2014, 80, 101–107. [Google Scholar] [CrossRef]
- Tosukhowong, P.; Boonla, C.; Dissayabutra, T.; Kaewwilai, L.; Muensri, S.; Chotipanich, C.; Joutsa, J.; Rinne, J.; Bhidayasiri, R. Biochemical and clinical effects of Whey protein supplementation in Parkinson’s disease: A pilot study. J. Neurol. Sci. 2016, 367, 162–170. [Google Scholar] [CrossRef]
- Zavorsky, G.S.; Kubow, S.; Grey, V.; Riverin, V.; Lands, L.C. An open-label dose-response study of lymphocyte glutathione levels in healthy men and women receiving pressurized whey protein isolate supplements. Int. J. Food Sci. Nutr. 2007, 58, 429–436. [Google Scholar] [CrossRef]
- Bumrungpert, A.; Pavadhgul, P.; Nunthanawanich, P.; Sirikanchanarod, A.; Adulbhan, A. Whey protein supplementation improves nutritional status, glutathione levels, and immune function in cancer patients: A randomized, double-blind controlled trial. J Med. Food 2018, 21, 612–616. [Google Scholar] [CrossRef]
- Zhou, X.; He, L.; Zuo, S.; Zhang, Y.; Wan, D.; Long, C.; Huang, P.; Wu, X.; Wu, C.; Liu, G.; et al. Serine prevented high-fat diet-induced oxidative stress by activating AMPK and epigenetically modulating the expression of glutathione synthesis-related genes. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 488–498. [Google Scholar] [CrossRef]
- Zhou, X.; He, L.; Wu, C.; Zhang, Y.; Wu, X.; Yin, Y. Serine alleviates oxidative stress via supporting glutathione synthesis and methionine cycle in mice. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Rahman, I. Inflammation and the regulation of glutathione level in lung epithelial cells. Antioxid. Redox. Signal 1999, 1, 425–447. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.L.; Lagopoulos, J.; Cockayne, N.; Lewis, S.J.; Hickie, I.B.; Hermens, D.F.; Naismith, S.L. The effect of 12-wk ω-3 fatty acid supplementation on in vivo thalamus glutathione concentration in patients “at risk” for major depression. Nutrition 2015, 31, 1247–1254. [Google Scholar] [CrossRef] [PubMed]
- Taghizadeh, M.; Tamtaji, O.R.; Dadgostar, E.; Daneshvar Kakhaki, R.; Bahmani, F.; Abolhassani, J.; Aarabi, M.H.; Kouchaki, E.; Memarzadeh, M.R.; Asemi, Z. The effects of omega-3 fatty acids and vitamin E co-supplementation on clinical and metabolic status in patients with Parkinson’s disease: A randomized, double-blind, placebo-controlled trial. Neurochem. Int. 2017, 108, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Gago-Dominguez, M.; Castelao, J.E.; Sun, C.-L.; Van Den Berg, D.; Koh, W.-P.; Lee, H.-P.; Yu, M.C. Marine n-3 fatty acid intake, glutathione S-transferase polymorphisms and breast cancer risk in post-menopausal Chinese women in Singapore. Carcinogenesis 2014, 25, 2143–2147. [Google Scholar] [CrossRef]
- Sepidarkish, M.; Akbari-Fakhrabadi, M.; Daneshzad, E.; Yavari, M.; Rezaeinejad, M.; Morvaridzadeh, M.; Heshmati, J. Effect of omega-3 fatty acid plus vitamin E Co-Supplementation on oxidative stress parameters: A systematic review and meta-analysis. Clin. Nutr. 2019. pii:S0261-561430215-8. [Google Scholar] [CrossRef] [PubMed]
- García-Rodríguez, C.E.; Mesa, M.D.; Olza, J.; Vlachava, M.; Kremmyda, L.S.; Diaper, N.D.; Noakes, P.S.; Miles, E.A.; Ramírez-Tortosa, M.C.; Liaset, B.; et al. Does consumption of two portions of salmon per week enhance the antioxidant defense system in pregnant women? Antioxid. Redox. Signal. 2012, 16, 1401–1406. [Google Scholar] [CrossRef]
- Stonehouse, W.; Pauga, M.R.; Kruger, R.; Thomson, C.D.; Wong, M.; Kruger, M.C. Consumption of salmon v. salmon oil capsules: effects on n-3 PUFA and selenium status. Br. J. Nutr. 2011, 106, 1231–1239. [Google Scholar] [CrossRef] [PubMed]
- Ashoori, M.; Saedisomeolia, A. Riboflavin (vitamin B₂) and oxidative stress: a review. Br. J. Nutr. 2014, 111, 1985–1991. [Google Scholar] [CrossRef]
