A Randomized Feeding Trial of Iron-Biofortified Beans in School Children in Mexico
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
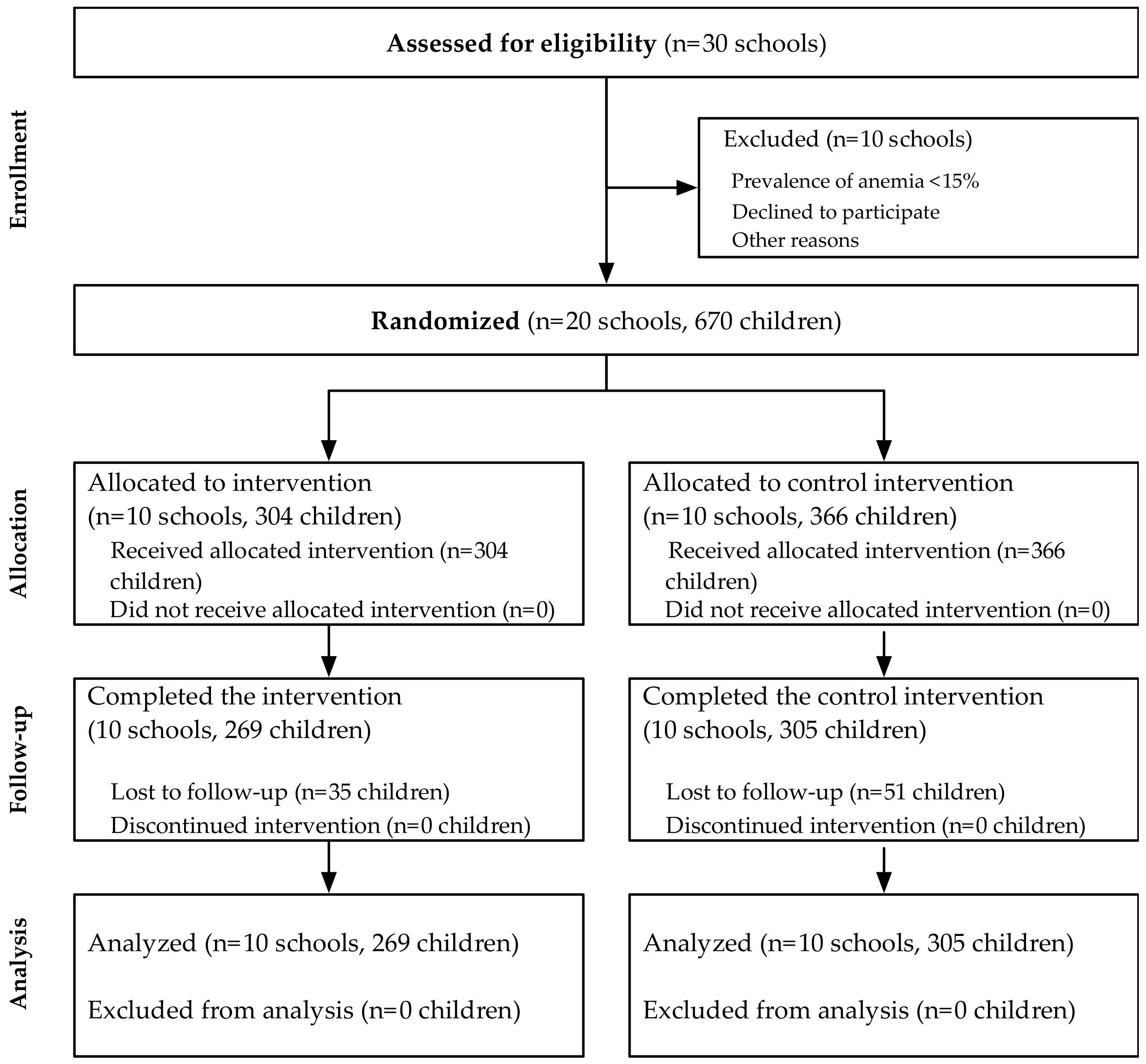
2.2. Study Design
2.3. Intervention
2.4. Power and Sample Size Calculations
3. Results
3.1. Baseline Characteristics
3.2. Effects of the Intervention on Iron Status
3.3. Plausability Analyses
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Iron Deficiency Anaemia Assessment, Prevention, and Control: A Guide for Programme Managers; WHO: Geneva, Switzerland, 2001. [Google Scholar]
- UNSCN. 4th Report—The World Nutrition Situation: Nutrition Throughout the Life Cycle; UNSCN: Rome, Italy, 2000. [Google Scholar]
- McLean, E.; Cogswell, M.; Egli, I.; Wojdyla, D.; de Benoist, B. Worldwide prevalence of anaemia, WHO Vitamin and Mineral Nutrition Information System, 1993–2005. Pub. Health Nutr. 2009, 12, 444–454. [Google Scholar] [CrossRef]
- ACC/SCN. Fourth Report on the World Nutrition Situation; ACC/SCN in collaboration with IFPRI: Geneva, Switzerland, 2000. [Google Scholar]
- WHO. Micronutrient Deficiencies: Iron Deficiency Anaemia; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Petry, N.; Olofin, I.; Hurrell, R.F.; Boy, E.; Wirth, J.P.; Moursi, M.; Moira, D.A.; Rohner, F. The proportion of anemia associated with iron deficiency in low, medium, and high Human Development Index countries: A systematic analysis of national surveys. Nutrients 2016, 8, 693. [Google Scholar] [CrossRef] [PubMed]
- WHO. The Global Prevalence of Anaemia in 2011; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Trumbo, P.; Yates, A.A.; Schlicker, S.; Poos, M. Dietary reference intakes: Vitamin A., vitamin K., arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J. Am. Diet Assoc. 2001, 101, 294–301. [Google Scholar] [CrossRef]
- Flores, M.; Macías, N.; Rivera, M.; Barquera, S.; Hernández, L.; García-Guerra, A.; Rivera, J.A. Energy and nutrient intake among Mexican school-aged children, Mexican National Health and Survey 2006. Salud Pub. Mex. 2009, 51, 540–550. [Google Scholar] [CrossRef]
- Finkelstein, J.L.; Layden, A.J.; Stover, P.J. Vitamin B-12 and perinatal health. Adv. Nutr. 2015, 6, 552–563. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, J.L.; Kurpad, A.V.; Thomas, T.; Duggan, C. Vitamin B12 status in pregnant women and their infants in South India. Eur. J. Clin. Nutr. 2017, 71, 1046–1053. [Google Scholar] [CrossRef]
