The Golden Egg: Nutritional Value, Bioactivities, and Emerging Benefits for Human Health
Abstract
1. Introduction
2. Egg Nutrients
2.1. Macronutrients
2.1.1. Proteins
2.1.2. Lipids
2.1.3. Carbohydrates
2.2. Micronutrients
2.2.1. Vitamins and Choline
2.2.2. Minerals and Trace Elements
2.3. Antinutritional Factors
3. Egg Nutraceuticals
3.1. Antimicrobials
3.2. Antioxidant Activities
3.3. Anti-Cancerous Molecules
3.4. Immunomodulatory Activities
3.5. Antihypertensive Activities
4. Factors Affecting Egg Quality
4.1. Genetics
4.2. Nutrition and Rearing Systems
4.3. Physiological Status
4.4. Egg Storage and Heat Treatment
4.5. Variability between Avian Domestic Species
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- McNamara, D.J. The Fifty Year Rehabilitation of the Egg. Nutrients 2015, 7, 8716–8722. [Google Scholar] [CrossRef]
- Pelletier, X.; Thouvenot, P.; Belbraouet, S.; Chayvialle, J.A.; Hanesse, B.; Mayeux, D.; Debry, G. Effect of egg consumption in healthy volunteers: Influence of yolk, white or whole-egg on gastric emptying and on glycemic and hormonal responses. Ann. Nutr. Metab. 1996, 40, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Lopez Sobaler, A.M.; Aparicio Vizuete, A.; Ortega, R.M. Role of the egg in the diet of athletes and physically active people. Nutr. Hosp. 2017, 34, 31–35. [Google Scholar] [CrossRef]
- Kim, J.E.; Campbell, W.W. Dietary Cholesterol Contained in Whole Eggs Is Not Well Absorbed and Does Not Acutely Affect Plasma Total Cholesterol Concentration in Men and Women: Results from 2 Randomized Controlled Crossover Studies. Nutrients 2018, 10, 1272. [Google Scholar] [CrossRef]
- Papanikolaou, Y.; Fulgoni, V.L., 3rd. Egg Consumption in Infants is Associated with Longer Recumbent Length and Greater Intake of Several Nutrients Essential in Growth and Development. Nutrients 2018, 10, 719. [Google Scholar] [CrossRef]
- Missimer, A.; DiMarco, D.M.; Andersen, C.J.; Murillo, A.G.; Vergara-Jimenez, M.; Fernandez, M.L. Consuming Two Eggs per Day, as Compared to an Oatmeal Breakfast, Decreases Plasma Ghrelin while Maintaining the LDL/HDL Ratio. Nutrients 2017, 9, 89. [Google Scholar] [CrossRef] [PubMed]
- Stanciuc, N.; Banu, I.; Turturica, M.; Aprodu, I. pH and heat induced structural changes of chicken ovalbumin in relation with antigenic properties. Int. J. Biol. Macromol. 2016, 93, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Kuang, H.; Yang, F.; Zhang, Y.; Wang, T.; Chen, G. The Impact of Egg Nutrient Composition and Its Consumption on Cholesterol Homeostasis. Cholesterol 2018, 2018, 6303810. [Google Scholar] [CrossRef] [PubMed]
- Mine, Y.; Yang, M. Recent advances in the understanding of egg allergens: Basic, industrial, and clinical perspectives. J. Agric. Food Chem. 2008, 56, 4874–4900. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Pereira, B.; Voigt, K.; Grundy, J.; Clayton, C.B.; Higgins, B.; Arshad, S.H.; Dean, T. Prevalence and cumulative incidence of food hypersensitivity in the first 3 years of life. Allergy 2008, 63, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Osterballe, M.; Hansen, T.K.; Mortz, C.G.; Host, A.; Bindslev-Jensen, C. The prevalence of food hypersensitivity in an unselected population of children and adults. Pediatric Allergy Immunol. Off. Publ. Eur. Soc. Pediatric Allergy Immunol. 2005, 16, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.L.; Koplin, J.J.; Gurrin, L.C.; Dharmage, S.C.; Wake, M.; Ponsonby, A.L.; Tang, M.L.K.; Lowe, A.J.; Matheson, M.; Dwyer, T.; et al. The prevalence of food allergy and other allergic diseases in early childhood in a population-based study: HealthNuts age 4-year follow-up. J. Allergy Clin. Immunol. 2017, 140, 145–153.e148. [Google Scholar] [CrossRef] [PubMed]
- Nys, Y.; Sauveur, B. Valeur nutritionnelle des oeufs. INRA Prod. Anim. 2004, 17, 385–393. [Google Scholar]
- Drewnowski, A. The Nutrient Rich Foods Index helps to identify healthy, affordable foods. Am. J. Clin. Nutr. 2010, 91, 1095s–1101s. [Google Scholar] [CrossRef]
- Abeyrathne, E.; Ahn, D.U. Isolation of value-added components from egg white and their potential uses in food, nutraceutical and pharmaceutical industries. In Handbook of Eggs in Human Function; Watson, R.R., DeMeester, F., Eds.; Wageningen Acad Publ: Wageningen, The Netherlands, 2015; pp. 35–52. [Google Scholar]
- Kovacs-Nolan, J.; Phillips, M.; Mine, Y. Advances in the value of eggs and egg components for human health. J. Agric. Food Chem. 2005, 53, 8421–8431. [Google Scholar] [CrossRef] [PubMed]
- Anton, M.; Nau, F.; Guerin-Dubiard, C. Bioactive fractions of eggs for human and animal health. In Improving the Safety and Quality of Eggs and Egg Products, Vol 2: Egg Safety and Nutritional Quality; VanImmerseel, F., Nys, Y., Bain, M., Eds.; Woodhead Publ Ltd.: Cambridge, UK, 2011; pp. 321–345. [Google Scholar]
- Windhorst, H.-W.; Grabkowsky, B.; Wilke, A. Atlas of the Global Egg Industry; International Egg Commission: London, UK, 2015. [Google Scholar]
- Tukur, H.M. Egg production in Africa. In Improving the Safety of Eggs and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 1, pp. 27–38. [Google Scholar]
- Evenepoel, P.; Claus, D.; Geypens, B.; Hiele, M.; Geboes, K.; Rutgeerts, P.; Ghoos, Y. Amount and fate of egg protein escaping assimilation in the small intestine of humans. Am. J. Physiol. 1999, 277, G935–G943. [Google Scholar] [CrossRef] [PubMed]
- Ramalho, H.M.; Santos, V.V.; Medeiros, V.P.; Silva, K.H.; Dimenstein, R. Effect of thermal processing on retinol levels of free-range and caged hen eggs. Int. J. Food Sci. Nutr. 2006, 57, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Evenepoel, P.; Geypens, B.; Luypaerts, A.; Hiele, M.; Ghoos, Y.; Rutgeerts, P. Digestibility of cooked and raw egg protein in humans as assessed by stable isotope techniques. J. Nutr. 1998, 128, 1716–1722. [Google Scholar] [CrossRef]
- Stanciuc, N.; Cretu, A.A.; Banu, I.; Aprodu, I. Advances on the impact of thermal processing on structure and antigenicity of chicken ovomucoid. J. Sci. Food Agric. 2018, 98, 3119–3128. [Google Scholar] [CrossRef]
- Van der Plancken, I.; Van Remoortere, M.; Van Loey, A.; Hendrickx, M.E. Trypsin inhibition activity of heat-denatured ovomucoid: A kinetic study. Biotechnol. Prog. 2004, 20, 82–86. [Google Scholar] [CrossRef]
- Giansanti, F.; Massucci, M.T.; Giardi, M.F.; Nozza, F.; Pulsinelli, E.; Nicolini, C.; Botti, D.; Antonini, G. Antiviral activity of ovotransferrin derived peptides. Biochem. Biophys. Res. Commun. 2005, 331, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Ledesma, B.; Hsieh, C.C. Chemopreventive role of food-derived proteins and peptides: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 2358–2376. [Google Scholar] [CrossRef] [PubMed]
- Liao, W.; Fan, H.B.; Wu, J.P. Egg White-Derived Antihypertensive Peptide IRW (Ile-Arg-Trp) Inhibits Angiotensin II-Stimulated Migration of Vascular Smooth Muscle Cells via Angiotensin Type I Receptor. J. Agric. Food Chem. 2018, 66, 5133–5138. [Google Scholar] [CrossRef] [PubMed]
- Dave, L.A.; Hayes, M.; Montoya, C.A.; Rutherfurd, S.M.; Moughan, P.J. Human gut endogenous proteins as a potential source of angiotensin-I-converting enzyme (ACE-I)-, renin inhibitory and antioxidant peptides. Peptides 2016, 76, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Seuss-Baum, I.; Nau, F.; Guérin-Dubiard, C. The nutritional quality of eggs. In Improving the Safety and Quality of Egg and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2 Egg Safety and Nutrional Quality; pp. 201–236. [Google Scholar]
- USDA National Nutrient Database for Standard Reference, Release 1; U.S. Department of Agriculture. Food Group: Dairy and Egg Products: Beltsville, MD, USA, 2018.
