Nutrition and Breast Cancer: A Literature Review on Prevention, Treatment and Recurrence
Abstract
1. Introduction
2. Selection of Studies
3. Dietary Factors in Breast Cancer Incidence and Recurrence
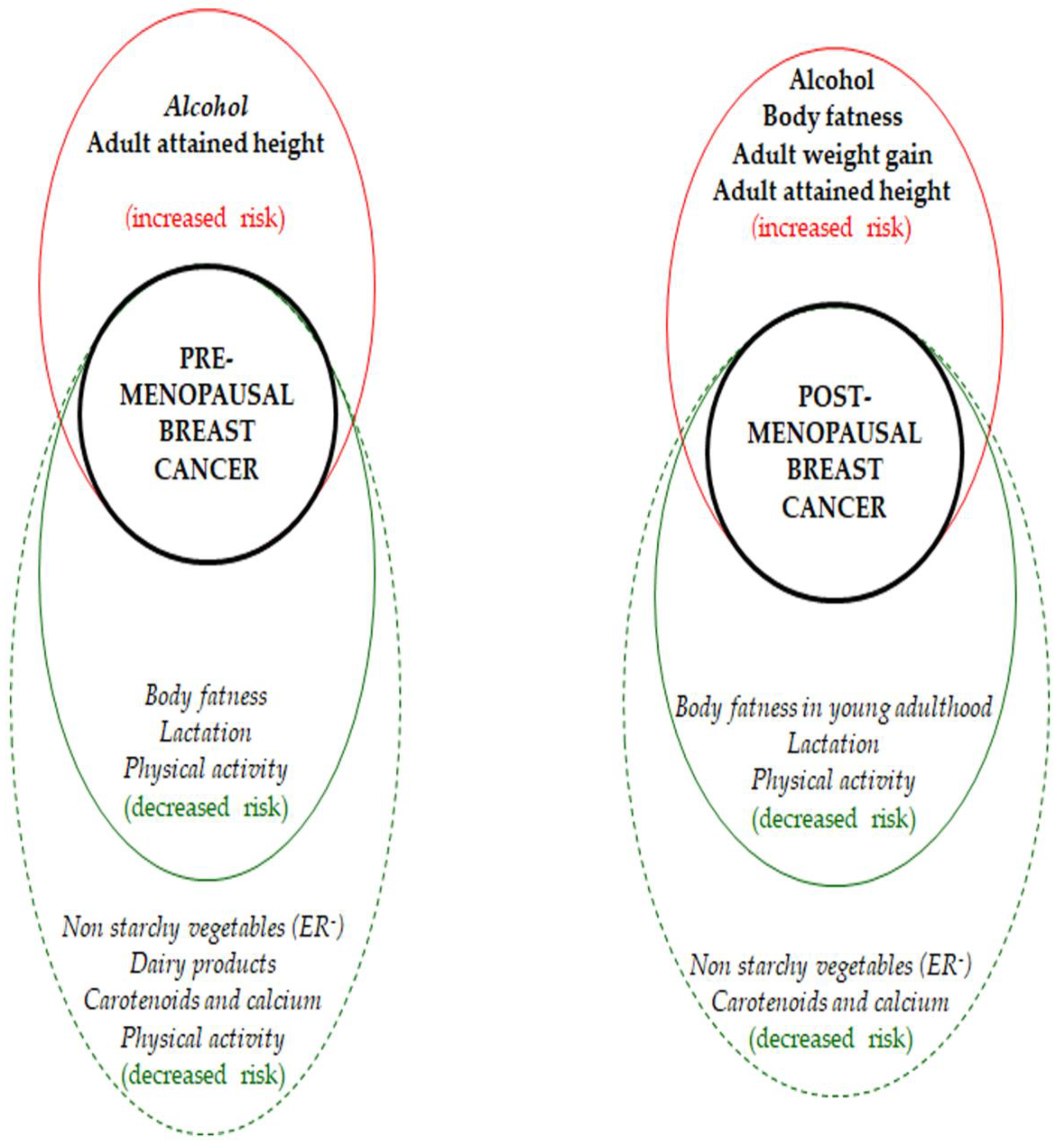
3.1. Fruits and Vegetables
3.2. Red Meat
3.3. Dietary Fat
3.4. Dairy Products
3.5. Carbohydrate and Glycaemic Index
3.6. Alcohol
3.7. Soy Products and Isoflavones
4. Impact of Therapy on Nutritional Status of Women with BC
5. Nutritional Interventions during BC Treatment
5.1. ω-3 Poly Unsaturated Fatty Acids (PUFAs)
5.2. Green Tea
5.3. Antioxidants Vitamins and Minerals
5.4. Intermittent Fasting
6. Nutritional Interventions to Reduce BC Recurrence and Mortality
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ferlay, J.; Hery, C.; Autier, P.; Sankaranarayanan, R. Global Burden of Breast Cancer. In Breast Cancer Epidemiology; Springer: New York, NY, USA, 2010; pp. 1–19. [Google Scholar]
- Seward, B.W.; Wild, C.P. International Agency for Research on Cancer. World Cancer Report 2014; Lyon International Agency for Research on Cancer: Lyon, France, 2014; pp. 16–69. [Google Scholar]
- Porter, P. “Westernizing” women’s risks? Breast cancer in lower-income countries. N. Engl. J. Med. 2008, 358, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Cancer Statistics Center. Available online: https://cancerstatisticscenter.cancer.org/#!/cancer-site/Breast (accessed on 27 September 2018).
- Global Cancer Observatory. Available online: http://gco.iarc.fr (accessed on 27 September 2018).
- Soerjomataram, I.; Louwman, W.J.; Lemmens, V.E.; de Vries, E.; Klokman, W.J.; Coebergh, J.W. Risks of second primary breast and urogenital cancer following female breast cancer in the south of The Netherlands, 1972–2001. Eur. J. Cancer 2005, 41, 2331–2337. [Google Scholar] [CrossRef] [PubMed]
- Haque, R.; Prout, M.; Geiger, A.M.; Kamineni, A.; Thwin, S.S.; Avila, C.; Silliman, R.A.; Quinn, V.; Yood, M.U. Comorbidities and cardiovascular disease risk in older breast cancer survivors. Am. J. Manag. Care 2014, 20, 86–92. [Google Scholar] [PubMed]
- Pasanisi, P.; Berrino, F.; De Petris, M.; Venturelli, E.; Mastroianni, A.; Panico, S. Metabolic syndrome as a prognostic factor for breast cancer recurrences. Int. J. Cancer 2006, 119, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Makari-Judson, G.; Braun, B.; Jerry, D.J.; Mertens, W.C. Weight gain following breast cancer diagnosis: Implication and proposed mechanisms. World J. Clin. Oncol. 2014, 5, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Althuis, M.D.; Fergenbaum, J.H.; Garcia-Closas, M.; Brinton, L.A.; Madigan, M.P.; Sherman, M.E. Etiology of hormone receptor-defined breast cancer: A systematic review of the literature. Cancer Epidemiol. Biomark. Prev. 2004, 13, 1558–1568. [Google Scholar]
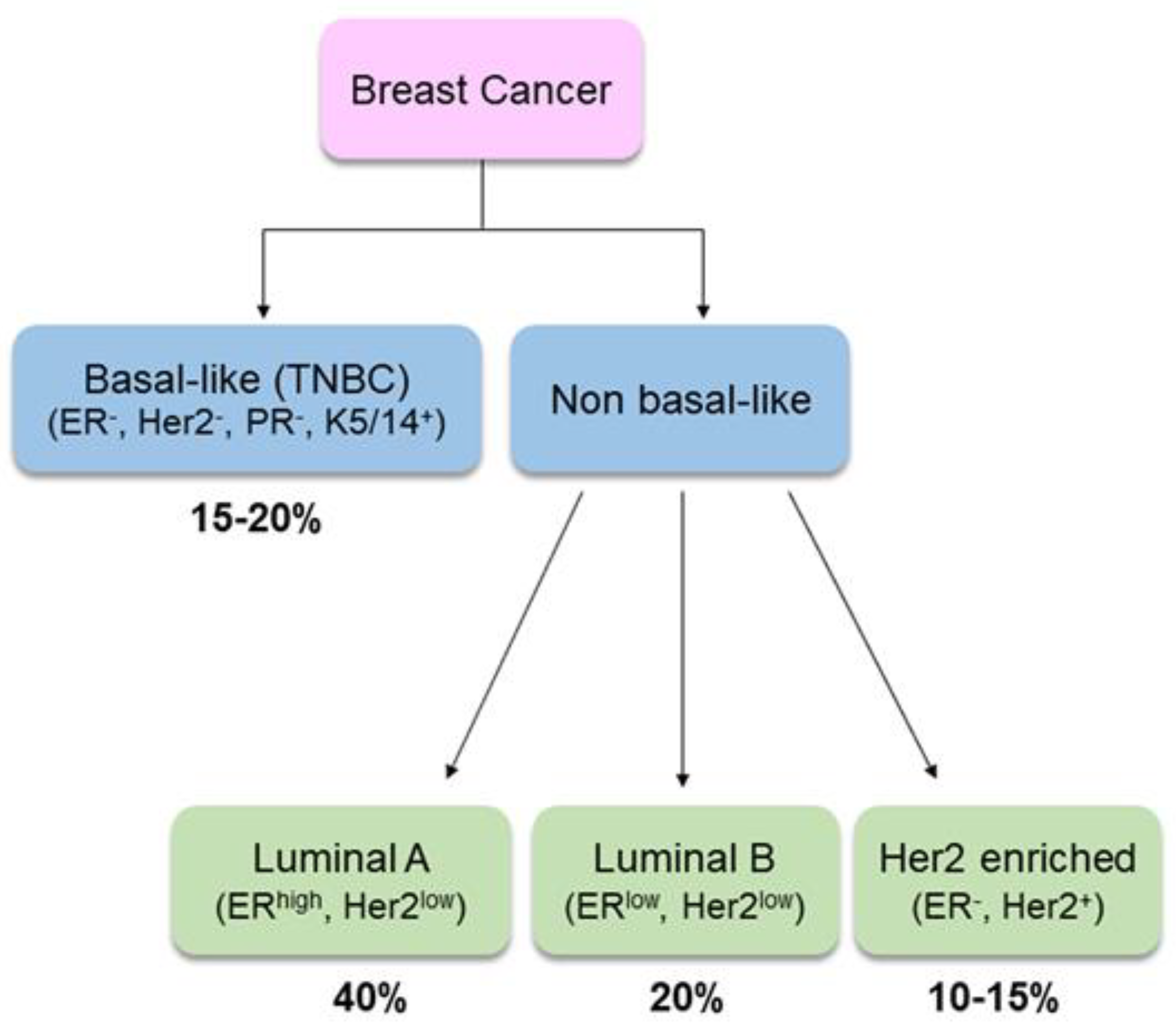
- Anderson, W.F.; Rosenber, P.S.; Prat, A.; Perou, C.M.; Sherman, M.E. How many etiological subtypes of breast cancer: Two, three, four, or more? J. Natl. Cancer Inst. 2014, 106, dju165. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Breast Cancer Facts & Figures 2017–2018. Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2017-2018.pdf (accessed on 1 June 2019).
