Rosmarinic and Sinapic Acids May Increase the Content of Reduced Glutathione in the Lenses of Estrogen-Deficient Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Drugs
2.2. Experimental Design
- sham-operated control rats (SHAM);
- ovariectomized control rats (OVX);
- ovariectomized rats treated with estradiol at a dose of 0.2 mg/kg (OVX+ESTR);
- ovariectomized rats treated with rosmarinic acid at a dose of 10 mg/kg (OVX+RA10);
- ovariectomized rats treated with rosmarinic acid at a dose of 50 mg/kg (OVX+RA50);
- ovariectomized rats treated with sinapic acid at a dose of 5 mg/kg (OVX+SA5);
- ovariectomized rats treated with sinapic acid at a dose of 25 mg/kg (OVX+SA25).
2.3. Determination of Soluble Protein in the Lenses
2.4. Determination of Superoxide Dismutase and Catalase Activities and Oxidative Damage Products Content in the Lenses
2.5. Determination of Glutathione-related Enzymes Activities in the Lenses
2.6. Determination of Glutathione in the Lenses
2.7. Determination of Amyloid β1-42 Content in the Lenses
2.8. Statistical Analysis
3. Results
3.1. Effect of Rosmarinic Acid and Sinapic Acid on the Lens Mass and Lens Soluble Protein Content
3.2. Effect of Rosmarinic Acid and Sinapic Acid on Superoxide Dismutase and Catalase Activities and on Oxidative Damage Products Content in the Lenses
3.3. Effect of Rosmarinic Acid and Sinapic Acid on Glutathione-Related Enzymes Activities in the Lenses
3.4. Effect of Rosmarinic Acid and Sinapic Acid on Glutathione Content in the Lenses
3.5. Effect of Rosmarinic Acid and Sinapic Acid on Amyloid β1-42 Content in the Lenses
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klein, B.E.K.; Klein, R.; Linton, K.L.P. Prevalence of age-related lens opacities in a population. The Beaver Dam Eye Study. Ophthalmology 1992, 99, 546–552. [Google Scholar] [CrossRef]
- Zhang, J.S.; Xu, L.; Wang, Y.X.; You, Q.S.; Da Wang, J.; Jonas, J.B. Five-year incidence of age-related cataract and cataract surgery in the adult population of Greater Beijing. The Beijing Eye Study. Ophthalmology 2011, 118, 711–718. [Google Scholar] [CrossRef]
- Lai, K.; Cui, J.; Ni, S.; Zhang, Y.; He, J.; Yao, K. The effects of postmenopausal hormone use on cataract: A meta-analysis. PLoS ONE 2013, 8, e78647. [Google Scholar] [CrossRef]
