Daily Intake of Fermented Milk Containing Lactobacillus casei Shirota (Lcs) Modulates Systemic and Upper Airways Immune/Inflammatory Responses in Marathon Runners
Abstract
1. Introduction
2. Material and Methods
2.1. Subjects and Study Design
2.2. Schedule of Fermented Milk Ingestion
2.3. Collection of the Samples
2.4. Determination of the Salivary Concentration of Immunoglobulin A (SIgA) and Antibacterial Peptides
2.5. Determination of Cytokine Concentrations in Nasal Mucosa Lavage
2.6. Statistical Analysis
3. Results
3.1. Effects of LcS on the Incidence and Duration of Upper Respiratory Symptoms (URS)
3.2. LcS Maintains Secretory IgA (SIgA) and Antimicrobial Peptides Salivary Levels after the Marathon
3.3. LcS Reduces Neutrophils Infiltration in Nasal Mucosa
3.4. LcS Modulates the Pro and Anti-inflammatory Cytokines Response in Upper Airways
3.5. LcS Modulates the Pro- and Anti-Inflammatory Cytokines Response in Systemic Circulation
3.6. LcS Increases the Anti-inflammatory Ratio in Upper Airways
4. Discussion
5. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nieman, D.C. Is infection risk linked to exercise workload? Med. Sci. Sports Exerc. 2000, 32, S406–S411. [Google Scholar] [CrossRef] [PubMed]
- Bermon, S. Airway inflammation and upper respiratory tract infection in athletes: Is there a link? Exerc. Immunol. Rev. 2007, 13, 6–14. [Google Scholar] [PubMed]
- Walsh, N.P.; Gleeson, M.; Shephard, R.J.; Gleeson, M.; Woods, J.A.; Bishop, N.C.; Fleshner, M.; Green, C.; Pedersen, B.K.; Hoffman-Goetz, L.; et al. Position statement. Part one: Immune function and exercise. Exerc. Immunol. Rev. 2011, 17, 6–63. [Google Scholar] [PubMed]
- Vaisberg, M.; Suguri, V.M.; Gregorio, L.C.; Lopes, J.D.; Bachi, A.L. Cytokine kinetics in nasal mucosa and sera: new insights in understanding upper-airway disease of marathon runners. Exerc. Immunol. Rev. 2013, 19, 49–59. [Google Scholar] [PubMed]
- Baken, K.A.; Ezendam, J.; Gremmer, E.R.; de Klerk, A.; Pennings, J.L.; Matthee, B.; Peijnenburg, A.A.; van Loveren, H. Evaluation of immunomodulation by Lactobacillus casei Shirota: immune function, autoimmunity and gene expression. Int. J. Food Microbiol. 2006, 112, 8–18. [Google Scholar] [CrossRef]
