Influence of Vitamin C on Antioxidant Capacity of In Vitro Perfused Porcine Kidneys
Abstract
1. Introduction
2. Methods
2.1. Animals
2.2. Test Circuits
2.3. Kidney Perfusion and Vitamin C Administration
2.4. Sampling and Measurements
2.5. Statistical Analysis
3. Results
3.1. Perfusion Parameters
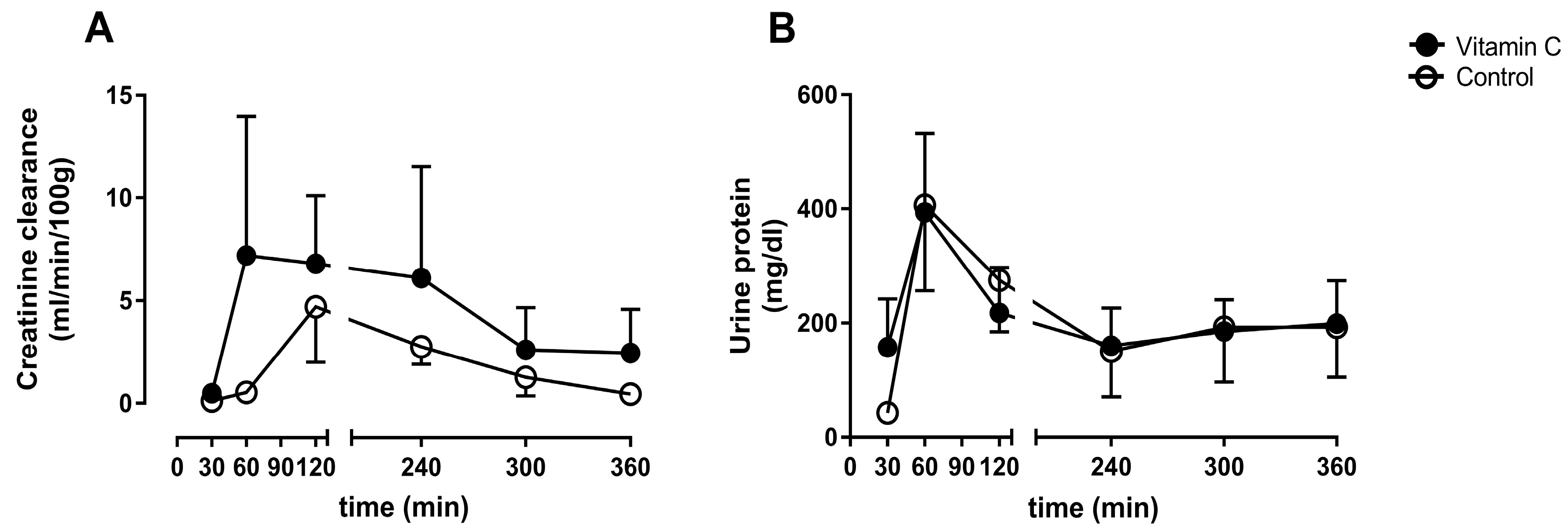
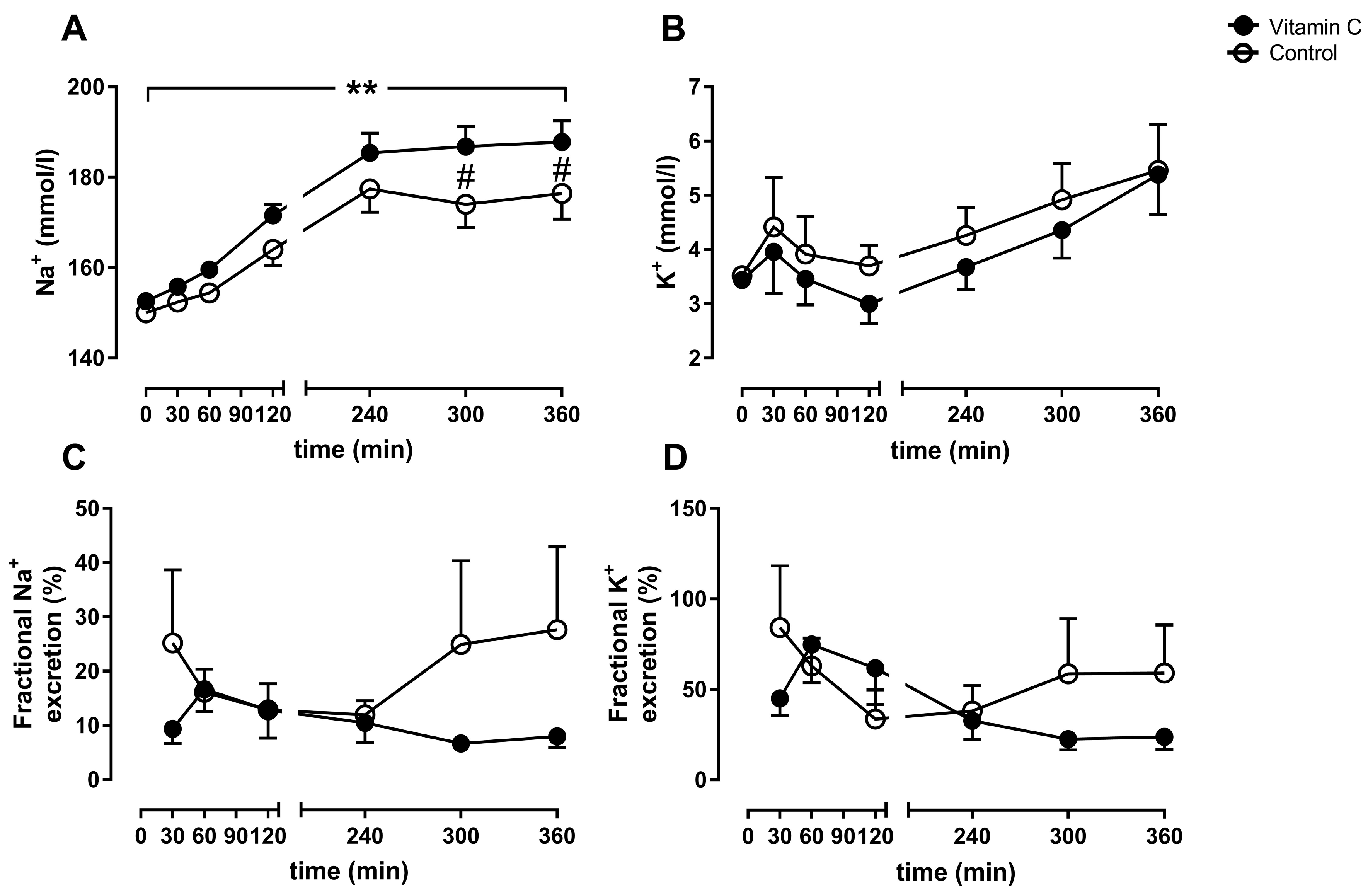
3.2. Kidney Function
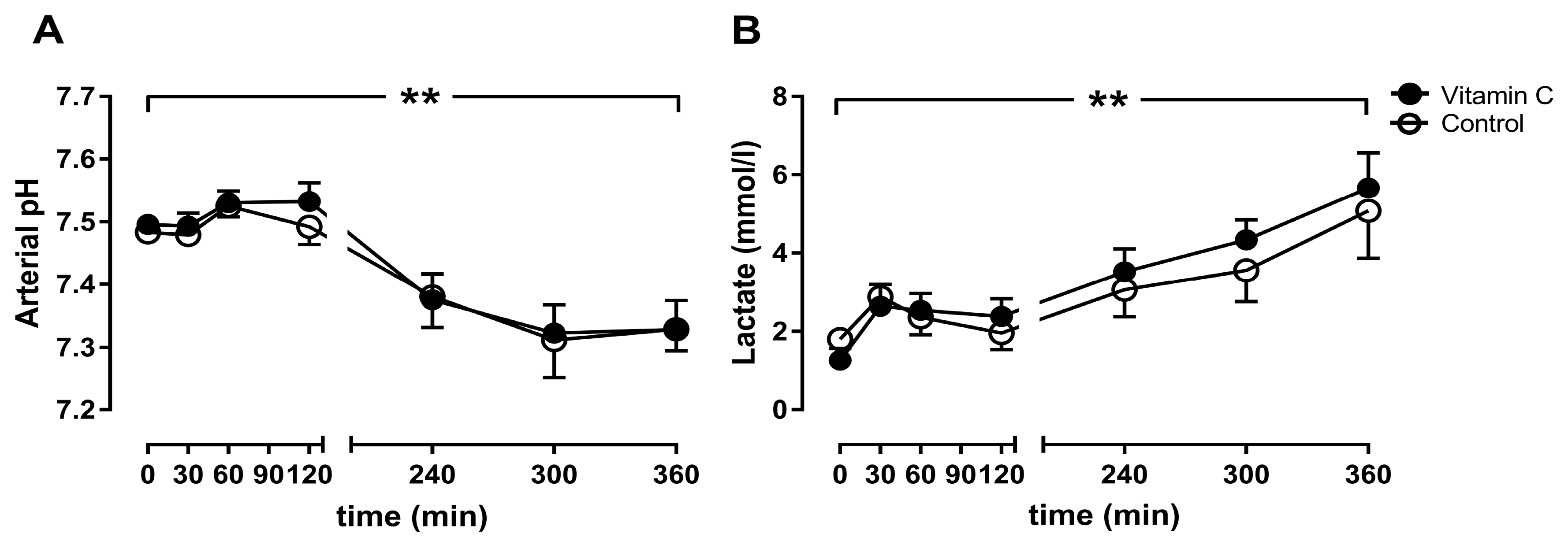
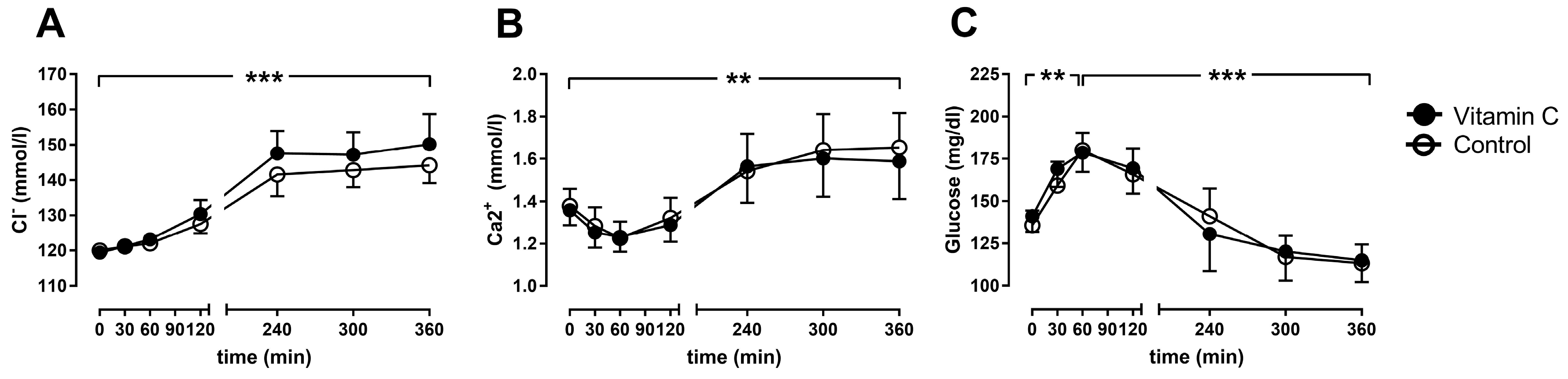
3.3. Blood Gas Analyses
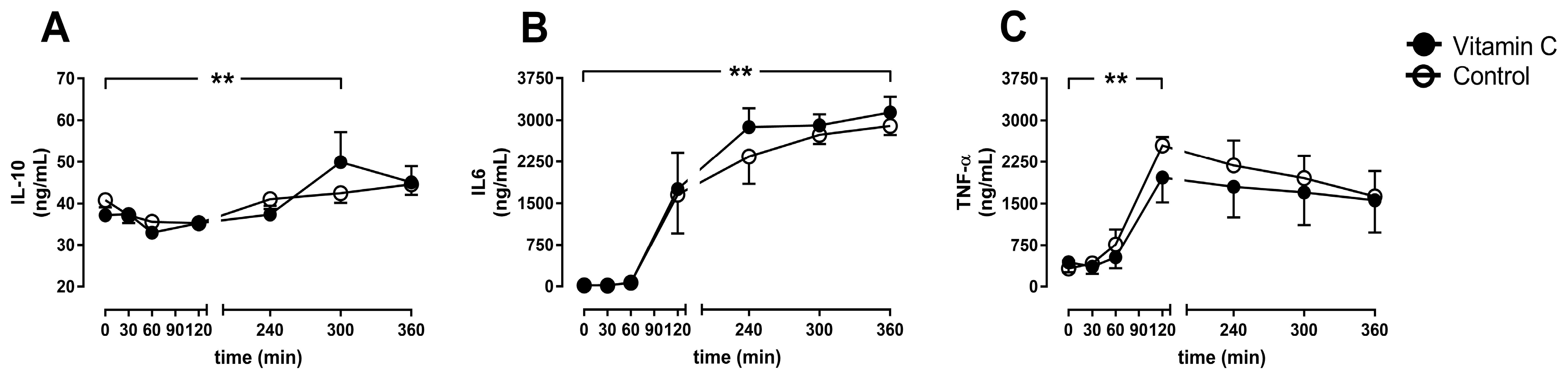
3.4. Inflammation
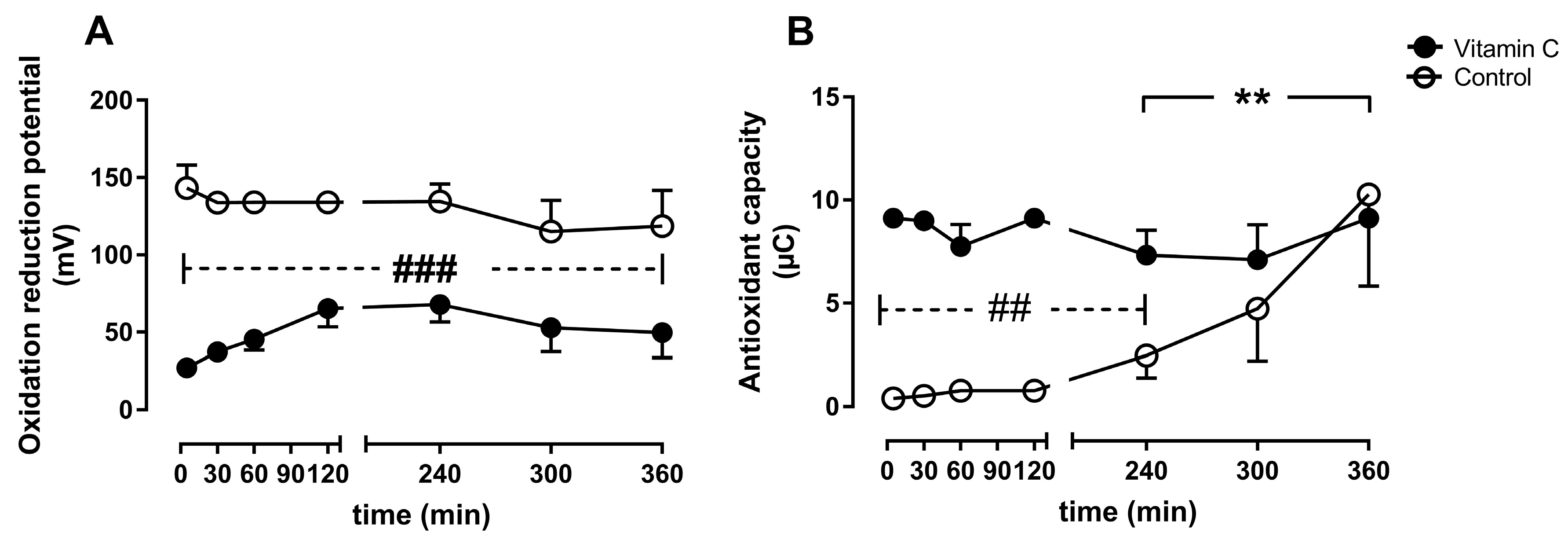
3.5. Oxidative Stress
3.6. Histological Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AC | Antioxidant Capacity |
| BW | Body Weight |
| HB | Hemoglobin |
| IL | Interleukin |
| I/R | Ischemia/Reperfusion |
| NMP | Normothermic Machine Perfusion |
| ORP | Oxidation reduction potential |
| RBC | Red blood Count |
| TNF | Tumor necrosis factor |
References
- Hart, A.; Smith, J.M.; Skeans, M.A.; Gustafson, S.K.; Wilk, A.R.; Robinson, A.; Wainright, J.L.; Haynes, C.R.; Snyder, J.J.; Kasiske, B.L.; et al. OPTN/SRTR 2016 Annual Data Report: Kidney. Am. J. Transplant. 2018, 18, 18–113. [Google Scholar] [CrossRef] [PubMed]
- Balk, R.A. Systemic inflammatory response syndrome (SIRS): Where did it Come from and is it still relevant today? Virulence 2014, 5, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Saat, T.C.; Akker, E.K.V.D.; Ijzermans, J.N.M.; Dor, F.J.M.F.; De Bruin, R.W.F. Improving