- Celojevic, D.; Petersen, A.; Karlsson, J.-O.; Behndig, A.; Zetterberg, M. Effects of 17β-estradiol on proliferation, cell viability and intracellular redox status in native human lens epithelial cells. Mol. Vis. 2011, 17, 1987–1996. [Google Scholar] [PubMed]
- Gottipati, S.; Cammarata, P.R. Mitochondrial superoxide dismutase activation with 17 β-estradiol-treated human lens epithelial cells. Mol. Vis. 2008, 14, 898–905. [Google Scholar] [PubMed]
- Dynlacht, J.R.; Valluri, S.; Lopez, J.; Greer, F.; DesRosiers, C.; Caperell-Grant, A.; Mendonca, M.S.; Bigsby, R.M. Estrogen protects against radiation-induced cataractogenesis. Radiat. Res. 2008, 170, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Dynlacht, J.R.; Tyree, C.; Valluri, S.; Desrosiers, C.; Mendonca, M.S.; Timmerman, R.; Bigsby, R.M.; Mendonca, M.S.; Desrosiers, C.; Tyree, C.; et al. Effect of estrogen on radiation-induced cataractogenesis. Radiat. Res. 2006, 165, 9–15. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bigsby, R.M.; Valluri, S.; Lopez, J.; Mendonca, M.S.; Caperell-Grant, A.; Desrosiers, C.; Dynlacht, J.R. Ovarian hormone modulation of radiation-induced cataractogenesis: Dose-response studies. Invest. Ophthalmol. Vis. Sci. 2009, 50, 3304–3310. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zetterberg, M.; Celojevic, D. Gender and cataract—The role of estrogen. Curr. Eye Res. 2015, 40, 176–190. [Google Scholar] [CrossRef]
- Cagnacci, A.; Cannoletta, M.; Palma, F.; Bellafronte, M.; Romani, C.; Palmieri, B. Relation between oxidative stress and climacteric symptoms in early postmenopausal women. Climacteric 2015, 18, 631–636. [Google Scholar] [CrossRef]
- Manolagas, S.C. From estrogen-centric to aging and oxidative stress: A revised perspective of the pathogenesis of osteoporosis. Endocr. Rev. 2010, 31, 266–300. [Google Scholar] [CrossRef]
- Cervellati, C.; Bergamini, C.M. Oxidative damage and the pathogenesis of menopause related disturbances and diseases. Clin. Chem. Lab. Med. 2016, 54, 739–753. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-C.; Wilkins, M.; Kim, T.; Malyugin, B.; Mehta, J.S. Cataracts. Lancet 2017, 390, 600–612. [Google Scholar] [CrossRef]
- Nita, M.B.; Grzybowski, A. The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxid. Med. Cell. Longev. 2016, 2016, 3164734. [Google Scholar] [CrossRef]
