Assessment of Vitamin C and Antioxidant Profiles in Saliva and Serum in Patients with Periodontitis and Ischemic Heart Disease
Abstract
1. Introduction
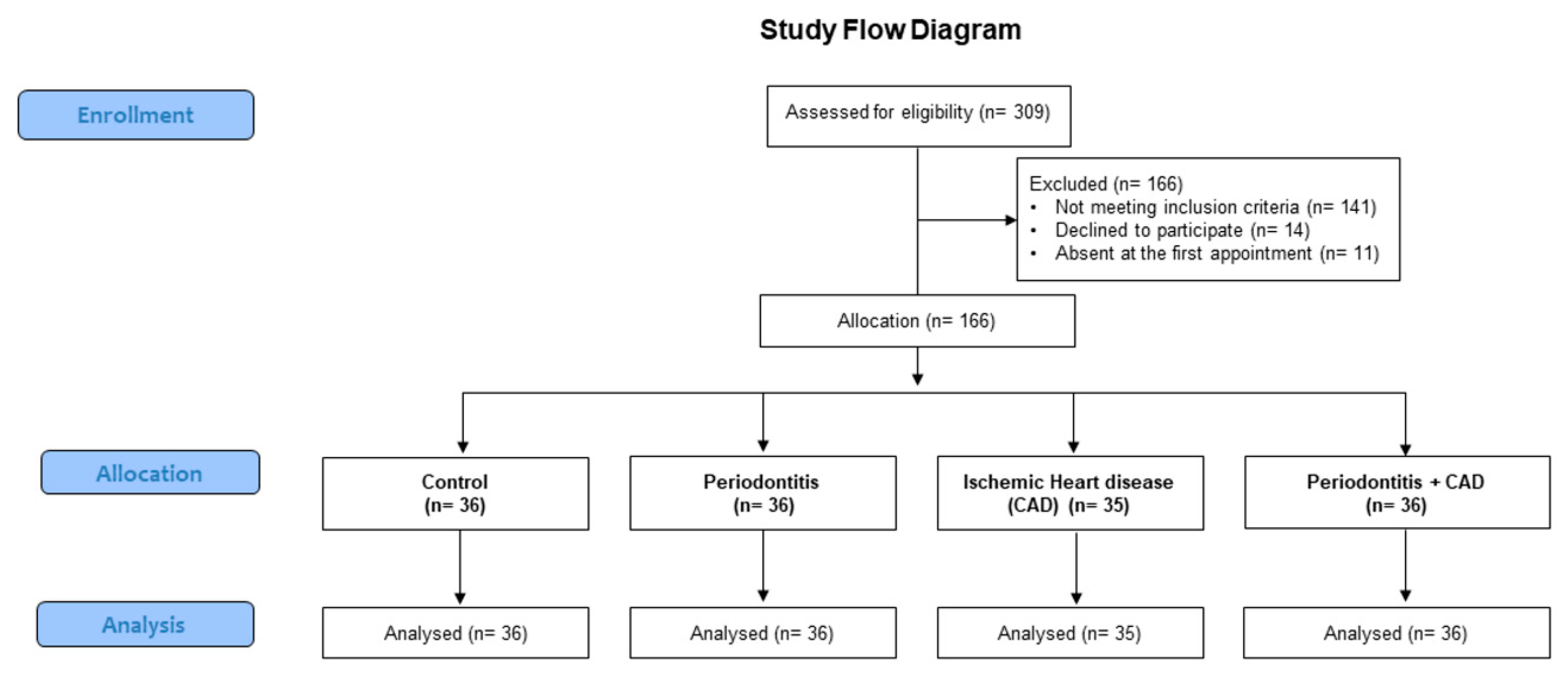
2. Materials and Methods
2.1. Study Design
2.2. Vitamin C Assessment in Saliva and Serum
2.3. Statistical Analysis
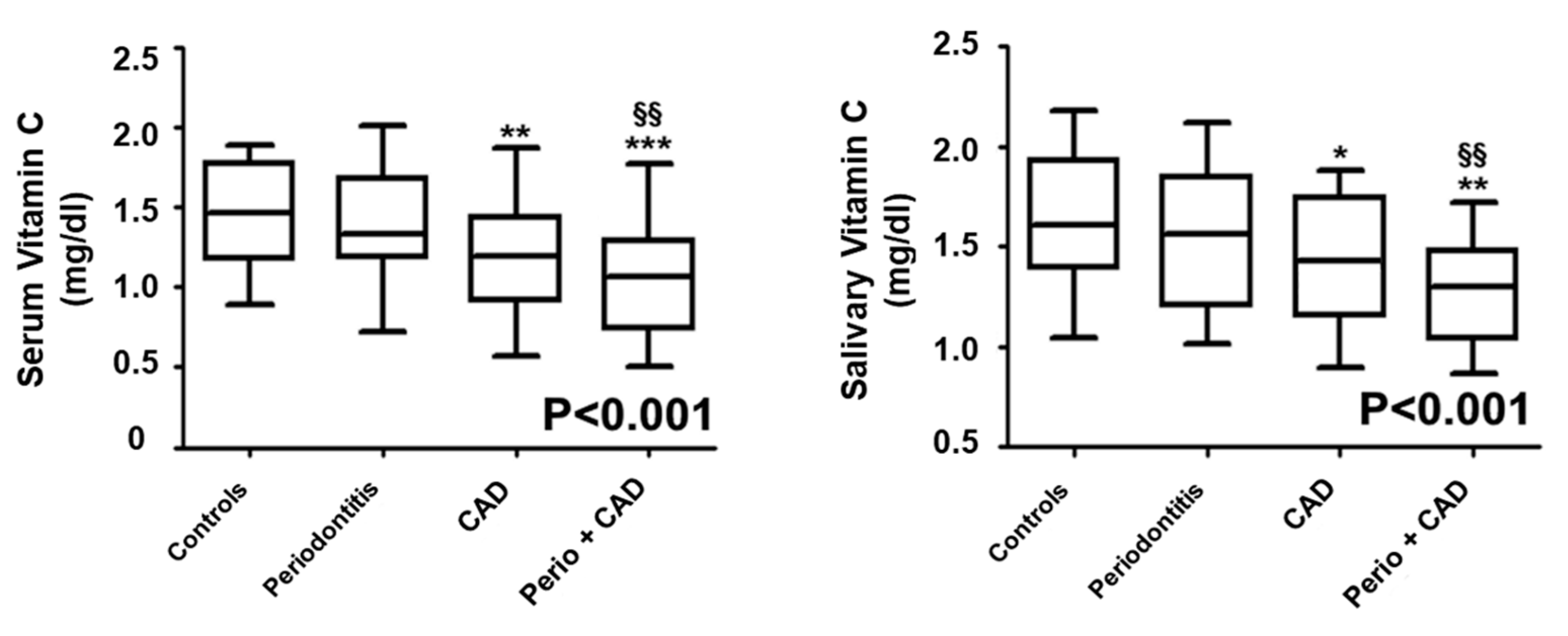
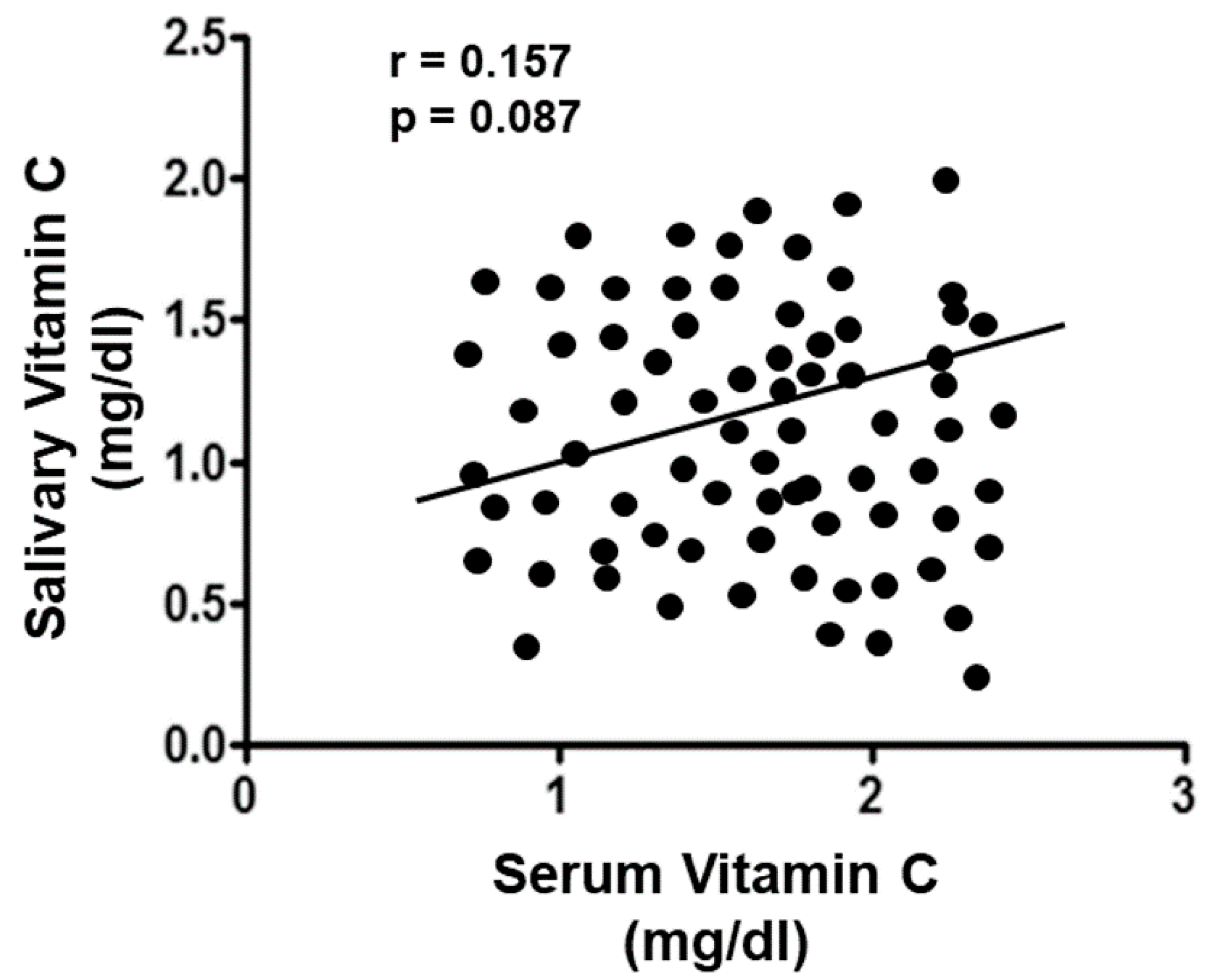
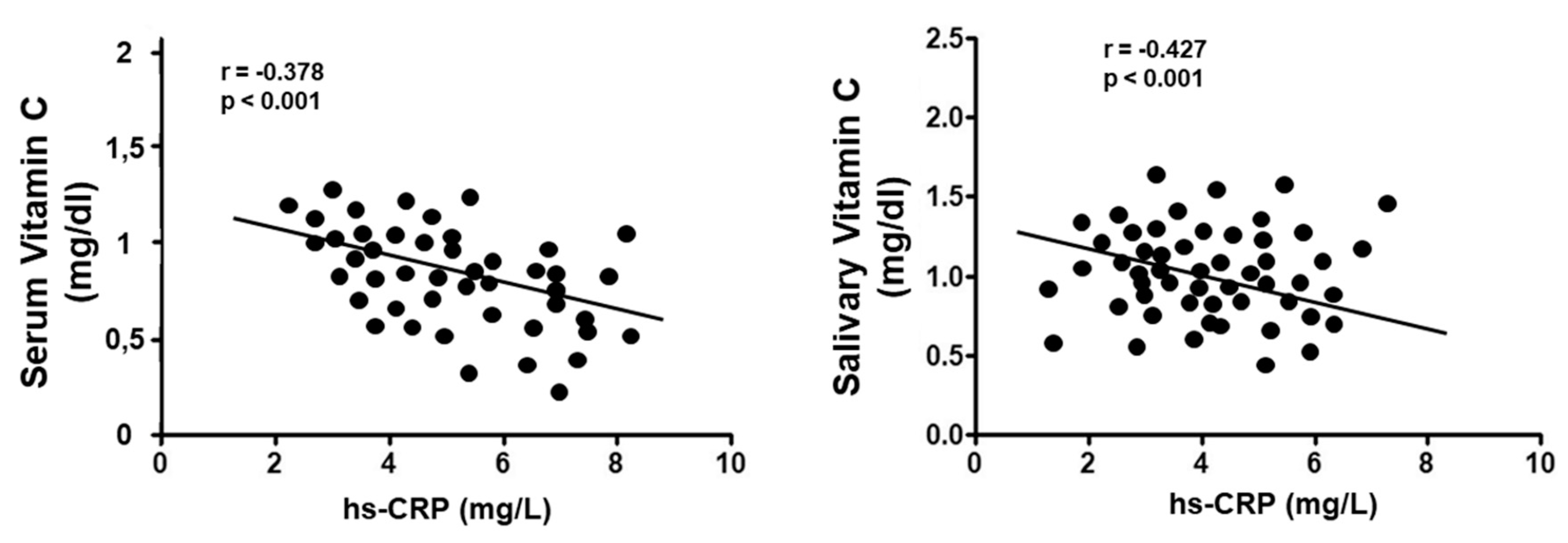
3. Results
Vitamin C Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Eke, P.I.; Wei, L.; Thornton-Evans, G.O.; Borrell, L.N.; Borgnakke, W.S.; Dye, B.; Genco, R.J. Risk Indicators for Periodontitis in US Adults: NHANES 2009 to 2012. J. Periodontol. 2016, 87, 1174–1185. [Google Scholar] [CrossRef] [PubMed]
- Isola, G.; Polizzi, A.; Santonocito, S.; Alibrandi, A.; Ferlito, S. Expression of Salivary And Serum Malondialdehyde And Lipid Profile Of Patients With Periodontitis And Coronary Heart Disease. Int. J. Mol. Sci. 2019, 20, 6061. [Google Scholar] [CrossRef]
- Bernabe, E.; Dahiya, M.; Bhandari, B.; Murray, C.J.; Marcenes, W. Global burden of severe periodontitis in 1990–2010: A systematic review and meta-regression. J. Dent. Res. 2014, 93, 1045–1053. [Google Scholar]
- Friedewald, V.E.; Kornman, K.S.; Beck, J.D.; Genco, R.; Goldfine, A.; Libby, P.; Offenbacher, S.; Ridker, P.M.; Van Dyke, T.E.; Roberts, W.C. American Journal of Cardiology; Journal of Periodontology. The American Journal of Cardiology and Journal of Periodontology editors’ consensus: Periodontitis and atherosclerotic cardiovascular disease. J. Periodontol. 2009, 80, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Seymour, G.J.; Palmer, J.E.; Leishman, S.J.; Do, H.L.; Westerman, B.; Carle, A.D.; Faddy, M.J.; West, M.J.; Cullinan, M.P. Influence of a triclosan toothpaste on periodontopathic bacteria and periodontitis progression in cardiovascular patients: A randomized controlled trial. J. Periodontal Res. 2017, 52, 61–73. [Google Scholar] [CrossRef]
- Holmlund, A.; Holm, G.; Lind, L. Number of teeth as a predictor of cardiovascular mortality in a cohort of 7,674 subjects followed for 12 years. J. Periodontol. 2010, 81, 870–876. [Google Scholar] [CrossRef]
