Combined Effect of Arginine, Valine, and Serine on Exercise-Induced Fatigue in Healthy Volunteers: A Randomized, Double-Blinded, Placebo-Controlled Crossover Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
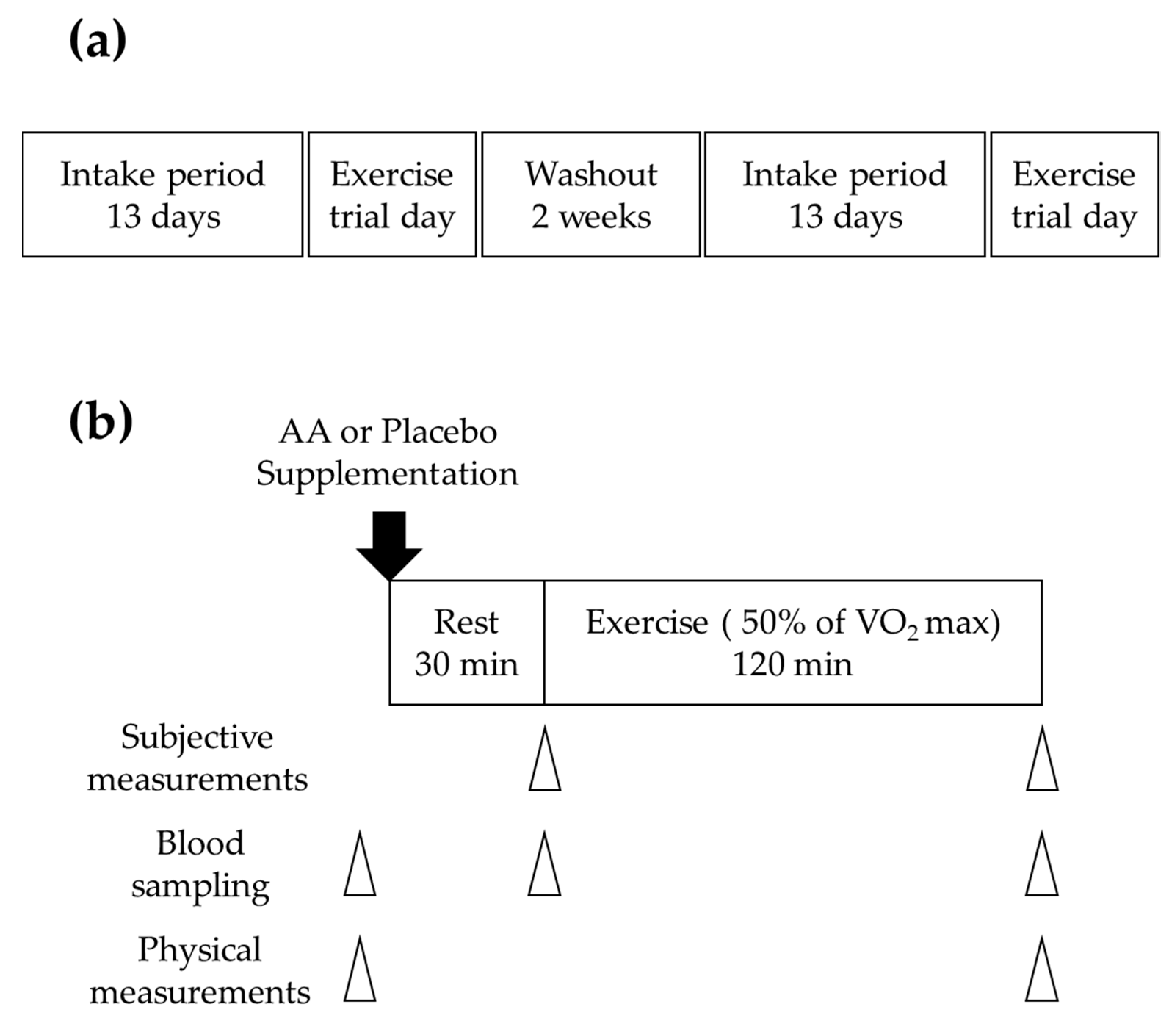
2.2. Study Design
2.3. Experimental Procedure
2.4. Subjective Measurements
2.5. Blood Sampling
2.6. Physical Measurements
2.7. Statistical Analysis
3. Results
3.1. Subjective Measurements
3.2. Blood Analysis
3.3. Physical Measurements
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grego, F.; Vallier, J.-M.; Collardeau, M.; Bermon, S.; Ferrari, P.; Candito, M.; Bayer, P.; Magnié, M.-N.; Brisswalter, J. Effects of long duration exercise on cognitive function, blood glucose, and counterregulatory hormones in male cyclists. Neurosci. Lett. 2004, 364, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Coyle, E.; Hagberg, J.; Hurley, B.; Martin, W.; Ehsani, A.; Holloszy, J. Carbohydrate feeding during prolonged strenuous exercise can delay fatigue. J. Appl. Physiol. 1983, 55, 230–235. [Google Scholar] [CrossRef]
- Coyle, E.F.; Coggan, A.R.; Hemmert, M.; Ivy, J.L. Muscle glycogen utilization during prolonged strenuous exercise when fed carbohydrate. J. Appl. Physiol. 1986, 61, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Jacks, D.E.; Sowash, J.; Anning, J.; Mcgloughlin, T.; Andres, F. Effect of exercise at three exercise intensities on salivary cortisol. J. Strength Cond. Res. 2002, 16, 286–289. [Google Scholar] [PubMed]
- Hill, E.; Zack, E.; Battaglini, C.; Viru, M.; Viru, A.; Hackney, A. Exercise and circulating cortisol levels: The intensity threshold effect. J. Endocrinol. Investig. 2008, 31, 587–591. [Google Scholar] [CrossRef]
- McGuigan, M.R.; Egan, A.D.; Foster, C. Salivary cortisol responses and perceived exertion during high intensity and low intensity bouts of resistance exercise. J. Sports Sci. Med. 2004, 3, 8. [Google Scholar] [PubMed]
- Caetano Júnior, P.; Castilho, M.; Raniero, L. Salivary cortisol responses and session ratings of perceived exertion to a rugby match and fatigue test. Percept. Mot. Skills 2017, 124, 649–661. [Google Scholar] [CrossRef]
- Mutch, B.; Banister, E. Ammonia metabolism in exercise and fatigue: A review. Med. Sci. Sports Exerc. 1983, 15, 41–50. [Google Scholar] [CrossRef]