- Zare, N.; Haem, E.; Lankarani, K.B.; Heydari, S.T.; Barooti, E. Breast cancer risk factors in a defined population: Weighted logistic regression approach for rare events. J. Breast Cancer 2013, 16, 214–219. [Google Scholar] [CrossRef]
- Sun, Y.S.; Zhao, Z.; Yang, Z.N.; Xu, F.; Lu, H.J.; Zhu, Z.Y.; Shi, W.; Jiang, J.; Yao, P.P.; Zhu, H.P. Risk factors and preventions of breast cancer. Int. J. Biol. Sci. 2017, 13, 1387–1397. [Google Scholar] [CrossRef]
- Giles, E.D.; Wellberg, E.A.; Astling, D.P.; Anderson, S.M.; Thor, A.D.; Jindal, S.; Tan, A.C.; Schedin, P.S.; Maclean, P.S. Obesity and overfeeding affecting both tumor and systemic metabolism activates the progesterone receptor to contribute to post-menopausal breast cancer. Cancer Res. 2012, 72, 6490–6501. [Google Scholar] [CrossRef]
- Mourouti, N.; Kontogianni, M.D.; Papavagelis, C.; Panagiotakos, D.B. Diet and breast cancer: A systematic review. Int. J. Food Sci. Nutr. 2015, 66, 1–42. [Google Scholar] [CrossRef] [PubMed]
- Protani, M.; Coory, M.; Martin, J.H. Effects of obesity on survival of women with breast cancer: Systematic review and meta-analysis. Breast Cancer Res. Treat. 2010, 123, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Kwan, M.L.; Weltzien, E.; Kushi, L.H.; Castillo, A.; Slattery, M.L.; Caan, B.J. Dietary patterns and breast cancer recurrence and survival among women with early-stage breast cancer. J. Clin. Oncol. 2009, 27, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Doyle, C.; Demark-Wahnefried, W.; Meyerhardt, J.; Courneya, K.S.; Schwartz, A.L.; Bandera, E.V.; Hamilton, K.K.; Grant, B.; McCullough, M.; et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J. Clin. 2012, 62, 243–274. [Google Scholar] [CrossRef] [PubMed]
- World Cancer Research Fund, Third Expert Report on “Diet, Nutrition, Physical Activity and Cancer: A Global Perspective”. Available online: https://www.wcrf.org/dietandcancer/breast-cancer (accessed on 27 September 2018).
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.S.; Vieira, A.R.; Aune, D.; Bandera, E.V.; Greenwood, D.C.; McTiernan, A.; Navarro Rosenblatt, D.; Thune, I.; Vieira, R.; Norat, T. Body mass index and survival in women with breast cancer—Systematic literature review and meta-analysis of 82 follow-up studies. Ann. Oncol. 2014, 25, 1901–1914. [Google Scholar] [CrossRef] [PubMed]
- George, S.M.; Bernstein, L.; Smith, A.W.; Neuhouser, M.L.; Baumgartner, K.B.; Baumgartner, R.N.; Ballard-Barbash, R. Central adiposity after breast cancer diagnosis is related to mortality in the Health, Eating, Activity, and Lifestyle study. Breast Cancer Res. Treat. 2014, 146, 647–655. [Google Scholar] [CrossRef]
- Skouroliakou, M.; Grosomanidis, D.; Massara, P.; Kostara, C.; Papandreou, P.; Ntountaniotis, D.; Xepapadakis, G. Serum antioxidant capacity, biochemical profile and body composition of breast cancer survivors in a randomized Mediterranean dietary intervention study. Eur. J. Nutr. 2018, 57, 2133–2145. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Chan, D.S.; Vieira, A.R.; Rosenblatt, D.A.; Vieira, R.; Greenwood, D.C.; Norat, T. Fruits, vegetables and breast cancer risk: A systematic review and meta-analysis of prospective studies. Breast Cancer Res. Treat. 2012, 134, 479–493. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Chiuve, S.E.; Willett, W.C.; Hankinson, S.E.; Hu, F.B.; Holmes, M.D. Intake of specific fruits and vegetables in relation to risk of estrogen receptor-negative breast cancer among post-menopausal women. Breast Cancer Res. Treat. 2013, 138, 925–930. [Google Scholar] [CrossRef]
- Masala, G.; Assedi, M.; Bendinelli, B.; Ermini, I.; Sieri, S.; Grioni, S.; Sacerdote, C.; Ricceri, F.; Panico, S.; Mattiello, A.; et al. Fruit and vegetables consumption and breast cancer risk: The EPIC Italy study. Breast Cancer Res. Treat. 2012, 132, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Farvid, M.S.; Stern, M.C.; Norat, T.; Sasazuki, S.; Vineis, P.; Weijenberg, M.P.; Wolk, A.; Wu, K.; Stewart, B.W.; Cho, E. Consumption of red and processed meat and breast cancer incidence: A systematic review and meta-analysis of prospective studies. Int. J. Cancer 2018, 143, 2787–2799. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.J.; Darwis, N.D.M.; Mackay, D.F.; Celis-Morales, C.A.; Lyall, D.M.; Sattar, N.; Gill, J.M.R.; Pell, J.P. Red and processed meat consumption and breast cancer: UK Biobank cohort study and meta-analysis. Eur. J. Cancer 2018, 90, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Prentice, R.L.; Caan, B.; Chlebowski, R.T.; Patterson, R.; Kuller, L.H.; Ockene, J.K.; Margolis, K.L.; Limacher, M.C.; Manson, J.E.; Parker, L.M.; et al. Low fat dietary pattern and risk of invasive breast cancer. The Women’s Health Initiative randomized controlled dietary modification trial. JAMA 2006, 295, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Turner, L.B. A meta-analysis of fat intake, reproduction, and breast cancer risk: An evolutionary perspective. Am. J. Hum. Biol. 2011, 23, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Makarem, N.; Chandran, U.; Bandera, E.V.; Parekh, N. Dietary fat in breast cancer survival. Annu. Rev. Nutr. 2013, 33, 319–348. [Google Scholar] [CrossRef] [PubMed]
- Sieri, S. Dietary fat intake and development of specific breast cancer subtypes. J. Natl. Cancer Inst. 2014, 106, dju068. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yang, L.; Zhang, D.; Jiang, W. Systematic review and meta-analysis suggest that dietary cholesterol intake increases risk of breast cancer. Nutr. Res. 2016, 36, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Missmer, S.A.; Smith-Warner, S.A.; Spiegelman, D.; Yaun, S.S.; Adami, H.O.; Beeson, W.L.; van den Brandt, P.A.; Fraser, G.E.; Freudenheim, J.L.; Goldbohm, R.A.; et al. Meat and dairy food consumption and breast cancer: A pooled analysis of cohort studies. Int. J. Epidemiol. 2002, 31, 78–85. [Google Scholar] [CrossRef]
- Dong, J.Y.; Zhang, L.; He, K.; Qin, L.Q. Dairy consumption and risk of breast cancer: A meta-analysis of prospective cohort studies. Breast Cancer Res. Treat. 2011, 127, 23–31. [Google Scholar] [CrossRef]
- Zang, J.; Shen, M.; Du, S.; Chen, T.; Zou, S. The association between dairy intake and breast cancer in western and asian populations: A systematic review and meta-analysis. J. Breast Cancer 2015, 18, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, S.; Chan, D.S.M.; Vingeliene, S.; Vieira, A.R.; Abar, L.; Polemiti, E.; Stevens, C.A.T.; Greenwood, D.C.; Aune, D.; Norat, T. Carbohydrates, glycemic index, glycemic load, and breast cancer risk: A systematic review and dose-response meta-analysis of prospective studies. Nutr. Rev. 2017, 75, 420–441. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.Q.; Xu, J.Y.; Wang, P.Y.; Hoshi, K. Soyfood intake in the prevention of breast cancer risk in women: A meta-analysis of observational epidemiological studies. J. Nutr. Sci. Vitam. 2006, 52, 428–436. [Google Scholar] [CrossRef][Green Version]