- Villalpando, S.; Perez-Exposito, A.B.; Shamah-Levy, T.; Rivera, J.A. Distribution of anemia associated with micronutrient deficiencies other than iron in a probabilistic sample of Mexican children. Ann. Nutr. Metab. 2006, 50, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Morales-Ruán, M.C.; Villalpando, S.; García-Guerra, A.; Shamah-Levy, T.; Robledo-Pérez, R.; Avila-Arcos, M.A.; Rivera, J. Iron, zinc, copper and magnesium nutritional status in Mexican children aged 1 to 11 years. Salud Pub. Mex. 2012, 54, 125–134. [Google Scholar] [CrossRef]
- WHO. Guideline: Daily Iron Supplementation in Infants and Children; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- McClung, J.P.; Murray-Kolb, L.E. Iron nutrition and premenopausal women: Effects of poor iron status on physical and neuropsychological performance. Annu. Rev. Nutr. 2013, 33, 271–288. [Google Scholar] [CrossRef]
- Haas, J.D.; Brownlie, T., IV. Iron deficiency and reduced work capacity: A critical review of the research to determine a causal relationship. J. Nutr. 2001, 131, 676S–688S. [Google Scholar] [CrossRef]
- Murray-Kolb, L.E.; Beard, J.L. Iron treatment normalizes cognitive functioning in young women. Am. J. Clin. Nutr. 2007, 85, 778–787. [Google Scholar] [CrossRef] [PubMed]
- Graham, R.S.D.; Beebe, S.; Iglesias, C.; Monasterio, I. Breeding for micronutrient density in edile portions of staple food crops: Conventional approaches. Field Crops Res. 1999, 60, 57–80. [Google Scholar] [CrossRef]
- Meenakshi, J.V. Cost-Effectiveness of Biofortification; Best Practice Paper; Copenhagen Consensus Center: Copenhagen, Denmark, 2008. [Google Scholar]
- Nestel, P.; Bouis, H.E.; Meenakshi, J.V.; Pfeiffer, W. Biofortification of staple food crops. J. Nutr. 2006, 136, 1064–1067. [Google Scholar] [CrossRef] [PubMed]
- Hotz, C.; McClafferty, B. From harvest to health: Challenges for developing biofortified staple foods and determining their impact on micronutrient status. Food Nutr. Bull. 2007, 28, S271–S279. [Google Scholar] [CrossRef] [PubMed]
- Haas, J.D.; Beard, J.L.; Murray-Kolb, L.E.; del Mundo, A.M.; Felix, A.; Gregorio, G.B. Iron-biofortified rice improves the iron stores of nonanemic Filipino women. J. Nutr. 2005, 135, 2823–2830. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, J.L.; Mehta, S.; Udipi, S.A.; Ghugre, P.S.; Luna, S.V.; Wenger, M.J.; Murray-Kolb, L.E.; Przybyszewski, E.M.; Haas, J.D. A randomized trial of iron-biofortified pearl millet in school children in India. J. Nutr. 2015, 145, 1576–1581. [Google Scholar] [CrossRef] [PubMed]
