Adherence to DASH-Style Dietary Pattern Impacts on Adiponectin and Clustered Metabolic Risk in Older Women
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Assessment of Inflammatory and Metabolic Risk Factors
2.3. Assessment of Dietary Quality
2.4. Assessment of Physical Activity (PA)
2.5. Statistical Analyses
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- GBD 2016 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- Ford, E.S. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: A summary of the evidence. Diabetes Care 2005, 28, 1769–1778. [Google Scholar] [CrossRef]
- Maury, E.; Brichard, S.M. Adipokine dysregulation, adipose tissue inflammation and metabolic syndrome. Mol. Cell Endocrinol. 2010, 314, 1–16. [Google Scholar] [CrossRef]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- The U.S. Department of Human Health Services. 2018 Physical Activity Guidelines Advisory Committee Scientific Report; Department of Health and Human Services: Washington, DC, USA, 2018.
- Nilsson, A.; Bergens, O.; Kadi, F. Physical Activity Alters Inflammation in Older Adults by Different Intensity Levels. Med. Sci. Sports Exerc. 2018, 50, 1502–1507. [Google Scholar] [CrossRef]
- Nilsson, A.; Montiel Rojas, D.; Kadi, F. Impact of Meeting Different Guidelines for Protein Intake on Muscle Mass and Physical Function in Physically Active Older Women. Nutrients 2018, 10, 1156. [Google Scholar] [CrossRef] [PubMed]
- AlEssa, H.B.; Malik, V.S.; Yuan, C.; Willett, W.C.; Huang, T.; Hu, F.B.; Tobias, D.K. Dietary patterns and cardiometabolic and endocrine plasma biomarkers in US women. Am. J. Clin. Nutr. 2017, 105, 432–441. [Google Scholar] [CrossRef]
- Livingstone, K.M.; McNaughton, S.A. Association between diet quality, dietary patterns and cardiometabolic health in Australian adults: A cross-sectional study. Nutr. J. 2018, 17, 19. [Google Scholar] [CrossRef]
- World Health Organization. Diet, Nutrition and the Prevention of Chronic Diseases; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Shivappa, N.; Steck, S.E.; Hurley, T.G.; Hussey, J.R.; Hebert, J.R. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. 2014, 17, 1689–1696. [Google Scholar] [CrossRef]
- Siervo, M.; Lara, J.; Chowdhury, S.; Ashor, A.; Oggioni, C.; Mathers, J.C. Effects of the Dietary Approach to Stop Hypertension (DASH) diet on cardiovascular risk factors: A systematic review and meta-analysis. Br. J. Nutr. 2015, 113, 1–15. [Google Scholar] [CrossRef]