- Nys, Y.; Guyot, N. Egg formation and chemistry. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 1, pp. 83–132. [Google Scholar]
- Mann, K. The chicken egg white proteome. Proteomics 2007, 7, 3558–3568. [Google Scholar] [CrossRef] [PubMed]
- Mann, K. Proteomic analysis of the chicken egg vitelline membrane. Proteomics 2008, 8, 2322–2332. [Google Scholar] [CrossRef] [PubMed]
- Mann, K.; Mann, M. The chicken egg yolk plasma and granule proteomes. Proteomics 2008, 8, 178–191. [Google Scholar] [CrossRef] [PubMed]
- Gautron, J.; Rehault-Godbert, S.; Nys, Y.; Mann, K.; Righetti, P.G. Use of high-throughput technology to identify new egg components. In Improving the Safety and Quality of Eggs and Egg Products, Vol 1: Egg Chemistry, Production and Consumption; Nys, Y., Bain, M., VanImmerseel, F., Eds.; Woodhead Publ Ltd.: Cambridge, UK, 2011; Volume 1, pp. 133–150. [Google Scholar]
- Gautron, J.; Nau, F.; Mann, K.; Guerin-Dubiard, C.; Rehault, S.; Hincke, M.T.; Nys, Y. Molecular approaches for the identification of novel egg components. Worlds Poult. Sci. J. 2007, 63, 82–90. [Google Scholar] [CrossRef]
- Farinazzo, A.; Restuccia, U.; Bachi, A.; Guerrier, L.; Fortis, F.; Boschetti, E.; Fasoli, E.; Citterio, A.; Righetti, P.G. Chicken egg yolk cytoplasmic proteome, mined via combinatorial peptide ligand libraries. J. Chromatogr. A 2009, 1216, 1241–1252. [Google Scholar] [CrossRef]
- Guerin-Dubiard, C.; Pasco, M.; Molle, D.; Desert, C.; Croguennec, T.; Nau, F. Proteomic analysis of hen egg white. J. Agric. Food Chem. 2006, 54, 3901–3910. [Google Scholar] [CrossRef] [PubMed]
- Mann, K.; Macek, B.; Olsen, J.V. Proteomic analysis of the acid-soluble organic matrix of the chicken calcified eggshell layer. Proteomics 2006, 6, 3801–3810. [Google Scholar] [CrossRef] [PubMed]
- Mann, K.; Olsen, J.V.; Macek, B.; Gnad, F.; Mann, M. Phosphoproteins of the chicken eggshell calcified layer. Proteomics 2007, 7, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Anton, M. Egg yolk: Structures, functionalities and processes. J. Sci. Food Agric. 2013, 93, 2871–2880. [Google Scholar] [CrossRef] [PubMed]
- Anton, M.; Gandemer, G. Composition, Solubility and Emulsifying Properties of Granules and Plasma of Egg Yolk. J. Food Sci. 1997, 62, 484–487. [Google Scholar] [CrossRef]
- Rehault-Godbert, S.; Guyot, N. Vitellogenesis and Yolk Proteins, Birds. In Encyclopedia of Reproduction; Skinner, M., Ed.; Elsevier: Amsterdam, The Netherlands; London, UK, 2018; Volume 6. [Google Scholar]
- Bellairs, R.; Harkness, M.; Harkness, R.D. The vitelline membrane of the hen’s egg: A chemical and electron microcopical study. J. Ultrastruct. Res. 1963, 8, 339–359. [Google Scholar] [CrossRef]
- Back, J.F.; Bain, J.M.; Vadehra, D.V.; Burley, R.W. Proteins of the outer layer of the vitelline membrane of hen’s eggs. Biochim. Et Biophys. Acta 1982, 705, 12–19. [Google Scholar] [CrossRef]
- Hawthorne, J.R. The action of egg white lysozyme on ovomucoid and ovomucin. Biochim. Et Biophys. Acta 1950, 6, 28–35. [Google Scholar] [CrossRef]
- Saxena, I.; Tayyab, S. Protein proteinase inhibitors from avian egg whites. Cell. Mol. Life Sci. CMLS 1997, 53, 13–23. [Google Scholar] [CrossRef]
- Trziszka, T.; Smolinska, T. Chemical characterization of the vitelline membrane of Hens eggs. Food Chem. 1982, 8, 61–70. [Google Scholar] [CrossRef]
- Shinn, S.E.; Liyanage, R.; Lay, J.O., Jr.; Proctor, A. Isolation and Characterization of Chicken Yolk Vitelline Membrane Lipids Using Eggs Enriched with Conjugated Linoleic Acid. Lipids 2016, 51, 769–779. [Google Scholar] [CrossRef]
- Hayes, K.C.; Khosla, P. Dietary fatty acid thresholds and cholesterolemia. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 1992, 6, 2600–2607. [Google Scholar] [CrossRef]
- Pronczuk, A.; Khosla, P.; Hayes, K.C. Dietary myristic, palmitic, and linoleic acids modulate cholesterolemia in gerbils. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 1994, 8, 1191–1200. [Google Scholar] [CrossRef]
- Chatterjee, I.B. Evolution and the biosynthesis of ascorbic acid. Science 1973, 182, 1271–1272. [Google Scholar] [CrossRef]
- Patterson, K.Y.; Bhagwat, S.A.; Williams, J.R.; Howe, J.C.; Holden, J.M. USDA Database for the Choline Content of Common Foods, Release 2; Nutrient Data Laboratory: Beltsville, MD, USA, 2008. [Google Scholar]
- Wiedeman, A.M.; Barr, S.I.; Green, T.J.; Xu, Z.; Innis, S.M.; Kitts, D.D. Dietary Choline Intake: Current State of Knowledge Across the Life Cycle. Nutrients 2018, 10, 1513. [Google Scholar] [CrossRef]
- Wallace, T.C.; Fulgoni, V.L. Usual Choline Intakes Are Associated with Egg and Protein Food Consumption in the United States. Nutrients 2017, 9, 839. [Google Scholar] [CrossRef]
- Leermakers, E.T.; Moreira, E.M.; Kiefte-de Jong, J.C.; Darweesh, S.K.; Visser, T.; Voortman, T.; Bautista, P.K.; Chowdhury, R.; Gorman, D.; Bramer, W.M.; et al. Effects of choline on health across the life course: A systematic review. Nutr. Rev. 2015, 73, 500–522. [Google Scholar] [CrossRef]
- Oyen, J.; Gjesdal, C.G.; Karlsson, T.; Svingen, G.F.; Tell, G.S.; Strand, E.; Drevon, C.A.; Vinknes, K.J.; Meyer, K.; Ueland, P.M.; et al. Dietary Choline Intake Is Directly Associated with Bone Mineral Density in the Hordaland Health Study. J. Nutr. 2017, 147, 572–578. [Google Scholar] [CrossRef]
- Staggs, C.G.; Sealey, W.M.; McCabe, B.J.; Teague, A.M.; Mock, D.M. Determination of the biotin content of select foods using accurate and sensitive HPLC/avidin binding. J. Food Compos. Anal. Off. Publ. U. N. Univ. Int. Netw. Food Data Syst. 2004, 17, 767–776. [Google Scholar] [CrossRef]
- Wang, J.; Um, P.; Dickerman, B.A.; Liu, J. Zinc, Magnesium, Selenium and Depression: A Review of the Evidence, Potential Mechanisms and Implications. Nutrients 2018, 10, 584. [Google Scholar] [CrossRef]
- Livnah, O.; Bayer, E.A.; Wilchek, M.; Sussman, J.L. Three-dimensional structures of avidin and the avidin-biotin complex. Proc. Natl. Acad. Sci. USA 1993, 90, 5076–5080. [Google Scholar] [CrossRef]
- Chang, C.; Lahti, T.; Tanaka, T.; Nickerson, M.T. Egg proteins: Fractionation, bioactive peptides and allergenicity. J. Sci. Food Agric. 2018, 98, 5547–5558. [Google Scholar] [CrossRef]
- Abeyrathne, E.D.; Lee, H.Y.; Ahn, D.U. Egg white proteins and their potential use in food processing or as nutraceutical and pharmaceutical agents—A review. Poult. Sci. 2013, 92, 3292–3299. [Google Scholar] [CrossRef] [PubMed]
- Andersen, C.J. Bioactive Egg Components and Inflammation. Nutrients 2015, 7, 7889–7913. [Google Scholar] [CrossRef] [PubMed]
- Giansanti, F.; Leboffe, L.; Angelucci, F.; Antonini, G. The Nutraceutical Properties of Ovotransferrin and Its Potential Utilization as a Functional Food. Nutrients 2015, 7, 9105–9115. [Google Scholar] [CrossRef] [PubMed]
