Efficacy of Arabica Versus Robusta Coffee in Improving Weight, Insulin Resistance, and Liver Steatosis in a Rat Model of Type-2 Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Animals
2.2. Study Design and Procedures
2.3. Experimental Measurements
2.4. Quantitative Real-Time Polymerase Chain Reaction
2.5. Statistical Analyses
3. Results
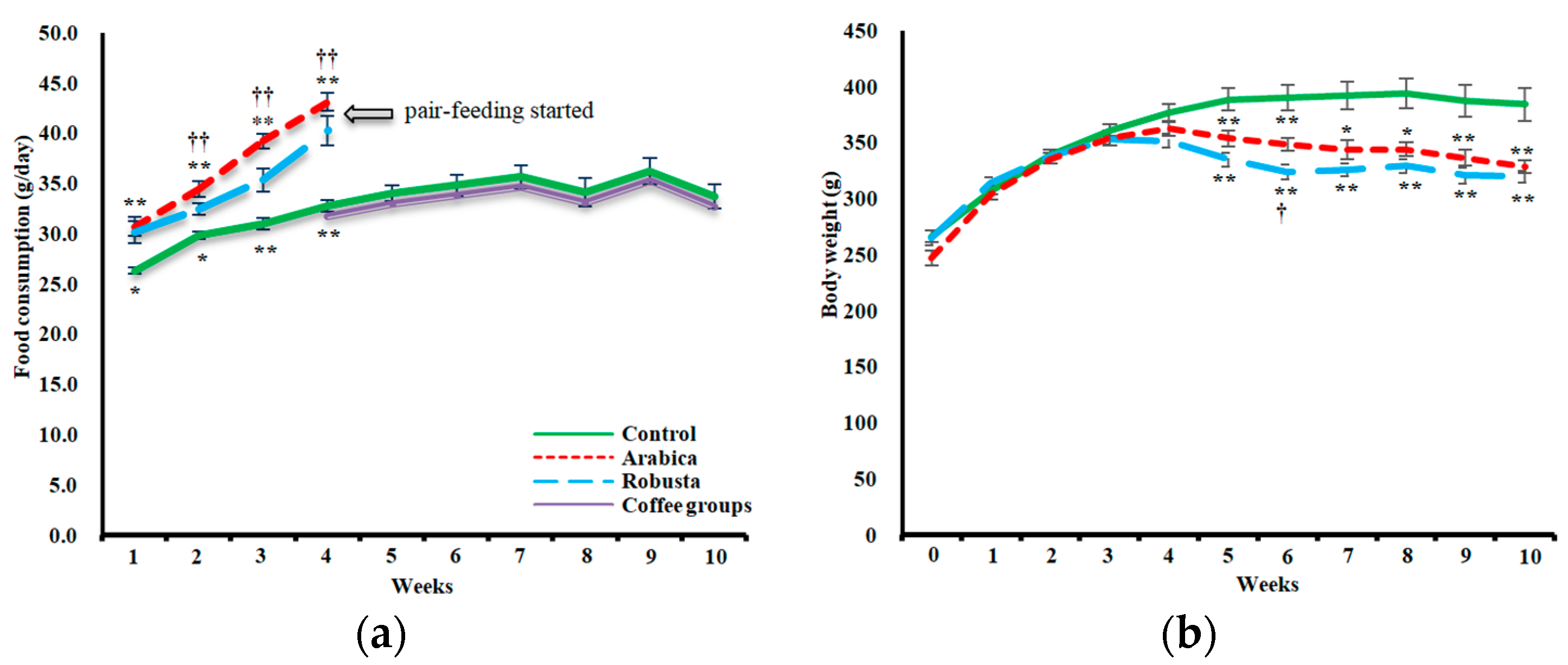
3.1. Food Consumption and Body Weight
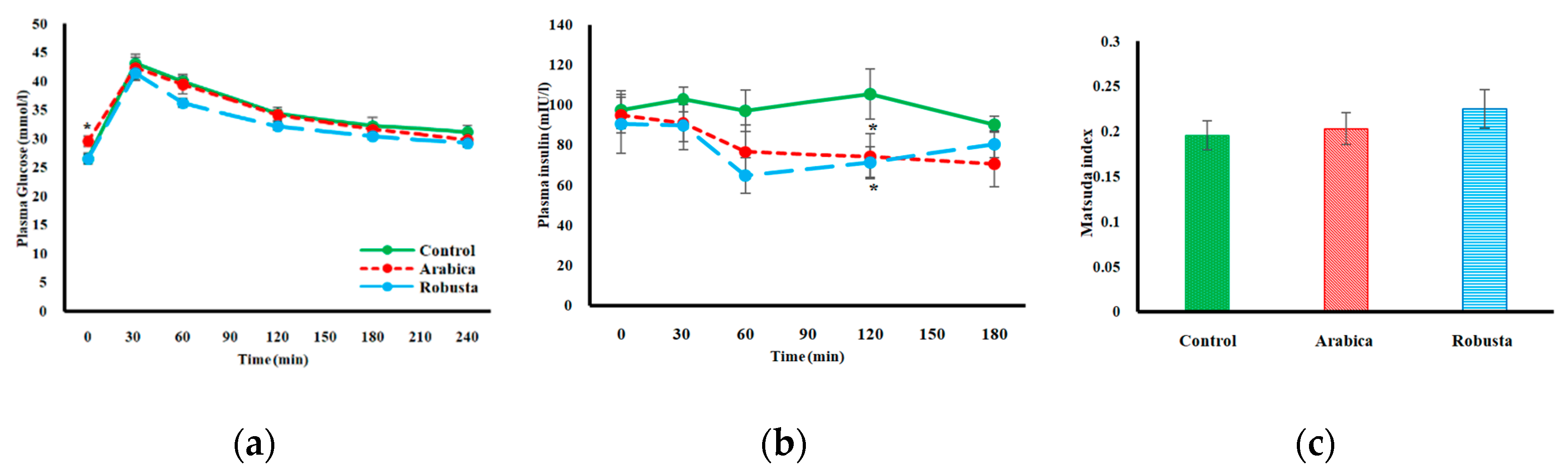
3.2. Glucose Homeostasis
3.3. Surrogate Indices of Insulin Resistance
3.4. Plasma Lipids
3.5. Plasma Adipokines
3.6. Liver Triglyceride Content
3.7. Gene Expression Analyses
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nieber, K. The Impact of Coffee on Health. Planta Med. 2017, 83, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Carlström, M.; Larsson, S. Coffee consumption and reduced risk of developing type 2 diabetes: A systematic review with meta-analysis. Nutr. Rev. 2018, 76, 395–417. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.J.; Pablos, F.; Gonzalez, A.G. Discrimination between arabica and robusta green coffee varieties according to their chemical composition. Talanta 1998, 46, 1259–1264. [Google Scholar] [CrossRef]
- Bicho, N.C.; Leitão, A.E.; Ramalho, J.C.; de Alvarenga, N.B.; Lidon, F.C. Identification of chemical clusters discriminators of Arabica and Robusta green coffee. Int. J. Food Prop. 2013, 16, 895–904. [Google Scholar] [CrossRef]
- Perrone, D.; Farah, A.; Donangelo, C.M.; de Paulis, T.; Martin, P.R. Comprehensive analysis of major and minor chlorogenic acids and lactones in economically relevant Brazilian coffee cultivars. Food Chem. 2008, 106, 859–867. [Google Scholar] [CrossRef]
- Tfouni, S.A.; Serrate, C.S.; Carreiro, L.B.; Camargo, M.C.; Teles, C.R.; Cipolli, K.M.; Furlani, R.P. Effect of roasting on chlorogenic acids, caffeine and polycyclic aromatic hydrocarbons levels in two Coffea cultivars: Coffea arabica cv. Catuaí Amarelo IAC-62 and Coffea canephora cv. Apoatã IAC-2258. J. Food Sci. Technol. 2012, 47, 406–415. [Google Scholar] [CrossRef]
- Farah, A.; Monteiro, M.C.; Calado, V.; Franca, A.S.; Trugo, L.C. Correlation between cup quality and chemical attributes of Brazilian coffee. Food Chem. 2006, 98, 373–380. [Google Scholar] [CrossRef]
