Effects of Olive Oil and Its Minor Components on Cardiovascular Diseases, Inflammation, and Gut Microbiota
Abstract
1. Introduction
2. Olive Oil
3. Cardiovascular Diseases and Olive Oil
4. Inflammatory Process and Olive Oil
5. Gut Microbiota
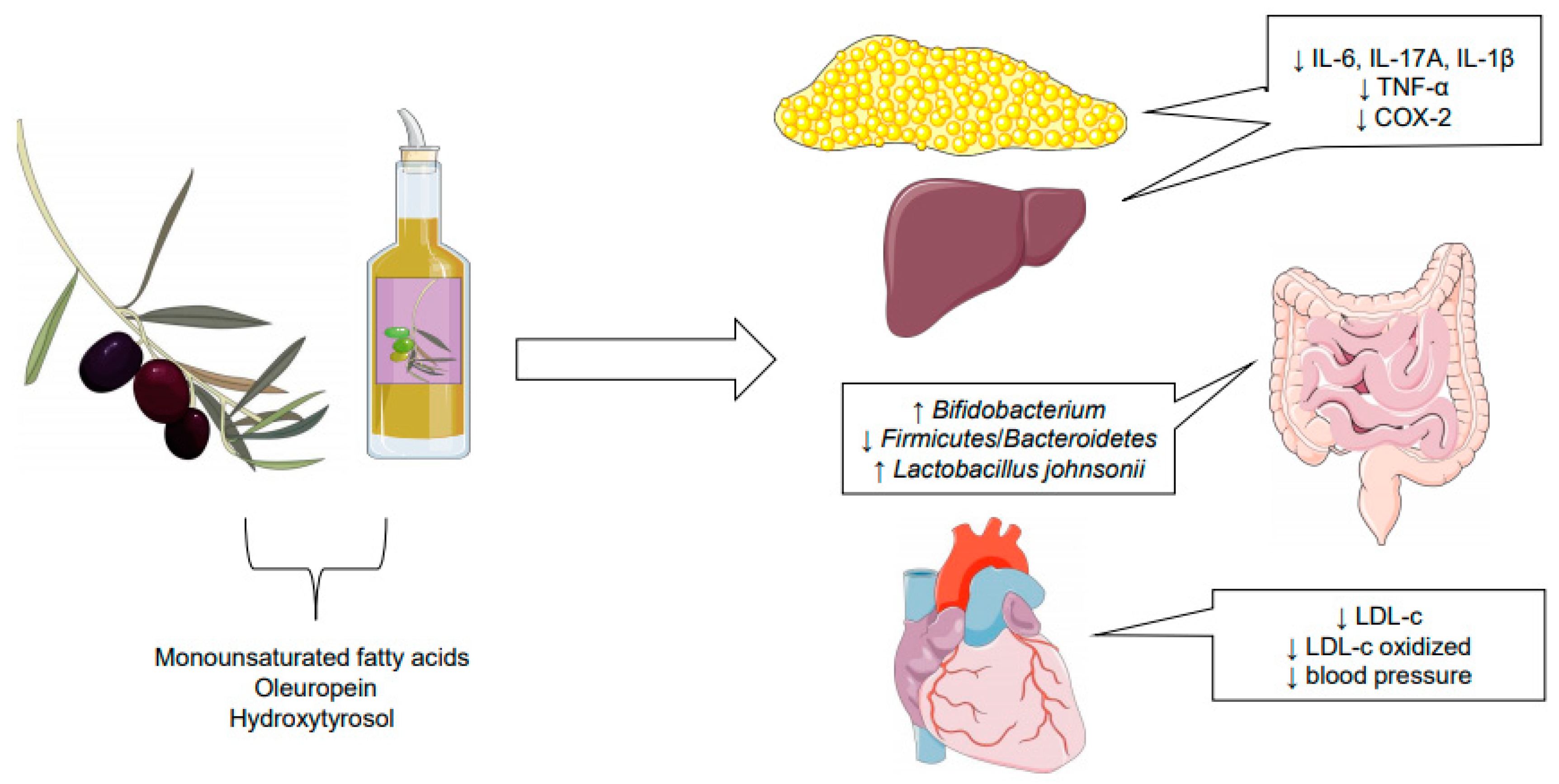
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Covas, M.I.; de la Torre, R.; Fitó, M. Virgin olive oil: A key food for cardiovascular risk protection. Br. J. Nutr. 2015, 113, 519–528. [Google Scholar] [CrossRef]
- Hohmann, C.D.; Cramer, H.; Michalsen, A.; Kessler, C.; Steckhan, N.; Choi, K.; Dobos, G. Effects of high phenolic olive oil on cardiovascular risk factors: A systematic review and meta-analysis. Phytomedicine 2016, 22, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Mastínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Ruiz-Gutiérrez, V.; Covas, M.I.; Fiol, M.; Gómez-Gracia, E.; López-Sabater, M.C.; Vinyoles, E.; et al. Effects of a Mediterranean-style diet on cardiovascular risk factors—A randomized trial. Ann. Intern. Med. 2006, 145, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Montserrat-de la Paz, S.; Bermidez, B.; Cardelo, M.P.; Lopez, S.; Abia, R.; Muriana, F.J.G. Olive oil and postprandial hyperlipidemia: Implications for atherosclerosis and metabolic syndrome. Food Funct. 2016, 7, 4743–4744. [Google Scholar] [CrossRef] [PubMed]
- Giner, E.; Recio, M.C.; Ríos, J.L.; Cerdá-Nicolás, J.M.; Giner, R.M. Chemopreventive effect of oleuropein in colitis-associated colorectal cancer in c57bl/6 mice. Mol. Nutr. Food Res. 2016, 60, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Foscolou, A.; Critselis, E.; Panagiotakos, D. Olive oil consumption and human health: A narrative review. Maturitas 2018, 118, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Castro-Barquero, S.; Lamuela-Raventós, R.M.; Doménech, M.; Estruch, R. Relationship between Mediterranean dietary polyphenol intake and obesity. Nutrients 2018, 10, 1523. [Google Scholar] [CrossRef] [PubMed]