- Nongonierma, A.B.; FitzGerald, R.J. Strategies for the discovery and identification of food protein-derived biologically active peptides. Trends Food Sci. Technol. 2017, 69, 289–305. [Google Scholar] [CrossRef]
- Arena, S.; Scaloni, A. An Extensive Description of the Peptidomic Repertoire of the Hen Egg Yolk Plasma. J. Agric. Food Chem. 2018, 66, 3239–3255. [Google Scholar] [CrossRef]
- Vilcacundo, R.; Mendez, P.; Reyes, W.; Romero, H.; Pinto, A.; Carrillo, W. Antibacterial Activity of Hen Egg White Lysozyme Denatured by Thermal and Chemical Treatments. Sci. Pharm. 2018, 86, 48. [Google Scholar] [CrossRef]
- Rehault-Godbert, S.; Herve-Grepinet, V.; Gautron, J.; Cabau, C.; Nys, Y.; Hincke, M. Molecules involved in chemical defence of the chicken egg. In Improving the Safety and Quality of Eggs and Egg Products, Vol 1: Egg Chemistry, Production and Consumption; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publ Ltd.: Cambridge, UK, 2011; Volume 1, pp. 183–208. [Google Scholar]
- Da Silva, M.; Dombre, C.; Brionne, A.; Monget, P.; Chesse, M.; De Pauw, M.; Mills, M.; Combes-Soia, L.; Labas, V.; Guyot, N.; et al. The unique features of proteins depicting the chicken amniotic fluid. Mol. Cell. Proteom. MCP 2018. [Google Scholar] [CrossRef] [PubMed]
- Mine, Y.; Ma, F.P.; Lauriau, S. Antimicrobial peptides released by enzymatic hydrolysis of hen egg white lysozyme. J. Agric. Food Chem. 2004, 52, 1088–1094. [Google Scholar] [CrossRef]
- Ibrahim, H.R.; Thomas, U.; Pellegrini, A. A helix-loop-helix peptide at the upper lip of the active site cleft of lysozyme confers potent antimicrobial activity with membrane permeabilization action. J. Biol. Chem. 2001, 276, 43767–43774. [Google Scholar] [CrossRef]
- Pellegrini, A.; Thomas, U.; Wild, P.; Schraner, E.; von Fellenberg, R. Effect of lysozyme or modified lysozyme fragments on DNA and RNA synthesis and membrane permeability of Escherichia coli. Microbiol. Res. 2000, 155, 69–77. [Google Scholar] [CrossRef]
- Pellegrini, A.; Thomas, U.; Bramaz, N.; Klauser, S.; von Fellenberg, R. Identification and isolation of a bactericidal domain in chicken egg white lysozyme. J. Appl. Microbiol. 1997, 82, 372–378. [Google Scholar] [CrossRef]
- Omana, D.A.; Wang, J.P.; Wu, J.P. Ovomucin—A glycoprotein with promising potential. Trends Food Sci. Technol. 2010, 21, 455–463. [Google Scholar] [CrossRef]
- Blankenvoorde, M.F.J.; van’t Hof, W.; Walgreen-Weterings, E.; van Steenbergen, T.J.M.; Brand, H.S.; Veerman, E.C.I.; Amerongen, A.V.N. Cystatin and cystatin-derived peptides have antibacterial activity against the pathogen Porphyromonas gingivalis. Biol. Chem. 1998, 379, 1371–1375. [Google Scholar]
- Guyot, N.; Labas, V.; Harichaux, G.; Chesse, M.; Poirier, J.C.; Nys, Y.; Rehault-Godbert, S. Proteomic analysis of egg white heparin-binding proteins: Towards the identification of natural antibacterial molecules. Sci. Rep. 2016, 6, 27974. [Google Scholar] [CrossRef]
- Herve-Grepinet, V.; Rehault-Godbert, S.; Labas, V.; Magallon, T.; Derache, C.; Lavergne, M.; Gautron, J.; Lalmanach, A.C.; Nys, Y. Purification and Characterization of Avian beta-Defensin 11, an Antimicrobial Peptide of the Hen Egg. Antimicrob. Agents Chemother. 2010, 54, 4401–4408. [Google Scholar] [CrossRef]
- Korpela, J.; Salonen, E.M.; Kuusela, P.; Sarvas, M.; Vaheri, A. Binding of avidin to bacteria and to the outer-membrane porin of Escherichia coli. FEMS Microbiol. Lett. 1984, 22, 3–10. [Google Scholar] [CrossRef]
- Krkavcova, E.; Kreisinger, J.; Hyankova, L.; Hyrsl, P.; Javurkova, V. The hidden function of egg white antimicrobials: Egg weight-dependent effects of avidin on avian embryo survival and hatchling phenotype. Biol. Open 2018, 7, 9. [Google Scholar] [CrossRef]
- Kolaczkowska, A.; Kolaczkowski, M.; Sokolowska, A.; Miecznikowska, H.; Kubiak, A.; Rolka, K.; Polanowski, A. The antifungal properties of chicken egg cystatin against Candida yeast isolates showing different levels of azole resistance. Mycoses 2010, 53, 314–320. [Google Scholar] [CrossRef]
- Wesierska, E.; Saleh, Y.; Trziszka, T.; Kopec, W.; Siewinski, M.; Korzekwa, K. Antimicrobial activity of chicken egg white cystatin. World J. Microbiol. Biotechnol. 2005, 21, 59–64. [Google Scholar] [CrossRef]
- Bjorck, L. Proteinase inhibition, immunoglobulin-binding proteins and a novel antimicrobial principle. Mol. Microbiol. 1990, 4, 1439–1442. [Google Scholar] [CrossRef] [PubMed]
- Korant, B.D.; Brzin, J.; Turk, V. Cystatin, a protein inhibitor of cysteine proteases alters viral protein cleavages in infected human cells. Biochem. Biophys. Res. Commun. 1985, 127, 1072–1076. [Google Scholar] [CrossRef]
- Ebina, T.; Tsukada, K. Protease inhibitors prevent the development of human rotavirus-induced diarrhea in suckling mice. Microbiol. Immunol. 1991, 35, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Lima, A.P.; dos Reis, F.C.; Serveau, C.; Lalmanach, G.; Juliano, L.; Menard, R.; Vernet, T.; Thomas, D.Y.; Storer, A.C.; Scharfstein, J. Cysteine protease isoforms from Trypanosoma cruzi, cruzipain 2 and cruzain, present different substrate preference and susceptibility to inhibitors. Mol. Biochem. Parasitol. 2001, 114, 41–52. [Google Scholar] [CrossRef]
- Yu, L.T.; Xiao, Y.P.; Li, J.J.; Ran, J.S.; Yin, L.Q.; Liu, Y.P.; Zhang, L. Molecular characterization of a novel ovodefensin gene in chickens. Gene 2018, 678, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Whenham, N.; Lu, T.C.; Maidin, M.B.M.; Wilson, P.W.; Bain, M.M.; Stevenson, M.L.; Stevens, M.P.; Bedford, M.R.; Dunn, I.C. Ovodefensins, an Oviduct-Specific Antimicrobial Gene Family, Have Evolved in Birds and Reptiles to Protect the Egg by Both Sequence and Intra-Six-Cysteine Sequence Motif Spacing. Biol. Reprod. 2015, 92, 13. [Google Scholar] [CrossRef]
- Herve, V.; Meudal, H.; Labas, V.; Rehault-Godbert, S.; Gautron, J.; Berges, M.; Guyot, N.; Delmas, A.F.; Nys, Y.; Landon, C. Three-dimensional NMR Structure of Hen Egg Gallin (Chicken Ovodefensin) Reveals a New Variation of the beta-Defensin Fold. J. Biol. Chem. 2014, 289, 7211–7220. [Google Scholar] [CrossRef]
- Gong, D.Q.; Wilson, P.W.; Bain, M.M.; McDade, K.; Kalina, J.; Herve-Grepinet, V.; Nys, Y.; Dunn, I.C. Gallin; an antimicrobial peptide member of a new avian defensin family, the ovodefensins, has been subject to recent gene duplication. BMC Immunol. 2010, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Kovacs-Nolan, J.; Mine, Y. Egg Yolk Antibodies for Passive Immunity. In Annual Review of Food Science and Technology, Volume 3; Doyle, M.P., Klaenhammer, T.R., Eds.; Annual Reviews: Palo Alto, CA, USA, 2012; Volume 3, pp. 163–182. [Google Scholar]
