Coffee Intake and Obesity: A Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Statistical Analyses
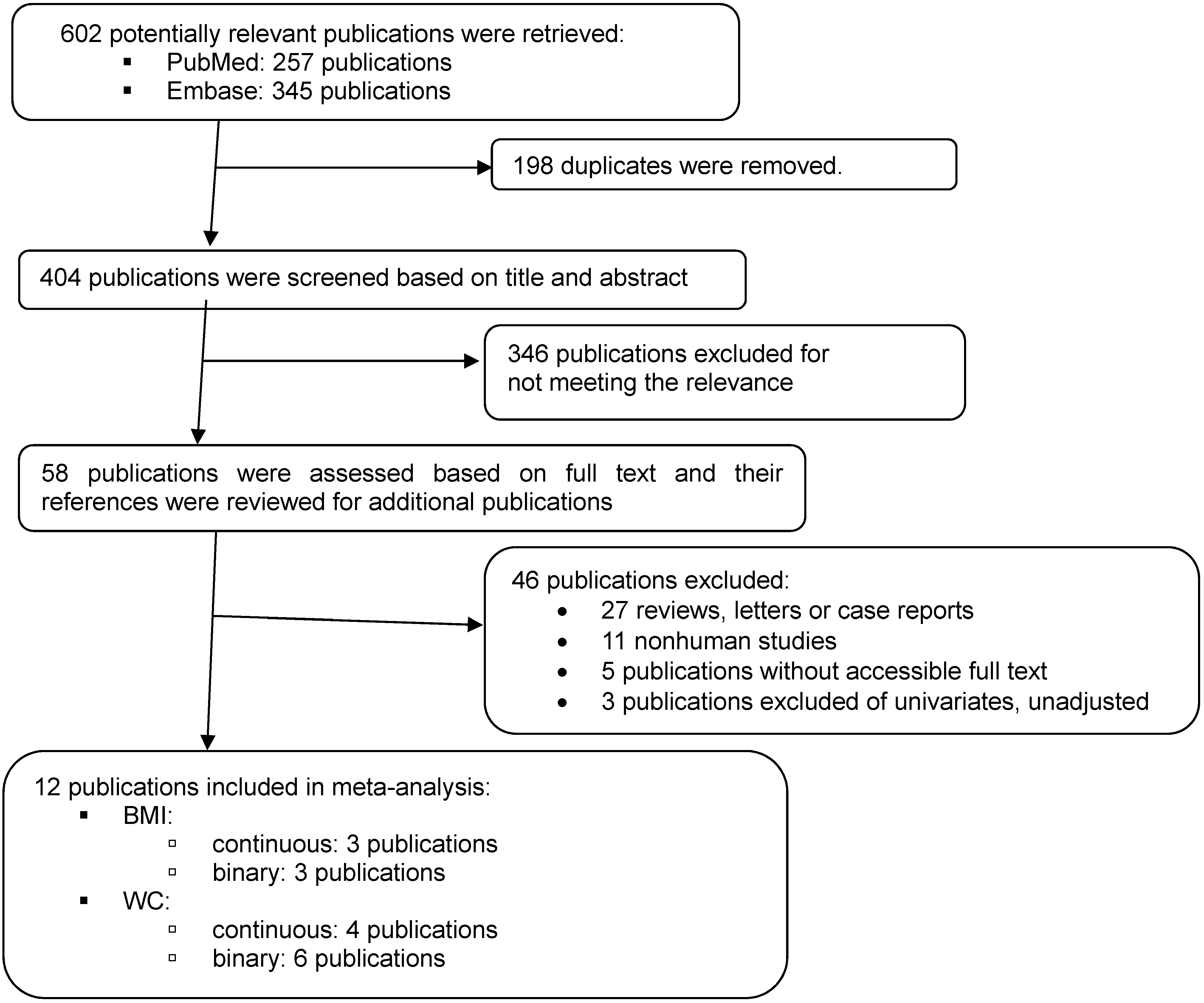
3. Results
3.1. Characteristics of Studies Included in the Meta-Analysis
3.1.1. For Overall Adiposity
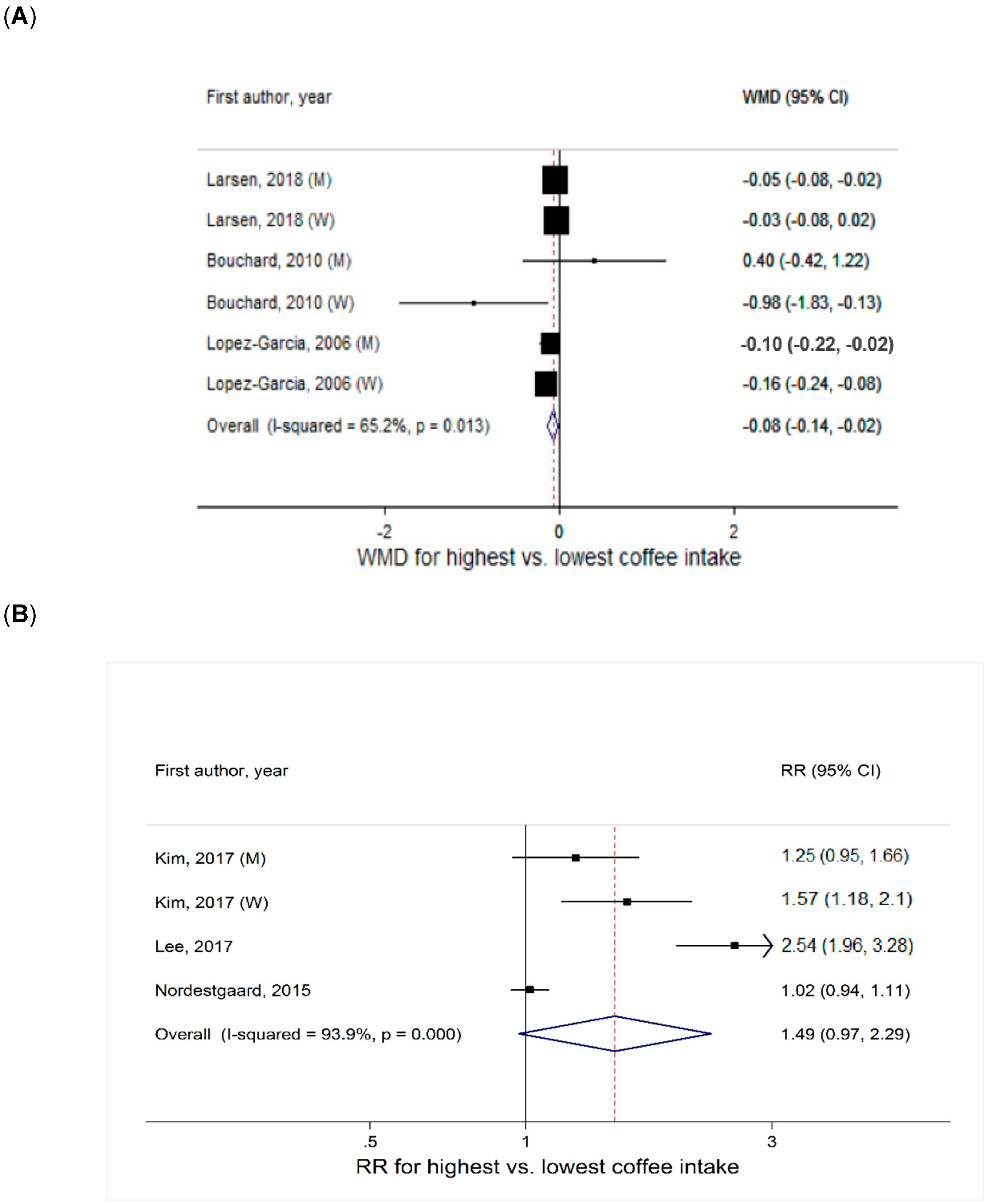
Coffee Intake and Continuous BMI
Coffee Intake and Overweight or Obesity Defined by BMI
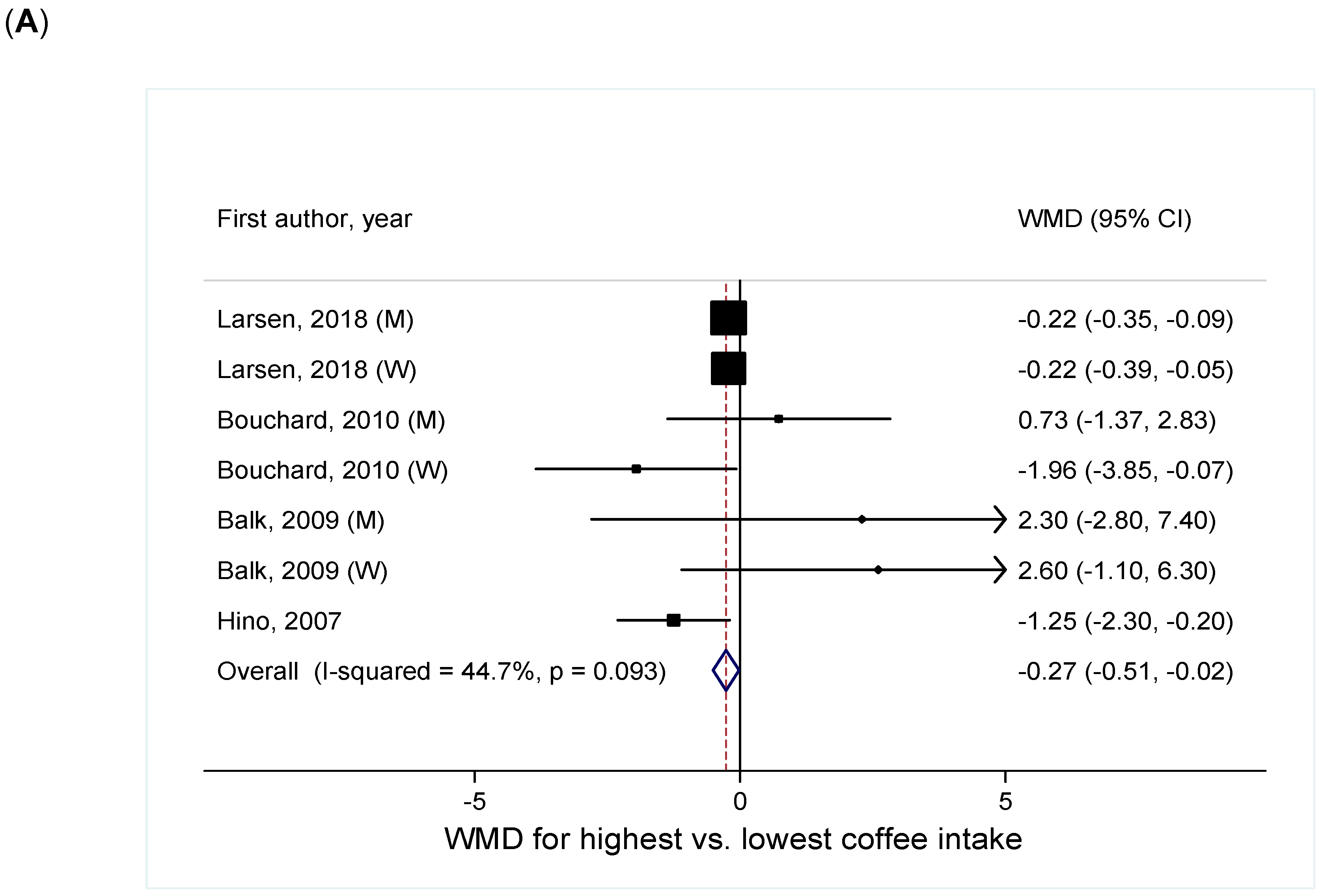
3.1.2. For Central Adiposity
Coffee Intake and Continuous WC
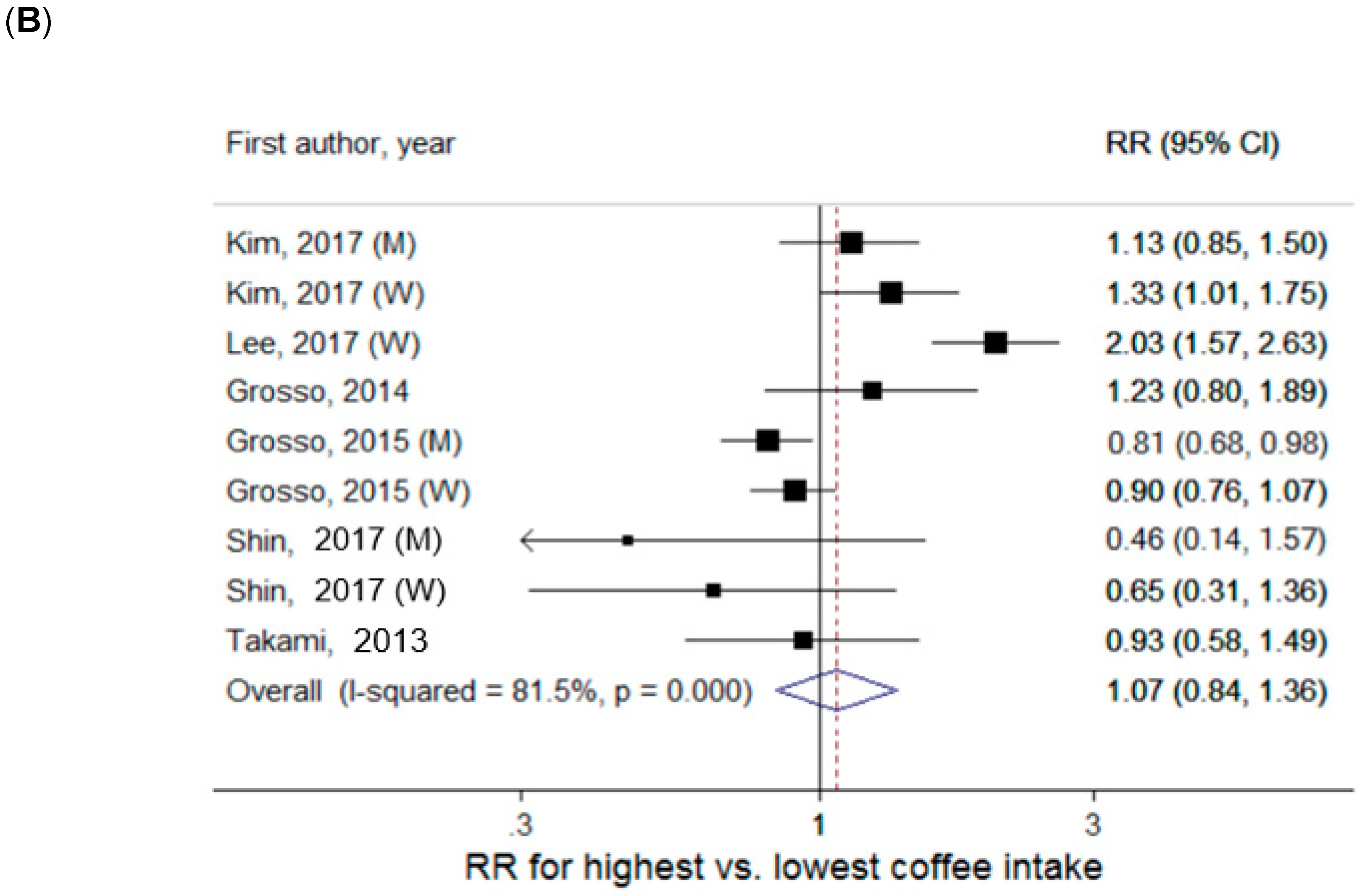
Coffee Intake and Central Obesity Defined by WC
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Higdon, J.V.; Frei, B. Coffee and health: A review of recent human research. Crit. Rev. Food Sci. Nutr. 2006, 46, 101–123. [Google Scholar] [CrossRef] [PubMed]
- Poole, R.; Kennedy, O.J.; Roderick, P.; Fallowfield, J.A.; Hayes, P.C.; Parkes, J. Coffee consumption and health: Umbrella review of meta-analyses of multiple health outcomes. BMJ 2017, 359, j5024. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Godos, J.; Galvano, F.; Giovannucci, E.L. Coffee, Caffeine, and Health Outcomes: An Umbrella Review. Annu. Rev. Nutr. 2017, 37, 131–156. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Stepaniak, U.; Micek, A.; Topor-Madry, R.; Pikhart, H.; Szafraniec, K.; Pajak, A. Association of daily coffee and tea consumption and metabolic syndrome: Results from the Polish arm of the HAPIEE study. Eur. J. Nutr. 2015, 54, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Garcia, E.; van Dam, R.M.; Rajpathak, S.; Willett, W.C.; Manson, J.E.; Hu, F.B. Changes in caffeine intake and long-term weight change in men and women. Am. J. Clin. Nutr. 2006, 83, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, H.Y.; Kim, J. Coffee Consumption and the Risk of Obesity in Korean Women. Nutrients 2017, 9, 1340. [Google Scholar] [CrossRef]
- Nordestgaard, A.T.; Thomsen, M.; Nordestgaard, B.G. Coffee intake and risk of obesity, metabolic syndrome and type 2 diabetes: A Mendelian randomization study. Int. J. Epidemiol. 2015, 44, 551–565. [Google Scholar] [CrossRef]
- Bouchard, D.R.; Ross, R.; Janssen, I. Coffee, tea and their additives: Association with BMI and waist circumference. Obes. Facts. 2010, 3, 345–352. [Google Scholar] [CrossRef]
- Grosso, G.; Marventano, S.; Galvano, F.; Pajak, A.; Mistretta, A. Factors associated with metabolic syndrome in a mediterranean population: Role of caffeinated beverages. J. Epidemiol. 2014, 24, 327–333. [Google Scholar] [CrossRef]
