Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis
Abstract
1. Introduction
2. Methods
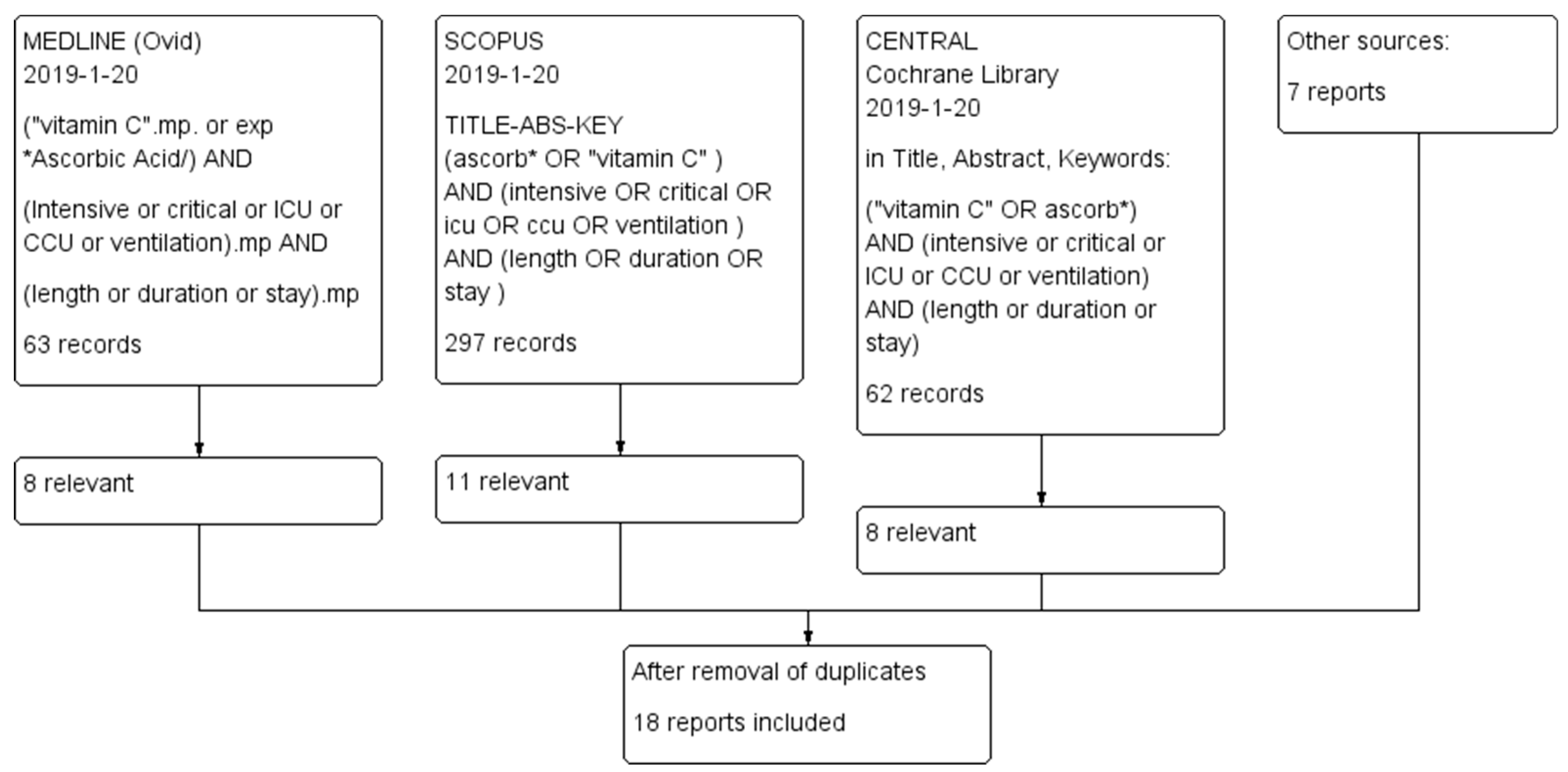
2.1. Selection Criteria for Trials and the Searches
2.2. Outcomes
2.3. Selection of Studies and Data Extraction
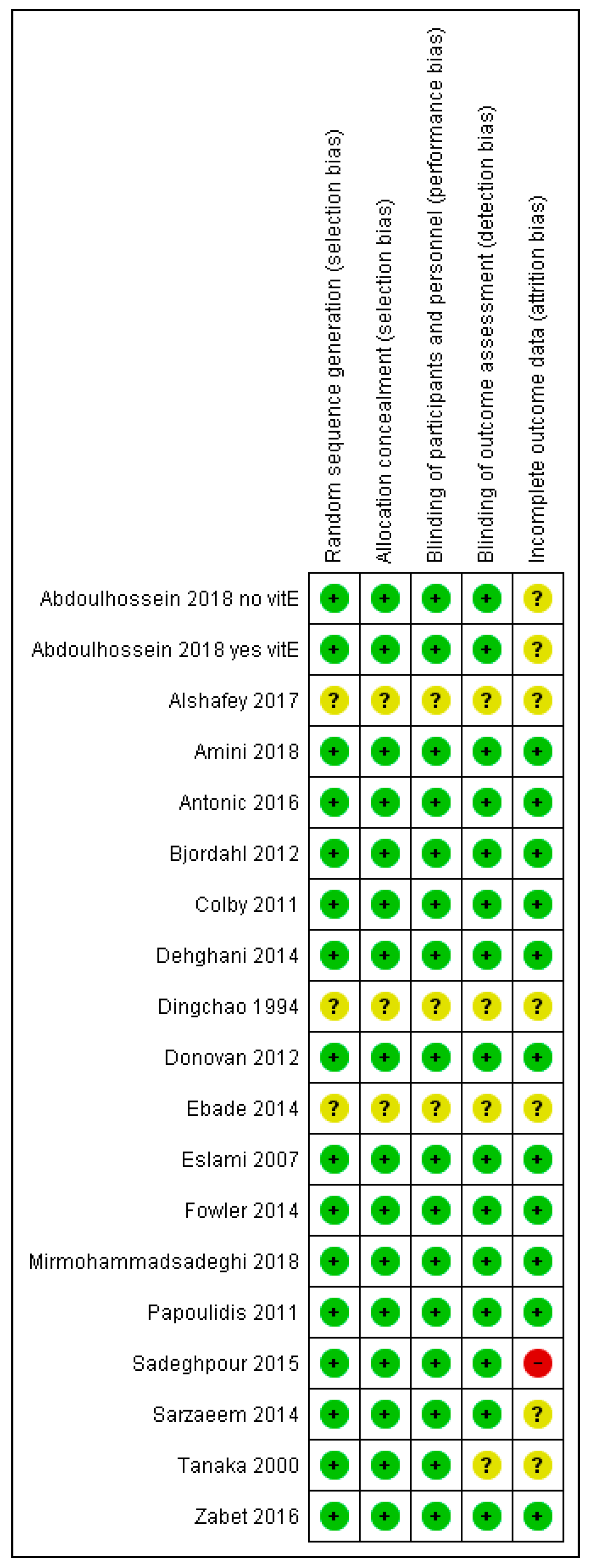
2.4. Quality Assessment of the Trials
2.5. Statistical Methods
3. Results
3.1. Description of the Trials
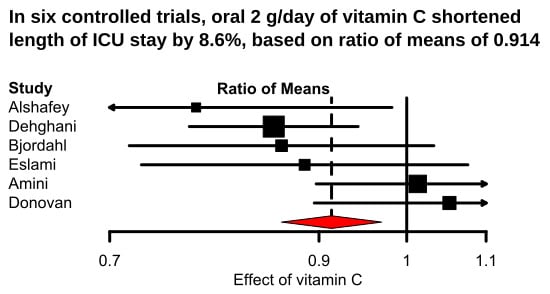
3.2. Effect of Vitamin C Administration on the Length of ICU Stay
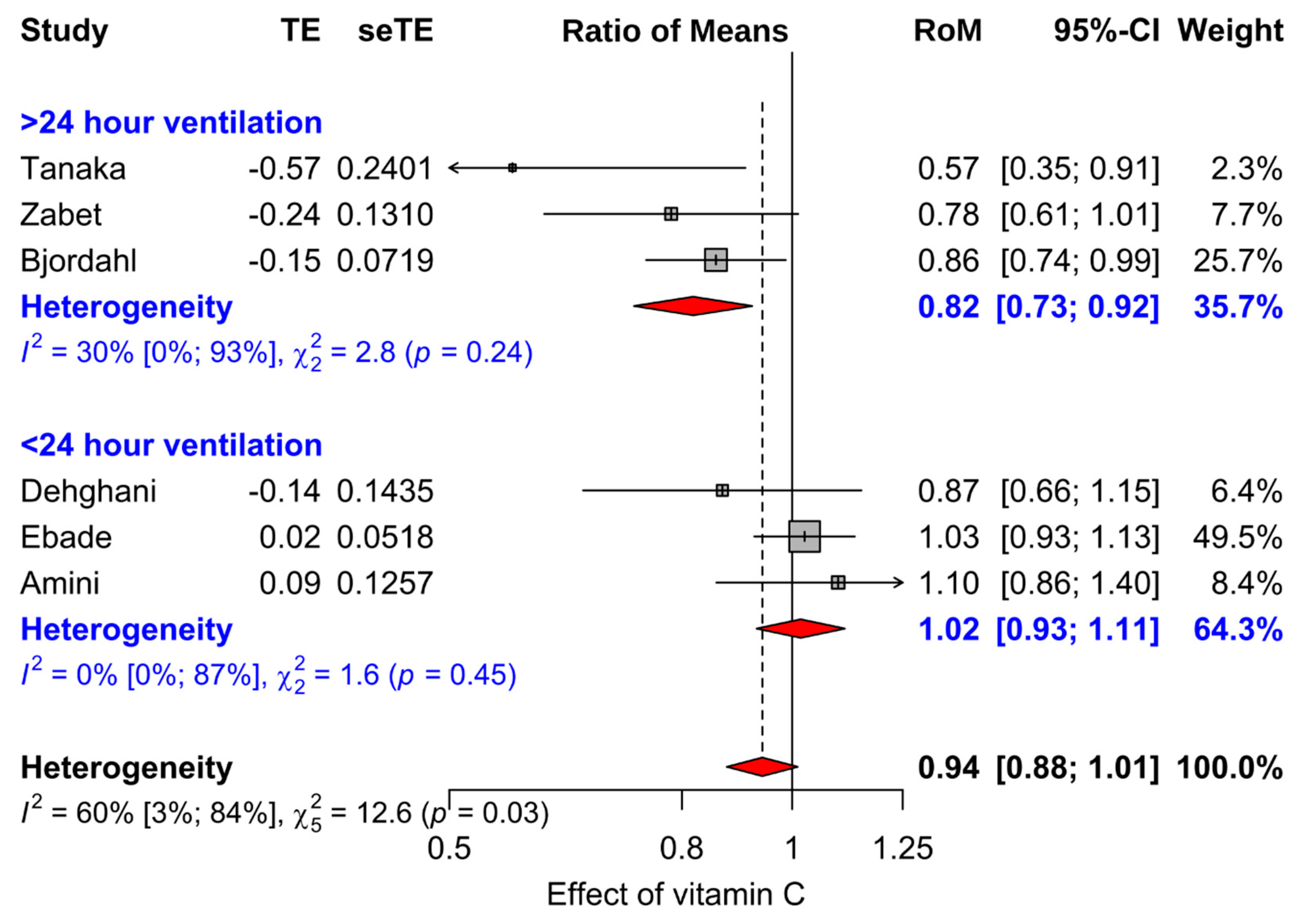
3.3. Effect of Vitamin C on Mechanical Ventilation
3.4. Other Effects in the Included Trials
4. Discussion
4.1. The Complex Biochemistry of Vitamin C
4.2. Early Evidence of Vitamin C Deficiency in Hospital Patients
4.3. Vitamin C and Pneumonia and the Common Cold
4.4. Vitamin C and the Cardiovascular System
4.5. Other Possible Effects of Vitamin C Relevant in the ICU Context
4.6. Pharmacokinetics of Vitamin C
4.7. Current Findings: Vitamin C and Length of ICU Stay and Duration of Mechanical Ventilation
4.8. Other Relevant Studies
4.9. Safety of Vitamin C
4.10. Preconceptions about Vitamin C: Why Is Vitamin C not Used Despite the Evidence?
5. Conclusions: The Way Forward
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hemilä, H.; Chalker, E. References to “Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis” with Web-Links. Available online: http://www.mv.helsinki.fi/home/hemila/N2019 (accessed on 9 March 2019).
