Preliminary Validation of a Food Frequency Questionnaire to Assess Long-Chain Omega-3 Fatty Acid Intake in Eye Care Practice
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Clinical Omega-3 Dietary Survey
2.3. Dietary Questionnaire for Epidemiology Studies
2.4. Dried Blood Spot Testing
2.5. Statistical Analysis
3. Results
3.1. Dietary Assessment of Long-Chain Omega-3 EFA Intake
3.2. Dried Blood Spot Analysis of Long-Chain Omega-3 EFAs Levels
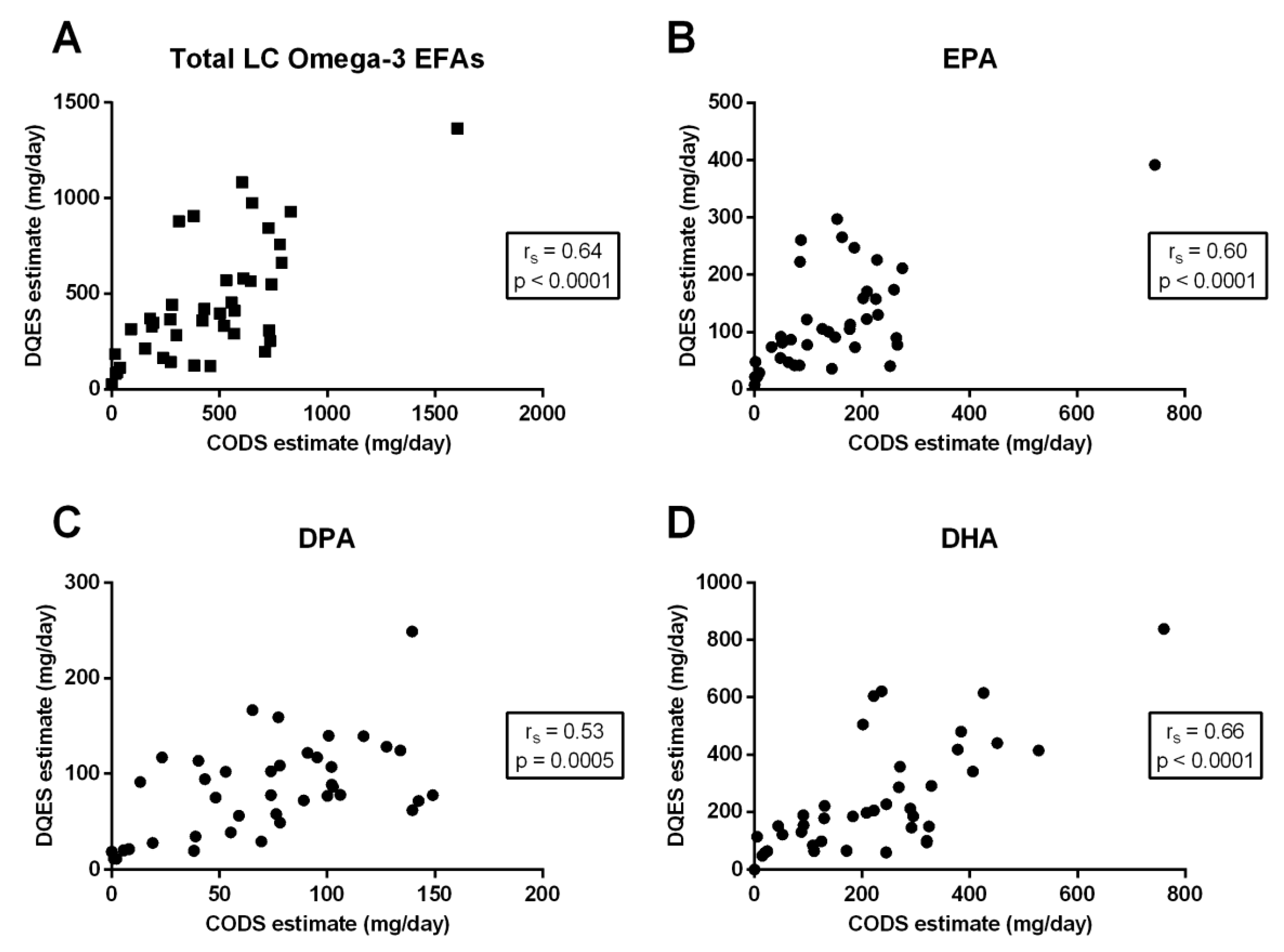
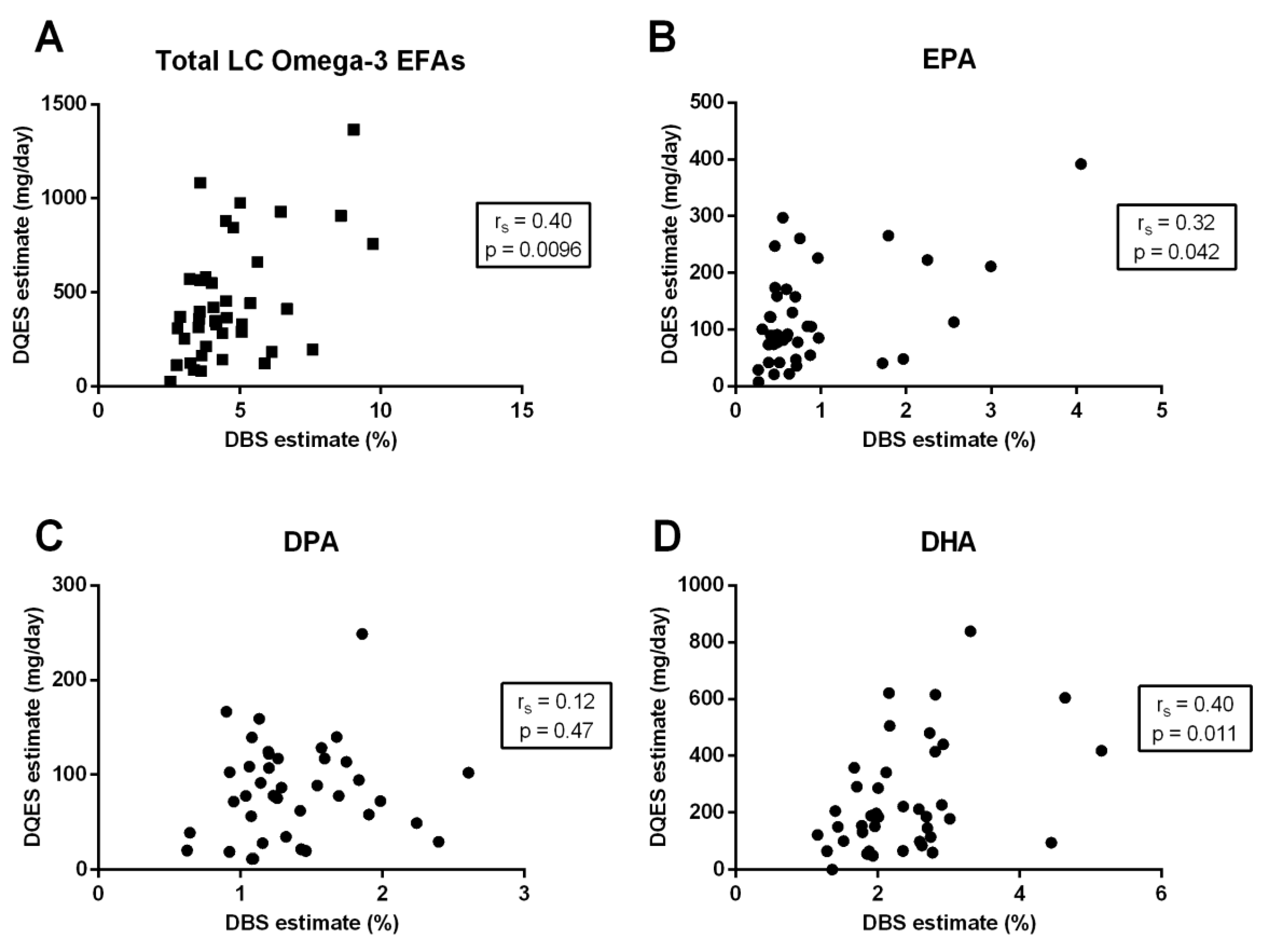
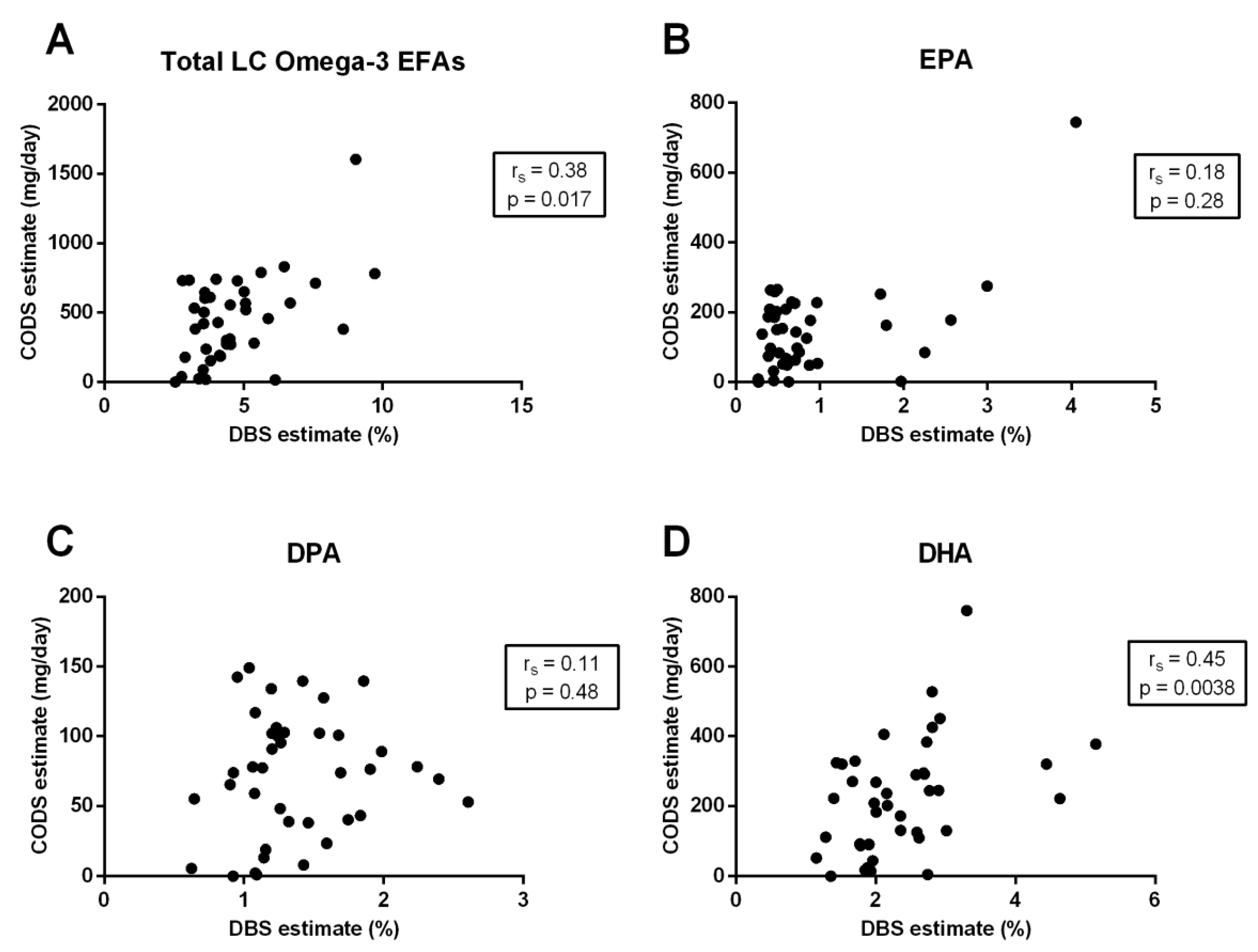
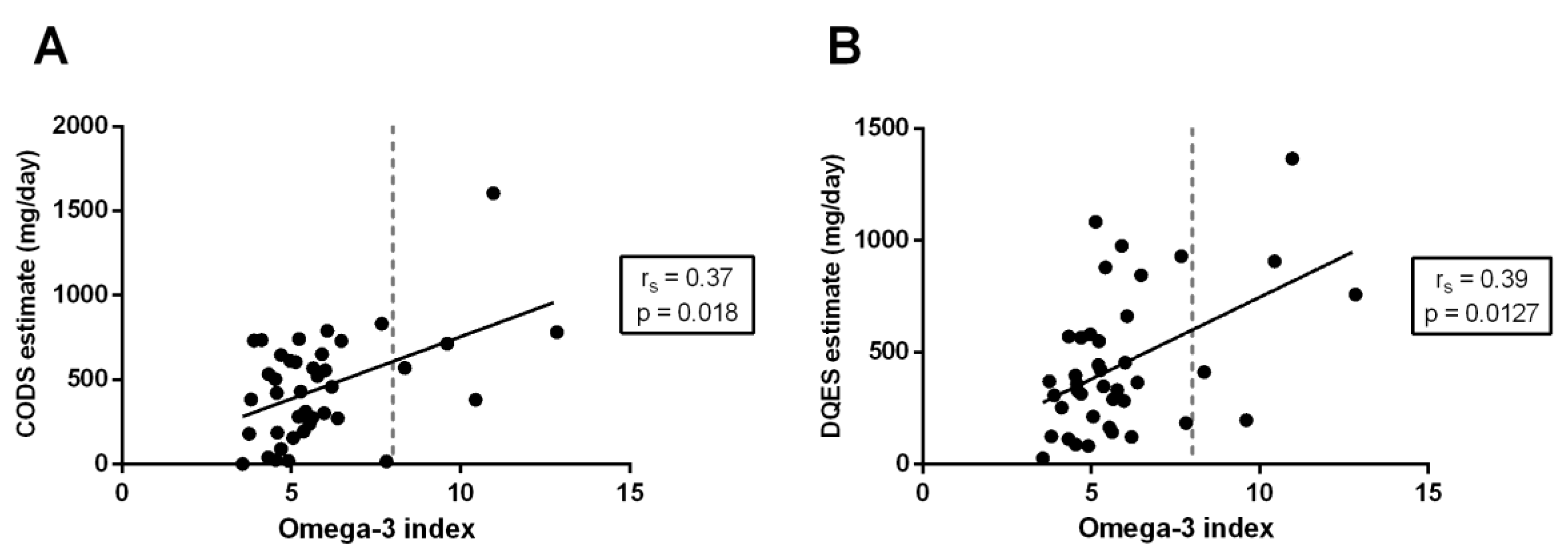
3.3. Correlation between Quantification Methods
3.4. Method of Triads Analysis
3.5. Bland–Altman Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rajaei, E.; Mowla, K.; Ghorbani, A.; Bahadoram, S.; Bahadoram, M.; Dargahi-Malamir, M. The Effect of Omega-3 Fatty Acids in Patients with Active Rheumatoid Arthritis Receiving