the outcome of kidney transplantation by ameliorating renal ischemia reperfusion injury: Lost in translation? J. Transl. Med. 2016, 14, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Alam, A.; Soo, A.P.; George, A.J.; Ma, D. Ischemia-Reperfusion Injury Reduces Long Term Renal Graft Survival: Mechanism and Beyond. EBioMedicine 2018, 28, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Salvadori, M.; Rosso, G.; Bertoni, E. Update on ischemia-reperfusion injury in kidney transplantation: Pathogenesis and treatment. World J. Transplant. 2015, 5, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Roy, J.; Galano, J.M.; Durand, T.; Le Guennec, J.Y.; Lee, J.C.Y. Physiological role of reactive oxygen species as promoters of natural defenses. FASEB J. 2017, 31, 3729–3745. [Google Scholar] [CrossRef] [PubMed]
- Jochmans, I.; Nicholson, M.L.; Hosgood, S.A. Kidney perfusion: Some like it hot others prefer to keep it Cool. Curr. Opin. Organ Transplant. 2017, 22, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Hamar, M.; Urbanellis, P.; Kaths, M.J.; Kollmann, D.; Linares, I.; Ganesh, S.; Wiebe, A.; Cen, J.Y.; Yip, P.M.; John, R.; et al. Normothermic Ex Vivo Kidney Perfusion Reduces Warm Ischemic Injury of Porcine Kidney Grafts Retrieved After Circulatory Death. Transplantation 2018, 102, 1262–1270. [Google Scholar] [CrossRef]
- Nicholson, M.L.; Hosgood, S.A. Renal Transplantation after Ex Vivo Normothermic Perfusion: The First Clinical Study. Arab. Archaeol. Epigr. 2013, 13, 1246–1252. [Google Scholar]
