Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration
Abstract
1. Introduction
2. Acute Dietary Exposure and the Gut Microbiome
2.1. The Gut Microbiota Responds Rapidly to Dietary Changes
2.2. Effect of Short-Term Dietary Interventions on the Gut Microbiota
2.3. The Impact of Probiotics on Microbial Communities Is Individualised and Transient
2.4. Prebiotics Induce Changes in Microbial Composition and Metabolite Production
3. Long-Term diet and the Gut Microbiome
3.1. Evidence for Long Lasting Effect of Diet on the Gut Microbiome
3.2. Capturing Long-Term Effect of Diet on the Gut Microbiota
3.2.1. Dietary Diversity
3.2.2. Singular and Combined Nutrients
3.2.3. Dietary Indices
4. Limitations of Knowledge and Recommendations for the Future
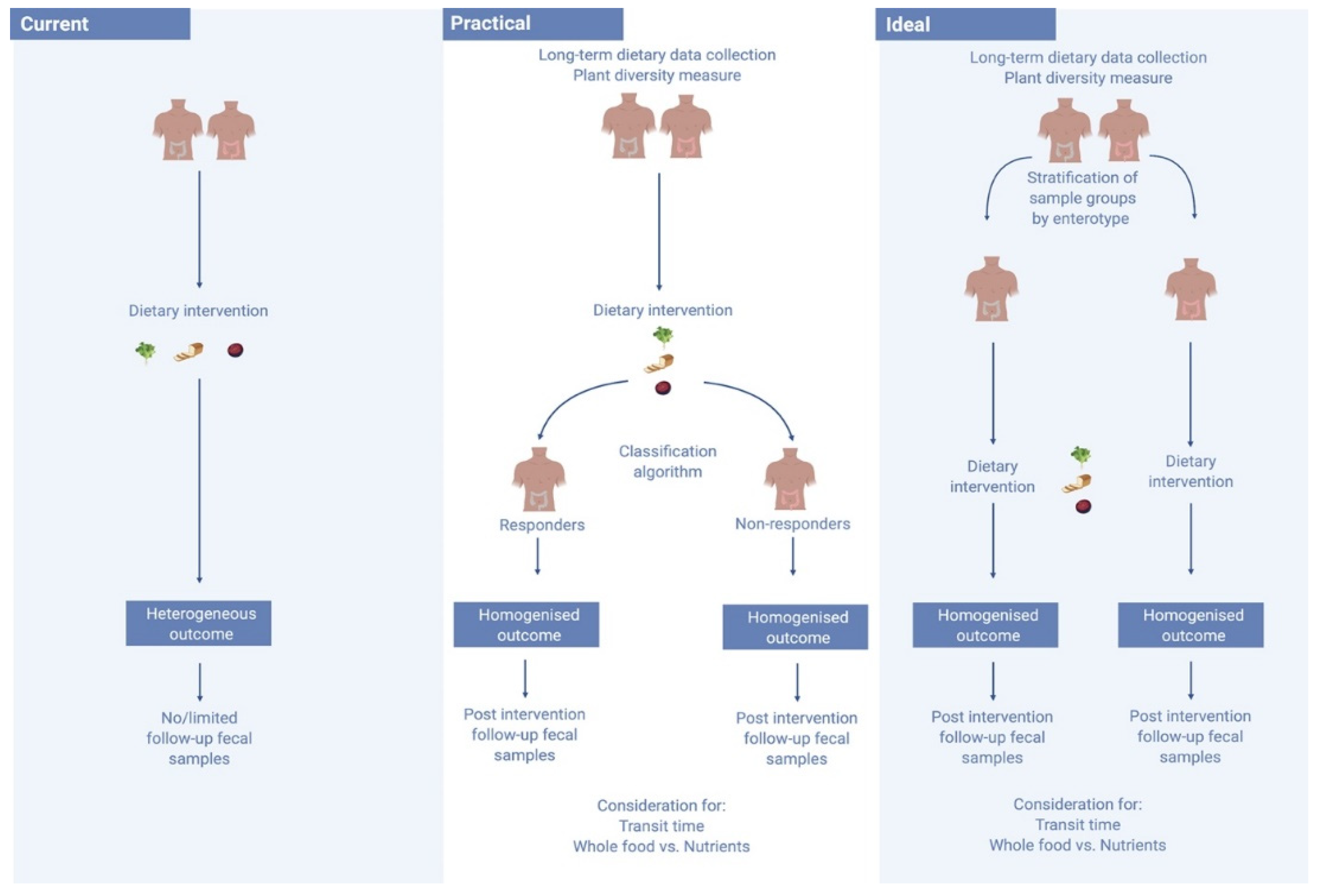
4.1. Increasing Emphasis on Habitual Diet Prior to Dietary Interventions and Analyses
4.2. Nutrient Centred Designs vs. Whole Food Approaches to Dietary Interventions
4.3. Symbiotics Provide an Opportunity to Selectively Alter Microbiome Composition
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jackson, M.A.; Verdi, S.; Maxan, M.E.; Shin, C.M.; Zierer, J.; Bowyer, R.C.E.; Martin, T.; Williams, F.M.K.; Menni, C.; Bell, J.T.; et al. Gut microbiota associations with common diseases and prescription medications in a population-based cohort. Nat. Commun. 2018, 9, 2655. [Google Scholar] [CrossRef]
- Valdes, A.M.; Walter, J.; Segal, E.; Spector, T.D. Role of the gut microbiota in nutrition and health. BMJ 2018, 361, k2179. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, M.; Wang, S.; Han, R.; Cao, Y.; Hua, W.; Mao, Y.; Zhang, X.; Pang, X.; Wei, C.; et al. Interactions between gut microbiota, host genetics and diet relevant to development of metabolic syndromes in mice. ISME J. 2010, 4, 232–241. [Google Scholar] [CrossRef]
- Rothschild, D.; Weissbrod, O.; Barkan, E.; Kurilshikov, A.; Korem, T.; Zeevi, D.; Costea, P.I.; Godneva, A.; Kalka, I.N.; Bar, N.; et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 2018, 555, 210–215. [Google Scholar] [CrossRef]
- David, L.A.; Materna, A.C.; Friedman, J.; Campos-Baptista, M.I.; Blackburn, M.C.; Perrotta, A.; Erdman, S.E.; Alm, E.J. Host lifestyle affects human microbiota on daily timescales. Genome Biol. 2014, 15, R89. [Google Scholar] [CrossRef]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef]
- Goodrich, J.K.; Davenport, E.R.; Beaumont, M.; Jackson, M.A.; Knight, R.; Ober, C.; Spector, T.D.; Bell, J.T.; Clark, A.G.; Ley, R.E. Genetic Determinants of the Gut Microbiome in UK Twins. Cell Host Microbe 2016, 19, 731–743. [Google Scholar] [CrossRef]
- Maier, L.; Typas, A. Systematically investigating the impact of medication on the gut microbiome. Curr. Opin. Microbiol. 2017, 39, 128–135. [Google Scholar] [CrossRef]
- Li, H.; Wang, J.; Wu, L.; Luo, J.; Liang, X.; Xiao, B.; Zhu, Y. The impacts of delivery mode on infant’s oral microflora. Sci. Rep. 2018, 8, 11938. [Google Scholar] [CrossRef]
- Ho, N.T.; Li, F.; Lee-Sarwar, K.A.; Tun, H.M.; Brown, B.P.; Pannaraj, P.S.; Bender, J.M.; Azad, M.B.; Thompson, A.L.; Weiss, S.T.; et al. Meta-analysis of effects of exclusive breastfeeding on infant gut microbiota across populations. Nat. Commun. 2018, 9, 4169. [Google Scholar] [CrossRef]
- Tanaka, M.; Nakayama, J. Development of the gut microbiota in infancy and its impact on health in later life. Allergol. Int. 2017, 66, 515–522. [Google Scholar] [CrossRef]
