Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption
Abstract
1. Introduction
2. Concept of Tea within an Integrative Theory of Depression
3. Antidepressant Effects of Tea Consumption in the Literature
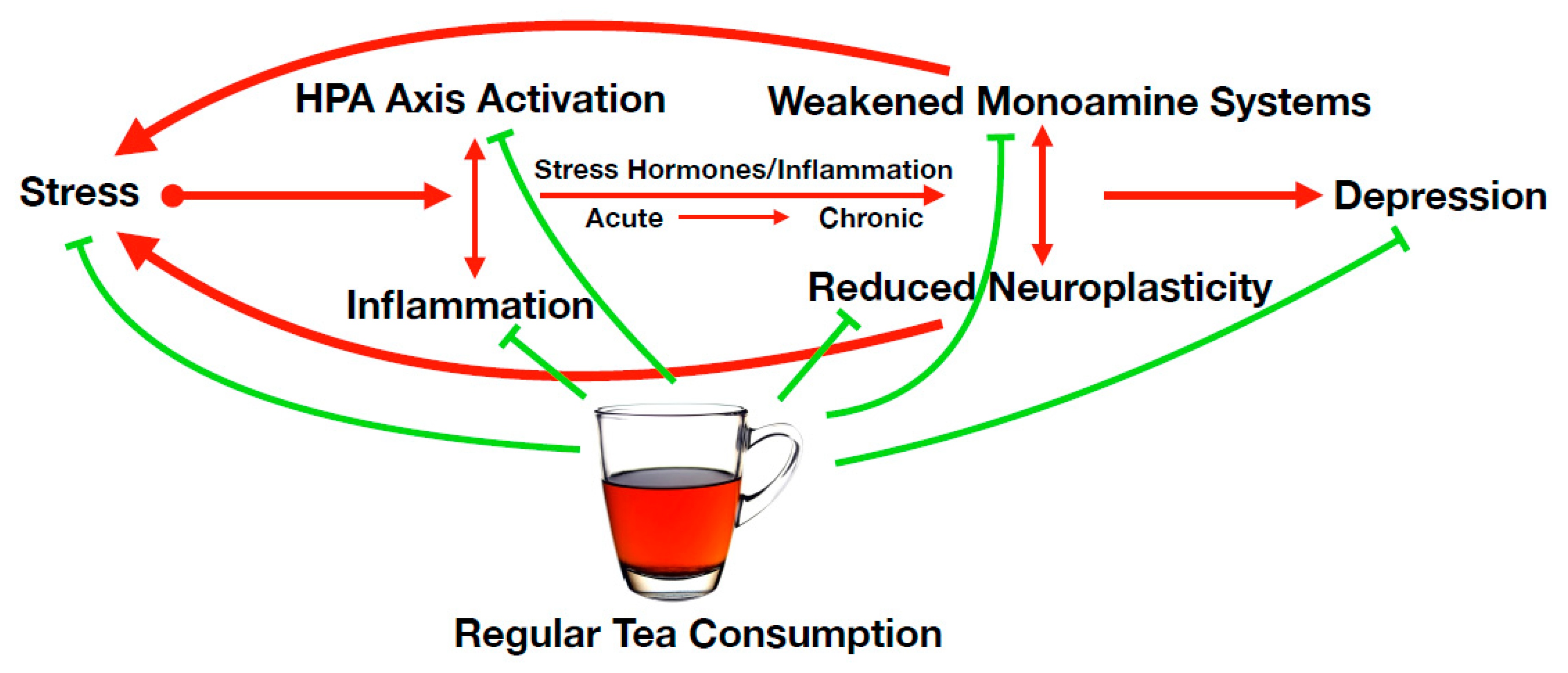
4. Mechanistic Considerations
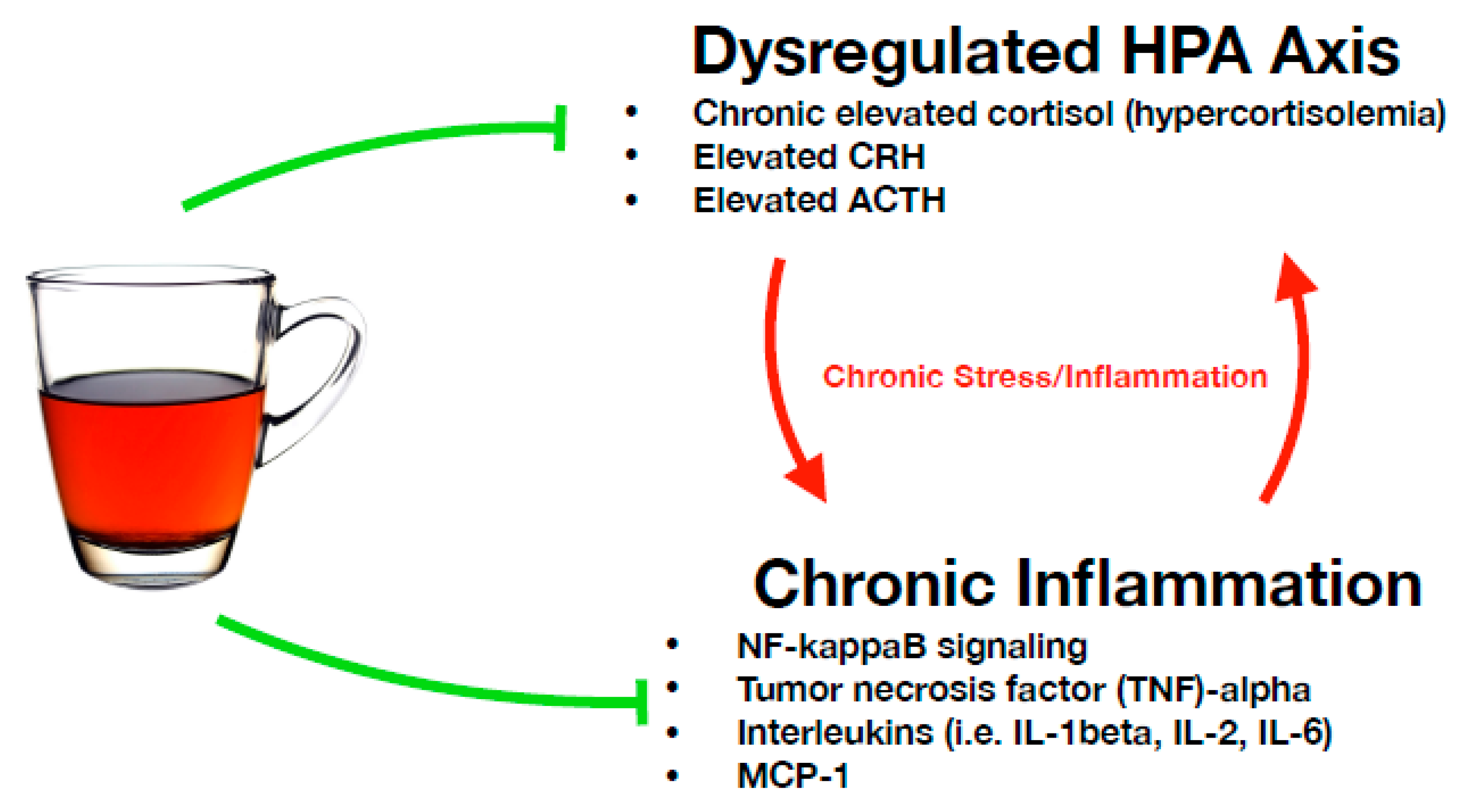
4.1. Reduction of HPA Axis Hyperactivity
4.2. Reducing Inflammation
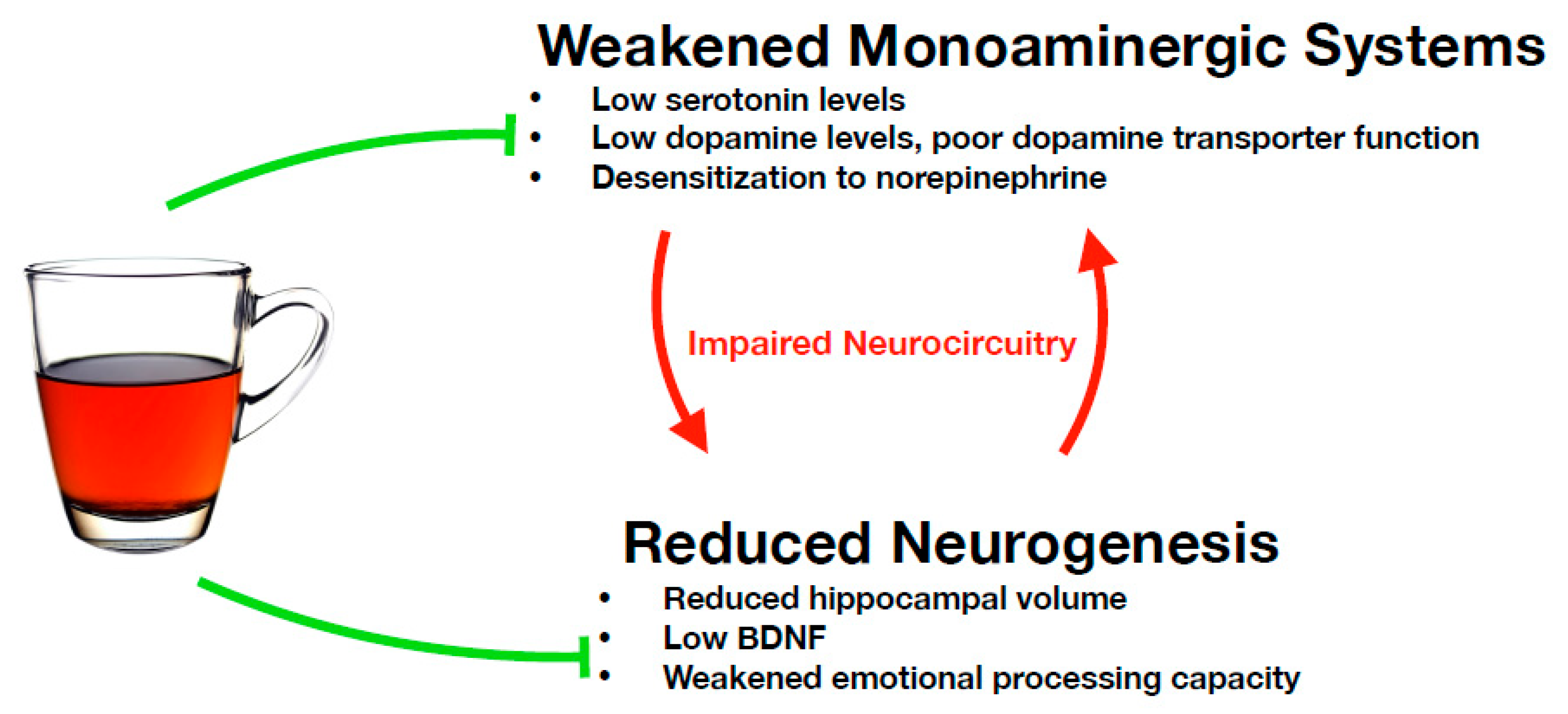
4.3. Restoration of Monoaminergic Systems
4.3.1. Monoamine Oxidase Inhibition
4.3.2. Modulating Monoamines via the Gut–Brain Axis
4.3.3. Generation of Short-Chain Fatty Acids
4.4. Restoring Neurogenesis and Neuroplasticity
4.5. Mood Enhancement
4.6. Determining Bioavailability, Pharmacokinetics and Pharmacodynamics
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Sinyor, M.; Rezmovitz, J.; Zaretsky, A. Screen all for depression. Br. Med. J. 2016, 352. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; McGongle, K.A.; Zhao, S.; Nelson, C.B.; Hughes, M.; Eshleman, S.; Wittchen, H.-U.; Kendler, K.S. Lifetime and 12-month prevalence of DSM-III-R psychiatric disorders in the United States. Arch. Gen. Psychiatry 1994, 51, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Tang, C.; Liow, C.S.; Ng, W.W.N.; Ho, C.S.H.; Ho, R.C.M. A regressional analysis of maladaptive rumination, illness perception and negative emotional outcomes in Asian patients suffering from depressive disorder. Asian J. Psychiatry 2014, 12, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Choo, C.; Diederich, J.; Song, I.; Ho, R. Cluster analysis reveals risk factors for repeated suicide attempts in a multi-ethnic Asian population. Asian J. Psychiatry 2014, 8, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Large, M. Suicide Risk and Intervention in Mental Illness Study on suicide risk assessment in mental illness underestimates inpatient suicide risk. Br. Med. J. 2016, 532, i267. [Google Scholar] [CrossRef] [PubMed]
- Cuijpers, P.; Smit, H. Excess mortality in depression: A meta-analysis of community studies. J. Affect. Disord. 2002, 72, 227–236. [Google Scholar] [CrossRef]
- Stewart, W.F.S.; Ricci, J.A.; Chee, E.; Hahn, S.R.; Morganstein, D. Cost of lost productive work time among US workers with depression. Eur. J. Public Health 2005, 15, 26. [Google Scholar]
- Daly, E.J.; Trivedi, M.H.; Wisniewski, S.R.; Nierenberg, A.A.; Gaynes, B.N.; Warden, D.; Morris, D.W.; Luther, J.F.; Farabaugh, A.; Cook, I.; et al. Health-related quality of life in depression: A STAR*D report. Ann. Clin. Psychiatry 2010, 22, 43–55. [Google Scholar]
- Mayor, S. Persistent depression doubles stroke risk despite treatment, study finds. BMJ Clin. Res. Ed. 2015, 350, h2611. [Google Scholar] [CrossRef]
- Lim, G.Y.; Tam, W.W.; Lu, Y.; Ho, C.S.; Zhang, M.W.; Ho, R.C. Prevalence of Depression in the Community from 30 Countries between 1994 and 2014. Sci. Rep. 2018, 8, 2861. [Google Scholar] [CrossRef]
- Cowen, P.J. Backing into the future: Pharmacological approaches to the management of resistant depression. Psychol. Med. 2017, 47, 2569–2577. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.E.; Khandker, R.K.; Ichikawa, L.; Operskalski, B.H. Recovery from depression predicts lower health services costs. J. Clin. Psychiatry 2006, 67, 1226–1231. [Google Scholar] [CrossRef] [PubMed]
- Kang, E.; Lee, J. A longitudinal study on the causal association between smoking and depression. J. Prev. Med. Public Health = Yebang Uihakhoe Chi 2010, 43, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, L.E.; Fiellin, D.A.; O’Connor, P.G. The prevalence and impact of alcohol problems in major depression: A systematic review. Am. J. Med. 2005, 118, 330–341. [Google Scholar] [CrossRef] [PubMed]
- Schachter, J.; Martel, J.; Lin, C.-S.; Chang, C.-J.; Wu, T.-R.; Lu, C.-C.; Ko, Y.-F.; Lai, H.-C.; Ojcius, D.M.; Young, J.D. Effects of obesity on depression: A role for inflammation and the gut microbiota. Brain Behav. Immun. 2018, 69, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dinas, P.C.; Koutedakis, Y.; Flouris, A.D. Effects of exercise and physical activity on depression. Ir. J. Med. Sci. 2011, 180, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Yang, C.; Cao, S.; Gan, Y.; Sun, H.; Gong, Y.; Yang, H.; Yin, X.; Lu, Z. Tea consumption and the risk of depression: A meta-analysis of observational studies. Aust. N. Z. J. Psychiatry 2015, 49, 334–345. [Google Scholar] [CrossRef]
- Khan, N.; Mukhtar, H. Tea and Health: Studies in Humans. Curr. Pharm. Des. 2013, 19, 6141–6147. [Google Scholar] [CrossRef]
- da Silva Pinto, M. Tea: A new perspective on health benefits. Food Res. Int. 2013, 53, 558–567. [Google Scholar] [CrossRef]
- Legeay, S.; Rodier, M.; Fillon, L.; Faure, S.; Clere, N. Epigallocatechin Gallate: A Review of Its Beneficial Properties to Prevent Metabolic Syndrome. Nutrients 2015, 7, 5443–5468. [Google Scholar] [CrossRef]
- Villas Boas, G.R.; Boerngen de Lacerda, R.; Paes, M.M.; Gubert, P.; Almeida, W.L.d.C.; Rescia, V.C.; de Carvalho, P.M.G.; de Carvalho, A.A.V.; Oesterreich, S.A. Molecular aspects of depression: A review from neurobiology to treatment. Eur. J. Pharmacol. 2019, 851, 99–121. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, F.; Villa, R.F. The Neurobiology of Depression: An Integrated Overview from Biological Theories to Clinical Evidence. Mol. Neurobiol. 2017, 54, 4847–4865. [Google Scholar] [CrossRef] [PubMed]
- Dean, J.; Keshavan, M. The neurobiology of depression: An integrated view. Asian J. Psychiatry 2017, 27, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Ruusunen, A.; Lehto, S.M.; Tolmunen, T.; Mursu, J.; Kaplan, G.A.; Voutilainen, S. Coffee, tea and caffeine intake and the risk of severe depression in middle-aged Finnish men: The Kuopio Ischaemic Heart Disease Risk Factor Study. Public Health Nutr. 2010, 13, 1215–1220. [Google Scholar] [CrossRef] [PubMed]
- Masana, M.F.; Maria Haro, J.; Mariolis, A.; Piscopo, S.; Valacchi, G.; Bountziouka, V.; Anastasiou, F.; Zeimbekis, A.; Tyrovola, D.; Gotsis, E.; et al. Mediterranean diet and depression among older individuals: The multinational MEDIS study. Exp. Gerontol. 2018, 110, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Kim, J. Green Tea, Coffee, and Caffeine Consumption Are Inversely Associated with Self-Report Lifetime Depression in the Korean Population. Nutrients 2018, 10, 1201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Y.; Yang, H.C.; Wang, J.; Li, A.W.; Zhang, W.T.; Cui, X.H.; Wang, K.L. Effect of green tea on reward learning in healthy individuals: A randomized, double-blind, placebo-controlled pilot study. Nutr. J. 2013, 12, 84. [Google Scholar] [CrossRef]
- Kendler, K.S. Toward a philosophical structure for psychiatry. Am. J. Psychiatry 2005, 162, 433–440. [Google Scholar] [CrossRef]
- Chang, S.C.; Cassidy, A.; Willett, W.C.; Rimm, E.B.; O’Reilly, E.J.; Okereke, O.I. Dietary flavonoid intake and risk of incident depression in midlife and older women. Am. J. Clin. Nutr. 2016, 104, 704–714. [Google Scholar] [CrossRef]
- Chang, X.R.; Wang, L.; Li, J.; Wu, D.S. Analysis of anti-depressant potential of curcumin against depression induced male albino wistar rats. Brain Res. 2016, 1642, 219–225. [Google Scholar] [CrossRef]
- Unno, K.; Hara, A.; Nakagawa, A.; Iguchi, K.; Ohshio, M.; Morita, A.; Nakamura, Y. Anti-stress effects of drinking green tea with lowered caffeine and enriched theanine, epigallocatechin and arginine on psychosocial stress induced adrenal hypertrophy in mice. Phytomedicine 2016, 23, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.X.; Cai, M.; Wang, T.T.; Zhao, G.S.; Huang, J.B.; Wang, H.S.; Qian, F.; Ho, C.T.; Wang, Y.J. Theanine supplementation prevents liver injury and heat shock response by normalizing hypothalamic-pituitaryadrenal axis hyperactivity in mice subjected to whole body heat stress. J. Funct. Foods 2018, 45, 181–189. [Google Scholar] [CrossRef]
- Wang, D.X.; Gao, Q.; Zhao, G.S.; Kan, Z.P.; Wang, X.X.; Wang, H.S.; Huang, J.B.; Wang, T.T.; Qian, F.; Ho, C.T.; et al. Protective Effect and Mechanism of Theanine on Lipopolysaccharide-Induced Inflammation and Acute Liver Injury in Mice. J. Agr. Food Chem. 2018, 66, 7674–7683. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.L.; Shi, H.S.; Wei, Y.M.; Wang, S.J.; Sun, C.Y.; Ding, Z.B.; Lu, L. Green tea polyphenols produce antidepressant-like effects in adult mice. Pharmacol. Res. 2012, 65, 74–80. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, F.; Jin, H.; Li, R.; Wang, Y.; Zhang, W.; Wang, H.; Chen, W. Involvement of PKC Alpha and Erk1/2 Signaling Pathways in EGCG’s Protection Against Stress-Induced Neural Injuries in Wistar Rats. Neuroscience 2017, 346, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Shim, I.; Lee, H.; Hahm, D.-H. Effects of Epigallocatechin Gallate on Behavioral and Cognitive Impairments, Hypothalamic-Pituitary-Adrenal Axis Dysfunction, and Alternations in Hippocampal BDNF Expression Under Single Prolonged Stress. J. Med. Food 2018, 21, 979–989. [Google Scholar] [CrossRef]
