Energy Status and Body Composition Across a Collegiate Women’s Lacrosse Season
Abstract
1. Introduction
2. Materials and Methods
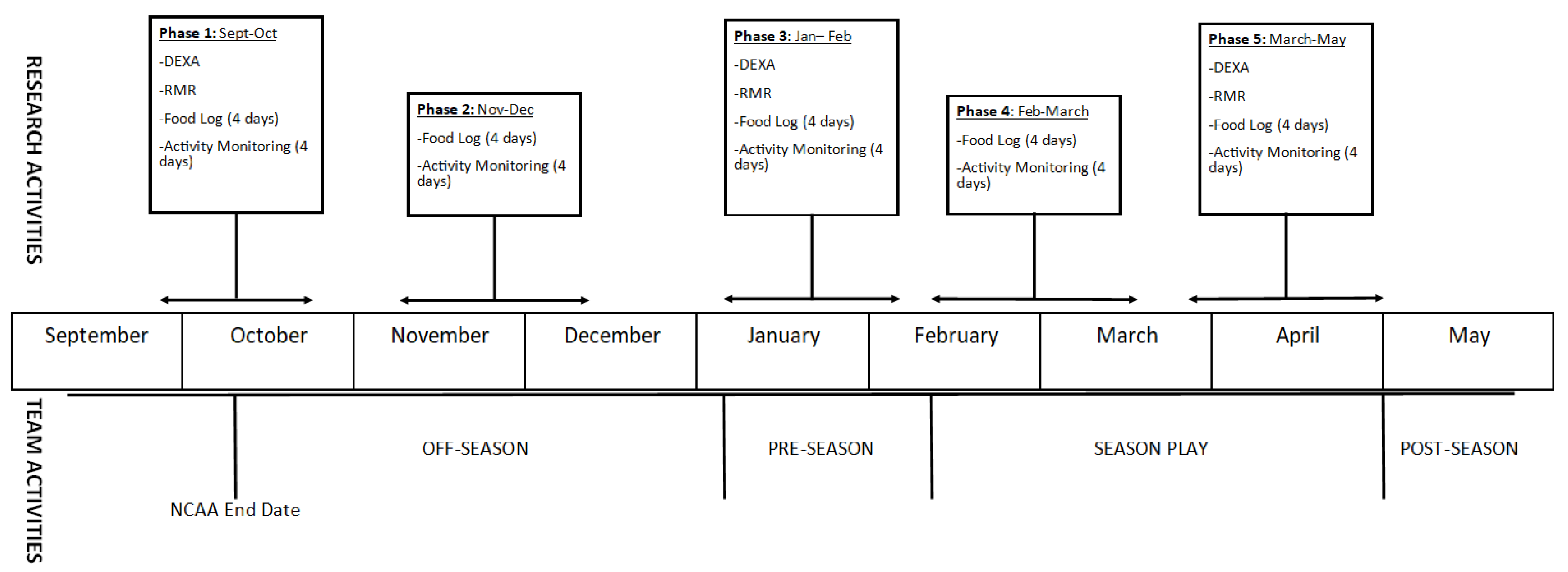
2.1. Research Design
2.2. Subjects
2.3. Methodology
2.3.1. Anthropometrics, Body Composition and Bone Health
2.3.2. Resting Metabolic Rate
2.3.3. Diet Record
2.3.4. Physical Activity Monitoring
2.3.5. Markers of Energy Deficiency
2.3.6. Assessment of Recovery
2.4. Statistical Analysis
3. Results
3.1. Body Composition and Bone Health
3.2. Metabolic Rate and Energy Expenditure
3.3. Energy Intake
3.4. Energy Balance, Energy Availability and Surrogate Markers of Energy Deficiency
3.5. Recovery Measures
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Scheers, T.; Philippaerts, R.; Lefevre, J. Variability in physical activity patterns as measured by the SenseWear Armband: How many days are needed? Eur. J. Appl. Physiol. 2012, 112, 1653–1662. [Google Scholar] [CrossRef] [PubMed]
- Drenowatz, C.; Hand, G.A.; Shook, R.P.; Jakicic, J.M.; Hebert, J.R.; Burgess, S.; Blair, S.N. The association between different types of exercise and energy expenditure in young nonoverweight and overweight adults. Appl. Physiol. Nutr. Metab. 2015, 40, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.B.; Madrigal, L.A.; Burnfield, J.M. Weight control practices of Division I National Collegiate Athletic Association athletes. Phys. Sportsmed. 2016, 44, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Pikosky, M.A.; Smith, T.J.; Grediagin, A.; Castaneda-Sceppa, C.; Byerley, L.; Glickman, E.L.; Young, A.J. Increased protein maintains nitrogen balance during exercise-induced energy deficit. Med. Sci. Sports Exerc. 2008, 40, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Joy, E.A.; Campbell, D. Stress fractures in the female athlete. Curr. Sports Med. Rep. 2005, 4, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Novas, A.; Rowbottom, D.; Jenkins, D. Total daily energy expenditure and incidence of upper respiratory tract infection symptoms in young females. Int. J. Sports Med. 2002, 23, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Logue, D.; Madigan, S.M.; Delahunt, E.; Heinen, M.; Mc Donnell, S.J.; Corish, C.A. Low Energy Availability in Athletes: A Review of Prevalence, Dietary Patterns, Physiological Health, and Sports Performance. Sports Med. 2018, 48, 73–96. [Google Scholar] [CrossRef] [PubMed]
- Melin, A.; Tornberg, A.B.; Skouby, S.; Moller, S.S.; Sundgot-Borgen, J.; Faber, J.; Sidelmann, J.J.; Aziz, M.; Sjodin, A. Energy availability and the female athlete triad in elite endurance athletes. Scand. J. Med. Sci. Sports 2015, 25, 610–622. [Google Scholar] [CrossRef] [PubMed]