- Mensink, R.P.; Lebbink, W.J.; Lobbezoo, I.E.; Weusten-Van der Wouw, M.P.; Zock, P.L.; Katan, M.B. Diterpene composition of oils from Arabica and Robusta coffee beans and their effects on serum lipids in man. J. Intern. Med. 1995, 237, 543–550. [Google Scholar] [CrossRef]
- van Rooij, J.; van der Stegen, G.H.; Schoemaker, R.C.; Kroon, C.; Burggraaf, J.; Hollaar, L.; Vroon, T.F.; Smelt, A.H.; Cohen, A.F. A placebo-controlled parallel study of the effect of two types of coffee oil on serum lipids and transaminases: Identification of chemical substances involved in the cholesterol-raising effect of coffee. Am. J. Clin. Nutr. 1995, 61, 1277–1283. [Google Scholar] [CrossRef]
- Abrahao, S.A.; Pereira, R.G.; de Sousa, R.V.; Lima, A.R.; Crema, G.P.; Barros, B.S. Influence of coffee brew in metabolic syndrome and type 2 diabetes. Plant Foods Hum. Nutr. 2013, 68, 184–189. [Google Scholar] [CrossRef]
- Yamauchi, R.; Kobayashi, M.; Matsuda, Y.; Ojika, M.; Shigeoka, S.; Yamamoto, Y.; Tou, Y.; Inoue, T.; Katagiri, T.; Murai, A.; et al. Coffee and caffeine ameliorate hyperglycemia, fatty liver, and inflammatory adipocytokine expression in spontaneously diabetic KK-Ay mice. J. Agric. Food Chem. 2010, 58, 5597–5603. [Google Scholar] [CrossRef]
- Ong, K.W.; Hsu, A.; Tan, B.K. Anti-diabetic and anti-lipidemic effects of chlorogenic acid are mediated by ampk activation. Biochem. Pharmacol. 2013, 85, 1341–1351. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez de Sotillo, D.V.; Hadley, M. Chlorogenic acid modifies plasma and liver concentrations of: Cholesterol, triacylglycerol, and minerals in (fa/fa) Zucker rats. J. Nutr. Biochem. 2002, 13, 717–726. [Google Scholar] [CrossRef]
- Yoshinari, O.; Igarashi, K. Anti-diabetic effect of trigonelline and nicotinic acid, on KK-A(y) mice. Curr. Med. Chem. 2010, 17, 2196–2202. [Google Scholar] [CrossRef]
- Wang, Y.W.; Sun, G.D.; Sun, J.; Liu, S.J.; Wang, J.; Xu, X.H.; Miao, L.N. Spontaneous type 2 diabetic rodent models. J. Diabetes Res. 2013, 401723. [Google Scholar] [CrossRef] [PubMed]
- Poole, R.; Ewings, S.; Parkes, J.; Fallowfield, J.A.; Roderick, P. Misclassification of coffee consumption data and the development of a standardised coffee unit measure. BMJ Nutr. 2019, 1–9. [Google Scholar] [CrossRef]