- Bae, J.H.; Mun, K.C.; Park, W.K.; Lee, S.R.; Suh, S.I.; Baek, W.K.; Yim, M.B.; Kwon, T.K.; Song, D.K. EGCG attenuates AMPA-induced intracellular calcium increase in hippocampal neurons. Biochem. Biophys. Res. Commun. 2002, 290, 1506–1512. [Google Scholar]
- Jeong, H.S.; Kim, Y.S.; Park, J.S. Modulation of neuronal activity by EGCG. Brain Res. 2005, 1047, 267–270. [Google Scholar] [CrossRef]
- Yi, Q.Y.; Li, H.B.; Qi, J.; Yu, X.J.; Huo, C.J.; Li, X.; Bai, J.; Gao, H.L.; Kou, B.; Liu, K.L.; et al. Chronic infusion of epigallocatechin-3-O-gallate into the hypothalamic paraventricular nucleus attenuates hypertension and sympathoexcitation by restoring neurotransmitters and cytokines. Toxicol. Lett. 2016, 262, 105–113. [Google Scholar] [CrossRef]
- Ye, Q.Y.; Ye, L.F.; Xu, X.J.; Huang, B.X.; Zhang, X.D.; Zhu, Y.G.; Chen, X.C. Epigallocatechin-3-gallate suppresses 1-methyl-4-phenyl-pyridine-induced oxidative stress in PC12 cells via the SIRT1/PGC-1 alpha signaling pathway. BMC Complement. Altern. Med. 2012, 12, 82. [Google Scholar] [CrossRef]
- Unno, K.; Noda, S.; Kawasaki, Y.; Yamada, H.; Morita, A.; Iguchi, K.; Nakamura, Y. Reduced Stress and Improved Sleep Quality Caused by Green Tea Are Associated with a Reduced Caffeine Content. Nutrients 2017, 9, 777. [Google Scholar] [CrossRef]
- Yoto, A.; Murao, S.; Nakamura, Y.; Yokogoshi, H. Intake of green tea inhibited increase of salivary chromogranin A after mental task stress loads. J. Physiol. Anthropol. 2014, 33, 20. [Google Scholar] [CrossRef] [PubMed]
- Yoto, A.; Fukui, N.; Kaneda, C.; Torita, S.; Goto, K.; Nanjo, F.; Yokogoshi, H. Black tea aroma inhibited increase of salivary chromogranin-A after arithmetic tasks. J. Physiol. Anthropol. 2018, 37. [Google Scholar] [CrossRef] [PubMed]
- Yoto, A.; Yokogoshi, H.; Moriyama, T.; Nakamura, Y.; Katsuno, T.; Nakayama, T. Effect of Smelling Green Tea Rich in Aroma Components on EEG Activity and Memory Task Performance. In Proceedings of the 2013 International Conference on Biometrics and Kansei Engineering (ICBAKE), Tokyo, Japan, 5–7 July 2013; pp. 76–81. [Google Scholar]
- Vargas, N.; Marino, F. Heat stress, gastrointestinal permeability and interleukin-6 signaling—Implications for exercise performance and fatigue. Temperature 2016, 3, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Evanson, N.K.; van Hooren, D.C.; Herman, J.P. GluR5-mediated glutamate signaling regulates hypothalamo-pituitary-adrenocortical stress responses at the paraventricular nucleus and median eminence. Psychoneuroendocrinology 2009, 34, 1370–1379. [Google Scholar] [CrossRef] [PubMed]
- Zelena, D.; Mergl, Z.; Makara, G.B. Glutamate agonists activate the hypothalamic-pituitary-adrenal axis through hypothalamic paraventricular nucleus but not through vasopressinerg neurons. Brain Res. 2005, 1031, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Kakuda, T. Neuroprotective effects of theanine and its preventive effects on cognitive dysfunction. Pharmacol. Res. 2011, 64, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Miyazaki, Y.; Unno, K.; Min, J.Z.; Todoroki, K.; Toyo’oka, T. Stable isotope dilution HILIC-MS/MS method for accurate quantification of glutamic acid, glutamine, pyroglutamic acid, GABA and theanine in mouse brain tissues. Biomed. Chromatogr. 2016, 30, 55–61. [Google Scholar] [CrossRef]
- Hyland, N.P.; Cryan, J.F. A gut feeling about GABA: Focus on GABA(B) receptors. Front. Pharmacol. 2010, 1, 124. [Google Scholar] [CrossRef]
- Unno, K.; Fujitani, K.; Takamori, N.; Takabayashi, F.; Maeda, K.; Miyazaki, H.; Tanida, N.; Iguchi, K.; Shimoi, K.; Hoshino, M. Theanine intake improves the shortened lifespan, cognitive dysfunction and behavioural depression that are induced by chronic psychosocial stress in mice. Free Radic. Res. 2011, 45, 966–974. [Google Scholar] [CrossRef]
- Unno, K.; Iguchi, K.; Tanida, N.; Fujitani, K.; Takamori, N.; Yamamoto, H.; Ishii, N.; Nagano, H.; Nagashima, T.; Hara, A.; et al. Ingestion of theanine, an amino acid in tea, suppresses psychosocial stress in mice. Exp. Physiol. 2013, 98, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, M.; Sugihara, N.; Tagashira, T.; Terao, K.; Furuno, K. Presence or absence of a gallate moiety on catechins affects their cellular transport. J. Pharm. Pharmacol. 2008, 60, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Annaba, F.; Kumar, P.; Dudeja, A.K.; Saksena, S.; Gill, R.K.; Alrefai, W.A. Green tea catechin EGCG inhibits ileal apical sodium bile acid transporter ASBT. Am. J. Physiol.-Gastrointest. Liver Physiol. 2010, 298, G467–G473. [Google Scholar] [CrossRef] [PubMed]
- Farabegoli, F.; Papi, A.; Bartolini, G.; Ostan, R.; Orlandi, M. (−)-Epigallocatechin-3-gallate downregulates Pg-P and BCRP in a tamoxifen resistant MCF-7 cell line. Phytomedicine 2010, 17, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Terashima, T.; Kawano, S.; Furuno, R.; Okubo, T.; Juneja, L.R.; Yokogoshi, H. Theanine, gamma-glutamylethylamide, a unique amino acid in tea leaves, modulates neurotransmitter concentrations in the brain striatum interstitium in conscious rats. Amino Acids 2009, 36, 21–27. [Google Scholar] [CrossRef]
- Legendre, P. The glycinergic inhibitory synapse. Cell. Mol. Life Sci. 2001, 58, 760–793. [Google Scholar] [CrossRef] [PubMed]
- Eulenburg, V.; Armsen, W.; Betz, H.; Gomeza, J. Glycine transporters: Essential regulators of neurotransmission. Trends Biochem. Sci. 2005, 30, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Yadid, G.; Pacak, K.; Golomb, E.; Harvey-White, J.D.; Lieberman, D.M.; Kopin, I.J.; Goldstein, D.S. Glycine stimulates striatal dopamine release in conscious rats. Br. J. Pharmacol. 1993, 110, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Schuster, J.; Mitchell, E.S. More than just caffeine: Psychopharmacology of methylxanthine interactions with plant-derived phytochemicals. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 89, 263–274. [Google Scholar] [CrossRef]
- Lee, H.; Bae, J.H.; Lee, S.R. Protective effect of green tea polyphenol EGCG against neuronal damage and brain edema after unilateral cerebral ischemia in gerbils. J. Neurosci. Res. 2004, 77, 892–900. [Google Scholar] [CrossRef]
- Nater, U.M.; Rohleder, N. Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: Current state of research. Psychoneuroendocrinology 2009, 34, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Almela, M.; Hidalgo, V.; Villada, C.; van der Meij, L.; Espin, L.; Gomez-Amor, J.; Salvador, A. Salivary alpha-amylase response to acute psychosocial stress: The impact of age. Biol. Psychol. 2011, 87, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Nater, U.M.; La Marca, R.; Florin, L.; Moses, A.; Langhans, W.; Koller, M.M.; Ehlert, U. Stress-induced changes in human salivary alpha-amylase activity-associations with adrenergic activity. Psychoneuroendocrinology 2006, 31, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Nater, U.M.; Rohleder, N.; Schlotz, W.; Ehlert, U.; Kirschbaum, C. Determinants of the diurnal course of salivary alpha-amylase. Psychoneuroendocrinology 2007, 32, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Lovato, N.; Gradisar, M. A meta-analysis and model of the relationship between sleep and depression in adolescents: Recommendations for future research and clinical practice. Sleep Med. Rev. 2014, 18, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Jo, K.; Hong, K.B.; Han, S.H.; Suh, H.J. GABA and l-theanine mixture decreases sleep latency and improves NREM sleep. Pharm. Biol. 2019, 57, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Daglia, M.; Di Lorenzo, A.; Nabavi, S.F.; Sureda, A.; Khanjani, S.; Moghaddam, A.H.; Braidy, N.; Nabavi, S.M. Improvement of Antioxidant Defences and Mood Status by Oral GABA Tea Administration in a Mouse Model of Post-Stroke Depression. Nutrients 2017, 9, 446. [Google Scholar] [CrossRef] [PubMed]
- Mahoney, C.R.; Giles, G.E.; Marriott, B.P.; Judelson, D.A.; Glickman, E.L.; Geiselman, P.J.; Lieberman, H.R. Intake of caffeine from all sources and reasons for use by college students. Clin. Nutr. (Edinburgh) 2019, 38, 668–675. [Google Scholar] [CrossRef]
- Unno, K.; Tanida, N.; Ishii, N.; Yamamoto, H.; Iguchi, K.; Hoshino, M.; Takeda, A.; Ozawa, H.; Ohkubo, T.; Juneja, L.R.; et al. Anti-stress effect of theanine on students during pharmacy practice: Positive correlation among salivary α-amylase activity, trait anxiety and subjective stress. Pharmacol. Biochem. Behav. 2013, 111, 128–135. [Google Scholar] [CrossRef]