- Voreades, N.; Kozil, A.; Weir, T.L. Diet and the development of the human intestinal microbiome. Front. Microbiol. 2014, 5. [Google Scholar] [CrossRef]
- Francino, P.M. Early Development of the Gut Microbiota and Immune Health. Pathogens 2014, 3, 769. [Google Scholar] [CrossRef]
- Vatanen, T.; Kostic, A.D.; D’Hennezel, E.; Siljander, H.; Franzosa, E.A.; Yassour, M.; Kolde, R.; Vlamakis, H.; Arthur, T.D.; Hamalainen, A.M.; et al. Variation in Microbiome LPS Immunogenicity Contributes to Autoimmunity in Humans. Cell 2016, 165, 842–853. [Google Scholar] [CrossRef]
- Uhr, G.T.; Dohnalová, L.; Thaiss, C.A. The Dimension of Time in Host-Microbiome Interactions. Msystems 2019, 4, e00216-18. [Google Scholar] [CrossRef]
- Tebani, A.; Bekri, S. Paving the Way to Precision Nutrition Through Metabolomics. Front. Nutr. 2019, 6. [Google Scholar] [CrossRef]
- Sonnenburg, J.L.; Bäckhed, F. Diet-microbiota interactions as moderators of human metabolism. Nature 2016, 535, 56–64. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Walker, A.W.; Ince, J.; Duncan, S.H.; Webster, L.M.; Holtrop, G.; Ze, X.; Brown, D.; Stares, M.D.; Scott, P.; Bergerat, A.; et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. Isme J. 2010, 5, 220–230. [Google Scholar] [CrossRef]
- Liang, X.; FitzGerald, G.A. Timing the Microbes: The Circadian Rhythm of the Gut Microbiome. J. Biol. Rhythm. 2017, 32, 505–515. [Google Scholar] [CrossRef]
- Parkar, G.S.; Kalsbeek, A.; Cheeseman, F.J. Potential Role for the Gut Microbiota in Modulating Host Circadian Rhythms and Metabolic Health. Microorganisms 2019, 7, 41. [Google Scholar] [CrossRef]
- Thaiss, C.A.; Zeevi, D.; Levy, M.; Zilberman-Schapira, G.; Suez, J.; Tengeler, A.C.; Abramson, L.; Katz, M.N.; Korem, T.; Zmora, N.; et al. Transkingdom Control of Microbiota Diurnal Oscillations Promotes Metabolic Homeostasis. Cell 2014, 159, 514–529. [Google Scholar] [CrossRef]
- Thaiss, C.A.; Itav, S.; Rothschild, D.; Meijer, M.; Levy, M.; Moresi, C.; Dohnalova, L.; Braverman, S.; Rozin, S.; Malitsky, S.; et al. Persistent microbiome alterations modulate the rate of post-dieting weight regain. Nature 2016, 540, 544. [Google Scholar] [CrossRef]
- Kuang, Z.; Wang, Y.; Li, Y.; Ye, C.; Ruhn, K.A.; Behrendt, C.L.; Olson, E.N.; Hooper, L.V. The intestinal microbiota programs diurnal rhythms in host metabolism through histone deacetylase 3. BioRxiv 2019, 580613. [Google Scholar] [CrossRef]
- Reynolds, A.C.; Broussard, J.; Paterson, J.L.; Wright, K.P., Jr.; Ferguson, S.A. Sleepy, circadian disrupted and sick: Could intestinal microbiota play an important role in shift worker health? Mol. Metab. 2016, 6, 12–13. [Google Scholar] [CrossRef]
- Oosterman, J.E.; Kalsbeek, A.; Fleur, S.E.L.; Belsham, D.D. Impact of nutrients on circadian rhythmicity. Am. J. Physiol. -Regul. Integr. Comp. Physiol. 2015, 308, R337–R350. [Google Scholar] [CrossRef]
- Kaczmarek, J.L.; Musaad, S.M.; Holscher, H.D. Time of day and eating behaviors are associated with the composition and function of the human gastrointestinal microbiota. Am. J. Clin. Nutr. 2017, 106, 1220–1231. [Google Scholar] [CrossRef]
- Collado, M.C.; Engen, P.A.; Bandín, C.; Cabrera-Rubio, R.; Voigt, R.M.; Green, S.J.; Naqib, A.; Keshavarzian, A.; Scheer, F.A.J.L.; Garaulet, M. Timing of food intake impacts daily rhythms of human salivary microbiota: A randomized, crossover study. FASEB J. 2018, 32, 2060–2072. [Google Scholar] [CrossRef]