- Majchrzak, D.; Singer, I.; Männer, M.; Rust, P.; Genser, D.; Wagner, K.H.; Elmadfa, I. B-vitamin status and concentrations of homocysteine in Austrian omnivores, vegetarians and vegans. Ann. Nutr. Metab. 2006, 50, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Moat, S.J.; Ashfield-Watt, P.A.; Powers, H.J.; Newcombe, R.G.; McDowell, I.F. Effect of riboflavin status on the homocysteine-lowering effect of folate in relation to the MTHFR (C677T) genotype. Clin Chem. 2003, 49, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Slyshenkov, V.S.; Dymkowska, D.; Wojtczak, L. Pantothenic acid and pantothenol increase biosynthesis of glutathione by boosting cell energetics. FEBS Lett. 2004, 569, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Misra, U.K.; Kalita, J.; Singh, S.K.; Rahi, S.K. Oxidative stress markers in Vitamin B12 deficiency. Mol. Neurobiol. 2017, 54, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Lenton, K.J.; Sané, A.T.; Therriault, H.; Cantin, A.M.; Payette, H.; Wagner, J.R. Vitamin C augments lymphocyte glutathione in subjects with ascorbate deficiency. Am. J. Clin. Nutr. 2003, 77, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Meyer, C.G.; Srilakshmi, J.C. Vitamin C elevates red blood cell glutathione in healthy adults. Am. J. Clin. Nutr. 1993, 58, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Chugh, S.N.; Kakkar, R.; Kalra, S.; Sharma, A. An evaluation of oxidative stress in diabetes mellitus during uncontrolled and controlled state and after vitamin E supplementation. J. Assoc. Physic. Ind. 1999, 47, 380–383. [Google Scholar]
- Sharma, A.; Kharb, S.; Chugh, S.N.; Kakkar, R.; Singh, G.P. Evaluation of oxidative stress before and after control of glycemia and after vitamin E supplementation in diabetic patients. Metabolism 2000, 49, 160–162. [Google Scholar] [CrossRef]
- Jain, S.K.; McVie, R.; Smith, T. Vitamin E supplementation restores glutathione and malondialdehyde to normal concentrations in erythrocytes of type 1 diabetic children. Diabetes Care. 2000, 23, 1389–1394. [Google Scholar] [CrossRef]
- Gupta, S.; Sharma, T.K.; Kaushik, G.G.; Shekhawat, V.P. Vitamin E supplementation may ameliorate oxidative stress in type 1 diabetes mellitus patients. Clin Lab. 2011, 57, 379–386. [Google Scholar]
- Aghadavod, E.; Soleimani, A.; Hamidi, G.; Keneshlou, F.; Heidari, A.; Asemi, Z. Effects of high-dose vitamin E supplementation on markers of cardiometabolic risk and oxidative stress in patients with diabetic nephropathy: A randomized double-blinded controlled trial. Iran J. Kidney Dis. 2018, 12, 156–162. [Google Scholar]
- Kolesnichenko, L.S.; Kulinskiĭ, V.I.; Shprakh, V.V.; Bardymov, V.V.; Verlan, N.V.; Gubina, L.P.; Pensionerova, G.A.; Sergeeva, M.P.; Stanevich, L.M.; Filippova, G.T. The blood glutathione system in cerebral vascular diseases and its treatment with alpha-lipoic acid. Zhurnal nevrologii i psikhiatrii imeni SS Korsakova 2008, 108, 36–40. [Google Scholar]
- Martins, V.D.; Manfredini, V.; Peralba, M.C.; Benfato, M.S. Alpha-lipoic acid modifies oxidative stress parameters in sickle cell trait subjects and sickle cell patients. Clin Nutr. 2009, 28, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Ansar, H.; Mazloom, Z.; Kazemi, F.; Hejazi, N. Effect of alpha-lipoic acid on blood glucose, insulin resistance and glutathione peroxidase of type 2 diabetic patients. Saudi Med. J. 2011, 32, 584–588. [Google Scholar] [PubMed]
- Georgakouli, K.; Deli, C.K.; Zalavras, A.; Fatouros, I.G.; Kouretas, D.; Koutedakis, Y.; Jamurtas, A.Z. A-lipoic acid supplementation up-regulates antioxidant capacity in adults with G6PD deficiency. Food Chem. Toxicol. 2013, 61, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Khalili, M.; Eghtesadi, S.; Mirshafiey, A.; Eskandari, G.; Sanoobar, M.; Sahraian, M.A.; Motevalian, A.; Norouzi, A.; Moftakhar, S.; Azimi, A. Effect of lipoic acid consumption on oxidative stress among multiple sclerosis patients: a randomized controlled clinical trial. Nutr. Neurosci. 2014, 17, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Hu, F.X. α-Lipoic acid treatment of aged type 2 diabetes mellitus complicated with acute cerebral infarction. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3715–3719. [Google Scholar]