- Wu, A.H.; Yu, M.C.; Tseng, C.C.; Pike, M.C. Epidemiology of soy exposures and breast cancer risk. Br. J. Cancer 2008, 98, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.Y.; Qin, L.Q. Soy isoflavones consumption and risk of breast cancer incidence or recurrence: A meta-analysis of prospective studies. Breast Cancer Res. Treat. 2011, 125, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Couto, E.; Sandin, S.; Löf, M.; Ursin, G.; Adami, H.O.; Weiderpass, E. Mediterranean dietary pattern and risk of breast cancer. PLoS ONE 2013, 8, e55374. [Google Scholar] [CrossRef] [PubMed]
- Psaltopoulou, T.; Kosti, R.I.; Haidopoulos, D.; Dimopoulos, M.; Panagiotakos, D.B. Olive oil intake is inversely related to cancer prevalence: A systematic review and a meta-analysis of 13,800 patients and 23,340 controls in 19 observational studies. Lipids Health Dis. 2011, 10, 127. [Google Scholar] [CrossRef]
- Castelló, A.; Boldo, E.; Pérez-Gómez, B.; Lope, V.; Altzibar, J.M.; Martín, V.; Castaño-Vinyals, G.; Guevara, M.; Dierssen-Sotos, T.; Tardón, A.; et al. Adherence to the Western, Prudent and Mediterranean dietary patterns and breast cancer risk: MCC-Spain study. Maturitas 2017, 103, 8–15. [Google Scholar] [CrossRef]
- Toklu, H.; Nogay, N.H. Effects of dietary habits and sedentary lifestyle on breast cancer among women attending the oncology day treatment center at a state university in Turkey. Niger. J. Clin. Pr. 2018, 21, 1576–1584. [Google Scholar]
- Toledo, E.; Salas-Salvado, J.; Donat-Vargas, C.; Buil-Cosiales, P.; Estruch, R.; Ros, E.; Corella, D.; Fitó, M.; Hu, F.B.; Arós, F.; et al. Mediterranean diet and invasive breast cancer risk among women at high cardiovascular risk in the PREDIMED trial: A randomized clinical trial. JAMA Intern. Med. 2015, 175, 1752–1760. [Google Scholar] [CrossRef]
- Khalis, M.; Chajès, V.; Moskal, A.; Biessy, C.; Huybrechts, I.; Rinaldi, S.; Dossus, L.; Charaka, H.; Mellas, N.; Nejjari, C.; et al. Healthy lifestyle and breast cancer risk: A case-control study in Morocco. Cancer Epidemiol. 2019, 58, 160–166. [Google Scholar] [CrossRef]
- Van den Brandt, P.A.; Schulpen, M. Mediterranean diet adherence and risk of post-menopausal breast cancer: Results of a cohort study and meta-analysis. Int. J. Cancer 2017, 140, 2220–2231. [Google Scholar] [CrossRef] [PubMed]
- Fararouei, M.; Iqbal, A.; Rezaian, S.; Gheibi, Z.; Dianatinasab, A.; Shakarami, S.; Dianatinasab, M. Dietary habits and physical activity are associated with the risk of breast cancer among young iranian women: A case-control study on 1010 premenopausal women. Clin. Breast Cancer 2019, 19, 127–134. [Google Scholar] [CrossRef]
- Kontou, N. The Mediterranean Diet in Cancer Prevention. In The Mediterranean Diet; Preedy, V.R., Watson, R.R., Eds.; Academic Press: San Diego, CA, USA, 2015; Chapter 36; pp. 393–406. [Google Scholar]
- Maskarinec, G.; Morimoto, Y.; Takata, Y.; Murphy, S.P.; Stanczyk, F.Z. Alcohol and dietary fibre intakes affect circulating sex hormones among premenopausal women. Public Health Nutr. 2006, 9, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Braakhuis, A.J.; Campion, P.; Bishop, K.S. Reducing breast cancer recurrence: The role of dietary polyphenolics. Nutrients 2016, 8, 547. [Google Scholar] [CrossRef] [PubMed]
- Kanaya, N.; Adams, L.; Takasaki, A.; Chen, S. Whole blueberry powder inhibits metastasis of triple negative breast cancer in a xenograft mouse model through modulation of inflammatory cytokines. Nutr. Cancer 2014, 66, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Wadsworth, T.L.; Koop, D.R. Effects of the wine polyphenolics quercetin and resveratrol on pro-inflammatory cytokine expression in RAW 264.7 macrophages. Biochem. Pharm. 1999, 57, 941–949. [Google Scholar] [CrossRef]
- Gerhäuser, C.; Klimo, K.; Heiss, E.; Neumann, I.; Gamal-Eldeen, A.; Knauft, J.; Liu, G.Y.; Sitthimonchai, S.; Frank, N. Mechanism-based in vitro screening of potential cancer chemopreventive agents. Mutat. Res. 2003, 523, 163–172. [Google Scholar] [CrossRef]
- Biswas, S.K.; McClure, D.; Jimenez, L.A.; Megson, I.L.; Rahman, I. Curcumin induces glutathione biosynthesis and inhibits NF-kappaB activation and interleukin-8 release in alveolar epithelial cells: Mechanism of free radical scavenging activity. Antioxid. Redox Signal. 2005, 7, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Dannenberg, A.J.; Subbaramaiah, K. Targeting cyclooxygenase-2 in human neoplasia: Rationale and promise. Cancer Cell 2003, 4, 431–436. [Google Scholar] [CrossRef]
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [PubMed]
- Brueggemeier, R.W.; Díaz-Cruz, E.S.; Li, P.K.; Sugimoto, Y.; Lin, Y.C.; Shapiro, C.L. Translational studies on aromatase.; cyclooxygenases.; and enzyme inhibitors in breast cancer. J. Steroid Biochem. Mol. Biol. 2005, 95, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.P.; Chien, M.H. Phytoestrogens induce differential effects on both normal and malignant human breast cells in vitro. Climacteric 2014, 17, 682–691. [Google Scholar] [CrossRef] [PubMed]
- Papoutsi, Z.; Kassi, E.; Tsiapara, A.; Fokialakis, N.; Chrousos, G.P.; Moutsatsou, P. Evaluation of estrogenic/antiestrogenic activity of ellagic acid via the estrogen receptor subtypes ERalpha and ERbeta. J. Agric. Food Chem. 2005, 53, 7715–7720. [Google Scholar] [CrossRef] [PubMed]
- Inoue-Choi, M.; Sinha, R.; Gierach, G.L.; Ward, M.H. Red and processed meat, nitrite, and heme iron intakes and post-menopausal breast cancer risk in the NIH-AARP Diet and Health Study. Int. J. Cancer 2016, 138, 1609–1618. [Google Scholar] [CrossRef] [PubMed]
- Lauber, S.N.; Ali, S.; Gooderham, N.J. The cooked food derived carcinogen 2-amino-1-methyl-6-phenylimidazo[4.;5-b] pyridine is a potent oestrogen: A mechanistic basis for its tissue-specific carcinogenicity. Carcinogenesis 2004, 25, 2509–2517. [Google Scholar] [CrossRef]
- Rose, D.P. Effects of dietary fatty acids on breast and prostate cancers: Evidence from in vitro experiments and animal studies. AJCN 1997, 6, 1513S–1522S. [Google Scholar] [CrossRef]
- Blackburn, G.L.; Wang, K.A. Dietary fat reduction and breast cancer outcome: Results from the Women’s Intervention Nutrition Study (WINS). Am. J. Clin. Nutr. 2007, 86, 878–881. [Google Scholar] [CrossRef]