- Fetissov, S.O. Role of the gut microbiota in host appetite control: Bacterial growth to animal feeding behaviour. Nat. Rev. Endocrinol. 2016, 13, 11–25. [Google Scholar] [CrossRef]
- Zarrinpar, A.; Chaix, A.; Yooseph, S.; Panda, S. Diet and Feeding Pattern Affect the Diurnal Dynamics of the Gut Microbiome. Cell Metab. 2014, 20, 1006–1017. [Google Scholar] [CrossRef]
- Garud, N.R.; Good, B.H.; Hallatschek, O.; Pollard, K.S. Evolutionary dynamics of bacteria in the gut microbiome within and across hosts. PLoS Biol. 2019, 17, e3000102. [Google Scholar] [CrossRef] [PubMed]
- Klimenko, N.S.; Tyakht, A.V.; Popenko, A.S.; Vasiliev, A.S.; Altukhov, I.A.; Ischenko, D.S.; Shashkova, T.I.; Efimova, D.A.; Nikogosov, D.A.; Osipenko, D.A.; et al. Microbiome Responses to an Uncontrolled Short-Term Diet Intervention in the Frame of the Citizen Science Project. Nutrients 2018, 10, 576. [Google Scholar] [CrossRef] [PubMed]
- Sommer, F.; Anderson, J.M.; Bharti, R.; Raes, J.; Rosenstiel, P. The resilience of the intestinal microbiota influences health and disease. Nat. Rev. Microbiol. 2017, 15, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Knight, R. Dietary effects on human gut microbiome diversity. Br. J. Nutr. 2015, 113 (Suppl. S1), S1–S5. [Google Scholar] [CrossRef]
- Johnson, A.J.; Vangay, P.; Al-Ghalith, G.A.; Hillmann, B.M.; Ward, T.L.; Shields-Cutler, R.R.; Kim, A.D.; Shmagel, A.K.; Syed, A.N.; Walter, J.; et al. Daily Sampling Reveals Personalized Diet-Microbiome Associations in Humans. Cell Host Microbe 2019, 25, 789–802. [Google Scholar] [CrossRef]
- Chassaing, B.; Vijay-Kumar, M.; Gewirtz, A.T. How diet can impact gut microbiota to promote or endanger health. Curr. Opin. Gastroenterol. 2017, 33, 417–421. [Google Scholar] [CrossRef]
- So, D.; Whelan, K.; Rossi, M.; Morrison, M.; Holtmann, G.; Kelly, J.T.; Shanahan, E.R.; Staudacher, H.M.; Campbell, K.L. Dietary fiber intervention on gut microbiota composition in healthy adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018, 107, 965–983. [Google Scholar] [CrossRef]
- Liu, F.; Li, P.; Chen, M.; Luo, Y.; Prabhakar, M.; Zheng, H.; He, Y.; Qi, Q.; Long, H.; Zhang, Y.; et al. Fructooligosaccharide (FOS) and Galactooligosaccharide (GOS) Increase Bifidobacterium but Reduce Butyrate Producing Bacteria with Adverse Glycemic Metabolism in healthy young population. Sci. Rep. 2017, 7, 11789. [Google Scholar] [CrossRef]
- Burton, K.J.; Rosikiewicz, M.; Pimentel, G.; Butikofer, U.; von Ah, U.; Voirol, M.J.; Croxatto, A.; Aeby, S.; Drai, J.; McTernan, P.G.; et al. Probiotic yogurt and acidified milk similarly reduce postprandial inflammation and both alter the gut microbiota of healthy, young men. Br. J. Nutr. 2017, 117, 1312–1322. [Google Scholar] [CrossRef]
- Kellingray, L.; Tapp, H.S.; Saha, S.; Doleman, J.F.; Narbad, A.; Mithen, R.F. Consumption of a diet rich in Brassica vegetables is associated with a reduced abundance of sulphate-reducing bacteria: A randomised crossover study. Mol. Nutr. Food Res. 2017. [Google Scholar] [CrossRef]
- Albenberg, L.G.; Wu, G.D. Diet and the Intestinal Microbiome: Associations, Functions, and Implications for Health and Disease. Gastroenterology 2014, 146, 1564–1572. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Amp Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Kunk, D. Probiotics: Elixir or empty promise? Lancet Gastroenterol. Hepatol. 2019, 4, 81. [Google Scholar] [CrossRef]
- Li, Y.T.; Xu, H.; Ye, J.Z.; Wu, W.R.; Shi, D.; Fang, D.Q.; Liu, Y.; Li, L.J. Efficacy of Lactobacillus rhamnosus GG in treatment of acute pediatric diarrhea: A systematic review with meta-analysis. World J. Gastroenterol. 2019, 25, 4999–5016. [Google Scholar] [CrossRef] [PubMed]