- Banister, E.; Cameron, B. Exercise-induced hyperammonemia: Peripheral and central effects. Int. J. Sports Med. 1990, 11, S129–S142. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, A.; Piquard, F.; Geny, B.; Doutreleau, S.; Lampert, E.; Mettauer, B.; Lonsdorfer, J. L-arginine reduces exercise-induced increase in plasma lactate and ammonia. Int. J. Sports Med. 2002, 23, 403–407. [Google Scholar] [CrossRef]
- Camic, C.L.; Housh, T.J.; Zuniga, J.M.; Hendrix, R.C.; Mielke, M.; Johnson, G.O.; Schmidt, R.J. Effects of arginine-based supplements on the physical working capacity at the fatigue threshold. J. Strength Cond. Res. 2010, 24, 1306–1312. [Google Scholar] [CrossRef] [PubMed]
- Bednarz, B.; Jaxa-Chamiec, T.; Gębalska, J.; Herbaczyńska-Cedro, K.; Ceremużyński, L. L-arginine supplementation prolongs duration of exercise in congestive heart failure. Kardiol. Pol. (Pol. Heart J.) 2004, 60, 351–353. [Google Scholar]
- Bailey, S.J.; Winyard, P.G.; Vanhatalo, A.; Blackwell, J.R.; DiMenna, F.J.; Wilkerson, D.P.; Jones, A.M. Acute l-arginine supplementation reduces the O2 cost of moderate-intensity exercise and enhances high-intensity exercise tolerance. J. Appl. Physiol. 2010, 109, 1394–1403. [Google Scholar] [CrossRef] [PubMed]
- Greer, B.K.; Woodard, J.L.; White, J.P.; Arguello, E.M.; Haymes, E.M. Branched-chain amino acid supplementation and indicators of muscle damage after endurance exercise. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 595–607. [Google Scholar] [CrossRef] [PubMed]
- De Palo, E.; Gatti, R.; Cappellin, E.; Schiraldi, C.; De Palo, C.; Spinella, P. Plasma lactate, gh and gh-binding protein levels in exercise following bcaa supplementation in athletes. Amino. Acids 2001, 20, 1–11. [Google Scholar] [CrossRef]
- Blomstrand, E. A role for branched-chain amino acids in reducing central fatigue. J. Nutr. 2006, 136, 544S–547S. [Google Scholar] [CrossRef]
- Newsholme, E.A.; Blomstrand, E. Branched-chain amino acids and central fatigue. J. Nutr. 2006, 136, 274S–276S. [Google Scholar] [CrossRef]
- Crozier, S.J.; Kimball, S.R.; Emmert, S.W.; Anthony, J.C.; Jefferson, L.S. Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J. Nutr. 2005, 135, 376–382. [Google Scholar] [CrossRef]
- Anthony, J.C.; Yoshizawa, F.; Anthony, T.G.; Vary, T.C.; Jefferson, L.S.; Kimball, S.R. Leucine stimulates translation initiation in skeletal muscle of postabsorptive rats via a rapamycin-sensitive pathway. J. Nutr. 2000, 130, 2413–2419. [Google Scholar] [CrossRef]