- Garcia-Arellano, A.; Ramallal, R.; Ruiz-Canela, M.; Salas-Salvado, J.; Corella, D.; Shivappa, N.; Schroder, H.; Hebert, J.R.; Ros, E.; Gomez-Garcia, E.; et al. Dietary Inflammatory Index and Incidence of Cardiovascular Disease in the PREDIMED Study. Nutrients 2015, 7, 4124–4138. [Google Scholar] [CrossRef]
- Bondonno, N.P.; Lewis, J.R.; Blekkenhorst, L.C.; Shivappa, N.; Woodman, R.J.; Bondonno, C.P.; Ward, N.C.; Hebert, J.R.; Thompson, P.L.; Prince, R.L.; et al. Dietary inflammatory index in relation to sub-clinical atherosclerosis and atherosclerotic vascular disease mortality in older women. Br. J. Nutr. 2017, 117, 1577–1586. [Google Scholar] [CrossRef]
- Park, S.Y.; Kang, M.; Wilkens, L.R.; Shvetsov, Y.B.; Harmon, B.E.; Shivappa, N.; Wirth, M.D.; Hebert, J.R.; Haiman, C.A.; Le Marchand, L.; et al. The Dietary Inflammatory Index and All-Cause, Cardiovascular Disease, and Cancer Mortality in the Multiethnic Cohort Study. Nutrients 2018, 10, 1844. [Google Scholar] [CrossRef]
- Soltani, S.; Chitsazi, M.J.; Salehi-Abargouei, A. The effect of dietary approaches to stop hypertension (DASH) on serum inflammatory markers: A systematic review and meta-analysis of randomized trials. Clin. Nutr. 2018, 37, 542–550. [Google Scholar] [CrossRef]
- Namazi, N.; Larijani, B.; Azadbakht, L. Dietary Inflammatory Index and its Association with the Risk of Cardiovascular Diseases, Metabolic Syndrome, and Mortality: A Systematic Review and Meta-Analysis. Horm. Metab. Res. 2018, 50, 345–358. [Google Scholar] [CrossRef]
- Vissers, L.E.; Waller, M.A.; van der Schouw, Y.T.; Hebert, J.R.; Shivappa, N.; Schoenaker, D.A.; Mishra, G.D. The relationship between the dietary inflammatory index and risk of total cardiovascular disease, ischemic heart disease and cerebrovascular disease: Findings from an Australian population-based prospective cohort study of women. Atherosclerosis 2016, 253, 164–170. [Google Scholar] [CrossRef]
- Shivappa, N.; Hebert, J.R.; Rietzschel, E.R.; De Buyzere, M.L.; Langlois, M.; Debruyne, E.; Marcos, A.; Huybrechts, I. Associations between dietary inflammatory index and inflammatory markers in the Asklepios Study. Br. J. Nutr. 2015, 113, 665–671. [Google Scholar] [CrossRef]
- Neale, E.P.; Batterham, M.J.; Tapsell, L.C. Consumption of a healthy dietary pattern results in significant reductions in C-reactive protein levels in adults: A meta-analysis. Nutr. Res. 2016, 36, 391–401. [Google Scholar] [CrossRef]
- Phillips, C.M.; Shivappa, N.; Hebert, J.R.; Perry, I.J. Dietary Inflammatory Index and Biomarkers of Lipoprotein Metabolism, Inflammation and Glucose Homeostasis in Adults. Nutrients 2018, 10, 1033. [Google Scholar] [CrossRef]
