Dietary Polyphenols—Important Non-Nutrients in the Prevention of Chronic Noncommunicable Diseases. A Systematic Review
Abstract
Highlights
1. Introduction
2. Chronic Noncommunicable Diseases
- cardio- and cerebrovascular diseases
- cancers with lifestyle components
- endocrine/metabolic disorders
- gastrointestinal disorders
- kidney disease
- mental/central nervous system (CNS) health
- musculoskeletal disorders
- respiratory diseases
- reproductive disorders
- dermatological disorders
2.1. Historical Background
2.2. Current Findings on NCDs
2.3. Nutritional Changes Associated with NCDs—Potential Role of Antioxidants
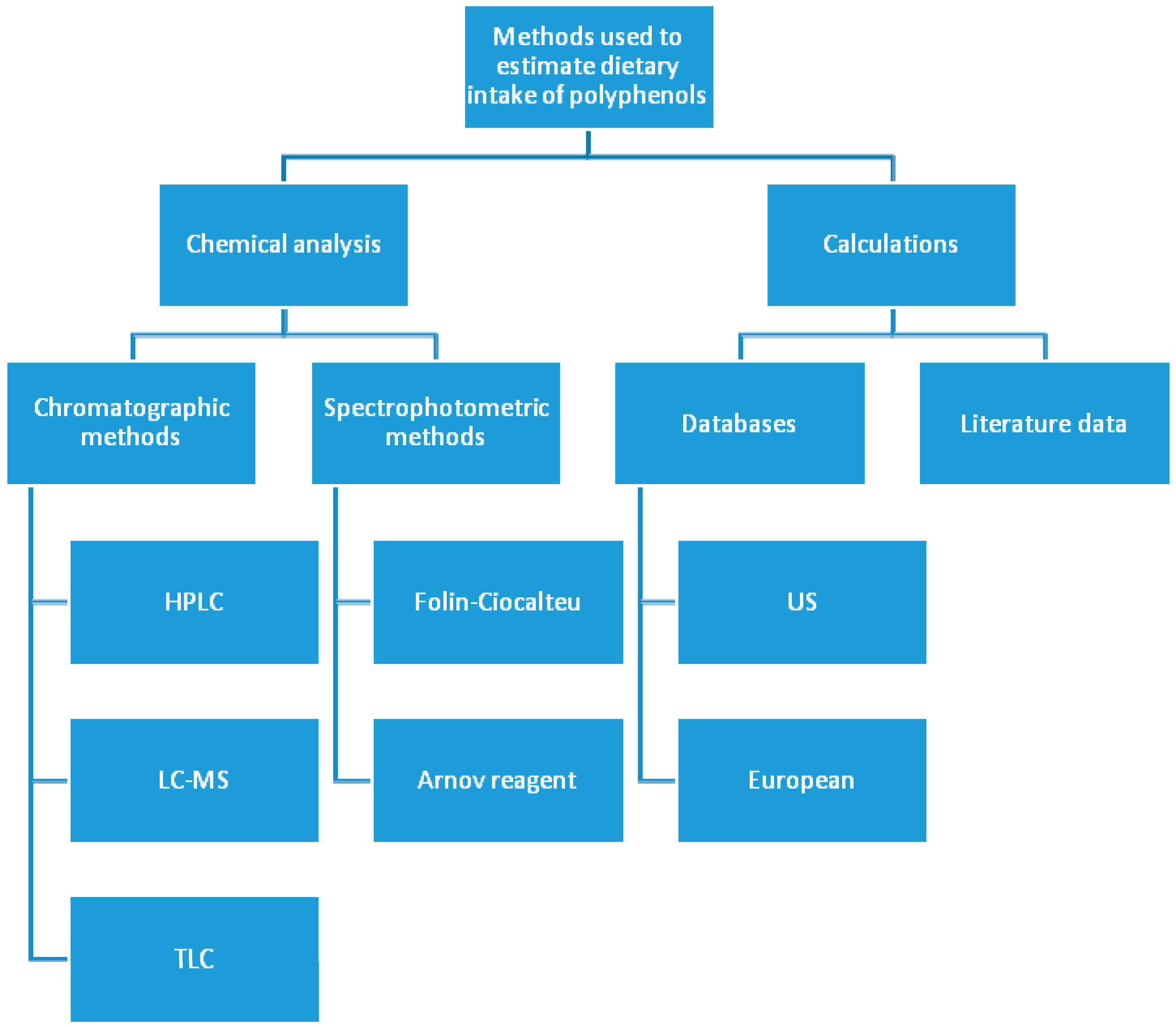
3. Polyphenols—Estimation of Daily Dietary Intake Problems
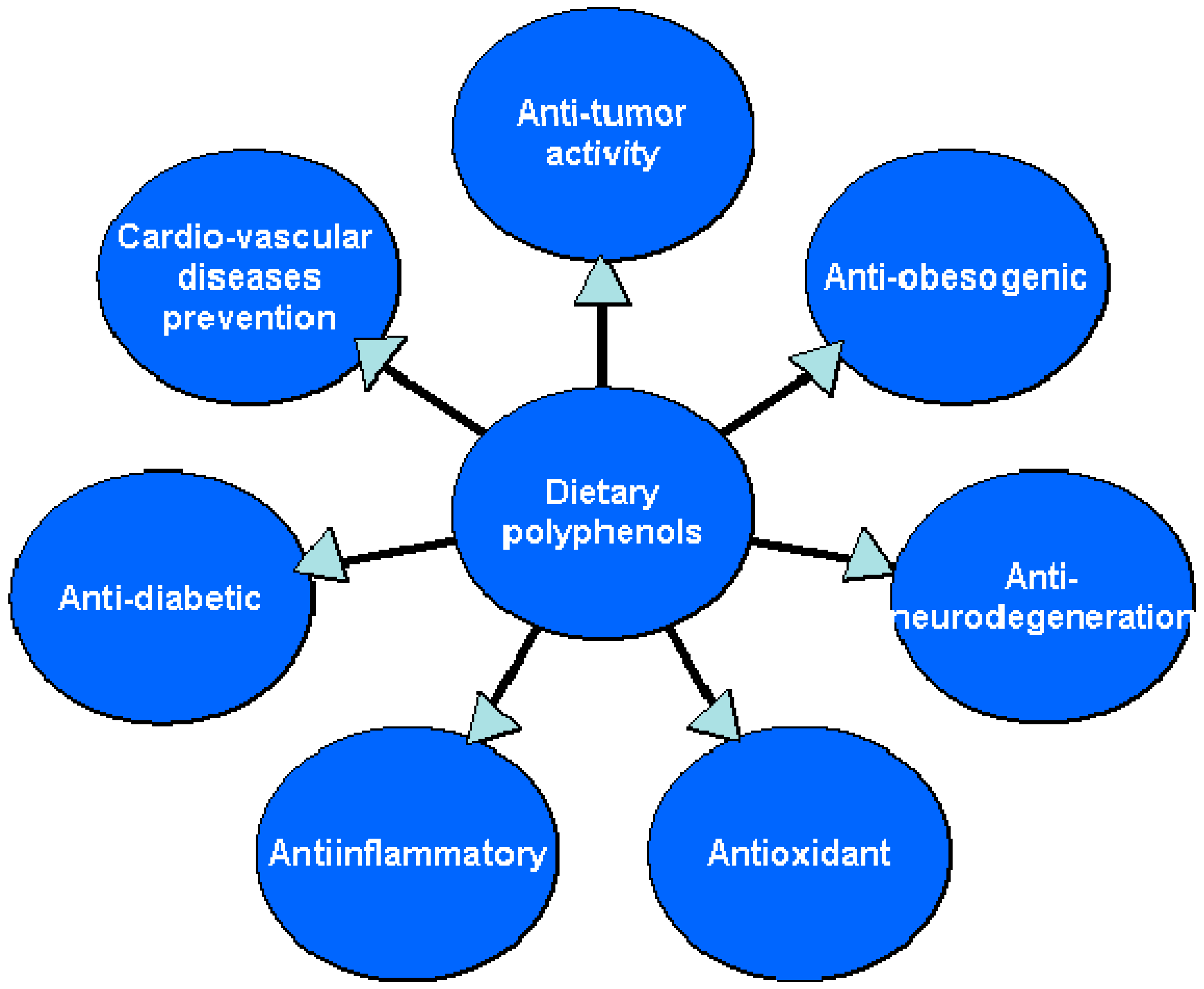
4. Dietary Polyphenols—Natural Substances Beneficial in the Prevention and Treatment of Lifestyle Diseases
4.1. Antioxidant Activities of Dietary Polyphenols
4.2. Anti-Inflammatory Effect
4.3. Anticancer Activity
4.4. Cardiovascular Diseases Prevention
4.5. Anti-Obesity Effect/Weight-Reducing Properties
4.6. Antidiabetic Activity
4.7. The Activity of Dietary Polyphenols toward CNS Disorders
4.7.1. Parkinson’s Disease
4.7.2. Alzheimer’s Disease
5. Conclusions
Funding
List of Abbreviations
| AD | Alzheimer’s disease |
| AMPK | 5’ adenosine monophosphate-activated protein kinase |
| APP | amyloid precursor protein |
| b.w. | body weight |
| C | (+)-catechin |
| CNS | central nervous system |
| ECG | epicatechin-3-gallate |
| EDRF | endothelium-derived relaxing factor |
| EGC | epigallocatechin |
| EGCG | (-)-epigallocatechin-3-gallate |
| G6Pase | glucose-6-phosphatase |
| GC | (-)-gallocatechin |
| GCG | (-)-gallocatechin gallate |
| GK | glucokinase |
| GLUT 2 and 4 | glucose transporters type 2 and 4 |
| HMG-CoA | 3-hydroxy-3-methylglutaryl-coenzyme |
| IRS-1 | insulin receptor substrate 1 |
| MPO | myeloperoxidase |
| MPTP | N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| NO | nitric oxide |
| N-Sase | neutral sphingomyelin-specific phospholipase C |
| 6-OHDA | 6-hydroxyydopamine |
| PD | Parkinson’s disease |
| PEPCK | Phosphoenolpyruvate carboxykinase |
| PI3K | phosphoinositide-3-kinase |
| PKC | protein kinase C |
| PPARγ | peroxisome proliferator-associated receptor |
| RCTs | randomized controlled trials |
| ROS | reactive oxygen species |
| sAPP | soluble form of APP |
| SGLT 1 | sodium-glucose co-transporter type 1 |
| SOD | SuperOxide Dismutase |
| STZ | streptozocin |
| TAGs | Triglycerides |
| TBARS | thiobarbituric acid reactive substances |
| TF | theaflavin |
| TFs | theaflavins |
| TRs | thearubigins |
References
- Carrera-Bastos, P.; Fontes-Villalba, M.; O’Keefe, J.H.; Lindeberg, S.; Cordain, L. The western diet and lifestyle and diseases of civilization. Res. Rep. Clin. Cardiol. 2011, 2, 15–35. [Google Scholar] [CrossRef]
- Betlejewski, S. Social diseases, civilization diseases or lifestyle diseases? Wiad. Lek. 2007, 60, 489–492. [Google Scholar]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef]
- Booth, F.W.; Lees, S.J. Fundamental questions about genes, inactivity, and chronic diseases. Physiol. Genom. 2007, 28, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Egger, G.; Dixon, J. Beyond obesity and lifestyle: A review of 21st century chronic diseases determinants. BioMed Res. Int. 2014, 2014, 731685. [Google Scholar] [CrossRef]
- Lindeberg, S. Food and Western Disease: Health and Nutrition from an Evolutionary Perspective; Wiley-Blackwell: Chichester, UK, 2010. [Google Scholar]
- Eaton, S.B.; Konner, M.; Shostak, M. Stone agers in the fast lane: Chronic degenerative diseases in evolutionary perspective. Am. J. Med. 1988, 84, 739–749. [Google Scholar] [CrossRef]
- Jönsson, T.; Olsson, S.; Ahrén, B.; Bøg-Hansen, T.C.; Dole, A.; Lindeberg, S. Agrarian diet and diseases of affluence: Do evolutionary novel dietary lectins cause leptin resistance? BMC Endocr. Disord. 2005, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Cordain, L. Implications of Plio-Pleistocene hominin diets for modern humans. In Evolution of the Human Diet: The Known, the Unknown, and the Unknowable; Ungar, P., Ed.; Oxford University Press: New York, NY, USA, 2007; pp. 363–383. [Google Scholar]
- Cordain, L. Cereal grains: Humanity’s double-edged sword. World Rev. Nutr. Diet 1999, 84, 19–73. [Google Scholar]
- Chaplin, G.; Jablonski, N.G. Vitamin D and the evolution of human depigmentation. Am. J. Phys. Anthropol. 2009, 139, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Vieth, R. What is the optimal vitamin D status for health? Prog. Biophys. Mol. Biol. 2006, 92, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Deutch, B.; Dyerberg, J.; Pedersen, H.S.; Aschlund, E.; Hansen, J.C. Traditional and modern Greenlandic food: Dietary composition, nutrients and contaminants. Sci. Total Environ. 2007, 384, 106–119. [Google Scholar] [CrossRef]
- Kuhnlein, H.V.; Receveur, O. Local cultural animal food contributes high levels of nutrients for Arctic Canadian indigenous adults and children. J. Nutr. 2007, 137, 1110–1114. [Google Scholar] [CrossRef]
- Wiley, T.S.; Formby, B. Lights Out: Sleep, Sugar and Survival; Pocket Books: New York, NY, USA, 2000. [Google Scholar]
- Rook, G.A. 99th Dahlem conference on infection, inflammation and chronic inflammatory disorders: Darwinian medicine and the ‘hygiene’ or ‘old friends’ hypothesis. Clin. Exp. Immunol. 2010, 160, 70–79. [Google Scholar] [CrossRef]
- Booth, F.W.; Chakravarthy, M.V.; Gordon, S.E.; Spangenburg, E.E. Waging war on physical inactivity: Using modern molecular ammunition against an ancient enemy. J. Appl. Physiol. 2002, 93, 3–30. [Google Scholar] [CrossRef] [PubMed]
- Pettee, K.K.; Ainsworth, B.E. The Building Healthy Lifestyles Conference: Modifying lifestyles to enhance physical activity, diet, and reduce cardiovascular disease. Am. J. Lifestyle Med. 2009, 3, 6–10. [Google Scholar] [CrossRef]
- Cordain, L.; Gotshall, R.W.; Eaton, S.B.; Eaton, S.B., 3rd. Physical activity, energy expenditure and fitness: An evolutionary perspective. Int. J. Sports Med. 1998, 19, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Eaton, S.B. The ancestral human diet: What was it and should it be a paradigm for contemporary nutrition? Proc. Nutr. Soc. 2006, 65, 1–6. [Google Scholar] [CrossRef]