- Wang, J.Q.; Mao, L. The ERK Pathway: Molecular Mechanisms and Treatment of Depression. Mol. Neurobiol. 2019. [Google Scholar] [CrossRef]
- Wang, L.; Peng, D.; Xie, B.; Jiang, K.; Fang, Y. The extracellular signal-regulated kinase pathway may play an important role in mediating antidepressant-stimulated hippocampus neurogenesis in depression. Med. Hypotheses 2012, 79, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Pahlevani, P.; Fatahi, Z.; Moradi, M.; Haghparast, A. Morphine-induced conditioned place preference and the alterations of p-ERK, p-CREB and c-fos levels in hypothalamus and hippocampus: The effects of physical stress. Cell. Mol. Biol. 2014, 60, 48–55. [Google Scholar] [PubMed]
- Dwivedi, Y.; Rizavi, H.S.; Conley, R.R.; Pandey, G.N. ERK MAP kinase signaling in post-mortem brain of suicide subjects: Differential regulation of upstream Raf kinases Raf-1 and B-Raf. Mol. Psychiatry 2006, 11, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Zhang, J.; Liu, G.; Chen, F.; Lin, Y. Neuroprotection by (-)-epigallocatechin-3-gallate in a rat model of stroke is mediated through inhibition of endoplasmic reticulum stress. Mol. Med. Rep. 2014, 9, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Johnston, G.A. Flavonoid nutraceuticals and ionotropic receptors for the inhibitory neurotransmitter GABA. Neurochem. Int. 2015, 89, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Alho, H.; Costa, E.; Ferrero, P.; Fujimoto, M.; Cosenza-Murphy, D.; Guidotti, A. Diazepam-binding inhibitor: A neuropeptide located in selected neuronal populations of rat brain. Science (New York) 1985, 229, 179–182. [Google Scholar] [CrossRef]
- Chang, K.-C.; Yang, J.J.; Wang-Hsu, E.C.-H.; Chiu, T.-H.; Hsu, F.-C. (-)Epigallocatechin-3-gallate inhibits the spontaneous firing of rat locus coeruleus neuron. Neurosci. Lett. 2009, 452, 141–145. [Google Scholar] [CrossRef]
- Lee, M.-S.; Lee, S.; Doo, M.; Kim, Y. Green Tea (-)-Epigallotocatechin-3-Gallate Induces PGC-1alpha Gene Expression in HepG2 Cells and 3T3-L1 Adipocytes. Prev. Nutr. Food Sci. 2016, 21, 62–67. [Google Scholar] [CrossRef]
- Zhan, G.; Huang, N.; Li, S.; Hua, D.; Zhang, J.; Fang, X.; Yang, N.; Luo, A.; Yang, C. PGC-1 alpha-FNDC5-BDNF signaling pathway in skeletal muscle confers resilience to stress in mice subjected to chronic social defeat. Psychopharmacology 2018, 235, 3351–3358. [Google Scholar] [CrossRef]
- Cunha, M.P.; Pazini, F.L.; Lieberknecht, V.; Rodrigues, A.L.S. Subchronic administration of creatine produces antidepressant-like effect by modulating hippocampal signaling pathway mediated by FNDC5/BDNF/Akt in mice. J. Psychiatr. Res. 2018, 104, 78–87. [Google Scholar] [CrossRef]
- Lucas, E.K.; Dougherty, S.E.; McMeekin, L.J.; Reid, C.S.; Dobrunz, L.E.; West, A.B.; Hablitz, J.J.; Cowell, R.M. PGC-1 alpha Provides a Transcriptional Framework for Synchronous Neurotransmitter Release from Parvalbumin-Positive Interneurons. J. Neurosci. 2014, 34, 14375–14387. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, S.E.; Bartley, A.F.; Lucas, E.K.; Hablitz, J.J.; Dobrunz, L.E.; Cowell, R.M. Mice Lacking the Transcriptional Coactivator Pgc-1 Alpha Exhibit Alterations in Inhibitory Synaptic Transmission in the Motor Cortex. Neuroscience 2014, 271, 137–148. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Felger, J.C.; Lotrich, F.E. Inflammatory Cytokines in Depression: Neurobiological Mechanisms and Therapeutic Implications. Neuroscience 2013, 246, 199–229. [Google Scholar] [CrossRef] [PubMed]
- Leonard, B.E. Inflammation and depression: A causal or coincidental link to the pathophysiology? Acta Neuropsychiatr. 2018, 30, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Tanaka, M.; Kinney, D.K. Depression as an evolutionary strategy for defense against infection. Brain Behav. Immun. 2013, 31, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Rosenblat, J.D.; Cha, D.S.; Mansur, R.B.; McIntyre, R.S. Inflamed moods: A review of the interactions between inflammation and mood disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014, 53, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.Q.; Wang, W.; Peng, Y.; Deng, Z.F. Antidepressive effects of kaempferol mediated by reduction of oxidative stress, proinflammatory cytokines and up-regulation of AKT/beta-catenin cascade. Metab. Brain Dis. 2019, 34, 485–494. [Google Scholar] [CrossRef]
- Xu, Y.; Lin, W.J. Anti-inflammatory Cytokines and Depression. Prog. Biochem. Biophys. 2014, 41, 1099–1108. [Google Scholar]
- Skaper, S.D.; Facci, L.; Zusso, M.; Giusti, P. An Inflammation-Centric View of Neurological Disease: Beyond the Neuron. Front. Cell. Neurosci. 2018, 12, 72. [Google Scholar] [CrossRef]
- Ano, Y.; Ohya, R.; Kita, M.; Taniguchi, Y.; Kondo, K. Theaflavins Improve Memory Impairment and Depression-Like Behavior by Regulating Microglial Activation. Molecules 2019, 24, 467. [Google Scholar] [CrossRef]
- Wu, Y.T.; Jin, F.J.; Wang, Y.L.; Li, F.; Wang, L.; Wang, Q.L.; Ren, Z.; Wang, Y.F. In vitro and in vivo anti-inflammatory effects of theaflavin-3,3‘-digallate on lipopolysaccharide-induced inflammation. Eur. J. Pharmacol. 2017, 794, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.J.; Lo, C.Y.; Wang, B.J.; Chiou, R.Y.Y.; Lin, S.M. Theaflavin-3,3′-digallate, a black tea polyphenol, attenuates adipocyte-activated inflammatory response of macrophage associated with the switch of M1/M2-like phenotype. J. Funct. Foods 2014, 11, 36–48. [Google Scholar] [CrossRef]
- Li, J.H.; Sasaki, G.Y.; Dey, P.; Chitchumroonchokchai, C.; Labyk, A.N.; McDonald, J.D.; Kim, J.B.; Bruno, R.S. Green tea extract protects against hepatic NF kappa B activation along the gut-liver axis in diet-induced obese mice with nonalcoholic steatohepatitis by reducing endotoxin and TLR4/MyD88 signaling. J. Nutr. Biochem. 2018, 53, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.S.; Qi, G.Y.; Fan, R.; Qiao, Q.L.; Sun, Y.L.; Gao, Y.Q.; Liu, X.B. EGCG ameliorates high-fat- and high-fructose-induced cognitive defects by regulating the IRS/AKT and ERK/CREB/BDNF signaling pathways in the CNS. FASEB J. 2017, 31, 4998–5011. [Google Scholar] [CrossRef] [PubMed]
- Fechtner, S.; Singh, A.; Chourasia, M.; Ahmed, S. Molecular insights into the differences in anti-inflammatory activities of green tea catechins on IL-1 beta signaling in rheumatoid arthritis synovial fibroblasts. Toxicol. Appl. Pharmacol. 2017, 329, 112–120. [Google Scholar] [CrossRef]
- Liang, Y.; Ip, M.S.M.; Mak, J.C.W. (-)-Epigallocatechin-3-gallate suppresses cigarette smoke-induced inflammation in human cardiomyocytes via ROS-mediated MAPK and NF-kappaB pathways. Phytomed. Int. J. Phytother. Phytopharmacol. 2019, 58, 152768. [Google Scholar]
- Li, K.K.; Peng, J.M.; Zhu, W.; Cheng, B.H.; Li, C.M. Gallocatechin gallate (GCG) inhibits 3T3-L1 differentiation and lipopolysaccharide induced inflammation through MAPK and NF-kappa B signaling. J. Funct. Foods 2017, 30, 159–167. [Google Scholar] [CrossRef]
- Novilla, A.; Djamhuri, D.S.; Nurhayati, B.; Rihibiha, D.D.; Afifah, E.; Widowati, W. Anti-inflammatory properties of oolong tea (Camellia sinensis) ethanol extract and epigallocatechin gallate in LPS-induced RAW 264.7 cells. Asian Pac. J. Trop. Biomed. 2017, 7, 1005–1009. [Google Scholar] [CrossRef]
- Seok, J.K.; Lee, J.W.; Kim, Y.M.; Boo, Y.C. Punicalagin and (-)-Epigallocatechin-3-Gallate Rescue Cell Viability and Attenuate Inflammatory Responses of Human Epidermal Keratinocytes Exposed to Airborne Particulate Matter PM10. Skin Pharmacol. Physiol. 2018, 31, 134–143. [Google Scholar] [CrossRef]
- Yoon, J.Y.; Kwon, H.H.; Min, S.U.; Thiboutot, D.M.; Suh, D.H. Epigallocatechin-3-Gallate Improves Acne in Humans by Modulating Intracellular Molecular Targets and Inhibiting P. acnes. J. Investig. Dermatol. 2013, 133, 429–440. [Google Scholar] [CrossRef]
- Fan, Y.J.; Piao, C.H.; Hyeon, E.; Jung, S.Y.; Eoin, J.E.; Shin, H.S.; Song, C.H.; Chai, O.H. Gallic acid alleviates nasal inflammation via activation of Th1 and inhibition of Th2 and Th17 in a mouse model of allergic rhinitis. Int. Immunopharmacol. 2019, 70, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.J.; Tan, Z.; Lai, X.F.; Xu, Y.N.; Mai, C.L.; Zhang, J.; Lin, Z.J.; Liu, X.G.; Sun, S.L.; Zhou, L.J. Topical delivery of L-theanine ameliorates TPA-induced acute skin inflammation via downregulating endothelial PECAM-1 and neutrophil infiltration and activation. Chem.-Biol. Interact. 2018, 284, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Jin, S.W.; Choi, J.H.; Choi, C.Y.; Kim, H.G.; Kim, S.J.; Kim, Y.; Lee, K.J.; Chung, Y.C.; Jeong, H.G. Inhibitory effects of L-theanine on airway inflammation in ovalbumin-induced allergic asthma. Food Chem. Toxicol. 2017, 99, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Huang, X.F.; Zhang, P.; Newell, K.A.; Wang, H.; Zheng, K.; Yu, Y. Dietary teasaponin ameliorates alteration of gut microbiota and cognitive decline in diet-induced obese mice. Sci. Rep. 2017, 7, 12203. [Google Scholar] [CrossRef] [PubMed]