- Haas, J.D.; Luna, S.V.; Lung’aho, M.G.; Wenger, M.J.; Murray-Kolb, L.E.; Beebe, S.; Gahutu, J.-B.; Egli, I.M. Consuming iron biofortified beans increases iron status in Rwandan women after 128 days in a randomized controlled feeding trial. J. Nutr. 2016, 146, 1586–1592. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, J.L.; Haas, J.D.; Mehta, S. Iron-biofortified staple food crops for improving iron status: A review of the current evidence. Curr. Opin. Biotechnol. 2017, 44, 138–145. [Google Scholar] [CrossRef]
- Olaiz-Fernández, G.; Rivera-Donmarco, J.; Shamah-Levy, T.; Rojas, R.; Villalpando-Hernández, S.; Hernández-Avila, M.; Sepúlveda-Amor, J. Encuesta National de Salud y Nutricion 2006; Instituto Nacional de Salud Pública: Cuernavaca, Mexico, 2006. (In Spanish) [Google Scholar]
- De Onis, M.; Blössner, M. The World Health Organization Global Database on Child Growth and Malnutrition: Methodology and applications. Int. J. Epidemiol. 2003, 32, 518–526. [Google Scholar] [CrossRef]
- Cook, J.D.; Flowers, C.H.; Skikne, B.S. The quantitative assessment of body iron. Blood 2003, 101, 3359–3364. [Google Scholar] [CrossRef]
- Cohen, J.H.; Haas, J.D. Hemoglobin correction factors for estimating the prevalence of iron deficiency anemia in pregnant women residing at high altitudes in Bolivia. Rev. Panam. Salud Publica 1999, 6, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Namaste, S.M.; Rohner, F.; Huang, J.; Bhushan, N.L.; Flores-Ayala, R.; Kupka, R.; Mei, Z.; Rawat, R.; Williams, A.M.; Raiten, D.J.; et al. Adjusting ferritin concentrations for inflammation: Biomarkers Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA) project. Am. J. Clin. Nutr. 2017, 106, 359S–371S. [Google Scholar] [PubMed]
- Thurnham, D.I.; McCabe, L.D.; Haldar, S.; Wieringa, F.T.; Northrop-Clewes, C.A.; McCabe, G.P. Adjusting plasma ferritin concentrations to remove the effects of subclinical inflammation in the assessment of iron deficiency: A meta-analysis. Am. J. Clin. Nutr. 2010, 92, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Thurnham, D.I.; Northrop-Clewes, C.A.; Knowles, J. The use of adjustment factors to address the impact of inflammation on vitamin A and iron status in humans. J. Nutr. 2015, 145, 1137S–1143S. [Google Scholar] [CrossRef] [PubMed]