- Fox, L.C.; Juan, J.; Albert, R.M. Phytolith analysis on dental calculus, enamel surface, and burial soil: Information about diet and paleoenviroment. Am. J. Phys. Anthropol. 1996, 101, 101–113. [Google Scholar] [CrossRef]
- Cordain, L.; Brand Miller, J.; Eaton, S.B.; Mann, N.; Holt, S.H.A.; Speth, J.D. Plant to animal subsistence ratios and macronutrient energy estimations in worldwide huntergatherer diets. Am. J. Clin. Nutr. 2000, 71, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Kuipers, R.S.; Luxwolda, M.F.; Janneke Dijck-Brouwer, D.A.; Eaton, S.B.; Crawford, M.A.; Cordain, L.; Muskiet, F.A. Estimated macronutrient and fatty acid intakes from an East African Paleolithic diet. Br. J. Nutr. 2010, 104, 1666–1687. [Google Scholar] [CrossRef] [PubMed]
- McKeown, T. The Origins of Human Disease; Basil Blackwell: New York, NY, USA, 1998. [Google Scholar]
- Sanders, J.W.; Fuhrer, G.S.; Johnson, M.D.; Riddle, M.S. The epidemiological transition: The current status of infectious diseases in the developed world versus the developing world. Sci. Prog. 2008, 91, 1–38. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose expression of tumor necrosis factor-α: Direct role in obesity-linked insulin resistance. Science 1993, 259, 87–91. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef]
- Libby, P. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 65, 140–146. [Google Scholar] [CrossRef]
- Gregor, M.F.; Hotamisligil, G.S. Inflammatory mechanisms in obesity. Ann. Rev. Immunol. 2011, 29, 415–445. [Google Scholar] [CrossRef]
- Hanson, G.K. Inflammation, atherosclerosis and coronary artery disease. N. Engl. J. Med. 2005, 352, 1658–1695. [Google Scholar] [CrossRef]
- Egger, G.; Dixon, J. Inflammatory effects of nutritional stimuli: Further support for the need for a big picture approach to tackling obesity and chronic disease. Obes. Rev. 2010, 11, 137–149. [Google Scholar] [CrossRef]
- Egger, G.; Dixon, J. Non-nutrient causes of low-grade, systemic inflammation: Support for a “canary in the mineshaft” view of obesity in chronic disease. Obes. Rev. 2011, 12, 339–345. [Google Scholar] [CrossRef]
- Egger, G. In search of a “germ theory” equivalent for chronic Disease. Prev. Chronic Dis. 2012, 9, 1–7. [Google Scholar] [CrossRef]
- Black, P.H. The inflammatory response is an integral part of the stress response: Implications for atherosclerosis, insulin resistance, type II diabetes and metabolic syndrome X. Brain Behav. Immun. 2003, 17, 350–364. [Google Scholar] [CrossRef]
- Mills, N.L.; Donaldson, K.; Hadoke, P.W.; Boon, N.A.; MacNee, W.; Cassee, F.R.; Sandström, T.; Blomberg, A.; Newby, D.E. Adverse cardiovascular effects of air pollution. Nat. Clin. Pract. Cardiovasc. Med. 2009, 6, 36–44. [Google Scholar] [CrossRef]
- Vardavas, C.I.; Panagiotakos, D.B. The causal relationship between passive smoking and inflammation on the development of cardiovascular disease: A review of the evidence. Inflamm. Allergy Drug Targets 2009, 8, 328–333. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R. Inflammatory mechanisms in the regulation of insulin resistance. Mol. Med. 2008, 14, 222–231. [Google Scholar] [CrossRef]
- Kolb, H.; Mandrup-Poulsen, T. The global diabetes epidemic as a consequence of lifestyle-induced low-grade inflammation. Diabetologia 2010, 53, 10–20. [Google Scholar] [CrossRef]
- Tasali, E.; Leproult, R.; Spiegel, K. Reduced sleep duration or quality: Relationships with insulin resistance and type 2 diabetes. Prog. Cardiovasc. Dis. 2009, 51, 381–391. [Google Scholar] [CrossRef]
- Krueger, P.M.; Friedman, E.M. Sleep duration in the United States: A cross-sectional population-based study. Am. J. Epidemiol. 2009, 169, 1052–1063. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- World Health Organization. Diet, Nutrition, and the Prevention of Chronic Diseases; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Nicholas, L.; Roberts, D.; Pond, D. The role of the general practitioner and the dietitian in patient nutrition management. Asia Pac. J. Clin. Nutr. 2003, 12, 3–8. [Google Scholar]
- Australian Institute of Health and Welfare, Chronic Diseases. Available online: http://www.aihw.gov.au/chronic-diseases (accessed on 3 March 2018).
- Galland, L. Diet and inflammation. Nutr. Clin. Pract. 2010, 25, 634–664. [Google Scholar] [CrossRef]
- Barbaresko, J.; Koch, M.; Schulze, M.B.; Nothlings, U. Dietary pattern analysis and biomarkers of low-grade inflammation: A systematic literature review. Nutr. Rev. 2013, 71, 511–527. [Google Scholar] [CrossRef]
- O’Neil, C.E.; Keast, D.R.; Fulgoni, V.L.; Nicklas, T.A. Food sources of energy and nutrients among adults in the US: NHANES 2003-2006. Nutrients 2012, 4, 2097–2120. [Google Scholar] [CrossRef]
- Cordain, L.; Eades, M.R.; Eades, M.D. Hyperinsulinemic diseases of civilization: More than just Syndrome X. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2003, 136, 95–112. [Google Scholar] [CrossRef]
- Barclay, A.W.; Petocz, P.; McMillan-Price, J.; Flood, V.M.; Prvan, T.; Mitchell, T.; Brand-Miller, J.C. Glycemic index, glycemic load, and chronic disease risk: A meta-analysis of observational studies. Am. J. Clin. Nutr. 2008, 87, 627–637. [Google Scholar] [CrossRef]
- Alas-Salvadó, J.; Farrés, X.; Luque, X.; Narejos, S.; Borrel, M.; Basora, J.; Anguera, A.; Torres, F.; Bullo, M.; Balanza, R. Effect of two doses of a mixture of soluble fibres on body weight and metabolic variables in overweight or obese patients: A randomised trial. Br. J. Nutr. 2008, 99, 1380–1387. [Google Scholar] [CrossRef]
- Tarini, J.; Wolever, T.M. The fermentable fibre inulin increases postprandial serum short-chain fatty acids and reduces free fatty acids and ghrelin in healthy subjects. Appl. Physiol. Nutr. Metab. 2010, 35, 9–16. [Google Scholar] [CrossRef]
- National Health and Nutrition Examination Survey. Available online: http://www.cdc.gov/nchs/nhanes.htm (accessed on 7 April 2018).