- VanWeelden, K.; Flanagan, L.; Binderup, L.; Tenniswood, M.; Welsh, J. Apoptotic regression of MCF-7 xenografts in nude mice treated with the vitamin D3 analog, EB1089. Endocrinology 1998, 139, 2102–2110. [Google Scholar] [CrossRef]
- Chiang, K.C.; Yeh, C.N.; Chen, S.C.; Shen, S.C.; Hsu, J.T.; Yeh, T.S.; Pang, J.H.; Su, L.J.; Takano, M.; Kittaka, A.; et al. MART-10, a new generation of vitamin D analog, is more potent than 1α,25-dihydroxyvitamin D(3) in inhibiting cell proliferation and inducing apoptosis in ER+ MCF-7 breast cancer cells. Evid. Based Complement. Altern. Med. 2012, 2012, 310872. [Google Scholar] [CrossRef]
- Welsh, J. Vitamin D and breast cancer: Insights from animal models. Am. J. Clin. Nutr. 2004, 80, 1721S–1724S. [Google Scholar] [CrossRef] [PubMed]
- Colston, K.W.; Perks, C.M.; Xie, S.P.; Holly, J.M. Growth inhibition of both MCF-7 and Hs578T human breast cancer cell lines by vitamin D analogues is associated with increased expression of insulin-like growth factor binding protein-3. J. Mol. Endocrinol. 1998, 20, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Cauley, J.A.; Chlebowski, R.T.; Wactawski-Wende, J.; Robbins, J.A.; Rodabough, R.J.; Chen, Z.; Johnson, K.C.; O’Sullivan, M.J.; Jackson, R.D.; Manson, J.E. Calcium plus vitamin D supplementation and health outcomes five years after active intervention ended: The Women’s Health Initiative. J. Womens Health 2013, 22, 915–929. [Google Scholar] [CrossRef] [PubMed]
- Schulz, M.; Hoffmann, K.; Weikert, C.; Nöthlings, U.; Schulze, M.B.; Boeing, H. Identification of a dietary pattern characterized by high-fat food choices associated with increased risk of breast cancer: The European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Br. J. Nutr. 2008, 100, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.D.; Liu, S.; Hankinson, S.E.; Colditz, G.A.; Hunter, D.J.; Willett, W. Dietary carbohydrates, fiber, and breast cancer risk. C Am. J. Epidemiol. 2004, 159, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, H.G.; Murray, L.J.; Cardwell, C.R.; Cantwell, M.M. Dietary glycaemic index.; glycaemic load and breast cancer risk: A systematic review and meta-analysis. Br. J. Cancer 2008, 99, 1170–1175. [Google Scholar] [CrossRef] [PubMed]
- Mullie, P.; Koechlin, A.; Boniol, M.; Autier, P.; Boyle, P. Relation between breast cancer and high glycemic index or glycemic load: A meta-analysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2016, 56, 152–159. [Google Scholar] [CrossRef]
- Romieu, I.; Ferrari, P.; Rinaldi, S.; Slimani, N.; Jenab, M.; Olsen, A.; Tjonneland, A.; Overvad, K.; Boutron-Ruault, M.C.; Lajous, M.; et al. Dietary glycemic index and glycemic load and breast cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC). Am. J. Clin. Nutr. 2012, 96, 345–355. [Google Scholar] [CrossRef]
- Kaaks, R.; Lukanova, A. Energy balance and cancer: The role of insulin and insulin-like growth factor-I. Proc. Nutr. Soc. 2001, 60, 91–106. [Google Scholar] [CrossRef]
- Helle, S.I.; Lonning, P.E. Insulin-like growth factors in breast cancer. Acta Oncol. 1996, 35, 19–22. [Google Scholar] [CrossRef]
- Key, T.J.; Appleby, P.N.; Reeves, G.K.; Roddam, A.W. Endogenous Hormones and Breast Cancer Collaborative Group. Insulin-like growth factor 1 (IGF1), IGF binding protein 3 (IGFBP3), and breast cancer risk: Pooled individual data analysis of 17 prospective studies. Lancet Oncol. 2010, 11, 530–542. [Google Scholar] [PubMed]
- Xu, M.; Wang, S.; Ren, Z.; Frank, J.A.; Yang, X.H.; Zhang, Z.; Ke, Z.; Shi, X.; Luo, J. Chronic ethanol exposure enhances the aggressiveness of breast cancer: The role of p38γ. Oncotarget 2016, 7, 3489–3505. [Google Scholar] [CrossRef] [PubMed]
- Roswall, N.; Weiderpass, E. Alcohol as a risk factor for Cancer: Existing evidence in a global perspective. J. Prev. Med. Public Health 2015, 48, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Nguyen, N.; Colditz, G.A. Links between alcohol consumption and breast cancer: A look at the evidence. Womens Health 2015, 11, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Seitz, H.K.; Pelucchi, C.; Bagnardi, V.; La Vecchia, C. Epidemiology and pathophysiology of alcohol and breast cancer: Update 2012. Alcohol 2012, 47, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Murphy, P.A.; Song, T.; Buseman, G.; Barua, K.; Beecher, G.R.; Trainer, D.; Holden, J. Isoflavones in retail and institutional soy foods. J. Agric. Food Chem. 1999, 47, 2697–2704. [Google Scholar] [CrossRef] [PubMed]
- Messina, M.J.; Wood, C.E. Soy isoflavones, estrogen therapy, and breast cancer risk: Analysis and commentary. Nutr. J. 2008, 7, 17. [Google Scholar] [CrossRef]
- Anampa, J.; Makower, D.; Sparano, J.A. Progress in adjuvant chemotherapy for breast cancer: An overview. BMC Med. 2015, 13, 195. [Google Scholar] [CrossRef]
- Kayl, A.E.; Meyers, C.A. Side-effects of chemotherapy and quality of life in ovarian and breast cancer patients. Curr. Opin. Obs. Gynecol. 2006, 18, 24–28. [Google Scholar] [CrossRef]
- Saquib, N.; Flatt, S.W.; Natarajan, L.; Thomson, C.A.; Bardwell, W.A.; Caan, B.; Rock, C.L.; Pierce, J.P. Weight gain and recovery of pre-cancer weight after breast cancer treatments: Evidence from the women’s healthy eating and living (WHEL) study. Breast Cancer Res. Treat. 2007, 105, 177–186. [Google Scholar] [CrossRef]
- Buch, K.; Gunmalm, V.; Andersson, M.; Schwarz, P.; Brøns, C. Effect of chemotherapy and aromatase inhibitors in the adjuvant treatment of breast cancer on glucose and insulin metabolism-A systematic review. Cancer Med. 2019, 8, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Caan, B.J.; Kwan, M.L.; Hartzell, G.; Castillo, A.; Slattery, M.L.; Sternfeld, B.; Weltzien, E. Pre-diagnosis body mass index, post-diagnosis weight change, and prognosis among women with early stage breast cancer. Cancer Causes Control. 2008, 19, 1319–1328. [Google Scholar] [CrossRef] [PubMed]
- Irwin, M.L.; McTiernan, A.; Baumgartner, R.N.; Baumgartner, K.B.; Bernstein, L.; Gilliland, F.D.; Ballard-Barbash, R. Changes in body fat and weight after a breast cancer diagnosis: Influence of demographic, prognostic, and lifestyle factors. J. Clin. Oncol. 2005, 23, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Nechuta, S.J.; Caan, B.J.; Chen, W.Y.; Flatt, S.W.; Lu, W.; Patterson, R.E.; Poole, E.M.; Kwan, M.L.; Chen, Z.; Weltzien, E.; et al. The After Breast Cancer Pooling Project: Rationale, methodology, and breast cancer survivor characteristics. Cancer Causes Control. 2011, 22, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- Chlebowski, R.T. Nutrition and physical activity influence on breast cancer incidence and outcome. Breast 2013, 22, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Boltong, A.; Aranda, S.; Keast, R.; Wynne, R.; Francis, P.A.; Chirgwin, J.; Gough, K. A prospective cohort study of the effects of adjuvant breast cancer chemotherapy on taste function, food liking, appetite and associated nutritional outcomes. PLoS ONE 2014, 9, e103512. [Google Scholar] [CrossRef] [PubMed]