- Tsuda, Y.; Iwasawa, K.; Yamaguchi, M. Acute supplementation of valine reduces fatigue during swimming exercise in rats. Biosci. Biotechnol. Biochem. 2018, 82, 856–861. [Google Scholar] [CrossRef]
- Wells, A.J.; Hoffman, J.R.; Gonzalez, A.M.; Stout, J.R.; Fragala, M.S.; Mangine, G.T.; McCormack, W.P.; Jajtner, A.R.; Townsend, J.R.; Robinson IV, E.H. Phosphatidylserine and caffeine attenuate postexercise mood disturbance and perception of fatigue in humans. Nutr. Res. 2013, 33, 464–472. [Google Scholar] [CrossRef]
- Kingsley, M.I.; Miller, M.; Kilduff, L.P.; McENENY, J.; Benton, D. Effects of phosphatidylserine on exercise capacity during cycling in active males. Med. Sci. Sports Exerc 2006, 38, 64–71. [Google Scholar] [CrossRef]
- Starks, M.A.; Starks, S.L.; Kingsley, M.; Purpura, M.; Jäger, R. The effects of phosphatidylserine on endocrine response to moderate intensity exercise. J. Int. Soc. Sports Nutr. 2008, 5, 11. [Google Scholar] [CrossRef]
- Gomez-Merino, D.; Bequet, F.; Berthelot, M.; Riverain, S.; Chennaoui, M.; Guezennec, C. Evidence that the branched-chain amino acid l-valine prevents exercise-induced release of 5-ht in rat hippocampus. Int. J. Sports Med. 2001, 22, 317–322. [Google Scholar] [CrossRef]
- Nishitani, S.; Matsumura, T.; Fujitani, S.; Sonaka, I.; Miura, Y.; Yagasaki, K. Leucine promotes glucose uptake in skeletal muscles of rats. Biochem. Biophys. Res. Commun. 2002, 299, 693–696. [Google Scholar] [CrossRef]
- Doi, M.; Yamaoka, I.; Fukunaga, T.; Nakayama, M. Isoleucine, a potent plasma glucose-lowering amino acid, stimulates glucose uptake in c2c12 myotubes. Biochem. Biophys. Res. Commun. 2003, 312, 1111–1117. [Google Scholar] [CrossRef]
- Watson, P.; Shirreffs, S.M.; Maughan, R.J. The effect of acute branched-chain amino acid supplementation on prolonged exercise capacity in a warm environment. Eur. J. Appl. Physiol. 2004, 93, 306–314. [Google Scholar] [CrossRef]
- Sugino, T.; Shirai, T.; Kajimoto, Y.; Kajimoto, O. L-ornithine supplementation attenuates physical fatigue in healthy volunteers by modulating lipid and amino acid metabolism. Nutr. Res. 2008, 28, 738–743. [Google Scholar] [CrossRef]
- Lee, K.A.; Hicks, G.; Nino-Murcia, G. Validity and reliability of a scale to assess fatigue. Psychiatry Res. 1991, 36, 291–298. [Google Scholar] [CrossRef]