- Scoditti, E.; Massaro, M.; Carluccio, M.A.; Pellegrino, M.; Wabitsch, M.; Calabriso, N.; Storelli, C.; De Caterina, R. Additive regulation of adiponectin expression by the mediterranean diet olive oil components oleic acid and hydroxytyrosol in human adipocytes. PLoS ONE 2015, 10, e0128218. [Google Scholar] [CrossRef]
- Commission Regulation (EU) No 432/2012 of 16 May 2012 establishing a list of permitted health claims made of foods, other than those referring to the reduction of disease risk and to children’s development and health. Off. J. Eur. Union 2012, 136, 1.
- Hidalgo, M.; Prieto, I.; Abriouel, H.; Villarejo, A.B.; Ramírez-Sánchez, M.; Cobo, A.; Benomar, N.; Gálvez, A.; Martínez-Cañamero, M. Changes in gut microbiota linked to a reduction in systolic blood pressure in spontaneously hypertensive rats fed an extra virgen olive oil-enriched diet. Plant Foods Hum. Nutr. 2018, 73, 1–6. [Google Scholar] [CrossRef]
- Deiana, M.; Serra, G.; Corona, G. Modulation of intestinal epithelium homeostasis by extra virgin olive oil phenolic compounds. Food Funct. 2018, 9, 4085–4099. [Google Scholar] [CrossRef]
- Shen, J.; Wilmot, K.A.; Ghasemzadeh, N.; Molloy, D.L.; Burkman, G.; Mekonnen, G.; Gongora, M.C.; Quyyumi, A.A.; Sperling, L.S. Mediterranean dietary patterns and cardiovascular health. Ann. Ver. Nutr. 2015, 35, 425–449. [Google Scholar] [CrossRef]
- Berroughui, H.; Ikhlef, S.; Khalil, A. Extra virgin olive oil polyphenols promote cholesterol efflux and improve HDL functionality. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The mediterranean diet, its components, and cardiovascular disease. Am. J. Med. 2015, 121, 229–238. [Google Scholar] [CrossRef]
- United States Department of Agriculture. Oilseeds: World Markets and Trade. 2019. Available online: https://apps.fas.usda.gov/psdonline/circulars/oilseeds.pdf (accessed on 14 April 2019).