- Erlinger, T.P.; Miller, E.R., 3rd; Charleston, J.; Appel, L.J. Inflammation modifies the effects of a reduced-fat low-cholesterol diet on lipids: Results from the DASH-sodium trial. Circulation 2003, 108, 150–154. [Google Scholar] [CrossRef]
- Alkerwi, A.; Shivappa, N.; Crichton, G.; Hebert, J.R. No significant independent relationships with cardiometabolic biomarkers were detected in the Observation of Cardiovascular Risk Factors in Luxembourg study population. Nutr. Res. 2014, 34, 1058–1065. [Google Scholar] [CrossRef]
- Vishram, J.K.; Borglykke, A.; Andreasen, A.H.; Jeppesen, J.; Ibsen, H.; Jorgensen, T.; Palmieri, L.; Giampaoli, S.; Donfrancesco, C.; Kee, F.; et al. Impact of age and gender on the prevalence and prognostic importance of the metabolic syndrome and its components in Europeans. The MORGAM Prospective Cohort Project. PLoS ONE 2014, 9, e107294. [Google Scholar] [CrossRef]
- Franceschi, C.; Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, S4–S9. [Google Scholar] [CrossRef]
- Nilsson, A.; Wahlin-Larsson, B.; Kadi, F. Physical activity and not sedentary time per se influences on clustered metabolic risk in elderly community-dwelling women. PLoS ONE 2017, 12, e0175496. [Google Scholar] [CrossRef]
- Fung, T.T.; Chiuve, S.E.; McCullough, M.L.; Rexrode, K.M.; Logroscino, G.; Hu, F.B. Adherence to a DASH-style diet and risk of coronary heart disease and stroke in women. Arch. Intern. Med. 2008, 168, 713–720. [Google Scholar] [CrossRef]
- Troiano, R.P.; Berrigan, D.; Dodd, K.W.; Masse, L.C.; Tilert, T.; McDowell, M. Physical activity in the United States measured by accelerometer. Med. Sci. Sports Exerc. 2008, 40, 181–188. [Google Scholar] [CrossRef]
- Gil-Campos, M.; Canete, R.R.; Gil, A. Adiponectin, the missing link in insulin resistance and obesity. Clin. Nutr. 2004, 23, 963–974. [Google Scholar] [CrossRef]
- Mantzoros, C.S.; Williams, C.J.; Manson, J.E.; Meigs, J.B.; Hu, F.B. Adherence to the Mediterranean dietary pattern is positively associated with plasma adiponectin concentrations in diabetic women. Am. J. Clin. Nutr. 2006, 84, 328–335. [Google Scholar] [CrossRef]
- Fargnoli, J.L.; Fung, T.T.; Olenczuk, D.M.; Chamberland, J.P.; Hu, F.B.; Mantzoros, C.S. Adherence to healthy eating patterns is associated with higher circulating total and high-molecular-weight adiponectin and lower resistin concentrations in women from the Nurses’ Health Study. Am. J. Clin. Nutr. 2008, 88, 1213–1224. [Google Scholar] [CrossRef]
