Aspects of Dietary Diversity Differ in Their Association with Atherosclerotic Cardiovascular Risk in a Racially Diverse US Adult Population
Abstract
1. Introduction
2. Methods
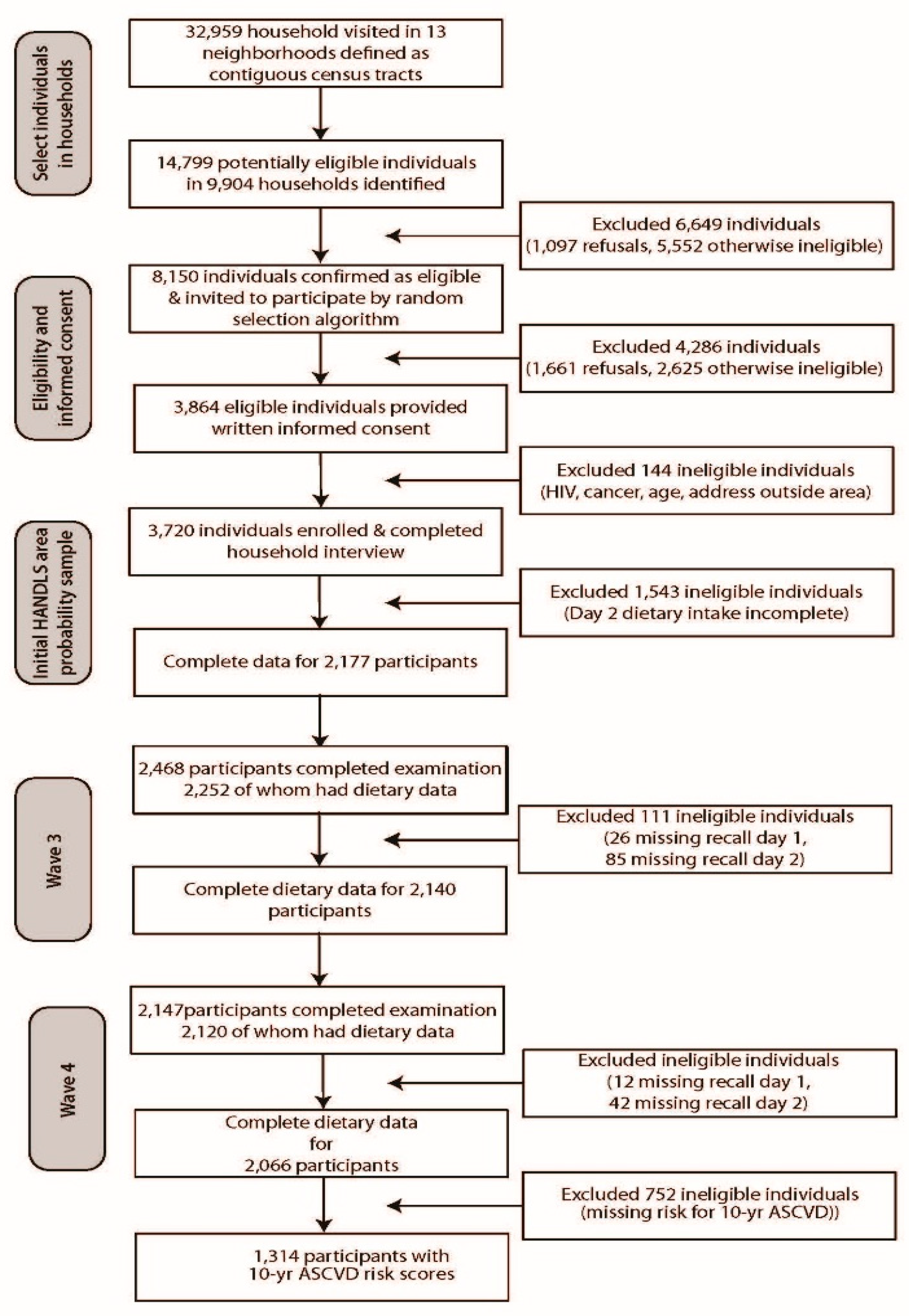
2.1. Healthy Aging in Neighborhoods of Diversity across the Life Span (HANDLS) Study Population
2.2. Dietary Method
2.3. Diversity Measurements
2.4. Diet Quality Measures
2.5. Demographic and Health-Related Measures
2.6. Statistical Analyses
3. Results
3.1. Population Characteristics
3.2. Dietary Characteristics
4. Discussions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the western diet: health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef]
- Lachat, C.; Raneri, J.E.; Smith, K.W.; Kolsteren, P.; Van Damme, P.; Verzelen, K.; Penafiel, D.; Vanhove, W.; Kennedy, G.; Hunter, D.; et al. Dietary species richness as a measure of food biodiversity and nutritional quality of diets. Proc. Natl. Acad. Sci. USA 2018, 115, 127–132. [Google Scholar] [CrossRef]
- Drewnowski, A.; Popkin, B.M. The nutrition transition: new trends in the global diet. Nutr. Rev 1997, 55, 31–43. [Google Scholar] [CrossRef]
- Maxwell, S.L.; Fuller, R.A.; Brooks, T.M.; Watson, J.E.M. Biodiversity: The ravages of guns, nets and bulldozers. Nat. Cell Boil. 2016, 536, 143–145. [Google Scholar] [CrossRef] [PubMed]
- Biodiversity. Available online: http://www.fao.org/biodiversity/overview/en/ (accessed on 27 April 2019).