- Babizhayev, M.A.; Yegorov, Y.E. Reactive oxygen species and the aging eye: Specific role of metabolically active mitochondria in maintaining lens function and in the initiation of the oxidation-induced maturity onset cataract—A novel platform of mitochondria-targeted antioxidants with broad therapeutic potential for redox regulation and detoxification of oxidants in eye diseases. Am. J. Ther. 2016, 23, 98–117. [Google Scholar]
- Miquel, J.; Ramírez-Boscá, A.; Ramírez-Bosca, J.V.; Alperi, J.D. Menopause: A review on the role of oxygen stress and favorable effects of dietary antioxidants. Arch. Gerontol. Geriatr. 2006, 42, 289–306. [Google Scholar] [CrossRef]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Amoah, S.K.S.; Sandjo, L.P.; Kratz, J.M.; Biavatti, M.W. Rosmarinic acid—Pharmaceutical and clinical aspects. Planta Med. 2016, 82, 388–406. [Google Scholar] [CrossRef] [PubMed]
- Nićiforović, N.; Abramovič, H. Sinapic acid and its derivatives: Natural sources and bioactivity. Compr. Rev. Food Sci. Food Saf. 2014, 13, 34–51. [Google Scholar] [CrossRef]
- Zych, M.; Kaczmarczyk-Sedlak, I.; Wojnar, W.; Folwarczna, J. Effect of rosmarinic acid on the serum parameters of glucose and lipid metabolism and oxidative stress in estrogen-deficient rats. Nutrients 2019, 11, 267. [Google Scholar] [CrossRef]
- Zych, M.; Kaczmarczyk-Sedlak, I.; Wojnar, W.; Folwarczna, J. The effects of sinapic acid on the development of metabolic disorders induced by estrogen deficiency in rats. Oxid. Med. Cell. Longev. 2018, 2018, 9274246. [Google Scholar] [CrossRef]
- Lim, V.; Schneider, E.; Wu, H.; Pang, I.-H. Cataract preventive role of isolated phytoconstituents: Findings from a decade of research. Nutrients 2018, 10, 1580. [Google Scholar] [CrossRef]
- Kaur, A.; Gupta, V.; Francis, A.; Ahmad, M.; Bansal, P. Nutraceuticals in prevention of cataract—An evidence based approach. Saudi J. Ophthalmol. 2017, 31, 30–37. [Google Scholar] [CrossRef]
- Sunkireddy, P.; Jha, S.N.; Kanwar, J.R.; Yadav, S.C. Natural antioxidant biomolecules promises future nanomedicine based therapy for cataract. Colloids Surf. B Biointerfaces 2013, 112, 554–562. [Google Scholar] [CrossRef]