- Larson, L.M.; Addo, O.; Sandalinas, F.; Faigao, K.; Kupka, R.; Flores-Ayala, R.; Suchdev, P. Accounting for the influence of inflammation on retinol-binding protein in a population survey of Liberian preschool-age children. Matern. Child Nutr. 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Petry, N.; Egli, I.; Zeder, C.; Walczyk, T.; Hurrell, R. Polyphenols and phytic acid contribute to the low iron bioavailability from common beans in young women. J. Nutr. 2010, 140, 1977–1982. [Google Scholar] [CrossRef] [PubMed]
- Kodkany, B.S.; Bellad, R.M.; Mahantshetti, N.S.; Westcott, J.E.; Krebs, N.F.; Kemp, J.F.; Hambidge, K.M. Biofortification of pearl millet with iron and zinc in a randomized controlled trial increases absorption of these minerals above physiologic requirements in young children. J. Nutr. 2013, 143, 1489–1493. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; Centers for Disease Control and Prevention. Assessing the Iron Status of Populations; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- Camaschella, C. New insights into iron deficiency and iron deficiency anemia. Blood Rev. 2017, 31, 225–233. [Google Scholar] [CrossRef]
- Suchdev, P.S.; William, A.M.; Mei, Z.; Flores-Ayala, R.; Pasricha, S.-R.; Rogers, L.M.; Namaste, S.M.L. Assessment of iron status in settings of inflammation: Challenges and potential approaches. Am. J. Clin. Nutr. 2017, 106, 1626S–1633S. [Google Scholar] [CrossRef]
- Ganz, T. Iron and infection. Int. J. Hematol. 2017, 107, 7–15. [Google Scholar] [CrossRef]
| Study Population 1 | Total (n = 574) | Fe-Beans (n = 269) | Control-Beans (n = 305) |
|---|---|---|---|
| Median ± IQR/n (%) | Median ± IQR/n (%) | Median ± IQR/n (%) | |
| Age, years | 9.57 (8.08, 10.89) | 9.46 (8.06, 10.67) | 9.67 (8.09, 11.04) |
| Girls, n (%) | 306 (53.31) | 134 (49.81) | 172 (56.39) |
| Weight, kg | 25.90 (22.10, 30.70) | 25.50 (21.80, 30.50) | 26.35 (22.50, 31.00) |
| Height, cm | 124.90 (118.00, 132.55) | 124.70 (117.60, 132.35) | 125.30 (118.40, 132.85) |
| Body Mass Index (BMI), kg/m2 | 16.63 (15.73, 17.66) | 16.36 (15.49, 17.53) | 16.77 (15.96, 17.78) |
| BMI z-score (BMIZ) | 0.16 (−0.30, 0.65) | 0.11 (−0.38, 0.65) | 0.20 (−0.20, 0.65) |
| BMIZ <−2 | 2 (0.35) | 1 (0.37) | 1 (0.33) |
| Biomarkers | |||
| Hemoglobin 2 g/dL | 12.82 (11.84, 13.83) | 12.89 (11.89, 14.09) | 12.74 (11.74, 13.64) |