- Popkin, B.M.; Adair, L.S.; Ng, S.W. Global nutrition transition and the pandemic of obesity in developing countries. Nutr. Rev. 2012, 70, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Romagnolo, D.F.; Selmin, O.I. Mediterranean diet and prevention of chronic diseases. Nutr. Today 2017, 52, 208–222. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Marventano, S.; Yang, J.; Micek, A.; Pajak, A.; Scalfi, L.; Galvano, F.; Kales, S.N. A comprehensive meta-analysis on evidence of Mediterranean diet and cardiovascular disease: are individual components equal? Crit. Rev. Food Sci. Nutr. 2017, 57, 3218–3232. [Google Scholar] [CrossRef]
- Yang, J.; Farioli, A.; Korre, M.; Kales, S.N. Modified Mediterranean diet score and cardiovascular risk in a north American working population. PLoS ONE 2014, 9, e87539. [Google Scholar] [CrossRef]
- Steffen, L.M.; Van Horn, L.; Daviglus, M.L.; Zhou, X.; Reis, J.P.; Loria, C.M.; Jacobs, D.R.; Duffey, K.J. A modified Mediterranean diet score is associated with a lower risk of incident metabolic syndrome over 25 years among young adults: The CARDIA (Coronary Artery Risk Development in Young Adults) study. Br. J. Nutr. 2014, 112, 1654–1661. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Gea, A.; Ruiz-Canela, M. The Mediterranean diet and cardiovascular health. Circ. Res. 2019, 124, 779–798. [Google Scholar] [CrossRef]
- Casas, R.; Castro-Barquero, S.; Estruch, R.; Sacanella, E. Nutrition and cardiovascular health. Int. J. Mol. Sci. 2018, 19, 3988. [Google Scholar] [CrossRef] [PubMed]
- Becerra-Tomás, N.; Blanco Mejía, S.; Viguiliouk, E.; Khan, T.; Kendall, C.W.C.; Kahleova, H.; Rahelić, D.; Sievenpiper, J.L.; Salas-Salvadó, J. Mediterranean diet, cardiovascular disease and mortality in diabetes: A systematic review and meta-analysis of prospective cohort studies and randomized clinical trials. Crit. Rev. Food Sci. Nutr. 2019, 1–21. [Google Scholar] [CrossRef]
- Chiavaroli, L.; Viguiliouk, E.; Nishi, S.K.; Blanco Mejia, S.; Rahelić, D.; Kahleová, H.; Salas-Salvadó, J.; Kendall, C.W.; Sievenpiper, J.L. DASH dietary pattern and cardiometabolic outcomes: An umbrella review of systematic reviews and meta-analyses. Nutrients 2019, 11, 338. [Google Scholar] [CrossRef]
- Rai, S.K.; Fung, T.T.; Lu, N.; Keller, S.F.; Curhan, G.C.; Choi, H.K. The Dietary Approaches to Stop Hypertension (DASH) diet, Western diet, and risk of gout in men: prospective cohort study. BMJ 2017, 357, j1794. [Google Scholar] [CrossRef]
- Rebholz, C.M.; Crews, D.C.; Grams, M.E.; Steffen, L.M.; Levey, A.S.; Miller, E.R.; Appel, L.J.; Coresh, J. DASH (dietary approaches to stop hypertension) diet and risk of subsequent kidney disease. Am. J. Kidney Dis. 2016, 68, 853–861. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Notara, V.; Kouvari, M.; Pitsavos, C. The Mediterranean and other dietary patterns in secondary cardiovascular disease prevention: A review. Curr. Vasc. Pharmacol. 2016, 14, 442–451. [Google Scholar] [CrossRef]
- Myles, I.A. Fast food fever: Reviewing the impacts of the Western diet on immunity. Nutr. J. 2014, 13, 61. [Google Scholar] [CrossRef]
- Larsson, S.C.; Akesson, A.; Wolk, A. Overall diet quality and risk of stroke: A prospective cohort study in women. Atherosclerosis 2014, 233, 27–29. [Google Scholar] [CrossRef]