- Priore, P.; Caruso, D.; Siculella, L.; Gnoni, G.V. Rapid down-regulation of hepatic lipid metabolism by phenolic fraction from extra virgin olive oil. Eur. J. Nutr. 2015, 54, 823–833. [Google Scholar] [CrossRef]
- Tarhan, I.; Ismail, A.A.; Kara, H. Quantitative determination of free fatty acids in extra virgin olive oils by multivariate methods and Fourier transform infrared spectroscopy considering different absorption modes. Int. J. Food Prop. 2017, 20, 790–797. [Google Scholar] [CrossRef]
- Roncero-Ramos, I.; Rangel-Zuñiga, O.A.; Lopez-Moreno, J.; Alcala-Diaz, J.F.; Perez-Martine, P.; Jimenez-Lucena, R.; Castaño, J.P.; Rochem, H.M.; Delgado-Lista, J.; Ordovas, J.M.; et al. Mediterranean diet, glucose homeostasis, and inflammasome genetic variants: The CORDIOPREV Study. Mol. Nutr. Food Res. 2018, 62, 1700960. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Castañer, O.; Konstantinidou, V.; Subirana, I.; Muñoz-Aguayo, D.; Blanchart, G.; Gaixas, S.; de la Torre, R.; Farré, M.; Sáez, G.T.; et al. Effect of olive oil phenolic compounds on the expression of blood pressure-related genes in healthy individuals. Eur. J. Nutr. 2017, 56, 663–670. [Google Scholar] [CrossRef]
- Piroddi, M.; Albini, A.; Fabiani, R.; Giovannelli, L.; Luceri, C.; Natella, F.; Rosignoli, P.; Rossi, T.; Taticchi, A.; Servili, M.; et al. Nutrigenomics of extra-virgin olive oils: A review. BioFactors 2016, 43, 17–41. [Google Scholar] [CrossRef]
- Venturini, D.; Simão, A.N.C.; Urbano, M.R.; Dichi, I. Effects of extra virgin olive oil and fish oil on lipid profile and oxidative stress in patients with metabolic syndrome. Nutrition 2015, 31, 834–840. [Google Scholar] [CrossRef]
- Soler-Rivas, C.; Espín, J.C.; Wichers, H.J. Oleuropein and related compounds. J. Sci. Food Agric. 2000, 80, 1013–1023. [Google Scholar] [CrossRef]
- Katsarou, A.I.; Kaliora, A.C.; Chiou, A.; Kalogeropoulos, N.; Papalois, A.; Agrogiannis, G.; Andrikopoulos, N.K. Amelioration of oxidative and inflammatory status in hearts of cholesterol-fed rats supplemented with oils or oil-products with extra virgem olive oil components. Eur. J. Nutr. 2016, 55, 1283–1296. [Google Scholar] [CrossRef]
- Crespo, M.C.; Tomé-Carneiro, J.; Dávalos, A.; Visioli, F. Pharma-nutritional properties of olive oil phenols. Transfer of new findings to human nutrition. Foods 2018, 7, 90. [Google Scholar] [CrossRef]
- Gungor, Z.B. Vascular Inflammation and genetic predisposition as risk factors for cardiovascular diseases. In Cholesterol—Good, Bad, and the Heart, 1st ed.; Nagpanl, M.L., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Avci, B.; Dolapoglu, A.; Akgun, D.E. Role of cholesterol as a risk factor in cardiovascular diseases. In Cholesterol—Good, Bad, and the Heart, 1st ed.; Nagpanl, M.L., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Hernáez, Á.; Sanllorente, A.; Castañer, O.; Martínez-González, M.Á.; Ros, E.; Pintó, X.; Estruch, R.; Salas-Salvadó, J.; Corella, D.; Alonso-Gómez, Á.M.; et al. Increased consumption of virgin olive oil, nuts, legumes, whole grains, and fish promotes HDL functions in humans. Mol. Nutr. Food Res. 2019, 63, 1800847. [Google Scholar] [CrossRef]