| Anemic | 102 (17.77) | 47 (17.47) | 55 (18.03) |
| Serum ferritin 4 µg/L | 28.70 (20.40, 38.90) | 28.00 (19.00, 38.10) | 29.20 (21.80, 39.50) |
| <15.0 µg/L | 65 (11.32) | 40 (14.87) 3 | 25 (8.20) 3 |
| <20.0 µg/L | 138 (24.04) | 77 (28.62) | 61 (20.00) |
| <30.0 µg/L | 309 (53.83) | 147 (54.65) | 162 (53.11) |
| Serum ferritin4 (BRINDA adjusted), µg/L | 24.51 (18.18, 33.80) | 24.20 (15.92, 32.12) | 25.10 (18.58, 35.61) |
| <15.0 µg/L | 90 (15.90) | 58 (22.05) 3 | 32 (10.56) 3 |
| <20.0 µg/L | 182 (32.16) | 97 (36.88) | 85 (28.05) |
| <30.0 µg/L | 383 (67.67) | 184 (69.96) | 199 (65.68) |
| sTfR 4 (Ramco corrected), mg/L | 4.31 (3.99, 4.68) | 4.33 (4.04, 4.69) | 4.31 (3.97, 4.65) |
| >8.3 mg/L | 0 (0.00) | 0 (0.00) | 0 (0.00) |
| Total body iron, mg/kg | 5.37 (4.14, 6.51) | 5.25 (3.80, 6.42) | 5.54 (4.40, 6.61) |
| <0 mg/kg | 4 (0.70) | 4 (1.49) | 0 (0.00) |
| Total body iron (BRINDA adjusted), mg/kg | 4.79 (3.63, 5.97) | 4.63 (3.20, 5.77) | 4.94 (3.86, 6.11) |
| <0 mg/kg | 5 (0.88) | 5 (1.90) | 0 (0.00) |
| CRP 4 mg/L | 0.37 (0.17, 0.99) | 0.38 (0.18, 1.04) | 0.35 (0.15, 0.80) |
| >5 mg/L | 36 (6.32) | 11 (4.12) 3 | 25 (8.25) 3 |
| AGP 4 g/L | 0.70 (0.56, 0.84) | 0.69 (0.56, 0.80) | 0.71 (0.57, 0.89) |
| >1 g/L | 66 (11.58) | 17 (6.42) 3 | 49 (16.07) 3 |
| Intervention | |||
| Iron concentration in experimental beans, µg/g | - | 94 | 54 |
| Maximum potential number of feeding days | 104.00 (100.00, 108.00) | 104.00 (100.00, 104.00) | 104.00 (100.00, 109.00) |
| Actual number of feeding days | 68.00 (52.00, 75.00) | 68.00 (51.00, 75.00) | 67.00 (54.00, 75.00) |
| Total beans experimentally consumed, g | 11965.00 (8042.00, 14090.00) | 11786.00 (8227.00, 14405.00) | 12022.50 (8006.50, 13876.00) |
| Total iron intake from dry experimental beans, mg | 337.42 (256.28, 493.20) | 503.58 (351.52, 615.49) | 295.10 (196.52, 340.59) |
| Total iron absorbed from dry beans (5.0%), mg | 16.87 (12.81, 24.66) | 25.18 (17.58, 30.77) | 14.75 (9.83, 17.03) |
| Fe-Beans | Control-Beans | Intervention | ||||
|---|---|---|---|---|---|---|
| Observed Outcomes at the Endline | n | Median ± IQR/n (%) | N | Median ± IQR/n (%) | β (SE) or RR (95% CI) | p-value 1 |
| Hemoglobin 2 g/dL | 269 | 12.89 (11.92, 13.59) | 305 | 13.00 (12.20, 13.84) | −0.18 (0.26) | 0.50 |
| Anemic | 41 (15.24) | 39 (12.79) | 1.19 (0.54, 2.60) | 0.66 | ||
| Serum ferritin 3 µg/L | 269 | 30.55 (23.26, 39.62) | 305 | 35.55 (26.23, 48.44) | −0.17 (0.08) | 0.04 |