- Foote, J.A.; Murphy, S.P.; Wilkens, L.R.; Basiotis, P.P.; Foote, J.A.; Carlson, A. Dietary variety increases the probability of nutrient adequacy among adults. J. Nutr. 2004, 134, 1779–1785. [Google Scholar] [CrossRef] [PubMed]
- Hatløy, A.; Torheim, L.E.; Oshaug, A. Food variety—a good indicator of nutritional adequacy of the diet? A case study from an urban area in Mali, West Africa. Eur. J. Clin. Nutr. 1998, 52, 891–898. [Google Scholar] [CrossRef]
- Mirmiran, P.; Azadbakht, L.; Azizi, F. Dietary diversity within Food Groups: An indicator of specific nutrient adequacy in Tehranian women. J. Am. Coll. Nutr. 2006, 25, 354–361. [Google Scholar] [CrossRef]
- Kant, A.K.; Schatzkin, A.; Harris, T.B.; Ziegler, R.G.; Block, G. Dietary diversity and subsequent mortality in the first national health and nutrition examination survey epidemiologic follow-up study. Am. J. Clin. Nutr. 1993, 57, 434–440. [Google Scholar] [CrossRef]
- Habte, T.Y.; Krawinkel, K. Dietary diversity score: A measure of nutritional adequacy or an indicator of healthy diet? J. Nutr. Health Sci. 2016, 3, 303. [Google Scholar] [CrossRef]
- Tavakoli, S.; Dorosty-Motlagh, A.R.; Hoshiar-Rad, A.; Eshraghian, M.R.; Sotoudeh, G.; Azadbakht, L.; Karimi, M.; Jalali-Farahani, S. Is dietary diversity a proxy measurement of nutrient adequacy in Iranian elderly women? Appetite 2016, 105, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Torheim, L.E.; Barikmo, I.; Parr, C.L.; Hatløy, A.; Ouattara, F.; Oshaug, A. Validation of food variety as an indicator of diet quality assessed with a food frequency questionnaire for Western Mali. Eur. J. Clin. Nutr. 2003, 57, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Savy, M.; Martin-Prével, Y.; Sawadogo, P.; Kameli, Y.; Delpeuch, F. Use of variety/diversity scores for diet quality measurement: relation with nutritional status of women in a rural area in Burkina Faso. Eur. J. Clin. Nutr. 2005, 59, 703–716. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.P.; Foote, J.A.; Wilkens, L.R.; Basiotis, P.P.; Carlson, A.; White, K.K.L.; Yonemori, K.M. Simple measures of dietary variety are associated with improved dietary quality. J. Acad. Nutri. Diet. 2006, 106, 425–429. [Google Scholar] [CrossRef]
- Ruel, M.T. Operationalizing dietary diversity: A review of measurement issues and research priorities. J. Nutr. 2003, 133, 3911S–3926S. [Google Scholar] [CrossRef] [PubMed]
- Otto, M.C.; Anderson, C.A.M.; Dearborn, J.L.; Ferranti, E.P.; Mozaffarian, D.; Rao, G.; Wylie-Rosett, J.; Lichtenstein, A.H.; American Heart Association Behavioral Change for Improving Health Factors Committee of the Council on Lifestyle and Cardiometabolic Health and Council on Epidemiology and Prevention; Council on Cardiovascular and Stroke Nursing; et al. Dietary diversity: Implications for obesity prevention in adult populations: A science advisory from the American heart association. Circulation 2018, 138, e160–e168. [Google Scholar]
- Vadiveloo, M.; Dixon, L.B.; Parekh, N. Associations between dietary variety and measures of body adiposity: A systematic review of epidemiological studies. Br. J. Nutr. 2013, 109, 1557–1572. [Google Scholar] [CrossRef]