- Hess, A.F. Scurvy: Past and Present; Lippincott: Philadelphia, PA, USA, 1920; Available online: http://chla.library.cornell.edu (accessed on 22 January 2019).
- Carpenter, K.J. The History of Scurvy and Vitamin C; Cambridge University Press: Cambridge, UK, 1986. [Google Scholar]
- Bown, S.R. Scurvy: How a Surgeon, a Mariner and a Gentleman Solved the Greatest Medical Mystery of the Age of Sail; Summersdale: Chichester, West Sussex, UK, 2003. [Google Scholar]
- Harvie, D.I. Limeys: The True Story of One Man’s War against Ignorance, the Establishment and the Deadly Scurvy; Sutton: Stroud, Gloucestershire, UK, 2002. [Google Scholar]
- Lamb, J. Scurvy: The Disease of Discovery; Princeton University Press: Princeton, NJ, USA, 2016. [Google Scholar]
- Wilson, L.G. The clinical definition of scurvy and the discovery of vitamin C. J. Hist. Med. Allied Sci. 1975, 30, 40–60. [Google Scholar] [CrossRef]
- Dickman, S.R. The search for the specific factor in scurvy. Perspect. Biol. Med. 1981, 24, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.P. Sailors, scurvy and science. J. R. Soc. Med. 1997, 90, 50–54. [Google Scholar] [CrossRef]
- Hirschmann, J.V.; Raugi, G.J. Adult scurvy. J. Am. Acad. Dermatol. 1999, 41, 895–906. [Google Scholar] [CrossRef]
- Carpenter, K.J. A short history of nutritional science. Part 2 (1885–1912). J. Nutr. 2003, 133, 975–984. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, K.J. A short history of nutritional science. Part 3 (1912–1944). J. Nutr. 2003, 133, 3023–3032. [Google Scholar] [CrossRef]
- Pimentel, L. Scurvy: Historical review and current diagnostic approach. Am. J. Emerg. Med. 2003, 21, 328–332. [Google Scholar] [CrossRef]
- Sutton, G. Putrid gums and ‘dead men’s cloaths’: James Lind aboard the Salisbury. J. R. Soc. Med. 2003, 96, 605–608. [Google Scholar] [CrossRef]
- Magiorkinis, E.; Beloukas, A.; Diamantis, A. Scurvy: Past, present and future. Eur. J. Intern Med. 2011, 22, 147–152. [Google Scholar] [CrossRef]
- Hemilä, H. Do Vitamins C and E Affect Respiratory Infections? Ph.D. Thesis, University of Helsinki, Helsinki, Finland, 2006; pp. 1–51, 101–104. Available online: https://hdl.handle.net/10138/20335 (accessed on 22 January 2019).
- Szent-Györgyi, A. Lost in the twentieth century. Annu. Rev. Biochem. 1963, 32, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ashor, A.W.; Lara, J.; Mathers, J.C.; Siervo, M. Effect of vitamin C on endothelial function in health and disease: A systematic review and meta-analysis of randomised controlled trials. Atherosclerosis 2012, 235, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Ashor, A.W.; Siervo, M.; Lara, J.; Oggioni, C.; Afshar, S.; Mathers, J.C. Effect of vitamin C and vitamin E supplementation on endothelial function: A systematic review and meta-analysis of randomised controlled trials. Br. J. Nutr. 2015, 113, 1182–1194. [Google Scholar] [CrossRef] [PubMed]
- Thosar, S.S.; Bielko, S.L.; Wiggins, C.C.; Klaunig, J.E.; Mather, K.J.; Wallace, J.P. Antioxidant vitamin C prevents decline in endothelial function during sitting. Med. Sci. Monit. 2015, 21, 1015–1021. [Google Scholar] [CrossRef]
- Juraschek, S.P.; Guallar, E.; Appel, L.J.; Miller, E.R. Effects of vitamin C supplementation on blood pressure: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 95, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Oktar, G.L.; Sinci, V.; Kalaycioglu, S.; Soncul, H.; Gökgöz, L.; Halit, V.; Ersöz, A. Biochemical and hemodynamic effects of ascorbic acid and alpha-tocopherol in coronary artery surgery. Scand. J. Clin. Lab. Investig. 2001, 61, 621–629. [Google Scholar] [CrossRef]
- Basili, S.; Tanzilli, G.; Mangieri, E.; Raparelli, V.; Di Santo, S.; Pignatelli, P.; Violi, F. Intravenous ascorbic acid infusion improves myocardial perfusion grade during elective percutaneous coronary intervention: Relationship with oxidative stress markers. JACC Cardiovasc. Interv. 2010, 3, 221–229. [Google Scholar] [CrossRef]
- Pignatelli, P.; Tanzilli, G.; Carnevale, R.; Di Santo, S.; Loffredo, L.; Celestini, A.; Proietti, M.; Tovaglia, P.; Mangieri, E.; Basili, S.; et al. Ascorbic acid infusion blunts CD40L upregulation in patients undergoing coronary stent. Cardiovasc. Ther. 2011, 29, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Valls, N.; Gormaz, J.G.; Aguayo, R.; Gonzalez, J.; Brito, R.; Hasson, D.; Libuy, M.; Ramos, C.; Carrasco, R.; Prieto, J.C.; Dussaillant, G.; et al. Amelioration of persistent left ventricular function impairment through increased plasma ascorbate levels following myocardial infarction. Redox Rep. 2016, 21, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Ramos, C.; Brito, R.; Gonzalez-Montero, J.; Valls, N.; Gormaz, J.G.; Prieto, J.C.; Aguayo, R.; Puentes, A.; Noriega, V.; Pereira, G.; et al. Effects of a novel ascorbate-based protocol on infarct size and ventricle function in acute myocardial infarction patients undergoing percutaneous coronary angioplasty. Arch. Med. Sci. 2017, 13, 558–567. [Google Scholar] [CrossRef]
- Polymeropoulos, E.; Bagos, P.; Papadimitriou, M.; Rizos, I.; Patsouris, E.; Τoumpoulis, I. Vitamin C for the prevention of postoperative atrial fibrillation after cardiac surgery: A meta-analysis. Adv. Pharm. Bull. 2016, 6, 243–250. [Google Scholar] [CrossRef]
- Hemilä, H.; Suonsyrjä, T. Vitamin C for preventing atrial fibrillation in high risk patients: A systematic review and meta-analysis. BMC Cardiovasc. Disord. 2017, 17, 49. [Google Scholar] [CrossRef]
- Shi, R.; Li, Z.H.; Chen, D.; Wu, Q.C.; Zhou, X.L.; Tie, H.T. Sole and combined vitamin C supplementation can prevent postoperative atrial fibrillation after cardiac surgery: A systematic review and meta-analysis of randomized controlled trials. Clin. Cardiol. 2018, 41, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Sadat, U.; Usman, A.; Gillard, J.H.; Boyle, J.R. Does ascorbic acid protect against contrast-induced acute kidney injury in patients undergoing coronary angiography: A systematic review with meta-analysis of randomized, controlled trials. J. Am. Coll. Cardiol. 2013, 62, 2167–2175. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zheng, X.; Liang, B.; Gao, J.; Gu, Z. Vitamins for prevention of contrast-induced acute kidney injury: A systematic review and trial sequential analysis. Am. J. Cardiovasc. Drugs 2018, 18, 373–386. [Google Scholar] [CrossRef] [PubMed]
- Ashor, A.W.; Werner, A.D.; Lara, J.; Willis, N.D.; Mathers, J.C.; Siervo, M. Effects of vitamin C supplementation on glycaemic control: A systematic review and meta-analysis of randomised controlled trials. Eur. J. Clin. Nutr. 2017, 71, 1371–1380. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Vitamin C may alleviate exercise-induced bronchoconstriction: A meta-analysis. BMJ Open 2013, 3, e002416. [Google Scholar] [CrossRef]
- Hemilä, H. The effect of vitamin C on bronchoconstriction and respiratory symptoms caused by exercise: A review and statistical analysis. Allergy Asthma Clin. Immunol. 2014, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Vitamin C and common cold-induced asthma: A systematic review and statistical analysis. Allergy Asthma Clin. Immunol. 2013, 9, 46. [Google Scholar] [CrossRef]
- Pauling, L. The significance of the evidence about ascorbic acid and the common cold. Proc. Natl. Acad. Sci. USA 1971, 68, 2678–2681. [Google Scholar] [CrossRef]
- Pauling, L. Ascorbic acid and the common cold. Am. J. Clin. Nutr. 1971, 24, 1294–1299. [Google Scholar] [CrossRef]
- Hemilä, H.; Herman, Z.S. Vitamin C and the common cold: A retrospective analysis of Chalmers’ review. J. Am. Coll. Nutr. 1995, 4, 116–123. Available online: https://helda.helsinki.fi/handle/10138/42358 (accessed on 9 March 2019). [CrossRef]
- Hemilä, H. Vitamin C supplementation and common cold symptoms: Problems with inaccurate reviews. Nutrition 1996, 12, 804–809. Available online: https://helda.helsinki.fi/handle/10138/225877 (accessed on 9 March 2019). [CrossRef]
- Hemilä, H. Vitamin C, the placebo effect, and the common cold: A case study of how preconceptions influence the analysis of results [comments in 1996, 49, 1985–1987]. J. Clin. Epidemiol. 1996, 49, 1079–1084. Available online: https://helda.helsinki.fi/handle/10138/225872 (accessed on 9 March 2019). [CrossRef]