- Becker, K.; Pons-Kühnemann, J.; Fechner, A.; Funk, M.; Gromer, S.; Gross, H.J.; Grünert, A.; Schirmer, R.H. Effects of antioxidants on glutathione levels and clinical recovery from the malnutrition syndrome kwashiorkor--a pilot study. Redox Rep. 2005, 10, 215–226. [Google Scholar] [CrossRef]
- Jariwalla, R.; Lalezari, J.; Cenko, D.; Mansour, S.E.; Kumar, A.; Gangapurkar, B.; Nakamura, D. Restoration of blood total glutathione status and lymphocyte function following alpha-lipoic acid supplementation in patients with HIV infection. J. Altern. Complement. Med. 2008, 14, 139–146. [Google Scholar] [CrossRef]
- Song, E.; Su, C.; Fu, J.; Xia, X.; Yang, S.; Xiao, C.; Lu, B.; Chen, H.; Sun, Z.; Wu, S.; et al. Selenium supplementation shows protective effects against patulin-induced brain damage in mice via increases in GSH-related enzyme activity and expression. Life Sci. 2014, 109, 37–43. [Google Scholar] [CrossRef]
- Richie, J.P., Jr.; Muscat, J.E.; Ellison, I.; Calcagnotto, A.; Kleinman, W.; El-Bayoumy, K. Association of selenium status and blood glutathione concentrations in blacks and whites. Nutr. Cancer 2011, 63, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Galan-Chilet, I.; Tellez-Plaza, M.; Guallar, E.; De Marco, G.; Lopez-Izquierdo, R.; Gonzalez-Manzano, I.; Carmen Tormos, M.; Martin-Nuñez, G.M.; Rojo-Martinez, G.; Saez, G.T.; et al. Plasma selenium levels and oxidative stress biomarkers: a gene-environment interaction population-based study. Free Radic. Biol. Med. 2014, 74, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Bohn, T. Exogenous antioxidants—Double-edged swords in cellular redox state: Health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxid. Med. Cell Longev. 2010, 3, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Dietary bioactive compounds and their health implications. J. Food Sci. 2013, 78 (Suppl. S1), A18–A25. [Google Scholar] [CrossRef] [PubMed]
- Fowke, J.H.; Morrow, J.D.; Motley, S.; Bostick, R.M.; Ness, R.M. Brassica vegetable consumption reduces urinary F2-isoprostane levels independent of micronutrient intake. Carcinogenesis 2006, 27, 2096–2102. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Riso, P.; Grande, S.; Galli, C.; Porrini, M. Protective activity of tomato products on in vivo markers of lipid oxidation. Eur J. Nutr. 2003, 42, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.J.; Heimendinger, J.; Sedlacek, S.; Haegele, A.; Diker, A.; O’Neill, C.; Meinecke, B.; Wolfe, P.; Zhu, Z.; Jiang, W. 8-Isoprostane F2alpha excretion is reduced in women by increased vegetable and fruit intake. Am. J. Clin. Nutr. 2005, 82, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.J.; Heimendinger, J.; Haegele, A.; Sedlacek, S.M.; Gillette, C.; O’Neill, C.; Wolfe, P.; Conry, C. Effect of increased vegetable and fruit consumption on markers of oxidative cellular damage. Carcinogenesis 1999, 20, 2261–2266. [Google Scholar] [CrossRef]
- Rink, S.M.; Mendola, P.; Mumford, S.L.; Poudrier, J.K.; Browne, J.R.; Eactawski-Wende, J.; Perkins, N.J.; Schisterman, E.F. Self-report of fruit and vegetable intake that meets the 5 a day recommendation is associated with reduced levels of oxidative stress biomarkers and increased levels of antioxidant defense in premenopausal women. J. Acad. Nutr. Diet. 2013, 113, 776–785. [Google Scholar] [CrossRef]
- Minich, D.M. A Review of the Science of Colorful, Plant-Based Food and Practical Strategies for Eating the Rainbow. J. Nutr. Metab. 2019, 2019, 19. [Google Scholar] [CrossRef]
- Wark, P.A.; Grubben, M.J.A.L.; Peters, W.H.M.; Nagengast, F.M.; Kampman, E.; Kok, F.J.; van ‘t Veer, P. Habitual consumption of fruits and vegetables: associations with human rectal glutathione S -transferase. Carcinogenesis 2014, 25, 2135–2142. [Google Scholar] [CrossRef] [PubMed]