- Azadbakht, L.; Mirmiran, P.; Azizi, F. Dietary diversity score is favorably associated with the metabolic syndrome in Tehranian adults. Int. J. Obes. 2005, 29, 1361–1367. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Esmaillzadeh, A. dietary diversity score is related to obesity and abdominal adiposity among Iranian female youth. Public Health Nutr. 2011, 14, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Farhangi, M.A.; Jahangiry, L. Dietary diversity score is associated with cardiovascular risk factors and serum adiponectin concentrations in patients with metabolic syndrome. BMC Cardiovasc. Disord. 2018, 18, 68. [Google Scholar] [CrossRef]
- Azadbakht, L.; Mirmiran, P.; Esmaillzadeh, A.; Azizi, F. Dietary diversity score and cardiovascular risk factors in Tehranian adults. Public Health. Nutr. 2006, 9, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Salehi-Abargouei, A.; Akbari, F.; Bellissimo, N.; Azadbakht, L. Dietary diversity score and obesity: A systematic review and meta-analysis of observational studies. Eur. J. Clin. Nutr. 2016, 70, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Otto, M.C.; Padhye, N.S.; Bertoni, A.G.; Jacobs, D.R., Jr.; Mozaffarian, D. Everything in moderation—Dietary diversity and quality, central obesity and risk of diabetes. PLoS ONE 2015, 10, e0141341. [Google Scholar] [CrossRef]
- Jayawardena, R.; Byrne, N.M.; Soares, M.J.; Katulanda, P.; Yadav, B.; Hills, A.P. High dietary diversity is associated with obesity in Sri Lankan adults: An evaluation of three dietary scores. BMC Public Health 2013, 13, 314. [Google Scholar] [CrossRef]
- Drescher, L.S.; Thiele, S.; Mensink, G.B. A new index to measure healthy food diversity better reflects a healthy diet than traditional measures. J. Nutr. 2007, 137, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Katanoda, K.; Kim, H.-S.; Matsumura, Y. New quantitative index for dietary diversity (QUANTIDD) and its annual changes in the Japanese. Nutrition 2006, 22, 283–287. [Google Scholar] [CrossRef]
- Evans, M.K.; Lepkowski, J.M.; Powe, N.R.; LaVeist, T.; Kuczmarski, M.F.; Zonderman, A.B. Healthy aging in neighborhoods of diversity across the life span (HANDLS): Overcoming Barriers to implementing a longitudinal, epidemiologic, urban study of health, race, and socioeconomic status. Ethn. Dis. 2010, 20, 267–275. [Google Scholar] [PubMed]
- Fanelli Kuczmarski, M.; Mason, M.A.; Beydoun, M.A.; Allegro, D.; Zonderman, A.B.; Evans, M.K. Dietary patterns and sarcopenia in an urban African American and White population in the United States. J. Nutr. Gerontol. Geriatr. 2013, 32, 291–316. [Google Scholar] [CrossRef] [PubMed]
- The 2004 HHS Poverty Guidelines. Available online: https://aspe.hhs.gov/2004-hhs-poverty-guidelines (accessed on 29 April 2019).
- Healthy Aging in Neighborhoods of Diversity across the Life Span. Available online: https://handls.nih.gov/06Coll-dataDoc.htm (accessed on 29 April 2019).
- Moshfegh, A.J.; Rhodes, D.G.; Baer, D.J.; Murayi, T.; Clemens, J.C.; Rumpler, W.V.; Paul, D.R.; Sebastian, R.S.; Kuczynski, K.J.; Ingwersen, L.A.; et al. The US Department of Agriculture Automated Multiple-Pass Method reduces bias in the collection of energy intakes. Am. J. Clin. Nutr. 2008, 88, 324–332. [Google Scholar] [CrossRef]
- Raper, N.; Perloff, B.; Ingwersen, L.; Steinfeldt, L.; Anand, J. An overview of USDA’s dietary intake data system. Subtrop. Plant Sci. 2004, 17, 545. [Google Scholar] [CrossRef]
- Food Surveys Research Group: Beltsville, MD. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/ (accessed on 27 April 2019).