- Hino, A.; Adachi, H.; Enomoto, M.; Furuki, K.; Shigetoh, Y.; Ohtsuka, M.; Kumagae, S.; Hirai, Y.; Jalaldin, A.; Satoh, A.; et al. Habitual coffee but not green tea consumption is inversely associated with metabolic syndrome: An epidemiological study in a general Japanese population. Diabetes Res. Clin. Pract. 2007, 76, 383–389. [Google Scholar] [CrossRef]
- Larsen, S.C.; Mikkelsen, M.L.; Frederiksen, P.; Heitmann, B.L. Habitual coffee consumption and changes in measures of adiposity: A comprehensive study of longitudinal associations. Sci. Rep. 2018, 42, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Takami, H.; Nakamoto, M.; Uemura, H.; Katsuura, S.; Yamaguchi, M.; Hiyoshi, M.; Sawachika, F.; Juta, T.; Arisawa, K. Inverse correlation between coffee consumption and prevalence of metabolic syndrome: Baseline survey of the Japan Multi-Institutional Collaborative Cohort (J-MICC) Study in Tokushima, Japan. J. Epidemiol. 2013, 23, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Park, Y.S. Light coffee consumption is protective against sarcopenia, but frequent coffee consumption is associated with obesity in Korean adults. Nutr. Res. 2017, 41, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Linton, J.A.; Kwon, Y.; Jung, Y.; Oh, B.; Oh, S. Relationship between Coffee Consumption and Metabolic Syndrome in Korean Adults: Data