Guava (Psidium guajava) Fruit Extract Prepared by Supercritical CO2 Extraction Inhibits Intestinal Glucose Resorption in a Double-Blind, Randomized Clinical Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Guava Fruit Extract
2.2. HPLC-MS, High Pressure Liquid Chromatography-High Resolution Mass Spectrometry
2.3. GC-MS, Gas Chromatography-Mass Spectrometry
2.4. NMR, Nuclear Magnetic Resonance Spectroscopy
2.5. Study Design
2.6. Sample Preparation
2.7. Plasma Blood Glucose and Insulin Determination
2.8. Calculations and Statistics
3. Results
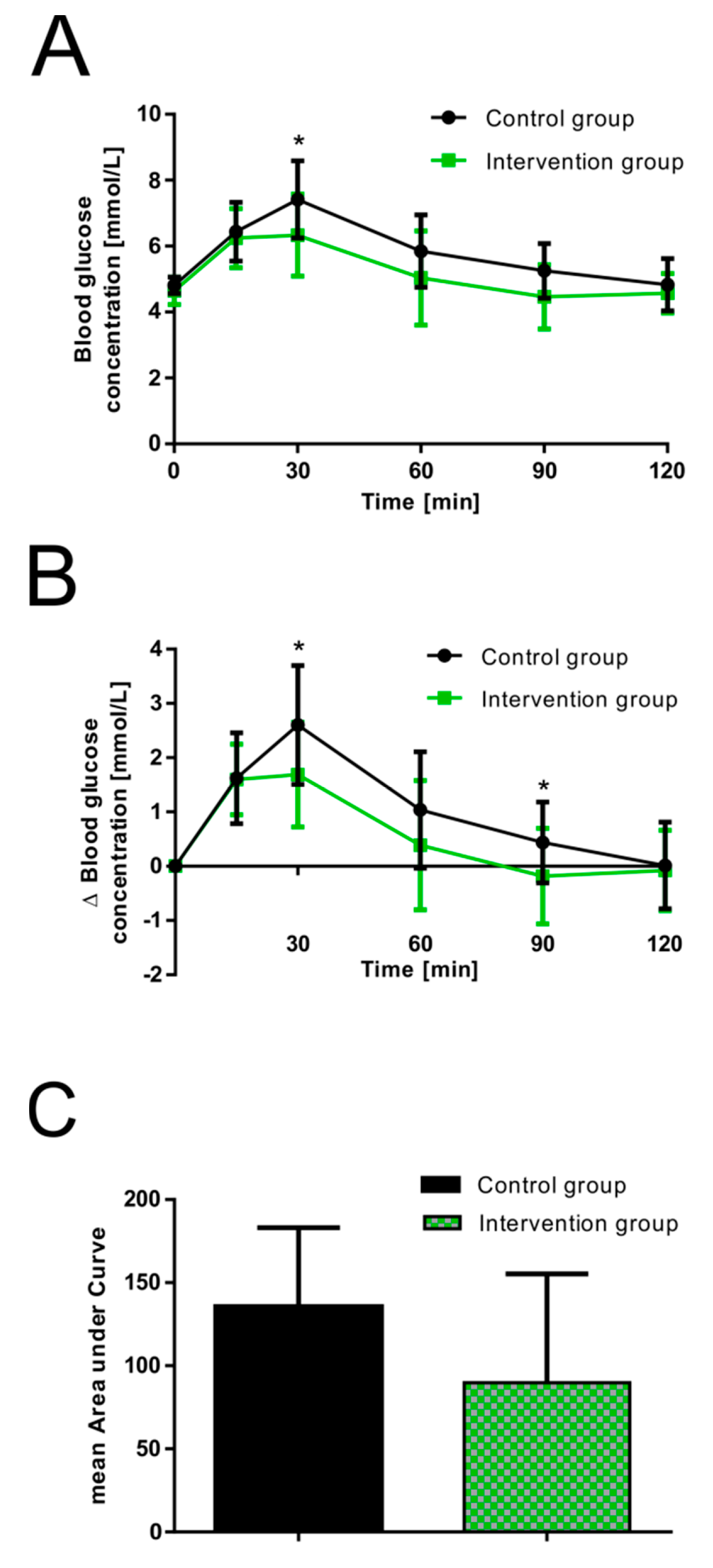
3.1. Influence of Guava Fruit Extract on Postprandial Plasma Glucose Response
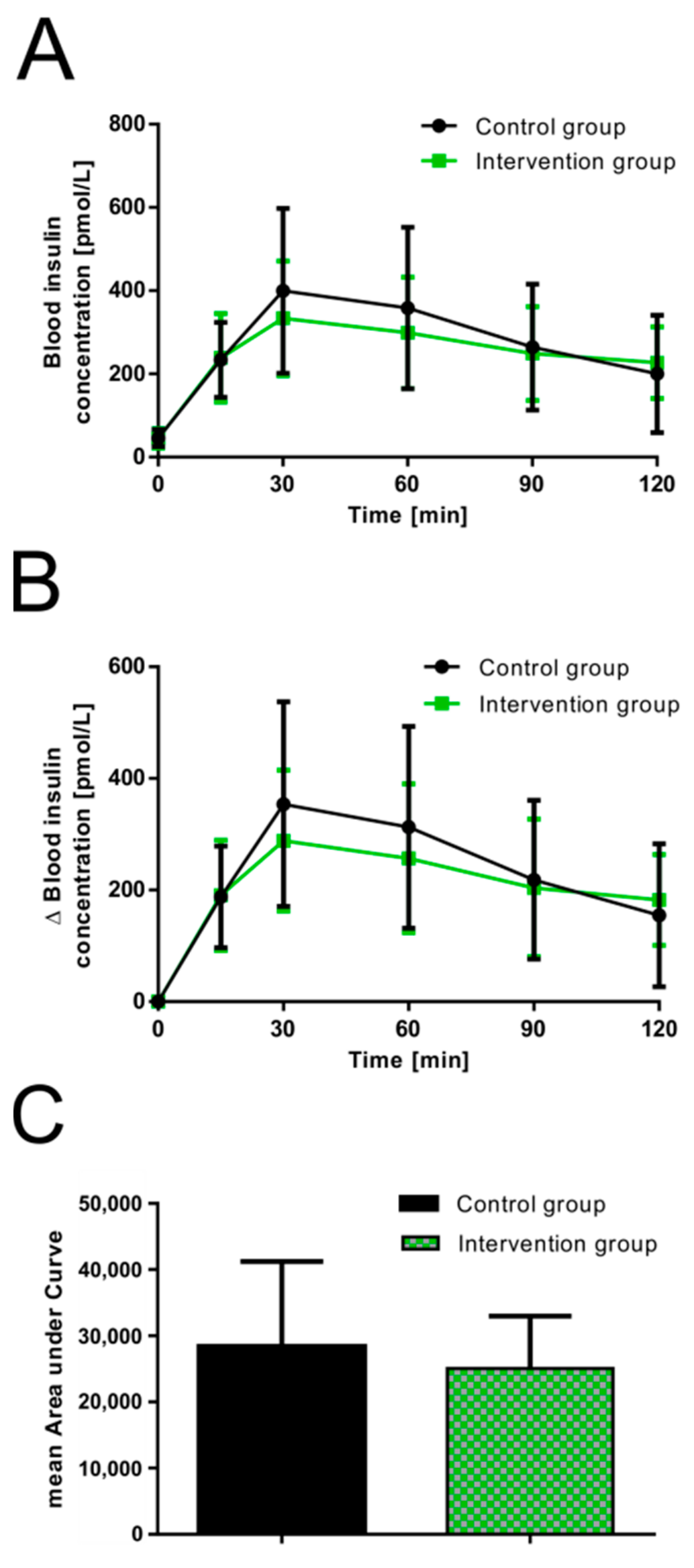
3.2. Influence of Guava Fruit Extract on Postprandial Plasma Insulin Response
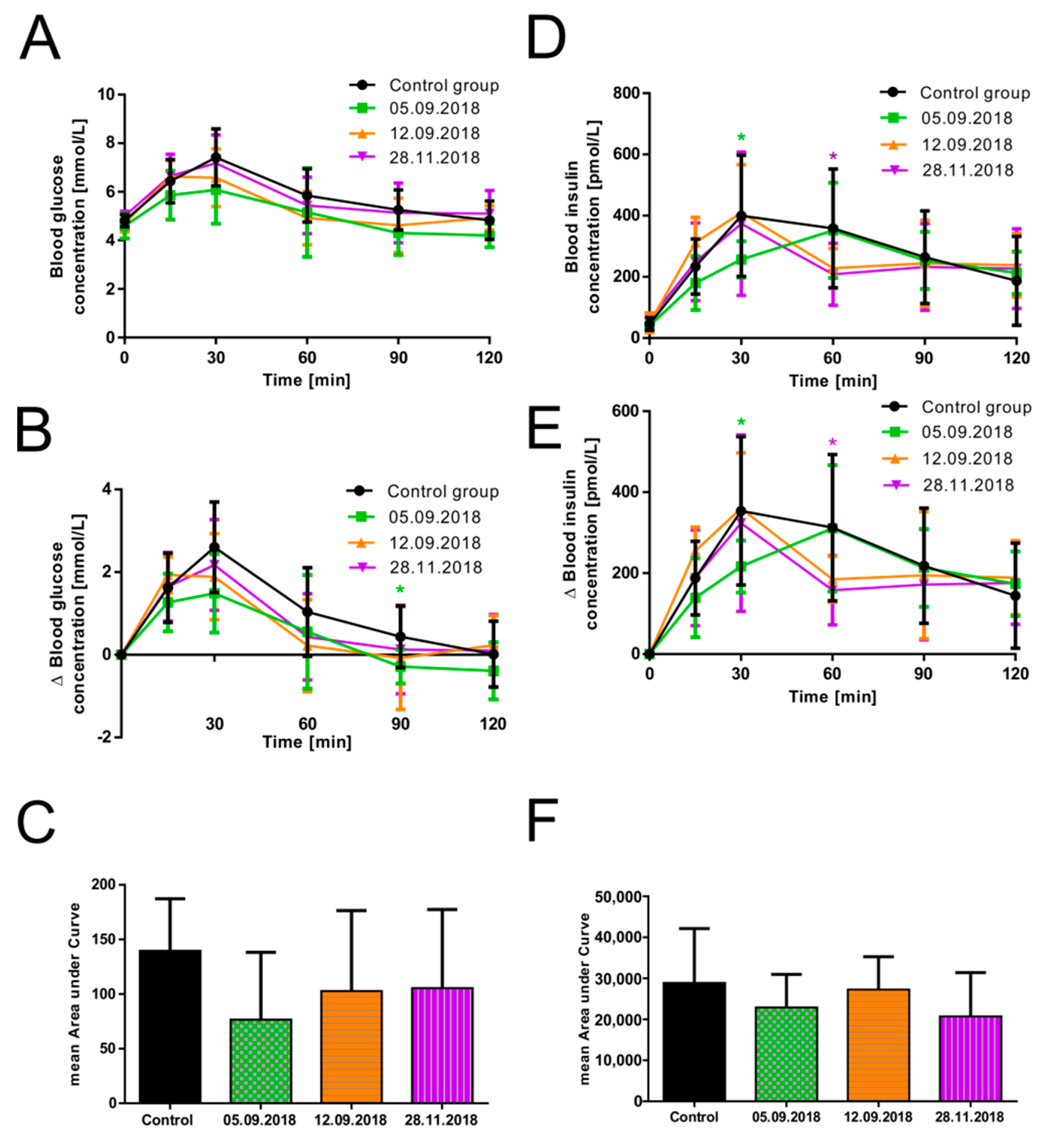
3.3. The Efficacy of Guava Fruit Extract Depends on Its Storage Time
3.4. HPLC-MS, GC-MS, and NMR Analysis of Guava Fruit Extract
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rosenberg, D.E.; Jabbour, S.A.; Goldstein, B.J. Insulin resistance, diabetes and cardiovascular risk: Approaches to treatment. Diabetes Obes. Metab. 2005, 7, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Schmutterer, I.; Delcour, J.; Griebler, R. Österreichischer Diabetesbericht 2017; Bundesministerium für Gesundheit und Frauen: Vienna, Austria, 2017.