- De Vries, Y.C.; Boesveldt, S.; Kelfkens, C.S.; Posthuma, E.E.; van den Berg, M.M.G.A.; de Kruif, J.T.C.M.; Haringhuizen, A.; Sommeijer, D.W.; Buist, N.; Grosfeld, S.; et al. Taste and smell perception and quality of life during and after systemic therapy for breast cancer. Breast Cancer Res. Treat. 2018, 170, 27–34. [Google Scholar] [CrossRef] [PubMed]
- De Vries, Y.C.; van den Berg, M.M.G.A.; de Vries, J.H.M.; Boesveldt, S.; de Kruif, J.T.C.M.; Buist, N.; Haringhuizen, A.; Los, M.; Sommeijer, D.W.; Timmer-Bonte, J.H.N.; et al. Differences in dietary intake during chemotherapy in breast cancer patients compared to women without cancer. Support. Care Cancer 2017, 25, 2581–2591. [Google Scholar] [CrossRef]
- Speck, R.M.; DeMichele, A.; Farrar, J.T.; Hennessy, S.; Mao, J.J.; Stineman, M.G.; Barg, F.K. Taste alteration in breast cancer patients treated with taxane chemotherapy: Experience, effect, and coping strategies. Support. Care Cancer 2013, 21, 549–555. [Google Scholar] [CrossRef]
- Murtaza, B.; Hichami, A.; Khan, A.S.; Ghiringhelli, F.; Khan, N.A. Alteration in taste perception in cancer: Causes and strategies of treatment. Front. Physiol. 2017, 8, 134. [Google Scholar] [CrossRef]
- Villarini, A.; Pasanisi, P.; Raimondi, M.; Gargano, G.; Bruno, E.; Morelli, D.; Evangelista, A.; Curtosi, P.; Berrino, F. Preventing weight gain during adjuvant chemotherapy for breast cancer: A dietary intervention study. Breast Cancer Res. Treat. 2012, 135, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Bougnoux, P.; Hajjaji, N.; Ferrasson, M.N.; Giraudeau, B.; Couet, C.; Le Floch, O. Improving outcome of chemotherapy of metastatic breast cancer by docosahexaenoic acid: A phase II trial. Br. J. Cancer 2009, 101, 1785–1978. [Google Scholar] [CrossRef] [PubMed]
- Hutchins-Wiese, H.L.; Picho, K.; Watkins, B.A.; Li, Y.; Tannenbaum, S.; Claffey, K.; Kenny, A.M. High-dose eicosapentaenoic acid and docosahexaenoic acid supplementation reduces bone resorption in post-menopausal breast cancer survivors on aromatase inhibitors: A pilot study. Nutr. Cancer 2014, 66, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.; Unger, J.M.; Crew, K.D.; Till, C.; Greenlee, H.; Gralow, J.; Dakhil, S.R.; Minasian, L.M.; Wade, J.L., 3rd; Fisch, M.J.; et al. Omega-3 fatty acid use for obese breast cancer patients with aromatase inhibitor-related arthralgia (SWOG S0927). Breast Cancer Res. Treat. 2018, 172, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Ghoreishi, Z.; Esfahani, A.; Djazayeri, A.; Djalali, M.; Golestan, B.; Ayromlou, H.; Hashemzade, S.; Asghari Jafarabadi, M.; Montazeri, V.; Keshavarz, S.A.; et al. Omega-3 fatty acids are protective against paclitaxel-induced peripheral neuropathy: A randomized double-blind placebo controlled trial. Bmc Cancer 2012, 12, 355. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Tajima, K.; Mizutani, M.; Iwata, H.; Iwase, T.; Miura, S.; Hirose, K.; Hamajima, N.; Tominaga, S. Regular consumption of green tea and the risk of breast cancer recurrence: Follow-up study from the Hospital-based Epidemiologic Research Program at Aichi Cancer Center (HERPACC).; Japan. Cancer Lett. 2001, 167, 175–182. [Google Scholar] [CrossRef]
- Nakachi, K.; Suemasu, K.; Suga, K.; Takeo, T.; Imai, K.; Higashi, Y. Influence of drinking green tea on breast cancer malignancy among Japanese patients. Jpn. J. Cancer Res. 1998, 89, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Bao, P.P.; Zhao, G.M.; Shu, X.O.; Peng, P.; Cai, H.; Lu, W.; Zheng, Y. Modifiable lifestyle factors and triple-negative breast cancer survival: A population-based prospective study. Epidemiology 2015, 26, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Babu, R.J.; Sundravel, S.; Arumugam, G.; Renuka, R.; Deepa, N.; Sachdanandam, P. Salubrious effect of vitamin C and vitamin E on tamoxifen-treated women in breast cancer with reference to plasma lipid and lipoprotein levels. Cancer Lett. 2000, 151, 1–5. [Google Scholar] [CrossRef]
- Suhail, N.; Bilal, N.; Khan, H.Y.; Hasan, S.; Sharma, S.; Khan, F.; Mansoor, T.; Banu, N. Effect of vitamins C and E on antioxidant status of breast-cancer patients undergoing chemotherapy. J. Clin. Pharm. 2012, 37, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Peralta, E.A.; Brewer, A.T.; Louis, S.; Dunnington, G.L. Vitamin E increases biomarkers of estrogen stimulation when taken with tamoxifen. J. Surg. Res. 2009, 153, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Alhambra, D.; Servitja, S.; Javaid, M.K.; Garrigós, L.; Arden, N.K.; Cooper, C.; Albanell, J.; Tusquets, I.; Diez-Perez, A.; Nogues, X. Vitamin D threshold to prevent aromatase inhibitor-related bone loss: The B-ABLE prospective cohort study. Breast Cancer Res. Treat. 2012, 133, 1159–1167. [Google Scholar] [CrossRef] [PubMed]
- Khan, Q.J.; Reddy, P.S.; Kimler, B.F.; Sharma, P.; Baxa, S.E.; O’Dea, A.P.; Klemp, J.R.; Fabian, C.J. Effect of vitamin D supplementation on serum 25-hydroxy vitamin D levels, joint pain, and fatigue in women starting adjuvant letrozole treatment for breast cancer. Breast Cancer Res. Treat. 2010, 119, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.S.; Hu, X.J.; Zhao, Y.M.; Yang, J.; Li, D. Intake of fish and marine n-3 polyunsaturated fatty acids and risk of breast cancer: Meta-analysis of data from 21 independent prospective cohort studies. BMJ 2013, 346, f3706. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ma, D.W. The role of n-3 polyunsaturated fatty acids in the prevention and treatment of breast cancer. Nutrients 2014, 6, 5184–5223. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Hraiki, A.; Bebawy, M.; Pazderka, C.; Rawling, T. Anti-tumor activities of lipids and lipid analogues and their development as potential anticancer drugs. Pharm. Ther. 2015, 150, 109–128. [Google Scholar] [CrossRef]
- D’Eliseo, D.; Velotti, F. Omega-3 fatty acids and cancer cell cytotoxicity: Implications for multi-targeted cancer therapy. J. Clin. Med. 2016, 5, 15. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov (accessed on 20 November 2018).