from the 2013-2014 Korea National Health and Nutrition Examination Survey. Korean J. Fam. Med. 2017, 38, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Balk, L.; Hoekstra, T.; Twisk, J. Relationship between long-term coffee consumption and components of the metabolic syndrome: The Amsterdam Growth and Health Longitudinal Study. Eur. J. Epidemiol. 2009, 24, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Maki, C.; Funakoshi-Tago, M.; Aoyagi, R.; Ueda, F.; Kimura, M.; Kobata, K.; Tago, K.; Tamura, H. Coffee extract inhibits adipogenesis in 3T3-L1 preadipocyes by interrupting insulin signaling through the downregulation of IRS1. PLoS ONE 2017, 12, e0173264. [Google Scholar] [CrossRef]
- Wille, N.; Erhart, M.; Petersen, C.; Ravens-Sieberer, U. The impact of overweight and obesity on health-related quality of life in childhood--results from an intervention study. BMC Public Health 2008, 8, 421. [Google Scholar] [CrossRef]
- Hruby, A.; Hu, F.B. The Epidemiology of Obesity: A Big Picture. PharmacoEconomics 2015, 33, 673–689. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Harbord, R.M.; Egger, M.; Sterne, J.A.C. A modified test for small-study effects in meta-analyses of controlled trials with binary endpoints. Stat. Med. 2006, 25, 3443–3457. [Google Scholar] [CrossRef] [PubMed]