- Crewe, H.; Tucker, R.; Noakes, T.D. The rate of increase in rating of perceived exertion predicts the duration of exercise to fatigue at a fixed power output in different environmental conditions. Eur. J. Appl. Physiol. 2008, 103, 569. [Google Scholar] [CrossRef]
- Newman, J.C.; Verdin, E. Ketone bodies as signaling metabolites. Trends Endocrinol. Metab. 2014, 25, 42–52. [Google Scholar] [CrossRef]
- Winder, W.W.; Holloszy, J.O.; Baldwin, K.M. Enzymes involved in ketone utilization in different types of muscle: Adaptation to exercise. Eur. J. Biochem. 1974, 47, 461–467. [Google Scholar] [CrossRef]
- Cox, P.J.; Clarke, K. Acute nutritional ketosis: Implications for exercise performance and metabolism. Extrem. Physiol. Med. 2014, 3, 17. [Google Scholar] [CrossRef]
- Volek, J.S.; Noakes, T.; Phinney, S.D. Rethinking fat as a fuel for endurance exercise. Eur. J. Sport Sci. 2015, 15, 13–20. [Google Scholar] [CrossRef]
- Pinckaers, P.J.; Churchward-Venne, T.A.; Bailey, D.; van Loon, L.J. Ketone bodies and exercise performance: The next magic bullet or merely hype? Sports Med. 2017, 47, 383–391. [Google Scholar] [CrossRef]
- Gerich, J.E.; Charles, M.A.; Grodsky, G.M. Characterization of the effects of arginine and glucose on glucagon and insulin release from the perfused rat pancreas. J. Clin. Investig. 1974, 54, 833–841. [Google Scholar] [CrossRef]
- Meijer, A.J.; Lamers, W.H.; Chamuleau, R. Nitrogen metabolism and ornithine cycle function. Physiol. Rev. 1990, 70, 701–748. [Google Scholar] [CrossRef]
- Suzuki, T.; Morita, M.; Kobayashi, Y.; Kamimura, A. Oral l-citrulline supplementation enhances cycling time trial performance in healthy trained men: Double-blind randomized placebo-controlled 2-way crossover study. J. Int. Soc. Sports Nutr. 2016, 13, 6. [Google Scholar] [CrossRef] [PubMed]
- Fernstrom, J.D.; Fernstrom, M.H. Exercise, serum free tryptophan, and central fatigue. J. Nutr. 2006, 136, 553S–559S. [Google Scholar] [CrossRef] [PubMed]
- Fernstrom, J.D.; Wurtman, R.J. Brain serotonin content: Physiological regulation by plasma neutral amino acids. Science 1972, 178, 414–416. [Google Scholar] [CrossRef]
- Fernstrom, J.D.; Faller, D.V. Neutral amino acids in the brain: Changes in response to food ingestion 1. J. Neurochem. 1978, 30, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Blood-brain barrier carrier-mediated transport and brain metabolism of amino acids. Neurochem. Res. 1998, 23, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Sureda, A.; Córdova, A.; Ferrer, M.D.; Pérez, G.; Tur, J.A.; Pons, A. L-citrulline-malate influence over branched chain amino acid utilization during exercise. Eur. J. Appl. Physiol. 2010, 110, 341–351. [Google Scholar] [CrossRef] [PubMed]