- Hemilä, H.; Chalker, E. Vitamin C for preventing and treating the common cold. Cochrane Database Syst. Rev. 2013, CD000980. Available online: https://helda.helsinki.fi/handle/10138/225864 (accessed on 9 March 2019). [CrossRef]
- Hemilä, H. Vitamin C and infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin C and common cold incidence: A review of studies with subjects under heavy physical stress. Int. J. Sports Med. 1996, 17, 379–383. Available online: https://helda.helsinki.fi/handle/10138/225881 (accessed on 9 March 2019). [CrossRef]
- Shibuya, N.; Humphers, J.M.; Agarwal, M.R.; Jupiter, D.C. Efficacy and safety of high-dose vitamin C on complex regional pain syndrome in extremity trauma and surgery: Systematic review and meta-analysis. J. Foot Ankle Surg. 2013, 52, 62–66. [Google Scholar] [CrossRef]
- Meena, S.; Sharma, P.; Gangary, S.K.; Chowdhury, B. Role of vitamin C in prevention of complex regional pain syndrome after distal radius fractures: A meta-analysis. Eur. J. Orthop. Surg. Traumatol. 2015, 25, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Roffey, D.M.; Dion, C.A.; Arab, A.; Wai, E.K. Effect of perioperative vitamin C supplementation on postoperative pain and the incidence of chronic regional pain syndrome: A systematic review and meta-analysis. Clin. J. Pain. 2016, 32, 179–185. [Google Scholar] [CrossRef]
- Carr, A.C.; McCall, C. The role of vitamin C in the treatment of pain: New insights. J. Transl. Med. 2017, 15, 77. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Louhiala, P. Vitamin C may affect lung infections. J. R. Soc. Med. 2007, 100, 495–498. [Google Scholar] [CrossRef]
- Hemilä, H.; Louhiala, P. Vitamin C for preventing and treating pneumonia. Cochrane Database Syst. Rev. 2013, CD005532. Available online: https://helda.helsinki.fi/handle/10138/225862 (accessed on 9 March 2019). [CrossRef]
- Crandon, J.H.; Lund, C.C. Vitamin C deficiency in an otherwise normal adult. N. Engl. J. Med. 1940, 222, 748–752. [Google Scholar] [CrossRef]
- Crandon, J.H.; Lund, C.C.; Dill, D.B. Experimental human scurvy. N. Engl. J. Med. 1940, 223, 353–369. [Google Scholar] [CrossRef]
- Lund, C.C.; Crandon, J.H. Human experimental scurvy and the relation of vitamin C deficiency to postoperative pneumonia and to wound healing. JAMA 1941, 116, 663–668. [Google Scholar] [CrossRef]
- Vitamin C Subcommittee. Vitamin C requirement of human adults. Experimental study of vitamin-C deprivation in man. Lancet 1948, 251, 853–858. [Google Scholar] [CrossRef]
- Bartley, W.; Krebs, H.A.; O’Brien, J.R.P. Vitamin C Requirement of Human Adults; A Report by the Vitamin C Subcommittee of the Accessory Food Factors Committee; Her Majesty’s Stationery Office (HMSO): London, UK, 1953. [Google Scholar]
- Krebs, H.A. The Sheffield experiment on the vitamin C requirement of human adults. Proc. Nutr. Soc. 1953, 12, 237–246. [Google Scholar] [CrossRef]
- Hodges, R.E.; Baker, E.M.; Hood, J.; Sauberlich, H.E.; March, S.C. Experimental scurvy in man. Am. J. Clin. Nutr. 1969, 22, 535–548. [Google Scholar] [CrossRef]
- Hood, J. Femoral neuropathy in scurvy. N. Engl. J. Med. 1969, 281, 1292–1293. [Google Scholar] [CrossRef]
- Hood, J.; Burns, C.A.; Hodges, R.E. Sjögren’s syndrome in scurvy. N. Engl. J. Med. 1970, 282, 1120–1124. [Google Scholar] [CrossRef]
- Hodges, R.E. What’s new about scurvy? Am. J. Clin. Nutr. 1971, 24, 383–384. [Google Scholar] [CrossRef]
- Hodges, R.E.; Hood, J.; Canham, J.E.; Sauberlich, H.E.; Baker, E.M. Clinical manifestations of ascorbic acid deficiency in man. Am. J. Clin. Nutr. 1971, 24, 432–443. [Google Scholar] [CrossRef]
- Kinsman, R.A.; Hood, J. Some behavioral effects of ascorbic acid deficiency. Am. J. Clin. Nutr. 1971, 24, 455–464. [Google Scholar] [CrossRef]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef]
- Food and Nutrition Board, Institute of Medicine: Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium and Carotenoids; National Academy Press: Washington, DC, USA, 2000; pp. 95–185. Available online: https://www.ncbi.nlm.nih.gov/books/NBK225483 (accessed on 22 January 2019).
- Kieffer, P.; Thannberger, P.; Wilhelm, J.M.; Kieffer, C.; Schneider, F. Multiple organ dysfunction dramatically improving with the infusion of vitamin C: More support for the persistence of scurvy in our “welfare” society. Intensive Care Med. 2001, 27, 448. [Google Scholar] [CrossRef]
- Holley, A.D.; Osland, E.; Barnes, J.; Krishnan, A.; Fraser, J.F. Scurvy: Historically a plague of the sailor that remains a consideration in the modern intensive care unit. Intern. Med. J. 2011, 41, 283–285. [Google Scholar] [CrossRef]
- Doll, S.; Ricou, B. Severe vitamin C deficiency in a critically ill adult: A case report. Eur. J. Clin. Nutr. 2013, 67, 881–882. [Google Scholar] [CrossRef]
- Weinstein, M.; Babyn, P.; Zlotkin, S. An orange a day keeps the doctor away: Scurvy in the year 2000. Pediatrics 2001, 108, E55. [Google Scholar] [CrossRef]
- Duvall, M.G.; Pikman, Y.; Kantor, D.B.; Ariagno, K.; Summers, L.; Sectish, T.C.; Mullen, M.P. Pulmonary hypertension associated with scurvy and vitamin deficiencies in an autistic child. Pediatrics 2013, 132, e1699–e1703. [Google Scholar] [CrossRef]
- Bennett, S.E.; Schmitt, W.P.; Stanford, F.C.; Baron, J.M. Case 22-2018: A 64-year-old man with progressive leg weakness, recurrent falls, and anemia. N. Engl. J. Med. 2018, 379, 282–289. [Google Scholar] [CrossRef]
- Shafar, J. Rapid reversion of electrocardiographic abnormalities after treatment in two cases of scurvy. Lancet 1967, 290, 176–178. [Google Scholar] [CrossRef]
- Singh, D.; Chan, W. Cardiomegaly and generalized oedema due to vitamin C deficiency. Singapore Med. J. 1974, 15, 60–63. Available online: http://smj.sma.org.sg/1501/1501smj11.pdf (accessed on 9 March 2019).
- Meisel, J.L.; McDowell, R.K. Case 39-1995: A 72-year-old man with exertional dyspnea, fatigue, and extensive ecchymoses and purpuric lesions. N. Engl. J. Med. 1995, 333, 1695–1702. [Google Scholar] [CrossRef]
- Mertens, M.T.; Gertner, E. Rheumatic manifestations of scurvy: A report of three recent cases in a major urban center and a review. Semin. Arthritis Rheum. 2011, 41, 286–290. [Google Scholar] [CrossRef]
- Kupari, M.; Rapola, J. Reversible pulmonary hypertension associated with vitamin C deficiency. Chest 2012, 142, 225–227. [Google Scholar] [CrossRef] [PubMed]
- Zipursky, J.S.; Alhashemi, A.; Juurlink, D. A rare presentation of an ancient disease: Scurvy presenting as orthostatic hypotension. BMJ Case Rep. 2014, 2014, bcr2013201982. [Google Scholar] [CrossRef] [PubMed]
- Abbas, F.; Ha, L.D.; Sterns, R.; von Doenhoff, L. Reversible right heart failure in scurvy: Rediscovery of an old observation. Circ. Heart Fail. 2016, 9, e003497. [Google Scholar] [CrossRef] [PubMed]
- Velandia, B.; Centor, R.M.; McConnell, V.; Shah, M. Scurvy is still present in developed countries. J. Gen. Intern. Med. 2008, 23, 1281–1284. [Google Scholar] [CrossRef] [PubMed]
- Woodier, N.; Koytzoumis, V. Scurvy: Presentation and skin manifestations of a not so uncommon condition. Emerg. Med. J. 2012, 29, 103. [Google Scholar] [CrossRef]
- Bonsall, A. Never surprise a patient with scurvy. Int. J. Dermatol. 2017, 56, 1488–1489. [Google Scholar] [CrossRef] [PubMed]
- Lux-Battistelli, C.; Battistelli, D. Latent scurvy with tiredness and leg pain in alcoholics: An underestimated disease three case reports. Medicine (Baltimore) 2017, 96, e8861. [Google Scholar] [CrossRef] [PubMed]
- Jiang, A.W.; Vijayaraghavan, M.; Mills, E.G.; Prisco, A.R.; Thurn, J.R. Scurvy, a not-so-ancient disease. Am. J. Med. 2018, 131, e185–e186. [Google Scholar] [CrossRef]
- Hunt, C.; Chakravorty, N.K.; Annan, G.; Habibzadeh, N.; Schorah, C.J. The clinical effects of vitamin C supplementation in elderly hospitalised patients with acute respiratory infections. Int. J. Vitam. Nutr. Res. 1994, 64, 212–219. Available online: http://www.mv.helsinki.fi/home/hemila/CP/Hunt_1994_ch.pdf (accessed on 22 January 2019).