- Shida, K.; Nanno, M.; Nagata, S. Flexible cytokine production by macrophages and T cells in response to probiotic bacteria: a possible mechanism by which probiotics exert multifunctional immune regulatory activities. Gut Microbes 2011, 2, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Kekkonen, R.A.; Vasankari, T.J.; Vuorimaa, T.; Haahtela, T.; Julkunen, I.; Korpela, R. The effect of probiotics on respiratory infections and gastrointestinal symptoms during training in marathon runners. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 352–363. [Google Scholar] [CrossRef]
- Gleeson, M.; Bishop, N.C.; Oliveira, M.; Tauler, P. Daily probiotic’s (Lactobacillus casei Shirota) reduction of infection incidence in athletes. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 55–64. [Google Scholar] [CrossRef]
- Harriss, D.J.; Atkinson, G. Ethical Standards in Sport and Exercise Science Research: 2016 Update. Int. J. Sports Med. 2015, 36, 1121–1124. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Akerstrom, T.C.; Nielsen, A.R.; Fischer, C.P. Role of myokines in exercise and metabolism. J. Appl. Physiol. 2007, 103, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Beal, A.L.; Cerra, F.B. Multiple organ failure syndrome in the 1990s. Systemic inflammatory response and organ dysfunction. JAMA 1994, 271, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Boisson, C.; Haccoun, M.; Thomachot, L.; Mege, J.L. Patterns of cytokine evolution (tumor necrosis factor-alpha and interleukin-6) after septic shock, hemorrhagic shock, and severe trauma. Crit. Care Med. 1997, 25, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- Steensberg, A.; Keller, C.; Starkie, R.L.; Osada, T.; Febbraio, M.A.; Pedersen, B.K. IL-6 and TNF-alpha expression in, and release from, contracting human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E1272–E1278. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [PubMed]
- Steensberg, A.; Fischer, C.P.; Keller, C.; Moller, K.; Pedersen, B.K. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am. J. Physiology Endocrinol. Metab. 2003, 285, E433–E437. [Google Scholar] [CrossRef] [PubMed]