DMARDs Therapy: Double-Blind Randomized Controlled Trial. Glob. J. Health Sci. 2015, 8, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Omega-3 fatty acids in inflammation and autoimmune diseases. J. Am. Coll. Nutr. 2002, 21, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wu, Y.; Meng, Y.F.; Xing, Q.; Tao, J.J.; Lu, J. Fish Consumption and Age-Related Macular Degeneration Incidence: A Meta-Analysis and Systematic Review of Prospective Cohort Studies. Nutrients 2016, 8, 743. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.E.; Keller, P.R. Nutrition and age-related macular degeneration: Research evidence in practice. Optom. Vis. Sci. 2014, 91, 821–831. [Google Scholar] [CrossRef] [PubMed]
- Miljanovic, B.; Trivedi, K.A.; Dana, M.R.; Gilbard, J.P.; Buring, J.E.; Schaumberg, D.A. Relation between dietary n-3 and n-6 fatty acids and clinically diagnosed dry eye syndrome in women. Am. J. Clin. Nutr. 2005, 82, 887–893. [Google Scholar] [CrossRef]
- Tan, J.S.; Wang, J.J.; Flood, V.; Mitchell, P. Dietary fatty acids and the 10-year incidence of age-related macular degeneration: The Blue Mountains Eye Study. Arch. Ophthalmol. 2009, 127, 656–665. [Google Scholar] [CrossRef]
- Chong, E.W.; Kreis, A.J.; Wong, T.Y.; Simpson, J.A.; Guymer, R.H. Dietary omega-3 fatty acid and fish intake in the primary prevention of age-related macular degeneration: A systematic review and meta-analysis. Arch. Ophthalmol. 2008, 126, 826–833. [Google Scholar] [CrossRef]
- Sangiovanni, J.P.; Agrón, E.; Meleth, A.D.; Reed, G.F.; Sperduto, R.D.; Clemons, T.E.; Chew, E.Y. Age-Related Eye Disease Study Research G. {omega}-3 Long-chain polyunsaturated fatty acid intake and 12-y incidence of neovascular age-related macular degeneration and central geographic atrophy: AREDS report 30, a prospective cohort study from the Age-Related Eye Disease Study. Am. J. Clin. Nutr. 2009, 90, 1601–1607. [Google Scholar] [CrossRef]
- Christen, W.G.; Schaumberg, D.A.; Glynn, R.J.; Buring, J.E. Dietary omega-3 fatty acid and fish intake and incident age-related macular degeneration in women. Arch. Ophthalmol. 2011, 129, 921–929. [Google Scholar] [CrossRef]
- Seddon, J.M.; George, S.; Rosner, B. Cigarette Smoking, Fish Consumption, Omega-3 Fatty Acid Intake, and Associations with Age-Related Macular Degeneration: The US Twin Study of Age-Related Macular Degeneration. Arch. Ophthalmol. 2006, 124, 995–1001. [Google Scholar] [CrossRef]
- Downie, L.E.; Vingrys, A.J. Oral Omega-3 Supplementation Lowers Intraocular Pressure in Normotensive Adults. Transl. Vis. Sci. Technol. 2018, 7, 1. [Google Scholar] [CrossRef]
- Fats: Total Fat & Fatty Acids; Nutrient Reference Values for Australia and New Zealand. Available online: https://www.nrv.gov.au/nutrients/fats-total-fat-fatty-acids (accessed on 20 February 2019).