- Brader, L.; Overgaard, A.; Christensen, L.P.; Jeppesen, P.B.; Hermansen, K. Polyphenol-rich bilberry ameliorates total cholesterol and LDL-cholesterol when implemented in the diet of Zucker diabetic fatty rats. Rev. Diabet. Stud. 2013, 10, 270–282. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Muniyappa, R.; Lee, S.; Chen, H.; Quon, M.J. Current approaches for assessing insulin sensitivity and resistance in vivo: Advantages, limitations, and appropriate usage. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E15–E26. [Google Scholar] [CrossRef]
- Jouihan, H. Measurement of Liver Triglyceride Content. Bio-Protocol. 2012, 2. [Google Scholar] [CrossRef]
- Hayter, A.J. The maximum familywise error rate of Fisher’s least significant difference test. J. Am. Stat. Assoc. 1986, 81, 1000–1004. [Google Scholar] [CrossRef]
- Rustenbeck, I.; Lier-Glaubitz, V.; Willenborg, M.; Eggert, F.; Engelhardt, U.; Jorns, A. Effect of chronic coffee consumption on weight gain and glycaemia in a mouse model of obesity and type 2 diabetes. Nutr. Diabetes 2014, 4, e123. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; DeFronzo, R.A. Insulin sensitivity indices obtained from oral glucose tolerance testing: Comparison with the euglycemic insulin clamp. Diabetes Care 1999, 22, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Zhang, D.; Jiang, W. Coffee and caffeine intake and incidence of type 2 diabetes mellitus: A meta-analysis of prospective studies. Eur. J. Nutr. 2014, 53, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wang, M. Development and application of rodent models for type 2 diabetes. Diabetes Obes. Metab. 2005, 7, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, H.; Seki, E.; Aitani, M. Inhibitory effect of green coffee bean extract on fat accumulation and body weight gain in mice. BMC Complement. Altern. Med. 2006, 6, 1–9. [Google Scholar] [CrossRef]
- Santos, R.M.; Lima, D.R. Coffee consumption, obesity and type 2 diabetes: A mini-review. Eur. J. Nutr. 2016, 55, 1345–1358. [Google Scholar] [CrossRef]
- Tokuyama, Y.; Sturis, J.; DePaoli, A.M.; Takeda, J.; Stoffel, M.; Tang, J.; Bell, G.I. Evolution of β-cell dysfunction in the male Zucker diabetic fatty rat. Diabetes 1995, 44, 1447–1457. [Google Scholar] [CrossRef]
- Leonard, B.L.; Watson, R.N.; Loomes, K.M.; Phillips, A.R.; Cooper, G.J. Insulin resistance in the Zucker diabetic fatty rat: A metabolic characterisation of obese and lean phenotypes. Acta Diabetol. 2005, 42, 162–170. [Google Scholar] [CrossRef]
- Ferrannini, E.; Mari, A. How to measure insulin sensitivity. J. Hypertens. 1998, 16, 895–906. [Google Scholar] [CrossRef] [PubMed]