- Cho, A.S.; Jeon, S.M.; Kim, M.J.; Yeo, J.; Seo, K.I.; Choi, M.S.; Lee, M.K. Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem. Toxicol. 2010, 48, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Ilavenil, S.; Arasu, M.V.; Lee, J.C.; Kim, D.H.; Roh, S.G.; Park, H.S.; Choi, G.J.; Mayakrishnan, V.; Choi, K.C. Trigonelline attenuates the adipocyte differentiation and lipid accumulation in 3T3-L1 cells. Phytomedicine 2014, 21, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ma, X.; Peng, D.; Zhang, R.; Sun, X.; Chen, M.; Yan, J.; Wang, S.; Yan, D.; He, Z.; et al. Effects of Obesity Related Genetic Variations on Visceral and Subcutaneous Fat Distribution in a Chinese Population. Sci. Rep. 2016, 6, 20691. [Google Scholar] [CrossRef]
- Leeners, B.; Geary, N.; Tobler, P.N.; Asarian, L. Ovarian hormones and obesity. Hum. Reprod. Update 2017, 23, 300–321. [Google Scholar] [CrossRef]
| Outcome | Among Men | Among Women | Pheterogeneity by Sex |
|---|---|---|---|
| Overall adiposity | |||
| BMI | |||
| No. of studies | 3 | 3 | |
| WMD (95% CI; I2) | −0.05 (−0.09, −0.02; 0%) | −0.12 (−0.27, 0.03; 84%) | 0.68 |
| Overweight or obesity as defined by BMI | |||
| No. of studies | 1 | 2 | |
| RR (95% CI; I2) | 1.25 (0.95, 1.65; NR) | 2.01 (1.25, 3.21; 83%) | 0.13 |