- Sugama, K.; Suzuki, K.; Yoshitani, K.; Shiraishi, K.; Kometani, T. Urinary excretion of cytokines versus their plasma levels after endurance exercise. Exerc. Immunol. Rev. 2013, 19, 29–48. [Google Scholar] [PubMed]
- Jee, H.; Jin, Y. Effects of prolonged endurance exercise on vascular endothelial and inflammation markers. J. Sports Sci. Med. 2012, 11, 719–726. [Google Scholar]
- Harbige, L.S.; Pinto, E.; Allgrove, J.; Thomas, L.V. Immune Response of Healthy Adults to the Ingested Probiotic Lactobacillus casei Shirota. Scand. J. Immunol. 2016, 84, 353–364. [Google Scholar] [CrossRef]
- Nieman, D.C.; Henson, D.A.; Smith, L.L.; Utter, A.C.; Vinci, D.M.; Davis, J.M.; Kaminsky, D.E.; Shute, M. Cytokine changes after a marathon race. J. Appl. Physiol. 2001, 91, 109–114. [Google Scholar] [CrossRef]
- Abedelmalek, S.; Souissi, N.; Takayuki, A.; Hadouk, S.; Tabka, Z. Effect of Acute Maximal Exercise on Circulating Levels of Interleukin-12 during Ramadan Fasting. Asian J. Sports Med. 2011, 2, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, M.; Matikainen, S.; Vuopio-Varkila, J.; Pirhonen, J.; Varkila, K.; Kurimoto, M.; Julkunen, I. Lactobacilli and streptococci induce interleukin-12 (IL-12), IL-18, and gamma interferon production in human peripheral blood mononuclear cells. Infect. Immun. 1998, 66, 6058–6062. [Google Scholar] [PubMed]
- Takeda, K.; Suzuki, T.; Shimada, S.I.; Shida, K.; Nanno, M.; Okumura, K. Interleukin-12 is involved in the enhancement of human natural killer cell activity by Lactobacillus casei Shirota. Clin. Exp. Immunol. 2006, 146, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Jayne, J.G.; Bensman, T.J.; Schaal, J.B.; Park, A.Y.J.; Kimura, E.; Tran, D.; Selsted, M.E.; Beringer, P.M. Rhesus theta-Defensin-1 Attenuates Endotoxin-induced Acute Lung Injury by Inhibiting Proinflammatory Cytokines and Neutrophil Recruitment. Am. J. Respir. Cell Mol. Biol. 2018, 58, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Wei-Xu, H.; Wen-Yun, Z.; Xi-Ling, Z.; Zhu, W.; Li-Hua, W.; Xiao-Mu, W.; Hui-Ping, W.; Wen-Ding, W.; Dan, H.; Qin, X.; et al. Anti-Interleukin-1 Beta/Tumor Necrosis Factor-Alpha IgY Antibodies Reduce Pathological Allergic Responses in Guinea Pigs with Allergic Rhinitis. Mediat. Inflamm. 2016, 2016, 3128182. [Google Scholar] [CrossRef] [PubMed]
