Association between Postpartum Nutritional Status and Postpartum Depression Symptoms
Abstract
1. Introduction
2. Participants and Methods
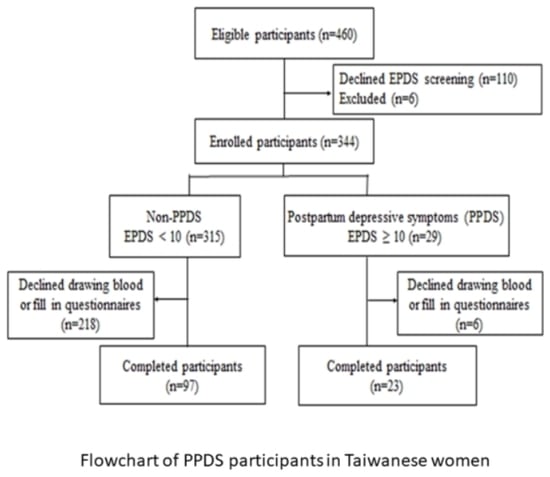
2.1. Study Participants
2.2. PPDS Screening and Questionnaires
2.3. Blood Sample Collections
2.4. Biochemical Analyses
2.5. Erythrocyte Fatty Acid Profiles Analysis
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Biochemical Analyses
3.3. Association of Nutritional Biomarkers with Postpartum Depression
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klainin, P.; Arthur, D.G. Postpartum depression in Asian cultures: A literature review. Int. J. Nurs. Stud. 2009, 46, 1355–1373. [Google Scholar] [CrossRef]
- Affonso, D.D.; De, A.K.; Horowitz, J.A.; Mayberry, L.J. An international study exploring levels of postpartum depressive symptomatology. J. Psychosom. Res. 2000, 49, 207–216. [Google Scholar] [CrossRef]
- Lanes, A.; Kuk, J.L.; Tamim, H. Prevalence and characteristics of postpartum depression symptomatology among Canadian women: A cross-sectional study. BMC Public Health 2011, 11, 302. [Google Scholar] [CrossRef] [PubMed]
- Norhayati, M.N.; Hazlina, N.H.; Asrenee, A.R.; Emilin, W.M. Magnitude and risk factors for postpartum symptoms: A literature review. J. Affect. Disord. 2015, 175, 34–52. [Google Scholar] [CrossRef]
- Lancaster, C.A.; Gold, K.J.; Flynn, H.A.; Yoo, H.; Marcus, S.M.; Davis, M.M. Risk factors for depressive symptoms during pregnancy: A systematic review. Am. J. Obs. Gynecol. 2010, 202, 5–14. [Google Scholar] [CrossRef]
- Feng, Z.; Jones, K.; Wang, W.W. An exploratory discrete-time multilevel analysis of the effect of social support on the survival of elderly people in China. Soc. Sci. Med. 2015, 130, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Melaki, V.; Sarri, K.; Apostolaki, I.; Roumeliotaki, T.; Georgiou, V.; Vassilaki, M.; Koutis, A.; Bitsios, P.; Kogevinas, M. Dietary patterns during pregnancy and the risk of postpartum depression: The mother-child ‘Rhea’ cohort in Crete, Greece. Public Health Nutr. 2011, 14, 1663–1670. [Google Scholar] [CrossRef]
- Gaillard, A.; Le Strat, Y.; Mandelbrot, L.; Keita, H.; Dubertret, C. Predictors of postpartum depression: Prospective study of 264 women followed during pregnancy and postpartum. Psychiatr. Res. 2014, 215, 341–346. [Google Scholar] [CrossRef]
- Huang, T.; Rifas-Shiman, S.L.; Ertel, K.A.; Rich-Edwards, J.; Kleinman, K.; Gillman, M.W.; Oken, E.; James-Todd, T. Pregnancy Hyperglycaemia and Risk of Prenatal and Postpartum Depressive Symptoms. Paediatr. Perinat. Epidemiol. 2015, 29, 281–289. [Google Scholar] [CrossRef]
- Douma, S.L.; Husband, C.; O’Donnell, M.E.; Barwin, B.N.; Woodend, A.K. Estrogen-related mood disorders: Reproductive life cycle factors. Adv. Nurs. Sci. 2005, 28, 364–375. [Google Scholar] [CrossRef]
- Harris, B.; Huckle, P.; Thomas, R.; Johns, S.; Fung, H. The use of rating scales to identify post-natal depression. Br. J. Psychiatr. J. Ment. Sci. 1989, 154, 813–817. [Google Scholar] [CrossRef]