- Danaei, G.; Ding, E.L.; Mozaffarian, D.; Taylor, B.; Rehm, J.; Murray, C.J.; Ezzati, M. The preventable causes of death in the United States: Comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med. 2009, 6, e1000058. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhang, Y.; Wang, M.; Li, X.; Xia, M.; Ling, W. Dietary protein and plasma total homocysteine, cysteine concentrations in coronary angiographic subjects. Nutr. J. 2013, 12, 144. [Google Scholar] [CrossRef]
- Miller, D.B.; O’Callaghan, J.P. Biomarkers of Parkinson’s disease: Present and future. Metabolism 2015, 64, 40–46. [Google Scholar] [CrossRef]
- Ceriello, A.; Motz, E. Is oxidative stress the pathogenic mechanism underlying insulin resistance, diabetes, and cardiovascular disease? The common soil hypothesis revisited. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 816–823. [Google Scholar] [CrossRef]
- De la Monte, S.M.; Neely, T.R.; Cannon, J. Oxidative stress and hypoxia, molecular abnormalities in central nervous system neurons. Cell. Mol. Life Sci. 2000, 57, 1471–1481. [Google Scholar] [CrossRef]
- Robertson, R.P.; Harmon, J.; Tran, P.O.; Tanaka, Y.; Takahashi, H. Glucose toxicity in-cells: Type 2 diabetes, good radicals gone bad, and the glutathione connection. Diabetes 2003, 52, 581–587. [Google Scholar] [CrossRef]
- Paolisso, G.; Giugliano, D. Oxidative stress and insulin action. Is there a relationship? Diabetologia 1996, 39, 357–363. [Google Scholar] [CrossRef]
- Kinkade, K.; Streeter, J.; Miller, F.J., Jr. Inhibition of NADPH oxidase by apocynin attenuates progression of atherosclerosis. Int. J. Mol. Sci. 2013, 14, 17017–17028. [Google Scholar] [CrossRef]
- Sugiyama, S.; Kugiyama, K.; Aikawa, M.; Nakamura, S.; Ogawa, H.; Libby, P. Hypochlorous acid, a macrophage product, induces endothelial apoptosis and tissue factor expression: Involvement of myeloperoxidase-mediated oxidant in plaque erosion and thrombogenesis. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1309–1314. [Google Scholar] [CrossRef]
- Csanyi, G.; Yao, M.; Rodriguez, A.I.; Al Ghouleh, I.; Sharifi-Sanjani, M.; Frazziano, G.; Huang, X.; Kelley, E.E.; Isenberg, J.S.; Pagano, P.J. Thrombospondin-1 regulates blood flow via CD47 receptor-mediated activation of NADPH oxidase 1. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2966–2973. [Google Scholar] [CrossRef]
- Touyz, R.M.; Briones, A.M. Reactive oxygen species and vascular biology: Implications in human hypertension. Hypertens. Res. 2011, 34, 5–14. [Google Scholar] [CrossRef]
- Heitzer, T.; Schlinzig, T.; Krohn, K.; Meinertz, T.; Münzel, T. Endothelial dysfunction, oxidative stress, and risk of cardiovascular events in patients with coronary artery disease. Circulation 2001, 104, 2673–2678. [Google Scholar] [CrossRef]
- Koch, W.; Baj, T.; Kukula-Koch, W.; Marzec, Z. Dietary intake of specific phenolic compounds and their effect on the antioxidant activity of daily food rations. Open Chem. 2015, 13, 869–876. [Google Scholar] [CrossRef]
- Huxley, R.R.; Neil, H.A. The relation between dietary flavonol intake and coronary heart disease mortality: A meta-analysis of prospective cohort studies. Eur. J. Clin. Nutr. 2003, 57, 904–908. [Google Scholar] [CrossRef]
- Jarząb, A.; Kukula-Koch, W. Recent advances in obesity: The role of turmeric tuber and its metabolites in the prophylaxis and therapeutical strategies. Curr. Med. Chem. 2017, 24, 1. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.J.; Myung, S.-K.; Chung, S.T.; Lee, Y.K.; Jeon, Y.-J.; Park, C.-H.; Seo, H.G.; Huh, B.Y. Effects of vitamin treatment or supplements with purported antioxidant properties on skin cancer prevention: A meta-analysis of randomized controlled trials. Dermatology 2011, 223, 36–44. [Google Scholar] [CrossRef]
- Puertollano, M.A.; Puertollano, E.; de Cienfuegos, G.Á.; de Pablo, M.A. Dietary antioxidants: Immunity and host defense. Curr. Top. Med. Chem. 2011, 11, 1752–1766. [Google Scholar] [CrossRef]
- Katta, R.; Brown, D.C. Diet and skin cancer: The potential role of dietary antioxidants in nonmelanoma skin cancer prevention. J. Skin Cancer 2015, 893149. [Google Scholar] [CrossRef] [PubMed]
- Lugasi, A.; Hóvári, J.; Sági, K.V.; Bíró, L. The role of antioxidant phytonutrients in the prevention of diseases. Acta Biol. Szeged. 2003, 47, 119–125. [Google Scholar]
- Grosso, G.; Micek, A.; Godos, J.; Pajak, A.; Sciacca, S.; Galvano, F.; Giovannucci, E.L. Dietary flavonoid and lignan intake and mortality in prospective cohort studies: Systematic review and dose-response meta-analysis. Am. J. Epidemiol. 2017, 1–13. [Google Scholar] [CrossRef]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef]
- Koes, R.; Verweij, W.; Quattrocchio, F. Flavonoids: A colourful model for the regulation and evolution of biochemical pathways. Trends Plant Sci. 2005, 10, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Hertog, M.G.L.; Hollman, P.C.H.; Venema, D.P. Optimization of a quantitative HPLC determination of potentially anticarcinogenic flavonoids in vegetables and fruits. J. Agric. Food Chem. 1992, 40, 1591–1598. [Google Scholar] [CrossRef]
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomás-Barberán, F.A.; Datta, N.; Singanusong, R.; Chen, S.S. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef]
- Kühnau, J. The flavonoids, a class of semi-essential food components: Their role in human nutrition. World Rev. Nutr. Diet. 1976, 24, 117–191. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Clifford, M.N. Flavanones, chalcones and dihydrochalcones-nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1073–1080. [Google Scholar] [CrossRef]
- Justesen, U.; Knuthsen, P.; Leth, T. Determination of plant polyphenols in Danish foodstuffs by HPLC-UV and LC-MS detection. Cancer Lett. 1997, 114, 165–167. [Google Scholar] [CrossRef]
- Knekt, P.; Jarvinen, R.; Revnanen, A.; Moatela, J. Flavonoid intake and coronary mortality in Finland: A cohort study. BMJ 1996, 312, 478–481. [Google Scholar] [CrossRef]
- Hertog, M.G.L.; Kromhout, D.; Aravanis, C.; Blackburn, H.; Buzina, R.; Fidanza, F.; Giampaoli, S.; Jansen, A.; Menotti, A.; Nedeljkovic, S. Flavonoid intake and long term risk of coronary heart disease and cancer in the Seven Countries Study. Arch. Intern. Med. 1995, 155, 381–386. [Google Scholar] [CrossRef]
- Rimm, E.B.; Katan, M.B.; Ascherio, A.; Stampfer, M.J.; Willett, W.C. Relation between intake of flavonoids and risk for coronary heart disease in male health professionals. Ann. Intern. Med. 1996, 12, 384–389. [Google Scholar] [CrossRef]
- Pozzo, V.; Goitre, I.; Fadda, M.; Gambion, R.; De Francesco, A.; Soldati, L.; Gentile, L.; Magistroni, P.; Cassader, M.; Bo, S. Dietary flavonoid intake and cardiovascular risk: A population-based cohort study. J. Transl. Med. 2015, 13, 218. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. A systematic analysis of the content of 502 polyphenols in 452 foods and beverages—An application of the Phenol-Explorer database. J. Agric. Food Chem. 2010, 58, 4959–4969. [Google Scholar] [CrossRef] [PubMed]
- Neveu, V.; Pérez-Jiménez, J.; Vos, F.; Crespy, V.; Du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010, 64, 112–120. [Google Scholar] [CrossRef]
- Mishra, A.; Kumar, S.; Pandey, A.K. Scientific validation of the medicinal efficacy of Tinospora cordifolia. Sci. World. J. 2013, 292934. [Google Scholar] [CrossRef]
- Kukula-Koch, W.; Aligiannis, N.; Halabalaki, M.; Skaltsounis, A.L.; Glowniak, K.; Kalpoutzakis, E. Influence of extraction procedures on phenolic content and antioxidant activity of Cretan barberry herb. Food Chem. 2013, 138, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mishra, A. Chemistry and biological activities of flavonoids: An overview. Sci. World. J. 2013, 162750. [Google Scholar] [CrossRef]
- Pinto, M.S. Tea: A new perspective on health benefits. Food Res. Int. 2013, 53, 558–567. [Google Scholar] [CrossRef]
- Bruno, R.S.; Bomser, J.A.; Ferruzzi, M.G. Antioxidant Capacity of Green Tea (Camellia sinensis). In Processing and Impact on Antioxidants in Beverages; Preedy, V.R., Ed.; Academic Press: Oxford, UK, 2014. [Google Scholar]
- Kukula-Koch, W.; Koch, W.; Angelis, A.; Halabalaki, M.; Aligiannis, N. Application of pH-zone refining hydrostatic countercurrent chromatography (hCCC) for the recovery of antioxidant phenolics and the isolation of alkaloids from Siberian barberry herb. Food Chem. 2016, 203, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Nah, J.; Chun, S.; Park, H.; Yang, S.E.; Min, W.K. In vivo antioxidant effect of green tea. Eur. J. Clin. Nutr. 2000, 54, 527–529. [Google Scholar] [CrossRef]
- Arent, S.M.; Senso, M.; Golem, D.L. The effects of theaflavin-enriched black tea extract on muscle soreness, oxidative stress, inflammation, and endocrine responses to acute anaerobic interval training: A randomized, double-blind, crossover study. J. Int. Soc. Sport. Nutr. 2010, 7, 11. [Google Scholar] [CrossRef]
- Imran, A.; Arshad, M.U.; Arshad, M.S.; Imran, M.; Saeed, F.; Sohaib, M. Lipid peroxidation diminishing perspective of isolated theaflavins and thearubigins from black tea in arginine induced renal malfunctional rats. Lipid. Health. Dis. 2018, 17, 157. [Google Scholar] [CrossRef]
- Tsai, P.H.; Kan, N.B.; Ho, S.C.; Liu, C.C.; Lin, C.C. Effects of oolong tea supplementationon lipid peroxidation of athletes at rest and post-exhaustive exercise. J. Food Sci. 2005, 70, 581–585. [Google Scholar] [CrossRef]
- Weerawatanakorn, M.; Hung, W.-L.; Pan, M.-H.; Li, S.; Li, D.; Wan, X.; Ho, C.-T. Chemistry and health beneficial effects of oolong tea and theasinensins. Food Sci. Hum. Well. 2015, 4, 133–146. [Google Scholar] [CrossRef]
- Pae, M.; Wu, D. Immunomodulating effect of epigallocatechin-3-gallate from green tea: Mechanisms and applications. Food Funct. 2013, 4, 1287–1303. [Google Scholar] [CrossRef]