- Hungin, A.P.S.; Mitchell, C.R.; Whorwell, P.; Mulligan, C.; Cole, O.; Agreus, L.; Fracasso, P.; Lionis, C.; Mendive, J.; Philippart de Foy, J.M.; et al. Systematic review: Probiotics in the management of lower gastrointestinal symptoms—An updated evidence-based international consensus. Aliment Pharm. 2018, 47, 1054–1070. [Google Scholar] [CrossRef] [PubMed]
- Derwa, Y.; Gracie, D.J.; Hamlin, P.J.; Ford, A.C. Systematic review with meta-analysis: The efficacy of probiotics in inflammatory bowel disease. Aliment Pharm. 2017, 46, 389–400. [Google Scholar] [CrossRef]
- Krumbeck, J.A.; Walter, J.; Hutkins, R.W. Synbiotics for Improved Human Health: Recent Developments, Challenges, and Opportunities. Annu. Rev. Food Sci. Technol. 2018, 9, 451–479. [Google Scholar] [CrossRef]
- Shepherd, E.S.; DeLoache, W.C.; Pruss, K.M.; Whitaker, W.R.; Sonnenburg, J.L. An exclusive metabolic niche enables strain engraftment in the gut microbiota. Nature 2018, 557, 434–438. [Google Scholar] [CrossRef]
- Zmora, N.; Zilberman-Schapira, G.; Suez, J.; Mor, U.; Dori-Bachash, M.; Bashiardes, S.; Kotler, E.; Zur, M.; Regev-Lehavi, D.; Brik, R.B.; et al. Personalized Gut Mucosal Colonization Resistance to Empiric Probiotics Is Associated with Unique Host and Microbiome Features. Cell 2018, 174, 1388–1405.e21. [Google Scholar] [CrossRef]
- Maldonado-Gómez, M.X.; Martínez, I.; Bottacini, F.; O’Callaghan, A.; Ventura, M.; van Sinderen, D.; Hillmann, B.; Vangay, P.; Knights, D.; Hutkins, R.W.; et al. Stable Engraftment of Bifidobacterium longum AH1206 in the Human Gut Depends on Individualized Features of the Resident Microbiome. Cell Host Microbe 2016, 20, 515–526. [Google Scholar] [CrossRef]
- Sanders, M.E.; Merenstein, D.; Merrifield, C.A.; Hutkins, R. Probiotics for human use. Nutr. Bull. 2018, 43, 212–225. [Google Scholar] [CrossRef]
- Kristensen, N.B.; Bryrup, T.; Allin, K.H.; Nielsen, T.; Hansen, T.H.; Pedersen, O. Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: A systematic review of randomized controlled trials. Genome Med. 2016, 8, 52. [Google Scholar] [CrossRef] [PubMed]
- Licciardi, P.V.; Wong, S.-S.; Tang, M.L.K.; Karagiannis, T.C. Epigenome targeting by probiotic metabolites. Gut Pathog. 2010, 2, 24. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meance, S.; Cayuela, C.; Raimondi, A.; Turchet, P.; Lucas, C.; Antoine, J.-M. Recent Advances in the Use of Functional Foods: Effects of the Commercial Fermented Milk with Bifidobacterium Animalis Strain DN-173 010 and Yoghurt Strains on Gut Transit Time in the Elderly. Microb. Ecol. Health Dis. 2003, 15, 15–22. [Google Scholar] [CrossRef]
- Ferrarese, R.; Ceresola, E.; Preti, A.; Canducci, F. Probiotics, prebiotics and synbiotics for weight loss and metabolic syndrome in the microbiome era. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7588–7605. [Google Scholar] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Amp Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Gurry, T.; Dannenberg, P.H.; Finlayson, S.G.; Hughes, T.K.; Macias-Trevino, C.; Owusu-Boaitey, K.; Shomorony, A.; Tuang, S.L.; Valenstein, M.L.; Wang, K.K.; et al. Predictability and persistence of prebiotic dietary supplementation in a healthy human cohort. Sci. Rep. 2018, 8, 12699. [Google Scholar] [CrossRef]