- Bedard, M.; McClure, C.D.; Schiller, N.L.; Francoeur, C.; Cantin, A.; Denis, M. Release of interleukin-8, interleukin-6, and colony-stimulating factors by upper airway epithelial cells: implications for cystic fibrosis. Am. J. Respir. Cell Mol. Biol. 1993, 9, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Luna, L.A., Jr.; Bachi, A.L.; Novaes e Brito, R.R.; Eid, R.G.; Suguri, V.M.; Oliveira, P.W.; Gregorio, L.C.; Vaisberg, M. Immune responses induced by Pelargonium sidoides extract in serum and nasal mucosa of athletes after exhaustive exercise: modulation of secretory IgA, IL-6 and IL-15. Phytomed. Int. J. Phytother. Phytopharm. 2011, 18, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, M., Jr.; London, N.R., Jr.; Tharakan, A.; Surya, N.; Sussan, T.E.; Rao, X.; Lin, S.Y.; Toskala, E.; Rajagopalan, S.; Biswal, S. Airborne Particulate Matter Induces Nonallergic Eosinophilic Sinonasal Inflammation in Mice. Am. J. Respir. Cell Mol. Biol. 2017, 57, 59–65. [Google Scholar] [CrossRef]
- Fireman, P. Understanding asthma pathophysiology. Allergy Asthma Proc. 2003, 24, 79–83. [Google Scholar]
- Nakagome, K.; Imamura, M.; Kawahata, K.; Harada, H.; Okunishi, K.; Matsumoto, T.; Sasaki, O.; Tanaka, R.; Kano, M.R.; Chang, H.; et al. High expression of IL-22 suppresses antigen-induced immune responses and eosinophilic airway inflammation via an IL-10-associated mechanism. J. Immunol. 2011, 187, 5077–5089. [Google Scholar] [CrossRef]
- Shida, K.; Suzuki, T.; Kiyoshima-Shibata, J.; Shimada, S.; Nanno, M. Essential roles of monocytes in stimulating human peripheral blood mononuclear cells with Lactobacillus casei to produce cytokines and augment natural killer cell activity. Clin. Vaccine Immunol. 2006, 13, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, D.; Dlugosz, E.; Zawistowska-Deniziak, A. Functional Properties of Food Origin Lactobacillus in the Gastrointestinal Ecosystem-In Vitro Study. Probiotics Antimicrob. Proteins 2018. [Google Scholar] [CrossRef] [PubMed]
- De Marco, S.; Sichetti, M.; Muradyan, D.; Piccioni, M.; Traina, G.; Pagiotti, R.; Pietrella, D. Probiotic Cell-Free Supernatants Exhibited Anti-Inflammatory and Antioxidant Activity on Human Gut Epithelial Cells and Macrophages Stimulated with LPS. Evid. Based Complement. Altern. Med. ECAM 2018, 2018, 1756308. [Google Scholar] [CrossRef] [PubMed]