- Wellman-Labadie, O.; Picman, J.; Hincke, M.T. Comparative antibacterial activity of avian egg white protein extracts. Br. Poult. Sci. 2008, 49, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Samaranayake, Y.H.; Samaranayake, L.P.; Pow, E.H.N.; Beena, V.T.; Yeung, K.W.S. Antifungal effects of lysozyme and lactoferrin against genetically similar, sequential Candida albicans isolates from a human immunodeficiency virus-infected southern Chinese cohort. J. Clin. Microbiol. 2001, 39, 3296–3302. [Google Scholar] [CrossRef]
- Rehault-Godbert, S.; Labas, V.; Helloin, E.; Herve-Grepinet, V.; Slugocki, C.; Berges, M.; Bourin, M.C.; Brionne, A.; Poirier, J.C.; Gautron, J.; et al. Ovalbumin-related protein X is a heparin-binding ov-serpin exhibiting antimicrobial activities. J. Biol. Chem. 2013, 288, 17285–17295. [Google Scholar] [CrossRef]
- Maehashi, K.; Ueda, M.; Matano, M.; Takeuchi, J.; Uchino, M.; Kashiwagi, Y.; Watanabe, T. Biochemical and functional characterization of transiently expressed in neural precursor (TENP) protein in emu egg white. J. Agric. Food Chem. 2014, 62, 5156–5162. [Google Scholar] [CrossRef]
- Whenham, N.; Wilson, P.W.; Bain, M.M.; Stevenson, L.; Dunn, I.C. Comparative biology and expression of TENP, an egg protein related to the bacterial permeability-increasing family of proteins. Gene 2014, 538, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Malicka-Blaszkiewicz, M.; Filipczak, N.; Golab, K.; Juszczynska, K.; Sebzda, T.; Gburek, J. Ovocystatin affects actin cytoskeleton organization and induces proapoptotic activity. Acta Biochim. Pol. 2014, 61, 753–758. [Google Scholar] [PubMed]
- Bourin, M.; Gautron, J.; Berges, M.; Attucci, S.; Le Blay, G.; Labas, V.; Nys, Y.; Rehault-Godbert, S. Antimicrobial Potential of Egg Yolk Ovoinhibitor, a Multidomain Kazal-like Inhibitor of Chicken Egg. J. Agric. Food Chem. 2011, 59, 12368–12374. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.H.; Lee, J.H.; Lee, Y.J.; Paik, J.Y.; Ahn, D.U.; Paik, H.D. Antioxidant, Antimicrobial, and Cytotoxic Activities of Ovotransferrin from Egg White. Korean J. Food Sci. 2012, 32, 612–617. [Google Scholar] [CrossRef]
- Wu, J.P.; Acero-Lopez, A. Ovotransferrin: Structure, bioactivities, and preparation. Food Res. Int. 2012, 46, 480–487. [Google Scholar] [CrossRef]
- Giansanti, F.; Giardi, M.F.; Massucci, M.T.; Botti, D.; Antonini, G. Ovotransferrin expression and release by chicken cell lines infected with Marek’s disease virus. Biochem. Cell Biol. 2007, 85, 150–155. [Google Scholar] [CrossRef]
- Khan, M.A.S.; Nakamura, S.; Ogawa, M.; Akita, E.; Azakami, H.; Kato, A. Bactericidal action of egg yolk phosvitin against Escherichia coli under thermal stress. J. Agric. Food Chem. 2000, 48, 1503–1506. [Google Scholar] [CrossRef]
- Svensson, S.L.; Pasupuleti, M.; Walse, B.; Malmsten, M.; Morgelin, M.; Sjogren, C.; Olin, A.I.; Collin, M.; Schmidtchen, A.; Palmer, R.; et al. Midkine and pleiotrophin have bactericidal properties: Preserved antibacterial activity in a family of heparin-binding growth factors during evolution. J. Biol. Chem. 2010, 285, 16105–16115. [Google Scholar] [CrossRef]
- Sun, S.; Niu, H.; Yang, T.; Lin, Q.; Luo, F.; Ma, M. Antioxidant and anti-fatigue activities of egg white peptides prepared by pepsin digestion. J. Sci. Food Agric. 2014, 94, 3195–3200. [Google Scholar] [CrossRef] [PubMed]
- Abeyrathne, E.; Huang, X.; Ahn, D.U. Antioxidant, angiotensin-converting enzyme inhibitory activity and other functional properties of egg white proteins and their derived peptides—A review. Poult. Sci. 2018, 97, 1462–1468. [Google Scholar] [CrossRef]
- Nimalaratne, C.; Bandara, N.; Wu, J.P. Purification and characterization of antioxidant peptides from enzymatically hydrolyzed chicken egg white. Food Chem. 2015, 188, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Nimalaratne, C.; Wu, J. Hen Egg as an Antioxidant Food Commodity: A Review. Nutrients 2015, 7, 8274–8293. [Google Scholar] [CrossRef]
- Yi, J.E.; Zhao, J.; Wu, J.P. Egg ovotransferrin derived IRW exerts protective effect against H2O2-induced oxidative stress in Caco-2 cells. J. Funct. Foods 2017, 39, 160–167. [Google Scholar] [CrossRef]
- Shen, S.W.; Chahal, B.; Majumder, K.; You, S.J.; Wu, J.P. Identification of Novel Antioxidative Peptides Derived from a Thermolytic Hydrolysate of Ovotransferrin by LC-MS/MS. J. Agric. Food Chem. 2010, 58, 7664–7672. [Google Scholar] [CrossRef]
- Kim, J.; Moon, S.H.; Ahn, D.U.; Paik, H.D.; Park, E. Antioxidant effects of ovotransferrin and its hydrolysates. Poult. Sci. 2012, 91, 2747–2754. [Google Scholar] [CrossRef]
- Chen, C.; Chi, Y.J.; Zhao, M.Y.; Lv, L. Purification and identification of antioxidant peptides from egg white protein hydrolysate. Amino Acids 2012, 43, 457–466. [Google Scholar] [CrossRef]
- Abeyrathne, E.; Lee, H.Y.; Jo, C.; Suh, J.W.; Ahn, D.U. Enzymatic hydrolysis of ovomucoid and the functional properties of its hydrolysates. Poult. Sci. 2015, 94, 2280–2287. [Google Scholar] [CrossRef]
- Chang, O.K.; Ha, G.E.; Han, G.S.; Seol, K.H.; Kim, H.W.; Jeong, S.G.; Oh, M.H.; Park, B.Y.; Ham, J.S. Novel Antioxidant Peptide Derived from the Ultrafiltrate of Ovomucin Hydrolysate. J. Agric. Food Chem. 2013, 61, 7294–7300. [Google Scholar] [CrossRef]
- Yousr, M.; Howell, N. Antioxidant and ACE Inhibitory Bioactive Peptides Purified from Egg Yolk Proteins. Int. J. Mol. Sci. 2015, 16, 29161–29178. [Google Scholar] [CrossRef]
- Young, D.; Fan, M.Z.; Mine, Y. Egg Yolk Peptides Up-regulate Glutathione Synthesis and Antioxidant Enzyme Activities in a Porcine Model of Intestinal Oxidative Stress. J. Agric. Food Chem. 2010, 58, 7624–7633. [Google Scholar] [CrossRef]
- Sava, G. Reduction of B16 melanoma metastases by oral administration of egg-white lysozyme. Cancer Chemother. Pharmacol. 1989, 25, 221–222. [Google Scholar] [CrossRef]
- Ibrahim, H.R.; Kiyono, T. Novel Anticancer Activity of the Autocleaved Ovotransferrin against Human Colon and Breast Cancer Cells. J. Agric. Food Chem. 2009, 57, 11383–11390. [Google Scholar] [CrossRef]
- Clemente, A.; Arques Mdel, C. Bowman-Birk inhibitors from legumes as colorectal chemopreventive agents. World J. Gastroenterol. 2014, 20, 10305–10315. [Google Scholar] [CrossRef]
- Lee, M.; Kovacs-Nolan, J.; Yang, C.; Archbold, T.; Fan, M.Z.; Mine, Y. Hen egg lysozyme attenuates inflammation and modulates local gene expression in a porcine model of dextran sodium sulfate (DSS)-induced colitis. J. Agric. Food Chem. 2009, 57, 2233–2240. [Google Scholar] [CrossRef]
- Sorrelle, N.; Dominguez, A.T.A.; Brekken, R.A. From top to bottom: Midkine and pleiotrophin as emerging players in immune regulation. J. Leukoc. Biol. 2017, 102, 277–286. [Google Scholar] [CrossRef]