- Wu, D. Green tea EGCG, T-cell function, and T-cell-mediated autoimmune encephalomyelitis. J. Investig. Med. 2016, 64, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Jung, Y.O.; Ryu, J.G.; Oh, H.J.; Son, H.J.; Lee, S.H.; Kwon, J.E.; Kim, E.K.; Park, M.K.; Park, S.H.; et al. Epigallocatechin-3-gallate ameliorates autoimmune arthritis by reciprocal regulation of T helper-17 regulatory T cells and inhibition of osteoclastogenesis by inhibiting STAT3 signaling. J. Leucoc. Biol. 2016, 100, 559–568. [Google Scholar] [CrossRef]
- Nikfarjam, B.A.; Adineh, M.; Hajiali, F.; Nassiri-Asl, M. Treatment with rutin—A therapeutic strategy for neutrophil-mediated inflammatory and autoimmune diseases: Anti-inflammatory effects of rutin on neutrophils. J. Pharmacopunct. 2017, 20, 52–56. [Google Scholar] [CrossRef]
- Nikfarjam, B.A.; Hajiali, F.; Adineh, M.; Nassiri-Asl, M. Anti-inflammatory Effects of Quercetin and Vitexin on Activated Human Peripheral Blood Neutrophils. J. Pharmacopunct. 2017, 20, 127–131. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, X.; Sun, C.; Yang, J.; Wang, L.; Liu, J.; Gong, L.; Jing, Y. Apigenin attenuates experimental autoimmune myocarditis by modulating Th1/Th2 cytokine balance in mice. Inflammation 2016, 39, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Duan, W.; Owusu, L.; Wu, D.; Xin, Y. Epigallocatechin-3-gallate induces the apoptosis of hepatocellular carcinoma LM6 cells but not non-cancerous liver cells. Int. J. Mol. Med. 2015, 35, 117–124. [Google Scholar] [CrossRef]
- Sur, S.; Pal, D.; Roy, R.; Barua, A.; Roy, A.; Saha, P.; Panda, C.K. Tea polyphenols EGCG and TF restrict tongue and liver carcinogenesis simultaneously induced by N-nitrosodiethylamine in mice. Toxicol. Appl. Pharmacol. 2016, 300, 34–46. [Google Scholar] [CrossRef]
- Kalra, N.; Seth, K.; Prasad, S.; Singh, M.; Pant, A.B.; Shukla, Y. Theaflavins induced apoptosis of LNCaP cells is mediated through induction of p53, down-regulation of NF-kappa B and mitogen-activated protein kinases pathways. Life Sci. 2007, 80, 2137–2146. [Google Scholar] [CrossRef]
- Prasad, S.; Kaur, J.; Roy, P.; Kalra, N.; Shukla, Y. Theaflavins induce G2/M arrest by modulating expression of p21waf1/cip1, cdc25C and cyclin B in human prostate carcinoma PC-3 cells. Life Sci. 2007, 81, 1323–1331. [Google Scholar] [CrossRef]
- He, H.-F. Research progress on theaflavins: Efficacy, formation, and preparation. Food Nutr. Res. 2017, 61, 1344521. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Zhao, X.; Xu, J.; Zhang, H. Quercetin inhibits angiogenesis-mediated human retinoblastoma growth by targeting vascular endothelial growth factor receptor. Oncol. Lett. 2017, 14, 3343–3348. [Google Scholar] [CrossRef] [PubMed]
- Hashemzaei, M.; Far, A.D.; Yari, A.; Heravi, R.E.; Tabrizian, K.; Taghdisi, S.M.; Sadegh, S.E.; Tsarouhas, K.; Kouretas, D.; Tzanakakis, G.; et al. Anticancer and apoptosis-inducing effects of quercetin in vitro and in vivo. Oncol. Rep. 2017, 38, 819–828. [Google Scholar] [CrossRef]
- Dai, J.; Van Wie, P.G.; Fai, L.Y.; Kim, D.; Wang, L.; Poyil, P.; Luo, J.; Zhang, Z. Downregulation of NEDD9 by apigenin suppresses migration, invasion, and metastasis of colorectal cancer cells. Toxicol. Appl. Pharmacol. 2016, 311, 106–112. [Google Scholar] [CrossRef]
- Tseng, T.H.; Chien, M.H.; Lin, W.L.; Wen, Y.C.; Chow, J.M.; Chen, C.K.; Kuo, T.C.; Lee, W.J. Inhibition of MDA-MB-231 breast cancer cell proliferation and tumor growth by apigenin through induction of G2/M arrest and histone H3 acetylation-mediated p21WAF1/CIP1 expression. Environ. Toxicol. 2017, 32, 434–444. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Fu, P.; Gupta, S. Apigenin induces apoptosis by targeting inhibitor of apoptosis proteins and Ku70-Bax interaction in prostate cancer. Apoptosis 2014, 19, 883–894. [Google Scholar] [CrossRef]
- Shukla, S.; Shankar, E.; Fu, P.; MacLennan, G.T.; Gupta, S. Suppression of NF-κB and NF-κB-Regulated gene expression by apigenin through IκBα and IKK pathway in TRAMP mice. PLoS ONE 2015, 10, e0138710. [Google Scholar] [CrossRef]
- Cao, H.H.; Chu, J.H.; Kwan, H.Y.; Su, T.; Yu, H.; Cheng, C.Y.; Fu, X.Q.; Guo, H.; Li, T.; Tse, A.K.; et al. Inhibition of the STAT3 signaling pathway contributes to apigenin-mediated anti-metastatic effect in melanoma. Sci. Rep. 2016, 6, 21731. [Google Scholar] [CrossRef]
- Hu, X.W.; Meng, D.; Fang, J. Apigenin inhibited migration and invasion of human ovarian cancer A2780 cells through focal adhesion kinase. Carcinogenesis 2008, 29, 2369–2376. [Google Scholar] [CrossRef]
- Meng, S.; Zhu, Y.; Li, J.F.; Wang, X.; Liang, Z.; Li, S.Q.; Xu, X.; Chen, H.; Liu, B.; Zheng, X.Y.; et al. Apigenin inhibits renal cell carcinoma cell proliferation. Oncotarget 2017, 8, 19834–19842. [Google Scholar] [CrossRef]
- Banjerdpongchai, R.; Wudtiwai, B.; Khaw-on, P.; Rachakhom, W.; Duangnil, N.; Kongtawelert, P. Hesperidin from Citrus seed induces human hepatocellular carcinoma HepG2 cell apoptosis via both mitochondrial and death receptor pathways. Tumour Biol. 2016, 37, 227–237. [Google Scholar] [CrossRef]
- Lee, C.J.; Wilson, L.; Jordan, M.A.; Nguyen, V.; Tang, J.; Smiyun, G. Hesperidin suppressed proliferations of both human breast cancer and androgen-dependent prostate cancer cells. Phytother. Res. 2010, 24, 15–19. [Google Scholar] [CrossRef]
- Zhao, J.; Li, Y.; Gao, J.; De, Y. Hesperidin inhibits ovarian cancer cell viability through endoplasmic reticulum stress signaling pathways. Oncol. Lett. 2017, 14, 5569–5574. [Google Scholar] [CrossRef][Green Version]
- González-Sarrías, A.; Combet, E.; Pinto, P.; Mena, P.; Dall’Asta, M.; Garcia-Aloy, M.; Rodríguez-Mateos, A.; Gibney, E.R.; Dumont, J.; Massaro, M.; et al. A systematic review and meta-analysis of the effects of flavanol-containing tea, cocoa and apple products on body composition and blood lipids: Exploring the factors responsible for variability in their efficacy. Nutrients 2017, 9, 746. [Google Scholar] [CrossRef]
- Bogdanski, P.; Suliburska, J.; Szulinska, M.; Stepien, M.; Pupek-Musialik, D.; Jablecka, A. Green tea extract reduces blood pressure, inflammary biomarkers, and oxidative stress and improves, parameters associated with insulin resistance in obese, hypertensive patients. Nutr. Res. 2012, 32, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Ademiluyi, A.O.; Oboh, G.; Ogunsuyi, O.B.; Oloruntoba, F.M. A comparative study on antihypertensive and antioxidant properties of phenolic extracts from fruit and leaf of some guava (Psidium guajava L.) varieties. Comp. Clin. Pathol. 2016, 25, 363–374. [Google Scholar] [CrossRef]
- Tounekti, T.; Joubert, E.; Hernandez, I.; Munne-Bosch, S. Improving the polyphenol content of tea. Crit. Rev. Plant Sci. 2013, 32, 192–215. [Google Scholar] [CrossRef]
- Gómez-Guzmán, M.; Jiménez, R.; Sánchez, M.; Zarzuelo, M.J.; Galindo, P.; Quintela, A.M.; Lopez-Sepulveda, R.; Romero, M.; Tamargo, J.; Vargas, F.; et al. Epicatechin lowers blood pressure, restores endothelial function, and decreases oxidative stress and endothelin-1 and NADPH oxidase activity in DOCA-salt hypertension. Free Radic. Biol. Med. 2012, 52, 70–79. [Google Scholar] [CrossRef]
- Mahajan, N.; Dhawan, V.; Sharma, G.; Jain, S.; Kaul, D. Induction of inflammatory gene expression by THP-1 macrophages cultured in normocholesterolaemic hypertensive sera and modulatory effects of green tea polyphenols. J. Hum. Hypertens. 2008, 22, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Q.; Hu, T.; Han, Y.; Huang, W.; Yuan, H.B.; Zhang, Y.T.; Du, Y.; Jiang, Y.W. Preventive Effects of Catechins on Cardiovascular Disease. Molecules 2016, 21, 1759. [Google Scholar] [CrossRef]
- Namkung, W.; Thiagarajah, J.R.; Phuan, P.W.; Verkman, A.S. Inhibition of Ca2+-activated Cl¯ channels by gallotannins as a possible molecular basis for health benefits of red wine and green tea. FASEB J. 2010, 24, 4178–4186. [Google Scholar] [CrossRef]
- Ohno, S.; Yokoi, H.; Mori, K.; Kasahara, M.; Kuwahara, K.; Fujikura, J.; Naito, M.; Kuwabara, T.; Imamaki, H.; Ishii, A.; et al. Ablation of the N-type calcium channel ameliorates diabetic nephropathy with improved glycemic control and reduced blood pressure. Sci. Rep. 2016, 6, 27192. [Google Scholar] [CrossRef]
- Potenza, M.A.; Marasciulo, F.L.; Tarquinio, M.; Tiravanti, E.; Colantuono, G.; Federici, A.; Kim, J.A.; Quon, M.J.; Montagnani, M. EGCG, a green tea polyphenol, improves endothelial function and insulin sensitivity, reduces blood pressure, and protects against myocardial I/R injury in SHR. Am. J. Physiol. Endocrinol. Metab. 2007, 292, 1378–1387. [Google Scholar] [CrossRef]
- Li, F.; Takahashi, Y.; Yamaki, K. Inhibitory effect of catechin-related compounds on renin activity. Biomed. Res. 2013, 34, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wang, Y.; Xie, Z.; Zhou, Y.; Zhang, Y.; Wan, X. The anti-obesity effects of green tea in human intervention and basic molecular studies. Eur. J. Clin. Nutr. 2014, 68, 1075–1087. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.S.; Lee, H.G.; Choi, Y.J.; Kim, T.G.; Cho, C.S. Proposed mechanisms of (-)-epigallocatechin-3-gallate for anti-obesity. Chem. Biol. Interact. 2007, 167, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Bose, M.; Lambert, J.D.; Ju, J.; Reuhl, K.R.; Shapses, S.A.; Yang, C.S. The major green tea polyphenol, (-)-epigallocatechin-3 gallate, inhibits obesity, metabolic syndrome, and fatty liver disease in high-fat-fed mice. J. Nutr. 2008, 138, 1677–1683. [Google Scholar] [CrossRef] [PubMed]
- Nagao, T.; Hase, T.; Tokimitsu, I. A green tea extract high in catechins reduces body fat and cardiovascular risks in humans. Obesity 2007, 15, 1473–1483. [Google Scholar] [CrossRef] [PubMed]
- Mehta, V.; Parashar, A.; Sharma, A.; Singh, T.R.; Udayabanu, M. Quercetin ameliorates chronic unpredicted stress-mediated memory dysfunction in male Swiss albino mice by attenuating insulin resistance and elevating hippocampal GLUT4 levels independent of insulin receptor expression. Horm Behav. 2017, 89, 13–22. [Google Scholar] [CrossRef]