- Cowan, T.E.; Palmnas, M.S.; Yang, J.; Bomhof, M.R.; Ardell, K.L.; Reimer, R.A.; Vogel, H.J.; Shearer, J. Chronic coffee consumption in the diet-induced obese rat: Impact on gut microbiota and serum metabolomics. J. Nutr. Biochem. 2014, 25, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Kobayashi, M.; Yamauchi, R.; Ojika, M.; Hiramitsu, M.; Inoue, T.; Katagiri, T.; Murai, A.; Horio, F. Coffee and caffeine improve insulin sensitivity and glucose tolerance in C57BL/6J mice fed a high-fat diet. Biosci. Biotechnol. Biochem. 2011, 75, 2309–2315. [Google Scholar] [CrossRef] [PubMed]
- Gross, G.; Jaccaud, E.; Huggett, A.C. Analysis of the content of the diterpenescafestol and kahweol in coffee brews. Food Chem. Toxicol. 1997, 35, 547–554. [Google Scholar] [CrossRef]
- Beynen, A.C.; Weusten-Van der Wouw, M.P.; de Roos, B.; Katan, M.B. Boiled coffee fails to raise serum cholesterol in hamsters and rats. Br. J. Nutr. 1996, 76, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Karthikesan, K.; Pari, L.; Menon, V.P. Antihyperlipidemic effect of chlorogenic acid and tetrahydrocurcumin in rats subjected to diabetogenic agents. Chem.-Biol. Interac. 2010, 188, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Hamden, K.; Mnafgui, K.; Amri, Z.; Aloulou, A.; Elfeki, A. Inhibition of key digestive enzymes related to diabetes and hyperlipidemia and protection of liver-kidney functions by trigonelline in diabetic rats. Sci. Pharm. 2013, 81, 233–246. [Google Scholar] [CrossRef]
- Williams, C.J.; Fargnoli, J.L.; Hwang, J.J.; van Dam, R.M.; Blackburn, G.L.; Hu, F.B.; Mantzoros, C.S. Coffee consumption is associated with higher plasma adiponectin concentrations in women with or without type 2 diabetes: A prospective cohort study. Diabetes Care 2008, 31, 504–507. [Google Scholar] [CrossRef]
- Wedick, N.M.; Brennan, A.M.; Sun, Q.; Hu, F.B.; Mantzoros, C.S.; van Dam, R.M. Effects of caffeinated and decaffeinated coffee on biological risk factors for type 2 diabetes: A randomized controlled trial. Nutr. J. 2011, 10, 1–9. [Google Scholar] [CrossRef]
- Lihn, A.S.; Pedersen, S.B.; Richelsen, B. Adiponectin: Action, regulation and association to insulin sensitivity. Obes. Rev. 2005, 6, 13–21. [Google Scholar] [CrossRef]
- Janssen, S.W.; Martens, G.J.; Sweep, C.G.; Ross, H.A.; Hermus, A.R. In Zucker diabetic fatty rats plasma leptin levels are correlated with plasma insulin levels rather than with body weight. Horm. Metab. Res. 1999, 31, 610–615. [Google Scholar] [CrossRef]