- Fain, O.; Mathieu, E.; Thomas, M. Scurvy in patients with cancer. BMJ 1998, 316, 1661–1662. [Google Scholar] [CrossRef]
- Teixeira, A.; Carrie, A.S.; Genereau, T.; Herson, S.; Cherin, P. Vitamin C deficiency in elderly hospitalized patients. Am. J. Med. 2001, 111, 502. [Google Scholar] [CrossRef]
- Fain, O.; Paries, J.; Jacquart, B.; Le Moel, G.; Kettaneh, A.; Stirnemann, J.; Heron, C.; Sitbon, M.; Taleb, C.; Letellier, E.; et al. Hypovitaminosis C in hospitalized patients. Eur. J. Intern. Med. 2003, 14, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Mayland, C.R.; Bennett, M.I.; Allan, K. Vitamin C deficiency in cancer patients. Palliat. Med. 2005, 19, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.; Eintracht, S.; Hoffer, L.J. Vitamin C deficiency in a university teaching hospital. J. Am. Coll. Nutr. 2008, 27, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Raynaud-Simon, A.; Cohen-Bittan, J.; Gouronnec, A.; Pautas, E.; Senet, P.; Verny, M.; Boddaert, J. Scurvy in hospitalized elderly patients. J. Nutr. Health Aging 2010, 14, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, P.; Wiltshire, S.; Das, K.; Wilson, R.B. Vitamin C deficiency in an Australian cohort of metropolitan surgical patients. Pathology 2018, 50, 654–658. [Google Scholar] [CrossRef]
- MacLennan, W.J.; Hamilton, J.C. The effect of acute illness on leucocyte and plasma ascorbic acid levels. Br. J. Nutr. 1977, 38, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Ballmer, P.E.; Reinhart, W.H.; Jordan, P.; Buhler, E.; Moser, U.K.; Gey, K.F. Depletion of plasma vitamin C but not of vitamin E in response to cardiac operations [correction in 1995, 110, 1972]. J. Thorac. Cardiovasc. Surg. 1994, 108, 311–320. [Google Scholar]
- Borrelli, E.; Roux-Lombard, P.; Grau, G.E.; Girardin, E.; Ricou, B.; Dayer, J.; Suter, P.M. Plasma concentrations of cytokines, their soluble receptors, and antioxidant vitamins can predict the development of multiple organ failure in patients at risk. Crit. Care Med. 1996, 24, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Schorah, C.J.; Downing, C.; Piripitsi, A.; Gallivan, L.; Al-Hazaa, A.H.; Sanderson, M.J.; Bodenham, A. Total vitamin C, ascorbic acid, and dehydroascorbic acid concentrations in plasma of critically ill patients. Am. J. Clin. Nutr. 1996, 63, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Galley, H.F.; Davies, M.J.; Webster, N.R. Ascorbyl radical formation in patients with sepsis: Effect of ascorbate loading. Free Radic. Biol. Med. 1996, 20, 139–143. [Google Scholar] [CrossRef]
- Metnitz, P.G.; Krenn, C.G.; Steltzer, H.; Lang, T.; Ploder, J.; Lenz, K.; Le Gall, J.R.; Druml, W. Effect of acute renal failure requiring renal replacement therapy on outcome in critically ill patients. Crit. Care Med. 2002, 30, 2051–2058. [Google Scholar] [CrossRef]
- Lassnigg, A.; Punz, A.; Barker, R.; Keznickl, P.; Manhart, N.; Roth, E.; Hiesmayr, M. Influence of intravenous vitamin E supplementation in cardiac surgery on oxidative stress: A double-blinded, randomized, controlled study. Br. J. Anaesth. 2003, 90, 148–154. [Google Scholar] [CrossRef]
- Doise, J.M.; Aho, L.S.; Quenot, J.P.; Guilland, J.C.; Zeller, M.; Vergely, C.; Aube, H.; Blettery, B.; Rochette, L. Plasma antioxidant status in septic critically ill patients: A decrease over time. Fundam. Clin. Pharmacol. 2008, 22, 203–209. [Google Scholar] [CrossRef]
- Evans-Olders, R.; Eintracht, S.; Hoffer, L.J. Metabolic origin of hypovitaminosis C in acutely hospitalized patients. Nutrition 2010, 26, 1070–1074. [Google Scholar] [CrossRef]
- Rodemeister, S.; Duquesne, M.; Adolph, M.; Nohr, D.; Biesalski, H.K.; Unertl, K. Massive and long-lasting decrease in vitamin C plasma levels as a consequence of extracorporeal circulation. Nutrition 2014, 30, 673–678. [Google Scholar] [CrossRef]
- Katundu, K.G.H.; Hill, L.T.; Davids, L.M.; Joubert, I.A.; Miller, M.G.A.; Piercy, J.L.; Michell, W.L. An observational study on the relationship between plasma vitamin C, blood glucose, oxidative stress, endothelial dysfunction and outcome in patients with septic shock. South. Afr. J. Crit. Care 2016, 32, 21–27. [Google Scholar] [CrossRef]
- Carr, A.C.; Rosengrave, P.C.; Bayer, S.; Chambers, S.; Mehrtens, J.; Shaw, G.M. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit. Care 2017, 21, 300. [Google Scholar] [CrossRef]
- Long, C.L.; Maull, K.I.; Krishnan, R.S.; Laws, H.L.; Geiger, J.W.; Borghesi, L.; Franks, W.; Lawson, T.C.; Sauberlich, H.E. Ascorbic acid dynamics in the seriously ill and injured. J. Surg. Res. 2003, 109, 144–148. [Google Scholar] [CrossRef]
- Rümelin, A.; Jaehde, U.; Kerz, T.; Roth, W.; Krämer, M.; Fauth, U. Early postoperative substitution procedure of the antioxidant ascorbic acid. J. Nutr. Biochem. 2005, 16, 104–108. [Google Scholar] [CrossRef]
- Rümelin, A.; Humbert, T.; Lühker, O.; Drescher, A.; Fauth, U. Metabolic clearance of the antioxidant ascorbic acid in surgical patients. J. Surg. Res. 2005, 129, 46–51. [Google Scholar] [CrossRef]
- De Grooth, H.J.; Manubulu-Choo, W.P.; Zandvliet, A.S.; Spoelstra-de Man, A.M.E.; Girbes, A.R.; Swart, E.L.; Oudemans-van Straaten, H.M. Vitamin C pharmacokinetics in critically ill patients: A randomized trial of four iv regimens. Chest 2018, 153, 1368–1377. [Google Scholar] [CrossRef]
- Dingchao, H.; Zhiduan, Q.; Liye, H.; Xiaodong, F. The protective effects of high-dose ascorbic acid on myocardium against reperfusion injury during and after cardiopulmonary bypass. Thorac. Cardiovasc. Surg. 1994, 42, 276–278. [Google Scholar] [CrossRef]
- Tanaka, H.; Matsuda, T.; Miyagantani, Y.; Yukioka, T.; Matsuda, H.; Shimazaki, S. Reduction of resuscitation fluid volumes in severely burned patients using ascorbic acid administration: A randomized, prospective study. Arch. Surg. 2000, 135, 326–331. [Google Scholar] [CrossRef]
- Eslami, M.; Badkoubeh, R.S.; Mousavi, M.; Radmehr, H.; Salehi, M.; Tavakoli, N.; Avadi, M.R. Oral ascorbic acid in combination with beta-blockers is more effective than beta-blockers alone in the prevention of atrial fibrillation after coronary artery bypass grafting. Tex. Heart Inst. J. 2007, 34, 268–274. [Google Scholar]
- Colby, J.A.; Chen, W.T.; Baker, W.L.; Coleman, C.I.; Reinhart, K.; Kluger, J.; White, C.M. Effect of ascorbic acid on inflammatory markers after cardiothoracic surgery. Am. J. Health-Syst. Pharm. 2011, 68, 1632–1639. [Google Scholar] [CrossRef]
- Papoulidis, P.; Ananiadou, O.; Chalvatzoulis, E.; Ampatzidou, F.; Koutsogiannidis, C.; Karaiskos, T.; Madesis, A.; Drossos, G. The role of ascorbic acid in the prevention of atrial fibrillation after elective on-pump myocardial revascularization surgery: A single-center experience, a pilot study. Interact. Cardiovasc. Thorac. Surg. 2011, 12, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Bjordahl, P.M.; Helmer, S.D.; Gosnell, D.J.; Wemmer, G.E.; O’Hara, W.W.; Milfeld, D.J. Perioperative supplementation with ascorbic acid does not prevent atrial fibrillation in coronary artery bypass graft patients. Am. J. Surg. 2012, 204, 862–867. [Google Scholar] [CrossRef]
- Donovan, P.C.; Kramer, R.S. Prophylaxis to Reduce Postoperative Atrial Fibrillation in Cardiac Surgery. ClinicalTrials.gov NCT00953212; 2012. Available online: https://clinicaltrials.gov/ct2/show/NCT00953212 (accessed on 21 March 2019).
- Dehghani, M.R.; Madjidi, N.; Rahmani, A.; Asgari, B.; Rezaei, Y. Effect of oral vitamin C on atrial fibrillation development after isolated coronary artery bypass grafting surgery: A prospective randomized clinical trial. Cardiol. J. 2014, 21, 492–499. [Google Scholar] [CrossRef]
- Ebade, A.; Taha, W.S.; Saleh, R.H.; Fawzy, A. Ascorbic acid versus magnesium for the prevention of atrial fibrillation after coronary artery bypass grafting surgery. Egypt. J. Cardiothorac. Anesth. 2014, 8, 59–65. [Google Scholar] [CrossRef]
- Fowler, A.A.; Syed, A.A.; Knowlson, S.; Sculthorpe, R.; Farthing, D.; DeWilde, C.; Farthing, C.A.; Larus, T.L.; Martin, E.; Brophy, D.F.; et al. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J. Transl. Med. 2014, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Sarzaeem, M.; Shayan, N. Vitamin C in prevention of atrial fibrillation after coronary artery bypass graft: Double blind randomized clinical trial. Tehran Univ. Med. J. 2014, 71, 787–793. Available online: http://www.mv.helsinki.fi/home/hemila/T14.pdf (accessed on 22 January 2019). (In Persian).
- Zabet, M.H.; Mohammadi, M.; Ramezani, M.; Khalili, H. Effect of high-dose ascorbic acid on vasopressor’s requirement in septic shock. J. Res. Pharm. Pract. 2016, 5, 94–100. [Google Scholar] [CrossRef]
- Alshafey, M.K.; Elrakhawy, H.M.; Rezk, M.E.; Moustafa, H.M. Role of ascorbic acid in reduction of the incidence of the atrial fibrillation in patients under beta-blocker and undergoing coronary artery bypass graft operation in early post-operative period. J. Egypt. Soc. Cardio-Thorac. Surg. 2017, 25, 198–203. [Google Scholar] [CrossRef]
- Antonic, M.; Lipovec, R.; Gregorcic, F.; Juric, P.; Kosir, G. Perioperative ascorbic acid supplementation does not reduce the incidence of postoperative atrial fibrillation in on-pump coronary artery bypass graft patients. J. Cardiol. 2017, 69, 98–102. [Google Scholar] [CrossRef]
- Amini, S.; Robabi, H.N.; Tashnizi, M.A.; Vakili, V. Selenium, vitamin C and N-acetylcysteine do not reduce the risk of acute kidney injury after off-pump CABG: A randomized clinical trial. Braz. J. Cardiovasc. Surg. 2018, 33, 129–134. [Google Scholar] [CrossRef]
- Mirmohammadsadeghi, M.; Mirmohammadsadeghi, A.; Mahmoudian, M. Preventive use of ascorbic acid for atrial fibrillation after coronary artery bypass graft surgery. Heart Surg. Forum 2018, 21, E415–E417. [Google Scholar] [CrossRef]
- Abdoulhossein, D.; Taheri, I.; Saba, M.A.; Akbari, H.; Shafagh, S.; Zataollah, A. Effect of vitamin C and vitamin E on lung contusion: A randomized clinical trial study. Ann. Med. Surg. (Lond.) 2018, 36, 152–157. [Google Scholar] [CrossRef]
- Sadeghpour, A.; Alizadehasl, A.; Kyavar, M.; Sadeghi, T.; Moludi, J.; Gholizadeh, F.; Totonchi, Z.; Ghadrdoost, B. Impact of vitamin C supplementation on post-cardiac surgery ICU and hospital length of stay. Anesth. Pain Med. 2015, 5, e25337. [Google Scholar] [CrossRef]
- Montori, V.M.; Guyatt, G.H. Intention-to-treat principle. CMAJ 2001, 165, 1339–1341. [Google Scholar]
- McCoy, C.E. Understanding the intention-to-treat principle in randomized controlled trials. West. J. Emerg. Med. 2017, 18, 1075–1078. [Google Scholar] [CrossRef]
- DeMets, D.L.; Cook, T. Challenges of non-intention-to-treat analyses. JAMA 2019, 321, 145–146. [Google Scholar] [CrossRef]
- Friedrich, J.O.; Adhikari, N.K.; Beyene, J. Ratio of means for analyzing continuous outcomes in meta-analysis performed as well as mean difference methods. J. Clin. Epidemiol. 2011, 64, 556–564. [Google Scholar] [CrossRef]
- Friedrich, J.O.; Adhikari, N.K.; Beyene, J. The ratio of means method as an alternative to mean differences for analyzing continuous outcome variables in meta-analysis: A simulation study. BMC Med. Res. Methodol. 2008, 8, 32. [Google Scholar] [CrossRef]
- Hemilä, H. Many continuous variables such as the duration of the common cold should be analyzed using the relative scale. J. Clin. Epidemiol. 2016, 78, 128–129. [Google Scholar] [CrossRef]
- Hemilä, H. Duration of the common cold and similar continuous outcomes should be analyzed on the relative scale: A case study of two zinc lozenge trials. BMC Med. Res. Methodol. 2017, 17, 82. [Google Scholar] [CrossRef]
- R Core Team. R Project for Statistical Computing. 2019. Available online: https://www.r-project.org (accessed on 22 January 2019).