- Hinz, M.; Stein, A.; Uncini, T. The discrediting of the monoamine hypothesis. Int. J. Gen. Med. 2012, 5, 135–142. [Google Scholar] [CrossRef]
- Clevenger, S.S.; Malhotra, D.; Dang, J.; Vanle, B.; IsHak, W.W. The role of selective serotonin reuptake inhibitors in preventing relapse of major depressive disorder. Ther. Adv. Psychopharmacol. 2018, 8, 49–58. [Google Scholar] [CrossRef]
- Brezun, J.M.; Daszuta, A. Serotonergic reinnervation reverses lesion-induced decreases in PSA-NCAM labeling and proliferation of hippocampal cells in adult rats. Hippocampus 2000, 10, 37–46. [Google Scholar] [CrossRef]
- Nestler, E.J.; Carlezon, W.A. The mesolimbic dopamine reward circuit in depression. Biol. Psychiatry 2006, 59, 1151–1159. [Google Scholar] [CrossRef]
- El Mansari, M.; Guiard, B.P.; Chernoloz, O.; Ghanbari, R.; Katz, N.; Blier, P. Relevance of Norepinephrine-Dopamine Interactions in the Treatment of Major Depressive Disorder. CNS Neurosci. Ther. 2010, 16, e1–e17. [Google Scholar] [CrossRef]
- Shen, M.J.; Yang, Y.; Wu, Y.; Zhang, B.B.; Wu, H.S.; Wang, L.; Tang, H.; Chen, J.D. L-theanine ameliorate depressive-like behavior in a chronic unpredictable mild stress rat model via modulating the monoamine levels in limbic-cortical-striatal-pallidal-thalamic-circuit related brain regions. Phytother. Res. 2019, 33, 412–421. [Google Scholar] [CrossRef]
- Zhu, G.; Yang, S.; Xie, Z.; Wan, X. Synaptic modification by L-theanine, a natural constituent in green tea, rescues the impairment of hippocampal long-term potentiation and memory in AD mice. Neuropharmacology 2018, 138, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Ikram, H.; Mirza, B.; Haleem, D.J. Neurochemical and behavioral effects of green tea (Camellia sinensis) as observed in animals exposed to restraint stress. Pak. J. Pharm. Sci. 2017, 30, 487–492. [Google Scholar] [PubMed]
- Stringer, T.P.; Guerrieri, D.; Vivar, C.; van Praag, H. Plant-derived flavanol (-)epicatechin mitigates anxiety in association with elevated hippocampal monoamine and BDNF levels, but does not influence pattern separation in mice. Transl. Psychiatry 2015, 5, e493. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jia, G.G.; Gou, L.S.; Sun, L.Y.; Fu, X.B.; Lan, N.; Li, S.; Yin, X.X. Antidepressant-like effects of tea polyphenols on mouse model of chronic unpredictable mild stress. Pharmacol. Biochem. Behav. 2013, 104, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Janssens, P.L.H.R.; Penders, J.; Hursel, R.; Budding, A.E.; Savelkoul, P.H.M.; Westerterp-Plantenga, M.S. Long-Term Green Tea Supplementation Does Not Change the Human Gut Microbiota. PLoS ONE 2016, 11, e0153134. [Google Scholar] [CrossRef] [PubMed]
- Henning, S.M.; Yang, J.; Hsu, M.; Lee, R.P.; Grojean, E.M.; Ly, A.; Tseng, C.H.; Heber, D.; Li, Z. Decaffeinated green and black tea polyphenols decrease weight gain and alter microbiome populations and function in diet-induced obese mice. Eur. J. Nutr. 2018, 57, 2759–2769. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhu, X.L.; Sun, Y.K.; Hu, B.; Sun, Y.; Jabbar, S.; Zeng, X.X. Fermentation in vitro of EGCG, GCG and EGCG3” Me isolated from Oolong tea by human intestinal microbiota. Food Res. Int. 2013, 54, 1589–1595. [Google Scholar] [CrossRef]
- Bustos, I.; Garcia-Cayuela, T.; Hernandez-Ledesma, B.; Pelaez, C.; Requena, T.; Martinez-Cuesta, M.C. Effect of flavan-3-ols on the adhesion of potential probiotic lactobacilli to intestinal cells. J. Agric. Food Chem. 2012, 60, 9082–9088. [Google Scholar] [CrossRef] [PubMed]
- Kita, M.; Uchida, S.; Yamada, K.; Ano, Y. Anxiolytic effects of theaflavins via dopaminergic activation in the frontal cortex. Biosci. Biotechnol. Biochem. 2019, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Anandhan, A.; Janakiraman, U.; Manivasagam, T. Theaflavin ameliorates behavioral deficits, biochemical indices and monoamine transporters expression against subacute 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced mouse model of Parkinson’s disease. Neuroscience 2012, 218, 257–267. [Google Scholar] [CrossRef]
- Tzounis, X.; Vulevic, J.; Kuhnle, G.G.C.; George, T.; Leonczak, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P.E. Flavanol monomer-induced changes to the human faecal microflora. Br. J. Nutr. 2008, 99, 782–792. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Zhang, X.; Miao, Y.; Cao, J.; Wu, Z.; Weng, P. The modulatory effect of (-)-epigallocatechin 3-O-(3-O-methyl) gallate (EGCG3”Me) on intestinal microbiota of high fat diet-induced obesity mice model. Food Res. Int. 2017, 92, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Drevets, W.C.; Price, J.L.; Furey, M.L. Brain structural and functional abnormalities in mood disorders: Implications for neurocircuitry models of depression. Brain Struct. Funct. 2008, 213, 93–118. [Google Scholar] [CrossRef] [PubMed]
- Hamon, M.; Blier, P. Monoamine neurocircuitry in depression and strategies for new treatments. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2013, 45, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Henn, F.A.; Vollmayr, B. Stress models of depression: Forming genetically vulnerable strains. Neurosci. Biobehav. Rev. 2005, 29, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Zang, L.Y.; Misra, H.P. Generation of reactive oxygen species during the monoamine oxidase-catalyzed oxidation of the neurotoxicant, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. J. Biol. Chem. 1993, 268, 16504–16512. [Google Scholar]
- Khan, H.; Perviz, S.; Sureda, A.; Nabavi, S.M.; Tejada, S. Current standing of plant derived flavonoids as an antidepressant. Food Chem. Toxicol. 2018, 119, 176–188. [Google Scholar] [CrossRef] [PubMed]
- Martins, J.; Brijesh, S. Phytochemistry and pharmacology of anti-depressant medicinal plants: A review. Biomed. Pharmacother. 2018, 104, 343–365. [Google Scholar] [CrossRef]
- Kolla, N.J.; Chiuccariello, L.; Wilson, A.A.; Houle, S.; Links, P.; Bagby, R.M.; McMain, S.; Kellow, C.; Patel, J.; Rekkas, P.V.; et al. Elevated Monoamine Oxidase-A Distribution Volume in Borderline Personality Disorder Is Associated with Severity Across Mood Symptoms, Suicidality, and Cognition. Biol. Psychiatry 2016, 79, 117–126. [Google Scholar] [CrossRef]
- Bandaruk, Y.; Mukai, R.; Kawamura, T.; Nemoto, H.; Terao, J. Evaluation of the inhibitory effects of quercetin-related flavonoids and tea catechins on the monoamine oxidase-A reaction in mouse brain mitochondria. J. Agric. Food Chem. 2012, 60, 10270–10277. [Google Scholar] [CrossRef]
- Mazzio, E.; Deiab, S.; Park, K.; Soliman, K.F.A. High throughput Screening to Identify Natural Human Monoamine Oxidase B Inhibitors. Phytother. Res. 2013, 27, 818–828. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.M.; Wang, S.W.; Ho, S.C.; Tang, Y.L. Protective effect of green tea (-)-epigallocatechin-3-gallate against the monoamine oxidase B enzyme activity increase in adult rat brains. Nutrition 2010, 26, 1195–1200. [Google Scholar] [CrossRef] [PubMed]
- Ademosun, A.O.; Oboh, G. Comparison of the inhibition of monoamine oxidase and butyrylcholinesterase activities by infusions from green tea and some citrus peels. Int. J. Alzheimers Dis. 2014, 2014, 586407. [Google Scholar] [CrossRef] [PubMed]
- Levites, Y.; Weinreb, O.; Maor, G.; Youdim, M.B.H.; Mandel, S. Green tea polyphenol (-)-epigallocatechin-3-gallate prevents N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced dopaminergic neurodegeneration. J. Neurochem. 2001, 78, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Cryan, J.F. The Microbiome-Gut-Brain Axis in Health and Disease. Gastroenterol. Clin. N. Am. 2017, 46, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Stilling, R.M.; Stanton, C.; Cryan, J.F. Collective unconscious: How gut microbes shape human behavior. J. Psychiatr. Res. 2015, 63, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Cryan, J.F. Mood by microbe: Towards clinical translation. Genome Med. 2016, 8, 36. [Google Scholar] [CrossRef]
- Sudo, N.; Chida, Y.; Kubo, C. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J. Psychosom. Res. 2005, 58, S60. [Google Scholar] [CrossRef]
- Neufeld, K.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 2011, 23, 255–264. [Google Scholar] [CrossRef]
- Heijtza, R.D.; Wang, S.G.; Anuar, F.; Qian, Y.; Bjorkholm, B.; Samuelsson, A.; Hibberd, M.L.; Forssberg, H.; Pettersson, S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 3047–3052. [Google Scholar] [CrossRef]