- Talalay, P.; Fahey, J.W.; Holtzclaw, W.D.; Prestera, T.; Zhang, Y. Chemoprotection against cancer by phase 2 enzyme induction. Toxicol. Lett. 1995, 82–83, 173–179. [Google Scholar] [CrossRef]
- Clapper, M.L.; Szarka, C.E.; Pfeiffer, G.R.; Graham, T.A.; Balshem, A.M.; Litwin, S.; Goosenberg, E.B.; Frucht, H.; Engstrom, P.F. Preclinical and clinical evaluation of broccoli supplements as inducers of glutathione S-transferase activity. Clin. Cancer Res. 1997, 3, 25–30. [Google Scholar] [PubMed]
- Nijhoff, W.A.; Grubben, M.J.A.L.; Nagengast, F.M.; Jansen, J.B.M.J.; Verhagen, H.; Van Poppel, G.; Peters, W.H.M. Effects of consumption of Brussels sprouts on intestinal and lymphocytic glutathione S-transferases in humans. Carcinogenesis 1995, 16, 2125–2128. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lampe, J.W.; Chen, C.; Li, S.; Prunty, J.; Grate, M.T.; Meehan, D.E.; Barale, K.V.; Dightman, D.A.; Feng, Z.; Potter, J.D. Modulation of human glutathione S-transferases by botanically defined vegetable diets. Cancer Epidemiol. Biomarkers 2000, 9, 787–793. [Google Scholar]
- Bogaards, J.J.P.; Verhagen, H.; Willems, M.I.; Van Poppel, G.; Van Bladeren, P.J. Consumption of Brussels sprouts results in elevated alpha-class glutathione S-transferase levels in human blood plasma. Carcinogenesis 1994, 15, 1073–1075. [Google Scholar] [CrossRef] [PubMed]
- Nijhoff, W.A.; Mulder, T.P.J.; Verhagen, H.; Van Poppel, G.; Peters, W.H.M. Effects of consumption of brussels sprouts on plasma and urinary glutathione S-transferase class-alpha and -pi in humans. Carcinogenesis 1995, 16, 955–957. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, S.; Miyake, M. Biochemistry and biological functions of citrus limonoids. Food Rev. Int. 1996, 12, 413–435. [Google Scholar] [CrossRef]
- Hayes, J.D.; Pulford, D.J. The glutathione S-transferase supergene family: Regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 445–600. [Google Scholar] [CrossRef] [PubMed]
- Hodges, R.E.; Minich, D.M. Modulation of metabolic detoxification pathways using foods and food-derived components: A scientific Review with clinical application. J. Nutr. Metab. 2015, 2015, 760689. [Google Scholar] [CrossRef] [PubMed]
- Vanduchova, A.; Anzenbacher, P.; Anzenbacherova, E. Isothiocyanate from Broccoli, Sulforaphane, and Its Properties. J. Med. Food 2019, 22, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Johnson, I.T. Cruciferous vegetables and risk of cancers of the gastrointestinal tract. Mol Nutr Food Res. 2018, 62, e1701000. [Google Scholar] [CrossRef] [PubMed]
- Saleh, D.O.; Mansour, D.F.; Hashad, I.M.; Bakeer, R.M. Effects of sulforaphane on D-galactose-induced liver aging in rats: Role of keap-1/nrf-2 pathway. Eur. J. Pharmacol. 2019, 855, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Abdull Razis, A.F.; Konsue, N.; Ioannides, C. Isothiocyanates and xenobiotic detoxification. Mol. Nutr. Food Res. 2018, 62, e1700916. [Google Scholar] [CrossRef]
- Navarro, S.L.; Chang, J.L.; Peterson, S.; Chen, C.; King, I.B.; Schwarz, Y.; Li, S.S.; Li, L.; Potter, J.D.; Lampe, J.W. Modulation of human serum glutathione S-transferase A1/2 concentration by cruciferous vegetables in a controlled feeding study is influenced by GSTM1 and GSTT1 genotypes. Cancer Epidemiol. Biomarkers Prev. 2009, 18, 2974–2978. [Google Scholar] [CrossRef] [PubMed]
- Navarro, S.L.; Schwarz, Y.; Song, X.; Wang, C.Y.; Chen, C.; Trudo, S.P.; Kristal, A.R.; Kratz, M.; Eaton, D.L.; Lampe, J.W. Cruciferous vegetables have variable effects on biomarkers of systemic inflammation in a randomized controlled trial in healthy young adults. J. Nutr. 2014, 144, 1850–1857. [Google Scholar] [CrossRef]