- Chemerovski-Glikman, M.; Mimouni, M.; Dagan, Y.; Haj, E.; Vainer, I.; Allon, R.; Blumenthal, E.Z.; Adler-abramovich, L.; Segal, D.; Gazit, E. Rosmarinic acid restores complete transparency of sonicated human cataract ex vivo and delays cataract formation in vivo. Sci. Rep. 2018, 8, 9341. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Friedlander, M.; Capeillère-Blandin, C.; Nguyen-Khoa, T.; Nguyen, A.T.; Zingraff, J.; Jungers, P.; Descamps-Latscha, B. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 1996, 49, 1304–1313. [Google Scholar] [CrossRef]
- Bassnett, S.; Shi, Y.; Vrensen, G.F.J.M. Biological glass: Structural determinants of eye lens transparency. Philos. Trans. R. Soc. B 2011, 366, 1250–1264. [Google Scholar] [CrossRef]
- Lim, J.C.; Umapathy, A.; Grey, A.C.; Vaghefi, E.; Donaldson, P.J. Novel roles for the lens in preserving overall ocular health. Exp. Eye Res. 2017, 156, 117–123. [Google Scholar] [CrossRef]
- Sella, R.; Afshari, N.A. Nutritional effect on age-related cataract formation and progression. Curr. Opin. Ophthalmol. 2019, 30, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Jayanthy, G.; Subramanian, S. Rosmarinic acid, a polyphenol, ameliorates hyperglycemia by regulating the key enzymes of carbohydrate metabolism in high fat diet—STZ induced experimental diabetes mellitus. Biomed. Prev. Nutr. 2014, 4, 431–437. [Google Scholar] [CrossRef]
- Bais, S.; Kumari, R.; Prashar, Y. Therapeutic effect of sinapic acid in aluminium chloride induced dementia of Alzheimer’s type in rats. J. Acute Dis. 2017, 6, 154–162. [Google Scholar] [CrossRef]
- Fan, X.; Monnier, V.M.; Whitson, J. Lens glutathione homeostasis: Discrepancies and gaps in knowledge standing in the way of novel therapeutic approaches. Exp. Eye Res. 2017, 156, 103–111. [Google Scholar] [CrossRef]
- Nye-Wood, M.G.; Spraggins, J.M.; Caprioli, R.M.; Schey, K.L.; Donaldson, P.J.; Grey, A.C. Spatial distributions of glutathione and its endogenous conjugates in normal bovine lens and a model of lens aging. Exp. Eye Res. 2017, 154, 70–78. [Google Scholar] [CrossRef]
- Lou, M.F. Redox regulation in the lens. Prog. Retin. Eye Res. 2003, 22, 657–682. [Google Scholar] [CrossRef]
- Dalle-Donne, I.; Milzani, A.; Gagliano, N.; Colombo, R.; Giustarini, D.; Rossi, R. Molecular mechanisms and potential clinical significance of S-glutathionylation. Antioxid. Redox Signal. 2008, 10, 445–473. [Google Scholar] [CrossRef]
- Xing, K.-Y.; Lou, M.F. Effect of age on the thioltransferase (glutaredoxin) and thioredoxin systems in the human lens. Investig. Ophthalmol. Vis. Sci. 2010, 51, 6598–6604. [Google Scholar] [CrossRef]