- Katsarou, A.I.; Kaliora, A.C.; Papalois, A.; Chiou, A.; Kalogeropoulos, N.; Agrogiannis, G.; Andrikopoulos, N.K. Serum lipid profile and inflammatory markers in the aorta of cholesterol-fed rats supplemented with extra virgin olive oil, sunflower oils and oil-products. Int. J. Food Sci. Nutr. 2015, 66, 766–773. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Mosele, J.I.; Pizarro, N.; Farràs, M.; de la Torre, R.; Subirana, I.; Pérez-Cano, F.J.; Castañer, O.; Solà, R.; Fernandez-Catillejo, S.; et al. Effect of virgin olive oil and thyme phenolic compounds on blood lipid profile: Implications of human gut microbiota. Eur. J. Nutr. 2017, 56, 119–131. [Google Scholar] [CrossRef]
- Cândido, F.G.; Valente, F.X.; Silva, L.E.; Coelho, O.G.L.; Peluzio, M.C.G.; Alfenas, R.C.G. Consumption of extra virgin olive oil improves body composition and blood pressure in women with excess body fat: A randomized, double-blinded, placebo-controlled clinical trial. Eur. J. Nutr. 2017, 57, 7. [Google Scholar]
- Prieto, I.; Hidalgo, M.; Segarra, A.B.; Martínez-Rodríguez, A.M.; Cobo, A.; Ramírez, M.; Abriouel, H.; Gálvez, A.; Martínez-Cañamero, M. Influence of a diet enriched with virgin olive oil or butter on mouse gut microbiota and its correlation to physiological and biochemical parameters related to metabolic syndrome. PLoS ONE 2018, 13, e0190368. [Google Scholar] [CrossRef]
- Sanchez-Rodriguez, E.; Lima-Cabello, E.; Biel-Glesson, S.; Fernandez-Navarro, J.R.; Calleja, M.A.; Roca, M.; Espejo-Calvo, J.A.; Gil-Extremera, B.; Soria-Florido, M.; de la Torre, R.; et al. Effects of virgin olive oils differing in their bioactive compound contents on metabolic syndrome and endothelial functional risk biomarkers in healthy adults: A randomized double-bling controlled trial. Nutrients 2018, 10, 626. [Google Scholar] [CrossRef]
- Hernáez, Á.; Castañer, O.L.; Goday, A.; Ros, E.; Pintó, X.; Estruch, R.; Salas-Salvadó, J.; Corella, D.; Arós, F.; Serra-Majem, L.; et al. The Mediterranean diet decreases LDL atherogenicity in high cardiovascular risk individuals: A randomized controlled trial. Mol. Nutr. Food Res. 2017, 61, 1601015. [Google Scholar] [CrossRef]
- Kouli, G.M.; Panagiotakos, D.B.; Kyrou, I.; Magriplis, E.; Georgousopoulou, E.N.; Chrysohoou, C.; Tsigos, C.; Tousoulis, D.; Pitsavos, C. Olive oil consumption and 10-year (2002–2012) cardiovascular disease incidence: The ATTICA study. Eur. J. Nutr. 2019, 58, 131–138. [Google Scholar] [CrossRef]
- Carrillo, J.L.M.; Del Campo, J.O.M.; Coronado, O.G.; Gutiérrez, P.T.V.; Cordero, J.F.C.; Juárez, J.V. Adipose tissue and inflammation. In Adipose Tissue, 1st ed.; Szablewski, L., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Finicelli, M.; Squillaro, T.; Di Cristo, F.; Di Salle, A.; Melone, M.A.B.; Galderisi, U.; Peluso, G. Metabolic syndrome, mediterranean diet, and polyphenols: Evidence and perspectives. J. Cell Physiol. 2019, 234, 5807–5826. [Google Scholar] [CrossRef]