- Food Patterns Equivalents Database 2013–14: Methodology and User Guide. Available online: https://www.ars.usda.gov/ARSUserFiles/80400530/pdf/fped/FPED_1314.pdf (accessed on 29 April 2019).
- Berry, C.H. Corporate growth and diversification. J. Law Econ. 1971, 14, 371–383. [Google Scholar] [CrossRef]
- Dietary Guidelines for Americans 2015–2020 8th Edition. Available online: https://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 5 March 2019).
- Zhu, R.; Zhang, Y.; Liu, B.; Liu, L. Information Computing and Applications. In Communications in Computer and Information Science; Zhu, R., Zhang, Y., Liu, B., Liu, C., Eds.; Springer: Berlin, Germany, 2010. [Google Scholar]
- Schweizer, K. A threshold-free approach to the study of the structure of binary data. Int. J. Stat. Probab. 2013, 2, 67–76. [Google Scholar] [CrossRef]
- Nutrient Recommendations: Dietary Reference Intakes (DRI). Available online: https://ods.od.nih.gov/Health_Information/Dietary_Reference_Intakes.aspx (accessed on 5 March 2019).
- Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Mellen, P.B.; Gao, S.K.; Vitolins, M.Z.; Goff, D.C., Jr. Deteriorating dietary habits among adults with hypertension: DASH dietary accordance, NHANES 1988–1994 and 1999–2004. Arch. Intern. Med. 2008, 168, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Goff, D.C., Jr.; Lloyd-Jones, D.M.; Bennett, G.; Coady, S.; D’Agostino, R.B., Sr.; Gibbons, R.; Greenland, P.; Lackland, D.T.; Levy, D.; O’Donnell, C.J.; et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: A report of the American college of cardiology/American heart association task force on practice guidelines. J. Am. Coll. Cardiol. 2014, 129, 2935–2959. [Google Scholar] [CrossRef]
- Test for Association/Correlation Between Paired Samples. Available online: https://www.rdocumentation.org/packages/stats/versions/3.5.2/topics/cor.test (accessed on 18 March 2019).
- Lehamnn, E.L. Elements of Large Sample Theory; Springer-Verlag New York, Inc.: New York, NY, USA, 1999. [Google Scholar]
- Conover, W.J. Practical Nonparametric Statistics, 2nd ed.; John Wiley & Sons, Inc.: New York, NY, USA, 1980. [Google Scholar]
- Hair, J.F., Jr.; Anderson, R.E.; Tatham, R.L.; Black, W.C. Multivariate Data Analysis, 3rd ed.; Macmillan: New York, NY, USA, 1995. [Google Scholar]
- Vadiveloo, M.; Dixon, L.B.; Mijanovich, T.; Elbel, B.; Parekh, N. Dietary variety is inversely associated with body adiposity among us adults using a novel food diversity index. J. Nutr. 2015, 145, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Vadiveloo, M.; Sacks, F.M.; Champagne, C.M.; Bray, G.A.; Mattei, J. Greater healthful dietary variety is associated with greater 2-year changes in weight and adiposity in the preventing overweight using novel dietary strategies (POUNDS Lost) trial. J. Nutr. 2016, 146, 1552–1559. [Google Scholar] [CrossRef]
- Zhao, W.; Zhang, J.; Zhao, A.; Wang, M.; Wu, W.; Tan, S.; Guo, M.; Zhang, Y. Using an introduced index to assess the association between food diversity and metabolic syndrome and its components in Chinese adults. BMC Cardiovasc. Disord. 2018, 18, 189. [Google Scholar] [CrossRef]
- Kuczmarski, M.F.; Bodt, B.A.; Shupe, E.S.; Zonderman, A.B.; Evans, M.K. Dietary patterns associated with lower 10-year atherosclerotic cardiovascular disease risk among urban African-American and white adults consuming western diets. Nutrients 2018, 10, 158. [Google Scholar] [CrossRef]
- Tian, X.; Wu, M.; Zang, J.; Zhu, Y.; Wang, H. Dietary diversity and adiposity in chinese men and women: An analysis of four waves of cross-sectional survey data. Eur. J. Clin. Nutr. 2017, 71, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Sustainable Delopmental Goals. Available online: http://www.fao.org/sustainable-development-goals/en/ (accessed on 27 April 2019).