- Hurria, A.; Rosen, C.; Hudis, C.; Zuckerman, E.; Panageas, K.S.; Lachs, M.S.; Witmer, M.; van Gorp, W.G.; Fornier, M.; D’Andrea, G.; et al. Cognitive function of older patients receiving adjuvant chemotherapy for breast cancer: A pilot prospective longitudinal study. J. Am. Geriatr. Soc. 2006, 54, 925–931. [Google Scholar] [CrossRef]
- Orchard, T.S.; Gaudier-Diaz, M.M.; Weinhold, K.R.; Courtney DeVries, A. Clearing the fog: A review of the effects of dietary omega-3 fatty acids and added sugars on chemotherapy-induced cognitive deficits. Breast Cancer Res. Treat. 2017, 161, 391–398. [Google Scholar] [CrossRef]
- Manni, A.; El-Bayoumy, K.; Thompson, H. Docosahexaenoic acid in combination with dietary energy restriction for reducing the risk of obesity related breast cancer. Int. J. Mol. Sci. 2017, 19, 28. [Google Scholar] [CrossRef]
- Lecumberri, E.; Dupertuis, Y.M.; Miralbell, R.; Pichard, C. Green tea polyphenol epigallocatechin-3-gallate (EGCG) as adjuvant in cancer therapy. Clin. Nutr. 2013, 32, 894–903. [Google Scholar] [CrossRef] [PubMed]
- Yiannakopoulou, E.C. Interaction of green tea catechins with breast cancer endocrine treatment: A systematic review. Pharmacology 2014, 94, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Beltz, L.A.; Bayer, D.K.; Moss, A.L.; Simet, I. M: Mechanisms of cancer prevention by green and black tea polyphenols. Anticancer Agents Med. Chem. 2006, 6, 389–406. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Han, J.; Xiao, H.; Qiao, J.; Han, M. Effect of tea polyphenol compounds on anticancer drugs in terms of anti-tumor activity, toxicology, and pharmacokinetics. Nutrients 2016, 8, 762. [Google Scholar] [CrossRef] [PubMed]
- Greenlee, H.; Balneaves, L.G.; Carlson, L.E.; Cohen, M.; Deng, G.; Hershman, D.; Mumber, M.; Perlmutter, J.; Seely, D.; Sen, A.; et al. Society for Integrative Oncology. Clinical practice guidelines on the use of integrative therapies as supportive care in patients treated for breast cancer. J. Natl. Cancer Inst. Monogr. 2014, 2014, 346–358. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M. Nutritional supplements and cancer: Potential benefits and proven harms. Am. Soc. Clin. Oncol. Educ. Book 2014, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Kwan, M.L.; Greenlee, H.; Lee, V.S.; Castillo, A.; Gunderson, E.P.; Habel, L.A.; Kushi, L.H.; Sweeney, C.; Tam, E.K.; Caan, B.J. Multivitamin use and breast cancer outcomes in women with early-stage breast cancer: The Life After Cancer Epidemiology study. Breast Cancer Res. Treat. 2011, 130, 195–205. [Google Scholar] [CrossRef]
- Meulepas, J.M.; Newcomb, P.A.; Burnett-Hartman, A.N.; Hampton, J.M.; Trentham-Dietz, A. Multivitamin supplement use and risk of invasive breast cancer. Public Health Nutr. 2010, 13, 1540–1545. [Google Scholar] [CrossRef]
- Chen, Q.; Espey, M.G.; Krishna, M.C.; Mitchell, J.B.; Corpe, C.P.; Buettner, G.R.; Shacter, E.; Levine, M. Pharmacologic ascorbic acid concentrations selectively kill cancer cells: Action as a pro-drug to deliver hydrogen peroxide to tissues. Proc. Natl. Acad. Sci. USA 2005, 102, 13604–13609. [Google Scholar] [CrossRef]
- Willcox, J.K.; Ash, S.L.; Catignani, G.L. Antioxidants and prevention of chronic disease. Crit. Rev. Food Sci. Nutr. 2004, 44, 275–295. [Google Scholar] [CrossRef]
- Harris, H.R.; Bergkvist, L.; Wolk, A. Vitamin C intake and breast cancer mortality in a cohort of Swedish women. Brit. J. Cancer 2013, 109, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.R.; Orsini, N.; Wolk, A. Vitamin C and survival among women with breast cancer: A meta-analysis. Eur. J. Cancer 2014, 50, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Vollbracht, C.; Schneider, B.; Leendert, V.; Weiss, G.; Auerbach, L.; Beuth, J. Intravenous vitamin C administration improves quality of life in breast cancer patients during chemo-/radiotherapy and aftercare: Results of a retrospective, multicenter, epidemiological cohort study in Germany. Vivo 2011, 25, 983–990. [Google Scholar]
- Carr, A.C.; Vissers, M.C.; Cook, J. Relief from cancer chemotherapy side effects with pharmacologic vitamin C. NZ Med. J. 2014, 127, 66–70. [Google Scholar]
- Saintot, M.; Mathieu-Daude, H.; Astre, C.; Grenier, J.; Simony-Lafontaine, J.; Gerber, M. Oxidant-antioxidant status in relation to survival among breast cancer patients. Int. J. Cancer 2002, 97, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Tam, K.W.; Ho, C.T.; Lee, W.J.; Tu, S.H.; Huang, C.S.; Chen, C.S.; Lee, C.H.; Wu, C.H.; Ho, Y.S. Alteration of α-tocopherol-associated protein (TAP) expression in human breast epithelial cells during breast cancer development. Food Chem. 2013, 138, 1015–1521. [Google Scholar] [CrossRef]
- Pawłowicz, Z.; Zachara, B.A.; Trafikowska, U.; Maciag, A.; Marchaluk, E.; Nowicki, A. Blood selenium concentrations and glutathione peroxidase activities in patients with breast cancer and with advanced gastrointestinal cancer. J. Trace Elem. Electrolytes Health Dis. 1991, 5, 275–277. [Google Scholar]
- Gröber, U. Antioxidants and other micronutrients in complementary oncology. Breast Care 2009, 4, 13–20. [Google Scholar] [CrossRef]
- Chung, M.; Balk, E.M.; Brendel, M.; Ip, S.; Lau, J.; Lee, J.; Lichtenstein, A.; Patel, K.; Raman, G.; Tatsioni, A.; et al. Vitamin D and calcium: A systematic review of health outcomes. Evid. Rep. Technol. Assess (Full Rep.) 2009, 183, 1–420. [Google Scholar]
- Picotto, G.; Liaudat, A.C.; Bohl, L.; Tolosa de Talamoni, N. Molecular aspects of vitamin D anticancer activity. Cancer Investig. 2012, 30, 604–614. [Google Scholar] [CrossRef]
- Imtiaz, S.; Siddiqui, N. Vitamin-D status at breast cancer diagnosis: Correlation with social and environmental factors and dietary intake. J. Ayub Med. Coll. Abbottabad 2014, 26, 186–190. [Google Scholar] [PubMed]
- Vrieling, A.; Seibold, P.; Johnson, T.S.; Heinz, J.; Obi, N.; Kaaks, R.; Flesch-Janys, D.; Chang-Claude, J. Circulating 25-hydroxyvitamin D and post-menopausal breast cancer survival: Influence of tumor characteristics and lifestyle factors? Int. J. Cancer 2014, 134, 2972–2983. [Google Scholar] [CrossRef]
- Kim, Y.; Je, Y. Vitamin D intake, blood 25(OH)D levels, and breast cancer risk or mortality: A meta-analysis. Br. J. Cancer 2014, 110, 2772–2784. [Google Scholar] [CrossRef] [PubMed]
- Rose, A.A.; Elser, C.; Ennis, M.; Goodwin, P.J. Blood levels of vitamin D and early stage breast cancer prognosis: A systematic review and meta-analysis. Breast Cancer Res. Treat. 2013, 141, 331–339. [Google Scholar] [CrossRef]
- Yao, S.; Kwan, M.L.; Ergas, I.J.; Roh, J.M.; Cheng, T.D.; Hong, C.C.; McCann, S.E.; Tang, L.; Davis, W.; Liu, S.; et al. Association of serum level of vitamin D at diagnosis with breast cancer survival: A case-cohort analysis in the pathways study. JAMA Oncol. 2017, 3, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Vaughan-Shaw, P.G.; O’Sullivan, F.; Farrington, S.M.; Theodoratou, E.; Campbell, H.; Dunlop, M.G.; Zgaga, L. The impact of vitamin D pathway genetic variation and circulating 25-hydroxyvitamin D on cancer outcome: Systematic review and meta-analysis. Br. J. Cancer 2017, 116, 1092–1110. [Google Scholar] [CrossRef] [PubMed]
- Crew, K.D.; Shane, E.; Cremers, S.; McMahon, D.J.; Irani, D.; Hershman, D.L. High prevalence of vitamin D deficiency despite supplementation in premenopausal women with breast cancer undergoing adjuvant chemotherapy. J. Clin. Oncol. 2009, 27, 2151–2156. [Google Scholar] [CrossRef] [PubMed]