- Isola, G.; Alibrandi, A.; Currò, M.; Matarese, M.; Ricca, S.; Matarese, G.; Kocher, T. Evaluation of salivary and serum ADMA levels in patients with periodontal and cardiovascular disease as subclinical marker of cardiovascular risk. J. Periodontol 2019, in press. [Google Scholar]
- Isola, G.; Matarese, G.; Ramaglia, L.; Pedullà, E.; Rapisarda, E.; Iorio-Siciliano, V. Association between periodontitis and glycosylated hemoglobin before diabetes onset: A cross-sectional study. Clin. Oral Investig. 2019. [Google Scholar] [CrossRef]
- Vidal, F.; Figueredo, C.M.; Cordovil, I.; Fischer, R.G. Periodontal therapy reduces plasma levels of interleukin-6, C-reactive protein, and fibrinogen in patients with severe periodontitis and refractory arterial hypertension. J. Periodontol. 2009, 80, 786–791. [Google Scholar] [CrossRef]
- Isola, G.; Lo Giudice, A.; Polizzi, A.; Alibrandi, A.; Patini, R.; Ferlito, S. Association of Circulating Progenitor Cells Levels during Periodontitis. Genes 2019, in press. [Google Scholar]
- Tyml, K. Vitamin C and Microvascular Dysfunction in Systemic Inflammation. Antioxidants (Basel) 2017, 6, 49. [Google Scholar] [CrossRef] [PubMed]
- Hampton, T.G.; Amende, I.; Fong, J.; Laubach, V.E.; Li, J.; Metais, C.; Simons, M. Basic FGF reduces stunning via a NOS2-dependent pathway in coronary perfused mouse hearts. Am. J. Physiol. Heart Circ. Physiol. 2000, 279, H260–H268. [Google Scholar] [CrossRef] [PubMed]
- Engler, M.M.; Engler, M.B.; Malloy, M.J.; Chiu, E.Y.; Schloetter, M.C.; Paul, S.M.; Stuehlinger, M.; Lin, K.Y.; Cooke, J.P.; Morrow, J.D.; et al. Antioxidant vitamins C and E improve endothelial function in children with hyperlipidemia: Endothelial Assessment of Risk from Lipids in Youth (EARLY) Trial. Circulation 2003, 108, 1059–1063. [Google Scholar] [CrossRef] [PubMed]
- Song, E.K.; Kang, S.M. Vitamin C Deficiency, High-Sensitivity C-Reactive Protein, and Cardiac Event-Free Survival in Patients with Heart Failure. J. Cardiovasc. Nurs. 2018, 33, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, R.; Hasson, D.; Prieto, J.C.; Dussaillant, G.; Ramos, C.; León, L.; Gárate, J.; Valls, N.; Gormaz, J.G. The effectiveness of antioxidant vitamins C and E in reducing myocardial infarct size in patients subjected to percutaneous coronary angioplasty (PREVEC Trial): Study protocol for a pilot randomized double-blind controlled trial. Trials 2014, 15, 192. [Google Scholar] [CrossRef] [PubMed]