| <15.0 µg/L | 16 (5.95) | 4 (1.31) | 3.42 (1.07, 10.97) | 0.04 | ||
| <20.0 µg/L | 43 (15.99) | 23 (7.54) | 1.88 (0.97, 3.62) | 0.06 | ||
| <30.0 µg/L | 129 (47.96) | 117 (38.36) | 1.24 (0.88, 1.74) | 0.21 | ||
| Serum ferritin (BRINDA) 3 µg/L | 267 | 29.03 (22.12, 37.00) | 305 | 33.13 (24.04, 43.18) | −0.10 (0.07) | 0.17 |
| <15.0 µg/L | 20 (7.49) | 7 (2.30) | 2.32 (0.92, 5.83) | 0.07 | ||
| <20.0 µg/L | 48 (17.98) | 37 (12.13) | 1.30 (0.75, 2.26) | 0.35 | ||
| <30.0 µg/L | 143 (53.56) | 134 (43.93) | 1.15 (0.86, 1.53) | 0.35 | ||
| sTfR (Ramco corrected) 3 mg/L | 269 | 4.28 (4.02, 4.57) | 305 | 4.42 (4.06, 4.89) | −0.05 (0.02) | 0.05 |
| Total body iron, mg/kg | 269 | 5.62 (4.64, 6.54) | 305 | 6.00 (5.07, 7.08) | −0.43 (0.21) | 0.05 |
| Total body iron (BRINDA), mg/kg | 267 | 5.40 (4.48, 6.32) | 305 | 5.61 (4.79, 6.65) | −0.18 (0.18) | 0.34 |
| CRP 3 mg/L | 268 | 0.27 (0.10, 0.63) | 305 | 0.36 (0.10, 0.90) | –0.33 (0.13) | 0.03 |
| >5.0 mg/L | 10 (3.73) | 18 (5.90) | 0.60 (0.27, 1.32) | 0.20 | ||
| AGP 3 g/L | 268 | 0.70 (0.62, 0.81) | 305 | 0.66 (0.57, 0.84) | 0.05 (0.03) | 0.12 |
| >1.0 g/L | 18 (6.72) | 41 (13.44) | 0.51 (0.25, 1.03) | 0.06 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finkelstein, J.L.; Mehta, S.; Villalpando, S.; Mundo-Rosas, V.; Luna, S.V.; Rahn, M.; Shamah-Levy, T.; Beebe, S.E.; Haas, J.D. A Randomized Feeding Trial of Iron-Biofortified Beans in School Children in Mexico. Nutrients 2019, 11, 381. https://doi.org/10.3390/nu11020381
Finkelstein JL, Mehta S, Villalpando S, Mundo-Rosas V, Luna SV, Rahn M, Shamah-Levy T, Beebe SE, Haas JD. A Randomized Feeding Trial of Iron-Biofortified Beans in School Children in Mexico. Nutrients. 2019; 11(2):381. https://doi.org/10.3390/nu11020381
Chicago/Turabian StyleFinkelstein, Julia L., Saurabh Mehta, Salvador Villalpando, Veronica Mundo-Rosas, Sarah V. Luna, Maike Rahn, Teresa Shamah-Levy, Stephen E. Beebe, and Jere D. Haas. 2019. "A Randomized Feeding Trial of Iron-Biofortified Beans in School Children in Mexico" Nutrients 11, no. 2: 381. https://doi.org/10.3390/nu11020381
APA StyleFinkelstein, J. L., Mehta, S., Villalpando, S., Mundo-Rosas, V., Luna, S. V., Rahn, M., Shamah-Levy, T., Beebe, S. E., & Haas, J. D. (2019). A Randomized Feeding Trial of Iron-Biofortified Beans in School Children in Mexico. Nutrients, 11(2), 381. https://doi.org/10.3390/nu11020381