- National Health and Medical Research Council. Nutrient Reference Values for Australia and New Zealand including Recommended Dietary Intakes; Australian Government Department of Health and Ageing: Canberra, Australia, 2006.
- Nestel, P.; Clifton, P.; Colquhoun, D.; Noakes, M.; Mori, T.A.; Sullivan, D.; Thomas, B. Indications for Omega-3 Long Chain Polyunsaturated Fatty Acid in the Prevention and Treatment of Cardiovascular Disease. Heart Lung Circ. 2015, 24, 769–779. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services; U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; Federal Government: Washington, DC, USA, 2015.
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef]
- Meyer, B.J. Australians are not Meeting the Recommended Intakes for Omega-3 Long Chain Polyunsaturated Fatty Acids: Results of an Analysis from the 2011-2012 National Nutrition and Physical Activity Survey. Nutrients 2016, 8, 111. [Google Scholar] [CrossRef]
- Danaei, G.; Ding, E.L.; Mozaffarian, D.; Taylor, B.; Rehm, J.; Murray, C.J.; Ezzati, M. The preventable causes of death in the United States: Comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med. 2009, 6, e1000058. [Google Scholar] [CrossRef]
- Overby, N.C.; Serra-Majem, L.; Andersen, L.F. Dietary assessment methods on n-3 fatty acid intake: A systematic review. Br. J. Nutr. 2009, 102 (Suppl. 1), S56–S63. [Google Scholar] [CrossRef]
- Baylin, A.; Kabagambe, E.K.; Siles, X.; Campos, H. Adipose tissue biomarkers of fatty acid intake. Am. J. Clin. Nutr. 2002, 76, 750–757. [Google Scholar] [CrossRef]
- Saadatian-Elahi, M.; Slimani, N.; Chajes, V.; Jenab, M.; Goudable, J.; Biessy, C.; Ferrari, P.; Byrnes, G.; Autier, P.; Peeters, P.H.; et al. Plasma phospholipid fatty acid profiles and their association with food intakes: Results from a cross-sectional study within the European Prospective Investigation into Cancer and Nutrition. Am. J. Clin. Nutr. 2009, 89, 331–346. [Google Scholar] [CrossRef]
- Harris, W.S. The omega-3 index as a risk factor for coronary heart disease. Am. J. Clin. Nutr. 2008, 87, 1997S–2002S. [Google Scholar] [CrossRef]
- Wrieden, W.; Peace, H.; Armstrong, J.; Barton, K. A short review of dietary assessment methods used in national and Scottish research studies. In Briefing Paper Prepared for the Working Group on Monitoring Scottish Dietary Targets Workshop; Edinburgh University Press: Edinburgh, UK, 2003. [Google Scholar]
- Institute of Medicine (US) Committee on Dietary Risk Assessment in the WIC Program. 5. Food-Based Assessment of Dietary Intake. In Dietary Risk Assessment in the WIC Program; National Academies Press (US): Washington, DC, USA, 2002. [Google Scholar]
- Swierk, M.; Williams, P.G.; Wilcox, J.; Russell, K.G.; Meyer, B.J. Validation of an Australian electronic food frequency questionnaire to measure polyunsaturated fatty acid intake. Nutrition 2011, 27, 641–646. [Google Scholar] [CrossRef][Green Version]
- Rahmawaty, S.; Charlton, K.; Lyons-Wall, P.; Meyer, B.J. Development and validation of a food frequency questionnaire to assess omega-3 long chain polyunsaturated fatty acid intake in Australian children aged 9–13 years. J. Hum. Nutr. Diet. 2017, 30, 429–438. [Google Scholar] [CrossRef]
- Dahl, L.; Maeland, C.A.; Bjorkkjaer, T. A short food frequency questionnaire to assess intake of seafood and n-3 supplements: Validation with biomarkers. Open Nutr. J. 2011, 10, 127. [Google Scholar] [CrossRef]
- Lucas, M.; Asselin, G.; Mérette, C.; Poulin, M.-J.; Dodin, S. Validation of an FFQ for evaluation of EPA and DHA intake. Public Health Nutr. 2009, 12, 1783–1790. [Google Scholar] [CrossRef]
- Sublette, M.E.; Segal-Isaacson, C.J.; Cooper, T.B.; Fekri, S.; Vanegas, N.; Galfalvy, H.C.; Oquendo, M.A.; Mann, J.J. Validation of a food frequency questionnaire to assess intake of n-3 polyunsaturated fatty acids in subjects with and without major depressive disorder. J. Am. Diet. Assoc. 2011, 111, 117–123. [Google Scholar] [CrossRef]
- Dickinson, K.M.; Delaney, C.L.; Allan, R.; Spark, I.; Miller, M.D. Validation of a Brief Dietary Assessment Tool for Estimating Dietary EPA and DHA Intake in Australian Adults at Risk of Cardiovascular Disease. J. Am. Coll. Nutr. 2015, 34, 333–339. [Google Scholar] [CrossRef]
- Stark, K.D.; Van Elswyk, M.E.; Higgins, M.R.; Weatherford, C.A.; Salem, N., Jr. Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog. Lipid Res. 2016, 63, 132–152. [Google Scholar] [CrossRef]
- Food Standards Australia New Zealand. AUSNUT 2011–13—Australian Food Composition Database; FSANZ: Canberra, Australia, 2014. Available online: www.foodstandards.gov.au (accessed on 4 April 2018).