- Mountjoy, M.; Sundgot-Borgen, J.; Burke, L.; Carter, S.; Constantini, N.; Lebrun, C.; Meyer, N.; Sherman, R.; Steffen, K.; Budgett, R.; et al. The IOC consensus statement: Beyond the Female Athlete Triad--Relative Energy Deficiency in Sport (RED-S). Br. J. Sports Med. 2014, 48, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.J.; Davies, P.S. Energy intake and energy expenditure in elite lightweight female rowers. Med. Sci. Sports Exerc. 2002, 34, 1823–1829. [Google Scholar] [CrossRef] [PubMed]
- Ebine, N.; Feng, J.Y.; Homma, M.; Saitoh, S.; Jones, P.J. Total energy expenditure of elite synchronized swimmers measured by the doubly labeled water method. Eur. J. Appl. Physiol. 2000, 83, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gibson, J.C.; Stuart-Hill, L.; Martin, S.; Gaul, C. Nutrition status of junior elite Canadian female soccer athletes. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.L.; De Souza, M.J.; Williams, N.I. Changes in energy availability across the season in Division I female soccer players. J. Sports Sci. 2013, 31, 314–324. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, S.J.; Meloche, R.D. Energy availability of female varsity volleyball players. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Mara, J.K.; Thompson, K.G.; Pumpa, K.L. Assessing the Energy Expenditure of Elite Female Soccer Players: A Preliminary Study. J. Strength Cond. Res. 2015, 29, 2780–2786. [Google Scholar] [CrossRef] [PubMed]
- Sell, K.M.; Ledesma, A.B. Heart Rate and Energy Expenditure in Division I Field Hockey Players During Competitive Play. J. Strength Cond. Res. 2016, 30, 2122–2128. [Google Scholar] [CrossRef] [PubMed]
- Zanders, B.R.; Currier, B.S.; Harty, P.S.; Zabriskie, H.A.; Smith, C.R.; Stecker, R.A.; Richmond, S.R.; Jagim, A.R.; Kerksick, C.M. Changes in Energy Expenditure, Dietary Intake, and Energy Availability Across an Entire Collegiate Women’s Basketball Season. J. Strength Cond. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Crouter, S.E.; Churilla, J.R.; Bassett, D.R., Jr. Accuracy of the Actiheart for the assessment of energy expenditure in adults. Eur. J. Clin. Nutr. 2007, 62, 704. [Google Scholar] [CrossRef] [PubMed]
- Loucks, A.B.; Kiens, B.; Wright, H.H. Energy availability in athletes. J. Sports Sci. 2011, 29 (Suppl. 1), S7–S15. [Google Scholar] [CrossRef]
- Staal, S.; Sjodin, A.; Fahrenholtz, I.; Bonnesen, K.; Melin, A. Low RMRratio as a Surrogate Marker for Energy Deficiency, the Choice of Predictive Equation Vital for Correctly Identifying Male and Female Ballet Dancers at Risk. Int. J. Sport Nutr. Exerc. Metab. 2018, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Ten Haaf, T.; Weijs, P.J. Resting energy expenditure prediction in recreational athletes of 18–35 years: Confirmation of Cunningham equation and an improved weight-based alternative. PLoS ONE 2014, 9, e108460. [Google Scholar] [CrossRef] [PubMed]