- Brauer, H.A.; Libby, T.E.; Mitchell, B.L.; Li, L.; Chen, C.; Randolph, T.W.; Lampe, P.D. Cruciferous vegetable supplementation in a controlled diet study alters the serum peptidome in a GSTM1-genotype dependent manner. Nutr. J. 2011, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Navarro, S.L.; Peterson, S.; Chen, C.; Makar, K.W.; Schwarz, Y.; King, I.B.; Li, S.S.; Li, L.; Kestin, M.; Lampe, J.W. Cruciferous vegetable feeding alters UGT1A1 activity: Diet- and genotype-dependent changes in serum bilirubin in a controlled feeding trial. Cancer Prev. Res. 2009, 2, 345–352. [Google Scholar] [CrossRef]
- Peterson, S.; Schwarz, Y.; Li, S.S.; Li, L.; King, I.B.; Chen, C.; Eaton, D.L.; Potter, J.D.; Lampe, J.W. CYP1A2, GSTM1, and GSTT1 polymorphisms and diet effects on CYP1A2 activity in a crossover feeding trial. Cancer Epidemiol. Biomarkers Prev. 2009, 18, 3118–3125. [Google Scholar] [CrossRef]
- Riso, P.; Brusamolino, A.; Moro, M.; Porrini, M. Absorption of bioactive compounds from steamed broccoli and their effect on plasma glutathione S-transferase activity. Int. J. Food Sci. Nutr. 2009, 60 (Suppl. S1), 56–71. [Google Scholar] [CrossRef]
- Riso, P.; Martini, D.; Møller, P.; Loft, S.; Bonacina, G.; Moro, M.; Porrini, M. DNA damage and repair activity after broccoli intake in young healthy smokers. Mutagenesis 2010, 25, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Zhang, M.; Xie, X.; Jin, J.; Holman, C.A.J. Green tea consumption and glutathione S-transferases genetic polymorphisms on the risk of adult leukemia. Eur. J. Nutr. 2017, 56, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Betts, N.M.; Mulugeta, A.; Tong, C.; Newman, E.; Lyons, T.J. Green tea supplementation increases glutathione and plasma antioxidant capacity in adults with the metabolic syndrome. Nutr. Res. 2013, 33, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Pourahmadi, Z.; Mahboob, S.; Saedisomeolia, A.; Reykandeh, M.T. The effect of tomato juice consumption on antioxidant status in overweight and obese females. Women Health 2015, 55, 795–804. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Huerta, O.D.; Aguilera, C.M.; Martin, M.V.; Soto, M.J.; Rico, M.C.; Vallejo, F.; Tomas-Barberan, F.; Perez-de-la-Cruz, A.J.; Gil, A.; Mesa, M.D. Normal or high polyphenol concentration in orange Juice affects antioxidant activity, blood pressure, and body weight in obese or overweight adults. J. Nutr. 2015, 145, 1808–1816. [Google Scholar] [CrossRef] [PubMed]
- Ghavipour, M.; Sotoudeh, G.; Ghorbani, M. Tomato juice consumption improves blood antioxidative biomarkers in overweight and obese females. Clin. Nutr. 2015, 34, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Wei, J.; Yang, J.; Xu, J.; Pang, W.; Jiang, Y. Pomegranate juice is potentially better than apple juice in improving antioxidant function in elderly subjects. Nutr. Res. 2008, 28, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.; Turki, M.; Hammouda, O.; Chtourou, H.; Trabelsi, K.; Bouaziz, M.; Abdelkarim, O.; Hoekelmann, A.; Ayadi, F.; Souissi, N.; et al. Effects of pomegranate juice supplementation on oxidative stress biomarkers following weightlifting exercise. Nutrients 2017, 9, 819. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.R.; Han, J.H.; Lee, H.J.; Park, Y.K.; Kang, M.H. Purple grape juice supplementation in smokers and antioxidant status according to different types of GST polymorphisms. J. Clin. Biochem. Nutr. 2015, 56, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Toaldo, I.M.; Cruz, F.A.; da Silva, E.L.; Bordignon-Luiz, M.T. Acute consumption of organic and conventional tropical grape juices (Vitis labrusca L.) increases antioxidants in plasma and erythrocytes, but not glucose and uric acid levels, in healthy individuals. Nutr. Res. 2016, 36, 808–817. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Han, J.H.; Park, Y.K.; Kang, M.H. Effects of glutathione s-transferase (GST) M1 and T1 polymorphisms on antioxidant vitamins and oxidative stress-related parameters in Korean subclinical hypertensive subjects after kale juice (Brassica oleracea acephala) supplementation. Nutr. Res. Pract. 2018, 12, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Singletary, K.W.; Rokusek, J.T. Tissue-specific enhancement of xenobiotic detoxification enzymes in mice by dietary rosemary extract. Plant Foods Hum. Nutr. 1997, 50, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Singletary, K.W. Rosemary extract and carnosol stimulate rat liver glutathione-S-transferase and quinone reductase activities. Cancer Lett. 1996, 100, 139–144. [Google Scholar] [CrossRef]