- Adamska-Patruno, E.; Billing-Marczak, K.; Orlowski, M.; Gorska, M.; Krotkiewski, M.; Kretowski, A. A Synergistic Formulation of Plant Extracts Decreases Postprandial Glucose and Insulin Peaks: Results from Two Randomized, Controlled, Cross-Over Studies Using Real-World Meals. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.K.; Leung, P.S. Multifaceted interplay among mediators and regulators of intestinal glucose absorption: Potential impacts on diabetes research and treatment. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E887–E899. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G. Possible effects of dietary polyphenols on sugar absorption and digestion. Molecul. Nutr. Food Res. 2013, 57, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.; Eck, P.; Chen, S.; Corpe, C.P.; Lee, J.H.; Kruhlak, M.; Levine, M. Inhibition of the intestinal glucose transporter GLUT2 by flavonoids. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2007, 21, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Jacques, P.F.; Cassidy, A.; Rogers, G.; Peterson, J.J.; Meigs, J.B.; Dwyer, J.T. Higher dietary flavonol intake is associated with lower incidence of type 2 diabetes. J. Nutr. 2013, 143, 1474–1480. [Google Scholar] [CrossRef] [PubMed]
- Castro-Acosta, M.L.; Stone, S.G.; Mok, J.E.; Mhajan, R.K.; Fu, C.I.; Lenihan-Geels, G.N.; Corpe, C.P.; Hall, W.L. Apple and blackcurrant polyphenol-rich drinks decrease postprandial glucose, insulin and incretin response to a high-carbohydrate meal in healthy men and women. J. Nutr. Biochem. 2017, 49, 53–62. [Google Scholar] [CrossRef]
- Paquette, M.; Medina Larque, A.S.; Weisnagel, S.J.; Desjardins, Y.; Marois, J.; Pilon, G.; Dudonne, S.; Marette, A.; Jacques, H. Strawberry and cranberry polyphenols improve insulin sensitivity in insulin-resistant, non-diabetic adults: A parallel, double-blind, controlled and randomised clinical trial. Br. J. Nutr. 2017, 117, 519–531. [Google Scholar] [CrossRef]
- Schulze, C.; Bangert, A.; Kottra, G.; Geillinger, K.E.; Schwanck, B.; Vollert, H.; Blaschek, W.; Daniel, H. Inhibition of the intestinal sodium-coupled glucose transporter 1 (SGLT1) by extracts and polyphenols from apple reduces postprandial blood glucose levels in mice and humans. Molecul. Nutr. Food Res. 2014, 58, 1795–1808. [Google Scholar] [CrossRef]
- Wang, X.; Tian, J.; Jiang, J.; Li, L.; Ying, X.; Tian, H.; Nie, M. Effects of green tea or green tea extract on insulin sensitivity and glycaemic control in populations at risk of type 2 diabetes mellitus: A systematic review and meta-analysis of randomised controlled trials. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2014, 27, 501–512. [Google Scholar] [CrossRef]
- Devalaraja, S.; Jain, S.; Yadav, H. Exotic Fruits as Therapeutic Complements for Diabetes, Obesity and Metabolic Syndrome. Food Res. Int. 2011, 44, 1856–1865. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.C.; Shen, S.C.; Wu, J.S. Effect of guava (Psidium guajava L.) leaf extract on glucose uptake in rat hepatocytes. J. Food Sci. 2009, 74, H132–H138. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, S.; Haseena Banu, H.; Mookambika Ramya Bai, R.; Shanmugavalli, R. Biochemical evaluation of antihyperglycemic and antioxidant nature of Psidium guajava leaves extract in streptozotocin-induced experimental diabetes in rats. Pharm Biol. 2009, 47, 298–303. [Google Scholar] [CrossRef]
- Bahrani, A.H.M.; Zaheri, H.; Soltani, N.; Kharazmi, F.; Keshavarz, M.; Kamalinajad, M. Effect of the administration of Psidium guava leaves on blood glucose, lipid profiles and sensitivity of the vascular mesenteric bed to Phenylephrine in streptozotocin-induced diabetic rats. J. Diabetes Mellit. 2012, 2, 138–145. [Google Scholar] [CrossRef]