- Giles, G.G.; Ireland, P.D. Dietary Questionnaire for Epidemiological Studies (Version 3.2); Cancer Council Victoria: Melbourne, Australia, 1996. [Google Scholar]
- Bassett, J.K.; English, D.R.; Fahey, M.T.; Forbes, A.B.; Gurrin, L.C.; Simpson, J.A.; Brinkman, M.T.; Giles, G.G.; Hodge, A.M. Validity and calibration of the FFQ used in the Melbourne Collaborative Cohort Study. Public Health Nutr. 2016, 19, 2357–2368. [Google Scholar] [CrossRef]
- Hopkins, A.H.; Hodge, A.M.; Fletcher, A.S.; Bruinsma, F.J.; Bassett, J.K.; Popowski, L.V.; Brinkman, M.T.; English, D.R.; Giles, G.G.; Jayasekara, H.; et al. Cohort Profile: The Melbourne Collaborative Cohort Study (Health 2020). Int. J. Epidemiol. 2017, 46, 1757–1757i. [Google Scholar] [CrossRef]
- Food Standards Australia New Zealand. AUSNUT 2007—Australian Food Composition Tables; FSANZ: Canberra, Australia, 2008.
- Food Standards Australia New Zealand. NUTTAB 2010—Australian Food Composition Tables; FSANZ: Canberra, Australia, 2011.
- Liu, G.; Muhlhausler, B.S.; Gibson, R.A. A method for long term stabilisation of long chain polyunsaturated fatty acids in dried blood spots and its clinical application. Prostaglandins Leukot. Essent. Fatty Acids 2014, 91, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, B.L.; Williams, P.G.; Meyer, B.J. Biomarker validation of a long-chain omega-3 polyunsaturated fatty acid food frequency questionnaire. Lipids 2006, 41, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, D.C.G.; Segheto, W.; de Lima, M.F.C.; Pessoa, M.C.; Pelúzio, M.C.G.; Marchioni, D.M.L.; Cunha, D.B.; Longo, G.Z. Using the method of triads in the validation of a food frequency questionnaire to assess the consumption of fatty acids in adults. J. Hum. Nutr. Diet. 2018, 31, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Ocke, M.C.; Kaaks, R.J. Biochemical markers as additional measurements in dietary validity studies: Application of the method of triads with examples from the European Prospective Investigation into Cancer and Nutrition. Am. J. Clin. Nutr. 1997, 65, 1240s–1245s. [Google Scholar] [CrossRef] [PubMed]
- Efron, B.; Tibshirani, R. Bootstrap Methods for Standard Errors, Confidence Intervals, and Other Measures of Statistical Accuracy. Stat. Sci. 1986, 1, 54–75. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Measuring agreement in method comparison studies. Stat. Methods Med. Res. 1999, 8, 135–160. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.E.; Keller, P.R. The self-reported clinical practice behaviors of Australian optometrists as related to smoking, diet and nutritional supplementation. PLoS ONE 2015, 10, e0124533. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.E.; Douglass, A.; Guest, D.; Keller, P.R. What do patients think about the role of optometrists in providing advice about smoking and nutrition? Ophthalmic Physiol. Opt. 2017, 37, 202–211. [Google Scholar] [CrossRef]
- Soltan, S.S.; Gibson, R.A. Levels of Omega 3 fatty acids in Australian seafood. Asia Pac. J. Clin. Nutr. 2008, 17, 385–390. [Google Scholar]