- Marventano, S.; Salomone, F.; Godos, J.; Pluchinotta, F.; Del Rio, D.; Mistretta, A.; Grosso, G. Coffee and tea consumption in relation with non-alcoholic fatty liver and metabolic syndrome: A systematic review and meta-analysis of observational studies. Clin. Nutr. 2016, 35, 1269–1281. [Google Scholar] [CrossRef] [PubMed]
- Clore, J.N.; Stillman, J.; Sugerman, H. Glucose-6-phosphatase flux in vitro is increased in type 2 diabetes. Diabetes 2000, 49, 969–974. [Google Scholar] [CrossRef]
- Mues, C.; Zhou, J.; Manolopoulos, K.N.; Korsten, P.; Schmoll, D.; Klotz, L.O.; Barthel, A. Regulation of glucose-6-phosphatase gene expression by insulin and metformin. Horm. Metab. Res. 2009, 41, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Cui, F.; Meng, L.; Chen, G.; Li, Z. Carnitine Palmitoyltransferase Inhibitor in Diabetes. J. Mol. Gen. Med. 2016, 10, 238. [Google Scholar] [CrossRef]
- Conti, R.; Mannucci, E.; Pessotto, P.; Tassoni, E.; Carminati, P.; Giannessi, F.; Arduini, A. Selective reversible inhibition of liver carnitinepalmitoyl-transferase 1 by teglicar reduces gluconeogenesis and improves glucose homeostasis. Diabetes 2011, 60, 644–651. [Google Scholar] [CrossRef]
- Keung, W.; Ussher, J.R.; Jaswal, J.S.; Raubenheimer, M.; Lam, V.H.; Wagg, C.S.; Lopaschuk, G.D. Inhibition of carnitine palmitoyltransferase-1 activity alleviates insulin resistance in diet-induced obese mice. Diabetes 2013, 62, 711–720. [Google Scholar] [CrossRef]
- Khamzina, L.; Veilleux, A.; Bergeron, S.; Marette, A. Increased activation of the mammalian target of rapamycin pathway in liver and skeletal muscle of obese rats: Possible involvement in obesity-linked insulin resistance. Endocrinology 2005, 146, 1473–1481. [Google Scholar] [CrossRef]
- Reinke, A.; Chen, J.C.; Aronova, S.; Powers, T. Caffeine targets TOR complex I and provides evidence for a regulatory link between the FRB and kinase domains of Tor1p. J. Biol. Chem. 2006, 281, 31616–31626. [Google Scholar] [CrossRef]
- Mellbye, F.B.; Jeppesen, P.B.; Hermansen, K.; Gregersen, S. Cafestol, a Bioactive Substance in Coffee, Stimulates Insulin Secretion and Increases Glucose Uptake in Muscle Cells: Studies in Vitro. J. Nat. Prod. 2015, 78, 2447–2451. [Google Scholar] [CrossRef]