- Van Hemert, S.; Meijerink, M.; Molenaar, D.; Bron, P.A.; de Vos, P.; Kleerebezem, M.; Wells, J.M.; Marco, M.L. Identification of Lactobacillus plantarum genes modulating the cytokine response of human peripheral blood mononuclear cells. BMC Microbiol. 2010, 10, 293. [Google Scholar] [CrossRef] [PubMed]
- Izadpanah, A.; Gallo, R.L. Antimicrobial peptides. J. Am. Acad. Dermatol. 2005, 52, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Cox, G.; Pyne, D.; Kyd, J.; McDonald, W.; Fricker, P.; Cripps, A. Salivary lactoferrin as a marker of immunocompetence in elite swimmers. Int. J. Sports Med. 1999, 21, S83–S84. [Google Scholar]
- Gill, S.K.; Teixeira, A.M.; Rosado, F.; Hankey, J.; Wright, A.; Marczak, S.; Murray, A.; Costa, R.J. The impact of a 24-h ultra-marathon on salivary antimicrobial protein responses. Int. J. Sports Med. 2014, 35, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Sakai, M.; Kaida, Y.; Kaibara, K. Blood lactoferrin release induced by running exercise in normal volunteers: antibacterial activity. Clin. Chim. Acta Int. J. Clin. Chem. 2004, 341, 165–172. [Google Scholar] [CrossRef] [PubMed]


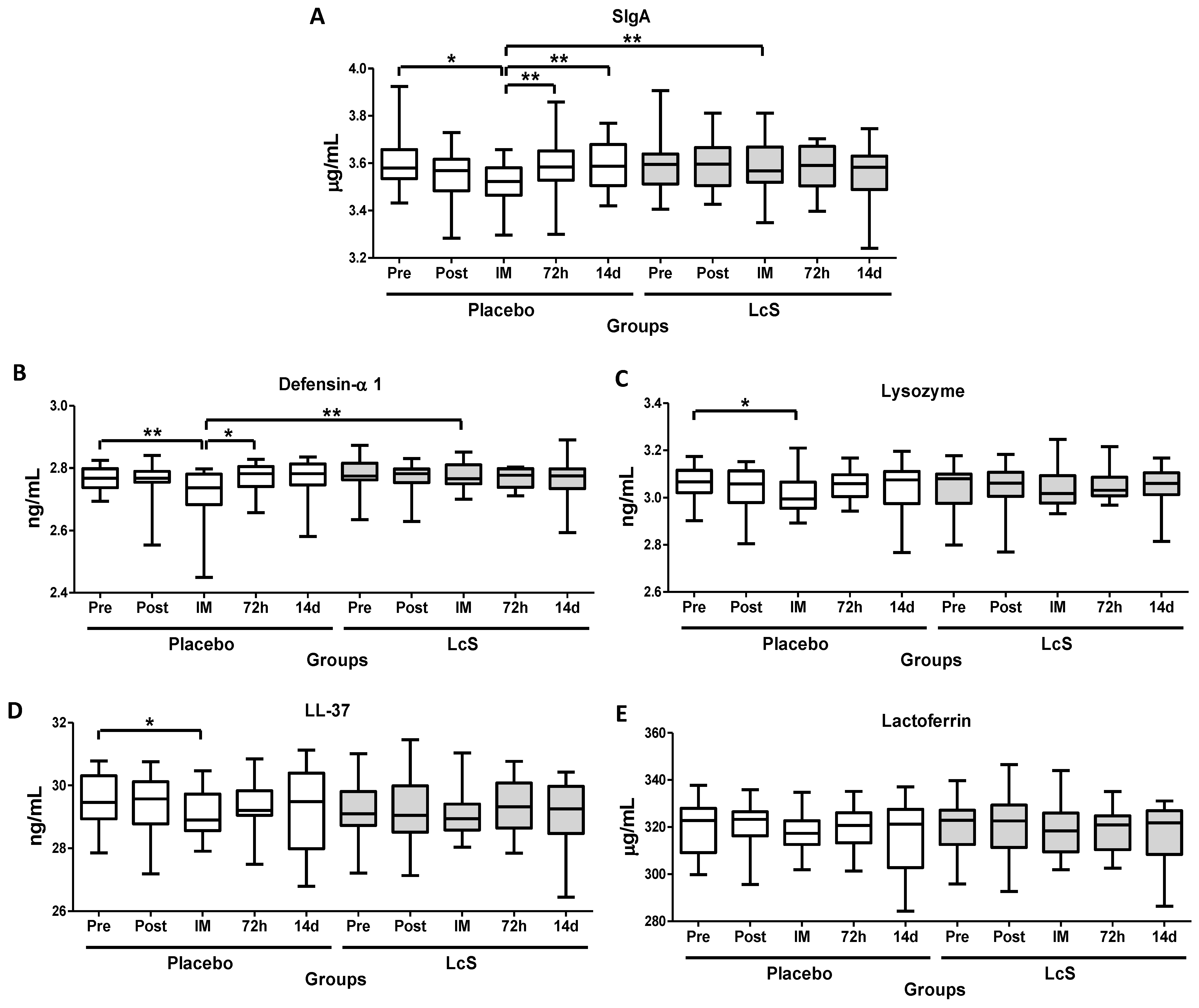

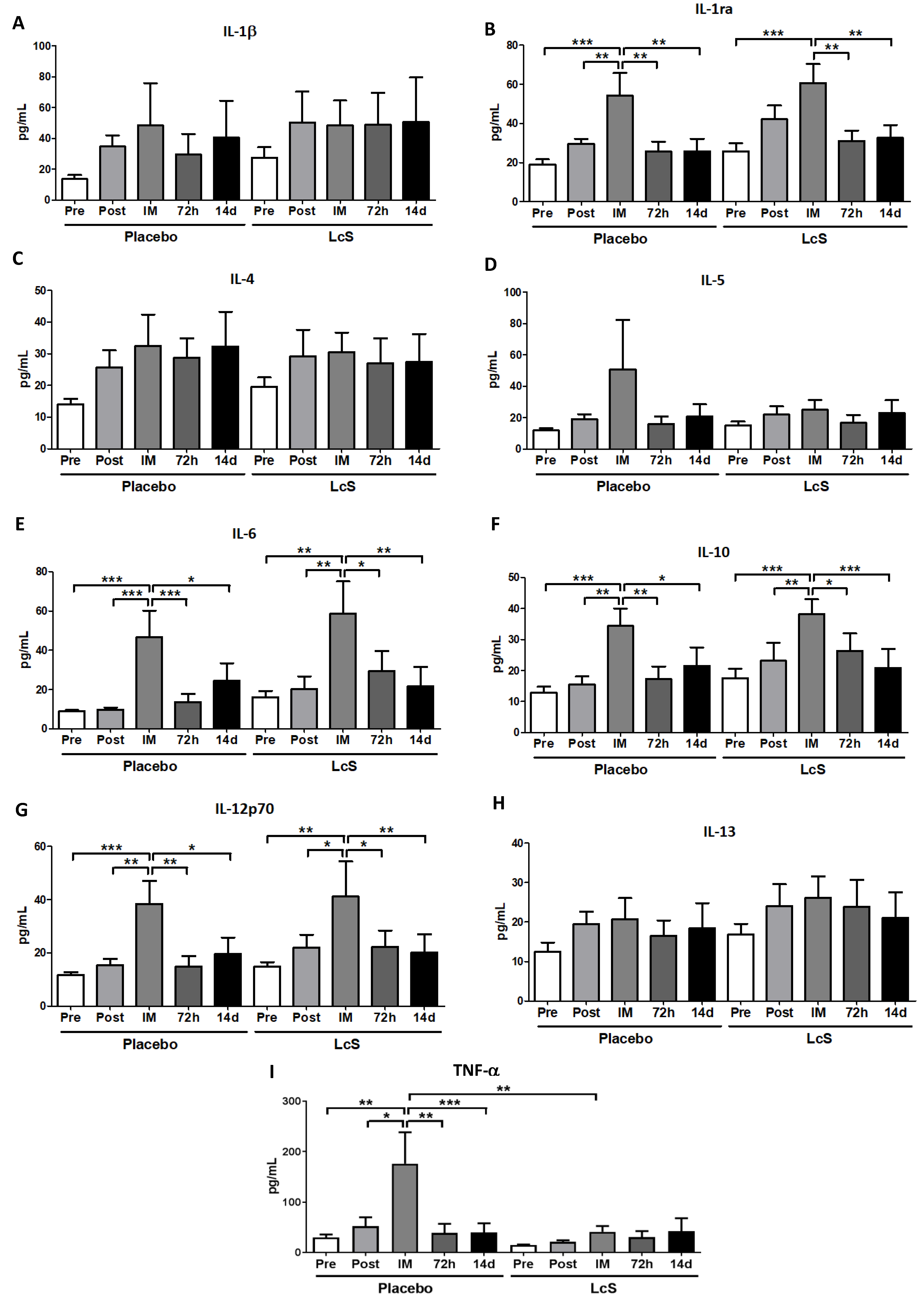
| Variables | Volunteers (n = 42) | |
|---|---|---|
| Placebo (n = 22) | LcS (n = 20) | |
| Age (year) | 40.1 ± 10.3 | 39.6 ± 8.8 |
| Weight (kg) | 76.5 ± 10.4 | 72.4 ± 7.8 |
| Height (cm) | 176.9 ± 7.6 | 173.8 ± 6.4 |
| Total body fat (%) | 18.6 ± 7.5 | 16.5 ± 6.6 |
| Total muscle mass (g) | 58.1 ± 4.7 | 54.7 ± 5.9 |
| BMI (kg/m2) | 24.4 ± 2.2 | 23.4 ± 2.4 |
| Relative VO2max (ml/kg/min) | 57.64 ± 6.89 | 57.86 ± 6.85 |
| Time of conclusion of the race (hours) | 04:09:20 ± 00:19:56 | 04:15:30 ± 00:15:22 |
| Variable | Volunteers (n = 42) | p Value | ||||
|---|---|---|---|---|---|---|
| Placebo (n = 22) | ||||||
| Pre | Post | IM | 72 h | 14 d | ||
| Neutrophil (%) | 50 ± 3 | 43 ± 6 * | 38 ± 3 *,# | 53 ± 4 * | 56 ± 3 | <0.05 |
| LcS (n = 20) | ||||||
| 45 ± 4 ø | 21 ± 3 | 28 ± 2 | 29 ± 3 | 48 ± 6 ø | <0.05 | |