- Jacot, W.; Pouderoux, S.; Thezenas, S.; Chapelle, A.; Bleuse, J.P.; Romieu, G.; Lamy, P.J. Increased prevalence of vitamin D insufficiency in patients with breast cancer after neoadjuvant chemotherapy. Breast Cancer Res. Treat. 2012, 134, 709–717. [Google Scholar] [CrossRef]
- Hatse, S.; Lambrechts, D.; Verstuyf, A.; Smeets, A.; Brouwers, B.; Vandorpe, T.; Brouckaert, O.; Peuteman, G.; Laenen, A.; Verlinden, L.; et al. Vitamin D status at breast cancer diagnosis: Correlation with tumor characteristics, disease outcome, and genetic determinants of vitamin D insufficiency. Carcinogenesis 2012, 33, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- McKay, J.D.; McCullough, M.L.; Ziegler, R.G.; Kraft, P.; Saltzman, B.S.; Riboli, E.; Barricarte, A.; Berg, C.D.; Bergland, G.; Bingham, S.; et al. Vitamin D receptor polymorphisms and breast cancer risk: Results from the National Cancer Institute Breast and Prostate Cancer Cohort Consortium. Cancer Epidemiol. Biomark. Prev. 2009, 18, 297–305. [Google Scholar] [CrossRef]
- Datta, M.; Schwartz, G.G. Calcium and vitamin D supplementation and loss of bone mineral density in women undergoing breast cancer therapy. Crit. Rev. Oncol. Hematol. 2013, 88, 613–624. [Google Scholar] [CrossRef] [PubMed]
- Almquist, M.; Anagnostaki, L.; Bondeson, L.; Bondeson, A.G.; Borgquist, S.; Landberg, G.; Malina, J.; Malm, J.; Manjer, J. Serum calcium and tumour aggressiveness in breast cancer: A prospective study of 7847 women. Eur. J. Cancer Prev. 2009, 18, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Bolland, M.J.; Avenell, A.; Baron, J.A.; Grey, A.; MacLennan, G.S.; Gamble, G.D.; Reid, I.R. Effect of calcium supplements on risk of myocardial infarction and cardiovascular events: Meta-analysis. BMJ 2010, 341, c3691. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Raffaghello, L.; Brandhorst, S.; Safdie, F.M.; Bianchi, G.; Martin-Montalvo, A.; Pistoia, V.; Wei, M.; Hwang, S.; Merlino, A.; et al. Fasting cycles retard growth of tumors and sensitize a range of cancer cell types to chemotherapy. Sci. Transl. Med. 2012, 4, 124ra27. [Google Scholar] [CrossRef] [PubMed]
- Safdie, F.M.; Dorff, T.; Quinn, D.; Fontana, L.; Wei, M.; Lee, C.; Cohen, P.; Longo, V.D. Fasting and cancer treatment in humans: A case series report. Aging 2009, 1, 988–1007. [Google Scholar] [CrossRef] [PubMed]
- De Groot, S.; Vreeswijk, M.P.; Welters, M.J.; Gravesteijn, G.; Boei, J.J.; Jochems, A.; Houtsma, D.; Putter, H.; van der Hoeven, J.J.; Nortier, J.W.; et al. The effects of short-term fasting on tolerance to (neo) adjuvant chemotherapy in HER2-negative breast cancer patients: A randomized pilot study. BMC Cancer 2015, 15, 652. [Google Scholar] [CrossRef]
- Chlebowski, R.T.; Blackburn, G.; Thomson, C.A.; Nixon, D.W.; Shapiro, A.; Hoy, M.K.; Goodman, M.T.; Giuliano, A.E.; Karanja, N.; McAndrew, P.; et al. Dietary fat reduction and breast cancer outcome: Interim efficacy results from the Women’s Intervention Nutrition Study (WINS). J. Natl. Cancer Inst. 2006, 98, 1767–1776. [Google Scholar] [CrossRef]
- Pierce, J.P.; Natarajan, L.; Caan, B.L.; Parker, B.A.; Greenberg, E.R.; Flatt, S.W.; Rock, C.L.; Kealey, S.; Al-Delaimy, W.K.; Bardwell, W.A.; et al. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: The Women’s Healthy Eating and Living (WHEL) randomized trial. JAMA 2007, 298, 289–298. [Google Scholar] [CrossRef]
- McCullough, M.L.; Gapstur, S.M.; Shah, R.; Campbell, P.T.; Wang, Y.; Doyle, C.; Gaudet, M.M. Pre- and postdiagnostic diet in relation to mortality among breast cancer survivors in the CPS-II Nutrition Cohort. Cancer Causes Control. 2016, 27, 1303–1314. [Google Scholar] [CrossRef]
- Kroenke, C.H.; Kwan, M.L.; Sweeney, C.; Castillo, A.; Caan, B.J. High- and low-fat dairy intake, recurrence, and mortality after breast cancer diagnosis. J. Natl Cancer Inst. 2013, 105, 616–623. [Google Scholar] [CrossRef]
- Belle, F.N.; Kampman, E.; McTiernan, A.; Bernstein, L.; Baumgartner, K.; Baumgartner, R.; Ambs, A.; Ballard-Barbash, R.; Neuhouser, M.L. Dietary fiber, carbohydrates, glycemic index, and glycemic load in relation to breast cancer prognosis in the HEAL cohort. Cancer Epidemiol. Biomark. Prev. 2011, 20, 890–899. [Google Scholar] [CrossRef] [PubMed]
- McEligot, A.J.; Largent, J.; Ziogas, A.; Peel, D.; Anton-Culver, H. Dietary fat, fiber, vegetable, and micronutrients are associated with overall survival in post-menopausal women diagnosed with breast cancer. Nutr. Cancer 2006, 55, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.D.; Chen, W.Y.; Hankinson, S.E.; Willett, W.C. Physical activity’s impact on the association of fat and fiber intake with survival after breast cancer. Am. J. Epidemiol. 2009, 170, 1250–1256. [Google Scholar] [CrossRef] [PubMed]
- Shu, X.O.; Zheng, Y.; Cai, H.; Gu, K.; Chen, Z.; Zheng, W.; Lu, W. Soy food intake and breast cancer survival. JAMA 2009, 302, 2437–2443. [Google Scholar] [CrossRef] [PubMed]
- Chi, F.; Wu, R.; Zeng, Y.C.; Xing, R.; Liu, Y.; Xu, Z.G. Post-diagnosis soy food intake and breast cancer survival: A meta-analysis of cohort studies. Asian Pac. J. Cancer Prev. 2013, 14, 2407–2412. [Google Scholar] [CrossRef] [PubMed]
- Nechuta, S.J.; Caan, B.J.; Chen, W.Y.; Lu, W.; Chen, Z.; Kwan, M.L.; Flatt, S.W.; Zheng, Y.; Zheng, W.; Pierce, J.P.; et al. Soy food intake after diagnosis of breast cancer and survival: An in-depth analysis of combined evidence from cohort studies of US and Chinese women. Am. J. Clin. Nutr. 2012, 96, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Haslam, D.E.; Terry, M.B.; Knight, J.A.; Andrulis, I.L.; Daly, M.B.; Buys, S.S.; John, E.M. Dietary isoflavone intake and all-cause mortality in breast cancer survivors: The Breast Cancer Family Registry. Cancer 2017, 123, 2070–2079. [Google Scholar] [CrossRef]
| Study | Results | Reference | |
|---|---|---|---|
| Fruits, vegetables | Meta-analysis (15 prospective studies) | RR = 0.89 (95% CI, 0.80–0.99, p = 0.67) fruits + vegetables; highest vs. lowest intake RR = 0.92 (95% CI, 0.86–0.98, p = 0.36) fruits; highest vs. lowest intake RR = 0.99 (95% CI, 0.92–1.06, p = 0.26) vegetables; highest vs. lowest intake | [25] |
| Prospective study (75,929 women, 38–63 years, 24 years follow-up) | RR = 0.82 (95% CI, 0.71–0.96, p = 0.01), 2 servings/week of total berries RR = 0.69 (95% CI, 0.50–0.95, p = 0.02), 1 serving/week of blueberries RR = 0.59 (95% CI, 0.37–0.93, p = 0.02), 2 servings/week of peaches/nectarines | [26] | |
| Prospective study (31,000 women, 36–64 years, 11.25 years follow-up) | HR = 0.70 (95% CI, 0.57–0.86, p = 0.0001) leafy vegetables, highest vs. lowest quintile HR = 0.75 (95% CI, 0.60–0.94, p = 0.01) fruiting vegetables, highest vs lowest quintile no association with fruit | [27] | |
| Red meat | Meta-analysis (13 cohort, 3 case-control, 2 clinical trials) | RR = 1.06 (95%CI, 0.99–1.14) unprocessed red meat, highest vs. lowest intake RR = 1.09 (95%CI, 1.03–1.16) processed red meat, highest vs. lowest intake | [28] |
| Cohort study (262,195 women, 7 years follow-up) Meta-analysis | HR = 1.21 (95% CI, 1.08–1.35, p = 0.001), >9 g/day processed red meat RR = 1.09 (95% CI 1.03–1.15, p = 0.662), >9 g/day processed red meat in post-menopausal women RR = 0.99 (95% CI 0.88–1.10, p = 0.570), >9 g/day processed red meat in pre-menopausal women | [29] | |
| Dietary Fat | Randomized controlled trial (48,835 post-menopausal women, 8.1 years follow-up) | HR = 0.91 (95% CI, 0.83–1.01, NS) intervention group vs. control group | [30] |