- Liu, X.; Yan, X.; Bi, J.; Liu, J.; Zhou, M.; Wu, X.; Chen, Q. Determination of phenolic compounds and antioxidant activities from peel, flesh, seed of guava (Psidium guajava L.). Electrophoresis 2018, 39, 1654–1662. [Google Scholar] [CrossRef] [PubMed]
- Muller, U.; Stubl, F.; Schwarzinger, B.; Sandner, G.; Iken, M.; Himmelsbach, M.; Schwarzinger, C.; Ollinger, N.; Stadlbauer, V.; Hoglinger, O.; et al. In Vitro and In Vivo Inhibition of Intestinal Glucose Transport by Guava (Psidium Guajava) Extracts. Molecul. Nutr. Food Res. 2018, 62, e1701012. [Google Scholar] [CrossRef] [PubMed]
- Spraul, M.; Schutz, B.; Humpfer, E.; Mortter, M.; Schafer, H.; Koswig, S.; Rinke, P. Mixture analysis by NMR as applied to fruit juice quality control. Magn. Reson. Chem. MRC 2009, 47, S130–S137. [Google Scholar] [CrossRef]
- Committee for Medicinal Products for Human Use (CHMP). Guideline on adjustment for baseline covariates in clinical trials. In EMA/CHMP/295050/2013; Agency, E.M., Ed.; Committee for Medicinal Products for Human Use (CHMP): London, UK, 2015. [Google Scholar]
- Friede, T.; Kieser, M. Blinded sample size recalculation for clinical trials with normal data and baseline adjusted analysis. Pharm. Stat. 2011, 10, 8–13. [Google Scholar] [CrossRef]
- World Health Organization. WHO Guidelines on Drawing Blood: Best Practices in Phlebotomy; WHO Press: Geneva, Switzerland, 2010. [Google Scholar]
- Chen, M.E.; Aguirre, R.S.; Hannon, T.S. Methods for Measuring Risk for Type 2 Diabetes in Youth: The Oral Glucose Tolerance Test (OGTT). Curr. Diabetes Rep. 2018, 18, 51. [Google Scholar] [CrossRef]
- Balijepalli, C.; Druyts, E.; Siliman, G.; Joffres, M.; Thorlund, K.; Mills, E.J. Hypoglycemia: A review of definitions used in clinical trials evaluating antihyperglycemic drugs for diabetes. Clin. Epidemiol. 2017, 9, 291–296. [Google Scholar] [CrossRef]
- International Hypoglycaemia Study Group. Glucose Concentrations of Less Than 3.0 mmol/L (54 mg/dL) Should Be Reported in Clinical Trials: A Joint Position Statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Car. 2017, 40, 155–157. [Google Scholar] [CrossRef] [PubMed]
- Lown, M.; Fuller, R.; Lightowler, H.; Fraser, A.; Gallagher, A.; Stuart, B.; Byrne, C.; Lewith, G. Mulberry-extract improves glucose tolerance and decreases insulin concentrations in normoglycaemic adults: Results of a randomised double-blind placebo-controlled study. PLoS ONE 2017, 12, e0172239. [Google Scholar] [CrossRef] [PubMed]
- Hanhineva, K.; Torronen, R.; Bondia-Pons, I.; Pekkinen, J.; Kolehmainen, M.; Mykkanen, H.; Poutanen, K. Impact of dietary polyphenols on carbohydrate metabolism. Int. J. Mol. Sci. 2010, 11, 1365–1402. [Google Scholar] [CrossRef] [PubMed]
- Castro-Vargas, H.I.; Rodriguez-Varela, L.I.; Parada-Alfonso, F. Guava (Psidium guajava L.) seed oil obtained with a homemade supercritical fluid extraction system using supercritical CO2 and co-solvent. J. Supercrit. Fluids 2011, 56, 238–242. [Google Scholar] [CrossRef]
- Shaw, E.F.; Charters, S. Chemistry and Technology of Soft Drinks and Fruit Juices, 3rd ed.; Ashurst, P.R., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 310–355. [Google Scholar]
- Da Silva, R.P.; Rocha-Santos, T.A.; Duarte, A.C. Supercritical fluid extraction of bioactive compounds. Trends Anal. Chem. 2016, 76, 40–51. [Google Scholar] [CrossRef]
- Renard, C. Extraction of bioactives from fruit and vegetables: State of the art and perspectives. Food Sci. Technol. 2018, 93, 390–395. [Google Scholar] [CrossRef]