- Dasari, S.; Ganjayi, M.S.; Yellanurkonda, P.; Basha, S.; Meriga, B. Role of glutathione S-transferases in detoxification of a polycyclic aromatic hydrocarbon, methylcholanthrene. Chem. Biol. Interact. 2018, 294, 81–90. [Google Scholar] [CrossRef]
- La Russa, D.; Brunelli, E.; Pellegrino, D. Oxidative imbalance and kidney damage in spontaneously hypertensive rats: Activation of extrinsic apoptotic pathways. Clin. Sci. 2017, 131, 1419–1428. [Google Scholar] [CrossRef]
- Simic, T.; Savic-Radojevic, A.; Pljesa-Ercegovac, M.; Matic, M.; Mimic-Oka, J. Glutathione S-transferases in kidney and urinary bladder tumors. Nat. Rev. Urol. 2009, 6, 281–289. [Google Scholar] [CrossRef]
- Chauhan, A.K.; Mittra, N.; Singh, B.K.; Singh, C. Inhibition of glutathione S-transferase-pi triggers c-jun N-terminal kinase-dependent neuronal death in Zn-induced Parkinsonism. Mol. Cell. Biochem. 2019, 452, 95–104. [Google Scholar] [CrossRef]
- Sun, W.; Su, L.; Sheng, Y.; Shen, Y.; Chen, G. Is there association between Glutathione S Transferases polymorphisms and cataract risk: A meta-analysis? BMC Ophthalmol. 2015, 15, 84. [Google Scholar] [CrossRef][Green Version]
- Qi, R.; Gu, Z.; Zhou, L. The effect of GSTT1, GSTM1 and GSTP1 gene polymorphisms on the susceptibility of age-related cataract in Chinese Han population. Int. J. Clin. Exp. Med. 2015, 8, 19448–19453. [Google Scholar]
- Sireesha, R.; Laxmi, S.G.B.; Mamata, M.; Reddy, P.Y.; Goud, P.U.; Rao, P.V.; Reddy, G.B.; Vishnupriya, S.; Padma, T. Total activity of glutathione-S-transferase (GST) and polymorphisms of GSTM1 and GSTT1 genes conferring risk for the development of age related cataracts. Exp. Eye Res. 2012, 98, 67–74. [Google Scholar] [CrossRef]
- Ganea, E.; Harding, J.J. Glutathione-related enzymes and the eye. Curr. Eye Res. 2006, 31, 1–11. [Google Scholar] [CrossRef]
- Umapathy, A.; Li, B.; Donaldson, P.J.; Lim, J.C. Functional characterisation of glutathione export from the rat lens. Exp. Eye Res. 2018, 166, 151–159. [Google Scholar] [CrossRef]
- Su, S.; Leng, F.; Guan, L.; Zhang, L.; Ge, J.; Wang, C.; Chen, S.; Liu, P. Differential proteomic analyses of cataracts from rat models of type 1 and 2 diabetes. Investig. Ophthalmol. Vis. Sci. 2014, 55, 7848–7861. [Google Scholar] [CrossRef]