- Meyer, B.J.; Swierk, M.; Russell, K.G. Assessing long-chain omega-3 polyunsaturated fatty acids: A tailored food-frequency questionnaire is better. Nutrition 2013, 29, 491–496. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Serra-Majem, L.; Frost Andersen, L.; Henrique-Sanchez, P.; Doreste-Alonso, J.; Sanchez-Villegas, A.; Ortiz-Andrelluchi, A.; Negri, E.; La Vecchia, C. Evaluating the quality of dietary intake validation studies. Br. J. Nutr. 2009, 102 (Suppl. 1), S3–S9. [Google Scholar] [CrossRef]
- Cade, J.; Thompson, R.; Burley, V.; Warm, D. Development, validation and utilisation of food-frequency questionnaires—A review. Public Health Nutr. 2002, 5, 567–587. [Google Scholar] [CrossRef]
- Thompson, F.E.; Byers, T. Dietary assessment resource manual. J. Nutr. 1994, 124, 2245s–2317s. [Google Scholar] [CrossRef]
- Jones, L.; Downie, L.E.; Korb, D.; Benitez-Del-Castillo, J.M.; Dana, R.; Deng, S.X.; Dong, P.N.; Geerling, G.; Hida, R.Y.; Liu, Y.; et al. TFOS DEWS II Management and Therapy Report. Ocul. Surf. 2017, 15, 575–628. [Google Scholar] [CrossRef]
- Deinema, L.A.; Vingrys, A.J.; Wong, C.Y.; Jackson, D.C.; Chinnery, H.R.; Downie, L.E. A Randomized, Double-Masked, Placebo-Controlled Clinical Trial of Two Forms of Omega-3 Supplements for Treating Dry Eye Disease. Ophthalmology 2017, 124, 43–52. [Google Scholar] [CrossRef]
- Asbell, P.A.; Maguire, M.G.; Pistilli, M.; Ying, G.S.; Szczotka-Flynn, L.B.; Hardten, D.R.; Lin, M.C.; Shtein, R.M. n-3 Fatty Acid Supplementation for the Treatment of Dry Eye Disease. N. Engl. J. Med. 2018, 378, 1681–1690. [Google Scholar] [CrossRef]
- Chinnery, H.R.; Naranjo Golborne, C.; Downie, L.E. Omega-3 supplementation is neuroprotective to corneal nerves in dry eye disease: A pilot study. Ophthalmic Physiol. Opt. 2017, 37, 473–481. [Google Scholar] [CrossRef]
- Downie, L.E.; Ng, S.M.; Lindsley, K.; Akpek, E.K. Omega-3 and omega-6 polyunsaturated fatty acids for dry eye disease. Cochrane Database Syst. Rev. 2019. In Press. [Google Scholar]
- Rice, H.B.; Bernasconi, A.; Maki, K.C.; Harris, W.S.; von Schacky, C.; Calder, P.C. Conducting omega-3 clinical trials with cardiovascular outcomes: Proceedings of a workshop held at ISSFAL 2014. Prostaglandins Leukot. Essent. Fatty Acids 2016, 107, 30–42. [Google Scholar] [CrossRef]
- Silva, V.; Singer, P. Membrane fatty acid composition of different target populations: Importance of baseline on supplementation. Clin. Nutr. Exp. 2015, 1, 1–9. [Google Scholar] [CrossRef][Green Version]
- Lawrenson, J.G.; Evans, J.R. Omega 3 fatty acids for preventing or slowing the progression of age-related macular degeneration. Cochrane Database Syst. Rev. 2015, 4, CD010015. [Google Scholar] [CrossRef]
- Harris, W.S.; Del Gobbo, L.; Tintle, N.L. The Omega-3 Index and relative risk for coronary heart disease mortality: Estimation from 10 cohort studies. Atherosclerosis 2017, 262, 51–54. [Google Scholar] [CrossRef]
- Gerster, H. Can adults adequately convert alpha-linolenic acid (18:3n-3) to eicosapentaenoic acid (20:5n-3) and docosahexaenoic acid (22:6n-3)? International journal for vitamin and nutrition research. Int. J. Vitam. Nutr. Res. 1998, 68, 159–173. [Google Scholar]
- Burdge, G.C.; Wootton, S.A. Conversion of alpha-linolenic acid to eicosapentaenoic, docosapentaenoic and docosahexaenoic acids in young women. Br. J. Nutr. 2002, 88, 411–420. [Google Scholar] [CrossRef]