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analysis. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Englard, S.; Seifter, S. The biochemical functions of ascorbic acid. Annu. Rev. Nutr. 1986, 6, 365–406. [Google Scholar] [CrossRef]
- Levine, M.; Morita, K. Ascorbic acid in endocrine systems. Vitam. Horm. 1985, 42, 1–64. [Google Scholar] [CrossRef] [PubMed]
- Levine, M. New concepts in the biology and biochemistry of ascorbic acid. N. Engl. J. Med. 1986, 314, 892–902. [Google Scholar] [CrossRef]
- Rice, M.E. Ascorbate regulation and its neuroprotective role in the brain. Trends Neurosci. 2000, 23, 209–216. [Google Scholar] [CrossRef]
- Holowatz, L.A. Ascorbic acid: What do we really NO? J. Appl. Physiol. 2011, 111, 1542–1543. [Google Scholar] [CrossRef]
- May, J.M.; Qu, Z.; Meredith, M.E. Mechanisms of ascorbic acid stimulation of norepinephrine synthesis in neuronal cells. Biochem. Biophys. Res. Commun. 2012, 426, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Figueroa-Méndez, R.; Rivas-Arancibia, S. Vitamin C in health and disease: Its role in the metabolism of cells and redox state in the brain. Front. Physiol. 2015, 6, 397. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Shaw, G.M.; Fowler, A.A.; Natarajan, R. Ascorbate-dependent vasopressor synthesis: A rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care 2015, 19, 418. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Mains, R.E.; Eipper, B.A. 60 YEARS OF POMC: From POMC and α-MSH to PAM, molecular oxygen, copper, and vitamin C. J. Mol. Endocrinol. 2016, 56, T63–T76. [Google Scholar] [CrossRef]
- Manning, J.; Mitchell, B.; Appadurai, D.A.; Shakya, A.; Pierce, L.J.; Wang, H.; Nganga, V.; Swanson, P.C.; May, J.M.; Tantin, D.; et al. Vitamin C promotes maturation of T-cells. Antioxid. Redox Signal. 2013, 19, 2054–2067. [Google Scholar] [CrossRef]
- May, J.M.; Harrison, F.E. Role of vitamin C in the function of the vascular endothelium. Antioxid. Redox Signal. 2013, 19, 2068–2083. [Google Scholar] [CrossRef]
- Monfort, A.; Wutz, A. Breathing-in epigenetic change with vitamin C. EMBO Rep. 2013, 14, 337–346. [Google Scholar] [CrossRef]
- Young, J.I.; Züchner, S.; Wang, G. Regulation of the epigenome by vitamin C. Annu. Rev. Nutr. 2015, 35, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Camarena, V.; Wang, G. The epigenetic role of vitamin C in health and disease. Cell. Mol. Life. Sci. 2016, 73, 1645–1658. [Google Scholar] [CrossRef]
- Ang, A.; Pullar, J.M.; Currie, M.J.; Vissers, M.C.M. Vitamin C and immune cell function in inflammation and cancer. Biochem. Soc. Trans. 2018, 46, 1147–1159. [Google Scholar] [CrossRef]
- Mandl, J.; Szarka, A.; Bánhegyi, G. Vitamin C: Update on physiology and pharmacology. Br. J. Pharmacol. 2009, 157, 1097–1110. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Levine, M. Vitamin C: The known and the unknown and Goldilocks. Oral Dis. 2016, 22, 463–493. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.L.; Villamor, E. Update: Effects of antioxidant and non-antioxidant vitamin supplementation on immune function. Nutr. Rev. 2007, 65, 181–217. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Wilson, J.X. Mechanism of action of vitamin C in sepsis: Ascorbate modulates redox signaling in endothelium. Biofactors 2009, 35, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Tyml, K. Vitamin C and microvascular dysfunction in systemic inflammation. Antioxidants 2017, 6, 49. [Google Scholar] [CrossRef] [PubMed]
- Harrison, F.E.; May, J.M. Vitamin C function in the brain: Vital role of the ascorbate transporter SVCT2. Free Radic. Biol. Med. 2009, 46, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.M. Neuropsychiatric scurvy. Psychosomatics 2015, 56, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Carnes, C.A.; Chung, M.K.; Nakayama, T.; Nakayama, H.; Baliga, R.S.; Piao, S.; Kanderian, A.; Pavia, S.; Hamlin, R.L.; McCarthy, P.M.; et al. Ascorbate attenuates atrial pacing-induced peroxynitrite formation and electrical remodeling and decreases the incidence of postoperative atrial fibrillation. Circ. Res. 2001, 89, E32–E38. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Bae, S.; Kim, Y.; Cho, C.H.; Kim, S.J.; Kim, Y.J.; Lee, S.P.; Kim, H.R.; Hwang, Y.I.; Kang, J.S.; et al. Vitamin C prevents stress-induced damage on the heart caused by the death of cardiomyocytes, through down-regulation of the excessive production of catecholamine, TNF-α, and ROS production in Gulo(-/-)Vit C-Insufficient mice. Free Radic. Biol. Med. 2013, 65, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Talkhabi, M.; Pahlavan, S.; Aghdami, N.; Baharvand, H. Ascorbic acid promotes the direct conversion of mouse fibroblasts into beating cardiomyocytes. Biochem. Biophys. Res. Commun. 2015, 463, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Akolkar, G.; da Silva Dias, D.; Ayyappan, P.; Bagchi, A.K.; Jassal, D.S.; Salemi, V.M.C.; Irigoyen, M.C.; De Angelis, K.; Singal, P.K. Vitamin C mitigates oxidative/nitrosative stress and inflammation in doxorubicin-induced cardiomyopathy. Am. J. Physiol. Heart. Circ. Physiol. 2017, 313, H795–H809. [Google Scholar] [CrossRef]
- Levin, N.A.; Greer, K.E. Scurvy in an unrepentant carnivore. Cutis 2000, 66, 39–44. Available online: https://www.mdedge.com/dermatology/article/66468/scurvy-unrepentant-carnivore (accessed on 9 March 2019).
- Pangan, A.L.; Robinson, D. Hemarthrosis as initial presentation of scurvy. J. Rheumatol. 2001, 28, 1923–1925. [Google Scholar]
- Bingham, A.C.; Kimura, Y.; Imundo, L. A 16-year-old boy with purpura and leg pain. J. Pediatr. 2003, 142, 560–563. [Google Scholar] [CrossRef]
- Reed, R.M. Captain Ignose to the rescue. Am. J. Med. 2010, 123, 704–706. [Google Scholar] [CrossRef]
- De Luna, R.H.; Colley, B.J.; Smith, K.; Divers, S.G.; Rinehart, J.; Marques, M.B. Scurvy: An often forgotten cause of bleeding. Am. J. Hematol. 2003, 74, 85–87. [Google Scholar] [CrossRef]
- Patrozou, E.; Opal, S. Scurvy masquerading as infectious cellulitis. Intern. Med. J. 2008, 38, 452–453. [Google Scholar] [CrossRef] [PubMed]
- Lau, H.; Massasso, D.; Joshua, F. Skin, muscle and joint disease from the 17th century: Scurvy. Int. J. Rheum. Dis. 2009, 12, 361–365. [Google Scholar] [CrossRef]
- Bernardino, V.R.; Mendes-Bastos, P.; Noronha, C.; Henriques, C.C. 2011: The scurvy Odyssey. BMJ Case Rep. 2012, 2012, bcr0220125819. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.; Di Primio, G.; Humphrey-Murto, S. Scurvy in the developed world. CMAJ 2011, 183, E752–E755. [Google Scholar] [CrossRef] [PubMed]
- Ciccocioppo, R.; Gallia, A.; Carugno, A.; Gamba, G.; Corazza, G.R. An unconventional case of scurvy. Eur. J. Clin. Nutr. 2013, 67, 1336–1337. [Google Scholar] [CrossRef]
- Fleming, J.D.; Martin, B.; Card, D.J.; Mellerio, J.E. Pain, purpura and curly hairs. Clin. Exp. Dermatol. 2013, 38, 940–942. [Google Scholar] [CrossRef]
- Zammit, P. Vitamin C deficiency in an elderly adult. J. Am. Geriatr. Soc. 2013, 61, 657–658. [Google Scholar] [CrossRef]
- Kluesner, N.H.; Miller, D.G. Scurvy: Malnourishment in the land of plenty. J. Emerg. Med. 2014, 46, 530–532. [Google Scholar] [CrossRef] [PubMed]
- Dufrost, V.; Risse, J.; Malgras, A.; Barraud, H.; Jaussaud, R.; Zuily, S.; Wahl, D. Unexpected cause of bleeding. Am. J. Med. 2017, 130, e387–e388. [Google Scholar] [CrossRef] [PubMed]
- Kinlin, L.M.; Blanchard, A.C.; Silver, S.; Morris, S.K. Scurvy as a mimicker of osteomyelitis in a child with autism spectrum disorder. Int. J. Infect. Dis. 2018, 69, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Berkram, P.; Bedano, P.M.; Kahi, C.J.; Illamperuma, C.; Matthews, D.E.; Antony, A.C. A landlubber with an ancient mariner’s leaky vessels. Gastrointest. Endosc. 2007, 66, 1065–1066. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, L.V.; Wilson, M.R.; Holmes, S. A historic disease still prevalent today. BMJ 2017, 356, j1013. [Google Scholar] [CrossRef] [PubMed]
- Schuman, R.W.; Rahmin, M.; Dannenberg, A.J. Scurvy and the gastrointestinal tract. Gastrointest. Endosc. 1997, 45, 195–196. [Google Scholar] [CrossRef]
- Blee, T.H.; Cogbill, T.H.; Lambert, P.J. Hemorrhage associated with vitamin C deficiency in surgical patients. Surgery 2002, 131, 408–412. [Google Scholar] [CrossRef]
- Choh, C.T.; Rai, S.; Abdelhamid, M.; Lester, W.; Vohra, R.K. Unrecognised scurvy. BMJ 2009, 339, b3580. [Google Scholar] [CrossRef] [PubMed]
- Ohta, A.; Yoshida, S.; Imaeda, H.; Ohgo, H.; Sujino, T.; Yamaoka, M.; Kanno, R.; Kobayashi, T.; Kinoshita, S.; Iida, S.; et al. Scurvy with gastrointestinal bleeding. Endoscopy 2013, 45 (Suppl. 2), E147–E148. [Google Scholar] [CrossRef]
- Poussier, M.; Osmak, L.; Naouri, A.; Rouber, X. Intestinal disorders caused by scurvy. Clin. Res. Hepatol. Gastroenterol. 2014, 38, e39–e40. [Google Scholar] [CrossRef]
- DeSantis, J. Scurvy and psychiatric symptoms. Perspect. Psychiatr. Care 1993, 29, 18–22. [Google Scholar] [CrossRef]
- Shavit, I.; Brown, T.M. Simultaneous scurvy and Wernicke’s encephalopathy in a patient with an ascorbate-responsive dyskinesia. Psychosomatics 2013, 54, 181–186. [Google Scholar] [CrossRef]
- Noble, M.; Healey, C.S.; McDougal-Chukwumah, L.D.; Brown, T.M. Old disease, new look? A first report of parkinsonism due to scurvy, and of refeeding-induced worsening of scurvy. Psychosomatics 2013, 54, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.D.; Stevens, E.; Ali, M.; Carroll, D.W.; Brown, T.M. The neuropsychiatry of scurvy. Psychosomatics 2014, 55, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Finkle, P. Vitamin C saturation levels in the body in normal subjects and in various pathological conditions. J. Clin. Investig. 1937, 16, 587–593. [Google Scholar] [CrossRef]
- Lund, C.C. The effect of surgical operations on the level of cevitamic acid in the blood plasma. N. Engl. J. Med. 1939, 221, 123–127. [Google Scholar] [CrossRef]
- Abbasy, M.A.; Harris, L.J.; Hill, N.G. Vitamin C and infection: Excretion of vitamin C in osteomyelitis. Lancet 1937, 230, 177–180. [Google Scholar] [CrossRef]
- Evans, W. Vitamin C in heart failure. Lancet 1938, 231, 308–309. [Google Scholar] [CrossRef]
- Editorial. Ascorbic acid as a diuretic. Lancet 1944, 244, 6310. [Google Scholar] [CrossRef]
- Hochwald, A. Vitamin C in the treatment of croupous pneumonia. Dtsch. Med. Wochenschr. 1937, 63, 182–184. Available online: http://www.mv.helsinki.fi/home/hemila/T8.pdf (accessed on 22 January 2019). (In German). [CrossRef]
- Gander, J.; Niederberger, W. Vitamin C in the treatment of pneumonia. Münch. Med. Wochenschr. 1936, 83, 2074–2077. Available online: http://www.mv.helsinki.fi/home/hemila/T1.pdf (accessed on 22 January 2019). (In German).