- Zhang, H.; Tsaio, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food. Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, N.; Ma, Y.; Wen, D. Hydroxytyrosol improves obesity and insulin resistance by modulating gut microbiota in high-fat diet-induced obese mice. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Castañer, O.; Solà, R.; Motilva, M.J.; Castell, M.; Pérez-Cano, F.J.; Fitó, M. Influence of phenol-enriched olive oils on human intestinal immune function. Nutrients 2016, 8, 213. [Google Scholar] [CrossRef]
- Liehr, M.; Mereu, A.; Pastor, J.J.; Quintela, J.C.; Staats, S.; Rimbach, G.; Ipharraguerre, R. Olive oil bioactives protect pigs against experimentally-induced chronic inflammation independently of alterations in gut microbiota. PLoS ONE 2017, 13, 3. [Google Scholar] [CrossRef]
- Larussa, T.; Oliverio, M.; Suraci, E.; Greco, M.; Placida, R.; Gervasi, S.; Marasco, R.; Imeneo, M.; Paolino, D.; Tucci, L.; et al. Oleuropein decreases cyclooxygenase-2 and interleukin-17 expression and attenuates inflammatory damage in colonic samples from ulcerative colitis patients. Nutrients 2017, 9, 391. [Google Scholar] [CrossRef]
- Carnevale, R.; Pastori, D.; Nocella, C.; Cammisotto, V.; Bartimoccia, S.; Novo, M.; Del Ben, M.; Farcomeni, A.; Angelico, F.; Violi, F. Gut-derived lipopolysaccharides increase post-prandial oxidative stress via Nox2 activation in patients with impaired fasting glucose tolerance: Effect of extra-virgin olive oil. Eur. J. Nutr. 2019, 58, 2. [Google Scholar] [CrossRef]
- Sanchez-Rodriguez, E.; Biel-Glesson, S.; Fernandez-Navarro, J.R.; Calleja, M.A.; Espejo-Calvo, J.A.; Gil-Extremera, B.; de la Torre, R.; Fito, M.; Covas, M.I.; Vilchez, P.; et al. Effects of virgin olive oils differing their bioactive compound contents on biomarkers of oxidative stress and inflammation in healthy adults: A randomized double-blind controlled trial. Nutrients 2019, 11, 561. [Google Scholar] [CrossRef]
- Camargo, A.; Rangel-Zuñiga, O.A.; Haro, C.; Meza-Miranda, E.R.; Peña-Orihuela, P.; Meneses, M.E.; Marin, C.; Yubero-Serrano, E.M.; Perez-Martinez, P.; Delgado-Lista, J.; et al. Olive oil phenolic compounds decrease the postprandial inflammatory responde by reducing postprandial plasma lipopolysaccharide levels. Food Chem. 2014, 162, 161–171. [Google Scholar] [CrossRef]
- Sánchez-Fidalgo, S.; Villegas, I.; Aparicio-Soto, M.; Cárdeno, A.; Rosilo, M.Á.; González-Benjumea, A.; Marset, A.; López, Ó.; Maya, I.; Fernández-Bolãnos, J.G.; et al. Effects of dietary virgin olive oil polyphenols: Hydroxytyrosyl acetate and 3,4-dihydroxyphenylglycol on DSS-induced acute colitis in mice. J. Nutr. Biochem. 2015, 26, 513–520. [Google Scholar] [CrossRef]
- Rial, S.A.; Karelis, A.D.; Bergeron, K.-F.; Mounier, C. Gut microbiota and metabolic health: The potential beneficial effects of a medium chain triglyceride diet in obese individuals. Nutrients 2016, 8, 281. [Google Scholar] [CrossRef]