- Choque Delgado, G.T.; Tamashiro, W.M.D.S.C. Role of prebiotics in regulation of microbiota and prevention of obesity. Food Res. Int. 2018, 113, 183–188. [Google Scholar] [CrossRef]
- Costabile, A.; Klinder, A.; Fava, F.; Napolitano, A.; Fogliano, V.; Leonard, C.; Gibson, G.R.; Tuohy, K.M. Whole-grain wheat breakfast cereal has a prebiotic effect on the human gut microbiota: A double-blind, placebo-controlled, crossover study. Br. J. Nutr. 2008, 99, 110–120. [Google Scholar] [CrossRef]
- Roager, H.M.; Vogt, J.K.; Kristensen, M.; Hansen, L.B.S.; Ibrügger, S.; Mærkedahl, R.B.; Bahl, M.I.; Lind, M.V.; Nielsen, R.L.; Frøkiær, H.; et al. Whole grain-rich diet reduces body weight and systemic low-grade inflammation without inducing major changes of the gut microbiome: A randomised cross-over trial. Gut 2019, 68, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Vanegas, S.M.; Meydani, M.; Barnett, J.B.; Goldin, B.; Kane, A.; Rasmussen, H.; Brown, C.; Vangay, P.; Knights, D.; Jonnalagadda, S.; et al. Substituting whole grains for refined grains in a 6-wk randomized trial has a modest effect on gut microbiota and immune and inflammatory markers of healthy adults. Am. J. Clin. Nutr. 2017, 105, 635–650. [Google Scholar] [CrossRef] [PubMed]
- Zeevi, D.; Korem, T.; Zmora, N.; Israeli, D.; Rothschild, D.; Weinberger, A.; Ben-Yacov, O.; Lador, D.; Avnit-Sagi, T.; Lotan-Pompan, M.; et al. Personalized Nutrition by Prediction of Glycemic Responses. Cell 2015, 163, 1079–1094. [Google Scholar] [CrossRef] [PubMed]
- Ukhanova, M.; Wang, X.; Baer, D.J.; Novotny, J.A.; Fredborg, M.; Mai, V. Effects of almond and pistachio consumption on gut microbiota composition in a randomised cross-over human feeding study. Br. J. Nutr. 2014, 111, 2146–2152. [Google Scholar] [CrossRef]
- Karl, J.P.; Meydani, M.; Barnett, J.B.; Vanegas, S.M.; Goldin, B.; Kane, A.; Rasmussen, H.; Saltzman, E.; Vangay, P.; Knights, D.; et al. Substituting whole grains for refined grains in a 6-wk randomized trial favorably affects energy-balance metrics in healthy men and postmenopausal women. Am. J. Clin. Nutr. 2017, 105, 589–599. [Google Scholar] [CrossRef]
- Sanz, Y. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult humans. Gut Microbes 2010, 1, 135–137. [Google Scholar] [CrossRef]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef]
- Kaczmarek, J.L.; Liu, X.; Charron, C.S.; Novotny, J.A.; Jeffery, E.H.; Seifried, H.E.; Ross, S.A.; Miller, M.J.; Swanson, K.S.; Holscher, H.D. Broccoli consumption affects the human gastrointestinal microbiota. J. Nutr. Biochem. 2019, 63, 27–34. [Google Scholar] [CrossRef]
- Duncan, S.H.; Belenguer, A.; Holtrop, G.; Johnstone, A.M.; Flint, H.J.; Lobley, G.E. Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl. Environ. Microbiol. 2007, 73, 1073–1078. [Google Scholar] [CrossRef]
- Russell, W.R.; Gratz, S.W.; Duncan, S.H.; Holtrop, G.; Ince, J.; Scobbie, L.; Duncan, G.; Johnstone, A.M.; Lobley, G.E.; Wallace, R.J.; et al. High-protein, reduced-carbohydrate weight-loss diets promote metabolite profiles likely to be detrimental to colonic health. Am. J. Clin. Nutr. 2011, 93, 1062–1072. [Google Scholar] [CrossRef]
- Bellikci-Koyu, E.; Sarer-Yurekli, P.B.; Akyon, Y.; Aydin-Kose, F.; Karagozlu, C.; Ozgen, G.A.; Brinkmann, A.; Nitsche, A.; Ergunay, K.; Yilmaz, E.; et al. Effects of Regular Kefir Consumption on Gut Microbiota in Patients with Metabolic Syndrome: A Parallel-Group, Randomized, Controlled Study. Nutrients 2019, 11, 2089. [Google Scholar] [CrossRef] [PubMed]