- Bohnholtzer, E. Contribution to the question of pneumonia treatment with vitamin C. Dtsch. Med. Wochenschr. 1937, 63, 1001–1003. Available online: http://www.mv.helsinki.fi/home/hemila/T7.pdf (accessed on 22 January 2019). (In German). [CrossRef]
- Klenner, F.R. Virus pneumonia and its treatment with vitamin C. South. Med. Surg. 1948, 110, 36–38. [Google Scholar]
- Klenner, F.R. Massive doses of vitamin C and the virus diseases. South. Med. Surg. 1951, 113, 101–107. [Google Scholar] [PubMed]
- Dalton, W.L. Massive doses of vitamin C in the treatment of viral diseases. J. Indiana State Med. Assoc. 1962, 55, 1151–1154. [Google Scholar]
- Stone, I. The Healing Factor: Vitamin C against Disease; Grosset Dunlap: New York, NY, USA, 1972. [Google Scholar]
- Mochalkin, N.I. Ascorbic acid in the complex therapy of acute pneumonia. Voen. Med. Zh. 1970, 9, 17–21. Available online: http://www.mv.helsinki.fi/home/hemila/T5.pdf (accessed on 22 January 2019). (In Russian). [PubMed]
- Kimbarowski, J.A.; Mokrow, N.J. Colored precipitation reaction of the urine according to Kimbarowski as an index of the effect of ascorbic acid during treatment of viral influenza. Dtsch. Gesundheitsw. 1967, 22, 2413–2418. Available online: http://www.mv.helsinki.fi/home/hemila/T4.pdf (accessed on 22 January 2019). (In German).
- Hemilä, H.; Louhiala, P. Vitamin C for preventing and treating pneumonia. Cochrane Database Syst. Rev. 2007, CD005532. [Google Scholar] [CrossRef]
- Hume, R.; Weyers, E. Changes in leucocyte ascorbic acid during the common cold. Scott. Med. J. 1973, 18, 3–7. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin C intake and susceptibility to the common cold. Br. J. Nutr. 1997, 77, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Bates, C.J.; Schorah, C.J.; Hemilä, H. Vitamin C intake and susceptibility to the common cold: Invited comments and reply. Br. J. Nutr. 1997, 78, 857–866. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin C supplementation and common cold symptoms: Factors affecting the magnitude of the benefit. Med. Hypotheses 1999, 52, 171–178. Available online: https://helda.helsinki.fi/handle/10138/223761 (accessed on 9 March 2019). [CrossRef]
- Karlowski, T.R.; Chalmers, T.C.; Frenkel, L.D.; Kapikian, A.Z.; Lewis, T.L.; Lynch, J.M. Ascorbic acid for the common cold: A prophylactic and therapeutic trial. JAMA 1975, 231, 1038–1042. [Google Scholar] [CrossRef]
- Anderson, T.W.; Suranyi, G.; Beaton, G.H. The effect on winter illness of large doses of vitamin C. Can. Med. Assoc. J. 1974, 111, 31–36. [Google Scholar]
- Anderson, T.W.; Reid, D.B.W.; Beaton, G.H. Vitamin C and the common cold: A double-blind trial. Can. Med. Assoc. J. 1972, 107, 503–508. [Google Scholar] [PubMed]
- Ludvigsson, J.; Hansson, L.O.; Tibbling, G. Vitamin C as a preventive medicine against common colds in children. Scand. J. Infect. Dis. 1977, 9, 91–98. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin C and sex differences in respiratory tract infections. Respir. Med. 2008, 102, 625–626. [Google Scholar] [CrossRef] [PubMed]
- Tyrrell, D.A.; Craig, J.W.; Meada, T.W.; White, T. A trial of ascorbic acid in the treatment of the common cold. Br. J. Prev. Soc. Med. 1977, 31, 189–191. [Google Scholar] [CrossRef]
- Baird, I.M.; Hughes, R.E.; Wilson, H.K.; Davies, J.E.; Howard, A.N. The effects of ascorbic acid and flavonoids on the occurrence of symptoms normally associated with the common cold. Am. J. Clin. Nutr. 1979, 32, 1686–1690. [Google Scholar] [CrossRef] [PubMed]
- Constantini, N.W.; Dubnov-Raz, G.; Eyal, B.B.; Berry, E.M.; Cohen, A.H.; Hemilä, H. The effect of vitamin C on upper respiratory infections in adolescent swimmers: A randomized trial. Eur. J. Pediatr. 2011, 170, 59–63. [Google Scholar] [CrossRef]
- Panagiotou, O.A.; Contopoulos-Ioannidis, D.G.; Ioannidis, J.P. Comparative effect sizes in randomised trials from less developed and more developed countries: Meta-epidemiological assessment. BMJ 2013, 346, f707. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.J.; Hu, W.K.; Liu, Y.Y.; Shi, D.M.; Cheng, W.J.; Guo, Y.H.; Yang, Q.; Zhao, Y.X.; Zhou, Y.J. The effect of intravenous vitamin C infusion on periprocedural myocardial injury for patients undergoing elective percutaneous coronary intervention. Can. J. Cardiol. 2014, 30, 96–101. [Google Scholar] [CrossRef]
- Spoelstra-de Man, A.M.E.; Elbers, P.W.G.; Oudemans-van Straaten, H.M. Making sense of early high-dose intravenous vitamin C in ischemia/reperfusion injury. Crit. Care 2018, 22, 70. [Google Scholar] [CrossRef]
- Angus, D.C.; van der Poll, T. Severe sepsis and septic shock. N. Engl. J. Med. 2013, 369, 840–851. [Google Scholar] [CrossRef]
- Wilson, J.X. Evaluation of vitamin C for adjuvant sepsis therapy. Antioxid. Redox Signal. 2013, 19, 2129–2140. [Google Scholar] [CrossRef]
- Oudemans-van Straaten, H.M.; Spoelstra-de Man, A.M.; de Waard, M.C. Vitamin C revisited. Crit. Care 2014, 18, 460. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Oudemans-van Straaten, H.M. Vitamin C supplementation in the critically ill patient. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Marik, P.E. Hydrocortisone, ascorbic acid and thiamine (HAT therapy) for the treatment of sepsis: Focus on ascorbic acid. Nutrients 2018, 10, 1762. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, A.; Andersen, L.W.; Huang, D.T.; Berg, K.M.; Grossestreuer, A.V.; Marik, P.E.; Sherwin, R.L.; Hou, P.C.; Becker, L.B.; Cocchi, M.N.; et al. Ascorbic acid, corticosteroids, and thiamine in sepsis: A review of the biologic rationale and the present state of clinical evaluation. Crit. Care 2018, 22, 283. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Oudemans-van Straaten, H.M.; Berger, M.M. Vitamin therapy in critically ill patients: Focus on thiamine, vitamin C, and vitamin D. Intensive Care Med. 2018, 44, 1940–1944. [Google Scholar] [CrossRef] [PubMed]
- Spoelstra-de Man, A.M.E.; Elbers, P.W.G.; Oudemans-Van Straaten, H.M. Vitamin C: Should we supplement? Curr. Opin. Crit. Care 2018, 24, 248–255. [Google Scholar] [CrossRef]
- Zhang, M.; Robitaille, L.; Eintracht, S.; Hoffer, L.J. Vitamin C provision improves mood in acutely hospitalized patients. Nutrition 2011, 27, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, X.J.; Robitaille, L.; Eintracht, S.; MacNamara, E.; Hoffer, L.J. Effects of vitamin C and vitamin D administration on mood and distress in acutely hospitalized patients. Am. J. Clin. Nutr. 2013, 98, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Sun, H.; Wang, Y.; Riordan, H.D.; Hewitt, S.M.; Katz, A.; Wesley, R.A.; Levine, M. Vitamin C pharmacokinetics: Implications for oral and intravenous use. Ann. Intern. Med. 2004, 140, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Padayatty, S.J.; Espey, M.G. Vitamin C: A concentration-function approach yields pharmacology and therapeutic discoveries. Adv. Nutr. 2011, 2, 78–88. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Sun, A.Y.; Chen, Q.; Espey, M.G.; Drisko, J.; Levine, M. Vitamin C: Intravenous use by complementary and alternative medicine practitioners and adverse effects. PLoS ONE 2010, 5, e11414. [Google Scholar] [CrossRef]
- Mikirova, N.; Casciari, J.; Rogers, A.; Taylor, P. Effect of high-dose intravenous vitamin C on inflammation in cancer patients. J. Transl. Med. 2012, 10, 189. [Google Scholar] [CrossRef]
- Schencking, M.; Vollbracht, C.; Weiss, G.; Lebert, J.; Biller, A.; Goyvaerts, B.; Kraft, K. Intravenous vitamin C in the treatment of shingles: Results of a multicenter prospective cohort study. Med. Sci. Monit. 2012, 18, CR215–CR224. [Google Scholar] [CrossRef]
- Pourmatroud, E.; Hormozi, L.; Hemadi, M.; Golshahi, R. Intravenous ascorbic acid (vitamin C) administration in myomectomy: A prospective, randomized, clinical trial. Arch. Gynecol. Obstet. 2012, 285, 111–115. [Google Scholar] [CrossRef]
- Hoffer, L.J.; Robitaille, L.; Zakarian, R.; Melnychuk, D.; Kavan, P.; Agulnik, J.; Cohen, V.; Small, D.; Miller, W.H. High-dose intravenous vitamin C combined with cytotoxic chemotherapy in patients with advanced cancer: A phase I-II clinical trial. PLoS ONE 2015, 10, e0120228. [Google Scholar] [CrossRef]