- Isola, G.; Alibrandi, A.; Rapisarda, E.; Matarese, G.; Williams, R.C.; Leonardi, R. Association of vitamin d in patients with periodontal and cardiovascular disease: A cross-sectional study. J. Periodontal Res. 2019, in press. [Google Scholar]
- Tonetti, M.S.; D’ Aiuto, F.; Nibali, L.; Donald, A.; Storry, C.; Parkar, M.; Suvan, J.; Hingorani, A.D.; Vallance, P.; Deanfield, J. Treatment of periodontitis and endothelial function. New Engl. J. Med. 2007, 356, 911–920. [Google Scholar] [CrossRef]
- Holtfreter, B.; Empen, K.; Glaser, S.; Gläser, S.; Lorbeer, R.; Völzke, H.; Ewert, R.; Kocher, T.; Dörr, M. Periodontitis is associated with endothelial dysfunction in a general population: A cross-sectional study. PLoS ONE 2013, 8, e84603. [Google Scholar] [CrossRef]
- Andrukhov, O.; Haririan, H.; Bertl, K.; Rausch, W.D.; Bantleon, H.P.; Moritz, A.; Rausch-Fan, X. Nitric oxide production, systemic inflammation and lipid metabolism in periodontitis patients: Possible gender aspect. J. Clin. Periodontol. 2013, 40, 916–923. [Google Scholar] [CrossRef]
- Huang, A.L.; Vita, J.A. Effects of systemic inflammation on endothelium-dependent vasodilation. Trends Cardiovasc. Med. 2006, 16, 15–20. [Google Scholar] [CrossRef]
- Gurav, A.N. The implication of periodontitis in vascular endothelial dysfunction. Eur. J. Clin. Investig. 2014, 44, 1000–1009. [Google Scholar] [CrossRef] [PubMed]
- Mah, E.; Matos, M.D.; Kawiecki, D.; Ballard, K.; Guo, Y.; Volek, J.S.; Bruno, R.S. Vitamin C status is related to proinflammatory responses and impaired vascular endothelial function in healthy, college-aged lean and obese men. J. Am. Diet. Assoc. 2011, 111, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Nishida, M.; Grossi, S.G.; Dunford, R.G.; Ho, A.W.; Trevisan, M.; Genco, R.J. Dietary vitamin C and the risk for periodontal disease. J. Periodontol. 2000, 71, 1215–1223. [Google Scholar] [CrossRef] [PubMed]
- Kendall, H.K.; Marshall, R.I.; Bartold, P.M. Nitric oxide and tissue destruction. Oral Dis. 2001, 7, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Amarasena, N.; Ogawa, H.; Yoshihara, A.; Hanada, N.; Miyazaki, H. Serum vitamin C-periodontal relationship in community-dwelling elderly Japanese. J. Clin. Periodontol. 2005, 32, 93–97. [Google Scholar] [CrossRef]