- Cialdella-Kam, L.; Ghosh, S.; Meaney, M.P.; Knab, A.M.; Shanely, R.A.; Nieman, D.C. Quercetin and green tea extract supplementation downregulated genes related to tissue inflammatory responses to a 12-week high fat-diet in mice. Nutrients 2017, 9, 773. [Google Scholar] [CrossRef]
- Henagan, T.M.; Lenard, N.R.; Gettys, T.W.; Stewart, L.K. Dietary quercetin supplementation in mice increases skeletal muscle PGC1alpha expression, improves mitochondrial function and attenuates insulin resistance in a time-specific manner. PLoS ONE 2014, 9, e89365. [Google Scholar] [CrossRef]
- Jung, C.H.; Cho, I.; Ahn, J.; Jeon, T.-I.; Ha, T.-Y. Quercetin reduces high-fat diet-induced fat accumulation in the liver by regulating lipid metabolism genes. Phytother. Res. 2013, 27, 139–143. [Google Scholar] [CrossRef]
- Ding, L.; Jin, D.; Chen, X. Luteolin enhances insulin sensitivity via activation of PPARγ transcriptional activity in adipocytes. J. Nutr. Biochem. 2010, 21, 941–947. [Google Scholar] [CrossRef]
- Jaacks, L.M.; Siegel, K.R.; Gujral, U.P.; Narayan, K.M. Type 2 diabetes: A 21st century epidemic. Best Pract. Res. Clin. Endocrinol. Metab. 2016, 30, 331–343. [Google Scholar] [CrossRef]
- Rauter, A.P.; Martins, A.; Borges, C.; Mota-Filipe, H.; Pinto, R.; Sepodes, B.; Justino, J. Antihyperglycaemic and protective effects of flavonoids on streptozotocin-induced diabetic rats. Phytother. Res. 2010, 24, 133–138. [Google Scholar] [CrossRef]
- Zang, M.; Xu, S.; Maitland-Toolan, K.A.; Zuccollo, A.; Hou, X.; Jiang, B.; Wierzbicki, M.; Verbeuren, T.J.; Cohen, R.A. Polyphenols stimulate amp-activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic ldl receptor-deficient mice. Diabetes 2006, 55, 2180–2191. [Google Scholar] [CrossRef]
- Hossain, M.K.; Dayem, A.A.; Han, J.; Yin, Y.; Kim, K.; Saha, S.K.; Yang, G.-M.; Choi, H.Y.; Cho, S.-G. Molecular mechanism of the anti-obesity and anti-diabetic properties of flavonoids. Int. J. Mol. Sci. 2016, 17, 569. [Google Scholar] [CrossRef]
- Panda, S.; Kar, A. Apigenin (4′,5,7-trihydroxyflavone) regulates hyperglycaemia, thyroid dysfunction and lipid peroxidation in alloxan induced diabetic mice. J. Pharm. Pharmacol. 2007, 59, 1543–1548. [Google Scholar] [CrossRef]
- Suh, K.S.; Oh, S.; Woo, J.-T.; Kim, S.-W.; Kim, J.-W.; Kim, Y.S.; Chon, S. Apigenin attenuates 2-deoxy-d-ribose-induced oxidative cell damage in HIT-T15 pancreatic. β-cells. Biol. Pharm. Bull. 2012, 35, 121–126. [Google Scholar] [CrossRef]
- Jung, U.J.; Lee, M.K.; Park, Y.B.; Kang, M.A.; Choi, M.S. Effect of citrus flavonoids on lipid metabolism and glucose-regulating enzyme mRNA levels in type-2 diabetic mice. Int. J. Biochem. Cell Biol. 2006, 38, 1134–1145. [Google Scholar] [CrossRef]
- Pu, P.; Gao, D.-M.; Mohamed, S.; Chen, J.; Zhang, J.; Zhou, X.-Y.; Zhou, N.-J.; Xie, J.; Jiang, H. Naringin ameliorates metabolic syndrome by activating amp-activated protein kinase in mice fed a high-fat diet. Arch. Biochem. Biophys. 2012, 518, 61–70. [Google Scholar] [CrossRef]
- Koch, C.E.; Ganjam, G.K.; Steger, J.; Legler, K.; Stohr, S.; Schumacher, D.; Hoggard, N.; Heldmaier, G.; Tups, A. The dietary flavonoids naringenin and quercetin acutely impair glucose metabolism in rodents possibly via inhibition of hypothalamic insulin signalling. Br. J. Nutr. 2013, 109, 1040–1051. [Google Scholar] [CrossRef]
- Akiyama, S.; Katsumata, S.; Suzuki, K.; Ishimi, Y.; Wu, J.; Uehara, M. Dietary hesperidin exerts hypoglycemic and hypolipidemic effects in streptozotocin-induced marginal type 1 diabetic rats. J. Clin. Biochem. Nutr. 2010, 46, 87–92. [Google Scholar] [CrossRef]
- Jung, U.J.; Lee, M.-K.; Jeong, K.-S.; Choi, M.-S. The hypoglycemic effects of hesperidin and naringin are partly mediated by hepatic glucose-regulating enzymes in C57BL/KsJ-db/db mice. J. Nutr. 2004, 134, 2499–2503. [Google Scholar] [CrossRef]
- Jung, U.J.; Choi, M.-S. Obesity and its metabolic complications: The role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 2014, 15, 6184–6223. [Google Scholar] [CrossRef]
- Agrawal, Y.O.; Sharma, P.K.; Shrivastava, B.; Ojha, S.; Upadhya, H.M.; Arya, D.S.; Goyal, S.N. Hesperidin produces cardioprotective activity via PPAR- pathway in ischemic heart disease model in diabetic rats. PLoS ONE 2014, 9, e111212. [Google Scholar] [CrossRef]
- Eid, H.M.; Martineau, L.C.; Saleem, A.; Muhammad, A.; Vallerand, D.; Benhaddou-Andaloussi, A.; Nistor, L.; Afshar, A.; Arnason, J.T.; Haddad, P.S. Stimulation of AMP-activated protein kinase and enhancement of basal glucose uptake in muscle cells by quercetin and quercetin glycosides, active principles of the antidiabetic medicinal plant vaccinium vitis-idaea. Mol. Nutr. Food Res. 2010, 54, 991–1003. [Google Scholar] [CrossRef]
- Alam, M.M.; Meerza, D.; Naseem, I. Protective effect of quercetin on hyperglycemia, oxidative stress and DNA damage in alloxan induced type 2 diabetic mice. Life Sci. 2014, 109, 8–14. [Google Scholar] [CrossRef]
- Kobori, M.; Masumoto, S.; Akimoto, Y.; Takahashi, Y. Dietary quercetin alleviates diabetic symptoms and reduces streptozotocin-induced disturbance of hepatic gene expression in mice. Mol. Nutr. Food Res. 2009, 53, 859–868. [Google Scholar] [CrossRef]
- Xu, M.; Hu, J.; Zhao, W.; Gao, X.; Jiang, C.; Liu, K.; Liu, B.; Huang, F. Quercetin differently regulates insulin-mediated glucose transporter 4 translocation under basal and inflammatory conditions in adipocytes. Mol. Nutr. Food Res. 2014, 58, 931–941. [Google Scholar] [CrossRef]
- Prince, P.; Kamalakkannan, N. Rutin improves glucose homeostasis in streptozotocin diabetic tissues by altering glycolytic and gluconeogenic enzymes. J. Biochem. Mol. Toxicol. 2006, 20, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.M.; Kang, M.J.; Choi, H.N.; Kim, J.H.; Kim, J.I. Quercetin ameliorates hyperglycemia and dyslipidemia and improves antioxidant status in type 2 diabetic db/db mice. Nutr. Res. Pract. 2012, 6, 201–207. [Google Scholar] [CrossRef]
- Arias, N.; Macarulla, M.T.; Aguirre, L.; Martinez-Castano, M.G.; Portillo, M.P. Quercetin can reduce insulin resistance without decreasing adipose tissue and skeletal muscle fat accumulation. Genes Nutr. 2014, 9, 361. [Google Scholar] [CrossRef]
- Yang, X.; Kong, F. Evaluation of the in vitro α-glucosidase inhibitory activity of green tea polyphenols and different tea types. J. Sci. Food Agric. 2016, 96, 777–782. [Google Scholar] [CrossRef]
- Johnston, K.; Sharp, P.; Clifford, M.; Morgan, L. Dietary polyphenols decrease glucose uptake by human intestinal Caco-2 cells. FEBS Lett. 2005, 579, 1653–1657. [Google Scholar] [CrossRef]
- Mandel, S.; Grunblatt, E.; Riederer, P.; Gerlach, M.; Levites, Y.; Youdim, M.B.H. Neuroprotective strategies in Parkinson’s disease: An update on progress. CNS Drugs 2003, 17, 729–762. [Google Scholar] [CrossRef] [PubMed]
- Berg, D.; Gerlach, M.; Youdim, M.B.H.; Double, K.L.; Zecca, L.; Riederer, P.; Becker, G. Brain iron pathways and their relevance to Parkinson’s disease. J. Neurochem. 2001, 79, 225–236. [Google Scholar] [CrossRef]
- Dauer, W.; Przedborski, S. Parkinson’s disease: Mechanisms and models. Neuron 2003, 39, 889–909. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Moreira, P.I.; Agostinho, P.; Pereira, C.; Oliveira, C.R. Neurodegenerative pathways in Parkinson’s disease: Therapeutic strategies. Curr. Drug Targets CNS Neurol. Disord. 2005, 4, 405–419. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Braak, H.; Ghebremedhin, E.; Rüb, U.; Bratzke, H.; Del Tredici, K. Stages in the development of Parkinson’s disease-related pathology. Cell Tissue Res. 2004, 318, 121–134. [Google Scholar] [CrossRef]
- McGeer, P.L.; Itagaki, S.; Akiyama, H.; McGeer, E.G. Rate of cell death in parkinsonism indicates active neuropathological process. Ann. Neurol. 1988, 24, 574–576. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.L.; James-Kracke, M.; Sun, G.Y.; Sun, A.Y. Oxidative and inflammatory pathways in Parkinson’s disease. Neurochem. Res. 2009, 34, 55–65. [Google Scholar] [CrossRef]
- Hamaguchi, T.; Ono, K.; Murase, A.; Yamada, M. Phenolic compounds prevent Alzheimer’s pathology through different effects on the amyloid-beta aggregation pathway. Am. J. Pathol. 2009, 175, 2557–2565. [Google Scholar] [CrossRef]
- Smith, A.; Giunta, B.; Bickford, P.C.; Fountain, M.; Tan, J.; Shytle, R.D. Nanolipidic particles improve the bioavailability and alpha-secretase inducing ability of epigallocatechin-3-gallate (EGCG) for the treatment of Alzheimer’s disease. Int. J. Pharm. 2010, 389, 207–212. [Google Scholar] [CrossRef]
- Janle, M.E.; Lila, M.A.; Grannan, M.; Wood, L.; Higgins, A.; Yousef, G.G.; Rogers, R.B.; Kim, H.; Jackson, G.S.; Ho, L.; et al. Pharmacokinetics and tissue distribution of 14C-labeled grape polyphenols in the periphery and the central nervous system following oral administration. J. Med. Food. 2010, 13, 926–933. [Google Scholar] [CrossRef]
- Stevenson, D.E.; Hurst, R.D. Polyphenolic phytochemicals—Just antioxidants or much more? Cell Mol. Life Sci. 2007, 64, 2900–2916. [Google Scholar] [CrossRef]
- Fraga, C.G. Plant polyphenols: How to translate their in vitro antioxidant actions to in vivo conditions. Life 2007, 59, 308–315. [Google Scholar] [CrossRef]
- Karpinar, D.P.; Balija, M.B.; Kügler, S.; Opazo, F.; Rezaei-Ghaleh, N.; Wender, N.; Kim, H.Y.; Taschenberger, G.; Falkenburger, B.H.; Heise, H.; et al. Pre-fibrillar alpha-synuclein variants with impaired beta-structure increase neurotoxicity in Parkinson’s disease models. EMBO J. 2009, 28, 3256–3268. [Google Scholar] [CrossRef]
- Marques, O.; Outeiro, T.F. Alpha-synuclein: From secretion to dysfunction and death. Cell Death Dis. 2012, 3, e350. [Google Scholar] [CrossRef]