- Azadbakht, L.; Surkan, P.J.; Esmaillzadeh, A.; Willett, W.C. The Dietary Approaches to Stop Hypertension eating plan affects C-reactive protein, coagulation abnormalities, and hepatic function tests among type 2 diabetic patients. J. Nutr. 2011, 141, 1083–1088. [Google Scholar] [CrossRef]
- Parikh, A.; Lipsitz, S.R.; Natarajan, S. Association between a DASH-like diet and mortality in adults with hypertension: Findings from a population-based follow-up study. Am. J. Hypertens. 2009, 22, 409–416. [Google Scholar] [CrossRef]
- Chiavaroli, L.; Viguiliouk, E.; Nishi, S.K.; Blanco Mejia, S.; Rahelic, D.; Kahleova, H.; Salas-Salvado, J.; Kendall, C.W.; Sievenpiper, J.L. DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Nutrients 2019, 11, 338. [Google Scholar] [CrossRef]
- Soltani, S.; Shirani, F.; Chitsazi, M.J.; Salehi-Abargouei, A. The effect of dietary approaches to stop hypertension (DASH) diet on weight and body composition in adults: A systematic review and meta-analysis of randomized controlled clinical trials. Obes. Rev. 2016, 17, 442–454. [Google Scholar] [CrossRef]
- Saneei, P.; Salehi-Abargouei, A.; Esmaillzadeh, A.; Azadbakht, L. Influence of Dietary Approaches to Stop Hypertension (DASH) diet on blood pressure: A systematic review and meta-analysis on randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1253–1261. [Google Scholar] [CrossRef]
- Ren, Z.; Zhao, A.; Wang, Y.; Meng, L.; Szeto, I.M.; Li, T.; Gong, H.; Tian, Z.; Zhang, Y.; Wang, P. Association between Dietary Inflammatory Index, C-Reactive Protein and Metabolic Syndrome: A Cross-Sectional Study. Nutrients 2018, 10, 831. [Google Scholar] [CrossRef]
- Alkerwi, A.; Vernier, C.; Crichton, G.E.; Sauvageot, N.; Shivappa, N.; Hebert, J.R. Cross-comparison of diet quality indices for predicting chronic disease risk: Findings from the Observation of Cardiovascular Risk Factors in Luxembourg (ORISCAV-LUX) study. Br. J. Nutr. 2015, 113, 259–269. [Google Scholar] [CrossRef]
- Boden, S.; Wennberg, M.; Van Guelpen, B.; Johansson, I.; Lindahl, B.; Andersson, J.; Shivappa, N.; Hebert, J.R.; Nilsson, L.M. Dietary inflammatory index and risk of first myocardial infarction; a prospective population-based study. Nutr. J. 2017, 16, 21. [Google Scholar] [CrossRef]
| Subject Characteristics | Tertile 1 | Tertile 2 | Tertile 3 | Total |
|---|---|---|---|---|
| Age (year) | 67.5 (1.7) | 67.3 (1.5) | 67.5 (1.7) | 67 (1.6) |
| Total energy intake (Kcal) | 1675 (332) | 1689 (372) | 1706 (427) | 1689 (372) |
| Daily time in MVPA (min) | 23 (16) | 31 (23) | 38 (32) | 30 (24) |
| Medication (% yes) | 32 | 31 | 34 | 32 |