- Darmon, N.; Drewnowski, A. Does social class predict diet quality? Am. J. Clin. Nutr. 2008, 87, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Wood, A.M.; Kaptoge, S.; Butterworth, A.S.; Willeit, P.; Warnakula, S.; Bolton, T.; Paige, E.; Paul, D.S.; Sweeting, M.; Burgess, S.; et al. Risk thresholds for alcohol consumption: combined analysis of individual-participant data for 599 912 current drinkers in 83 prospective studies. Lancet 2018, 391, 1513–1523. [Google Scholar] [CrossRef]
- Rhodes, D.G.; Murayi, T.; Clemens, J.C.; Baer, D.J.; Sebastian, R.S.; Moshfegh, A.J. The USDA Automated Multiple-Pass Method accurately assesses population sodium intakes. Am. J. Clin. Nutr. 2013, 97, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Naska, A.; Lagiou, A.; Lagiou, P. Dietary assessment methods in epidemiological research: Current state of the art and future prospects. F1000Res. 2017, 6, 926. [Google Scholar] [CrossRef]
| Characteristic | Race | Income | ||||
|---|---|---|---|---|---|---|
| AA n = 1259 | W n = 807 | p | <125% Poverty n = 842 | >125% Poverty n = 1224 | p | |
| Age, year | 56.7 ± 0.3 | 56.6 ± 0.3 | 0.875 | 56.0 ± 0.3 | 57.1 ± 0.3 | 0.008 |
| Sex, % female | 59.1 | 58.9 | 0.916 | 63.4 | 56.0 | 0.001 |
| Education, % <high school | 30.9 | 32.7 | 0.386 | 32.3 | 31.1 | 0.572 |
| Food Insecurity, % insecure1 | 27.1 n = 1125 | 24.2 n = 744 | 0.159 | 31.3 n = 739 | 22.5 n = 1130 | <0.001 |
| Energy, kcal | 1948 ± 23 | 1992 ± 29 | 0.237 | 1945 ± 29 | 1979 ± 23 | 0.350 |
| Diversity: Count | 0.4446 | 0.4445 | 0.934 | 0.4287 | 0.4558 | <0.001 |
| Diversity: Evenness (HFBI) | 0.1284 | 0.1285 | 0.700 | 0.1277 | 0.1290 | 0.295 |
| Diversity: Evenness (BI) | 0.8078 | 0.8093 | 0.824 | 0.8034 | 0.8119 | 0.007 |
| Diversity: Dissimilarity2 | 0.8063 | 0.7944 | <0.001 | 0.8056 | 0.7990 | 0.056 |
| Mean Nutrient Adequacy | 73.1 ± 0.4 | 75.2 ± 0.5 | 0.001 | 72.6 ± 0.5 | 74.8 ± 0.4 | 0.001 |
| DASH score | 1.90 ± 0.03 | 2.25 ± 0.04 | <0.001 | 1.94 ± 0.04 | 2.11 ± 0.04 | 0.001 |
| DASH, % adherent3 | 4.1 | 5.8 | 0.064 | 3.8 | 5.4 | 0.094 |
| ASCVD risk, % | 56.2 ± 0.3 n = 778 | 56.3 ± 0.3 n = 536 | 0.864 | 55.5 ± 0.4 n = 509 | 56.7 ± 0.3 n = 805 | 0.012 |
| Food Group | Mean Equivalents | Food Group | Mean Equivalents |
|---|---|---|---|
| Total Fruit | 0.126 cup | Total Protein foods | 2.129 oz |
| Citrus, melons, berries | 0.032 cup | Total Meat, poultry, fish1 | 1.488 oz |
| Other fruits | 0.065 cup | Meat | 0.181 oz |
| Juices | 0.030 cup | Cured meat1 | 0.466 oz |
| Total vegetables | 0.767 cup | Organ meat1 | 0.006 oz |
| Dark green | 0.125 cup | Poultry | 0.757 oz |
| Total red and orange | 0.212 cup | Seafood high in n-3 fatty acids | 0.014 oz |
| Total starchy | 0.051 cup | Seafood low in n-3 fatty acids | 0.064 oz |
| Other vegetables | 0.368 cup | Eggs | 0.065 oz |
| Legumes | 0.010 cup | Soy products | 0.012 oz |
| Total grains | 1.360 oz | Nuts and seeds | 0.564 oz |
| Whole grains | 0.238 oz | ||
| Refined grains | 1.122 oz | Oils | 20.120 g |
| Total Dairy | 2.990 cup | ||
| Milk | 0.138 cup | Solid fats1 | 45.571 g |