| Meta-analysis (cohort + case-control studies) | RR = 1.091 (95% CI, 1.001–1.184) cohort PUFA RR = 1.042 (95%CI, 1.013–1.073) case-control total fat RR = 1.22 (95% CI, 1.08–1.38) case-control PUFA | [31] | |
| Systematic review (18 studies) | 45–78% increased risk of death with increased intake of trans fats | [32] | |
| EPIC study (337,327 women, 11.5 years follow-up) | HR = 1.20 (95% CI, 1.0–1.45, p = 0.05), highest vs. lowest quintile of total fat intake (ER+PR+ BC) HR = 1.2 (95% CI, 1.09–1.52, p = 0.009), highest vs. lowest quintile of saturated fat intake (ER+PR+ BC) HR = 1.29 (95% CI, 1.01–1.64, p = 0.04), highest vs. lowest quintile of saturated fat intake (HER2− BC) | [33] | |
| Meta-analysis (6 cohort studies + 3 case-control studies) | RR = 1.29 (95% CI, 1.06–1.56), highest vs. lowest cholesterol intake | [34] | |
| Dairy products | Pooled analysis (8 prospective cohort studies) (351,041 women, 15 years follow-up) | NS | [35] |
| Meta-analysis (18 prospective cohort studies, n = 1,063,471) | RR = 0.91 (95% CI, 0.80–1.02, p = 0.003), milk consumption RR = 0.85 (95% CI, 0.76–0.95, p = 0.01), highest vs. lowest total dairy food | [36] | |
| Meta-analysis (22 cohort + 5 case-control studies) | RR = 0.90 (95% CI, 0.83–0.98, p = 0.111), highest vs. lowest dairy products RR = 0.91 (95% CI, 0.83–0.99, p = 0.991), yogurt consumption RR = 0.85 (95% CI, 0.75–0.96, p = 0.121), low-fat dairy consumption | [37] | |
| Carbohydrate, Glycaemic Index | Meta-analysis (19 prospective studies) | RR = 1.04 (95% CI, 1.00–1.07, p = 0.19), 10 units/d for glycemic index RR = 1.01 (95% CI, 0.98–1.04, p = 0.07), 50 units/d for glycemic load RR = 1.00 (95% CI, 0.96–1.05, p = 0.01), 50 g/d for carbohydrate intake | [38] |
| Soy products, isoflavones | Meta-analysis (14 case-control + 7 cohort studies) | RR = 0.75 (95% CI, 0.59–0.95, p = 0.023), soyfood intake RR = 0.81 (95% CI, 0.67–0.99), isoflavone intake | [39] |
| Meta-analysis (1 cohort + 7 case-control studies) | OR = 0.71 (95% CI, 0.60–0.85, p = 0.023), highest vs. lowest soy intake in Asians OR = 0.88 (95% CI, 0.78–0.98, p = 0.60), moderate vs. lowest soy intake in Asians OR = 1.04 (95% CI, 0.97–1.11, p = 0.42), highest vs. lowest soy isoflavone intake in Western populations | [40] | |
| Meta-analysis (18 prospective studies) | RR = 0.89 (95% CI, 0.79–0.99, p = 0.001), highest vs. lowest isoflavone intake (RR = 0.76, 95% CI: 0.65–0.86, p = 0.136 in Asian population; RR = 0.97, 95% CI: 0.87–1.06, p = 0.083 in Western population) | [41] |
| Study | Intervention | Results | Reference | |
|---|---|---|---|---|
| ω-3 PUFAs | Phase II clinical trial (n = 25 breast cancer patients, 31 months follow-up) | 1.8 g DHA/day anthracycline | Improvement of chemo-therapy outcome: median TTP = 6 months (95% CI, 2.8–8.7 months); median OS = 22 months (95% CI, 17–33 months) No severe adverse side effects (grade 3 or 4 toxicity only for neutropenia and alopecia, 80%) | [99] |
| Pilot study (n = 38 postmenopausal breast cancer patients) | 4 g/day EPA + DHA for 3 months AI therapy | Inhibition of bone resorption in the fish oil responders vs. placebo (p < 0.05) | [100] | |
| Controlled clinical trial (n = 249 postmenopausal breast cancer patients) | 3.3 g/day ω3 PUFA (560 mg EPA + DHA, 40:20 ratio) 24 weeks AI therapy | Reduction of arthralgia (4.36 vs. 5.70, p = 0.02) obese BC patients vs. placebo | [101] | |
| Controlled clinical trial (n = 20 breast cancer patients) | EPA (0.19 g/day) + DHA (1.04 g/day) paclitaxel | Reduction of paclitaxel-induced peripheral neuropathy incidence (OR = 0.3; 95% CI, 0.10–0.88, p = 0.029), but not severity (0.95% CI = (−2.06–0.02), p = 0.054) EPA + DHA vs. placebo | [102] | |
| Green tea | Prospective cohort study (n = 1160 breast cancer patients, 8 years follow-up) | Regular consumption of green tea | Inverse association between regular green tea consumption (≥3 cups/day) and BC recurrence for stage I/II patients (HR = 0.69; 95% CI, 0.47–1.00, p < 0.05) | [103] |
| Prospective cohort study (n = 472 breast cancer patients, 7 years follow-up) | Regular consumption of green tea | Inverse association between regular green tea consumption (≥5 cups/day) and BC recurrence for stage I/II patients (RR = 0.564; 95% CI, 0.350–0.911, p < 0.05) | [104] | |
| Prospective cohort study (n = 5042, 9.1 years follow-up) | Regular consumption of green tea | Reduced risk of total mortality (HR = 0.57; 95% CI: 0.34–0.93) and recurrence (HR = 0.54; 95% CI: 0.31–0.96) for the first 60-month post-diagnosis period | [105] | |
| Vitamin C | Controlled clinical trial (n = 54 post-menopausal breast cancer patients) | Vitamin C (500 mg) and E (400 mg) +tamoxifen (10 mg twice a day) for 90 days | Decrease of total cholesterol, TG, VLDL (p < 0.001) and LDL (p < 0.01) vs. tamoxifen alone Increase of HDL (p < 0.01) vs. tamoxifen alone | [106] |
| Controlled clinical trial (n = 40 breast cancer patients) | Vitamin C (500 mg) and E (400 mg) + 5-fluorouracil (500 mg/m2) + doxorubicin (50 mg/m2) + cyclophosphamide (500 mg/m2) (every 3 weeks for six cycles) | Increase of SOD, CAT, GST, GPx, GSH (p < 0.01) vs. chemotherapy alone Decrease of MDA, DNA damage (p < 0.01) vs. chemotherapy alone | [107] | |
| Vitamin E | Prospective cohort study (n = 7 breast cancer patients, 30 days follow-up) | Vitamin E (400 mg) + tamoxifen (20 mg daily) for 30 days | Vitamin E supplement interferes with the therapeutic effects of tamoxifen (increase expression of biomarkers of estrogen-stimulation (ER, PR, p-ERK in breast biopsies) | [108] |
| Vitamin D | Prospective cohort study (n = 232 post-menopausal breast cancer patients, 1-year follow-up) | Calcium (1 g) + vitamin D3 (800 IU/d and additional 16,000 IU, every 2 weeks) + AI therapy for 1 year | Reduction of AI-associated lumbar spine bone loss: 1.70% (95% CI, 0.4–3.0%; p = 0.005) (women with 25(OH)D serum levels ≥40 ng/ml vs. women with serum levels <30 ng/ml) | [109] |
| Prospective cohort study (n = 60 post-menopausal breast cancer patients, 16 weeks follow-up) | 50,000 IU/week + AI therapy for 12 weeks | Decrease of disability from joint pain (52 vs. 19%; p = 0.026); reduction of fatigue (BFI scores 1.4 vs. 2.9; NS); reduction of menopausal symptoms (MENQOL scores 2.2 vs. 3.2, p = 0.035) (women with 25OHD levels > 66 ng/ml vs. women with levels < 66 ng/ml) | [110] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Cicco, P.; Catani, M.V.; Gasperi, V.; Sibilano, M.; Quaglietta, M.; Savini, I. Nutrition and Breast Cancer: A Literature Review on Prevention, Treatment and Recurrence. Nutrients 2019, 11, 1514. https://doi.org/10.3390/nu11071514
De Cicco P, Catani MV, Gasperi V, Sibilano M, Quaglietta M, Savini I. Nutrition and Breast Cancer: A Literature Review on Prevention, Treatment and Recurrence. Nutrients. 2019; 11(7):1514. https://doi.org/10.3390/nu11071514
Chicago/Turabian StyleDe Cicco, Paola, Maria Valeria Catani, Valeria Gasperi, Matteo Sibilano, Maria Quaglietta, and Isabella Savini. 2019. "Nutrition and Breast Cancer: A Literature Review on Prevention, Treatment and Recurrence" Nutrients 11, no. 7: 1514. https://doi.org/10.3390/nu11071514
APA StyleDe Cicco, P., Catani, M. V., Gasperi, V., Sibilano, M., Quaglietta, M., & Savini, I. (2019). Nutrition and Breast Cancer: A Literature Review on Prevention, Treatment and Recurrence. Nutrients, 11(7), 1514. https://doi.org/10.3390/nu11071514