- Harej, A.; Macan, A.M.; Stepanić, V.; Klobučar, M.; Pavelić, K.; Pavelić, S.K.; Raić-Malić, S. The Antioxidant and Antiproliferative Activities of 1,2,3-Triazolyl-L-Ascorbic Acid Derivatives. Int. J. Mol. Sci. 2019, 20, 4735. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting ofObservational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Periodontol. 2008, 61, 344–349. [Google Scholar]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Periodontol. 2018, 89, S159–S172. [Google Scholar] [CrossRef]
- Isola, G.; Matarese, M.; Ramaglia, L.; Iorio-Siciliano, V.; Cordasco, G.; Matarese, G. Efficacy of a drug composed of herbal extracts on postoperative discomfort after surgical removal of impacted mandibular third molar: A randomized, triple-blind, controlled clinical trial. Clin. Oral Investig. 2019, 23, 2443–2453. [Google Scholar] [CrossRef]
- Bassand, J.P.; Hamm, C.W.; Ardissino, D.; Boersma, E.; Budaj, A.; Fernandez-Aviles, F.; Fox, K.A.; Hasdai, D.; Ohman, E.M.; Wallentin, L.; et al. Guidelines for the diagnosis and treatment of non-ST-segment elevation acute coronary syndromes: The Task Force for the Diagnosis and Treatment of Non-ST-Segment Elevation Acute Coronary Syndromes of the European Society of Cardiology. Eur. Heart J. 2007, 28, 1598–1660. [Google Scholar] [CrossRef]
- O’Leary, T.J.; Drake, R.B.; Naylor, J.E. The plaque control record. J. Periodontol. 1972, 43, 38. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: A systematic review and dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2018, 108, 1069–1091. [Google Scholar] [CrossRef] [PubMed]
- Polidori, M.C.; Ruggiero, C.; Croce, M.F.; Raichi, T.; Mangialasche, F.; Cecchetti, R.; Pelini, L.; Paolacci, L.; Ercolani, S.; Mecocci, P. Association of increased carotid intima-media thickness and lower plasma levels of vitamin C and vitamin E in old age subjects: Implications for Alzheimer’s disease. J. Neural. Transm. (Vienna) 2015, 122, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Mathias, T.M.; Silva, J.F.; Sapata, V.M.; Marson, F.C.; Zanoni, J.N.; Silva, C.O. Evaluation of the effects of periodontal treatment on levels of ascorbic acid in smokers. J. Int. Acad. Periodontol. 2014, 16, 109–114. [Google Scholar] [PubMed]
- Ellulu, M.S.; Rahmat, A.; Patimah, I.; Khaza’ai, H.; Abed, Y. Effect of vitamin C on inflammation and metabolic markers in hypertensive and/or diabetic obese adults: A randomized controlled trial. Drug Des. Dev. Ther. 2015, 9, 3405–3412. [Google Scholar] [CrossRef] [PubMed]