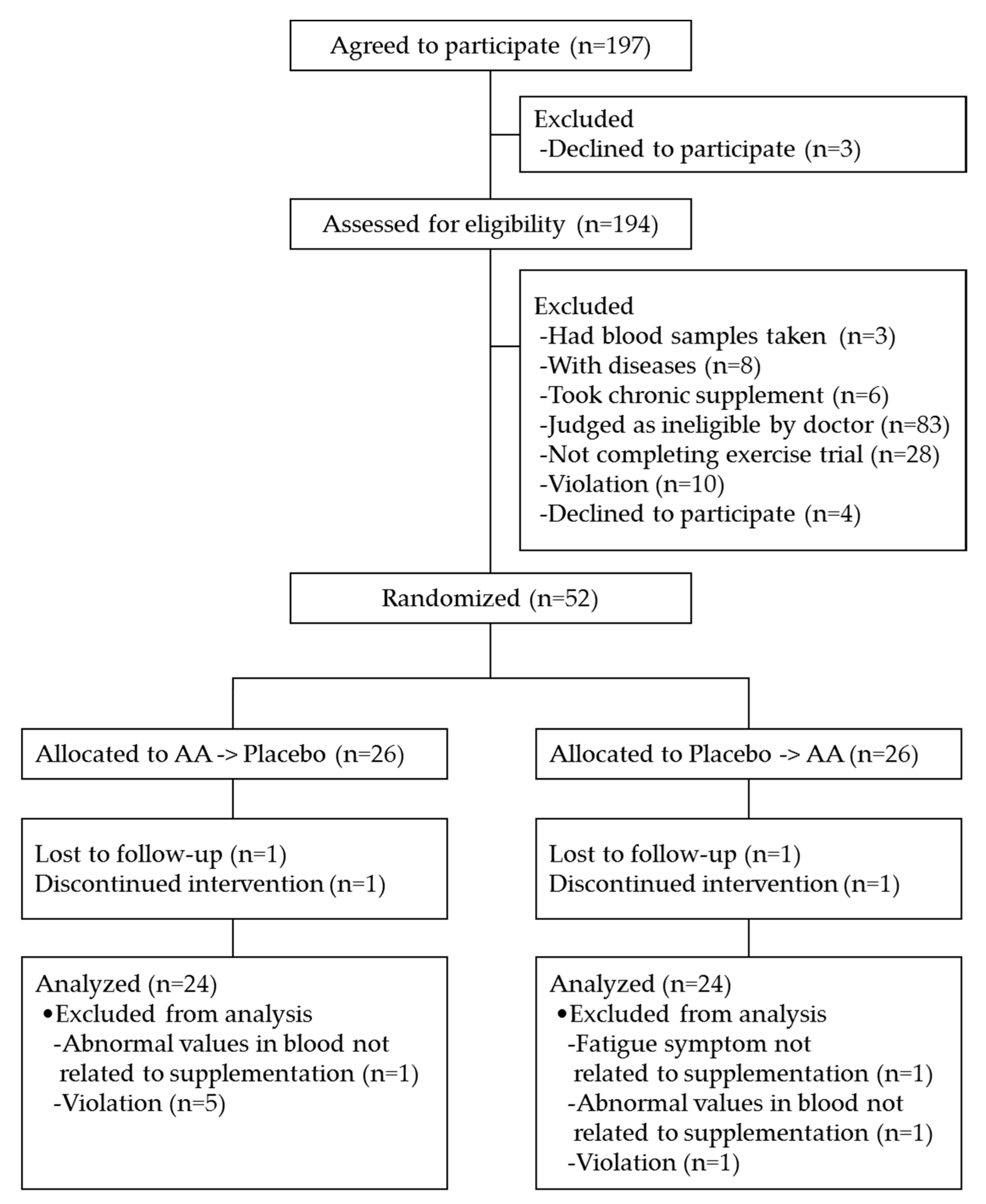
| AA -> Placebo | Placebo -> AA | |
|---|---|---|
| n = 18 | n = 21 | |
| Age (years) | 50.6 ± 6.5 | 49.8 ± 5.9 |
| Height (cm) | 168.8 ± 6.3 | 170.3 ± 6.0 |
| Body weight (kg) | 65.8 ± 6.9 | 68.8 ± 8.7 |
| VO2 max (mL/min/kg) | 34.7 ± 5.6 | 32.1 ± 4.5 |
| Before Exercise | After Exercise | p-Value for Interaction | p-Value for Treatment | p-Value for Time | |
|---|---|---|---|---|---|
| AA | 18.8 ± 11.4 | 58.2 ± 16.5 * | 0.132 | 0.006 | <0.001 |
| Placebo | 21.0 ± 12.5 | 62.6 ± 15.7 | |||
| Difference (95% CI) | −2.2 (−4.6, 0.1) | −4.4 (−7.4, −1.4) |
| Before Exercise | After Exercise | p-Value for Interaction | p-Value for Treatment | p-Value for Time | |
|---|---|---|---|---|---|
| AA | 7.7 ± 1.5 * | 13.7 ± 2.0 | 0.501 | 0.046 | <0.001 |
| Placebo | 8.1 ± 1.5 | 14.0 ± 2.3 | |||
| Difference (95% CI) | −0.5 (−0.8, −0.1) | −0.2 (−0.8, 0.3) |
| After Visit | Before Exercise | After Exercise | Change before and after Exercise (95% CI) | p-Value for Interaction | p-Value for Treatment | p-Value for Time | ||
|---|---|---|---|---|---|---|---|---|
| Plasma glucose (mg/dL) | AA | 88.8 ± 9.6 | 89.6 ± 6.6 | 80.4 ± 8.1 | −9.2 (−11.9, −6.5) | 0.454 | 0.323 | <0.001 |
| Placebo | 87.2 ± 9.1 | 89.7 ± 7.3 | 79.9 ± 6.4 | −9.7 (−11.7, −7.7) | ||||
| Blood lactate (mg/dL) | AA | 8.8 ± 3.1 | 7.8 ± 4.4 | 11.5 ± 3.6 | 3.7 (2.1, 5.3) | 0.531 | 0.191 | <0.001 |
| Placebo | 9.7 ± 3.6 | 8.3 ± 2.9 | 11.7 ± 3.6 | 3.4 (2.1, 4.7) | ||||
| Serum free fatty acid (mEq/L) | AA | 0.49 ± 0.21 | 0.42 ± 0.18 | 1.62 ± 0.43 | 1.20 (1.07, 1.33) | 0.210 | 0.841 | <0.001 |
| Placebo | 0.49 ± 0.27 | 0.38 ± 0.18 | 1.68 ± 0.41 | 1.29 (1.15, 1.44) | ||||
| Serum total ketone body (µmol/L) | AA | 172 ± 163 | 101 ± 85 | 582 ± 242 | 481 (419, 543) * | 0.034 | 0.739 | <0.001 |
| Placebo | 134 ± 135 | 72 ± 53 | 630 ± 255 | 559 (479, 638) | ||||
| Serum cortisol (µg/dL) | AA | 11.9 ± 3.9 | 10.9 ± 3.5 | 16.7 ± 6.3 | 5.8 (3.7, 7.9) | 0.644 | 0.672 | <0.001 |
| Placebo | 12.2 ± 4.1 | 10.7 ± 3.6 | 17.3 ± 6.7 | 6.6 (4.0, 9.2) |
| After Visit | Before Exercise | After Exercise | Change before and after Exercise (95% CI) | p-Value for Interaction | p-Value for Treatment | p-Value for Time | ||