- Hosgood, S.A.; Thompson, E.; Moore, T.; Wilson, C.H.; Nicholson, M.L. Normothermic machine perfusion for the assessment and transplantation of declined human kidneys from donation after Circulatory death donors. Br. J. Surg. 2018, 105, 388–394. [Google Scholar] [CrossRef]
- Yong, C.; Hosgood, S.A.; Nicholson, M.L. Ex-vivo normothermic perfusion in renal transplantation: Past, present and future. Curr. Opin. Organ. Transplant. 2016, 21, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Adams, T.D.; Patel, M.; Hosgood, S.A.; Nicholson, M.L. Lowering Perfusate Temperature from 37 degrees C to 32 degrees C Diminishes Function in a Porcine Model of Ex Vivo Kidney Perfusion. Transplant. Direct 2017, 3, e140. [Google Scholar] [CrossRef] [PubMed]
- Hosgood, S.A.; Moore, T.; Kleverlaan, T.; Adams, T.; Nicholson, M.L. Haemoadsorption reduces the inflammatory response and improves blood flow during ex vivo renal perfusion in an experimental model. J. Transl. Med. 2017, 15, 216. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Shaw, G.M.; Fowler, A.A.; Natarajan, R. Ascorbate-dependent vasopressor synthesis: A rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care 2015, 19, 418. [Google Scholar] [CrossRef] [PubMed]
- Oudemans-van Straaten, H.M.; Spoelstra-de Man, A.M.; de Waard, M.C. Vitamin C revisited. Crit. Care 2014, 18, 460. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.; Wendt, S.; Benstoem, C.; Neubauer, C.; Meybohm, P.; Langlois, P.; Adhikari, N.K.; Heyland, D.K.; Stoppe, C. Vitamin C to Improve Organ Dysfunction in Cardiac Surgery Patients—Review and Pragmatic Approach. Nutrients 2018, 10, 974. [Google Scholar] [CrossRef]