| Central adiposity | |||
| WC | |||
| No. of studies | 3 | 3 | |
| WMD (95% CI; I2) | −0.21 (−0.35, −0.08; 0%) | −0.36 (−2.00, 1.28; 64%) | 0.58 |
| Central obesity as defined by WC | |||
| No. of studies | 3 | 4 | |
| RR (95% CI; I2) | 0.90 (0.66, 1.23; 59%) | 1.18 (0.75, 1.86; 90%) | 0.59 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, A.; Lim, W.; Kim, S.; Khil, H.; Cheon, E.; An, S.; Hong, S.; Lee, D.H.; Kang, S.-S.; Oh, H.; et al. Coffee Intake and Obesity: A Meta-Analysis. Nutrients 2019, 11, 1274. https://doi.org/10.3390/nu11061274
Lee A, Lim W, Kim S, Khil H, Cheon E, An S, Hong S, Lee DH, Kang S-S, Oh H, et al. Coffee Intake and Obesity: A Meta-Analysis. Nutrients. 2019; 11(6):1274. https://doi.org/10.3390/nu11061274
Chicago/Turabian StyleLee, Ariel, Woobin Lim, Seoyeon Kim, Hayeong Khil, Eugene Cheon, Soobin An, SungEun Hong, Dong Hoon Lee, Seok-Seong Kang, Hannah Oh, and et al. 2019. "Coffee Intake and Obesity: A Meta-Analysis" Nutrients 11, no. 6: 1274. https://doi.org/10.3390/nu11061274
APA StyleLee, A., Lim, W., Kim, S., Khil, H., Cheon, E., An, S., Hong, S., Lee, D. H., Kang, S.-S., Oh, H., Keum, N., & Hsieh, C.-C. (2019). Coffee Intake and Obesity: A Meta-Analysis. Nutrients, 11(6), 1274. https://doi.org/10.3390/nu11061274