| Variable/Occasion | IL-10/IL-12p70 ratio | |||||
|---|---|---|---|---|---|---|
| Group/Sample | Pre | Post | IM | 72 h | 14 d | |
| Placebo/Serum | 1.05 ± 0.10 | 1.12 ± 0.16 | 1.63 ± 0.43 | 1.28 ± 0.19 | 1.16 ± 0.11 | |
| LcS/Serum | 1.09 ± 0.11 | 1.07 ± 0.13 | 1.40 ± 0.15 | 1.30 ± 0.13 | 1.11 ± 0.09 | |
| Placebo/Nasal mucosa lavage | 1.20 ± 0.47 | 0.83 ± 0.10 | 1.12 ± 0.13 | 0.84 ± 0.11 | 1.00 ± 0.11 | |
| LcS/Nasal mucosa lavage | 0.81 ± 0.10 | 0.94 ± 0.13 | 5.84 ± 2.74 *,# | 0.95 ± 0.12 | 0.76 ± 0.10 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vaisberg, M.; Paixão, V.; Almeida, E.B.; Santos, J.M.B.; Foster, R.; Rossi, M.; Pithon-Curi, T.C.; Gorjão, R.; Momesso, C.M.; Andrade, M.S.; et al. Daily Intake of Fermented Milk Containing Lactobacillus casei Shirota (Lcs) Modulates Systemic and Upper Airways Immune/Inflammatory Responses in Marathon Runners. Nutrients 2019, 11, 1678. https://doi.org/10.3390/nu11071678
Vaisberg M, Paixão V, Almeida EB, Santos JMB, Foster R, Rossi M, Pithon-Curi TC, Gorjão R, Momesso CM, Andrade MS, et al. Daily Intake of Fermented Milk Containing Lactobacillus casei Shirota (Lcs) Modulates Systemic and Upper Airways Immune/Inflammatory Responses in Marathon Runners. Nutrients. 2019; 11(7):1678. https://doi.org/10.3390/nu11071678
Chicago/Turabian StyleVaisberg, Mauro, Vitoria Paixão, Ewin B. Almeida, Juliana M. B. Santos, Roberta Foster, Marcelo Rossi, Tania C. Pithon-Curi, Renata Gorjão, Cesar M. Momesso, Marília S. Andrade, and et al. 2019. "Daily Intake of Fermented Milk Containing Lactobacillus casei Shirota (Lcs) Modulates Systemic and Upper Airways Immune/Inflammatory Responses in Marathon Runners" Nutrients 11, no. 7: 1678. https://doi.org/10.3390/nu11071678
APA StyleVaisberg, M., Paixão, V., Almeida, E. B., Santos, J. M. B., Foster, R., Rossi, M., Pithon-Curi, T. C., Gorjão, R., Momesso, C. M., Andrade, M. S., Araujo, J. R., Garcia, M. C., Cohen, M., Perez, E. C., Santos-Dias, A., Vieira, R. P., & Bachi, A. L. L. (2019). Daily Intake of Fermented Milk Containing Lactobacillus casei Shirota (Lcs) Modulates Systemic and Upper Airways Immune/Inflammatory Responses in Marathon Runners. Nutrients, 11(7), 1678. https://doi.org/10.3390/nu11071678