- Arnal, E.; Miranda, M.; Almansa, I.; Muriach, M.; Barcia, J.M.; Romero, F.J.; Diaz-Llopis, M.; Bosch-Morell, F. Lutein prevents cataract development and progression in diabetic rats. Graefe’s Arch. Clin. Exp. Ophthalmol. 2009, 247, 115–120. [Google Scholar] [CrossRef]
- Kim, J.; Choung, S. Pinus densiflora bark extract prevents selenite-induced cataract formation in the lens of Sprague Dawley rat pups. Mol. Vis. 2017, 23, 638–648. [Google Scholar]
- Cao, S.; Gao, M.; Wang, N.; Liu, N.; Du, G.; Lu, J. Prevention of selenite-induced cataratogenesis by Ginkgo biloba extract (Egb761) in Wistar rats. Curr. Eye Res. 2015, 40, 1028–1033. [Google Scholar] [CrossRef]
- Ozkol, H.U.; Koyuncu, I.; Tuluce, Y.; Dilsiz, N.; Soral, S.; Ozkol, H. Anthocyanin-rich extract from Hibiscus sabdariffa calyx counteracts UVC-caused impairments in rats. Pharm. Biol. 2015, 53, 1435–1441. [Google Scholar] [CrossRef]
- Nazıroğlu, M.; Güler, M.; Küçükayaz, M.; Övey, İ.S.; Özgül, C. Apple cider vinegar supplementation modulates lipid peroxidation and glutathione peroxidase values in lens of ovariectomized mice. Cell Membr. Free Radic. Res. 2012, 3, 209–214. [Google Scholar]
- Acer, S.; Pekel, G.; Küçükatay, V.; Karabulut, A.; Yağcı, R.; Çetin, E.N.; Akyer, Ş.P.; Şahın, B. Oxidative stress of crystalline lens in rat menopausal model. Arq. Bras. Oftalmol. 2016, 79, 222–225. [Google Scholar] [CrossRef]
- Lu, C.; Zou, Y.; Liu, Y.; Niu, Y. Rosmarinic acid counteracts activation of hepatic stellate cells via inhibiting the ROS-dependent MMP-2 activity: Involvement of Nrf2 antioxidant system. Toxicol. Appl. Pharmacol. 2017, 318, 69–78. [Google Scholar] [CrossRef]
- Tavafi, M.; Ahmadvand, H. Effect of rosmarinic acid on inhibition of gentamicin induced nephrotoxicity in rats. Tissue Cell 2011, 43, 392–397. [Google Scholar] [CrossRef]
- Vanithadevi, B.; Anuradha, C.V. Effect of rosmarinic acid on insulin sensitivity, glyoxalase system and oxidative events in liver of fructose-fed mice. Int. J. Diabetes Metab. 2008, 16, 35–44. [Google Scholar]
- Silambarasan, T.; Manivannan, J.; Raja, B.; Chatterjee, S. Prevention of cardiac dysfunction, kidney fibrosis and lipid metabolic alterations in L-NAME hypertensive rats by sinapic acid—Role of HMG-CoA reductase. Eur. J. Pharmacol. 2016, 777, 113–123. [Google Scholar] [CrossRef]