- Esteban, M.A.; Wang, T.; Qin, B.; Yang, J.; Qin, D.; Cai, J.; Li, W.; Weng, Z.; Chen, J.; Ni, S.; et al. Vitamin C Enhances the Generation of Mouse and Human Induced Pluripotent Stem Cells. Cell Stem Cell 2010, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Syed, A.A.; Knowlson, S.; Sculthorpe, R.; Farthing, D.; DeWilde, C.; Farthing, C.A.; Larus, T.L.; Martin, E.; Brophy, D.F.; Gupta, S.; et al. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J. Transl. Med. 2014, 12, 32. [Google Scholar]
- Han, M.; Pendem, S.; Teh, S.L.; Sukumaran, D.K.; Wu, F.; Wilson, J.X. Ascorbate protects endothelial barrier function during septic insult: Role of protein phosphatase type 2A. Free Radic. Biol. Med. 2010, 48, 128–135. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Frei, B.; Stocker, R.; England, L.; Ames, B.N. Ascorbate: The Most Effective Antioxidant in Human Blood Plasma. Results Probl. Cell Differ. 1990, 264, 155–163. [Google Scholar]
- Man, A.M.E.S.D.; Elbers, P.W.G.; Straaten, H.M.O. Making Sense of Early High-dose Intravenous Vitamin C in Ischemia/Reperfusion Injury. Crit. Care 2018, 22, 70. [Google Scholar]
- Flecknell, P. Replacement, reduction and refinement. ALTEX 2002, 19, 73–78. [Google Scholar]