- Telle-Hansen, V.H.; Holven, K.B.; Ulven, S.M. Impact of a health dietary pattern on gut microbiota and systemic inflammation in humans. Nutrients 2018, 10, 1783. [Google Scholar] [CrossRef]
- Skrypnik, K.; Bogdański, P.; Łoniewski, I.; Reguła, J.; Suliburska, J. Effect of probiot supplementation on liver function and lipid status in rats. Acta Sci. Pol. Technol. Aliment. 2018, 17, 2. [Google Scholar]
- Velasco, P.; Ferreira, A.; Crovesy, L.; Marine, T.; Carmo, M.G.T. Fatty acids, gut microbiota, and the genesis of obesity. In Biochemistry and Health Benefits of Fatty Acids, 1st ed.; Waisundara, V., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Bailey, M.A.; Holscher, H.D. Microbiome-mediated effects of the Mediterranean diet on inflammation. Adv. Nutr. 2018, 9, 193–206. [Google Scholar] [CrossRef]
- Mitsou, E.K.; Kakali, A.; Antonopoulou, S.; Mountzouris, K.C.; Yannakoulia, M.; Panagiotakos, D.B.; Jyriacou, A. Adherence to the Mediterranean diet is associated with the gut microbiota pattern and gastrointestinal characteristics in an adult population. Br. J. Nutr. 2017, 117, 1645–1655. [Google Scholar] [CrossRef]
- Incani, A.; Serra, G.; Atzeri, A.; Melis, M.P.; Serreli, G.; Bandino, G.; Sedda, P.; Campus, M.; Tuberoso, C.I.G.; Deiana, M. Extra virgin olive oil phenolic extracts counteract the pro-oxidant effect of dietary oxidized lipids in human intestinal cells. Food Chem. Toxicol. 2016, 90, 171–180. [Google Scholar] [CrossRef]
| Host | Diet | Effects | References |
|---|---|---|---|
| Metabolic syndrome Adult (n = 102) |
| (3) ↓ abdominal adiposity (4) ↓ LDL-c, TC/HDL-c and LDL-c/HDL-c | [21] |
| Male Wistar rats (n = 64) |
| (3,6,7 and 8)↑ TC and LDL-c | [28] |
| Male Wistar rats (n = 64) |
| (3, 5, 6 and 8)↑ TC and LDL-c ↓ HDL-c when compared with (2) ↑ MUFA when compared with (1) and (4) (4 and 7) Without alteration in TC and LDL-c ↓ HDL-c when compared with (2) ↑ PUFA | [23] |
| Male healthy adult (n = 18) |
| (1) ↓ systolic blood pressure ↓ LDL-c and TV. Negative regulation of the genes ACE and NR1H2 (2) ↑ diastolic blood pressure | [19] |
| Hypercholesterolemic adult (n = 12) |
| (3) ↓ oxidated LDL-c | [29] |
| Adult women with excess body fat (n = 41) |
| (2) ↓ body fat and diastolic blood pressure | [30] |
| Male Swiss Webster mice (n = 26) |
| (3) Control of blood pressure and ↓ triglycerides | [31] |
| Healthy adult (n = 51) |
| (1,2 and 3) ↓ endothelin-1 plasma (1) ↑ fasting plasma triacylglycerol and ↓ systolic blood pressure (2) ↑ fasting plasma triacylglycerol (3) ↑ TC and ↑ systolic blood pressure | [32] |
| Adults with risk of cardiovascular diseases (n = 210) |
| (1) ↑ resistance of LDL-c to oxidize, ↓ changes caused by oxidation of LDL-c and ↑ LDL-c cell size | [27] |
| Host | Diet | Effects | References |
|---|---|---|---|
| Male Wistar albino rats (n = 64) |
| (3, 4, 7 and 8) ↓ E-selectin (3 and 4) ↓ VCAM-1 | [28] |