- Heh, S.-S. Validation of the Chinese version of the Edinburgh Postnatal Depression Scale: Detecting postnatal depression in Taiwanese women. Hu Li Yan Jiu Nurs. Res. 2001, 9, 105–113. [Google Scholar]
- Chen, C.M.; Kuo, S.F.; Chou, Y.H.; Chen, H.C. Postpartum Taiwanese women: Their postpartum depression, social support and health-promoting lifestyle profiles. J. Clin. Nurs. 2007, 16, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- Heh, S.-S.; Coombes, L.; Bartlett, H. The association between depressive symptoms and social support in Taiwanese women during the month. Int. J. Nurs. Stud. 2004, 41, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.-L.; Chang, L.-I.; Wan, K.-S. The relationships between postpartum adaptation and postpartum depression symptoms of first pregnancy mothers in Taiwan. Int. J. Psychiat. Med. 2013, 45, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ellsworth-Bowers, E.R.; Corwin, E.J. Nutrition and the psychoneuroimmunology of postpartum depression. Nutr. Res. Rev. 2012, 25, 180–192. [Google Scholar] [CrossRef] [PubMed]
- Bieri, J.G.; Tolliver, T.J.; Catignani, G.L. Simultaneous determination of alpha-tocopherol and retinol in plasma or red cells by high pressure liquid chromatography. Am. J. Clin. Nutr. 1979, 32, 2143–2149. [Google Scholar] [CrossRef] [PubMed]
- Forrest, K.Y.; Stuhldreher, W.L. Prevalence and correlates of vitamin D deficiency in US adults. Nutr. Res. 2011, 31, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Sasaki, S.; Tanaka, K.; Yokoyama, T.; Ohya, Y.; Fukushima, W.; Saito, K.; Ohfuji, S.; Kiyohara, C.; Hirota, Y. Dietary folate and vitamins B 12, B 6, and B 2 intake and the risk of postpartum depression in Japan: The Osaka Maternal and Child Health Study. J. Affect. Disord. 2006, 96, 133–138. [Google Scholar] [CrossRef]
- Yang, H.W.; Luo, F. 2005-2008 Nutrition and Health Survey in Taiwan (NAHSIT): Thiamin and riboflavin biochemical status and associated factors of 19-64 year old adults. Minist. Health Welf. 2011, 20, 309–318. [Google Scholar]
- Fu, C.W.; Liu, J.T.; Tu, W.J.; Yang, J.Q.; Cao, Y. Association between serum 25-hydroxyvitamin D levels measured 24 hours after delivery and postpartum depression. Int. J. Obs. Gynaecol. 2015, 122, 1688–1694. [Google Scholar] [CrossRef] [PubMed]
- Murphy, P.K.; Mueller, M.; Hulsey, T.C.; Ebeling, M.D.; Wagner, C.L. An exploratory study of postpartum depression and vitamin d. J. Am. Psychiatr. Nurses Assoc. 2010, 16, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Gur, E.B.; Gokduman, A.; Turan, G.A.; Tatar, S.; Hepyilmaz, I.; Zengin, E.B.; Eskicioglu, F.; Guclu, S. Mid-pregnancy vitamin D levels and postpartum depression. Eur. J. Obs. Gynecol. Reprod. Biol. 2014, 179, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.; Whitehouse, A.J.; Newnham, J.P.; Gorman, S.; Jacoby, P.; Holt, B.J.; Serralha, M.; Tearne, J.E.; Holt, P.G.; Hart, P.H. Low maternal serum vitamin D during pregnancy and the risk for postpartum depression symptoms. Arch. Women Ment. Health 2014, 17, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Hsu, H.J.; Wu, I.W.; Sun, C.Y.; Ting, M.K.; Lee, C.C. Vitamin D deficiency in northern Taiwan: A community-based cohort study. BMC Public Health 2019, 19, 337. [Google Scholar] [CrossRef] [PubMed]