| Yogurt | 0.001 cup | Sugars + Beverages1,2 | 26.648 tsp |
| Cheese | 2.845 cup | Alcoholic drinks1 | 0.009 drinks |
| Food Group | Energy, % Total1 | Race | Income | ||||
|---|---|---|---|---|---|---|---|
| AA | W | p | <125% Poverty | >125% Poverty | p | ||
| Total grains | 32.16 | 0.7077 | 0.7088 | 0.8392 | 0.6900 | 0.7206 | <0.0001 |
| Total protein foods2 | 27.33 | 0.3344 | 0.3197 | <0.0001 | 0.3207 | 0.3342 | 0.0015 |
| Total vegetables | 8.80 | 0.4558 | 0.4539 | 0.7581 | 0.4305 | 0.472 | <0.0001 |
| Total dairy | 8.31 | 0.4617 | 0.5118 | <0.0001 | 0.4677 | 0.4906 | 0.0035 |
| Total fruit | 3.65 | 0.3119 | 0.3036 | 0.1958 | 0.2819 | 0.3271 | <0.0001 |
| Oils | 2.04 | 0.9805 | 0.9734 | 0.1269 | 0.9762 | 0.9788 | 0.5909 |
| Covariate | Estimate | SE | p |
|---|---|---|---|
| Education (<high school vs. ≥high school) | −0.458 | 0.467 | 0.327 |
| Energy per kg body weight | −0.033 | 0.023 | 0.148 |
| Food security (insecure vs. secure) 1 | 2.615 | 0.53 | <0.001 |
| Income (>125% poverty vs. <125% poverty) | 0.848 | 0.468 | 0.07 |
| Count | 11.746 | 2.666 | <0.001 |
| Evenness—Health Factor-adjusted Berry Index | 9.055 | 4.736 | 0.056 |
| Dissimilarity2 | −6.301 | 3.051 | 0.039 |
| Mean Adequacy Ratio | −0.127 | 0.022 | <0.001 |
| Covariate | Estimate | SE | p |
|---|---|---|---|
| Education (<high school vs. ≥high school) | −0.536 | 0.472 | 0.256 |
| Energy per kg body weight | −0.101 | 0.02 | <0.001 |
| Food security (insecure vs. secure) 1 | 2.442 | 0.535 | <0.001 |
| Income (>125% poverty vs. <125% poverty) | 0.748 | 0.474 | 0.115 |
| Count | 5.289 | 2.427 | 0.030 |
| Evenness- Health Factor-adjusted Berry Index | 8.146 | 4.861 | 0.094 |
| Dissimilarity 2 | −8.875 | 3.25 | 0.006 |
| DASH score 3 | −0.395 | 0.216 | 0.067 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fanelli Kuczmarski, M.; Brewer, B.C.; Rawal, R.; Pohlig, R.T.; Zonderman, A.B.; Evans, M.K. Aspects of Dietary Diversity Differ in Their Association with Atherosclerotic Cardiovascular Risk in a Racially Diverse US Adult Population. Nutrients 2019, 11, 1034. https://doi.org/10.3390/nu11051034
Fanelli Kuczmarski M, Brewer BC, Rawal R, Pohlig RT, Zonderman AB, Evans MK. Aspects of Dietary Diversity Differ in Their Association with Atherosclerotic Cardiovascular Risk in a Racially Diverse US Adult Population. Nutrients. 2019; 11(5):1034. https://doi.org/10.3390/nu11051034
Chicago/Turabian StyleFanelli Kuczmarski, Marie, Benjamin C. Brewer, Rita Rawal, Ryan T. Pohlig, Alan B. Zonderman, and Michele K. Evans. 2019. "Aspects of Dietary Diversity Differ in Their Association with Atherosclerotic Cardiovascular Risk in a Racially Diverse US Adult Population" Nutrients 11, no. 5: 1034. https://doi.org/10.3390/nu11051034
APA StyleFanelli Kuczmarski, M., Brewer, B. C., Rawal, R., Pohlig, R. T., Zonderman, A. B., & Evans, M. K. (2019). Aspects of Dietary Diversity Differ in Their Association with Atherosclerotic Cardiovascular Risk in a Racially Diverse US Adult Population. Nutrients, 11(5), 1034. https://doi.org/10.3390/nu11051034