|---|---|---|---|---|---|---|---|---|
| Aspartic acid (nmol/mL) | AA | 3.8 ± 1.2 ** | 3.7 ± 1.3 | 3.5 ± 1.2 | −0.1 (−0.6, 0.3) | 0.027 | 0.379 | <0.001 |
| Placebo | 4.3 ± 1.2 | 3.5 ± 1.1 | 3.6 ± 1.2 | 0.1 (−0.2, 0.5) | ||||
| Threonine (nmol/mL) | AA | 119.9 ± 22.5 ** | 125.5 ± 25.4 | 114.3 ± 22.1 ** | −11.2 (−17.6, −4.8) * | 0.010 | <0.001 | 0.008 |
| Placebo | 135.0 ± 26.2 | 128.8 ± 19.4 | 128.0 ± 21.8 | −0.8 (−6.2, 4.6) | ||||
| Serine (nmol/mL) | AA | 123.2 ± 25.2 | 128.3 ± 23.5 ** | 115.3 ± 17.3 | −13.0 (−18.9, −7.0) ** | 0.003 | 0.657 | 0.001 |
| Placebo | 126.4 ± 27.6 | 118.9 ± 20.5 | 118.6 ± 19.6 | −0.3 (−5.7, 5.0) | ||||
| Asparagine (nmol/mL) | AA | 48.0 ± 8.4 | 49.9 ± 8.8 | 45.9 ± 6.2 | −4.0 (−6.6, −1.4) | 0.234 | 0.376 | 0.030 |
| Placebo | 49.5 ± 10.2 | 49.1 ± 7.4 | 47.9 ± 8.3 | −1.2 (−3.7, 1.3) | ||||
| Glutamic acid (nmol/mL) | AA | 62.2 ± 23.6 | 54.2 ± 21.3 | 55.9 ± 22.2 | 1.7 (−4.1, 7.5) | 0.146 | 0.254 | <0.001 |
| Placebo | 67.7 ± 21.4 | 52.7 ± 17.7 | 58.4 ± 18.7 | 5.7 (0.9, 10.6) | ||||
| Glutamine (nmol/mL) | AA | 632.8 ± 91.6 | 675.0 ± 107.5 | 648.8 ± 67.2 | −26.2 (−57.2, 4.8) | 0.105 | 0.911 | 0.038 |
| Placebo | 643.2 ± 98.1 | 648.4 ± 56.3 | 667.9 ± 76.5 | 19.5 (−5.4, 44.4) | ||||
| Proline (nmol/mL) | AA | 151.2 ± 58.8 | 148.3 ± 49.3 | 142.6 ± 41.7 | −5.7 (−13.1, 1.7) | 0.214 | 0.233 | 0.004 |
| Placebo | 158.3 ± 46.0 | 147.9 ± 38.5 | 147.4 ± 40.1 | −0.5 (−6.2, 5.3) | ||||
| Glycine (nmol/mL) | AA | 250.4 ± 53.6 | 247.3 ± 52.4 | 227.2 ± 45.1 ** | −20.2 (−31.0, −9.4) | 0.383 | 0.020 | <0.001 |
| Placebo | 261.8 ± 64.8 | 251.9 ± 54.2 | 244.2 ± 54.1 | −7.7 (−21.7, 6.2) | ||||
| Alanine (nmol/mL) | AA | 420.9 ± 75.6 | 426.2 ± 85.5 | 401.2 ± 66.2 | −25.0 (−49.9, −0.1) | 0.174 | 0.174 | 0.009 |
| Placebo | 425.7 ± 89.8 | 406.5 ± 69.5 | 386.3 ± 66.0 | −20.2 (−41.5, 1.1) | ||||
| Citrulline (nmol/mL) | AA | 42.6 ± 8.8 ** | 44.5 ± 9.8 ** | 51.6 ± 9.2 ** | 7.1 (4.2, 10.1) | 0.004 | <0.001 | <0.001 |
| Placebo | 38.2 ± 7.9 | 34.3 ± 6.3 | 43.9 ± 6.8 | 9.6 (7.8, 11.4) | ||||
| Valine (nmol/mL) | AA | 488.3 ± 121.6 ** | 805.7 ± 190.1 ** | 596.1 ± 102.4 ** | −209.6 (−263.4, −155.8) ** | <0.001 | <0.001 | <0.001 |
| Placebo | 242.0 ± 53.4 | 253.6 ± 114.5 | 243.7 ± 37.8 | −9.9 (−48.9, 29.2) | ||||
| Cystine (nmol/mL) | AA | 32.6 ± 7.8 | 31.9 ± 7.3 | 33.3 ± 6.3 | 1.4 (−0.4, 3.2) | 0.903 | 0.823 | 0.079 |