- Rašković, A.; Milanović, I.; Pavlović, N.; Ćebović, T.; Vukmirović, S.; Mikov, M. Antioxidant activity of rosemary (Rosmarinus officinalis L.) essential oil and its hepatoprotective potential. BMC Complement. Altern Med. 2014, 14, 225. [Google Scholar] [CrossRef]
- Abrahams, S.; Haylett, W.L.; Johnson, G.; Carr, J.A.; Bardien, S. Antioxidant effects of curcumin in models of neurodegeneration, aging, oxidative and nitrosative stress: A review. Neuroscience 2019, 406, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Mendoza, N.; Madrigal-Santillán, E.; Morales-González, A.; Esquivel-Soto, J.; Esquivel-Chirino, C.; García-Luna, Y.; González-Rubio, M.; Gayosso-de-Lucio, J.A.; Morales-González, J.A. Hepatoprotective effect of silymarin. World J. Hepatol. 2014, 6, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Hatta, S.; Wada, K.; Ueda, N.; Yoshimura, T.; Endo, T.; Sakata, M.; Tanaka, T.; Haga, M. Effects of extract of Ginkgo biloba leaves and its constituents on carcinogen-metabolizing enzyme activities and glutathione levels in mouse liver. Life Sci. 2002, 70, 1657–1667. [Google Scholar] [CrossRef]
- Lee, H.Y.; Kim, S.W.; Lee, G.H.; Choi, M.K.; Jung, H.W.; Kim, Y.J.; Kwon, H.J.; Chae, H.J. Turmeric extract and its active compound, curcumin, protect against chronic CCl4-induced liver damage by enhancing antioxidation. BMC Complement. Altern Med. 2016, 16, 316. [Google Scholar] [CrossRef] [PubMed]
- Demirkol, O.; Adams, C.; Ercal, N. Biologically important thiols in various vegetables and fruits. J. Agric Food Chem. 2004, 52, 8151–8154. [Google Scholar] [CrossRef] [PubMed]
- Sarvan, I.; Kramer, E.; Bouwmeester, H.; Dekker, M.; Verkerk, R. Sulforaphane formation and bioaccessibility are more affected by steaming time than meal composition during in vitro digestion of broccoli. Food Chem. 2017, 214, 580–586. [Google Scholar] [CrossRef]
- Dosz, E.B.; Jeffery, E.H. Modifying the processing and handling of frozen broccoli for increased sulforaphane formation. J. Food Sci. 2013, 78, H1459–H1463. [Google Scholar] [CrossRef] [PubMed]
- Ghawi, S.K.; Methven, L.; Niranjan, K. The potential to intensify sulforaphane formation in cooked broccoli (Brassica oleracea var. italica) using mustard seeds (Sinapis alba). Food Chem. 2013, 138, 1734–1741. [Google Scholar] [CrossRef] [PubMed]
- Okunade, O.; Niranjan, K.; Ghawi, S.K.; Kuhnle, G.; Methven, L. Supplementation of the diet by exogenous myrosinase via mustard seeds to increase the bioavailability of sulforaphane in healthy human subjects after the consumption of cooked broccoli. Mol. Nutr. Food Res. 2018, 62, e1700980. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, G.F.; Lee, K.; Lopez, R.; Previs, S.F.; Willard, B.; McCullough, A.; Kasumov, T. A Western diet induced NAFLD in LDLR(-/)(-) mice is associated with reduced hepatic glutathione synthesis. Free Radic. Biol. Med. 2016, 96, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Bettermann, E.L.; Hartman, T.J.; Easley, K.A.; Ferranti, E.P.; Jones, D.P.; Quyyumi, A.A.; Vaccarino, V.; Ziegler, T.R.; Alvarez, J.A. Higher mediterranean diet quality scores and lower body mass index are associated with a less-oxidized plasma glutathione and cysteine redox status in adults. J. Nutr. 2018, 148, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Gaby, A. 62: Alpha-lipoic acid. Available online: https://www.ncbi.nlm.nih.gov/pubmed/18315507 (accessed on 28 June 2019).