- Masuda, M.; Suzuki, N.; Taniguchi, S.; Oikawa, T.; Nonaka, T.; Iwatsubo, T.; Hisanaga, S.; Goedert, M.; Hasegawa, M. Small molecule inhibitors of alpha-synuclein filament assembly. Biochemistry 2006, 45, 6085–6094. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Munishkina, L.A.; Fink, A.L.; Uversky, V.N. Effects of various flavonoids on the alpha-synuclein fibrillation process. Parkinsons Dis. 2010, 650794, 1–16. [Google Scholar] [CrossRef]
- Ehrnhoefer, D.E.; Bieschke, J.; Boeddrich, A.; Herbst, M.; Masino, L.; Lurz, R.; Engemann, S.; Pastore, A.; Wanker, E.E. EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nat. Struct. Mol. Biol. 2008, 15, 558–566. [Google Scholar] [CrossRef]
- Bae, S.Y.; Kim, S.; Hwang, H.; Kim, H.K.; Yoon, H.C.; Kim, J.H.; Lee, S.; Kim, T.D. Amyloid formation and disaggregation of alpha-synuclein and its tandem repeat (alpha-TR). Biochem. Biophys. Res. Commun. 2010, 400, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Bieschke, J.; Russ, J.; Friedrich, R.P.; Ehrnhoefer, D.E.; Wobst, H.; Neugebauer, K.; Wanker, E.E. EGCG remodels mature alpha-synuclein and amyloid-beta fibrils and reduces cellular toxicity. Proc. Natl. Acad. Sci. USA 2010, 107, 7710–7715. [Google Scholar] [CrossRef]
- Lu, H.; Meng, X.; Yang, C.S. Enzymology of methylation of tea catechins and inhibition of catechol-O-methyltransferase by (−)-epigallocatechin gallate. Drug Metab. Dispos. 2003, 31, 572–579. [Google Scholar] [CrossRef]
- Levites, Y.; Weinreb, O.; Maor, G.; Youdim, M.B.H.; Mandel, S. Green tea polyphenol (-)-epigallocatechin-3-gallate prevents N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced dopaminergic neurodegeneration. J. Neurochem. 2001, 78, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Mandel, S.; Maor, G.; Youdim, M.B.H. Iron and alpha synuclein in the substantia nigra of MPTP-treated mice: Effect of neuroprotective drugs R-apomorphine and green tea polyphenol epigallocatechin-3-gallate. J. Mol. Neurosci. 2004, 24, 401–416. [Google Scholar] [CrossRef]
- Chaturvedi, R.K.; Shukla, S.; Seth, K.; Chauhan, S.; Sinha, C.; Shukla, Y.; Agrawal, A.K. Neuroprotective and neurorescue effect of black tea extract in 6-hydroxydopamine-lesioned rat model of Parkinson’s disease. Neurobiol. Dis. 2006, 22, 421–434. [Google Scholar] [CrossRef] [PubMed]
- Checkoway, H.; Powers, K.; Smith-Weller, T.; Franklin, G.M.; Longstreth, W.T., Jr.; Swanson, P.D. Parkinson’s disease risks associated with cigarette smoking, alcohol consumption, and caffeine intake. Am. J. Epidemiol. 2002, 155, 732–738. [Google Scholar] [CrossRef]
- Gao, X.; Cassidy, A.; Schwarzschild, M.A.; Rimm, E.B.; Ascherio, A. Habitual intake of dietary flavonoids and risk of Parkinson disease. Neurology 2012, 78, 1138–1145. [Google Scholar] [CrossRef]
- Baptista, F.I.; Henriques, A.G.; Silva, A.M.S.; Wiltfang, J.; da Cruz a Silva, O.A.B. Flavonoids as therapeutic compounds targeting key proteins involved in Alzehimer’s disease. ACS Chem. Neurosci. 2014, 5, 83–92. [Google Scholar] [CrossRef]
- Van Cauwenberghe, C.; Van Broeckhoven, C.; Sleegers, K. The genetic landscape of Alzheimer disease: Clinical implications and perspectives. Genet. Med. 2015, 18, 421–430. [Google Scholar] [CrossRef]
- Mendiola-Precoma, J.; Berumen, L.C.; Padilla, K.; Garcia-Alcocer, G. Therapies for prevention and treatment of Alzheimer’s disease. Biomed. Res. Int. 2016, 2589276. [Google Scholar] [CrossRef]
- Contestabile, A. The history of the cholinergic hypothesis. Behav. Brain Res. 2011, 221, 334–340. [Google Scholar] [CrossRef]
- Cervellati, C.; Wood, P.L.; Romani, A.; Valacchi, G.; Squerzanti, M.; Sanz, J.M.; Ortolani, B.; Zuliani, G. Oxidative challenge in Alzheimer’s disease: State of knowledge and future needs. J. Investig. Med. 2016, 64, 21–32. [Google Scholar] [CrossRef]
- Folch, J.; Patraca, I.; Martínez, N.; Pedrós, I.; Petrov, D.; Ettcheto, M.; Abad, S.; Marin, M.; Beas-Zarate, C.; Camins, A. The role of leptin in the sporadic form of Alzheimer’s disease. Interactions with the adipokines amylin, ghrelin and the pituitary hormone prolactin. Life Sci. 2015, 140, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Talbot, K.; Wang, H.Y.; Kazi, H.; Han, L.Y.; Bakshi, K.P.; Stucky, A.; Fuino, R.L.; Kawaguchi, K.R.; Samoyedny, A.J.; Wilson, R.S.; et al. Demonstrated brain insulin resistance in Alzheimer’s disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. J. Clin. Investig. 2012, 122, 1316–1338. [Google Scholar] [CrossRef]
- Commenges, D.; Scotet, V.; Renaud, S.; Jacqmin-Gadda, H.; Barberger-Gateau, P.; Dartigues, J.F. Intake of flavonoids and risk of dementia. J. Epidemiol. 2000, 16, 357–363. [Google Scholar] [CrossRef]
- Le Bars, P.L.; Kieser, M.; Itil, K.Z. A 26-week analysis of a double-blind, placebo-controlled trial of the ginkgo biloba extract EGb 761 in dementia. Dement. Geriatr. Cognit. Disord. 2000, 11, 230–237. [Google Scholar] [CrossRef]
- Youdim, K.A.; Joseph, J.A. A possible emerging role of phytochemicals in improving age-related neurological dysfunctions: A multiplicity of effects. Free Radic. Biol. Med. 2001, 30, 583–594. [Google Scholar] [CrossRef]
- Joseph, J.A.; Denisova, N.A.; Arendash, G.; Gordon, M.; Diamond, D.; Shukitt-Hale, B.; Morgan, D. Blueberry supplementation enhances signaling and prevents behavioral deficits in an Alzheimer disease model. Nat. Neurosci. 2003, 6, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Levites, Y.; Amit, T.; Mandel, S.; Youdim, M.B.H. Neuroprotection and neurorescue against amyloid beta toxicity and PKC-dependent release of non-amyloidogenic soluble precursor protein by green tea polyphenol (-)-epigallocatechin-3-gallate. FASEB J. 2003, 17, 952–954. [Google Scholar] [CrossRef]
- Ono, K.; Yoshiike, Y.; Takashima, A.; Hasegawa, K.; Naiki, H.; Yamada, M. Potent anti-amyloidogenic and fibril-destabilizing effects of polyphenols in vitro: Implications for the prevention and therapeutics of Alzheimer’s disease. J. Neurochem. 2003, 87, 172–181. [Google Scholar] [CrossRef]
- Morris, M.S. Homocysteine and Alzheimer’s disease. Lancet. Neurol. 2003, 7, 425–428. [Google Scholar] [CrossRef]
- Wojtunik-Kulesza, K.A.; Oniszczuk, A.; Oniszczuk, T.; Waksmundzka-Hajnos, M. The influence of common free radicals and antioxidants on development of Alzheimer’s Disease. Biomed. Pharmacother. 2016, 78, 39–45. [Google Scholar] [CrossRef]
- Dai, Q.; Borenstein, A.R.; Wu, Y.; Jackson, J.C.; Larson, E.B. Fruit and vegetable juices and Alzheimer’s disease: The Kame Project. Am. J. Med. 2006, 119, 751–759. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, Z.; Ho, L.; Seror, I.; Humala, N.; Percival, S. Moderate consumption of Cabernet Sauvignon attenuates β-amyloid neuropathology in a mouse model of Alzheimer’s disease. FASEB J. 2006, 20, 2313–2320. [Google Scholar] [CrossRef] [PubMed]
- Luchsinger, J.A.; Tang, M.-X.; Siddiqui, M.; Shea, S.; Mayeux, R. Alcohol intake and risk of dementia. J. Am. Geriatr. Soc. 2004, 52, 540–546. [Google Scholar] [CrossRef]
| No | Characteristic | References |
|---|---|---|
| 1 | regular sun exposure (except the populations with high intake of vitamin D3 from fish and marine mammals, for which endogenous synthesis was less relevant, e.g., the Inuit) | [11,12,13,14] |
| 2 | sleep patterns according to natural day/night rhythm | [15] |
| 3 | lack of chronic stress/presence of acute stress | [16] |
| 4 | regular high physical activity—required to get food, to protect against predators or enemies, to building shelters, and to make social interactions | [17,18,19] |
| 5 | low environmental exposure to anthropological pollutants (e.g., pesticides, toxic heavy metals, cancerogenic carbohydrates etc.) | [16] |
| 6 | fresh (generally unprocessed) food a. plant leaves, seaweed, sea grasses, and algae b. roots and tubers c. berries and wild fruits d. insects, fish, marine animals, reptiles, and birds e. wild terrestrial mammals and eggs f. nuts and seeds g. honey (occasional intake) | [3,9,10,20,21,22,23] |
| Bioactivity | Experimental Model | Compound | Mechanism of Action/Effect | Ref. | |
|---|---|---|---|---|---|
| In Vitro | In Vivo | ||||
| Antioxidant | DPPH, ABTS, FRAP assays | Quercetin, rutin, EC, | —direct ROS scavenging —trace elements chelation —increasing the endogenic antioxidant enzymes production | [66,88,89,90,91,92,93] | |
| Ten healthy Korean subjects | Green tea polyphenols | —increased total capacity of blood plasma and serum | [90] | ||
| People subjected to anaerobic trainning | Theaflavins | —decreasing the oxidative stress in humans subjected to acute anaerobic interval training | [95] | ||
| Rats | Theaflavins and thearubigins | —increasing the antioxidant parameters of blood —significant reduction in lipid profile, glucose content and renal function —increasing the insulin level —the best results after administration of both theaflavins and thearubigins simultaneously | [96] | ||
| Athletes | Theasinensins from oolong tea | —strong reduction of malondialdehyde at rest and after exercise —decreased levels of SOD —reduced lipid peroxidation and cholesterol profile | [97] | ||
| Anti-inflammatory | Different immune cells | EGCG | —decreasing the production of proinflammatory chemokines —inhibition of the proliferation of autoreactive T cells —reducing the production of autoimmune agents | [99,100] | |
| Mice with collagen-induced arthritis | EGCG | —significant reduction in cytokines, phosphorylated-signal transducer and activator of transcription-3, 705 and 727 and oxidative stress proteins production —inhibition of osteoclast and Th 17 production —increased regulatory T cells production | [101] | ||
| Human peripheral blood neutrophils | Rutin, quercetin and vitexin | —significant reduction of NO, MPO and TNF-α (inflammatory mediators) production | [102,103] | ||
| Mice with an experimental autoimmune myocardiatitis | Apigenin | —inhibition of the proinflammatory cytokines production - Th1 cytokines, TNF-α, interferon-gamma (IFN-γ), and interleukin-2 (IL-2) | [104] | ||