| Placebo | 32.7 ± 7.7 | 31.4 ± 6.1 | 33.1 ± 6.4 | 1.7 (0.3, 3.1) | ||||
| Methionine (nmol/mL) | AA | 22.6 ± 3.8 ** | 22.9 ± 4.0 | 25.8 ± 3.4 ** | 2.9 (1.6, 4.3) * | 0.048 | <0.001 | <0.001 |
| Placebo | 24.6 ± 5.5 | 23.5 ± 3.2 | 28.4 ± 3.7 | 4.9 (3.8, 6.1) | ||||
| Isoleucine (nmol/mL) | AA | 58.0 ± 11.6 | 59.9 ± 13.9 | 68.1 ± 12.4 | 8.2 (4.2, 12.2) | 0.400 | 0.436 | <0.001 |
| Placebo | 60.1 ± 17.2 | 59.3 ± 11.9 | 69.6 ± 10.9 | 10.3 (7.2, 13.3) | ||||
| Leucine (nmol/mL) | AA | 123.0 ± 20.3 | 127.2 ± 26.6 | 140.6 ± 24.7 | 13.4 (5.4, 21.4) | 0.292 | 0.366 | <0.001 |
| Placebo | 123.8 ± 28.5 | 121.3 ± 20.8 | 138.7 ± 19.3 | 17.4 (11.6, 23.1) | ||||
| Tyrosine (nmol/mL) | AA | 54.7 ± 11.7 ** | 54.8 ± 11.3 | 62.1 ± 10.4 ** | 7.3 (4.1, 10.6) * | 0.079 | <0.001 | <0.001 |
| Placebo | 60.6 ± 15.2 | 57.3 ± 11.3 | 68.9 ± 10.1 | 11.7 (9.0, 14.4) | ||||
| Phenylalanine (nmol/mL) | AA | 59.4 ± 9.2 | 60.9 ± 11.2 | 64.9 ± 8.3 * | 4.0 (0.0, 7.9) | 0.285 | 0.040 | <0.001 |
| Placebo | 61.3 ± 9.5 | 61.1 ± 7.2 | 68.7 ± 7.5 | 7.5 (4.9, 10.1) | ||||
| β-Alanine (nmol/mL) | AA | 2.6 ± 1.3 | 3.1 ± 1.8 ** | 3.2 ± 1.5 ** | 0.1 (−0.6, 0.7) | 0.545 | <0.001 | 0.043 |
| Placebo | 2.2 ± 1.0 | 2.3 ± 1.2 | 2.5 ± 1.0 | 0.2 (−0.2, 0.5) | ||||
| Ornithine (nmol/mL) | AA | 86.7 ± 22.9 ** | 107.1 ± 35.2 ** | 100.7 ± 21.2 ** | −6.3 (−16.3, 3.6) | <0.001 | <0.001 | 0.003 |
| Placebo | 60.1 ± 16.0 | 58.0 ± 17.9 | 54.4 ± 10.1 | −3.6 (−9.1, 1.9) | ||||
| Tryptophan (nmol/mL) | AA | 38.1 ± 6.9 | 39.3 ± 8.6 | 28.0 ± 5.0 | −11.4 (−13.9, −8.8) | 0.481 | 0.257 | <0.001 |
| Placebo | 39.8 ± 8.9 | 39.3 ± 5.3 | 29.0 ± 6.3 | −10.3 (−12.2, −8.4) | ||||
| Lysine (nmol/mL) | AA | 188.9 ± 29.2 ** | 197.8 ± 40.3 | 188.6 ± 28.5 ** | −9.2 (−19.8, 1.4) ** | 0.005 | <0.001 | 0.914 |
| Placebo | 213.4 ± 40.5 | 201.6 ± 30.2 | 212.8 ± 28.5 | 11.2 (2.8, 19.6) | ||||
| Histidine (nmol/mL) | AA | 91.0 ± 13.3 | 92.9 ± 16.5 | 89.0 ± 12.4 | −3.9 (−8.3, 0.5) * | 0.095 | 0.573 | 0.317 |
| Placebo | 94.1 ± 17.0 | 89.6 ± 9.7 | 91.7 ± 13.2 | 2.1 (−1.5, 5.7) | ||||
| Arginine (nmol/mL) | AA | 144.5 ± 35.6 ** | 217.1 ± 55.2 ** | 168.9 ± 30.3 ** | −48.2 (−64.0, −32.4) ** | <0.001 | <0.001 | <0.001 |
| Placebo | 94.5 ± 22.8 | 95.6 ± 26.7 | 100.4 ± 17.8 | 4.8 (−2.7, 12.3) | ||||
| Ammonia (nmol/mL) | AA | 58.8 ± 12.9 | 56.4 ± 18.1 | 60.5 ± 14.3 | 4.0 (−0.8, 8.9) | 0.292 | 0.181 | 0.004 |