- Liperoti, R.; Landi, F.; Fusco, O.; Bernabei, R.; Onder, G. Omega-3 polyunsaturated fatty acids and depression: A review of the evidence. Curr. Pharm. Des. 2009, 15, 4165–4172. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ya, L.; Mannong, L.; Zhang, Q.; Wang, Z.; Duan, Y. Research of correlation between polyunsaturated fatty acids and postpartum depression. Int. J. Lab. Med. 2015, 6, 765–767. [Google Scholar]
- Su, H.; Sun, H.L.; Zhang, J.; Fan, W. Analysis of Correlation between Serum ω-3 Polyunsaturated Fatty Acids of Pregnant Women and Postpartum Depression. J. Kunming Med. Univ. 2014, 35, 138–144. [Google Scholar]
- Lu, D.-Y.; Tsao, Y.-Y.; Leung, Y.-M.; Su, K.-P. Docosahexaenoic acid suppresses neuroinflammatory responses and induces heme oxygenase-1 expression in BV-2 microglia: Implications of antidepressant effects for omega-3 fatty acids. Neuropsychopharmacology 2010, 35, 2238. [Google Scholar] [CrossRef]
- Su, K.-P. Biological Mechanism of Antidepressant Effect of Omega–3 Fatty Acids: How Does Fish Oil Act as a ‘Mind-Body Interface’? Neurosignals 2009, 17, 144–152. [Google Scholar] [CrossRef]
- Bove, M.; Mhillaj, E.; Tucci, P.; Giardino, I.; Schiavone, S.; Morgese, M.G.; Trabace, L. Effects of n-3 PUFA enriched and n-3 PUFA deficient diets in naïve and Aβ-treated female rats. Biochem. Pharmacol. 2018, 155, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Morgese, M.G.; Tucci, P.; Mhillaj, E.; Bove, M.; Schiavone, S.; Trabace, L.; Cuomo, V. Lifelong Nutritional Omega-3 Deficiency Evokes Depressive-Like State Through Soluble Beta Amyloid. Mol. Neurobiol. 2017, 54, 2079–2089. [Google Scholar] [CrossRef] [PubMed]
- de Gomes, M.G.; Souza, L.C.; Goes, A.R.; Del Fabbro, L.; Filho, C.B.; Donato, F.; Prigol, M.; Luchese, C.; Roman, S.S.; Puntel, R.L.; et al. Fish oil ameliorates sickness behavior induced by lipopolysaccharide in aged mice through the modulation of kynurenine pathway. J. Nutr. Biochem. 2018, 58, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Lotrich, F.E.; Sears, B.; McNamara, R.K. Elevated ratio of arachidonic acid to long-chain omega-3 fatty acids predicts depression development following interferon-alpha treatment: Relationship with interleukin-6. Brain Behav. Immun. 2013, 31, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Mamalakis, G.; Kiriakakis, M.; Tsibinos, G.; Kafatos, A. Depression and adipose polyunsaturated fatty acids in an adolescent group. Prostaglandins Leukot. Essent. Fat. Acids 2004, 71, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Otto, S.; De Groot, R.; Hornstra, G. Increased risk of postpartum depressive symptoms is associated with slower normalization after pregnancy of the functional docosahexaenoic acid status. Prostaglandins Leukot. Essent. Fat. Acids 2003, 69, 237–243. [Google Scholar] [CrossRef]