- Amaliya, A.; Laine, M.L.; Loos, B.G.; Van der Velden, U. Java project on periodontal diseases: Effect of vitamin C/calcium threonate/citrus flavonoids supplementation on periodontal pathogens, CRP and HbA1c. J. Clin. Periodontol. 2015, 42, 1097–1104. [Google Scholar] [CrossRef]
- Golatowski, C.; Salazar, M.G.; Dhople, V.M.; Hammer, E.; Kocher, T.; Jehmlich, N.; Völker, U. Comparative evaluation of saliva collection methods for proteome analysis. Clin. Chim. Acta 2013, 419, 42–46. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.J. Total antioxidant status in plasma and body fluids. Methods Enzymol. 1994, 234, 279–293. [Google Scholar]
- Battino, M.; Ferreiro, M.S.; Gallardo, I.; Newman, H.N.; Bullon, P. The antioxidant capacity of saliva. J. Clin. Periodontol. 2002, 29, 189–194. [Google Scholar] [CrossRef]
- Rausch-Fan, X.; Matejka, M. From plaque formation to periodontal disease, is there a role for nitric oxide? Eur. J. Clin. Investig. 2001, 31, 833–835. [Google Scholar] [CrossRef]
- Matejka, M.; Patyka, L.; Ulm, C.; Solar, P.; Sinzinger, H. Nitric oxide synthesis is increased in periodontal disease. J. Period. Res. 1998, 33, 517–518. [Google Scholar] [CrossRef] [PubMed]
- Gullu, C.; Ozmeric, N.; Tokman, B.; Elgun, S.; Balos, K. Effectiveness of scaling and root planing versus modified Widman flap on nitric oxide synthase and arginase activity in patients with chronic periodontitis. J. Period. Res. 2005, 40, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Ozer, L.; Elgun, S.; Ozdemir, B.; Pervane, B.; Ozmeric, N. Arginine-nitric oxide-polyamine metabolism in periodontal disease. J. Periodontol. 2011, 82, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Patini, R.; Staderini, E.; Gallenzi, P. Multidisciplinary surgical management of Cowden syndrome: Report of a case. J. Clin. Exp. Dent. 2016, 8, e472–e474. [Google Scholar] [CrossRef] [PubMed]
- Ekuni, D.; Tomofuji, T.; Sanbe, T.; Irie, K.; Azuma, T.; Maruyama, T.; Tamaki, N.; Murakami, J.; Kokeguchi, S.; Yamamoto, T. Vitamin C intake attenuates the degree of experimental atherosclerosis induced by periodontitis in the rat by decreasing oxidative stress. Arch. Oral Biol. 2009, 54, 495–502. [Google Scholar] [CrossRef]