- Cotillard, A.; Kennedy, S.P.; Kong, L.C.; Prifti, E.; Pons, N.; Le Chatelier, E.; Almeida, M.; Quinquis, B.; Levenez, F.; Galleron, N.; et al. Dietary intervention impact on gut microbial gene richness. Nature 2013, 500, 585–588. [Google Scholar] [CrossRef] [PubMed]
- Salonen, A.; Lahti, L.; Salojärvi, J.; Holtrop, G.; Korpela, K.; Duncan, S.H.; Date, P.; Farquharson, F.; Johnstone, A.M.; Lobley, G.E.; et al. Impact of diet and individual variation on intestinal microbiota composition and fermentation products in obese men. Isme J. 2014, 8, 2218–2230. [Google Scholar] [CrossRef] [PubMed]
- Dao, M.C.; Everard, A.; Aron-Wisnewsky, J.; Sokolovska, N.; Prifti, E.; Verger, E.O.; Kayser, B.D.; Levenez, F.; Chilloux, J.; Hoyles, L.; et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: Relationship with gut microbiome richness and ecology. Gut 2016, 65, 426–436. [Google Scholar] [CrossRef]
- Roager, H.M.; Licht, T.R.; Poulsen, S.K.; Larsen, T.M.; Bahl, M.I. Microbial enterotypes, inferred by the prevotella-to-bacteroides ratio, remained stable during a 6-month randomized controlled diet intervention with the new nordic diet. Appl. Environ. Microbiol. 2014, 80, 1142–1149. [Google Scholar] [CrossRef]
- Smits, S.A.; Leach, J.; Sonnenburg, E.D.; Gonzalez, C.G.; Lichtman, J.S.; Reid, G.; Knight, R.; Manjurano, A.; Changalucha, J.; Elias, J.E.; et al. Seasonal cycling in the gut microbiome of the Hadza hunter-gatherers of Tanzania. Science 2017, 357, 802–806. [Google Scholar] [CrossRef]
- Griffin, N.W.; Ahern, P.P.; Cheng, J.; Heath, A.C.; Ilkayeva, O.; Newgard, C.B.; Fontana, L.; Gordon, J.I. Prior Dietary Practices and Connections to a Human Gut Microbial Metacommunity Alter Responses to Diet Interventions. Cell Host Microbe 2017, 21, 84–96. [Google Scholar] [CrossRef]
- McDonald, D.; Hyde, E.; Debelius, J.W.; Morton, J.T.; Gonzalez, A.; Ackermann, G.; Aksenov, A.A.; Behsaz, B.; Brennan, C.; Chen, Y.; et al. American Gut: An Open Platform for Citizen Science Microbiome Research. mSystems 2018, 3. [Google Scholar] [CrossRef]
- Le Roy, C.I.; Wells, P.M.; Si, J.; Raes, J.; Bell, J.T.; Spector, T.D. Red Wine Consumption Associated With Increased Gut Microbiota α-diversity in 3 Independent Cohorts. Gastroenterology 2019. [Google Scholar] [CrossRef]
- Shikany, J.M.; Demmer, R.T.; Johnson, A.J.; Fino, N.F.; Meyer, K.; Ensrud, K.E.; Lane, N.E.; Orwoll, E.S.; Kado, D.M.; Zmuda, J.M.; et al. Association of dietary patterns with the gut microbiota in older, community-dwelling men. Am. J. Clin. Nutr. 2019, 110, 1003–1014. [Google Scholar] [CrossRef]
- Claesson, M.J.; Jeffery, I.B.; Conde, S.; Power, S.E.; O’Connor, E.M.; Cusack, S.; Harris, H.M.; Coakley, M.; Lakshminarayanan, B.; O’Sullivan, O.; et al. Gut microbiota composition correlates with diet and health in the elderly. Nature 2012, 488, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Ringel-Kulka, T.; Cheng, J.; Ringel, Y.; Salojärvi, J.; Carroll, I.; Palva, A.; de Vos, W.M.; Satokari, R. Intestinal Microbiota in Healthy U.S. Young Children and Adults—A High Throughput Microarray Analysis. PLoS ONE 2013, 8, e64315. [Google Scholar] [CrossRef] [PubMed]
- Agans, R.; Rigsbee, L.; Kenche, H.; Michail, S.; Khamis, H.J.; Paliy, O. Distal gut microbiota of adolescent children is different from that of adults. FEMS Microbiol. Ecol. 2011, 77, 404–412. [Google Scholar] [CrossRef]