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013, 15, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Gaby, A. 51: N-Acetylcysteine. In Nutritional Medicine; Concord, N.H., Ed.; Fritz Perlberg Publishing: Concord, NH, USA, 2011. [Google Scholar]
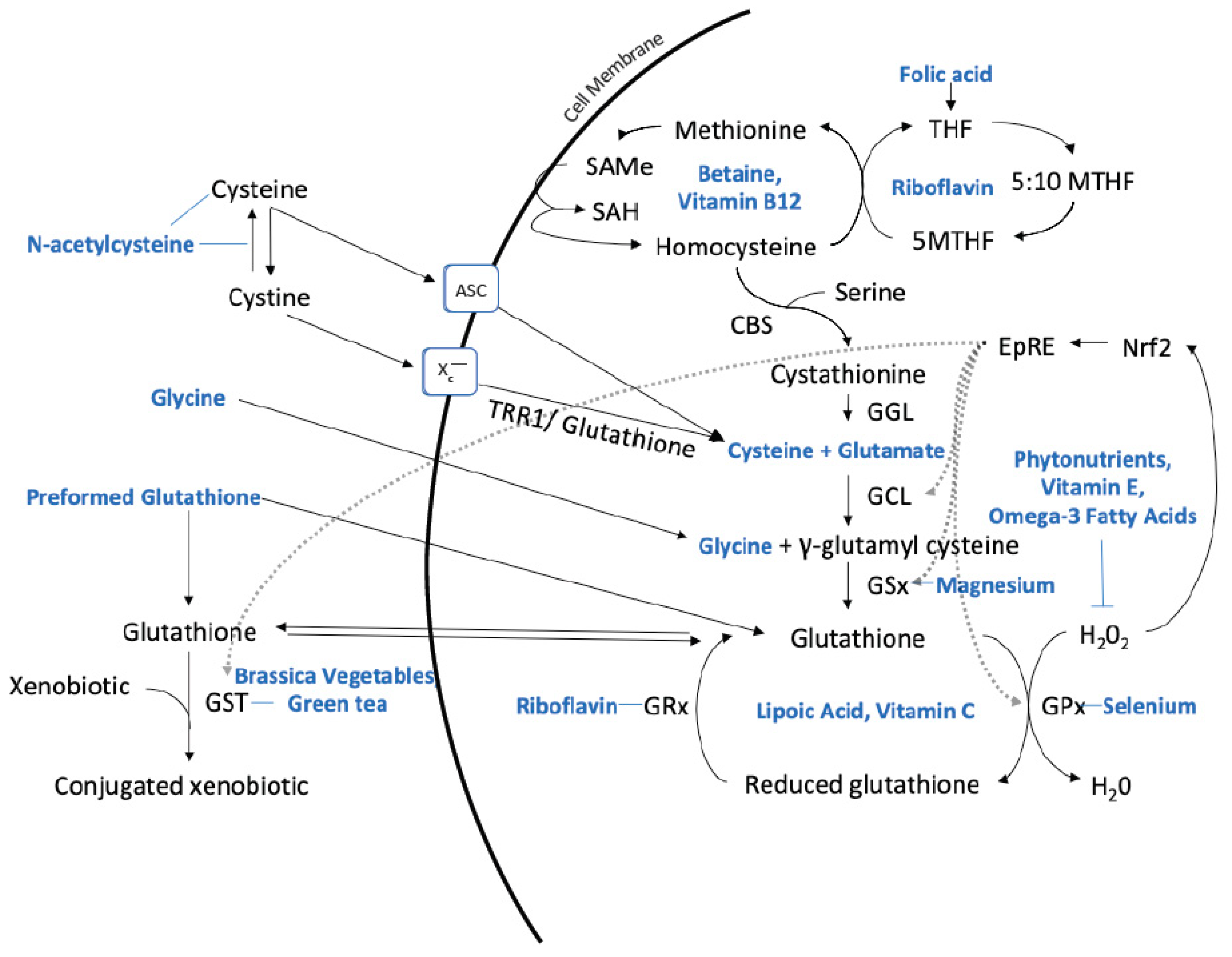
| Research has found that many chronic diseases are associated with a reduction in glutathione levels, leading to the hypothesis that increasing glutathione levels can help prevent and/or mitigate the progression of these diseases. Below is a list of some of the diseases [2] and issues associated with glutathione dysregulation or deficiency [3]: |
| • aging [15] and related disorders [3] |
| • Alzheimer’s disease [16] |
| • cancer [17] |
| • chronic liver disease [18] |
| • cognitive impairment [19] |
| • cystic fibrosis [20] |
| • diabetes [21], especially uncontrolled diabetes [22] |
| • human immunodeficiency virus (HIV)/ acquired immune deficiency syndrome (AIDS) [23] |
| • hypertension [24] |
| • infertility in both men and women [25] |
| • lupus [26] |
| • mental health disorders [27] |
| • multiple sclerosis [28] |
| • neurodegenerative disorders [29] |
| • Parkinson’s disease [30] |
| Food | Glutathione | NAC | Cysteine |
|---|---|---|---|
| Asparagus | 349 ± 26 | 46 ± 1 | 122 ± 1 |
| Avocado | 339 ± 10 | ND | 4 ± 1 |
| Banana | ND | ND | 7 ± 0 |
| Broccoli | 4 ± 1 | ND | ND |
| Carrot | 4 ± 0 | ND | ND |
| Cauliflower | 6 ± 1 | ND | 7 ± 1 |
| Cucumber | 123 ± 38 | 6 ± 1 | 11 ± 3 |
| Grapefruit | 13 ± 3 | 4 ± 0 | 15 ± 2 |
| Green Beans | 230 ± 2 | ND | 67 ± 11 |
| Green Pepper | 8 ± 1 | 12 ± 2 | 9 ± 1 |
| Green Squash | 47 ± 11 | ND | 6 ± 1 |
| Lemon | 5 ± 0 | 4 ± 0 | 6 ± 0 |
| Mango | 59 ± 6 | ND | 10 ± 0 |
| Orange | 5 ± 11 | ND | 41 ± 2 |
| Papaya | 136 ± 12 | ND | 58 ± 5 |
| Parsley | 17 ± 9 | 9 ± 1 | 8 ± 1 |
| Potato | 5 ± 0 | ND | ND |