- Lyte, M.; Li, W.; Opitz, N.; Gaykema, R.P.A.; Goehler, L.E. Induction of anxiety-like behavior in mice during the initial stages of infection with the agent of murine colonic hyperplasia Citrobacter rodentium. Physiol. Behav. 2006, 89, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Luczynski, P.; Neufeld, K.A.M.; Oriach, C.S.; Clarke, G.; Dinan, T.G.; Cryan, J.F. Growing up in a Bubble: Using Germ-Free Animals to Assess the Influence of the Gut Microbiota on Brain and Behavior. Int. J. Neuropsychopharmacol. 2016, 19, pyw020. [Google Scholar] [CrossRef] [PubMed]
- Bercik, P.; Denou, E.; Collins, J.; Jackson, W.; Lu, J.; Jury, J.; Deng, Y.K.; Blennerhassett, P.; Macri, J.; McCoy, K.D.; et al. The Intestinal Microbiota Affect Central Levels of Brain-Derived Neurotropic Factor and Behavior in Mice. Gastroenterology 2011, 141, U599–U609. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.; Grenham, S.; Scully, P.; Fitzgerald, P.; Moloney, R.D.; Shanahan, F.; Dinan, T.G.; Cryan, J.F. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol. Psychiatry 2013, 18, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.C.; Jenner, A.M.; Low, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, P.J.; Murphy, A.B.; Cryan, J.F.; Ross, P.R.; Dinan, T.G.; Stanton, C. Microbiome in brain function and mental health. Trends Food Sci. Technol. 2016, 57, 289–301. [Google Scholar] [CrossRef]
- Duda-Chodak, A.; Tarko, T.; Satora, P.; Sroka, P. Interaction of dietary compounds, especially polyphenols, with the intestinal microbiota: A review. Eur. J. Nutr. 2015, 54, 325–341. [Google Scholar] [CrossRef] [PubMed]
- Okubo, T.; Ishihara, N.; Oura, A.; Serit, M.; Kim, M.; Yamamoto, T.; Mitsuoka, T. In Vivo Effects of Tea Polyphenol Intake on Human Intestinal Microflora and Metabolism. Biosci. Biotechnol. Biochem. 1992, 56, 588–591. [Google Scholar] [CrossRef]
- Yates, A.A.; Erdman, J.W., Jr.; Shao, A.; Dolan, L.C.; Griffiths, J.C. Bioactive nutrients—Time for tolerable upper intake levels to address safety. Regul. Toxicol. Pharmacol. 2017, 84, 94–101. [Google Scholar] [CrossRef]
- Axling, U.; Olsson, C.; Xu, J.; Fernandez, C.; Larsson, S.; Strom, K.; Ahrne, S.; Holm, C.; Molin, G.; Berger, K. Green tea powder and Lactobacillus plantarum affect gut microbiota, lipid metabolism and inflammation in high-fat fed C57BL/6J mice. Nutr. Metab. (London) 2012, 9, 105. [Google Scholar] [CrossRef]
- Allen, A.P.; Hutch, W.; Borre, Y.E.; Kennedy, P.J.; Temko, A.; Boylan, G.; Murphy, E.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Bifidobacterium longum 1714 as a translational psychobiotic: Modulation of stress, electrophysiology and neurocognition in healthy volunteers. Transl. Psychiatry 2016, 6, e939. [Google Scholar] [CrossRef] [PubMed]
- Hara, Y. Influence of tea catechins on the digestive tract. J. Cell. Biochem. 1997, 27, 52–58. [Google Scholar] [CrossRef]
- Barrett, E.; Ross, R.P.; O’Toole, P.W.; Fitzgerald, G.F.; Stanton, C. Gamma-Aminobutyric acid production by culturable bacteria from the human intestine. J. Appl. Microbiol. 2012, 113, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.W.; Liong, M.T.; Tsai, Y.C. New perspectives of Lactobacillus plantarum as a probiotic: The gut-heart-brain axis. J. Microbiol. 2018, 56, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Strandwitz, P.; Kim, K.H.; Terekhova, D.; Liu, J.K.; Sharma, A.; Levering, J.; McDonald, D.; Dietrich, D.; Ramadhar, T.R.; Lekbua, A.; et al. GABA-modulating bacteria of the human gut microbiota. Nat. Microbiol. 2019, 4, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Etxeberria, U.; Fernandez-Quintela, A.; Milagro, F.I.; Aguirre, L.; Martinez, J.A.; Portillo, M.P. Impact of polyphenols and polyphenol-rich dietary sources on gut microbiota composition. J. Agric. Food Chem. 2013, 61, 9517–9533. [Google Scholar] [CrossRef] [PubMed]
- dos Santos Pereira Indiani, C.M.; Rizzardi, K.F.; Castelo, P.M.; Caldas Ferraz, L.F.; Darrieux, M.; Parisotto, T.M. Childhood Obesity and Firmicutes/Bacteroidetes Ratio in the Gut Microbiota: A Systematic Review. Child. Obes. 2018, 14, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.E.; Myslicki, J.P.; Bomhof, M.R.; Belke, D.D.; Shearer, J.; Reimer, R.A. Exercise training modifies gut microbiota in normal and diabetic mice. Appl. Physiol. Nutr. Metab. 2015, 40, 749–752. [Google Scholar] [CrossRef]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef]
- Rothenberg, D.O.; Zhou, C.B.; Zhang, L.Y. A Review on the Weight-Loss Effects of Oxidized Tea Polyphenols. Molecules 2018, 23, 1176. [Google Scholar] [CrossRef] [PubMed]
- Frost, G.; Sleeth, M.L.; Sahuri-Arisoylu, M.; Lizarbe, B.; Cerdan, S.; Brody, L.; Anastasovska, J.; Ghourab, S.; Hankir, M.; Zhang, S.; et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 2014, 5, 3611. [Google Scholar] [CrossRef] [PubMed]
- Vijay, N.; Morris, M.E. Role of monocarboxylate transporters in drug delivery to the brain. Curr. Pharm. Des. 2014, 20, 1487–1498. [Google Scholar] [CrossRef] [PubMed]
- van de Wouw, M.; Boehme, M.; Lyte, J.M.; Wiley, N.; Strain, C.; O’Sullivan, O.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Short-chain fatty acids: Microbial metabolites that alleviate stress-induced brain-gut axis alterations. J. Physiol.-Lond. 2018, 596, 4923–4944. [Google Scholar] [CrossRef] [PubMed]
- Stilling, R.M.; van de Wouw, M.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. The neuropharmacology of butyrate: The bread and butter of the microbiota-gut-brain axis? Neurochem. Int. 2016, 99, 110–132. [Google Scholar] [CrossRef] [PubMed]
- Odaira, T.; Nakagawasai, O.; Takahashi, K.; Nemoto, W.; Sakuma, W.; Lin, J.-R.; Tan-No, K. Mechanisms underpinning AMP-activated protein kinase-related effects on behavior and hippocampal neurogenesis in an animal model of depression. Neuropharmacology 2019, 150, 121–133. [Google Scholar] [CrossRef]
- Bremner, J.D.; Narayan, M.; Anderson, E.R.; Staib, L.H.; Miller, H.L.; Charney, D.S. Hippocampal volume reduction in major depression. Am. J. Psychiatry 2000, 157, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Sheline, Y.I. Neuroimaging studies of mood disorder effects on the brain. Biol. Psychiatry 2003, 54, 338–352. [Google Scholar] [CrossRef]
- Andrade, J.P.; Rodrigues, J.; Cardoso, A. Protective effects of chronic green tea consumption on dendritic trees during ageing. Agro Food Ind. Hi-Tech 2013, 24, 6–9. [Google Scholar]
- Assuncao, M.; Santos-Marques, M.J.; Carvalho, F.; Andrade, J.P. Green tea averts age-dependent decline of hippocampal signaling systems related to antioxidant defenses and survival. Free Radic. Biol. Med. 2010, 48, 831–838. [Google Scholar] [CrossRef]
- Assuncao, M.; Santos-Marques, M.J.; Carvalho, F.; Lukoyanov, N.V.; Andrade, J.P. Chronic green tea consumption prevents age-related changes in rat hippocampal formation. Neurobiol. Aging 2011, 32, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Qian, X.; Xing, J.; Wang, J.; Sun, Y.; Wang, Q.G.; Li, H. Particulate Matter Triggers Depressive-Like Response Associated with Modulation of Inflammatory Cytokine Homeostasis and Brain-Derived Neurotrophic Factor Signaling Pathway in Mice. Toxicol. Sci. 2018, 164, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Dugan, L.L.; Sensi, S.L.; Canzoniero, L.M.T.; Handran, S.D.; Rotham, S.M.; Lin, T.S.; Goldberg, M.P.; Choi, D.W. Mitochondrial production of reactive oxygen species in cortical neurons following exposure to N-methyl-D-aspartate. J. Neurosci. 1995, 15, 6377–6388. [Google Scholar] [CrossRef] [PubMed]
- Black, C.N.; Bot, M.; Scheffer, P.G.; Cuijpers, P.; Penninx, B.W. Is depression associated with increased oxidative stress? A systematic review and meta-analysis. Psychoneuroendocrinology 2015, 51, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Mayberg, H.S. Positron emission tomography imaging in depression: A neural systems perspective. Neuroimaging Clin. N. Am. 2003, 13, 805–815. [Google Scholar] [CrossRef]
- Wang, L.H.; Labar, K.S.; Smoski, M.; Rosenthal, M.Z.; Dolcos, F.; Lynch, T.R.; Krishnan, R.R.; McCarthy, G. Prefrontal mechanisms for executive control over emotional distraction are altered in major depression. Psychiatry Res.-Neuroimaging 2008, 163, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Groenewold, N.A.; Opmeer, E.M.; de Jonge, P.; Aleman, A.; Costafreda, S.G. Emotional valence modulates brain functional abnormalities in depression: Evidence from a meta-analysis of fMRI studies. Neurosci. Biobehav. Rev. 2013, 37, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Santarelli, L.; Saxe, M.; Gross, C.; Surget, A.; Battaglia, F.; Dulawa, S.; Weisstaub, N.; Lee, J.; Duman, R.; Arancio, O.; et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 2003, 301, 805–809. [Google Scholar] [CrossRef]
- Zagrebelsky, M.; Korte, M. Form follows function: BDNF and its involvement in sculpting the function and structure of synapses. Neuropharmacology 2014, 76, 628–638. [Google Scholar] [CrossRef]