- Brenna, J.T. Efficiency of conversion of alpha-linolenic acid to long chain n-3 fatty acids in man. Curr. Opin. Clin. Nutr. 2002, 5, 127–132. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Kaur, G.; Cameron-Smith, D.; Garg, M.; Sinclair, A.J. Docosapentaenoic acid (22:5n-3): A review of its biological effects. Prog. Lipid Res. 2011, 50, 28–34. [Google Scholar] [CrossRef]
- Miller, E.; Kaur, G.; Larsen, A.; Loh, S.P.; Linderborg, K.; Weisinger, H.S.; Turchini, G.M.; Cameron-Smith, D.; Sinclair, A.J. A short-term n-3 DPA supplementation study in humans. Eur. J. Nutr. 2013, 52, 895–904. [Google Scholar] [CrossRef]

| CODS (mg/day) | DQES v3.2 (mg/day) | p-value | |||||
|---|---|---|---|---|---|---|---|
| Median | IQR | Median | IQR | ||||
| p25 | p75 | p25 | p75 | ||||
| Total LC omega-3 EFAs | 443.7 | 204.1 | 650.0 | 362.6 | 200.7 | 577.9 | 0.83 |
| EPA (20:5-n3) | 140.5 | 55.9 | 209.2 | 91.4 | 49.9 | 168.0 | 0.37 |
| DPA (22:5-n3) | 75.2 | 39.3 | 102.2 | 77.8 | 41.3 | 116.2 | 0.41 |
| DHA (22:6-n3) | 221.7 | 95.8 | 320.2 | 184.9 | 98.2 | 353.7 | 0.98 |
| Median | IQR | ||
|---|---|---|---|
| p25 | p75 | ||
| Total LC omega-3 EFAs (%) | 4.15 | 3.58 | 5.30 |
| EPA (20:5-n3) (%) | 0.60 | 0.45 | 0.88 |
| DPA (22:5-n3) (%) | 1.25 | 1.08 | 1.66 |
| DHA (22:6-n3) (%) | 2.16 | 1.80 | 2.77 |
| Omega-3 index | 5.32 | 4.58 | 6.17 |
| CODS vs. DQES | DBS vs. DQES | DBS vs. CODS | ||||
|---|---|---|---|---|---|---|
| rs | p-value | rs | p-value | rs | p-value | |
| Total LC omega-3 EFAs | 0.64 | <0.0001 | 0.40 | 0.0096 | 0.38 | 0.017 |
| EPA (20:5-n3) | 0.60 | <0.0001 | 0.32 | 0.042 | 0.18 | 0.28 |
| DPA (22:5-n3) | 0.53 | 0.0005 | 0.12 | 0.47 | 0.11 | 0.48 |
| DHA (22:6-n3) | 0.66 | <0.0001 | 0.40 | 0.011 | 0.45 | 0.0038 |
| CODS Validity Coefficient vs. T [ρQT] (95% CI) | DQES Validity Coefficient vs. T [ρRT] (95% CI) | DBS Validity Coefficient vs. T [ρBT] (95% CI) | |
|---|---|---|---|
| Total LC omega-3 EFAs | 0.77 (0.31–0.98) | 0.83 (0.39–0.98) | 0.49 (0.12–0.73) |
| EPA (20:5n-3) | 0.57 (0.21–0.93) | 1.00 * (0.39–1.00) | 0.31 (0.07–0.65) |
| DPA (22:5n-3) | 0.72 (0.14–0.97) | 0.73 (0.16–0.97) | 0.16 (0.03–0.50) |
| DHA (22:6n-3) | 0.86 (0.54–0.99) | 0.77 (0.39–0.97) | 0.52 (0.21–0.74) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, A.C.; Downie, L.E. Preliminary Validation of a Food Frequency Questionnaire to Assess Long-Chain Omega-3 Fatty Acid Intake in Eye Care Practice. Nutrients 2019, 11, 817. https://doi.org/10.3390/nu11040817
Zhang AC, Downie LE. Preliminary Validation of a Food Frequency Questionnaire to Assess Long-Chain Omega-3 Fatty Acid Intake in Eye Care Practice. Nutrients. 2019; 11(4):817. https://doi.org/10.3390/nu11040817
Chicago/Turabian StyleZhang, Alexis Ceecee, and Laura E. Downie. 2019. "Preliminary Validation of a Food Frequency Questionnaire to Assess Long-Chain Omega-3 Fatty Acid Intake in Eye Care Practice" Nutrients 11, no. 4: 817. https://doi.org/10.3390/nu11040817
APA StyleZhang, A. C., & Downie, L. E. (2019). Preliminary Validation of a Food Frequency Questionnaire to Assess Long-Chain Omega-3 Fatty Acid Intake in Eye Care Practice. Nutrients, 11(4), 817. https://doi.org/10.3390/nu11040817