- Levine, M.; Rumsey, S.C.; Daruwala, R.; Park, J.B.; Wang, Y. Criteria and Recommendations for Vitamin C Intake. JAMA 1999, 281, 1415–1423. [Google Scholar] [CrossRef]
- Tanaka, H.; Matsuda, T.; Miyagantani, Y.; Yukioka, T.; Matsuda, H.; Shimazaki, S. Reduction of resuscitation fluid volumes in severely burned patients using ascorbic acid administration: A randomized, prospective study. Arch. Surg. 2000, 135, 326–331. [Google Scholar] [CrossRef]
- Dingchao, H.; Zhiduan, Q.; Liye, H.; Xiadong, F. The Protective Effects of High-Dose Ascorbic Acid on Myocardium against Reperfusion Injury during and after Cardiopulmonary Bypass. Thorac. Cardiovasc. Surg. 1994, 42, 276–278. [Google Scholar] [CrossRef]
- Stoppe, C.; Averdunk, L.; Goetzenich, A.; Soppert, J.; Marlier, A.; Kraemer, S.; Vieten, J.; Coburn, M.; Kowark, A.; Kim, B.S.; et al. The protective role of macrophage migration inhibitory factor in acute kidney injury after cardiac surgery. Sci. Transl. Med. 2018, 10, eaan4886. [Google Scholar] [CrossRef]
- Dennis, J.M.; Witting, P.K. Protective Role for Antioxidants in Acute Kidney Disease. Nutrients 2017, 9, 718. [Google Scholar] [CrossRef]
- Johnston, L.; Laverty, G. Vitamin C transport and SVCT1 transporter expression in chick renal proximal tubule cells in culture. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2007, 146, 327–334. [Google Scholar] [CrossRef]
- Corpe, C.P.; Tu, H.; Eck, P.; Wang, J.; Faulhaber-Walter, R.; Schnermann, J.; Margolis, S.; Padayatty, S.; Sun, H.; Wang, Y.; et al. Vitamin C transporter Slc23a1 links renal reabsorption, vitamin C tissue accumulation, and perinatal survival in mice. J. Clin. Investig. 2010, 120, 1069–1083. [Google Scholar] [CrossRef]