| Anticancer | hepatocellular carcinoma LM6 cells (HCCLM6) | EGCG | —induction of apoptosis —reduction of mitochondrial membrane potential and promotion of G0/G1 phase cell cycle arrest —significant decrease in Bcl-2 and NF-κB expression. | [105] | |
| Mice with tongue and liver cancer induced by N-nitrosodiethylamine | Theaflavins and EGCG | —modulation of cellular proliferation/apoptosis and prevalence of CD 44 positive regulation | [106] | ||
| human prostate carcinoma (LNaCP and PC-3 cells) | Theaflavins | —tumor cell death by induction of p53 suppressor expression and inhibition of nuclear transcription factor NF-κβ and mitogen-activated MAPK-kinase, —the cell cycle blocking in the phase G2/M | [107,108] | ||
| human retinoblastoma (RB Y79 cell line) | Quercetin | —inhibition of tumor progression and invasion —angiogenesis inhibition | [110] | ||
| 9 different cancer cell lines (i.a. colon, prostate, breast and ovarian carcinomas). These results were confirmed in animal model, as | Mice with induced colon and breast cancers | Quercetin | —cell growth inhibition of all tested cell lines due to the induction of apoptosis —reduction of the tumor volume in a mouse model and increased animal survival | [111] | |
| Animal models with induced colorectal, breast, prostate, melanoma, renal cell and ovarian cancers | Apigenin | —reduction of proliferation, invasion and migration —increased apoptosis | [112,113,114,115,116,117,118] | ||
| human hepatic cancer HepG2 cells | Hesperidin | —activation of caspase-9, -8, and -3 —downregulation of Bcl-xL protein —upregulation of Bax, Bak, and tBid protein levels | [119] | ||
| MCF-7 human breast, androgen-independent PC-3 and DU-145 prostate cancer cells, and androgen-dependent LNCa cancer cells | Hesperidin | —inhibition of the proliferation of MCF-7-GFP-Tubulin cells and both basal and testosterone-induced proliferation of LNCaP cells | [120] | ||
| A2780 ovarian cancer cell line | Hesperidin | —induction of apoptosis —increasing cleaved caspase-3 protein expression levels —elevating the expression of proteins of antigrowth arrest- and DNA damage-inducible gene 153 —increasing anti-CCAAT’ enhancer-binding protein homologous protein expression —increasing the expression of glucose-regulated protein 78 and cytochrome c | [121] | ||
| Prevention of cardiovascular diseases | population-based cohort study on 1658 individuals | different subclasses of dietary flavonoids | —significant reduction of a risk of nonfatal CV events - the reduction of all-cause mortality parameters was the most significantly correlated with the intake of flavan-3-ols, anthocyanidins and flavanones | [84] | |
| a double-blind, placebo-controlled trial, on 56 obese, hypertensive subjects | Green tea polyphenols | —reduction of both systolic and diastolic blood pressures —significant reduction of TNF-α, C-reactive protein levels, fasting serum glucose, insulin levels and insulin resistance | [123] | ||
| Rats with induced hypertension caused by deoxycorticosterone acetate (DOCA) | EC | —inhibition of blood pressure increase —improving the impaired endothelium-dependent relaxation response to acetylcholine —reduction of plasma endothelin-1 and malondialdehyde levels | [126] | ||
| FRT cells transfected with human TMEM16A | Green tea polyphenols | —strong inhibition of calcium-activated Cl− channels —ECG and EGCG more strongly inhibited Cl− currents than the other polyphenols | [129] | ||
| Rats with induced hypertension | EGCG | —significant decrease of blood pressure —stimulated nitric oxide production from endothelium through a PI-3-kinase pathway | [131] | ||
| human recombinant renin inhibitor screening assay kit | EGCG | —inhibition of renin with IC50 value of 44.53 µg | [132] | ||
| Anti-obesity effect/weight reducing properties | 24 clinical, placebo-controlled trials, performed on obese patients | Green tea polyphenols (mostly 600–900 mg/day, which is equal to 3–4 cups of green tea) | —significant decrease in total abdominal fat are —inhibition of lipid synthesis —reduced serum triglycerides —reduction of food intake —deterioration of lipid emulsification and absorption, —induction of adipogenesis —increase of energy expenditure —Acceleration of fat oxidation —improvement of fecal lipid excretion | [133,134,135,136] | |
| Mice fed a high fat diet for 12 weeks | Mixture of quercetin and green tea extract administrated for four weeks | —downregulation of over 100 genes in adipose tissue, including those involved in leukocyte extravasation and phagocytosis —downregulation of cholesterol metabolism —reduction of cholesterol congeries in adipose and muscle tissue —mild improvement of glucose tolerance | [138] | ||
| Mice fed a high fat diet for 9 weeks | Diet enriched with quercetin in the concentration of 0.025% (w/w) | —regulation of hepatic gene expression related to lipid —significant reduction of liver and white adipose tissue weight —decrease in hepatic lipid accumulation —reduction of the size of lipid droplets in the animal epididymal fat pads | [140] | ||
| 3T3-L1 cells/isolated mouse epididymal adipose cells from C57BL/6J mice | Luteolin (20 μmol/L) | —insulin sensitivity increase —decrease of low-grade chronic inflammation —improved uptake of glucose by adipocytes —reduction of proinflammatory chemokines —reduction of mRNA levels of tumor necrosis factor α, IL-6 and MCP-1 —increasing the gene expression of adiponectin and leptin —activation of PPARγ target genes in animal adipose cells | [141] | ||
| Antidiabetic activity | streptozotocin-induced diabetic Wistar rats | Apigenin/rutin/geinistein/luteolin administrated intraperitoneal-ly for seven days (4 mg/kg body weight/day) | —strong hypoglycemic effect, especially observed in the oral glucose tolerance test —protective effect towards liver and kidneys | [143] | |
| HepG2 hepatocytes | type 1 diabetic LDL receptor-deficient mice (DMLDLR(-/-) | Apigenin <0.1% in DMSO (in vitro) 130 mg/kg/day (in vivo) for six weeks | —regulation of lipid and glucose metabolism through AMP-dependent kinase —prevention of lipid accumulation in the liver —inhibition of hyperlipidemia —slowing the aortic lesion development | [144] | |
| HIT-T15 pancreatic β-cells | Apigenin (20 mM in DMSO) | —inhibition of pancreatic β-cells damage through different oxidative stress related signaling pathways (decreasing ROS production, apoptosis and increasing the mitochondrial membrane potential) | [147] | ||
| Alloxan-treated diabetic mice | Apigenin (0.78 mg/kg/day in DMSO), s.c. for ten consecutive days | —increased serum insulin and thyroid hormone levels —reduction of blood glucose —reduction of hepaticG-6-Pase activity —reduction of serum cholesterol level —hepatoprotective activity (observed as decreased levels of lipid peroxidation and increased levels of catalase, SOD or glutathione) | [146] | ||
| Mice fed with high-fat diet | Naringin (0.2 g/kg diet, administrated orally) | —inhibition of inflammation and insulin resistance by blocking activation of the MAPKs pathways and by activating IRS-1 —lipid reduction by synthesis inhibition and increased fatty acid oxidation —hypoglycemic effect through the regulation of PEPCK and G6pase activity in the liver | [149] | ||
| Male diabetic C57BL/KsJ-db/db mice | Naringin and hesperidin (0.2 g/kg diet, administrated orally, separately) | —increasing the glucokinase mRNA level (both flavonoids) —reducing the mRNA expression of PEPCK and G6pase in the liver (naringin) —reducing the expression of hepatic glucose transporter 2 protein in the liver —upregulation of the expression of adipocyte GLUT-4 and hepatic and adipocyte PPARγ (both flavonoids) —reducing free fatty acid and TAGs plasma levels (both flavonoids) —inhibition of hepatic (HMG-CoA) reductase activity —increasing fecal cholesterol | [148] | ||
| Adult Djungarian hamsters/ Female C57BL/6JRj-mic | Naringenin and quercetin (10 mg/kg administered orally) | —impaired glucose tolerane —no influence on circulating levels of insulin and insulin-like growth factor-binding proteins —reduced respiratory quotient (impaired glucose utilization) (oral quercetin) | [150] | ||
| Rats with streptozotocin (STZ)-induced marginal type 1 diabetes | Hesperidin (10 g/kg diet) | —reduced blood glucose level by decreased activity of G6Pase and increased expression of GK —decreased glucose export through glucose transporter membrane proteins | [151] | ||
| Male C57BL/KsJ-db/db mice | Diet supplemented with hesperidin (0.2 g/kg diet) or naringin (0.2 g/kg diet) | —significant reduction of blood glucose (both flavonoids) —significant increase of GK activity and glycogen concentration (both flavonoids) —reduced activity of hepatic G6pase and PEPCK (naringin) —increased levels of Plasma insulin, C-peptide, and leptin (both flavonoids) | [152] | ||
| Male Wistar diabetic rats | Hesperidin (100 mg/kg/day) administrated orally | —significant mean arterial pressure improvement —reduced left ventricular end-diastolic pressure —improved both inotropic and lusitropic function of the heart —decreased level of TBARs —anti-apoptotic and protective effect | [154] | ||
| C2C12 murine skeletal myoblasts and H4IIE murine hepatocytes | Quercetin and its glycosylated forms isolated from Vaccinium vitis-idaea extract | —significant glucose uptake through the insulin-independent AMPK pathway —mild inhibition of mitochondrial ADP-stimulated oxygen consumption —inhibition of ATP synthase in mitochondria (only the aglycone) | [155] | ||
| Alloxan induced Swiss albino mice | Oral supplementation with quercetin (20 mg/kg body weight) for three weeks | —significant decrease of fasting blood glucose level —reduced markers of liver and kidneys injury —increased levels of antioxidant enzymes (SOD, glutathione, catalase and glutathione-S-transferase) —decreased concentration of TBARs —increased level of GLUT4 expression | [156] | ||