- Assies, J.; Lieverse, R.; Vreken, P.; Wanders, R.J.; Dingemans, P.M.; Linszen, D.H. Significantly reduced docosahexaenoic and docosapentaenoic acid concentrations in erythrocyte membranes from schizophrenic patients compared with a carefully matched control group. Biol. Psychiatry 2001, 49, 510–522. [Google Scholar] [CrossRef]

| Non-PPDS | PPDS | |||
|---|---|---|---|---|
| N = 97 | N = 23 | |||
| n (%) or mean ± SD | p value | |||
| Age (year) | 32.6 ± 4.5 | 31.6 ± 4.3 | 0.353 | |
| Anthropometric | ||||
| Pre-pregnancy BMI (kg/m2) | 21.6 ± 3.2 | 21.5 ± 3.2 | 0.853 | |
| Gestational weight gain (kg) | 12.9 ± 4.4 | 13.1 ± 3.9 | 0.842 | |
| Current BMI (kg/m2) | 23.4 ± 3.5 | 23.2 ± 3.9 | 0.789 | |
| Years of education | ||||
| ≤12 | 19 (20) | 5 (22) | 0.817 | |
| ≥16 | 79 (80) | 18 (78) | ||
| Working state | ||||
| Homemaking | 31 (32) | 11 (48) | 0.529 | |
| Full-time | 49 (51) | 9 (39) | ||
| Part-time | 16 (17) | 3 (13) | ||
| Parity | ||||
| Primipara | 60 (62) | 15 (65) | 0.765 | |
| Multipara | 37 (38) | 8 (35) | ||
| Baby’s gender | ||||
| Male | 58 (60) | 12 (52) | 0.505 | |
| Type of delivery | ||||
| Vaginal | 67 (69) | 13 (57) | 0.251 | |
| Caesarian section | 30 (31) | 10 (43) | ||
| Baby’s caregiver | ||||
| Self | 72 (75) | 15 (65) | 0.342 | |
| Family members | 24 (25) | 8 (35) | ||
| Breast-feeding | ||||
| Exclusive breast feed | 34 (35) | 6 (26) | 0.533 | |
| Mix feed | 53 (55) | 13 (57) | ||
| Formula feed | 10 (10) | 4 (17) | ||
| Nutritional supplements | ||||
| Pregnancy first trimester | 88 (91) | 23 (100) | 0.129 | |
| Pregnancy second trimester | 82 (85) | 19 (83) | 0.820 | |
| Pregnancy third trimester | 78 (81) | 18 (78) | 0.817 | |
| Lactation | 40 (41) | 9 (39) | 0.853 | |
| Postpartum confinement | ||||
| Home care | 44 (45) | 16 (70) * | 0.037 | |
| Postpartum care center | 53 (55) | 7 (30) | ||
| Self-perceived psychological stress | ||||
| The ability to care for the baby | 49 (51) | 15 (65) | 0.204 | |
| Families unable to provide support | 8 (8) | 7 (30) * | 0.004 | |
| Husband unable to provide support | 1 (1) | 5 (22) * | 0.000 | |
| Self-health status | 8 (8) | 5 (22) | 0.061 | |
| Baby health status | 35 (36) | 10 (44) | 0.510 | |
| Financial ability | 15 (16) | 4 (17) | 0.820 | |
| Other | 3 (3) | 3 (13) * | 0.049 | |
| Overall psychological stress scores | 1.2 ± 1.0 | 2.1 ± 1.0* | 0.000 | |
| Postpartum care satisfaction scores | 6.1 ± 1.0 | 5.2 ± 1.2* | 0.005 | |
| Non-PPDS | PPDS | ||
|---|---|---|---|
| N = 97 | N = 23 | p Value | |
| HGB (g/dL) | 13.0 ± 1.2 | 13.3 ± 0.9 | 0.363 |
| Ferritin (ng/mL) | 58.0 ± 51.9 | 57.7 ± 48.4 | 0.977 |
| 25(OH)D3 (ng/mL) | 24.0 ± 8.0 | 26.3 ± 7.1 | 0.197 |
| Retinol (μmol/L) | 1.3 ± 0.4 | 1.5 ± 0.5 | 0.050 |
| α-tocopherol (μmol/L) | 26.0 ± 6.6 | 24.9 ± 8.6 | 0.496 |