- Jeon, Y.; Park, J.S.; Moon, S.; Yeo, J. Effect of intravenous high dose vitamin C on postoperative pain and morphine use after laparoscopic colectomy: A randomized controlled trial. Pain Res. Manag. 2016, 2016, 9147279. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Kim, D.J.; Na, C.H.; Shin, B.S. A study of intravenous administration of vitamin C in the treatment of acute herpetic pain and postherpetic neuralgia. Ann. Dermatol. 2016, 28, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Nauman, G.; Gray, J.C.; Parkinson, R.; Levine, M.; Paller, C.J. Systematic review of intravenous ascorbate in cancer clinical trials. Antioxidants 2018, 7, 89. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D.; CONSORT Group. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.L.; LaValley, M.P.; Felson, D.T. Discrepancy between published report and actual conduct of randomized clinical trials. J. Clin. Epidemiol. 2002, 55, 783–786. [Google Scholar] [CrossRef]
- Devereaux, P.J.; Choi, P.T.; El-Dika, S.; Bhandari, M.; Montori, V.M.; Schunemann, H.J.; Garg, A.X.; Busse, J.W.; Heels-Ansdell, D.; Ghali, W.A.; et al. An observational study found that authors of randomized controlled trials frequently use concealment of randomization and blinding, despite the failure to report these methods. J. Clin. Epidemiol. 2004, 57, 1232–1236. [Google Scholar] [CrossRef] [PubMed]
- Mhaskar, R.; Djulbegovic, B.; Magazin, A.; Soares, H.P.; Kumar, A. Published methodological quality of randomized controlled trials does not reflect the actual quality assessed in protocols. J. Clin. Epidemiol. 2012, 65, 602–609. [Google Scholar] [CrossRef]
- Hrobjartsson, A.; Gøtzsche, P.C. Is the placebo powerless? An analysis of clinical trials comparing placebo with no treatment. N. Engl. J. Med. 2001, 344, 1594–1602. [Google Scholar] [CrossRef] [PubMed]
- Jahan, K.; Ahmad, K.; Ali, M.A. Effect of ascorbic acid in the treatment of tetanus. Bangladesh Med. Res. Counc. Bull. 1984, 10, 24–28. [Google Scholar]
- Du, W.D.; Yuan, Z.R.; Sun, J.; Tang, J.X.; Cheng, A.Q.; Shen, D.M.; Huang, C.J.; Song, X.H.; Yu, X.F.; Zheng, S.B. Therapeutic efficacy of high-dose vitamin C on acute pancreatitis and its potential mechanisms. World J. Gastroenterol. 2003, 9, 2565–2569. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Koivula, T. Vitamin C for preventing and treating tetanus. Cochrane Database Syst. Rev. 2013, CD006665. Available online: https://helda.helsinki.fi/handle/10138/225863 (accessed on 9 March 2019). [CrossRef] [PubMed]
- Dey, P.K. Efficacy of vitamin C in counteracting tetanus toxin toxicity. Naturwissenschaften 1966, 53, 310. [Google Scholar] [CrossRef]
- Clemetson, C.A.B. Barlow’s disease. Med. Hypotheses 2002, 59, 52–56. [Google Scholar] [CrossRef]
- Hemilä, H.; Kaprio, J. Modification of the effect of vitamin E supplementation on the mortality of male smokers by age and dietary vitamin C. Am. J. Epidemiol. 2009, 169, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Sisto, T.; Paajanen, H.; Metsä-Ketelä, T.; Harmoinen, A.; Nordback, I.; Tarkka, M. Pretreatment with antioxidants and allopurinol diminishes cardiac onset events in coronary artery bypass grafting. Ann. Thorac. Surg. 1995, 59, 1519–1523. [Google Scholar] [CrossRef]
- Galley, H.F.; Howdle, P.D.; Walker, B.E.; Webster, N.R. The effects of intravenous antioxidants in patients with septic shock. Free Radic. Biol. Med. 1997, 23, 768–774. [Google Scholar] [CrossRef]
- Nathens, A.B.; Neff, M.J.; Jurkovich, G.J.; Klotz, P.; Farver, K.; Ruzinski, J.T.; Radella, F.; Garcia, I.; Maier, R.V. Randomized, prospective trial of antioxidant supplementation in critically ill surgical patients. Ann. Surg. 2002, 236, 814–822. [Google Scholar] [CrossRef]
- Crimi, E.; Liguori, A.; Condorelli, M.; Cioffi, M.; Astuto, M.; Bontempo, P.; Pignalosa, O.; Vietri, M.T.; Molinari, A.M.; Sica, V.; et al. The beneficial effects of antioxidant supplementation in enteral feeding in critically ill patients: A prospective, randomized, double-blind, placebo-controlled trial. Anesth. Analg. 2004, 99, 857–863. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Soguel, L.; Shenkin, A.; Revelly, J.P.; Pinget, C.; Baines, M.; Chiolero, R.L. Influence of early antioxidant supplements on clinical evolution and organ function in critically ill cardiac surgery, major trauma, and subarachnoid hemorrhage patients. Crit. Care 2008, 12, R101. [Google Scholar] [CrossRef]
- Barbosa, E.; Faintuch, J.; Machado Moreira, E.A.; Goncalves da Silva, V.R.; Lopes Pereima, M.J.; Martins Fagundes, R.L.; Filho, D.W. Supplementation of vitamin E, vitamin C, and zinc attenuates oxidative stress in burned children: A randomized, double-blind, placebo-controlled pilot study. J. Burn Care Res. 2009, 30, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.P.; Clochesy, J.M.; Goldstein, L.S.; Owen, H. Mechanical ventilation antioxidant trial. Am. J. Crit. Care 2015, 24, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Collier, B.R.; Giladi, A.; Dossett, L.A.; Dyer, L.; Fleming, S.B.; Cotton, B.A. Impact of high-dose antioxidants on outcomes in acutely injured patients. JPEN J. Parenter. Enteral Nutr. 2008, 32, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Giladi, A.M.; Dossett, L.A.; Fleming, S.B.; Abumrad, N.N.; Cotton, B.A. High-dose antioxidant administration is associated with a reduction in post-injury complications in critically ill trauma patients. Injury 2011, 42, 78–82. [Google Scholar] [CrossRef]
- Kahn, S.A.; Beers, R.J.; Lentz, C.W. Resuscitation after severe burn injury using high-dose ascorbic acid: A retrospective review. J. Burn Care Res. 2011, 32, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Bedreag, O.H.; Rogobete, A.F.; Sarandan, M.; Cradigati, A.C.; Papurica, M.; Rosu, O.M.; Luca, L.; Vernic, C.; Nartita, R.; Sandesc, D. Influence of antioxidant therapy on the clinical status of multiple trauma patients; a retrospective single center study. Rom. J. Anaesth. Intensive Care 2015, 22, 89–96. [Google Scholar]
- Marik, P.E.; Khangoora, V.; Rivera, R.; Hooper, M.H.; Catravas, J. Hydrocortisone, vitamin C, and thiamine for the treatment of severe sepsis and septic shock: A retrospective before-after study. Chest 2017, 151, 1229–1238. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, M.; Gandhi, H.; Shah, K.; Pandya, H.; Patel, R.; Keshvani, S.; Yadav, N. Hydrocortisone, vitamin C and thiamine for the treatment of sepsis and septic shock following cardiac surgery. Indian J. Anaesth. 2018, 62, 934–939. [Google Scholar] [PubMed]
- Kim, W.Y.; Jo, E.J.; Eom, J.S.; Mok, J.; Kim, M.H.; Kim, K.U.; Park, H.K.; Lee, M.K.; Lee, K. Combined vitamin C, hydrocortisone, and thiamine therapy for patients with severe pneumonia who were admitted to the intensive care unit: Propensity score-based analysis of a before-after cohort study. J. Crit. Care 2018, 47, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Spectacular reduction in the mortality of acutely injured patients by the administration of vitamins C and E and selenium [reply in 2009, 33, 449]. JPEN J. Parenter. Enteral Nutr. 2009, 33, 447–448. [Google Scholar] [CrossRef] [PubMed]
- Cathcart, R.F. Vitamin C, titrating to bowel tolerance, anascorbemia, and acute induced scurvy. Med. Hypotheses 1981, 7, 1359–1376. [Google Scholar] [CrossRef]
- Buehner, M.; Pamplin, J.; Studer, L.; Hughes, R.L.; King, B.T.; Graybill, J.C.; Chung, K.K. Oxalate nephropathy after continuous infusion of high-dose vitamin C as an adjunct to burn resuscitation. J. Burn Care Res. 2016, 37, e374–e379. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, T.C. Effects of ascorbic acid on the common cold: An evaluation of the evidence. Am. J. Med. 1975, 58, 532–536. [Google Scholar] [CrossRef]
- Hemilä, H. Do Vitamins C and E Affect Respiratory Infections? Ph.D. Thesis, University of Helsinki, Helsinki, Finland, 2006; pp. 21–45, 59–66. Available online: https://hdl.handle.net/10138/20335 (accessed on 22 January 2019).