- Monteleone, P.; Serritella, C.; Martiadis, V.; Maj, M. Decreased levels of serum brain-derived neurotrophic factor in both depressed and euthymic patients with unipolar depression and in euthymic patients with bipolar I and II disorders. Bipolar Disord. 2008, 10, 95–100. [Google Scholar] [CrossRef]
- Taliaz, D.; Stall, N.; Dar, D.E.; Zangen, A. Knockdown of brain-derived neurotrophic factor in specific brain sites precipitates behaviors associated with depression and reduces neurogenesis. Mol. Psychiatry 2010, 15, 80–92. [Google Scholar] [CrossRef] [PubMed]
- Nitta, A.; Ohmiya, M.; Sometani, A.; Itoh, M.; Nomoto, H.; Furukawa, Y.; Furukawa, S. Brain-derived neurotrophic factor prevents neuronal cell death induced by corticosterone. J. Neurosci. Res. 1999, 57, 227–235. [Google Scholar] [CrossRef]
- Lee, J.; Duan, W.; Mattson, M.P. Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J. Neurochem. 2002, 82, 1367–1375. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, E.; Hashimoto, K.; Iyo, M. Major depressive disorders and BDNF (brain-derived neurotrophic factor). Nihon Shinkei Seishin Yakurigaku Zasshi = Jpn. J. Psychopharmacol. 2004, 24, 147–150. [Google Scholar]
- Duman, R.S.; Monteggia, L.M. A neurotrophic model for stress-related mood disorders. Biol. Psychiatry 2006, 59, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Shimizu, E.; Iyo, M. Critical role of brain-derived neurotrophic factor in mood disorders. Brain Res. Rev. 2004, 45, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Sakamoto, K.; Tamano, H.; Fukura, K.; Inui, N.; Suh, S.W.; Won, S.-J.; Yokogoshi, H. Facilitated Neurogenesis in the Developing Hippocampus After Intake of Theanine, an Amino Acid in Tea Leaves, and Object Recognition Memory. Cell. Mol. Neurobiol. 2011, 31, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Lopez, L.; Marquez-Valadez, B.; Gomez-Sanchez, A.; Silva-Lucero, M.D.C.; Torres-Perez, M.; Tellez-Ballesteros, R.I.; Ichwan, M.; Meraz-Rios, M.A.; Kempermann, G.; Ramirez-Rodriguez, G.B. Green Tea Compound Epigallo-Catechin-3-Gallate (Egcg) Increases Neuronal Survival in Adult Hippocampal Neurogenesis in Vivo and in Vitro. Neuroscience 2016, 322, 208–220. [Google Scholar] [CrossRef]
- Gundimeda, U.; McNeill, T.H.; Fan, T.K.; Deng, R.; Rayudu, D.; Chen, Z.; Cadenas, E.; Gopalakrishna, R. Green tea catechins potentiate the neuritogenic action of brain-derived neurotrophic factor: Role of 67-kDa laminin receptor and hydrogen peroxide. Biochem. Biophys. Res. Commun. 2014, 445, 218–224. [Google Scholar] [CrossRef]
- Feng, S.; Liu, J.; Cheng, B.; Deng, A.P.; Zhang, H. (-)-Epigallocatechin-3-gallate protects PC12 cells against corticosterone-induced neurotoxicity via the hedgehog signaling pathway. Exp. Ther. Med. 2018, 15, 4284–4290. [Google Scholar] [CrossRef]
- Pervin, M.; Unno, K.; Nakagawa, A.; Takahashi, Y.; Iguchi, K.; Yamamoto, H.; Hoshino, M.; Hara, A.; Takagaki, A.; Nanjo, F.; et al. Blood brain barrier permeability of (-)-epigallocatechin gallate, its proliferation-enhancing activity of human neuroblastoma SH-SY5Y cells, and its preventive effect on age-related cognitive dysfunction in mice. Biochem. Biophys. Rep. 2017, 9, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.-l.; Ma, H.; Man, Y.-G.; Lv, H.-Y. Protective effects of a green tea polyphenol, epigallocatechin-3-gallate, against sevoflurane-induced neuronal apoptosis involve regulation of CREB/BDNF/TrkB and PI3K/Akt/mTOR signalling pathways in neonatal mice. Can. J. Physiol. Pharmacol. 2017, 95, 1396–1405. [Google Scholar] [CrossRef] [PubMed]
- Nath, S.; Bachani, M.; Harshavardhana, D.; Steiner, J.P. Catechins protect neurons against mitochondrial toxins and HIV proteins via activation of the BDNF pathway. J. Neurovirol. 2012, 18, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Tian, W.; Han, X.-G.; Liu, Y.-J.; Tang, G.-Q.; Liu, B.; Wang, Y.-Q.; Xiao, B.; Xu, Y.-F. Intrathecal Epigallocatechin Gallate Treatment Improves Functional Recovery After Spinal Cord Injury by Upregulating the Expression of BDNF and GDNF. Neurochem. Res. 2013, 38, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Pervin, M.; Nakagawa, A.; Iguchi, K.; Hara, A.; Takagaki, A.; Nanjo, F.; Minami, A.; Nakamura, Y. Blood-Brain Barrier Permeability of Green Tea Catechin Metabolites and their Neuritogenic Activity in Human Neuroblastoma SH-SY5Y Cells. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, J.; Assuncao, M.; Lukoyanov, N.; Cardoso, A.; Carvalho, F.; Andrade, J.P. Protective effects of a catechin-rich extract on the hippocampal formation and spatial memory in aging rats. Behav. Brain Res. 2013, 246, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Moghadas, M.; Edalatmanesh, M.A.; Robati, R. Histopathological Analysis from Gallic Acid Administration on Hippocampal Cell Density, Depression, and Anxiety Related Behaviors in A Trimethyltin Intoxication Model. Cell J. 2016, 17, 659–667. [Google Scholar] [PubMed]
- Zhou, H.; Li, X.J.; Gao, M. Curcumin Protects PC12 Cells from Corticosterone-Induced Cytotoxicity: Possible Involvement of the ERK 1/2 Pathway (vol 104, pg 236, 2009). Basic Clin. Pharmacol. Toxicol. 2009, 104, 417. [Google Scholar]
- Bhattacharya, T.K.; Pence, B.D.; Ossyra, J.M.; Gibbons, T.E.; Perez, S.; McCusker, R.H.; Kelley, K.W.; Johnson, R.W.; Woods, J.A.; Rhodes, J.S. Exercise but not (-)-epigallocatechin-3-gallate or beta-alanine enhances physical fitness, brain plasticity, and behavioral performance in mice. Physiol. Behav. 2015, 145, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Pervin, M.; Unno, K.; Ohishi, T.; Tanabe, H.; Miyoshi, N.; Nakamura, Y. Beneficial Effects of Green Tea Catechins on Neurodegenerative Diseases. Molecules 2018, 23, 1297. [Google Scholar] [CrossRef] [PubMed]
- Guedj, F.; Sebrie, C.; Rivals, I.; Ledru, A.; Paly, E.; Bizot, J.C.; Smith, D.; Rubin, E.; Gillet, B.; Arbones, M.; et al. Green Tea Polyphenols Rescue of Brain Defects Induced by Overexpression of DYRK1A. PLoS ONE 2009, 4, e4606. [Google Scholar] [CrossRef] [PubMed]
- Lyubomirsky, S.; King, L.; Diener, E. The benefits of frequent positive affect: Does happiness lead to success? Psychol. Bull. 2005, 131, 803–855. [Google Scholar] [CrossRef] [PubMed]
- Baas, M.; de Dreu, C.K.W.; Nijstad, B.A. A Meta-Analysis of 25 Years of Mood-Creativity Research: Hedonic Tone, Activation, or Regulatory Focus? Psychol. Bull. 2008, 134, 779–806. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.O.; Dellarco, D.V.; Breiner, K.; Helion, C.; Heller, A.S.; Rahdar, A.; Pedersen, G.; Chein, J.; Dyke, J.P.; Galvan, A.; et al. The Impact of Emotional States on Cognitive Control Circuitry and Function. J. Cogn. Neurosci. 2016, 28, 446–459. [Google Scholar] [CrossRef]
- Froeber, K.; Dreisbach, G. The differential influences of positive affect, random reward, and performance-contingent reward on cognitive control. Cogn. Affect. Behav. Neurosci. 2014, 14, 530–547. [Google Scholar] [CrossRef]
- Vanlessen, N.; De Raedt, R.; Mueller, S.C.; Rossi, V.; Pourtois, G. Happy and less inhibited? Effects of positive mood on inhibitory control during an antisaccade task revealed using topographic evoked potential mapping. Biol. Psychol. 2015, 110, 190–200. [Google Scholar] [CrossRef]
- Dolcos, F.; Iordan, A.D.; Dolcos, S. Neural correlates of emotion-cognition interactions: A review of evidence from brain imaging investigations. J. Cogn. Psychol. (Hove) 2011, 23, 669–694. [Google Scholar] [CrossRef]
- Ritchey, M.; Dolcos, F.; Eddington, K.M.; Strauman, T.J.; Cabeza, R. Neural correlates of emotional processing in depression: Changes with cognitive behavioral therapy and predictors of treatment response. J. Psychiatr. Res. 2011, 45, 577–587. [Google Scholar] [CrossRef]
- Steptoe, A.; Wardle, J. Positive affect and biological function in everyday life. Neurobiol. Aging 2005, 26, S108–S112. [Google Scholar] [CrossRef]
- Turner, M.A.; Andrewes, D.G. The relationship between mood state, interpersonal attitudes and psychological distress in stroke patients. International journal of rehabilitation research. Internationale Zeitschrift fur Rehabilitationsforschung. Revue Internationale de Recherches de Readaptation 2010, 33, 43–48. [Google Scholar] [PubMed]
- Einöther, S.J.; Martens, V.E. Acute effects of tea consumption on attention and mood. Am. J. Clin. Nutr. 2013, 98, 1700S–1708S. [Google Scholar] [CrossRef] [PubMed]
- Bryan, J. Psychological effects of dietary components of tea: Caffeine and L-theanine. Nutr. Rev. 2008, 66, 82–90. [Google Scholar] [CrossRef]