| Riboflavin (ng/mL) | 7.2 ± 2.0 | 6.2 ± 2.1 * | 0.036 |
| Fatty Acids | Non-PPDS | PPDS | |
|---|---|---|---|
| % of total FAs | N = 97 | N = 23 | p value |
| Total saturated fatty acids | 41.45 ± 0.81 | 41.59 ± 0.63 | 0.432 |
| C12:0 | 0.08 ± 0.02 | 0.08 ± 0.02 | 0.492 |
| C14:0 | 0.17 ± 0.05 | 0.17 ± 0.04 | 0.986 |
| C15:0 | 0.09 ± 0.02 | 0.09 ± 0.02 | 0.875 |
| C16:0 | 22.63 ± 0.70 | 22.62 ± 0.50 | 0.926 |
| C17:0 | 0.27 ± 0.03 | 0.27 ± 0.03 | 0.940 |
| C18:0 | 14.41 ± 0.75 | 14.48 ± 0.54 | 0.740 |
| C20:0 | 0.30 ± 0.04 | 0.30 ± 0.03 | 0.699 |
| C22:0 | 1.09 ± 0.12 | 1.09 ± 0.12 | 0.939 |
| C24:0 | 2.40 ± 0.29 | 2.50 ± 0.32 | 0.971 |
| Total monounsaturated fatty acids | 17.50 ± 0.79 | 17.37 ± 0.60 | 0.469 |
| C14:1n-5 | 0.04 ± 0.03 | 0.04 ± 0.02 | 0.757 |
| C16:1n-9 | 0.08 ± 0.01 | 0.07 ± 0.01* | 0.007 |
| C16:1n-7 | 0.25 ± 0.11 | 0.25 ± 0.04 | 0.745 |
| C18:1n-9 | 12.46 ± 0.64 | 12.47 ± 0.50 | 0.933 |
| C18:1n-7 | 1.02 ± 0.08 | 1.01 ± 0.11 | 0.668 |
| C20:1n-9 | 0.27 ± 0.04 | 0.28 ±0.04 | 0.359 |
| C24:1n-9 | 3.38 ± 0.28 | 3.24 ±0.22* | 0.032 |
| Total polyunsaturated fatty acids | |||
| Σn-6 | 30.35 ± 1.45 | 30.97 ± 1.16 | 0.059 |
| C18:2n-6 | 11.14 ± 1.47 | 11.28 ± 0.98 | 0.668 |
| C18:3n-6 | 0.05 ± 0.02 | 0.04 ± 0.02* | 0.001 |
| C20:2n-6 | 0.48 ± 0.05 | 0.47 ± 0.05 | 0.684 |
| C20:3n-6 | 1.24 ± 0.19 | 1.34 ± 0.22* | 0.030 |
| C20:4n-6 | 14.25 ± 1.01 | 14.44 ± 0.83 | 0.408 |
| C22:4n-6 | 2.63 ± 0.38 | 2.77 ± 0.50 | 0.134 |
| C22:5n-6 | 0.56 ± 0.12 | 0.63 ± 0.14* | 0.013 |
| Σn-3 | 10.07 ± 1.06 | 9.50 ± 0.93* | 0.020 |
| C18:3n-3 | 0.15 ± 0.03 | 0.15 ± 0.04 | 0.100 |
| C20:5n-3 (EPA) | 1.61 ± 0.43 | 1.34 ± 0.34* | 0.006 |
| C22:5n-3 | 1.94 ± 0.22 | 1.91 ± 0.21 | 0.543 |
| C22:6n-3 (DHA) | 6.40 ± 0.76 | 6.11 ± 0.72 | 0.102 |
| Σn-6/Σn-3 ratio | 3.05 ± 0.42 | 3.30 ± 0.44* | 0.015 |
| C20:3n-6/C18:3n-6 | 30.57 ± 23.42 | 54.93 ± 36.76* | 0.005 |
| C20:4n-6/(C20:5n-3 + C22:6n-3) | 1.81 ± 0.27 | 1.97 ± 0.29* | 0.012 |
| C22:6n-3/C22:5n-6 | 11.90 ± 2.88 | 10.05 ± 2.33* | 0.005 |
| β | S.E a | OR | p Value | 95% CI | |
|---|---|---|---|---|---|
| 25(OH)D3 (ng/mL) | |||||
| Model 1b | 0.037 | 0.029 | 1.038 | 0.198 | 0.981–1.099 |
| Model 2 | 0.031 | 0.033 | 1.032 | 0.352 | 0.966–1.101 |
| Retinol (μmol/L) | |||||
| Model 1 | 0.998 | 0.517 | 2.714 | 0.054 | 0.985–7.480 |
| Model 2 | 1.207 | 0.618 | 3.343 | 0.051 | 0.996–11.228 |
| α-tocopherol (μmol/L) | |||||
| Model 1 | −0.024 | 0.035 | 0.976 | 0.493 | 0.912–1.045 |
| Model 2 | −0.013 | 0.040 | 0.987 | 0.739 | 0.912–1.068 |
| Riboflavin (ng/mL) | |||||
| Model 1 | −0.291 | 0.142 | 0.747* | 0.040 | 0.566–0.987 |
| Model 2 | −0.379 | 0.157 | 0.684* | 0.015 | 0.504–0.930 |
| β | S.E | OR | p Value | 95% CI | |
|---|---|---|---|---|---|
| Total saturated fatty acids | |||||
| Model 1 b | 0.002 | 0.003 | 1.002 | 0.432 | 0.997–1.008 |
| Model 2 | 0.003 | 0.003 | 1.003 | 0.364 | 0.997–1.009 |
| Total monounsaturated fatty acids | |||||
| Model 1 | −0.002 | 0.003 | 0.998 | 0.466 | 0.992–1.004 |