- Achour, A.; M’Bika, J.P.; Baudouin, F.; Caruelle, D.; Courty, J. Pleiotrophin induces expression of inflammatory cytokines in peripheral blood mononuclear cells. Biochimie 2008, 90, 1791–1795. [Google Scholar] [CrossRef]
- Liu, L.; Xu, M.S.; Tu, Y.G.; Du, H.Y.; Zhou, Y.L.; Zhu, G.X. Immunomodulatory effect of protease hydrolysates from ovotransferrin. Food Funct. 2017, 8, 1452–1459. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, Y.; Yao, Y.; Xu, M.S.; Du, H.Y.; Zhang, M.Y.; Tu, Y.G. Anti-inflammatory activity of di-peptides derived from ovotransferrin by CrossMark simulated peptide-cut in TNF-alpha-induced Caco-2 cells. J. Funct. Foods 2017, 37, 424–432. [Google Scholar] [CrossRef]
- Kearney, P.M.; Whelton, M.; Reynolds, K.; Whelton, P.K.; He, J. Worldwide prevalence of hypertension: A systematic review. J. Hypertens. 2004, 22, 11–19. [Google Scholar] [CrossRef]
- Yoshii, H.; Tachi, N.; Ohba, R.; Sakamura, O.; Takeyama, H.; Itani, T. Antihypertensive effect of ACE inhibitory oligopeptides from chicken egg yolks. Comp. Biochem. Physiol. C-Toxicol. Pharmacol. 2001, 128, 27–33. [Google Scholar] [CrossRef]
- Moon, S.H.; Lee, J.H.; Kim, J.H.; Paik, H.D.; Ahn, D.U. In vitro cytotoxic and ACE-inhibitory activities of promod 278P hydrolysate of ovotransferrin from chicken egg white. Poult. Sci. 2017, 96, 1982–1987. [Google Scholar] [CrossRef]
- Majumder, K.; Wu, J.P. Purification and characterisation of angiotensin I converting enzyme (ACE) inhibitory peptides derived from enzymatic hydrolysate of ovotransferrin. Food Chem. 2011, 126, 1614–1619. [Google Scholar] [CrossRef]
- Liao, W.; Chakrabarti, S.; Davidge, S.T.; Wu, J.P. Modulatory Effects of Egg White Ovotransferrin-Derived Tripeptide IRW (Ile-Arg-Trp) on Vascular Smooth Muscle Cells against Angiotensin II Stimulation. J. Agric. Food Chem. 2016, 64, 7342–7347. [Google Scholar] [CrossRef]
- Majumder, K.; Chakrabarti, S.; Morton, J.S.; Panahi, S.; Kaufman, S.; Davidge, S.T.; Wu, J.P. Egg-derived ACE-inhibitory peptides IQW and LKP reduce blood pressure in spontaneously hypertensive rats. J. Funct. Foods 2015, 13, 50–60. [Google Scholar] [CrossRef]
- Chen, S.; Jiang, H.M.; Peng, H.H.; Wu, X.S.; Fang, J. The Utility of Ovotransferrin and Ovotransferrin-Derived Peptides as Possible Candidates in the Clinical Treatment of Cardiovascular Diseases. Oxid. Med. Cell. Longev. 2017. [Google Scholar] [CrossRef]
- Dunn, I.C. Breeding strategies to improve the egg’s natural defence. Worlds Poult. Sci. J. 2004, 60, 458–468. [Google Scholar] [CrossRef]
- Dunn, I.C. Poultry breeding for egg quality. In Improving the Safety and Quality of Eggs and Egg Products, Volume 1: Egg Chemistry, Production and Consumption; Nys, Y., Bain, M., VanImmerseel, F., Eds.; Woodhead Publ Ltd.: Cambridge, UK, 2011; Volume 1, pp. 245–260. [Google Scholar]
- Bilkova, B.; Swiderska, Z.; Zita, L.; Laloe, D.; Charles, M.; Benes, V.; Stopka, P.; Vinkler, M. Domestic Fowl Breed Variation in Egg White Protein Expression: Application of Proteomics and Transcriptomics. J. Agric. Food Chem. 2018, 66, 11854–11863. [Google Scholar] [CrossRef]
- Hocking, P.M.; Bain, M.; Channing, C.E.; Fleming, R.; Wilson, S. Genetic variation for egg production, egg quality and bone strength in selected and traditional breeds of laying fowl. Br. Poult. Sci. 2003, 44, 365–373. [Google Scholar] [CrossRef]
- Sellier, N.; Vidal, M.L.; Baron, F.; Michel, J.; Gautron, J.; Protais, M.; Beaumont, C.; Gautier, M.; Nys, Y. Estimations of repeatability and heritability of egg albumen antimicrobial activity and of lysozyme and ovotransferrin concentrations. Br. Poult. Sci. 2007, 48, 559–566. [Google Scholar] [CrossRef]
- Anderson, K.E. Comparison of fatty acid, cholesterol, vitamin A and E composition, and trans fats in eggs from brown and white egg strains that were molted or nonmolted. Poult. Sci. 2013, 92, 3259–3265. [Google Scholar] [CrossRef]
- Silversides, F.G.; Budgell, K. The relationships among measures of egg albumen height, pH, and whipping volume. Poult. Sci. 2004, 83, 1619–1623. [Google Scholar] [CrossRef]
- Bouvarel, I.; Nys, Y.; Lescoat, P. Hen nutrition for sustained egg quality. In Improving the Safety and Quality of Eggs and Egg Products, Vol 1: Egg Chemistry, Production and Consumption; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publ Ltd.: Cambridge, UK, 2011; pp. 261–299. [Google Scholar]
- Nys, Y.; Le Roy, N. Calcium Homeostasis and Eggshell Biomineralization in Female Chicken. In Vitamin D, 4th ed.; Feldman, D., Ed.; Academic Press: London, UK, 2018; Volume 1, pp. 361–382. [Google Scholar]
- Grobas, S.; Mendez, J.; De Blas, C.; Mateos, G.G. Influence of dietary energy, supplemental fat and linoleic acid concentration on performance of laying hens at two ages. Br. Poult. Sci. 1999, 40, 681–687. [Google Scholar] [CrossRef]
- Bregendahl, K.; Roberts, S.A.; Kerr, B.; Hoehler, D. Ideal ratios of isoleucine, methionine, methionine plus cystine, threonine, tryptophan, and valine relative to lysine for white leghorn-type laying hens of twenty-eight to thirty-four weeks of age. Poult. Sci. 2008, 87, 744–758. [Google Scholar] [CrossRef]
- Recoules, E.; Sabboh-Jourdan, H.; Narcy, A.; Lessire, M.; Harichaux, G.; Labas, V.; Duclos, M.J.; Rehault-Godbert, S. Exploring the in vivo digestion of plant proteins in broiler chickens. Poult. Sci. 2017, 96, 1735–1747. [Google Scholar] [CrossRef]
- Nain, S.; Renema, R.A.; Korver, D.R.; Zuidhof, M.J. Characterization of the n-3 polyunsaturated fatty acid enrichment in laying hens fed an extruded flax enrichment source. Poult. Sci. 2012, 91, 1720–1732. [Google Scholar] [CrossRef]
- Coorey, R.; Novinda, A.; Williams, H.; Jayasena, V. Omega-3 fatty acid profile of eggs from laying hens fed diets supplemented with chia, fish oil, and flaxseed. J. Food Sci. 2015, 80, S180–S187. [Google Scholar] [CrossRef]
- Sirri, F.; Meluzzi, A. Modifying egg lipids for human health. In Improving the Safety and Quality of Eggs and Egg Products; Van Immerseel, F., Nys, Y., Bain, M., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 272–288. [Google Scholar]
- Baeza, E.; Chartrin, P.; Lessire, M.; Meteau, K.; Chesneau, G.; Guillevic, M.; Mourot, J. Is it possible to increase the n-3 fatty acid content of eggs without affecting their technological and/or sensorial quality and the laying performance of hens? Br. Poult. Sci. 2015, 56, 748–754. [Google Scholar] [CrossRef]
- Grcevic, M.; Kralik, Z.; Kralik, G.; Galovic, O. Effects of dietary marigold extract on lutein content, yolk color and fatty acid profile of omega-3 eggs. J. Sci. Food Agric. 2019, 99, 2292–2299. [Google Scholar] [CrossRef]