- Legendre, C.; Canaud, G.; Martinez, F. Factors influencing long-term outcome after kidney transplantation. Transpl. Int. 2014, 27, 19–27. [Google Scholar] [CrossRef]
- Leonard, S.W.; Bobe, G.; Traber, M.G. Stability of antioxidant vitamins in whole human blood during overnight storage at 4 degrees C and frozen storage up to 6 months. Int. J. Vitam. Nutr. Res. 2018, 88, 151–157. [Google Scholar] [CrossRef]
- Radosinska, J.; Jasenovec, T.; Puzserova, A.; Krajcir, J.; Lacekova, J.; Kucerova, K.; Kalnovicova, T.; Tothova, L.; Kovacicova, I.; Vrbjar, N. Promotion of whole blood rheology after vitamin C supplementation: Focus on red blood cells. Can. J. Physiol. Pharmacol. 2019, 13, 1–7. [Google Scholar] [CrossRef]
- Wu, S.; Wu, G.; Wu, H. Hemolytic jaundice induced by pharmacological dose ascorbic acid in glucose-6-phosphate dehydrogenase deficiency A Case report. Medicine (Baltimore) 2018, 97, e13588. [Google Scholar] [CrossRef]
- Sadat, U.; Usman, A.; Gillard, J.H.; Boyle, J.R. Does ascorbic acid protect against Contrast-induced acute kidney injury in patients undergoing Coronary angiography: A systematic review with meta-analysis of randomized, Controlled trials. J. Am. Coll. Cardiol. 2013, 62, 2167–2175. [Google Scholar] [CrossRef]
- Gurm, H.; Sheta, M.A.; Nivera, N.; Tunkel, A. Vitamin C-induced oxalate nephropathy: A case report. J. Community Hosp. Intern. Med. Perspect. 2012, 2, 17718. [Google Scholar] [CrossRef]