- Kahathuduwa, C.N.; Dhanasekara, C.S.; Chin, S.H.; Davis, T.; Weerasinghe, V.S.; Dassanayake, T.L.; Binks, M. l-Theanine and caffeine improve target-specific attention to visual stimuli by decreasing mind wandering: A human functional magnetic resonance imaging study. Nutr. Res. 2018, 49, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Dietz, C.; Dekker, M.; Piqueras-Fiszman, B. An intervention study on the effect of matcha tea, in drink and snack bar formats, on mood and cognitive performance. Food Res. Int. 2017, 99, 72–83. [Google Scholar] [CrossRef]
- Wightman, E.L.; Haskell, C.F.; Forster, J.S.; Veasey, R.C.; Kennedy, D.O. Epigallocatechin gallate, cerebral blood flow parameters, cognitive performance and mood in healthy humans: A double-blind, placebo-controlled, crossover investigation. Hum. Psychopharmacol.-Clin. Exp. 2012, 27, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.; Downey, L.A.; Ciorciari, J.; Pipingas, A.; Nolidin, K.; Finn, M.; Wines, M.; Catchlove, S.; Terrens, A.; Barlow, E.; et al. Acute neurocognitive effects of epigallocatechin gallate (EGCG). Appetite 2012, 58, 767–770. [Google Scholar] [CrossRef]
- Einöther, S.J.L.; Baas, M.; Rowson, M.; Giesbrecht, T. Investigating the effects of tea, water and a positive affect induction on mood and creativity. Food Qual. Prefer. 2015, 39, 56–61. [Google Scholar] [CrossRef]
- Einother, S.J.L.; Rowson, M.; Ramaekers, J.G.; Giesbrecht, T. Infusing pleasure: Mood effects of the consumption of a single cup of tea. Appetite 2016, 103, 302–308. [Google Scholar] [CrossRef]
- Dohle, S.; Rail, S.; Siegrist, M. I cooked it myself: Preparing food increases liking and consumption. Food Qual. Prefer. 2014, 33, 14–16. [Google Scholar] [CrossRef]
- Vohs, K.D.; Wang, Y.; Gino, F.; Norton, M.I. Rituals enhance consumption. Psychol. Sci. 2013, 24, 1714–1721. [Google Scholar] [CrossRef] [PubMed]
- Burgdorf, J.; Panksepp, J. The neurobiology of positive emotions. Neurosci. Biobehav. Rev. 2006, 30, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Berridge, K.C.; Robinson, T.E. Parsing reward. Trends Neurosci. 2003, 26, 507–513. [Google Scholar] [CrossRef]
- Dawkins, L.; Shahzad, F.-Z.; Ahmed, S.S.; Edmonds, C.J. Expectation of having consumed caffeine can improve performance and mood. Appetite 2011, 57, 597–600. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.E.; Bargh, J.A. Experiencing physical warmth promotes interpersonal warmth. Science 2008, 322, 606–607. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, T.K.; Irwin, M.R.; Moieni, M.; Jevtic, I.; Eisenberger, N.I. A Pilot Study Examining Physical and Social Warmth: Higher (Non-Febrile) Oral Temperature Is Associated with Greater Feelings of Social Connection. PLoS ONE 2016, 11, e0156873. [Google Scholar] [CrossRef] [PubMed]
- Willemse, C.J.A.M.; Heylen, D.K.J.; van Erp, J.B.F. Communication via warm haptic interfaces does not increase social warmth. J. Multimodal User Interfaces 2018, 12, 329–344. [Google Scholar] [CrossRef]
- Dubovski, N.; Ert, E.; Niv, M.Y. Bitter mouth-rinse affects emotions. Food Qual. Prefer. 2017, 60, 154–164. [Google Scholar] [CrossRef]
- Wiener, A.; Shudler, M.; Levit, A.; Niv, M.Y. BitterDB: A database of bitter compounds. Nucleic Acids Res. 2012, 40, D413–D419. [Google Scholar] [CrossRef]
- Di Pizio, A.; Niv, M.Y. Computational Studies of Smell and Taste Receptors. Isr. J. Chem. 2014, 54, 1205–1218. [Google Scholar] [CrossRef]
- Di Pizio, A.; Niv, M.Y. Promiscuity and selectivity of bitter molecules and their receptors. Bioorg. Med. Chem. 2015, 23, 4082–4091. [Google Scholar] [CrossRef] [PubMed]
- Bobowski, N.; Reed, D.R.; Mennella, J.A. Variation in the TAS2R31 bitter taste receptor gene relates to liking for the nonnutritive sweetener Acesulfame-K among children and adults. Sci. Rep. 2016, 6, 39135. [Google Scholar] [CrossRef] [PubMed]
- Roura, E.; Aldayyani, A.; Thavaraj, P.; Prakash, S.; Greenway, D.; Thomas, W.G.; Meyerhof, W.; Roudnitzky, N.; Foster, S.R. Variability in Human Bitter Taste Sensitivity to Chemically Diverse Compounds Can Be Accounted for by Differential TAS2R Activation. Chem. Senses 2015, 40, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Bobowski, N.K. The sweetness and bitterness of childhood: Insights from basic research on taste preferences. Physiol. Behav. 2015, 152, 502–507. [Google Scholar] [CrossRef]
- Weisburger, J.H. Tea and health: A historical perspective. Cancer Lett. 1997, 114, 315–317. [Google Scholar] [CrossRef]
- Korir, M.W.; Wachira, F.N.; Wanyoko, J.K.; Ngure, R.M.; Khalid, R. The fortification of tea with sweeteners and milk and its effect on in vitro antioxidant potential of tea product and glutathione levels in an animal model. Food Chem. 2014, 145, 145–153. [Google Scholar] [CrossRef]
- Lorenz, M.; Jochmann, N.; von Krosigk, A.; Martus, P.; Baumann, G.; Stangl, K.; Stangl, V. Addition of milk prevents vascular protective effects of tea. Eur. Heart J. 2007, 28, 219–223. [Google Scholar] [CrossRef]
- Gose, M.; Krems, C.; Heuer, T.; Hoffmann, I. Trends in food consumption and nutrient intake in Germany between 2006 and 2012: Results of the German National Nutrition Monitoring (NEMONIT). Br. J. Nutr. 2016, 115, 1498–1507. [Google Scholar] [CrossRef]
- Drewnowski, A.; Rehm, C.D. Sources of Caffeine in Diets of US Children and Adults: Trends by Beverage Type and Purchase Location. Nutrients 2016, 8, 154. [Google Scholar] [CrossRef]
- Del Rio, D.; Calani, L.; Cordero, C.; Salvatore, S.; Pellegrini, N.; Brighenti, F. Bioavailability and catabolism of green tea flavan-3-ols in humans. Nutrition 2010, 26, 1110–1116. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Troufflard, S.; Serafini, M.; Crozier, A. Absorption, metabolism and excretion of Choladi green tea flavan-3-ols by humans. Mol. Nutr. Food Res. 2009, 53, S44–S53. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Espin, J.C.; Tomas-Barberan, F.A. Interaction between Phenolics and Gut Microbiota: Role in Human Health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wang, P.; Wang, P.; Hu, X.; Chen, F. The gut microbiota: A treasure for human health. Biotechnol. Adv. 2016, 34, 1210–1224. [Google Scholar] [CrossRef] [PubMed]
- Carlos Espin, J.; Gonzalez-Sarrias, A.; Tomas-Barberan, F.A. The gut microbiota: A key factor in the therapeutic effects of (poly) phenols. Biochem. Pharmacol. 2017, 139, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Oteiza, P.I.; Fraga, C.G.; Mills, D.A.; Taft, D.H. Flavonoids and the gastrointestinal tract: Local and systemic effects. Mol. Asp. Med. 2018, 61, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.; Wang, F.; Dai, S.; Lu, J.; Wu, X.; Zhang, J. Profiling and identification of (-)-epicatechin metabolites in rats using ultra-high performance liquid chromatography coupled with linear trap-Orbitrap mass spectrometer. Drug Test. Anal. 2017, 9, 1224–1235. [Google Scholar] [CrossRef]
- Borges, G.; Ottaviani, J.I.; van der Hooft, J.J.J.; Schroeter, H.; Crozier, A. Absorption, metabolism, distribution and excretion of (-)-epicatechin: A review of recent findings. Mol. Asp. Med. 2018, 61, 18–30. [Google Scholar] [CrossRef]
- Wang, J.; Ferruzzi, M.G.; Ho, L.; Blount, J.; Janle, E.M.; Gong, B.; Pan, Y.; Gowda, G.A.N.; Raftery, D.; Arrieta-Cruz, I.; et al. Brain-Targeted Proanthocyanidin Metabolites for Alzheimer’s Disease Treatment. J. Neurosci. 2012, 32, 5144–5150. [Google Scholar] [CrossRef]
- Schimidt, H.L.; Garcia, A.; Martins, A.; Mello-Carpes, P.B.; Carpes, F.P. Green tea supplementation produces better neuroprotective effects than red and black tea in Alzheimer-like rat model. Food Res. Int. 2017, 100 Pt 1, 442–448. [Google Scholar] [CrossRef]
- Wang, Y.; Kan, Z.; Thompson, H.J.; Ling, T.; Ho, C.-T.; Li, D.; Wan, X. Impact of Six Typical Processing Methods on the Chemical Composition of Tea Leaves Using a Single Camellia sinensis Cultivar, Longjing 43. J. Agric. Food Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-K.; Lin, C.-L.; Liang, Y.-C.; Lin-Shiau, S.-Y.; Juan, I.M. Survey of catechins, gallic acid, and methylxanthines in green, oolong, pu-erh, and black teas. J. Agric. Food Chem. 1998, 46, 3635–3642. [Google Scholar] [CrossRef]
- Lee, V.S.Y.; Dou, J.; Chen, R.J.Y.; Lin, R.-S.; Lee, M.-R.; Tzen, J.T.C. Massive accumulation of gallic acid and unique occurrence of myricetin, quercetin, and kaempferol in preparing old oolong tea. J. Agric. Food Chem. 2008, 56, 7950–7956. [Google Scholar] [CrossRef] [PubMed]
- Teng, J.; Zhou, W.; Zeng, Z.; Zhao, W.; Huang, Y.; Zhang, X. Quality components and antidepressant-like effects of GABA green tea. Food Funct. 2017, 8, 3311–3318. [Google Scholar] [CrossRef] [PubMed]
| Tea Compound | Effect on Physiological Stress Response |
|---|---|
| L-theanine | Reduction in stress as measured by attenuated adrenal hypertrophy [31] Reduced sAA and subjective stress levels [32] Reduction in elevated plasma ACTH and CORT [32,33] |