- Kaur, G.; Kathariya, R.; Bansal, S.; Singh, A.; Shahakar, D. Dietary antioxidants and their indispensable role in periodontal health. J. Food Drug Anal. 2016, 24, 239–246. [Google Scholar] [CrossRef]
- Caccianiga, G.; Paiusco, A.; Perillo, L.; Nucera, R.; Pinsino, A.; Maddalone, M.; Cordasco, G.; Lo Giudice, A. Does low-level laser therapy enhance the efficiency of orthodontic dental alignment? Results from a randomized pilot study. Photomed. Laser Surg. 2017, 35, 421–426. [Google Scholar] [CrossRef]
- Caccianiga, G.; Crestale, C.; Cozzani, M.; Piras, A.; Mutinelli, S.; Lo Giudice, A.; Cordasco, G. Low-level laser therapy and invisible removal aligners. J. Biol. Regul. Homeost. Agents 2016, 30, 107–113. [Google Scholar]
- Takahama, U.; Hirota, S.; Oniki, T. Quercetin-dependent scavenging of reactive nitrogen species derived from nitric oxide and nitrite in the human oral cavity: Interaction of quercetin with salivary redox components. Arch. Oral Biol. 2006, 51, 629–639. [Google Scholar] [CrossRef]
| Controls (N = 36) | Periodontiti (N = 36) | CAD (N = 35) | Periodontitis + CAD (N = 36) | |
|---|---|---|---|---|
| Age (years) | 54 (51; 56) | 55 (51; 57) | 54 (48; 57) | 55 (50; 56) |
| Gender (male/female) | 17/19 | 18/18 | 19/17 | 17/19 |
| Education level | ||||
| Primary school, n (%) | 13 (36.1) | 12 (33.3) | 11 (31.4) | 13 (36.1) |
| High school, n (%) | 12 (33.3) | 14 (38.9) | 13 (37.1) | 13 (36.1) |
| College/university, n (%) | 11 (30.5) | 10 (27.8) | 11 (31.4) | 10 (27.8) |
| Body mass index (kg/m2) | 25.6 (21.9; 27.7) | 25.1 (22.8; 26.7) | 25.9 (21.8; 28.4) | 25.5 (21.7; 27.1) |
| Fasting glucose (mg/dL) | 93.5 (87.7; 97.9) | 94.4 (84.3; 106.2) | 92.6 (87.9; 109.1) | 93.1 (88.7; 111.2) |
| Current smokers, n (%) | 3 (8.3) | 3 (8.3) | 2 (5.7) | 3 (8.3) |
| Comorbidities | ||||
| Diabetes, n (%) | 2 (14.2) ** | 3 (8.6) ** | 3 (8.3) ** | |
| Previous CVD | ||||
| Atrial fibrillation, n (%) | 7 (20) **,§§ | 10 (27.8) **,§§ | ||
| Angina pectoris, n (%) | 16 (45.7) **,§§ | 17 (48.6) **,§§ | ||
| Stroke, n (%) | 7 (20) **,§§ | 9 (25.7) **,§§ | ||
| Heart failure, n (%) | 9 (25.7) **,§§ | 10 (27.8) **,§§ | ||
| Drug treatment of CVD | ||||
| Antihypertensive, n (%) | 13 (37.1) **,§§ | 14 (38.9) **,§§ | ||
| Statins, n (%) | 9 (25.7) **,§§ | 10 (27.8) **,§§ | ||