- Enns, G.M.; Cowan, T.M. Glutathione as a redox biomarker in mitochondrial disease—Implications for therapy. J. Clin. Med. 2017, 6, 50. [Google Scholar] [CrossRef]
- Sengupta, P. The laboratory rat: Relating its age with human’s. Int. J. Prev. Med. 2013, 4, 624–630. [Google Scholar]
- Guo, Z.J.; Niu, H.X.; Hou, F.F.; Zhang, L.; Fu, N.; Nagai, R.; Lu, X.; Chen, B.H.; Shan, Y.X.; Tian, J.W.; et al. Advanced oxidation protein products activate vascular endothelial cells via a RAGE-mediated signaling pathway. Antioxid. Redox Signal. 2008, 10, 1699–1712. [Google Scholar] [CrossRef]
- Wojnar, W.; Kaczmarczyk-Sedlak, I.; Zych, M. Diosmin ameliorates the effects of oxidative stress in lenses of streptozotocin-induced type 1 diabetic rats. Pharmacol. Rep. 2017, 69, 995–1000. [Google Scholar] [CrossRef]
- Wojnar, W.; Zych, M.; Kaczmarczyk-Sedlak, I. Antioxidative effect of flavonoid naringenin in the lenses of type 1 diabetic rats. Biomed. Pharmacother. 2018, 108, 974–984. [Google Scholar] [CrossRef]
- Sedlak, L.; Wojnar, W.; Zych, M.; Mrukwa-Kominek, E.; Kaczmarczyk-Sedlak, I. Effect of resveratrol, a dietary-derived polyphenol, on the oxidative stress and polyol pathway in the lens of rats with streptozotocin-induced diabetes. Nutrients 2018, 10, 1423. [Google Scholar] [CrossRef]
- Xu, J.; Li, D.; Zheng, T.; Lu, Y. β-amyloid expression in age-related cataract lens epithelia and the effect of β-amyloid on oxidative damage in human lens epithelial cells. Mol. Vis. 2017, 23, 1015–1028. [Google Scholar]
- Nagai, N.; Ito, Y. Excessive hydrogen peroxide enhances the attachment of amyloid β1-42 in the lens epithelium of UPL rats, a hereditary model for cataracts. Toxicology 2014, 315, 55–64. [Google Scholar] [CrossRef]
- Özcura, F.; Dündar, S.O.; Çetin, E.D.; Beder, N.; Dündar, M. Effect of estrogen replacement therapy on lens epithelial cell apoptosis in an experimental rat model. Int. Ophthalmol. 2010, 30, 279–284. [Google Scholar] [CrossRef]
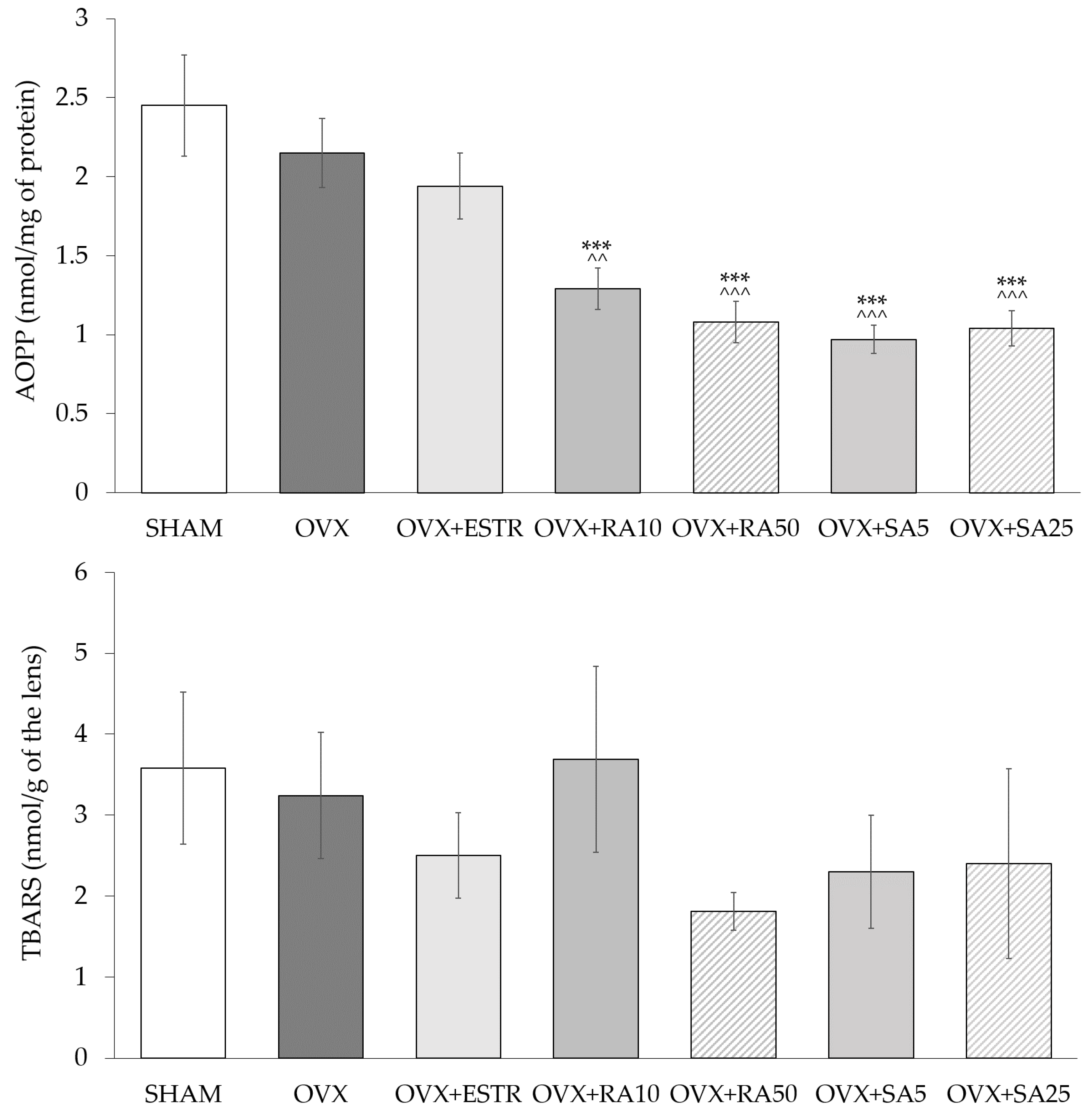
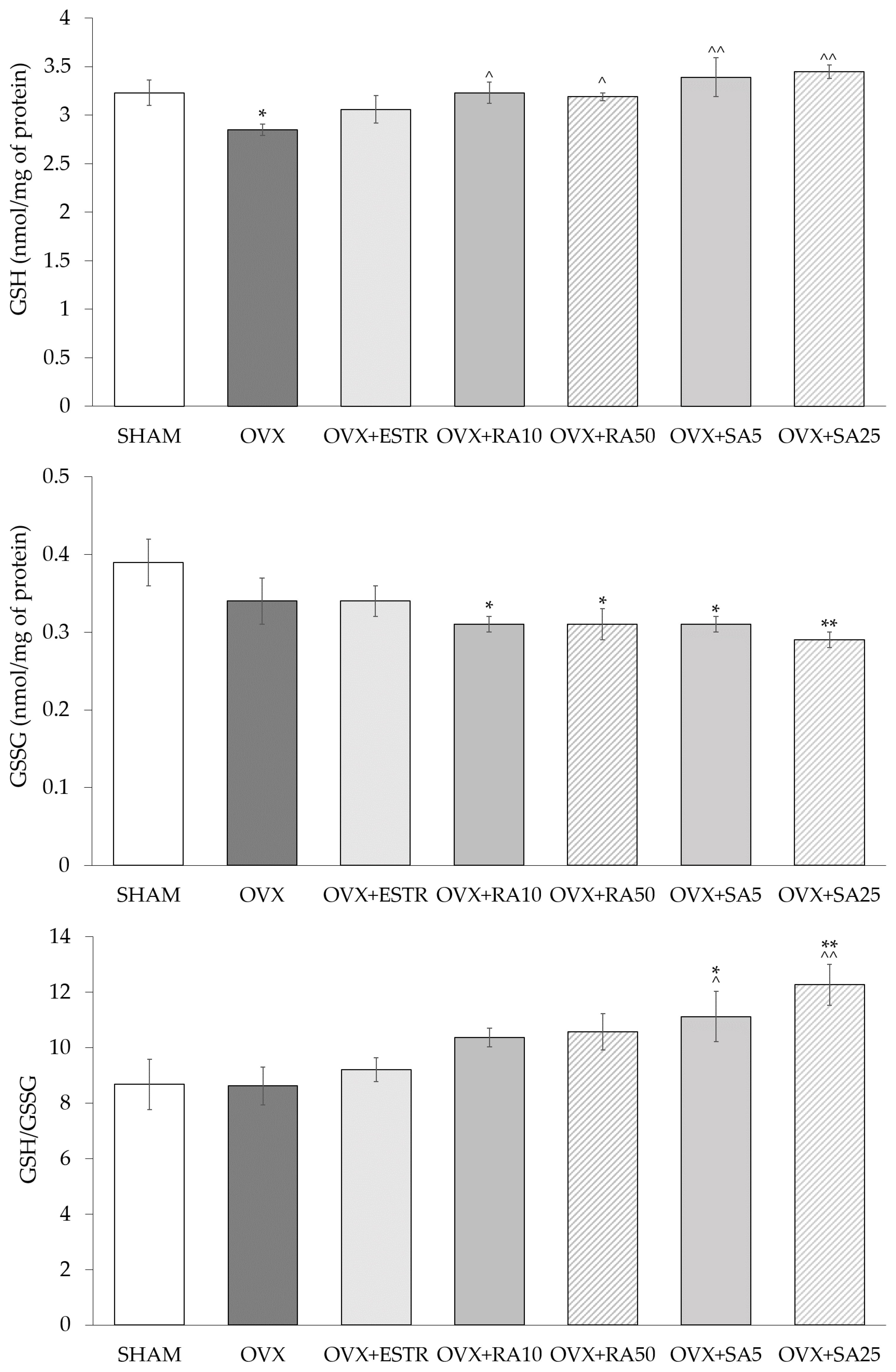
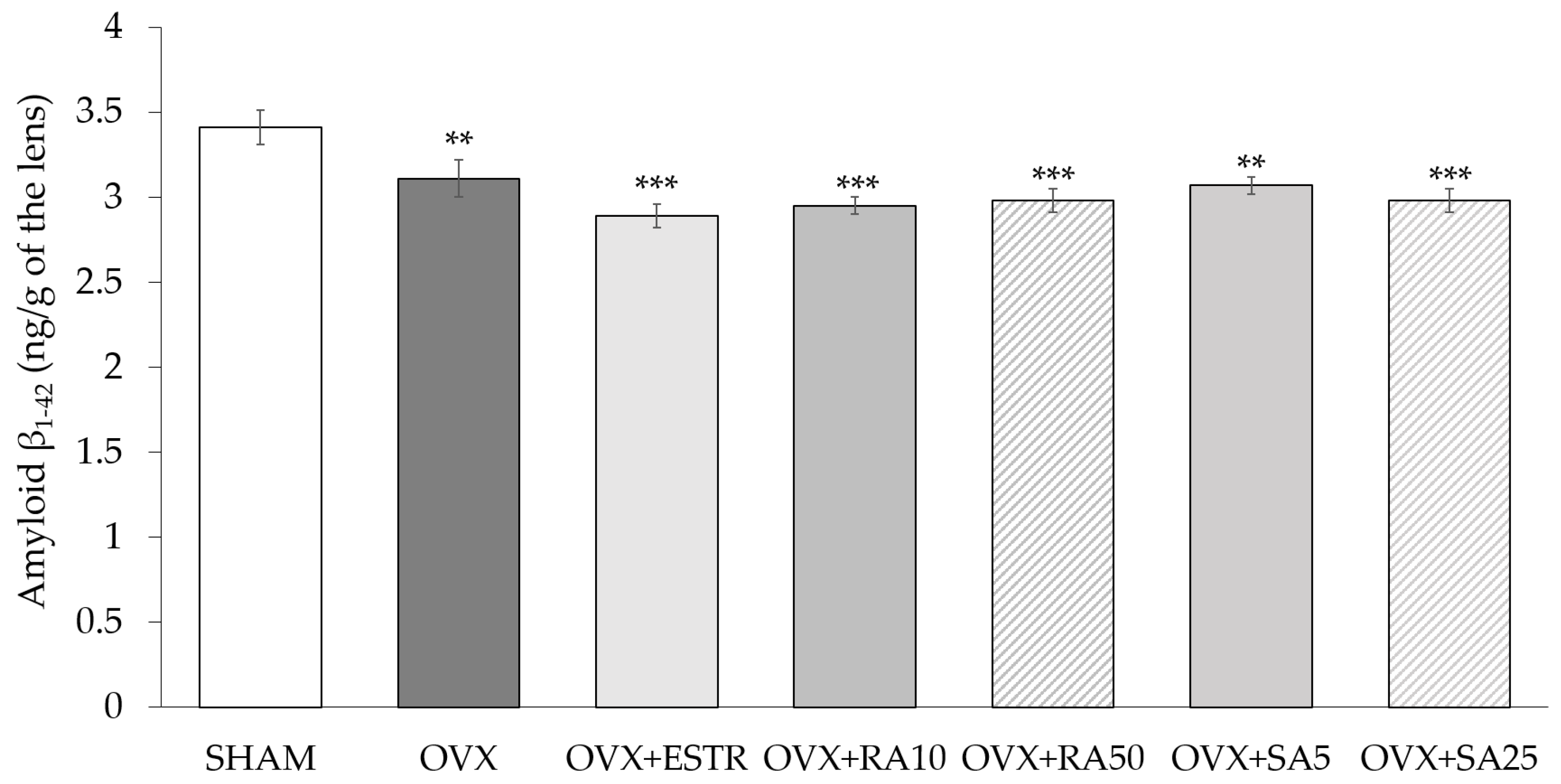
| Parameter/Group | SHAM | OVX | OVX + ESTR | OVX + RA10 | OVX + RA50 | OVX + SA5 | OVX + SA25 |
|---|---|---|---|---|---|---|---|
| Average mass of the lens (g) | 0.059 ± 0.002 | 0.058 ± 0.003 | 0.055 ± 0.002 | 0.055 ± 0.001 | 0.056 ± 0.002 | 0.056 ± 0.002 | 0.055 ± 0.001 |