| L-arginine | Reduction in stress as measured by attenuated adrenal hypertrophy [31] |
| Green tea polyphenol mixture | Reduced CORT and ACTH, reduced immobility in FST and TST [34] Restored HPA activity via ERK upregulation [35] |
| EGCG (anti-stress effects) | Restored HPA activity via ERK upregulation [35] Lowered corticosterone/CRH/ACTH [36] stress-reduction via reduction of neuron over-excitation [37,38,39] improved GABA transmission via activation of the SIRT1/PGC-1α pathway [40] Antagonized stress-reduction effects of L-theanine/L-arginine [31] |
| Epigallocatechin | Restored anti-stress effects of L-theanine/L-arginine [31] through competitive inhibition of EGCG [31] |
| Low-Caffeine Green Tea | Lowered sAA levels, improved sleep quality more effectively than standard caffeinated green tea [41] Reduction in adrenal hypertrophic stress response, more significant than standard green tea [31] |
| Green Tea, Shade-Grown White Tea | Lowered CgA levels following stress load task for both tea types, more effect for Shade-Grown White Tea [42] |
| Green Tea Aroma | Lowered CgA levels following stress load task [43] |
| Black Tea Aroma | Lowered CgA levels following stress load task [44] |
| Tea Compound | Effect on Inflammatory Response |
|---|---|
| Theaflavins (TF) | Reduced LPS-induced neural inflammation, suppressed cytokine production, reduced immobility in TST. TF showed better anti-inflammatory capacity than common polyphenols, but comparable to EGCG [91]. |
| Theaflavin-3,3′-digallate (TF3) | Inhibited LPS-induced expression TNF-α, IL-1β, and IL-6 [92,93] |
| Green Tea Extract | Reduced hepatic inflammation by attenuating NFκB activation via down-regulation on TNFR1 and TLR4 in HFD model [94] |
| Epigallocatechin gallate (EGCG) | Reduced neuroinflammation via inhibition of MAPK and NFκB pathways in HFFD model [95]. Reduced SPS-induced increase in IL-1β and TNF-α in mouse hippocampi, decreased expression level of IL-1β mRNA with RT-PCR analysis [36]. Downstream inhibitor of inflammatory signaling through occupation of TAK1 site, inhibition of p38 and NFκB [96]. Reduced CSM-induced IL-8 production via inhibition of p38, MAPK and NFκB in AC16 cardiomyocytes [97]. Inhibited phosphorylation of p65, showed in vitro NFκB regulation [98]. Inhibited activity of NO, COX-2, TNF-α, IL-6 and IL-1β in LPS-induced murine macrophage cell line [99] In cultured human epidermal keratinocytes exposed to airborne PM10, suppressed TNF-α, IL-1β, IL-6, IL-8 and MMP-1 [100]. Topical application in 8-week RCT improved acne via suppression of NFκB/AP-1 pathway [101] |
| EGC and EC | Effective downstream inhibitors of inflammatory signaling through occupation of TAK1 site [96] |
| Gallic acid | Reduced airway inflammation by decreasing IL-4, IL-5, IL-13, IL-17 in nasal lavage fluid of mice with allergic rhinitis [102] |
| Gallocatechin gallate | Inhibited LPS-induced expression of MCP-1 and IL-6 as effectively as EGCG in 3T3-L1 cells [98]. |
| Oolong tea ethanol extract | Inhibited activity of NO, COX-2, TNF-α, IL-6 and IL-1β in LPS-induced murine macrophage cell line [99] |
| L-theanine | Topically delivered, reduced skin inflammation via inhibition of IL-1β, TNF-α and COX-2 [103]. Alleviated airway inflammation via suppression of NFκB pathway, reduced production of MCP-1, IL-4, IL-5, IL-13, TNF-α and interferon-gamma, attenuated trafficking of inflammatory cells into bronchoalveolar lavage fluid [104]. |
| Teasaponin | Attenuated TLR-4, NF-κB, IL-1β, IL-6 and TNF-α in HFD mouse model [105]. |
| Tea Compound | Effect on Monoaminergic Systems |
|---|---|
| L-Theanine | Increased levels of 5-HT, NE and DA in the PFC, NAC, and HIP. Increased levels of 5-HT and DA in the ST. Increased DA levels in the HIP [111]. Promoted DA transmission in HIP via DA D1/5 receptor-PKA pathway activation in AD mouse model [112] |
| Green tea | Administration for 5 weeks, tone of 5-HT was normalized, reduced stress response [113]. |
| Epicatechin | Functioned as anxiolytic in OF and EPM via decreased expression of MAO-A in cortex, and increased pro-BDNF and BDNF via Akt pathway [114]. |
| Tea Polyphenol Mixture | Reversed CUMS-induced reduction in 5-HT and NE in the HIP, PFC [115]. Partial restoration of normal DA and 5-HT following stress-induced neural injury [35]. Null effect on human GM in 12-week RCT [116]. BTP supplementation more than doubled butyric acid levels. Significant increases were observed for GTP, but to a lesser extent than BTP. Increases in SCFA upregulated AMPK activation by 70% and 289% for GTP and BTP, respectively [117]. |
| EGCG | Partial restoration of normal DA and 5-HT following stress-induced neural injury [35]. Promoted growth of Bifidobacterium spp. and Lactobacillus/Enterococcus groups, induced higher concentrations of SCFA when incubated with human GM [118]. Enhanced adhesion of certain Lactobacillus strains to human epithelial intestinal lines [119]. |
| Theaflavins | Increased DA turnover in FC, as measured by increased DOPAC and DOPAC/DA ratio [120]. Reduced oxidative stress, preserved DA levels in ST via and protection of dopaminergic neurons against degeneration by MPTP, improving motor behavior and expression of DAT and VMAT2 in ST and substantia nigra [121]. |
| Catechin | Significantly increased Bifidobacterium spp. following in vitro incubation with human fecal samples [122]. |
| EGCG3”Me | Significantly increased Bifidobacterium spp. and SCFA-generating GM species in vivo in mouse model [123]. Promoted growth of Bifidobacterium spp. and Lactobacillus/Enterococcus groups, induced higher concentrations of SCFA when incubated with human GM [118]. |
| Gallocatechin gallate | Promoted growth of Bifidobacterium spp. and Lactobacillus/Enterococcus groups, induced higher concentrations of SCFA when incubated with human GM [118]. |
| Epigallocatechin | Enhanced adhesion of certain Lactobacillus strains to human epithelial intestinal lines [119]. |
| Tea Compound | Effect on Neurogenesis/Neuroplasticity |
|---|---|
| Teasaponin | Six-week supplementation in HFD model attenuated BDNF deficits in the HIP, prevented recognition memory impairment [105]. |
| L-theanine | Increased exploratory activity in OFT, enhanced object recognition memory, significantly increased BDNF levels and BrdU-, Ki67, and DCX-labeled cells in the granule cell layer [188]. |
| EGCG | DCX-positive neurons showed more elaborate dendritic trees, accompanied by significantly increased HIP neurogenesis [189]. Attenuated HFFD-induced neuronal damage, reduced cognitive disorder via upregulation of CREB/BDNF pathway [95]. Submicromolar concentrations potentiated the neuritogenic ability of BDNF in PC12 cells [190]. Attenuated corticosterone-induced cytotoxicity, up-regulated Shh pathway [191]. Induced SH-SY5Y cell growth in vitro [192]. RT-PCR showed enhanced BDNF and TrkB mRNA levels, activated Akt and CREB/BDNF pathways, inhibited sevoflurane-induced neurodegeneration, improved learning and memory retention [193]. Targeted BDNF and proBDNF signaling pathways, normalized tat-mediated increases in proapoptotic proBDNF, normalized tat-mediated decreases in mature BDNF protein in hippocampal neurons [194]. Following spinal cord injury in rats, increased expression of BDNF, GDNF, improved locomotor recovery [195]. |
| Epigallocatechin | Enhanced the neurogenic properties of EGCG [190]. Primary EGC metabolite, EGC-M5, and several conjugated forms improved neurite length and neurite number in SH-SY5Y cells [196]. |
| Epicatechin | Enhanced the neurogenic properties of EGCG [190]. Targeted BDNF and proBDNF signaling pathways, normalized tat-mediated increases in proapoptotic proBDNF, normalized tat-mediated decreases in mature BDNF protein in hippocampal neurons [194]. |
| Non-EGCG GTP | Plastic changes in dendritic arborizations of dentate granule cells, improved spatial learning in Morris water maze [197]. |
| Gallic acid | Ameliorated TMT-induced anxiety and depression, improved cell densities in the CA1, CA2, CA3 and DG hippocampal subdivisions [198]. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rothenberg, D.O.; Zhang, L. Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption. Nutrients 2019, 11, 1361. https://doi.org/10.3390/nu11061361
Rothenberg DO, Zhang L. Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption. Nutrients. 2019; 11(6):1361. https://doi.org/10.3390/nu11061361
Chicago/Turabian StyleRothenberg, Dylan O’Neill, and Lingyun Zhang. 2019. "Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption" Nutrients 11, no. 6: 1361. https://doi.org/10.3390/nu11061361
APA StyleRothenberg, D. O., & Zhang, L. (2019). Mechanisms Underlying the Anti-Depressive Effects of Regular Tea Consumption. Nutrients, 11(6), 1361. https://doi.org/10.3390/nu11061361