- Sunkara, V.; Pelkowski, T.D.; Dreyfus, D.; Satoskar, A. Acute Kidney Disease Due to Excessive Vitamin C Ingestion and Remote Roux-en-Y Gastric Bypass Surgery Superimposed on CKD. Am. J. Kidney Dis. 2015, 66, 721–724. [Google Scholar] [CrossRef]
- Beers, R.J.; Lentz, C.W.; Kahn, S.A. Resuscitation after Severe Burn Injury Using High-Dose Ascorbic Acid: A Retrospective Review. J. Burn. Care Res. 2011, 32, 110–117. [Google Scholar]
- Lin, J.; Falwell, S.; Greenhalgh, D.; Palmieri, T.; Sen, S. High-Dose Ascorbic Acid for Burn Shock Resuscitation May Not Improve Outcomes. J. Burn Care Res. 2018, 39, 708–712. [Google Scholar] [CrossRef]
- Nathens, A.B.; Neff, M.J.; Jurkovich, G.J.; Klotz, P.; Farver, K.; Ruzinski, J.T.; Radella, F.; Garcia, I.; Maier, R.V. Randomized, Prospective Trial of Antioxidant Supplementation in Critically Ill Surgical Patients. Ann. Surg. 2002, 236, 814–822. [Google Scholar] [CrossRef]
- Antonic, M. Effect of Ascorbic Acid on Postoperative Acute Kidney Injury in Coronary Artery Bypass Graft Patients: A Pilot Study. Hear. Surg. Forum 2017, 20, E214–E218. [Google Scholar] [CrossRef]
- Bucala, R. Lipid and lipoprotein oxidation: Basic mechanisms and unresolved questions in vivo. Redox Rep. 1996, 2, 291–307. [Google Scholar] [CrossRef]
- Halliwell, B. Vitamin C: Antioxidant or pro-oxidant in vivo? Free Radic. Res. 1996, 25, 439–454. [Google Scholar] [CrossRef]