- Council of Scientific Affairs, American Medical Association. Vitamin preparations as dietary supplements and as therapeutic agents. JAMA 1987, 257, 1929–1936. [Google Scholar] [CrossRef]
- Dykes, M.H.M.; Meier, P. Ascorbic acid and the common cold: Evaluation of its efficacy and toxicity. JAMA 1975, 231, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Pauling, L. Ascorbic acid and the common cold: Evaluation of its efficacy and toxicity. Part I. Med. Tribune 1976, 17, 18–19. [Google Scholar]
- Pauling, L. Ascorbic acid and the common cold. Part II. Med. Tribune 1976, 17, 37–38. [Google Scholar]
- Chalmers, T.C. Dissent to the preceding article by H. Hemilä. J. Clin. Epidemiol. 1996, 49, 1085. [Google Scholar] [CrossRef]
- Hemilä, H. To the dissent by Thomas Chalmers. J. Clin. Epidemiol. 1996, 49, 1087. Available online: https://helda.helsinki.fi/handle/10138/225873 (accessed on 9 March 2019). [CrossRef]
- Richards, E. The politics of therapeutic evaluation: The vitamin C and cancer controversy. Soc. Stud. Sci. 1988, 18, 653–701. [Google Scholar] [CrossRef]
- Richards, E. Vitamin C and Cancer: Medicine or Politics? St. Martins Press: New York, NY, USA, 1991. [Google Scholar]
- Segerstråle, U. Beleaguering the cancer establishment. Science 1992, 255, 613–615. [Google Scholar] [CrossRef]
- Goodwin, J.S.; Tangum, M.R. Battling quackery: Attitudes about micronutrient supplements in American Academic medicine. Arch. Intern. Med. 1998, 158, 2187–2191. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, J.S.; Goodwin, J.M. Failure to recognize efficacious treatments: A history of salicylate therapy in rheumatoid arthritis. Persp. Biol. Med. 1981, 31, 78–92. [Google Scholar] [CrossRef]
- Goodwin, J.S.; Goodwin, J.M. The tomato effect: Rejection of highly efficacious therapies. JAMA 1984, 251, 2387–2390. [Google Scholar] [CrossRef] [PubMed]
- Louhiala, P.; Hemilä, H. Can CAM treatments be evidence-based? Focus Altern. Complement. Ther. 2014, 19, 84–89. Available online: https://helda.helsinki.fi/handle/10138/228056 (accessed on 9 March 2019). [CrossRef]
| Trial [Reference] | Country | Settings | N | Route | Vitamin C Administration | Control Group Time | ||
|---|---|---|---|---|---|---|---|---|
| Dose (g/day) | Duration (days) | ICU Stay (days) | Ventilation (hours) | |||||
| Mirmohammadsadeghi 2018 [121] | Iran | Cardiac | 314 | iv | 1 | 4 | 2.2 | |
| Donovan 2012 [112] | USA | Cardiac | 304 | po | 2 | 5 | 1.9 | |
| Bjordahl 2012 [111] | USA | Cardiac | 185 | po | 2 | 5 | 4.3 | 33.6 |
| Papoulidis 2011 [110] | Greece | Cardiac | 170 | iv | 1 | 5 | 2.1 | |
| Sarzaeem 2014 [116] | Iran | Cardiac | 170 | iv | 1 | 5 | 3.0 | |
| Amini 2018 [120] | Iran | Cardiac | 138 | po | 3 | 2 | 2.3 | 6.7 |
| Antonic 2017 [119] | Slovenia | Cardiac | 105 | iv | 2 | 5 | 1.3 | |
| Alsfahey 2017 [118] | Egypt | Cardiac | 100 | po | 2 | 1 | 3.6 | |
| Dehghani 2014 [113] | Iran | Cardiac | 100 | po | 1 | 5 | 2.1 | 15.4 |
| Eslami 2007 [108] | Iran | Cardiac | 100 | po | 2 | 5 | 2.6 | |
| Dingchao 1994 [106] | China | Cardiac | 85 | iv | 17 * | 1 | 1.9 | |
| Abdoulhossein 2018 no vitE [122] | Iran | Lung contusion | 40 | iv | 0.5 | 2 | 5.2 | |
| Abdoulhossein 2018 vitE [122] | Iran | Lung contusion | 40 | iv | 0.5 | 2 | 5.2 | |
| Ebade 2014 [114] | Egypt | Cardiac | 40 | iv | 3 | 5 | 3.2 | 2.0 |
| Tanaka 2000 [107] | Japan | Burns | 37 | iv | 110 * | 1 | 511 | |
| Zabet 2016 [117] | Iran | Sepsis | 28 | iv | 7 * | 3 | 20.6 | 46.8 |
| Colby 2011 [109] | USA | Cardiac | 24 | po | 1 | 5 | 2.0 | |
| Fowler 2014 [115] | USA | Sepsis | 24 | iv | 3.5–14 * | 4 | 11 | |
| Selection of Trials | N Trials | N Patients | Estimate of Effect | Heterogeneity | |||
|---|---|---|---|---|---|---|---|
| RoM | 95% CI | p | I2 | p | |||
| All | 17 | 1967 | 0.835 | 0.81–0.86 | 10−26 | 90% | 10−24 |
| Exclusion1 (a) | 16 | 1882 | 0.908 | 0.87–0.94 | 10−6 | 58% | 0.002 |
| Exclusion2 (a) | 15 | 1842 | 0.923 | 0.89–0.96 | 0.00003 | 35% | 0.088 |
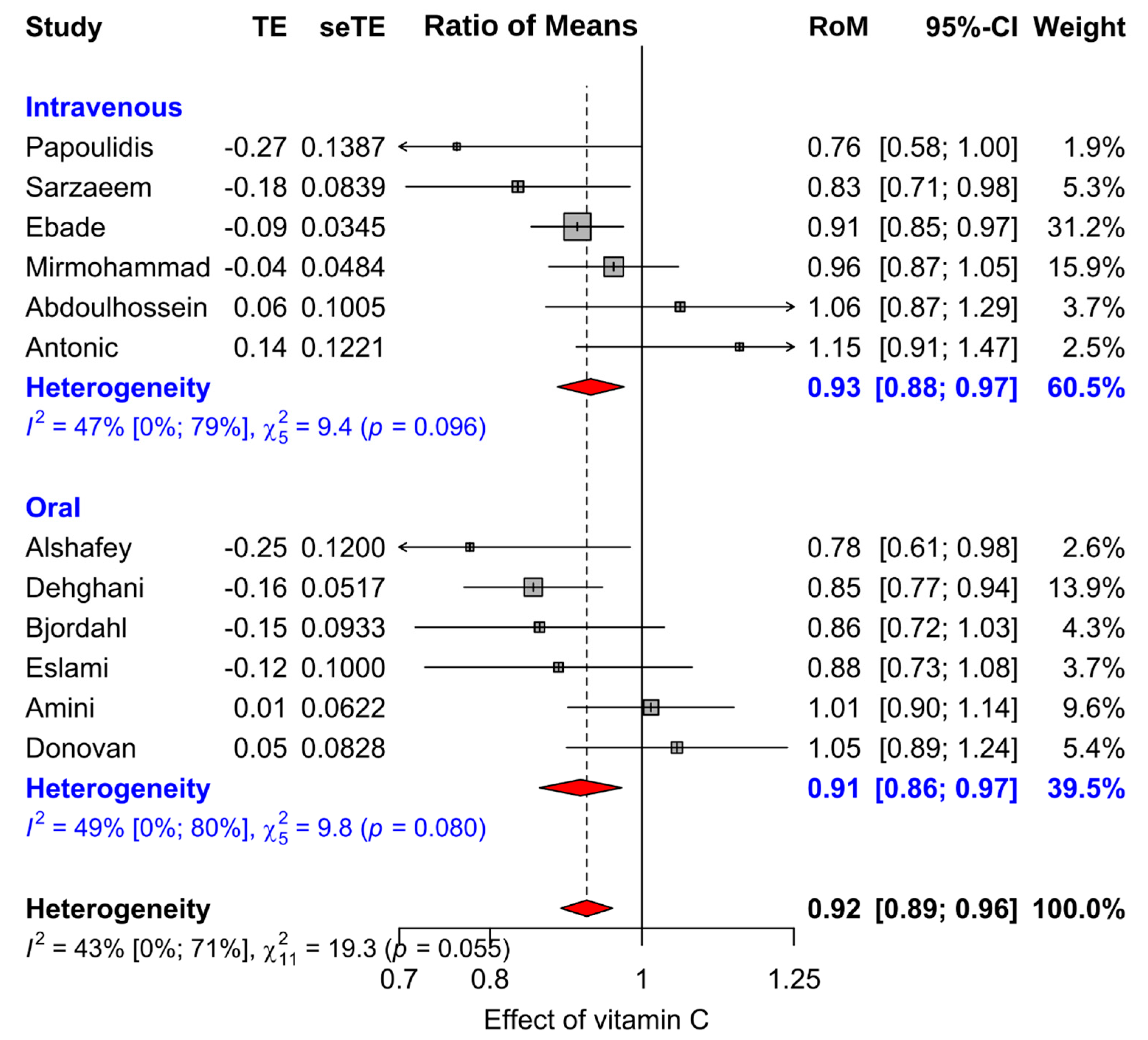
| Final meta-analysis (b) | 12 | 1766 | 0.922 | 0.89–0.96 | 0.00003 | 43% | 0.055 |
| Sensitivity analysis (c) | 9 | 1312 | 0.927 | 0.88–0.98 | 0.005 | 51% | 0.036 |
| Cardiac trials | 11 | 1726 | 0.918 | 0.88–0.95 | 0.00001 | 43% | 0.066 |
| Oral vitamin C | 6 | 927 | 0.914 | 0.86–0.97 | 0.003 | 49% | 0.08 |
| Intravenous vitamin C | 6 | 839 | 0.928 | 0.88–0.97 | 0.003 | 47% | 0.10 |
| Trials in Iran | 6 | 862 | 0.927 | 0.88–0.98 | 0.005 | 43% | 0.12 |
| Trials out of Iran | 6 | 904 | 0.917 | 0.87–0.97 | 0.002 | 53% | 0.06 |
| 1-2 days ICU (d) | 7 | 1231 | 0.943 | 0.89–0.99 | 0.027 | 52% | 0.05 |
| 3-5 days ICU (d) | 5 | 535 | 0.899 | 0.85–0.95 | 0.0001 | 23% | 0.3 |
| Vitamin E | Vitamin C | Difference (95% CI) | |
|---|---|---|---|
| No | Yes | ||
| No | 5.2 (1.67) | 5.5 (1.73) | +0.3 (−0.8 to +1.4) |
| Yes | 5.2 (1.74) | 3.5 (0.5) | −1.7 (−2.5 to −0.9) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hemilä, H.; Chalker, E. Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis. Nutrients 2019, 11, 708. https://doi.org/10.3390/nu11040708
Hemilä H, Chalker E. Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis. Nutrients. 2019; 11(4):708. https://doi.org/10.3390/nu11040708
Chicago/Turabian StyleHemilä, Harri, and Elizabeth Chalker. 2019. "Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis" Nutrients 11, no. 4: 708. https://doi.org/10.3390/nu11040708
APA StyleHemilä, H., & Chalker, E. (2019). Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis. Nutrients, 11(4), 708. https://doi.org/10.3390/nu11040708