- Park, J.H.; Upadhaya, S.D.; Kim, I.H. Effect of dietary marine microalgae (schizochytrium) powder on egg production, blood lipid profiles, egg quality, and Fatty Acid composition of egg yolk in layers. Asian-Australas. J. Anim. Sci. 2015, 28, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.G.; Sieo, C.C.; Kalavathy, R.; Saad, W.Z.; Yong, S.T.; Wong, H.K.; Ho, Y.W. Chemical Compositions of Egg Yolks and Egg Quality of Laying Hens Fed Prebiotic, Probiotic, and Synbiotic Diets. J. Food Sci. 2015, 80, C1686–C1695. [Google Scholar] [CrossRef] [PubMed]
- Schiavone, A.; Barroeta, A.C. Egg enrichment with vitamins and trace minerals. In Improving the Safety and Quality of Eggs and Egg Products; Van Immerseel, F., Nys, Y., Bain, M., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 289–320. [Google Scholar]
- Nys, Y.; Schlegel, P.; Durosoy, S.; Jondreville, C.; Narcy, A. Adapting trace mineral nutrition of birds for optimising the environment and poultry product quality. Worlds Poult. Sci. J. 2018, 74, 225–238. [Google Scholar] [CrossRef]
- Ranard, K.M.; Jeon, S.; Mohn, E.S.; Griffiths, J.C.; Johnson, E.J.; Erdman, J.W., Jr. Dietary guidance for lutein: Consideration for intake recommendations is scientifically supported. Eur. J. Nutr. 2017, 56, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Pignoli, G.; Rodriguez-Estrada, M.T.; Mandrioli, M.; Barbanti, L.; Rizzi, L.; Lercker, G. Effects of different rearing and feeding systems on lipid oxidation and antioxidant capacity of freeze-dried egg yolks. J. Agric. Food Chem. 2009, 57, 11517–11527. [Google Scholar] [CrossRef] [PubMed]
- Samiullah; Roberts, J.R.; Chousalkar, K.K. Effect of production system and flock age on egg quality and total bacterial load in commercial laying hens. J. Appl. Poult. Res. 2014, 23, 59–70. [Google Scholar] [CrossRef]
- Bedrani, L.; Helloin, E.; Guyot, N.; Rehault-Godbert, S.; Nys, Y. Passive maternal exposure to environmental microbes selectively modulates the innate defences of chicken egg white by increasing some of its antibacterial activities. BMC Microbiol. 2013, 13, 13. [Google Scholar] [CrossRef] [PubMed]
- Travel, A.; Nys, Y.; Bain, M. Effect of hens age, moult, laying environment and egg storage on egg quality. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 1, pp. 300–329. [Google Scholar]
- Rossi, M.; Pompei, C. Changes in some egg components and analytical values due to hen age. Poult. Sci. 1995, 74, 152–160. [Google Scholar] [CrossRef]
- Ngoka, D.A.; Froning, G.W.; Babji, A.S. Effect of temperature on egg-yolk characteristics of eggs from young and old laying hens. Poult. Sci. 1983, 62, 718–720. [Google Scholar] [CrossRef]
- Cherian, G. Egg quality and yolk polyunsaturated fatty acid status in relation to broiler breeder hen age and dietary n-3 oils. Poult. Sci. 2008, 87, 1131–1137. [Google Scholar] [CrossRef]
- Liu, X.T.; Lin, X.; Mi, Y.L.; Zeng, W.D.; Zhang, C.Q. Age-related changes of yolk precursor formation in the liver of laying hens. J. Zhejiang Univ. Sci. B 2018, 19, 390–399. [Google Scholar] [CrossRef]
- Roberts, J.R.; Souillard, R.; Bertin, J. Avian diseases which affect egg production and quality. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 1, pp. 376–393. [Google Scholar]
- Heerkens, J.L.; Delezie, E.; Kempen, I.; Zoons, J.; Ampe, B.; Rodenburg, T.B.; Tuyttens, F.A. Specific characteristics of the aviary housing system affect plumage condition, mortality and production in laying hens. Poult. Sci. 2015, 94, 2008–2017. [Google Scholar] [CrossRef] [PubMed]
- Kaab, H.; Bain, M.M.; Bartley, K.; Turnbull, F.; Wright, H.W.; Nisbet, A.J.; Birchmore, R.; Eckersall, P.D. Serum and acute phase protein changes in laying hens, infested with poultry red mite. Poult. Sci. 2018. [Google Scholar] [CrossRef]
- Sigognault Flochlay, A.; Thomas, E.; Sparagano, O. Poultry red mite (Dermanyssus gallinae) infestation: A broad impact parasitological disease that still remains a significant challenge for the egg-laying industry in Europe. Parasites Vectors 2017, 10, 357. [Google Scholar] [CrossRef]
- Tomley, F.M.; Sparagano, O. Spotlight on avian pathology: Red mite, a serious emergent problem in layer hens. Avian Pathol. J. 2018, 47, 533–535. [Google Scholar] [CrossRef]
- De Vylder, J.; Raspoet, R.; Dewulf, J.; Haesebrouck, F.; Ducatelle, R.; Van Immerseel, F. Salmonella Enteritidis is superior in egg white survival compared with other Salmonella serotypes. Poult. Sci. 2013, 92, 842–845. [Google Scholar] [CrossRef] [PubMed]
- Lublin, A.; Sela, S. The Impact of Temperature During the Storage of Table Eggs on the Viability of Salmonella enterica Serovars Enteritidis and Virchow in the Eggs. Poult. Sci. 2008, 87, 2208–2214. [Google Scholar] [CrossRef]
- Chemaly, M.; Salvat, G. Foodborne disease associated with eggs: Microbial hazards and Salmonella Enteritidis risk assessment. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Immersel, F.V., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2. [Google Scholar]
- European Centre for Disease Prevention and Control. Salmonellosis—Annual Epidemiological Report 2016 [2014 Data]; ECDC: Solna, Sweden, 2016.
- Davies, R. Detection and monotoring of Salmonella in laying hens flocks. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Immersel, F.V., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 83–106. [Google Scholar]
- Gast, R.K. Pre-harvest measures to control Salmonella in laying hens. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 120–145. [Google Scholar]
- Ducatelle, R.; Van Immerseel, F. Management and sanitation procedures to control Salmonella in laying hens flocks. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Immersel, F.V., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 146–162. [Google Scholar]
- Messens, W.; Gittins, J.; Leleu, S.; Sparks, N. Egg decontamination by washing. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 163–180. [Google Scholar]
- Berardinelli, A.; Ragni, L.; Giunchi, A.; Gradari, P.; Guarnieri, A. Physical-mechanical modifications of eggs for food-processing during storage. Poult. Sci. 2008, 87, 2117–2125. [Google Scholar] [CrossRef] [PubMed]
- Guyot, N.; Rehault-Godbert, S.; Nys, Y.; Baron, F. Understanding the natural antibacterial defences of egg white and their regulation. In Achieving Sustainable Production of Eggs; Roberts, J., Ed.; Burleigh Dodds Science Publishin Limited: Cambridge, UK, 2016; Volume 1, pp. 161–193. [Google Scholar]
- Rehault-Godbert, S.; Baron, F.; Mignon-Grasteau, S.; Labas, V.; Gautier, M.; Hincke, M.T.; Nys, Y. Effect of Temperature and Time of Storage on Protein Stability and Anti-Salmonella Activity of Egg White. J. Food Prot. 2010, 73, 1604–1612. [Google Scholar] [CrossRef]