| Model 2 | −0.009 | 0.004 | 0.991* | 0.003 | 0.983–0.999 |
| C16:1n-9 | |||||
| Model 1 | −0.522 | 0.200 | 0.593* | 0.009 | 0.401–0.878 |
| Model 2 | −0.686 | 0.241 | 0.504* | 0.004 | 0.314–0.808 |
| C24:1n-9 | |||||
| Model 1 | −0.019 | 0.009 | 0.981* | 0.036 | 0.964–0.999 |
| Model 2 | −0.034 | 0.012 | 0.967* | 0.006 | 0.943–0.990 |
| n-6 Polyunsaturated fatty acids | |||||
| Σn-6 | |||||
| Model 1 | 0.004 | 0.002 | 1.004 | 0.055 | 1.000–1.008 |
| Model 2 | 0.005 | 0.002 | 1.005* | 0.038 | 1.000–1.009 |
| C18:3n-6 | |||||
| Model 1 | −0.386 | 0.125 | 0.680* | 0.002 | 0.532–0.869 |
| Model 2 | −0.461 | 0.148 | 0.630* | 0.002 | 0.472–0.842 |
| C20:3n-6 | |||||
| Model 1 | 0.025 | 0.012 | 1.025* | 0.036 | 1.002–1.050 |
| Model 2 | 0.021 | 0.015 | 1.022 | 0.141 | 0.993–1.051 |
| C22:5n-6 | |||||
| Model 1 | 0.042 | 0.018 | 1.043* | 0.018 | 1.007–1.079 |
| Model 2 | 0.043 | 0.021 | 1.044* | 0.045 | 1.001–1.089 |
| n-3 Polyunsaturated fatty acids | |||||
| Σn-3 | |||||
| Model 1 | −0.006 | 0.002 | 0.994* | 0.023 | 0.990–0.999 |
| Model 2 | −0.004 | 0.003 | 0.996 | 0.155 | 0.991–1.001 |
| C20:5n-3 | |||||
| Model 1 | −0.020 | 0.008 | 0.980* | 0.008 | 0.966–0.995 |
| Model 2 | −0.019 | 0.008 | 0.982* | 0.020 | 0.966–0.997 |
| C22:6n-3 | |||||
| Model 1 | −0.005 | 0.003 | 0.995 | 0.104 | 0.989–1.001 |
| Model 2 | −0.003 | 0.004 | 0.997 | 0.356 | 0.990–1.004 |
| Σn-6/Σn-3 | |||||
| Model 1 | 0.013 | 0.005 | 1.013* | 0.019 | 1.002–1.024 |
| Model 2 | 0.010 | 0.006 | 1.010 | 0.113 | 0.998–1.023 |
| C20:3n-6/C18:3n-6 | |||||
| Model 1 | 0.025 | 0.008 | 1.025* | 0.001 | 1.010–1.041 |
| Model 2 | 0.031 | 0.009 | 1.031* | 0.001 | 1.014–1.049 |
| C20:4n-6/(C20:5n-3 + C22:6n-3) | |||||
| Model 1 | 0.022 | 0.009 | 1.022* | 0.015 | 1.004–1.040 |
| Model 2 | 0.020 | 0.010 | 1.020* | 0.048 | 1.000–1.041 |
| C22:6n-3/C22:5n-6 | |||||
| Model 1 | −0.270 | 0.100 | 0.764* | 0.007 | 0.627–0.930 |
| Model 2 | −0.241 | 0.116 | 0.786* | 0.037 | 0.626–0.986 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, Y.-H.; Chen, C.-M.; Su, H.-M.; Mu, S.-C.; Chang, M.-L.; Chu, P.-Y.; Li, S.-C. Association between Postpartum Nutritional Status and Postpartum Depression Symptoms. Nutrients 2019, 11, 1204. https://doi.org/10.3390/nu11061204
Lin Y-H, Chen C-M, Su H-M, Mu S-C, Chang M-L, Chu P-Y, Li S-C. Association between Postpartum Nutritional Status and Postpartum Depression Symptoms. Nutrients. 2019; 11(6):1204. https://doi.org/10.3390/nu11061204
Chicago/Turabian StyleLin, Yu-Hung, Chiao-Ming Chen, Hui-Min Su, Shu-Ci Mu, Mei-Ling Chang, Pei-Yin Chu, and Sing-Chung Li. 2019. "Association between Postpartum Nutritional Status and Postpartum Depression Symptoms" Nutrients 11, no. 6: 1204. https://doi.org/10.3390/nu11061204
APA StyleLin, Y.-H., Chen, C.-M., Su, H.-M., Mu, S.-C., Chang, M.-L., Chu, P.-Y., & Li, S.-C. (2019). Association between Postpartum Nutritional Status and Postpartum Depression Symptoms. Nutrients, 11(6), 1204. https://doi.org/10.3390/nu11061204