- Wang, B.; Chandrasekera, P.C.; Pippin, J.J. Leptin- and leptin receptor-deficient rodent models: Relevance for human type 2 diabetes. Curr. Diabetes Rev. 2014, 10, 131–145. [Google Scholar] [CrossRef] [PubMed]


| Gene Symbol | Gene Full Name | TaqMan Assay ID |
|---|---|---|
| AdipoR1 | Adiponectin receptor 1 | Rn01483784-m1 |
| Prkab1 | Protein kinase AMP-activated non-catalytic subunit beta 1 | Rn01499630-m1 |
| Prkaa1 | Protein kinase AMP-activated catalytic subunit alfa 1 | Rn00665045-m1 |
| Insr | Insulin receptor | Rn01637243-m1 |
| Irs1 | Insulin receptor substrate 1 | Rn02132493-s1 |
| Irs2 | Insulin receptor substrate 2 | Rn01482270-s1 |
| Slc2a2 | Solute carrier family 2 member 2 | Rn00563565-m1 |
| G6pc | Glucose-6-phosphatase, catalytic subunit | Rn00565347-m1 |
| Akt2 | AKT serine/threonine kinase 2 | Rn00690900-m1 |
| Acaca | Acetyl-CoA carboxylase alpha | Rn00573474-m1 |
| Cpt1a | Carnitine palmitoyltransferase1A | Rn00580702-m1 |
| Gys2 | Glycogen synthase 2 | Rn00565296-m1 |
| Gcgr | Glucagon receptor | Rn00597158-m1 |
| mTOR | Mechanistic target of rapamycin | Rn00571541-m1 |
| Pik3cg | Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit gamma | Rn01289357-g1 |
| Ppara | Peroxisome proliferator-activated receptor alfa | Rn00566193-m1 |
| Ppargc1a | Peroxisome proliferator-activated receptor gamma coactivator 1 alpha | Rn00580241-m1 |
| Srebf1 | Sterol regulatory element binding transcription factor 1 | Rn01495769-m1 |
| Nr1h3 | Nuclear receptor subfamily 1, group H, member 3 | Rn00581185-m1 |
| Tnfrsf1a | Tumor necrosis factor receptor superfamily, member 1A | Rn01492348-m1 |
| Lipc | Lipase C, hepatic type | Rn00561474-m1 |
| Hprt1 | Hypoxanthine phosphoribosyltransferase 1 | Rn01527840-m1 |
| Gapdh | Glyceraldehyde-3-phosphate dehydrogenase | Rn01775763-g1 |
| Haemoglobin A1C (mmol/mol) | Triglyceride (mmol/L) | Total Cholesterol (mmol/L) | HDL Cholesterol (mmol/L) | LDL Cholesterol (mmol/L) | Free Fatty Acids (mmol/L) | Liver TG Content (mmol/L) | Adiponectin (µg/mL) | ||
|---|---|---|---|---|---|---|---|---|---|
| Control | Baseline | 20.9 (19.62–22.18) | 1.76 (1.25–2.27) | 2.90 (2.67–3.12) | 1.61 (1.41–1.81) | 0.34 (0.26–0.41) | ˗ | ˗ | ˗ |
| Endpoint | 61.8 (55.69–67.91) | 2.35 (1.82–2.88) | 4.40 (3.97–4.84) | 2.96 (2.48–3.43) | 0.69 (0.51–0.87) | 0.55 (0.46–0.64) | 0.79 (0.68–0.90) | 4.01 (3.71–4.31) | |
| Difference | 40.4 (33.05–47.75) | 0.51 (0.41–1.43) | 1.51 (1.09–1.92) | 1.35 (0.84–1.85) | 0.36 (0.18–0.53) | ˗ | ˗ | ˗ | |