- Heiman, M.L.; Greenway, F.L. A healthy gastrointestinal microbiome is dependent on dietary diversity. Mol. Metab. 2016, 5, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Hollister, E.B.; Riehle, K.; Luna, R.A.; Weidler, E.M.; Rubio-Gonzales, M.; Mistretta, T.-A.; Raza, S.; Doddapaneni, H.V.; Metcalf, G.A.; Muzny, D.M.; et al. Structure and function of the healthy pre-adolescent pediatric gut microbiome. Microbiome 2015, 3, 36. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.S.B.; Raes, J.; Bork, P. The Human Gut Microbiome: From Association to Modulation. Cell 2018, 172, 1198–1215. [Google Scholar] [CrossRef]
- Hu, X.; Liu, G.; Li, Y.; Wei, Y.; Lin, S.; Liu, S.; Zheng, Y.; Hu, D. High-Throughput Analysis Reveals Seasonal Variation of the Gut Microbiota Composition Within Forest Musk Deer (Moschus berezovskii). Front. Microbiol. 2018, 9, 1674. [Google Scholar] [CrossRef]
- Orkin, J.D.; Campos, F.A.; Myers, M.S.; Cheves Hernandez, S.E.; Guadamuz, A.; Melin, A.D. Seasonality of the gut microbiota of free-ranging white-faced capuchins in a tropical dry forest. ISME J. 2019, 13, 183–196. [Google Scholar] [CrossRef]
- Davenport, E.R.; Mizrahi-Man, O.; Michelini, K.; Barreiro, L.B.; Ober, C.; Gilad, Y. Seasonal Variation in Human Gut Microbiome Composition. PLoS ONE 2014, 9, e90731. [Google Scholar] [CrossRef]
- Bailey, M.T.; Walton, J.C.; Dowd, S.E.; Weil, Z.M.; Nelson, R.J. Photoperiod modulates gut bacteria composition in male Siberian hamsters (Phodopus sungorus). Brain Behav. Immun. 2010, 24, 577–584. [Google Scholar] [CrossRef]
- Bowyer, R.C.E.; Jackson, M.A.; Pallister, T.; Skinner, J.; Spector, T.D.; Welch, A.A.; Steves, C.J. Use of dietary indices to control for diet in human gut microbiota studies. Microbiome 2018, 6, 77. [Google Scholar] [CrossRef] [PubMed]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Hill, P.; Muir, J.G.; Gibson, P.R. Controversies and Recent Developments of the Low-FODMAP Diet. Gastroenterol. Hepatol. 2017, 13, 36–45. [Google Scholar]
- Reddel, S.; Putignani, L.; Del Chierico, F. The Impact of Low-FODMAPs, Gluten-Free, and Ketogenic Diets on Gut Microbiota Modulation in Pathological Conditions. Nutrients 2019, 11, 373. [Google Scholar] [CrossRef]
- Burke, D.G.; Fouhy, F.; Harrison, M.J.; Rea, M.C.; Cotter, P.D.; O’Sullivan, O.; Stanton, C.; Hill, C.; Shanahan, F.; Plant, B.J.; et al. The altered gut microbiota in adults with cystic fibrosis. BMC Microbiol. 2017, 17, 58. [Google Scholar] [CrossRef]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill-Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 2009, 137, 1716–1724.e1-2. [Google Scholar] [CrossRef]
- Zinöcker, M.K.; Lindseth, I.A. The Western Diet-Microbiome-Host Interaction and Its Role in Metabolic Disease. Nutrients 2018, 10, 365. [Google Scholar] [CrossRef]
- Gentile, C.L.; Weir, T.L. The gut microbiota at the intersection of diet and human health. Science 2018, 362, 776–780. [Google Scholar] [CrossRef]
- Berding, K.; Holscher, H.D.; Arthur, A.E.; Donovan, S.M. Fecal microbiome composition and stability in 4- to 8-year old children is associated with dietary patterns and nutrient intake. J. Nutr. Biochem. 2018, 56, 165–174. [Google Scholar] [CrossRef]
- Herman, D.R.; Rhoades, N.; Mercado, J.; Argueta, P.; Lopez, U.; Flores, G.E. Dietary Habits of 2- to 9-Year-Old American Children Are Associated with Gut Microbiome Composition. J. Acad. Nutr. Diet. 2019. [Google Scholar] [CrossRef]