| Placebo | 63.3 ± 13.6 | 56.1 ± 12.0 | 63.3 ± 13.5 | 7.2 (2.6, 11.8) | ||||
| Tryptophan/BCAA | AA | 0.060 ± 0.019 ** | 0.041 ± 0.013 ** | 0.035 ± 0.008 ** | −0.006 (−0.010, −0.002) ** | <0.001 | <0.001 | <0.001 |
| Placebo | 0.095 ± 0.019 | 0.095 ± 0.020 | 0.065 ± 0.013 | −0.030 (−0.035, −0.025) |
| After Visit | After Exercise | p-Value for Interaction | p-Value for Treatment | p-Value for Time | ||
|---|---|---|---|---|---|---|
| Systolic blood pressure (mmHg) | AA | 111.7 ± 9.4 | 112.3 ± 9.9 | 0.075 | 0.144 | 0.673 |
| Placebo | 114.2 ± 10.8 | 112.6 ± 10.3 | ||||
| Diastolic blood pressure (mmHg) | AA | 73.9 ± 7.3 | 71.5 ± 9.1 | 0.850 | 0.427 | 0.022 |
| Placebo | 74.4 ± 7.7 | 72.3 ± 9.0 | ||||
| Pulse rate (bpm) | AA | 70.5 ± 9.4 | 95.5 ± 13.6 | 0.298 | 0.619 | <0.001 |
| Placebo | 71.0 ± 8.3 | 93.8 ± 13.6 | ||||
| Body temperature (°C) | AA | 36.1 ± 0.4 | 36.0 ± 0.4 | 0.147 | 0.551 | 0.780 |
| Placebo | 36.1 ± 0.4 | 36.1 ± 0.6 | ||||
| Body weight (kg) | AA | 65.8 ± 7.7 | 65.7 ± 7.6 | 0.670 | 0.216 | <0.001 |
| Placebo | 66.0 ± 7.7 | 65.8 ± 7.6 | ||||
| Body fat (%) | AA | 17.7 ± 4.5 | 18.4 ± 4.3 | 0.398 | 0.345 | <0.001 |
| Placebo | 17.6 ± 4.7 | 18.3 ± 4.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsuda, Y.; Yamaguchi, M.; Noma, T.; Okaya, E.; Itoh, H. Combined Effect of Arginine, Valine, and Serine on Exercise-Induced Fatigue in Healthy Volunteers: A Randomized, Double-Blinded, Placebo-Controlled Crossover Study. Nutrients 2019, 11, 862. https://doi.org/10.3390/nu11040862
Tsuda Y, Yamaguchi M, Noma T, Okaya E, Itoh H. Combined Effect of Arginine, Valine, and Serine on Exercise-Induced Fatigue in Healthy Volunteers: A Randomized, Double-Blinded, Placebo-Controlled Crossover Study. Nutrients. 2019; 11(4):862. https://doi.org/10.3390/nu11040862
Chicago/Turabian StyleTsuda, Yuichi, Makoto Yamaguchi, Teruyuki Noma, Eiji Okaya, and Hiroyuki Itoh. 2019. "Combined Effect of Arginine, Valine, and Serine on Exercise-Induced Fatigue in Healthy Volunteers: A Randomized, Double-Blinded, Placebo-Controlled Crossover Study" Nutrients 11, no. 4: 862. https://doi.org/10.3390/nu11040862
APA StyleTsuda, Y., Yamaguchi, M., Noma, T., Okaya, E., & Itoh, H. (2019). Combined Effect of Arginine, Valine, and Serine on Exercise-Induced Fatigue in Healthy Volunteers: A Randomized, Double-Blinded, Placebo-Controlled Crossover Study. Nutrients, 11(4), 862. https://doi.org/10.3390/nu11040862