| Arabica | Baseline | 22.4 (20.92–23.88) | 1.93 (1.43–2.44) | 3.23 (2.79–3.67) | 1.66 (1.39–1.92) | 0.35 (0.22–0.48) | ˗ | ˗ | ˗ |
| Endpoint | 66.6 (62.81–70.39) †† | 2.72 (2.23–3.20) | 4.63 (4.24–5.02) †† | 3.45 (2.95–3.94) † | 0.61 (0.49–0.73) | 0.57 (0.47–0.66) | 0.56 (0.47–0.64) ** | 4.51 (4.00–5.03) | |
| Difference | 43.7 (38.73–48.67) † | 0.78 (0.14–1.43) | 1.40 (0.67–2.13) | 1.79 (1.24–2.33) †† | 0.26 (0.04–0.48) | ˗ | ˗ | ˗ | |
| Robusta | Baseline | 22.82 (21.07–24.57) | 1.72 (1.49–1.95) | 3.11 (2.76–3.47) | 1.78 (1.59–1.96) | 0.31 (0.20–0.43) | ˗ | ˗ | ˗ |
| Endpoint | 57.27 (53.29–61.26) †† | 3.06 (2.41–3.71) | 5.45 (4.94–5.95) **†† | 4.19 (3.59–4.79) **† | 0.77 (0.65–0.89) | 0.58 (0.50–0.65) | 0.62 (0.53–0.71) ** | 5.08 (4.41–5.74) ** | |
| Difference | 34.27 (30.22–38.33) † | 1.34 (0.65–2.04) | 2.33 (1.60–3.07) | 2.41 (1.80–3.01) †† | 0.46 (0.30–0.61) | ˗ | ˗ | ˗ | |
| F-test p-value | Baseline | 0.133 | 0.773 | 0.343 | 0.458 | 0.875 | ˗ | ˗ | ˗ |
| Endpoint | 0.013 | 0.680 | 0.002 | 0.004 | 0.196 | 0.902 | 0.002 | 0.012 | |
| Difference | 0.033 | 0.837 | 0.056 | 0.017 | 0.238 | ˗ | ˗ | ˗ | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shokouh, P.; Jeppesen, P.B.; Christiansen, C.B.; Mellbye, F.B.; Hermansen, K.; Gregersen, S. Efficacy of Arabica Versus Robusta Coffee in Improving Weight, Insulin Resistance, and Liver Steatosis in a Rat Model of Type-2 Diabetes. Nutrients 2019, 11, 2074. https://doi.org/10.3390/nu11092074
Shokouh P, Jeppesen PB, Christiansen CB, Mellbye FB, Hermansen K, Gregersen S. Efficacy of Arabica Versus Robusta Coffee in Improving Weight, Insulin Resistance, and Liver Steatosis in a Rat Model of Type-2 Diabetes. Nutrients. 2019; 11(9):2074. https://doi.org/10.3390/nu11092074
Chicago/Turabian StyleShokouh, Pedram, Per B Jeppesen, Christine B Christiansen, Fredrik B Mellbye, Kjeld Hermansen, and Søren Gregersen. 2019. "Efficacy of Arabica Versus Robusta Coffee in Improving Weight, Insulin Resistance, and Liver Steatosis in a Rat Model of Type-2 Diabetes" Nutrients 11, no. 9: 2074. https://doi.org/10.3390/nu11092074
APA StyleShokouh, P., Jeppesen, P. B., Christiansen, C. B., Mellbye, F. B., Hermansen, K., & Gregersen, S. (2019). Efficacy of Arabica Versus Robusta Coffee in Improving Weight, Insulin Resistance, and Liver Steatosis in a Rat Model of Type-2 Diabetes. Nutrients, 11(9), 2074. https://doi.org/10.3390/nu11092074