| Risk Outcomes | Total | Tertile 1 | Tertile 2 | Tertile 3 | p-Value |
|---|---|---|---|---|---|
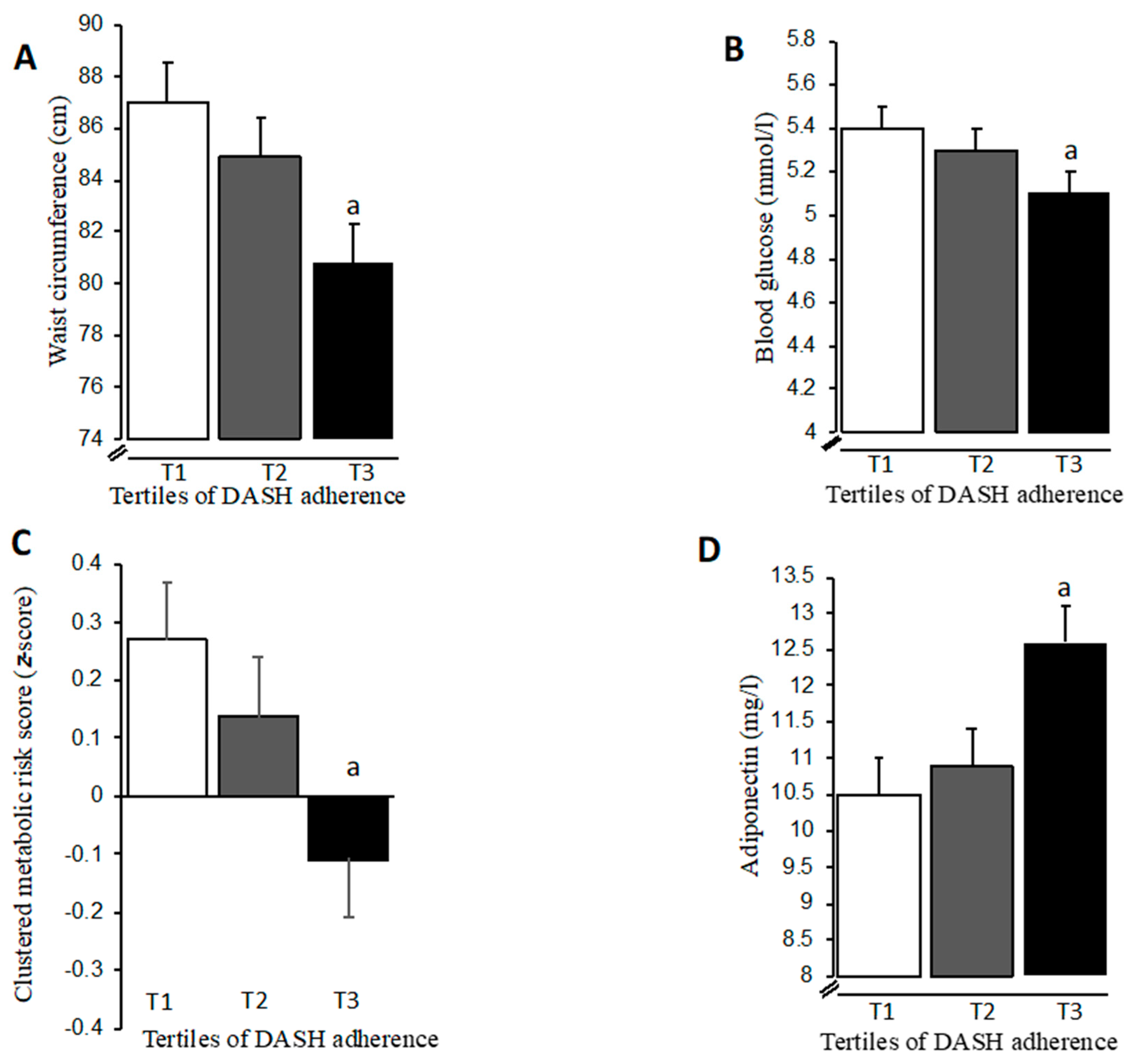
| Waist circumference (cm) | 84 (11) | 88 (9) | 84 (11) | 79 (11) a | p = 0.002 |
| Triglycerides (mmol/L) | 1.2 (0.4) | 1.2 (0.5) | 1.1 (0.4) | 1 (0.3) | p = 0.21 |
| HDL-cholesterol (mmol/L) | 1.6 (0.4) | 1.5 (0.3) | 1.5 (0.3) | 1.7 (0.4) | p = 0.12 |
| Glucose (mmol/L) | 5.3 (0.6) | 5.5 (0.6) | 5.3 (0.5) | 5 (0.7) a | p = 0.005 |
| Systolic blood pressure (mmHg) | 136 (15) | 134 (15) | 134 (16) | 139 (14) | p = 0.32 |
| Diastolic blood pressure (mmHg) | 78 (8) | 79 (9) | 76 (9) | 79 (7) | p = 0.24 |
| CRP (mg/L) * | 1.1 (1.5) | 1.3 (1.6) | 1 (1.7) | 1 (0.8) | p = 0.26 |
| Fibrinogen (g/L) | 3.2 (0.6) | 3.2 (0.5) | 3.2 (0.7) | 3.3 (0.6) | p = 0.64 |
| Adiponectin (mg/L) | 11.5 (3.4) | 10.6 (3.4) | 11.2 (3.3) | 12.9 (3.3) a | p = 0.008 |
| Risk Outcomes | Tertile 1 | Tertile 2 | Tertile 3 | p-Value |
|---|---|---|---|---|
| Waist circumference (cm) | 84 (12) | 82 (11) | 86 (9) | p = 0.39 |
| Triglycerides (mmol/L) | 1.1 (0.4) | 1.1 (0.4) | 1.2 (0.5) | p = 0.79 |
| HDL-cholesterol (mmol/L) | 1.6 (0.4) | 1.5 (0.3) | 1.6 (0.4) | p = 0.62 |
| Glucose (mmol/L) | 5.3 (0.7) | 5.2 (0.5) | 5.3 (0.5) | p = 0.95 |
| Systolic blood pressure (mmHg) | 136 (15) | 138 (15) | 133 (15) | p = 0.26 |
| Diastolic blood pressure (mmHg) | 77 (7) | 78 (10) | 77 (8) | p = 0.95 |
| CRP (mg/L) * | 1 (1.8) | 1.1 (1.3) | 1.2 (1.2) | p = 0.65 |
| Fibrinogen (g/L) | 3.2 (0.6) | 3.3 (0.5) | 3.2 (0.8) | p = 0.72 |
| Adiponectin (mg/L) | 11.3 (3.6) | 11.4 (3.2) | 11.7 (3.4) | p = 0.88 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nilsson, A.; Halvardsson, P.; Kadi, F. Adherence to DASH-Style Dietary Pattern Impacts on Adiponectin and Clustered Metabolic Risk in Older Women. Nutrients 2019, 11, 805. https://doi.org/10.3390/nu11040805
Nilsson A, Halvardsson P, Kadi F. Adherence to DASH-Style Dietary Pattern Impacts on Adiponectin and Clustered Metabolic Risk in Older Women. Nutrients. 2019; 11(4):805. https://doi.org/10.3390/nu11040805
Chicago/Turabian StyleNilsson, Andreas, Patrik Halvardsson, and Fawzi Kadi. 2019. "Adherence to DASH-Style Dietary Pattern Impacts on Adiponectin and Clustered Metabolic Risk in Older Women" Nutrients 11, no. 4: 805. https://doi.org/10.3390/nu11040805
APA StyleNilsson, A., Halvardsson, P., & Kadi, F. (2019). Adherence to DASH-Style Dietary Pattern Impacts on Adiponectin and Clustered Metabolic Risk in Older Women. Nutrients, 11(4), 805. https://doi.org/10.3390/nu11040805