- Collins, A.; Cadet, J.; Epe, B.; Gedik, C. Problems in the measurement of 8-oxoguanine in human DNA. Report of a workshop, DNA oxidation, held in Aberdeen, UK, 19–21 January, 1997. Carcinogenesis 1997, 18, 1833–1836. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bleilevens, C.; Doorschodt, B.M.; Fechter, T.; Grzanna, T.; Theißen, A.; Liehn, E.A.; Breuer, T.; Tolba, R.H.; Rossaint, R.; Stoppe, C.; et al. Influence of Vitamin C on Antioxidant Capacity of In Vitro Perfused Porcine Kidneys. Nutrients 2019, 11, 1774. https://doi.org/10.3390/nu11081774
Bleilevens C, Doorschodt BM, Fechter T, Grzanna T, Theißen A, Liehn EA, Breuer T, Tolba RH, Rossaint R, Stoppe C, et al. Influence of Vitamin C on Antioxidant Capacity of In Vitro Perfused Porcine Kidneys. Nutrients. 2019; 11(8):1774. https://doi.org/10.3390/nu11081774
Chicago/Turabian StyleBleilevens, Christian, Benedict M. Doorschodt, Tamara Fechter, Tim Grzanna, Alexander Theißen, Elisa A. Liehn, Thomas Breuer, René H. Tolba, Rolf Rossaint, Christian Stoppe, and et al. 2019. "Influence of Vitamin C on Antioxidant Capacity of In Vitro Perfused Porcine Kidneys" Nutrients 11, no. 8: 1774. https://doi.org/10.3390/nu11081774
APA StyleBleilevens, C., Doorschodt, B. M., Fechter, T., Grzanna, T., Theißen, A., Liehn, E. A., Breuer, T., Tolba, R. H., Rossaint, R., Stoppe, C., Boor, P., Hill, A., & Fabry, G. (2019). Influence of Vitamin C on Antioxidant Capacity of In Vitro Perfused Porcine Kidneys. Nutrients, 11(8), 1774. https://doi.org/10.3390/nu11081774