| Red Pepper | 42 ± 2 | 25 ± 4 | 349 ± 18 |
| Spinach | 313 ± 33 | ND | 84 ± 2 |
| Strawberry | 39 ± 8 | 5 ± 1 | 59 ± 5 |
| Tomato | 64 ± 10 | 3 ± 1 | 55 ± 3 |
| Yellow Squash | 39 ± 8 | ND | 27 ± 6 |
| Nutrient and Foods | Recommended Dosage |
|---|---|
| Alpha lipoic-acid | 300 mg 3× day; 200–600 mg/day [158] |
| Brassica vegetables | 250 g/day |
| Curcumin | Doses up to 12 g/day safe; 1–2 g/day found to benefit antioxidant capacity; increased bioavailability with piperine [159] |
| Fruit and vegetable juices | 300–400 mL/day |
| Glutathione (Liposomal) | 500–1000 mg/day [43] |
| Glutathione (Oral) | 500–1000 mg/day [41,42] |
| Glycine | 100 mg/kg/day [63] |
| Green tea | 4 cups/day |
| N-acetylcysteine | 600–1200 mg/day in divided doses, but up to 6000 mg/day have been shown effective in studies [30,53,56,160] |
| Omega-3 fatty acids | 4000 mg/day [76] |
| Salmon | 150 g twice a week [80] |
| Selenium | 247 μg/day of selenium enriched yeast; 100–200 ug/day. Anything above 400 ug/day watch for toxicity [103,160] |
| Vitamin C | 500–2000 mg/day [87,88] |
| Vitamin E | 100–400 IU/day [77,91] |
| Whey Protein | 40 g/day [72] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minich, D.M.; Brown, B.I. A Review of Dietary (Phyto)Nutrients for Glutathione Support. Nutrients 2019, 11, 2073. https://doi.org/10.3390/nu11092073
Minich DM, Brown BI. A Review of Dietary (Phyto)Nutrients for Glutathione Support. Nutrients. 2019; 11(9):2073. https://doi.org/10.3390/nu11092073
Chicago/Turabian StyleMinich, Deanna M., and Benjamin I. Brown. 2019. "A Review of Dietary (Phyto)Nutrients for Glutathione Support" Nutrients 11, no. 9: 2073. https://doi.org/10.3390/nu11092073
APA StyleMinich, D. M., & Brown, B. I. (2019). A Review of Dietary (Phyto)Nutrients for Glutathione Support. Nutrients, 11(9), 2073. https://doi.org/10.3390/nu11092073