| STZ-induced diabetic mice | Quercetin administrated orally (0.1 and 0.5% of the diet) | —decreased blood glucose —increased insulin plasma level —suppression of the STZ-induced alteration of gene expression (0.5% of quercetin) —the strongest inhibitory effect towards elevated expression of cyclin-dependent kinase inhibitor p21(WAF1/Cip1) (Cdkn1a) | [157] | ||
| 3T3-L1 adipocyte cells | Quercetin | —inhibition of insulin-mediated GLUT4 translocation —improved glucose metabolism through the regulation AMPK activity —improvement of insulin cell sensitivity by inhibition of IκB kinase β (IKKβ) phosphorylation and insulin signaling facilitation | [158] | ||
| Diabetic mice | Quercetin administrated orally (0.04 and 0.08% of the diet) for six weeks | —dose dependent decrease of blood level glucose —decreased values of the HOMA-IR parameter —increased plasma adiponectin level (0.08% of quercetin) —reduced levels of LDL, total cholesterol, TAGs and increased HDL —decreased TBARs —increased concentration of the liver SOD, catalase and glutathione peroxidase | [160] | ||
| Rats fed with high sucrose diet | Oral supplementation with quercetin (30 mg/kg body weight per day) for six weeks | —no reduction on body weight or adipose tissue size was observed —reduced levels of fructosamine, basal glucose, insulin, and HOMA-IR —no influence on lipogenic enzymes and lipoprotein lipase activity was noticed —no change in muscle TAGs content was observed | [161] | ||
| STZ-induced diabetic rats | Rutin administrated orally (100 mg/kg body weight) for 45 days | —decrease of plasma glucose concentration —increased blood insulin level —restoration of glycogen content —protective effect towards pancreatic cells (reduced fatty infiltrate of the Langerhans islets) | [159] | ||
| α-glucosidase inhibition assay | Green, black and oolong tea extracts | —dose dependent inhibitory potential towards α-glucosidase —the strongest activity was revealed for oolong tea extract, followed by black and green tea —the activity of particular extract was attributed to the content of polyphenols | [162] | ||
| polarized Caco-2 intestinal cells | Green tea catechins | —inhibition of intestinal glucose uptake —nonglycosylated dietary polyphenols were the most effective in reduction of glucose uptake under both sodium-dependent and sodium-free conditions | [163] | ||
| Protective activity towards Parkinson’s Disease | Human α-synuclein assembly inhibitory test | Several dietary polyphenols (e.g., Tea catechins, apigenin, quercetin, rutin) | —dietary polyphenols are a major class of compounds active towards the inhibition of α-synuclein —tea catechins were characterized by the highest inhibitory activity —common dietary polyphenols (rutin, quercetin or naringenin) were less active | [178] | |
| α-synuclein fibrillation inhibitory assay | 48 flavonoids dissolved in DMSO at concentration of 5, 10, and 20 mM | —majority of flavonoids inhibited the fibrillation of α-synuclein (EGCG, rutin, luteolin) —the inhibition of α-synuclein fibrillogenesis was due to the inhibition of nucleus formation and the inhibition of fibril elongation —structure-activity dependency was noticed (a vicinal dihydroxyphenyl moiety was crucial for the inhibitory activity) —EGCG, myricetin, baicalein, eriodictyol and quercetin were the strongest inhibitors —diosmetin, hesperetin or hesperidin possessed no inhibitory activity —diosmetin, hesperetin or hesperidin possessed no inhibitory activity | [179] | ||
| α-synuclein fibrillation inhibitory assay | EGCG (50 µM) | —EGCG directly converted fibrillar species into benign protein aggregates —inhibition of α-synuclein and amyloid-β fibrillogenesis by EGCG through a direct binding to β-sheet-rich aggregates was proofed —EGCG did not reverse the process of fibril formation —no increase in toxic intermediates formation was noticed | [182] | ||
| Rat liver cytosol | Green tea catechins (EGCG and EGC) | —significant inhibition of catechol-O-methyltransferase enzyme (COMT) —dose-dependent methylation of EGCG by COMT was proofed —EGCG inhibited methylation of L-DOPA, a common drug used in the therapy of PD, and improved its bioavailability | [183] | ||
| Mice with MPTP induced Parkinsonism | Green tea extract and EGCG (0.5 and 1 mg/kg i.p.) | —increased activity of striatal antioxidant enzymes (SOD and catalase) —significant decrease loss of neurons and increase production of dopamine in substantia nigra —protective mechanism was proofed to be other than inhibition of MPTP transformation to its active metabolite —the protective effect of green tea polyphenols was identified with brain penetrating activity of polyphenols, its antioxidant activity and iron chelating properties | [184] | ||
| Male C57-BL Mice with MPTP induced Parkinsonism | EGCG (2mg/kg/day, per os, for 10 days) | —decreased levels of striatal tyrosine hydroxylase protein and increased activity of this enzyme —increased in PKCα protein expression inside the striatal dopaminergic fibers —neuroprotective activity through iron-chelating and antioxidant action | [185] | ||
| Rats with 6-OHDA-induced PD | Black tea extract 1.5% (administrated orally before and after 6-OHDA injection) | —a significant neuroprotective and neurorescue effects were observed (increased nigral gluthathione and striatal SOD catalase activity and decreased lipid peroxidation level) —improved spontaneous locomotor activity and dopamine (DA)-D2 receptor binding, —increased striatal DA and 3-4 dihydroxy phenyl acetic acid (DOPAC) level —significant increase of antiapoptotic and proapoptotic protein level —higher improvement in motor and neurochemical deficiency was more visible in rats receiving polyphenols before 6-OHDA administration | [186] | ||
| A case-control study conducted in western Washington State in 1992–2000 on 557 individuals | Tea (black or green) | —significant reduced risk of PD development in population drinking 2 cups/day or more of tea | [187] | ||
| Health Professional Follow-up Study performed on 49,281 men and the Nurses’ Health Study performed on 80,336 women | 5 classes of flavonoids (intake based on calculations using database and food frequency questionnaire) | —significant negative correlation between flavonoid intake and PD incidence —the strongest correlation between flavonoids intake and PD risk was found for anthocyanins —the significance of the obtained results was more visible in men, compared to women | [188] | ||
| Protective activity towards Alzheimer’s Disease | A cohort study on 1367 subjects above 65 years of age | Dietary flavonoid intake based on calculations | —significant inverse correlation between flavonoid intake and risk of dementia | [196] | |
| Transgenic mice with genetically induced AD | Diet supplemented with 2% of water blue berry extract | —significant improvement in memory and cognitive functions —improved neuronal signaling through enhanced extracellular signal-regulated kinase and decreased activity of N-Sase in the hippocampus and striatum —diet rich in antioxidant from blue berry extract may overpass genetic predispositions to AD —no influence of particular extract constituents were investigated | [199] | ||
| Human SH-SY5Y neuroblastoma cells/Rat pheochromocytoma PC12 cells | Male C57/BL mice | EGCG (2 mg/kg, per os, for 14 days) (in vivo) | —activation of activates PKCα and PKCε in the membrane and cytosolic fractions of mice hippocampus, which lead to increased production of neuroprotective, nonamyloidogenic sAPPα —reduction of cellular APP holoprotein production, which further reduces the progress of amyloidogenic failure —protective effect against β-amyloid-induced neurotoxicity | [200] | |
| Fresh, nonaggregated β-amyloid fractions | Wine derived polyphenols (myricetin, morin, quercetin, kaempferol, catechin, epicatechin) | —dose-dependent inhibition of β-amyloid formation —dose-dependent destabilization of preformed β-amyloid aggregation —the activity of particular polyphenols was varied in order: myricetin = morin = quercetin > kaempferol > catechin = epicatechin —the effective concentration was within the range of 0.1–1 µM | [201] | ||
| A cohort, population-based prospective study of 1836 Japanese Americans in King County, Washington | Fruit and vegetable juices, containing a high concentration of polyphenols (>3 times per week) | —significant reduction of the hazard ratio for probable Alzheimer’s disease, especially for population drinking more than 3 times per week —the strongest inverse association was revealed for people with an apolipoprotein Eε-4 allele and those who were not physically active | [204] | ||
| Tg2576 mice (model AD-type amyloid beta-protein neuropathology) | Red wine (Cabernet Sauvignon) diluted in drinking water | —improved cognitive functions —attenuation of β-amyloid deposition in neocortex and hippocampus —increased α-secretase activity —significant activity of red wine towards the inhibition of AD development was associated with its polyphenolic composition | [205] | ||
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koch, W. Dietary Polyphenols—Important Non-Nutrients in the Prevention of Chronic Noncommunicable Diseases. A Systematic Review. Nutrients 2019, 11, 1039. https://doi.org/10.3390/nu11051039
Koch W. Dietary Polyphenols—Important Non-Nutrients in the Prevention of Chronic Noncommunicable Diseases. A Systematic Review. Nutrients. 2019; 11(5):1039. https://doi.org/10.3390/nu11051039
Chicago/Turabian StyleKoch, Wojciech. 2019. "Dietary Polyphenols—Important Non-Nutrients in the Prevention of Chronic Noncommunicable Diseases. A Systematic Review" Nutrients 11, no. 5: 1039. https://doi.org/10.3390/nu11051039
APA StyleKoch, W. (2019). Dietary Polyphenols—Important Non-Nutrients in the Prevention of Chronic Noncommunicable Diseases. A Systematic Review. Nutrients, 11(5), 1039. https://doi.org/10.3390/nu11051039