- Schofield, W.N. Predicting basal metabolic rate, new standards and review of previous work. Hum. Nutr. Clin. Nutr. 1985, 39 (Suppl. 1), 5–41. [Google Scholar] [PubMed]
- Capling, L.; Beck, K.L.; Gifford, J.A.; Slater, G.; Flood, V.M.; O’Connor, H. Validity of Dietary Assessment in Athletes: A Systematic Review. Nutrients 2017, 9, 1313. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.L.; De Souza, M.J.; Kindler, J.M.; Williams, N.I. Nutritional practices associated with low energy availability in Division I female soccer players. J. Sports Sci. 2014, 32, 1499–1509. [Google Scholar] [CrossRef] [PubMed]
- Barrack, M.T.; Gibbs, J.C.; De Souza, M.J.; Williams, N.I.; Nichols, J.F.; Rauh, M.J.; Nattiv, A. Higher Incidence of Bone Stress Injuries with Increasing Female Athlete Triad–Related Risk Factors: A Prospective Multisite Study of Exercising Girls and Women. Am. J. Sports Med. 2014, 42, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.D.; Zabriskie, H.A.; Witherbee, K.E.; Sulavik, A.; Gieske, B.T.; Kerksick, C.M. Determining a Resting Metabolic Rate Prediction Equation for Collegiate Female Athletes. J. Strength Cond. Res. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Tomten, S.E.; Hostmark, A.T. Energy balance in weight stable athletes with and without menstrual disorders. Scand. J. Med. Sci. Sports 2006, 16, 127–133. [Google Scholar] [CrossRef] [PubMed]
| Phase 1 (Off-Season) | Phase 2 (Off-Season) | Phase 3 (Pre-Season) | Phase 4 (In-Season) | Phase 5 (In-Season) | Overall p-Value | |
|---|---|---|---|---|---|---|
| Body Weight (kg) | 68.8 ± 8.9 | 69.6 ± 9.5 | 69.6 ± 10.0 | 69.3 ± 10.0 | 68.9 ± 10.1 | 0.20 |
| FFM (kg) | 47.0 ± 5.3 | -- | 47.9 ± 5.4 | -- | 47.4 ± 5.6 | 0.054 |
| FM (kg) | 18.4 ± 4.4 | -- | 18.3 ± 4.7 | -- | 17.8 ± 4.8 | 0.118 |
| Percent Fat (%) * | 27.9 ± 3.0 | -- | 27.3 ± 2.7 | -- | 27.0 ± 3.2 | 0.037 |
| BMC (g) * | 2575 ± 230 5 | -- | 2572 ± 230 5 | -- | 2610 ± 247 1,3 | <0.001 |
| BMD (g/cm3) | 1.20 ± 0.07 | -- | 1.20 ± 0.07 | -- | 1.24 ± 0.14 | 0.167 |
| z-Score * | 1.30 ± 0.76 5 | -- | 1.28 ± 0.80 5 | -- | 1.46 ± 0.75 1,3 | 0.004 |
| Phase 1 (Off-Season) | Phase 2 (Off-Season) | Phase 3 (Pre-Season) | Phase 4 (In-Season) | Phase 5 (In-Season) | Overall p-Value | |
|---|---|---|---|---|---|---|
| RMR (kcal/day) * | 1536 ± 152 3,5 | -- | 1683 ± 162 1 | -- | 1732 ± 244 1 | <0.001 |
| TDEE (kcal/day) * | 2608 ± 378 3 | 2579 ± 376 | 2798 ± 391 1 | 2513 ± 248 3 | 2582 ± 303 3 | <0.001 |