| Hypercholesterolemic adult (n = 33) |
| (2) ↑ CRP and IgA | [39] |
| Male Wistar rats (n = 64) |
| (2) Higher content of TNF-α when compared with (1) (3) ↓ IL-6 TNF-α when compared with (2) | [23] |
| Female C57BL/6 mice |
| (3,4 and 5) ↑ IL-17A (7 and 8) ↓ IL-6, IFN-γ, TNF-α (7 and 8) ↓ incidence of colonic neoplasias and inhibited the formation of new tumors. | [5] |
| Male piglets with subclinical chronic inflammation (n = 31) |
| (3) Impeded increased of IL-1β and improved the intestinal integrity | [40] |
| Cells of patients with ulcerative colitis (n = 14) |
| (2) ↑ IL-17 (3) ↓ COX-2, IL-17 and infiltration of leukocytes (3) inhibited activation NF-kβ (3) ↓ production of TNF-α and IL-1β | [41] |
| Male C57BL/6J mice (n = 28) |
| (2) ↑ LPS and TLR-4, TNF-α, IL-1β, IL-6 and p-JNK (3,4) ↓ LPS, IL-1β and IL-6 ↓ expression TLR-4, TNF-α, IL-1β, IL-6, p-JNK | [38] |
| Impaired fasting glucose adults (n = 30) |
| (2) Stabilization LPS, ↓ oxidated LDL-c and ↓ Nox2 | [42] |
| Healthy adult (n = 51) |
| (2) ↓ TNF-α and IL-8 | [43] |
| Host | Diet | Effects | References |
|---|---|---|---|
| Hypercholesterolemic adult (n = 12) |
| (3) ↑ Bifidobacterium spp. and ↑ Parascardovia. | [19] |
| Male Swiss Webster mice (n = 26) |
| (3) ↑ Bacteroides (B. fragilis) and ↑ Clostridium ↓ Lactobacillus (L. animalis, L. taiwanensis and Lactococcus). | [31] |
| Male spontaneously hypertensive rats (n = 16) |
| (2) Higher biodiversity of gut bacteria (Clostridios XIVa and Lactobacillus). | [10] |
| Male C57BL/6J mice (n = 28) |
| (1) ↓ Firmicutes/Bacteroides (3 and 4) ↓ Proteobacteria, Deferribacteres and Rikenella (3 and 4) ↑ Lactobacillus johnsonii (4) ↓ Anaeltotruncus sp. G3 | [38] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcelino, G.; Hiane, P.A.; Freitas, K.d.C.; Santana, L.F.; Pott, A.; Donadon, J.R.; Guimarães, R.d.C.A. Effects of Olive Oil and Its Minor Components on Cardiovascular Diseases, Inflammation, and Gut Microbiota. Nutrients 2019, 11, 1826. https://doi.org/10.3390/nu11081826
Marcelino G, Hiane PA, Freitas KdC, Santana LF, Pott A, Donadon JR, Guimarães RdCA. Effects of Olive Oil and Its Minor Components on Cardiovascular Diseases, Inflammation, and Gut Microbiota. Nutrients. 2019; 11(8):1826. https://doi.org/10.3390/nu11081826
Chicago/Turabian StyleMarcelino, Gabriela, Priscila Aiko Hiane, Karine de Cássia Freitas, Lidiani Figueiredo Santana, Arnildo Pott, Juliana Rodrigues Donadon, and Rita de Cássia Avellaneda Guimarães. 2019. "Effects of Olive Oil and Its Minor Components on Cardiovascular Diseases, Inflammation, and Gut Microbiota" Nutrients 11, no. 8: 1826. https://doi.org/10.3390/nu11081826
APA StyleMarcelino, G., Hiane, P. A., Freitas, K. d. C., Santana, L. F., Pott, A., Donadon, J. R., & Guimarães, R. d. C. A. (2019). Effects of Olive Oil and Its Minor Components on Cardiovascular Diseases, Inflammation, and Gut Microbiota. Nutrients, 11(8), 1826. https://doi.org/10.3390/nu11081826