| Low-dose aspirin, n (%) | 9 (25.7) **,§§ | 11 (30.5) **,§§ | ||
| Beta blockers, n (%) | 10 (28.5) **,§§ | 12 (33.3) **,§§ | ||
| hs-CRP (mg/L) | 2.8 (2.2; 3.1) | 3.7 (2.9; 4.2) * | 6.4 (5.1; 6.9) ** | 6.8 (5.9; 7.7) **,§§,# |
| Total cholesterol (mg/dL) | 174 (131; 189) | 178 (152; 194) | 181 (155; 197) *,§§ | 180 (149; 200) |
| Triglycerides (mg/dL) | 130 (111; 144) | 131 (57; 151) | 134 (109; 157) *,§§ | 136 (117; 161) |
| Retinol (µmol/L) | 2.12 (1.75; 2.47) | 2.08 (1.89; 2.09) | 2.05 (1.81; 2.07) *,§§ | 2.04 (1.78; 2.15) *,§§ |
| α-carotene (µmol/L) | 0.081 (0.051;0.123) | 0.075 (0.041; 0.101) | 0.073 (0.051; 0.123) *,§§ | 0.071 (0.049; 0.112) *,§§ |
| β-carotene (µmol/L) | 0.321 (0.22; 0.367) | 0.317 (0.209; 0.361) | 0.311 (0.278; 0.302) *,§§ | 0.309 (0.214; 0.355) *,§§ |
| β-cryptoxanthin (µmol/L) | 0.066 (0.043; 0.78) | 0.059 (0.033; 0.71) | 0.051 (0.029; 0.82) *,§§ | 0.048 (0.039; 0.64) *,§§ |
| γ-tocopherol (µmol/L) | 2.66 (2.35; 2.99) | 2.63 (2.21; 2.73) | 2.59 (2.12; 2.85) *,§§ | 2.57 (2.04; 2.78) *,§§ |
| Lutein (µmol/L) | 0.16 (0.11; 0.26) | 0.14 (0.1; 0.19) | 0.12 (0.08; 0.18) *,§§ | 0.11 (0.09; 0.21) *,§§ |
| Zeaxanthin (µmol/L) | 0.037 (0.024; 0.045) | 0.034 (0.018; 0.039) | 0.029 (0.021; 0.027) *,§§ | 0.031 (0.021; 0.042) *,§§ |
| Lycopene (µmol/L) | 0.36 (0.21; 0.42) | 0.35 (0.22; 0.41) | 0.32 (0.21; 0.39) *,§§ | 0.31 (0.19; 0.36) * |
| Controls (N = 36) | Periodontitis (N = 36) | CAD (N = 35) | Periodontitis + CAD (N = 36) | |
|---|---|---|---|---|
| N of teeth | 26 (24; 28) | 19 (17; 20) ** | 23 (20; 24) **,§§ | 18 (13; 20) **,## |
| CAL (mm) | 1.1 (0.8; 1.3) | 4 (3.5; 4.2) ** | 2.1 (1.7; 2.4) **,§§ | 4.1 (3.6; 4.8) **,## |
| CAL 4–5 mm (% sites) | 38.7 (36.2; 43.4) ** | 42.2 (38.9; 48.7) **,## | ||
| CAL ≥6 mm (% sites) | 20.2 (16.8; 21.7) ** | 18.2 (16.4; 24.2) **,## | ||
| PD (mm) | 1.4 (1.1; 1.8) | 4.5 (4.1; 5.2) ** | 2 (1.9; 2.3) **,§§ | 4.1 (3.8; 4.7) **,## |
| PD 4–5 mm (% sites) | 42.1 (40.1; 46.4) ** | 44.8 (41.5; 51.1) **,## | ||
| PD ≥6 mm (% sites) | 22.3 (17.9; 23.1) ** | 23.9 (21.6; 27.6) **,§§,## | ||
| BOP (%) | 8.8 (6.1; 10.9) | 47.1 (45.1; 48.9) ** | 8.7 (5.2; 9.2) **,§§ | 45.7 (44.6; 56.2) **,§§,## |
| PI (%) | 6.9 (5.3; 10.8) | 34.9 (33.3; 36.1) ** | 12.9 (12.1; 13.4) **,§§ | 34.3 (31.2; 35.1) **,## |