- Samli, H.E.; Agma, A.; Senkoylu, N. Effects of storage time and temperature on egg quality in old laying hens. J. Appl. Poult. Res. 2005, 14, 548–553. [Google Scholar] [CrossRef]
- Garcia, F.J.; Pons, A.; Alemany, M.; Palou, A. Permeability of chicken egg vitelline membrane to glucose, carbohydrate gradients between albumen and yolk. Comp. Biochem. Physiology. Bcomp. Biochem. 1983, 75, 137–140. [Google Scholar] [CrossRef]
- Rehault-Godbert, S.; Mann, K.; Bourin, M.; Brionne, A.; Nys, Y. Effect of embryonic development on the chicken egg yolk plasma proteome after 12 days of incubation. J. Agric. Food Chem. 2014, 62, 2531–2540. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Qiu, N.; Liu, Y.P.; Ma, M.H. Comparative proteome analysis of egg yolk plasma proteins during storage. J. Sci. Food Agric. 2017, 97, 2392–2400. [Google Scholar] [CrossRef]
- Qiu, N.; Ma, M.; Zhao, L.; Liu, W.; Li, Y.; Mine, Y. Comparative proteomic analysis of egg white proteins under various storage temperatures. J. Agric. Food Chem. 2012, 60, 7746–7753. [Google Scholar] [CrossRef] [PubMed]
- Bush, L.; White, H.B., 3rd. Avidin traps biotin diffusing out of chicken egg yolk. Comp. Biochem. Physiol. Bcomp. Biochem. 1989, 93, 543–547. [Google Scholar] [CrossRef]
- Nimalaratne, C.; Schieber, A.; Wu, J. Effects of storage and cooking on the antioxidant capacity of laying hen eggs. Food Chem. 2016, 194, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Martos, G.; Lopez-Fandino, R.; Molina, E. Immunoreactivity of hen egg allergens: Influence on in vitro gastrointestinal digestion of the presence of other egg white proteins and of egg yolk. Food Chem. 2013, 136, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Vanga, S.K.; Wang, J.; Raghavan, V. Impact of food processing on the structural and allergenic properties of egg white. Trends Food Sci. Technol. 2018, 78, 188–196. [Google Scholar] [CrossRef]
- Juhaimi, F.A.; Uslu, N.; Ozcan, M.M. Oil content and fatty acid composition of eggs cooked in drying oven, microwave and pan. J. Food Sci. Technol. 2017, 54, 93–97. [Google Scholar] [CrossRef]
- Nyemb, K.; Guerin-Dubiard, C.; Pezennec, S.; Jardin, J.; Briard-Bion, V.; Cauty, C.; Rutherfurd, S.M.; Dupont, D.; Nau, F. The structural properties of egg white gels impact the extent of in vitro protein digestion and the nature of peptides generated. Food Hydrocoll. 2016, 54, 315–327. [Google Scholar] [CrossRef]
- Wang, X.F.; Qiu, N.; Liu, Y.P. Effect of Different Heat Treatments on In Vitro Digestion of Egg White Proteins and Identification of Bioactive Peptides in Digested Products. J. Food Sci. 2018, 83, 1140–1148. [Google Scholar] [CrossRef]
- Remanan, M.K.; Wu, J. Antioxidant activity in cooked and simulated digested eggs. Food Funct. 2014, 5, 1464–1474. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.F.; Lin, C.C. Production, composition and quality of duck eggs. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Immersel, F.V., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 487–508. [Google Scholar]
- Tserveni-Goussi, A.; Fortomaris, P. Production and quality of quail, pheasant, goose and turkey eggs for uses other than human consumption. In Improving the Safety and Quality of Eggs and Egg Products; Nys, Y., Bain, M., Van Immerseel, F., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 509–537. [Google Scholar]
- Ali, A.H.; Zou, X.; Lu, J.; Abed, S.M.; Yao, Y.; Tao, G.; Jin, Q.; Wang, X. Identification of phospholipids classes and molecular species in different types of egg yolk by using UPLC-Q-TOF-MS. Food Chem. 2017, 221, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Bair, C.W.; Marion, W.W. Yolk cholesterol in eggs from various avian species. Poult. Sci. 1978, 57, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Golzar Adabi, S.H.; Ahbab, M.; Fani, A.R.; Hajbabaei, A.; Ceylan, N.; Cooper, R.G. Egg yolk fatty acid profile of avian species--influence on human nutrition. J. Anim. Physiol. Anim. Nutr. 2013, 97, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Qiu, N.; Liu, Y.; Zhao, H.; Gao, D.; Song, R.; Ma, M. Identification and comparative proteomic study of quail and duck egg white protein using 2-dimensional gel electrophoresis and matrix-assisted laser desorption/ionization time-of-flight tandem mass spectrometry analysis. Poult. Sci. 2016, 95, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.P.; Qiu, N.; Gao, D.; Ma, M.H. Comparative proteomic analysis of chicken, duck, and quail egg yolks. Int. J. Food Prop. 2018, 21, 1311–1321. [Google Scholar] [CrossRef]
- Sun, C.J.; Liu, J.N.; Li, W.B.; Xu, G.Y.; Yang, N. Divergent Proteome Patterns of Egg Albumen from Domestic Chicken, Duck, Goose, Turkey, Quail and Pigeon. Proteomics 2017, 17, 12. [Google Scholar] [CrossRef]
- Bacon, W.L.; Brown, K.I.; Musser, M.A. Low density lipoproteins of chicken, turkey and quail egg yolk. Poult. Sci. 1973, 52, 1741–1744. [Google Scholar] [CrossRef]
- Appleby, P.N.; Key, T.J. The long-term health of vegetarians and vegans. Proc. Nutr. Soc. 2016, 75, 287–293. [Google Scholar] [CrossRef]
- Key, T.J.; Davey, G.K.; Appleby, P.N. Health benefits of a vegetarian diet. Proc. Nutr. Soc. 1999, 58, 271–275. [Google Scholar] [CrossRef]
- Dinu, M.; Abbate, R.; Gensini, G.F.; Casini, A.; Sofi, F. Vegetarian, vegan diets and multiple health outcomes: A systematic review with meta-analysis of observational studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3640–3649. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Lv, J.; Guo, Y.; Bian, Z.; Si, J.; Yang, L.; Chen, Y.; Zhou, Y.; Zhang, H.; Liu, J.; et al. Associations of egg consumption with cardiovascular disease in a cohort study of 0.5 million Chinese adults. Heart (Br. Card. Soc.) 2018, 104, 1756–1763. [Google Scholar] [CrossRef]
- Orlich, M.J.; Jaceldo-Siegl, K.; Sabate, J.; Fan, J.; Singh, P.N.; Fraser, G.E. Patterns of food consumption among vegetarians and non-vegetarians. Br. J. Nutr. 2014, 112, 1644–1653. [Google Scholar] [CrossRef] [PubMed]
- Thuenemann, E.C.; Mandalari, G.; Rich, G.T.; Faulks, R.M. Dynamic Gastric Model (DGM). In The Impact of Food Bioactives on Health: In Vitro and Ex Vivo Models; Verhoeckx, K., Cotter, P., Lopez-Exposito, I., Kleiveland, C., Lea, T., Mackie, A., Requena, T., Swiatecka, D., Wichers, H., Eds.; Springer: Cham, Switzerland, 2015; pp. 47–59. [Google Scholar]
- Wang, X.; Ye, A.; Lin, Q.; Han, J.; Singh, H. Gastric digestion of milk protein ingredients: Study using an in vitro dynamic model. J. Dairy Sci. 2018, 101, 6842–6852. [Google Scholar] [CrossRef] [PubMed]