| Soluble protein (mg/g of the lens) | 280.6 ± 6.8 | 302.5 ± 7.8 | 295.8 ± 5.8 | 290.4 ± 3.2 | 291.7 ± 7.2 | 291.9 ± 5.2 | 286.7 ± 4.2 |
| Parameter/Group | SHAM | OVX | OVX + ESTR | OVX + RA10 | OVX + RA50 | OVX + SA5 | OVX + SA25 |
|---|---|---|---|---|---|---|---|
| SOD (U/mg of protein) | 0.194 ± 0.017 | 0.156 ± 0.003 | 0.170 ± 0.007 | 0.164 ± 0.005 | 0.174 ± 0.006 | 0.171 ± 0.012 | 0.167 ± 0.002 |
| CAT (nmol/min/mg of protein) | 0.085 ± 0.012 | 0.033 ± 0.009 | 0.052 ± 0.014 | 0.075 ± 0.014 | 0.076 ± 0.020 | 0.060 ± 0.018 | 0.063 ± 0.010 |
| Parameter/Group | SHAM | OVX | OVX + ESTR | OVX + RA10 | OVX + RA50 | OVX + SA5 | OVX + SA25 |
|---|---|---|---|---|---|---|---|
| GPx (nmol/min/mg of protein) | 2.31 ± 0.08 | 2.15 ± 0.08 | 2.04 ± 0.07 | 2.26 ± 0.11 | 2.21 ± 0.06 | 2.24 ± 0.08 | 2.28 ± 0.05 |
| GR (nmol/min/mg of protein) | 0.367 ± 0.047 | 0.220 ± 0.036 ** | 0.291 ± 0.034 | 0.357 ± 0.027 ^^ | 0.352 ± 0.026 ^^ | 0.323 ± 0.026 ^ | 0.346 ± 0.021 ^ |
| G6PD (nmol/min/mg of protein) | 1.99 ± 0.10 | 1.30 ± 0.14* | 1.39 ± 0.17 * | 1.52 ± 0.19 | 1.83 ± 0.16 | 1.47 ± 0.13 | 2.00 ± 0.27 ^ |
| GGT (nmol/min/mg of protein) | 0.039 ± 0.005 | 0.024 ± 0.004* | 0.028 ± 0.003 | 0.024 ± 0.004 * | 0.020 ± 0.002 ** | 0.025 ± 0.005 * | 0.031 ± 0.002 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zych, M.; Wojnar, W.; Dudek, S.; Kaczmarczyk-Sedlak, I. Rosmarinic and Sinapic Acids May Increase the Content of Reduced Glutathione in the Lenses of Estrogen-Deficient Rats. Nutrients 2019, 11, 803. https://doi.org/10.3390/nu11040803
Zych M, Wojnar W, Dudek S, Kaczmarczyk-Sedlak I. Rosmarinic and Sinapic Acids May Increase the Content of Reduced Glutathione in the Lenses of Estrogen-Deficient Rats. Nutrients. 2019; 11(4):803. https://doi.org/10.3390/nu11040803
Chicago/Turabian StyleZych, Maria, Weronika Wojnar, Sławomir Dudek, and Ilona Kaczmarczyk-Sedlak. 2019. "Rosmarinic and Sinapic Acids May Increase the Content of Reduced Glutathione in the Lenses of Estrogen-Deficient Rats" Nutrients 11, no. 4: 803. https://doi.org/10.3390/nu11040803
APA StyleZych, M., Wojnar, W., Dudek, S., & Kaczmarczyk-Sedlak, I. (2019). Rosmarinic and Sinapic Acids May Increase the Content of Reduced Glutathione in the Lenses of Estrogen-Deficient Rats. Nutrients, 11(4), 803. https://doi.org/10.3390/nu11040803