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| Variable | B | 95% CI | p | B | 95% CI | p |
| Serum Vitamin C levels | ||||||
| CAD | −0.378 | −0.222; 0.578 | <0.001 | −0.069 | −0.341; 0.498 | 0.644 |
| Periodontitis | −0.223 | −0.016; 0.404 | 0.034 | −0.141 | −0.039; 0.389 | 0.112 |
| hs-CRP | −0.119 | −0.075; 0.137 | <0.001 | −0.112 | 0.065; 0.149 | <0.001 |
| Age (years) | 0.078 | 0.29; 0.004 | 0.081 | 0.039 | −0.148; 0.187 | 0.436 |
| Female gender | −0.149 | −0.041; 0.327 | 0.149 | −0.178 | −0.112; 0.344 | 0.078 |
| Triglycerides | −0.066 | −0.199; 0.078 | 0.209 | 0.074 | −0.167; 0.366 | 0.141 |
| Salivary Vitamin C levels | ||||||
| CAD | −0.236 | 0.134; 0.41 | <0.001 | −0.029 | −0.433; 0.312 | 0.655 |
| Periodontitis | −0.064 | −0.087; 0.214 | 0.387 | 0.005 | −0.151; 0.184 | 0.972 |
| hs-CRP | −0.078 | 0.038; 0.132 | <0.001 | 0.077 | 0.061; 0.146 | <0.001 |
| Age (years) | 0.041 | −0.041; 0.012 | 0.207 | 0.012 | −0.029; 0.036 | 0.786 |
| Female gender | −0.038 | −0.114; 0.226 | 0.419 | 0.079 | −0.047; 0.239 | 0.178 |
| Triglycerides | 0.079 | −0.178; 0.006 | 0.039 | −0.714 | −0.058; 0.241 | 0.028 |
| Serum vitamin C | −0.149 | −0.031; 0.378 | 0.079 | −0.029 | −0.223; 0.154 | 0.599 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isola, G.; Polizzi, A.; Muraglie, S.; Leonardi, R.; Lo Giudice, A. Assessment of Vitamin C and Antioxidant Profiles in Saliva and Serum in Patients with Periodontitis and Ischemic Heart Disease. Nutrients 2019, 11, 2956. https://doi.org/10.3390/nu11122956
Isola G, Polizzi A, Muraglie S, Leonardi R, Lo Giudice A. Assessment of Vitamin C and Antioxidant Profiles in Saliva and Serum in Patients with Periodontitis and Ischemic Heart Disease. Nutrients. 2019; 11(12):2956. https://doi.org/10.3390/nu11122956
Chicago/Turabian StyleIsola, Gaetano, Alessandro Polizzi, Simone Muraglie, Rosalia Leonardi, and Antonino Lo Giudice. 2019. "Assessment of Vitamin C and Antioxidant Profiles in Saliva and Serum in Patients with Periodontitis and Ischemic Heart Disease" Nutrients 11, no. 12: 2956. https://doi.org/10.3390/nu11122956
APA StyleIsola, G., Polizzi, A., Muraglie, S., Leonardi, R., & Lo Giudice, A. (2019). Assessment of Vitamin C and Antioxidant Profiles in Saliva and Serum in Patients with Periodontitis and Ischemic Heart Disease. Nutrients, 11(12), 2956. https://doi.org/10.3390/nu11122956