- Cao, G.; Cai, H.; Cai, B.; Tu, S. Effect of 5-hydroxymethylfurfural derived from processed Cornus officinalis on the prevention of high glucose-induced oxidative stress in human umbilical vein endothelial cells and its mechanism. Food Chem. 2013, 140, 273–279. [Google Scholar] [CrossRef]
- Wei, Y.B.; Yang, X.D. Synthesis, characterization and anti-diabetic therapeutic potential of a new benzyl acid-derivatized kojic acid vanadyl complex. Biometals 2012, 25, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Brunner, G. Supercritical fluids: Technology and application to food processing. J. Food Eng. 2005, 67, 21–33. [Google Scholar] [CrossRef]
- Khosravi-Darani, K. Research activities on supercritical fluid science in food biotechnology. Crit. Rev. Food Sci. Nutr. 2010, 50, 479–488. [Google Scholar] [CrossRef]
- Wishart, D.S.; Tzur, D.; Knox, C.; Eisner, R.; Guo, A.C.; Young, N.; Cheng, D.; Jewell, K.; Arndt, D.; Sawhney, S.; et al. HMDB: The Human Metabolome Database. Nucleic Acids Res. 2007, 35, D521–D526. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, E.L.; Akutsu, H.; Doreleijers, J.F.; Harano, Y.; Ioannidis, Y.E.; Lin, J.; Livny, M.; Mading, S.; Maziuk, D.; Miller, Z.; et al. BioMagResBank. Nucleic Acids Res. 2008, 36, D402–D408. [Google Scholar] [CrossRef] [PubMed]
| Control Group | September Intervention | p-Value | November Intervention | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| Mean | ± SD | Mean | ± SD | Mean | ± SD | |||
| Subjects [n] | 15 | 10 | 6 | |||||
| Males/Females [n] | 6/9 | 3/7 | 2/4 | |||||
| Height [cm] | 172.73 | 7.85 | 168.74 | 8.69 | 0.2582 | 176.3 | 8.18 | 0.3842 |
| Weight [kg] | 69.1 | 11.36 | 62.52 | 9.38 | 0.129 | 67.93 | 7.48 | 0.7875 |
| Body mass index [kg/m2] | 23.06 | 2.73 | 21.88 | 2.19 | 0.1936 | 21.82 | 0.67 | 0.36812 |
| Abdominal girth [cm] | 78.5 | 10.49 | 72.5 | 6.75 | 0.1936 | 75.58 | 7.31 | 0.56192 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
König, A.; Schwarzinger, B.; Stadlbauer, V.; Lanzerstorfer, P.; Iken, M.; Schwarzinger, C.; Kolb, P.; Schwarzinger, S.; Mörwald, K.; Brunner, S.; et al. Guava (Psidium guajava) Fruit Extract Prepared by Supercritical CO2 Extraction Inhibits Intestinal Glucose Resorption in a Double-Blind, Randomized Clinical Study. Nutrients 2019, 11, 1512. https://doi.org/10.3390/nu11071512
König A, Schwarzinger B, Stadlbauer V, Lanzerstorfer P, Iken M, Schwarzinger C, Kolb P, Schwarzinger S, Mörwald K, Brunner S, et al. Guava (Psidium guajava) Fruit Extract Prepared by Supercritical CO2 Extraction Inhibits Intestinal Glucose Resorption in a Double-Blind, Randomized Clinical Study. Nutrients. 2019; 11(7):1512. https://doi.org/10.3390/nu11071512
Chicago/Turabian StyleKönig, Alice, Bettina Schwarzinger, Verena Stadlbauer, Peter Lanzerstorfer, Marcus Iken, Clemens Schwarzinger, Peter Kolb, Stephan Schwarzinger, Katharina Mörwald, Susanne Brunner, and et al. 2019. "Guava (Psidium guajava) Fruit Extract Prepared by Supercritical CO2 Extraction Inhibits Intestinal Glucose Resorption in a Double-Blind, Randomized Clinical Study" Nutrients 11, no. 7: 1512. https://doi.org/10.3390/nu11071512
APA StyleKönig, A., Schwarzinger, B., Stadlbauer, V., Lanzerstorfer, P., Iken, M., Schwarzinger, C., Kolb, P., Schwarzinger, S., Mörwald, K., Brunner, S., Höglinger, O., Weghuber, D., & Weghuber, J. (2019). Guava (Psidium guajava) Fruit Extract Prepared by Supercritical CO2 Extraction Inhibits Intestinal Glucose Resorption in a Double-Blind, Randomized Clinical Study. Nutrients, 11(7), 1512. https://doi.org/10.3390/nu11071512