- Do, D.H.T.; Kong, F.B.; Penet, C.; Winetzky, D.; Gregory, K. Using a dynamic stomach model to study efficacy of supplemental enzymes during simulated digestion. Lwt-Food Sci. Technol. 2016, 65, 580–588. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Holmes, E.; Kinross, J.; Burcelin, R.; Gibson, G.; Jia, W.; Pettersson, S. Host-gut microbiota metabolic interactions. Science 2012, 336, 1262–1267. [Google Scholar] [CrossRef]
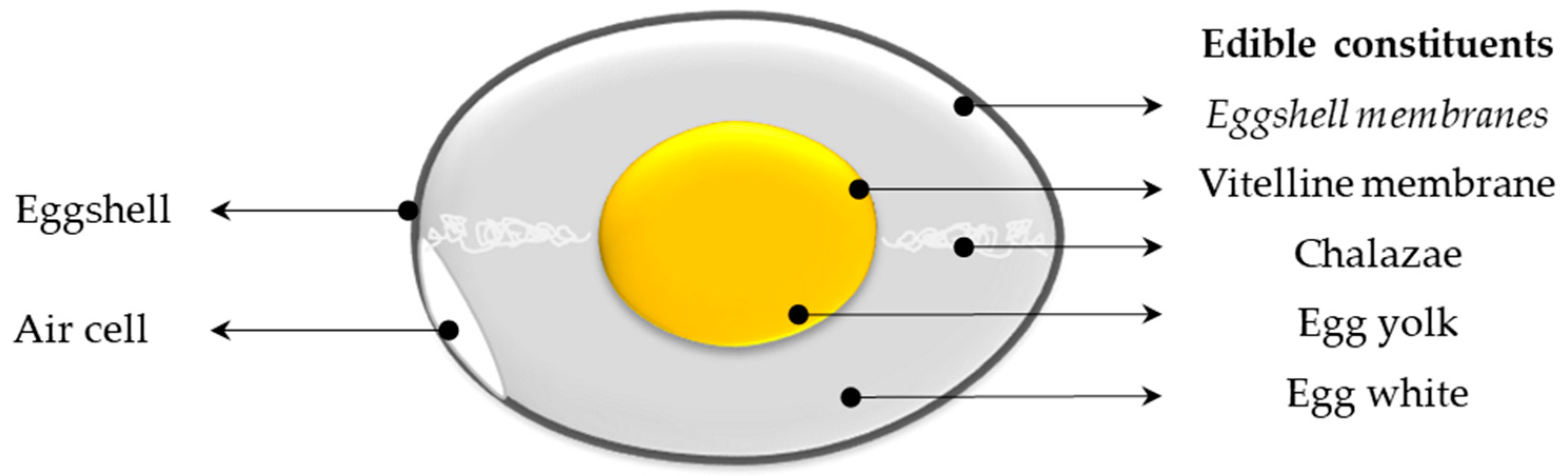
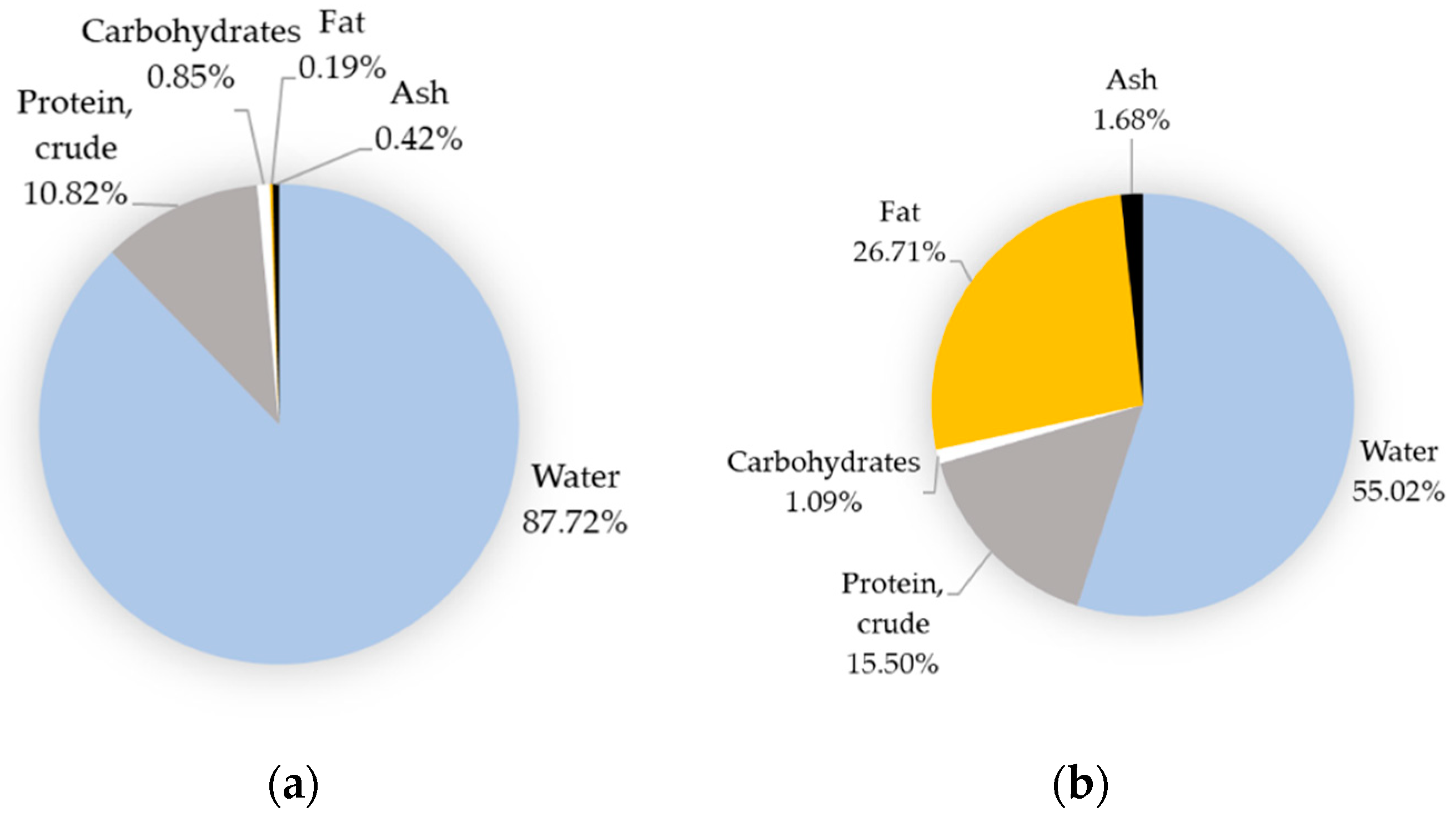
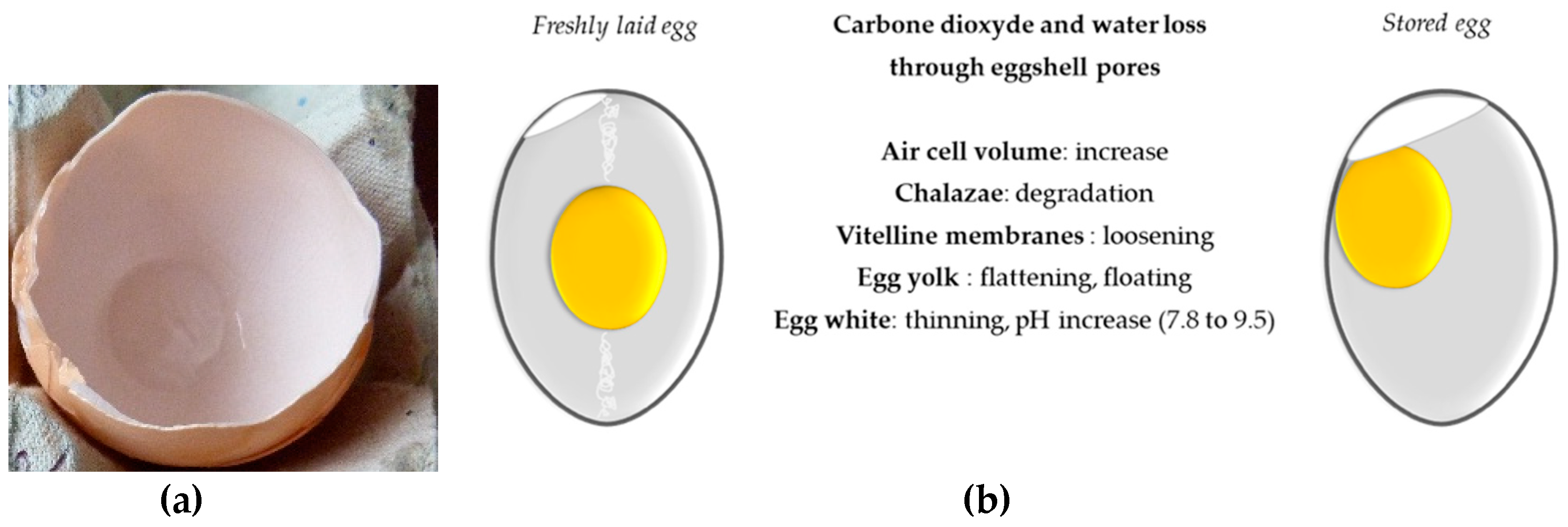
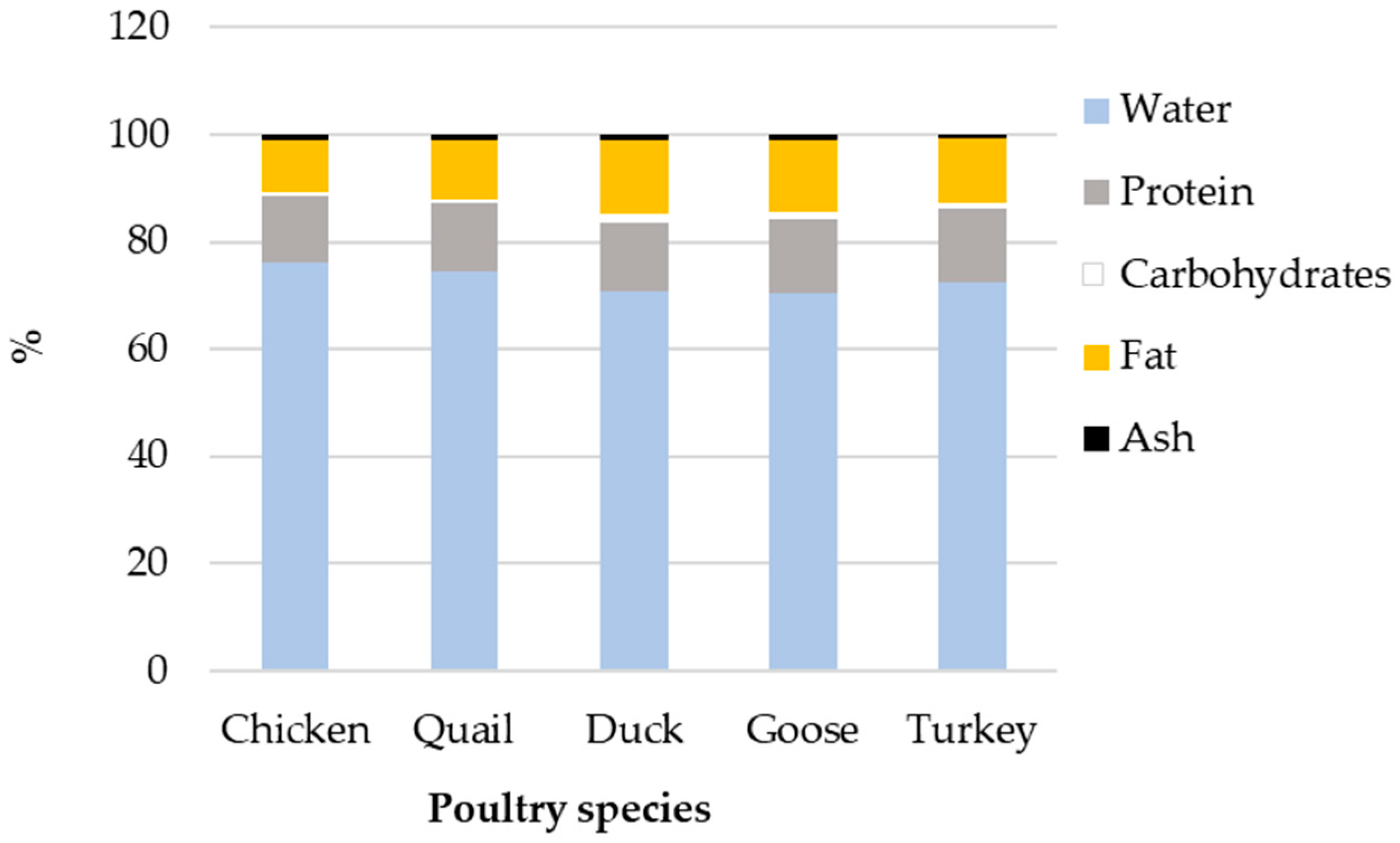
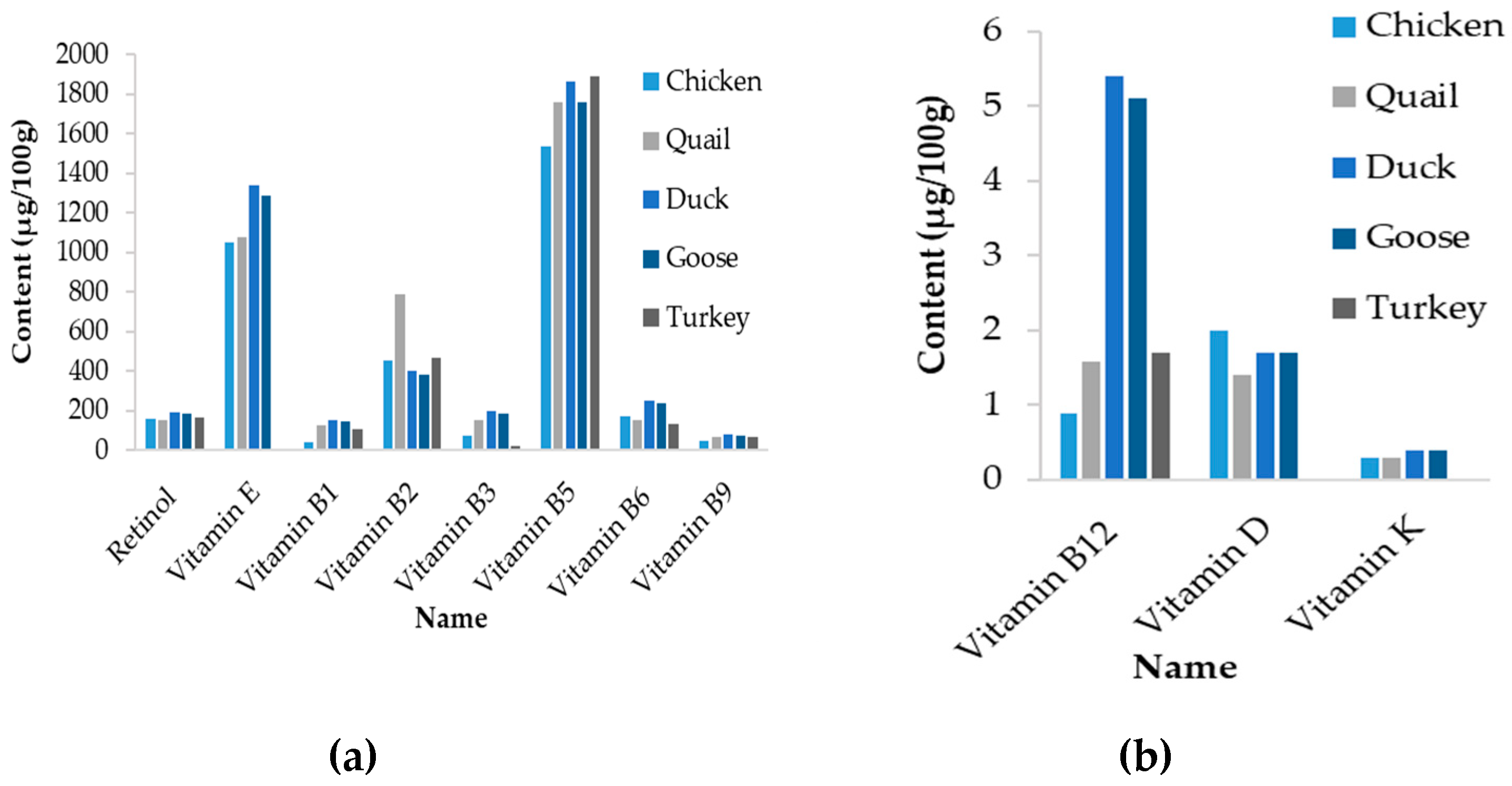
| Name | Egg, Whole, Raw | Egg Yolk, Raw | ||||
|---|---|---|---|---|---|---|
| Average Content (g/100g) | Min. Value (g/100g) | Max. Value (g/100g) | Average Content (g/100g) | Min. Value (g/100g) | Max. Value (g/100g) | |
| FA saturated | 2.64 | 0.05 | 3.13 | 8.47 | 7.13 | 9.55 |
| FA 4:0 | <0.05 | 0 | - 2 | 0 | - | - |
| FA 6:0 | <0.05 | 0 | - | 0 | - | - |
| FA 8:0 | <0.05 | 0 | - | 0.009 | - | - |
| FA 10:0 | <0.05 | 0 | - | 0.009 | - | - |
| FA 12:0 | <0.05 | 0 | - | 0.009 | - | - |
| FA 14:0 | 0.024 | 0 | 0.038 | 0.091 | 0.077 | 0.1 |
| FA 16:0 | 1.96 | 0.05 | 2.43 | 6.04 | 5.03 | 6.86 |
| FA 18:0 | 0.65 | 0.05 | 0.89 | 1.73 | - | 2.42 |
| FA monounsaturated | 3.66 | 0.05 | 6.73 | 11.9 | 10.2 | 13.8 |
| FA 18:1 n-9 cis | 3.51 | 3.03 | 3.65 | 10.4 | 9.69 | 11.2 |
| FA polyunsaturated | 1.65 | 0.05 | 3.39 | 4.07 | 3.33 | 4.66 |
| FA 18:2 9c,12c (n-6) | 1.38 | 1.18 | 2.7 | 3.28 | - | 3.62 |
| FA 18:3 9c,12c,15c (n-3) | 0.061 | 0.02 | 0.58 | 0.15 | - | 0.27 |
| FA 20:4 5c,8c,11c,14c (n-6) | 0.12 | - | 0.13 | 0.37 | - | 0.4 |
| FA 20:5 5c,8c,11c,14c,17c (n-3) EPA | 0 | - | 0.003 | 0.01 | - | 0.011 |
| FA 22:6 4c,7c,10c,13c,16c,19c (n-3) DHA | 0.09 | 0.045 | 0.18 | 0.25 | 0.11 | 0.46 |
| Cholesterol | 0.398 | 0.344 | 0.423 | 0.939 | 1.280 | |
| Name | Egg, Whole, Raw | Egg Yolk, Raw | Egg White, Raw |
|---|---|---|---|
| Vitamin A or Retinol | 160 | 371 | 0 |
| Vitamin A precursor or Beta-carotene | 0 | 88 | 0 |
| Vitamin D or Cholecalciferol | 2.0 | 5.4 | 0 |
| Vitamin E or Alpha-tocopherol | 1050 | 2580 | 0 |
| Vitamin K or Phylloquinone | 0.3 | 0.7 | 0 |
| Vitamin C | 0 | 0 | 0 |
| Vitamin B1 or Thiamin | 40 | 176 | 4 |
| Vitamin B2 or Riboflavin | 457 | 528 | 439 |
| Vitamin B3 or Niacin | 75 | 24 | 105 |
| Vitamin B5 or Pantothenic acid | 1533 | 2990 | 190 |
| Vitamin B6 | 170 | 350 | 5 |
| Vitamin B8 or Biotin | 16.5–53.8 2 | 27.2–49.4 2 | 5.7–7.9 2 |
| Vitamin B9 or Folate | 47 | 146 | 4 |
| Vitamin B12 or Colabamin | 0.89 | 1.95 | 0.09 |
| Name | Egg, Whole, Raw | Egg Yolk, Raw | Egg White, Raw |
|---|---|---|---|
| Calcium | 56 | 129 | 7 |
| Copper | 0.072 | 0.077 | 0.023 |
| Iodine | 0.021 | 0.18 | 0.002 |
| Iron | 1.75 | 2.73 | 0.08 |
| Magnesium | 12 | 5 | 11 |
| Manganese | 0.028 | 0.055 | 0.011 |
| Phosphorus | 198 | 390 | 15 |
| Potassium | 138 | 109 | 163 |
| Selenium | 0.030 | 0.056 | 0.020 |
| Sodium | 142 | 48 | 166 |
| Zinc | 1.29 | 2.30 | 0.03 |
| Gene ID/Gene Symbol | Target Organisms | Localization 1 | References | |
|---|---|---|---|---|
| Avian beta-defensin 11 | 414876/AVBD11 | Bacteria | EW, VM | [76,77] |
| Avidin | 396260/AVD | Bacteria | EW, VM, EY | [78,79] |
| Beta-microseminoprotein-like | 101750704 | Bacteria | EW | [76] |
| Cystatin | 396497/CST3 | Bacteria, viruses, fungi, parasites | EW, VM, EY | [75,80,81,82,83,84,85] |
| Gallin | 422030/OvoDA1 | Bacteria | EW | [86,87,88,89] |
| Immunoglobulin Y | - | EY | [90] | |
| Lysozyme | 396218/LYZ | Bacteria, viruses, fungi | EW, VM, EY | [67,91,92] |
| Ovalbumin-related protein X | 420898/OVALX | Bacteria | EW, VM, EY | [93] |
| OvoglobulinG2/TENP | 395882/BPIFB2 | Bacteria | EW, VM, EY | [94,95,96] |
| Ovoinhibitor | 416235/SPIK5 | Bacteria | EW, EY | [97] |
| Ovomucin (alpha and beta subunits) | 395381/LOC395381 (alpha) 414878/MUC6 (beta) | Bacteria, viruses | EW, VM | [74] |
| Ovotransferrin | 396241/TF | Bacteria, viruses | EW, VM, EY | [98,99,100] |
| Phosvitin | 424547/VTG1 | Bacteria | EY | [101] |
| Pleiotrophin | 418125/PTN | Bacteria | EW | [76,102] |
| Vitelline membrane outer layer protein 1 | 418974/VMO1 | Bacteria | EW, VM, EY | [76] |
| Name | Egg, Whole, Raw, Fresh | Egg, Whole, Soft-Boiled | Egg, Whole, Hard-Boiled |
|---|---|---|---|
| Energy (kcal/100g) | 140 1; 143 2 | 142; 143 | 134; 155 |
| Protein (g/100g) | 12.7; 12.56 | 12.2; 12.51 | 13.5; 12.58 |
| Carbohydrate (g/100g) | 0.27; 0.72 | 1.08; 0.71 | 0.52; 1.12 |
| Fat (g/100g) | 9.83; 9.51 | 9.82; 9.47 | 8.62; 10.61 |
| FA saturated (g/100g) | 2.64; 3.126 | 3.11; 3.11 | 2.55; 3.267 |
| FA monounsaturated (g/100g) | 3.66; 3.658 | 4.42; 3.643 | 3.57; 4.077 |
| FA polyunsaturated (g/100g) | 1.65; 1.911 | 1.28; 1.904 | 1.03; 1.414 |
| Cholesterol (mg/100g) | 398; 372 | 222; 370 | 355; 373 |
| Salt (g/100g) | 0.31 | 0.2 | 0.31 |
| Calcium (mg/100g) | 76.8; 56 | 150; 56 | 41; 50 |
| Potassium (mg/100g) | 134; 138 | 164; 138 | 120; 126 |
| Selenium (µg/100g) | 30 | 23.8 | 7.01 |
| Vitamin A, Retinol (µg/100g) | 182; 160 | 132; 160 | 61.5; 149 |
| Vitamin D (µg/100g) | 1.88; 2.0 | 1.28; 2.0 | 1.12; 2.2 |
| Vitamin E (mg/100g) | 1.43; 1.05 | 2.17; 1.04 | 1.03; 1.03 |
| Choline (mg/100g) | 250; 293.8 | - | 230; 293.8 |
| Name | Chicken | Quail | Duck | Goose | Turkey |
|---|---|---|---|---|---|
| Calcium | 56 | 64 | 64 | 60 | 99 |
| Copper | 0.072 | 0.062 | 0.062 | 0.062 | 0.062 |
| Iron | 1.75 | 3.65 | 3.85 | 3.64 | 4.1 |
| Magnesium | 12 | 13 | 17 | 16 | 13 |
| Manganese | 0.028 | 0.038 | 0.038 | 0.038 | 0.038 |
| Phosphorus | 198 | 226 | 220 | 208 | 170 |
| Potassium | 138 | 132 | 222 | 210 | 142 |
| Selenium | 0.0307 | 0.032 | 0.0364 | 0.0369 | 0.0343 |
| Sodium | 142 | 141 | 146 | 138 | 151 |
| Zinc | 1.29 | 1.47 | 1.41 | 1.33 | 1.58 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Réhault-Godbert, S.; Guyot, N.; Nys, Y. The Golden Egg: Nutritional Value, Bioactivities, and Emerging Benefits for Human Health. Nutrients 2019, 11, 684. https://doi.org/10.3390/nu11030684
Réhault-Godbert S, Guyot N, Nys Y. The Golden Egg: Nutritional Value, Bioactivities, and Emerging Benefits for Human Health. Nutrients. 2019; 11(3):684. https://doi.org/10.3390/nu11030684
Chicago/Turabian StyleRéhault-Godbert, Sophie, Nicolas Guyot, and Yves Nys. 2019. "The Golden Egg: Nutritional Value, Bioactivities, and Emerging Benefits for Human Health" Nutrients 11, no. 3: 684. https://doi.org/10.3390/nu11030684
APA StyleRéhault-Godbert, S., Guyot, N., & Nys, Y. (2019). The Golden Egg: Nutritional Value, Bioactivities, and Emerging Benefits for Human Health. Nutrients, 11(3), 684. https://doi.org/10.3390/nu11030684