| AEE (kcal/day) * | 842 ± 267 3 | 804 ± 244 | 1001 ± 267 1 | 749 ± 161 3 | 817 ± 235 3 | 0.001 |
| PAL * | 1.75 ± 0.19 3 | 1.72 ± 0.14 3 | 1.87 ± 0.15 1 | 1.69 ± 0.15 3 | 1.73 ± 0.18 3 | 0.001 |
| Phase 1 (Off-Season) | Phase 2 (Off-Season) | Phase 3 (Pre-Season) | Phase 4 (In-Season) | Phase 5 (In-Season) | Overall p-Value | |
|---|---|---|---|---|---|---|
| Calories (kcal/day) | 2242 ± 462 | 2015 ± 451 | 2079 ± 435 | 2124 ± 505 | 2161 ± 392 | 0.247 |
| Carbohydrate (g/day) | 262 ± 61 | 231 ± 59 | 247 ± 74 | 248 ± 66 | 236 ± 74 | 0.262 |
| Protein (g/day) | 80 ± 19 | 72 ± 20 | 82 ± 22 | 84 ± 16 | 79 ± 20 | 0.168 |
| Fat (g/day) * | 78 ± 20 | 70 ± 25 5 | 74 ± 23 | 81 ± 26 | 88 ± 23 2 | 0.03 |
| Relative Carbohydrate (g/kg/day) | 3.9 ± 1.1 | 3.4 ± 0.9 | 3.6 ± 1.2 | 3.6 ± 0.9 | 3.5 ± 1.2 | 0.146 |
| Relative Protein (g/kg/day) | 1.2 ± 0.3 | 1.1 ± 0.3 | 1.2 ± 0.4 | 1.2 ± 0.3 | 1.2 ± 0.4 | 0.163 |
| Phase 1 (Off-Season) | Phase 2 (Off-Season) | Phase 3 (Pre-Season) | Phase 4 (In-Season) | Phase 5 (In-Season) | Overall p-Value | |
|---|---|---|---|---|---|---|
| Energy Balance (kcal/day) * | −366 ± 527 ‡ | −564 ± 484 | −719 ± 440 | −389 ± 432 3 | −421 ± 418 | 0.01 |
| Energy Availability (kcal/kg FFM) * | 30.4 ± 11.0 ‡ | 26.2 ± 10.5 | 22.9 ± 8.5 | 28.7 ± 9.5 ‡ | 28.9 ± 9.2 | 0.017 |
| RMR Ratio, Schofield (Measured/Predicted) * | 1.02 ± 0.7 | -- | 1.11 ± 0.1 1 | -- | 1.15 ± 0.1 1 | <0.001 |
| RMR Ratio, Cunningham (Measured/Predicted) * | 1.0 ± 0.1 | -- | 1.08 ± 0.1 1 | -- | 1.1 ± 0.1 1 | 0.001 |
| Free Mass Index (kg/m2) | 16.6 ± 1.2 | -- | 16.7 ± 1.2 | -- | 16.5 ± 1.3 | 0.175 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zabriskie, H.A.; Currier, B.S.; Harty, P.S.; Stecker, R.A.; Jagim, A.R.; Kerksick, C.M. Energy Status and Body Composition Across a Collegiate Women’s Lacrosse Season. Nutrients 2019, 11, 470. https://doi.org/10.3390/nu11020470
Zabriskie HA, Currier BS, Harty PS, Stecker RA, Jagim AR, Kerksick CM. Energy Status and Body Composition Across a Collegiate Women’s Lacrosse Season. Nutrients. 2019; 11(2):470. https://doi.org/10.3390/nu11020470
Chicago/Turabian StyleZabriskie, Hannah A., Bradley S. Currier, Patrick S. Harty, Richard A. Stecker, Andrew R. Jagim, and Chad M. Kerksick. 2019. "Energy Status and Body Composition Across a Collegiate Women’s Lacrosse Season" Nutrients 11, no. 2: 470. https://doi.org/10.3390/nu11020470
APA StyleZabriskie, H. A., Currier, B. S., Harty, P. S., Stecker, R. A., Jagim, A. R., & Kerksick, C. M. (2019). Energy Status and Body Composition Across a Collegiate Women’s Lacrosse Season. Nutrients, 11(2), 470. https://doi.org/10.3390/nu11020470