- Haro, C.; García-Carpintero, S.; Rangel-Zúñiga, O.A.; Alcalá-Díaz, J.F.; Landa, B.B.; Clemente, J.C.; Pérez-Martínez, P.; López-Miranda, J.; Pérez-Jiménez, F.; Camargo, A. Consumption of Two Healthy Dietary Patterns Restored Microbiota Dysbiosis in Obese Patients with Metabolic Dysfunction. Mol. Nutr. Food Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Ye, C.; Yan, B.; He, X.; Xing, K. Assessing the Influence of Dietary History on Gut Microbiota. Curr. Microbiol. 2019, 76, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, E.D.; Smits, S.A.; Tikhonov, M.; Higginbottom, S.K.; Wingreen, N.S.; Sonnenburg, J.L. Diet-induced extinctions in the gut microbiota compound over generations. Nature 2016, 529, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Vangay, P.; Johnson, A.J.; Ward, T.L.; Al-Ghalith, G.A.; Shields-Cutler, R.R.; Hillmann, B.M.; Lucas, S.K.; Beura, L.K.; Thompson, E.A.; Till, L.M.; et al. US Immigration Westernizes the Human Gut Microbiome. Cell 2018, 175, 962–972. [Google Scholar] [CrossRef]
- Falony, G.; Vieira-Silva, S.; Raes, J. Richness and ecosystem development across faecal snapshots of the gut microbiota. Nat. Microbiol. 2018, 3, 526–528. [Google Scholar] [CrossRef]
- Vandeputte, D.; Falony, G.; Vieira-Silva, S.; Wang, J.; Sailer, M.; Theis, S.; Verbeke, K.; Raes, J. Prebiotic inulin-type fructans induce specific changes in the human gut microbiota. Gut 2017, 66, 1968–1974. [Google Scholar] [CrossRef]
- Lewis, S.J.; Heaton, K.W. Stool form scale as a useful guide to intestinal transit time. Scand. J. Gastroenterol. 1997, 32, 920–924. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef]
- Wilson, K.; Situ, C. Systematic Review on Effects of Diet on Gut Microbiota in Relation to Metabolic Syndromes. J. Clin. Nutr. Metab. 2017, 1, 1–2. [Google Scholar]
- Wu, G.D.; Compher, C.; Chen, E.Z.; Smith, S.A.; Shah, R.D.; Bittinger, K.; Chehoud, C.; Albenberg, L.G.; Nessel, L.; Gilroy, E.; et al. Comparative metabolomics in vegans and omnivores reveal constraints on diet-dependent gut microbiota metabolite production. Gut 2016, 65, 63–72. [Google Scholar] [CrossRef]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R., Jr.; Gross, M.D.; Tapsell, L.C. Food synergy: An operational concept for understanding nutrition. Am. J. Clin. Nutr. 2009, 89, 1543S–1548S. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Hullar, M.A.J.; Schwarz, Y.; Lampe, J.W. Human gut bacterial communities are altered by addition of cruciferous vegetables to a controlled fruit- and vegetable-free diet. J. Nutr. 2009, 139, 1685–1691. [Google Scholar] [CrossRef] [PubMed]
- Holscher, H.D.; Guetterman, H.M.; Swanson, K.S.; An, R.; Matthan, N.R.; Lichtenstein, A.H.; Novotny, J.A.; Baer, D.J. Walnut Consumption Alters the Gastrointestinal Microbiota, Microbially Derived Secondary Bile Acids, and Health Markers in Healthy Adults: A Randomized Controlled Trial. J. Nutr. 2018, 148, 861–867. [Google Scholar] [CrossRef]
- Holscher, H.D.; Taylor, A.M.; Swanson, K.S.; Novotny, J.A.; Baer, D.J. Almond Consumption and Processing Affects the Composition of the Gastrointestinal Microbiota of Healthy Adult Men and Women: A Randomized Controlled Trial. Nutrients 2018, 10, 126. [Google Scholar] [CrossRef]
- Reese, A.T.; Kartzinel, T.R.; Petrone, B.L.; Turnbaugh, P.J.; Pringle, R.M.; David, L.A. Using DNA Metabarcoding To Evaluate the Plant Component of Human Diets: A Proof of Concept. MSystems 2019, 4, e00458-19. [Google Scholar] [CrossRef]
- Pandey, K.R.; Naik, S.R.; Vakil, B.V. Probiotics, prebiotics and synbiotics—A review. J. Food Sci. Technol. 2015, 52, 7577–7587. [Google Scholar] [CrossRef]
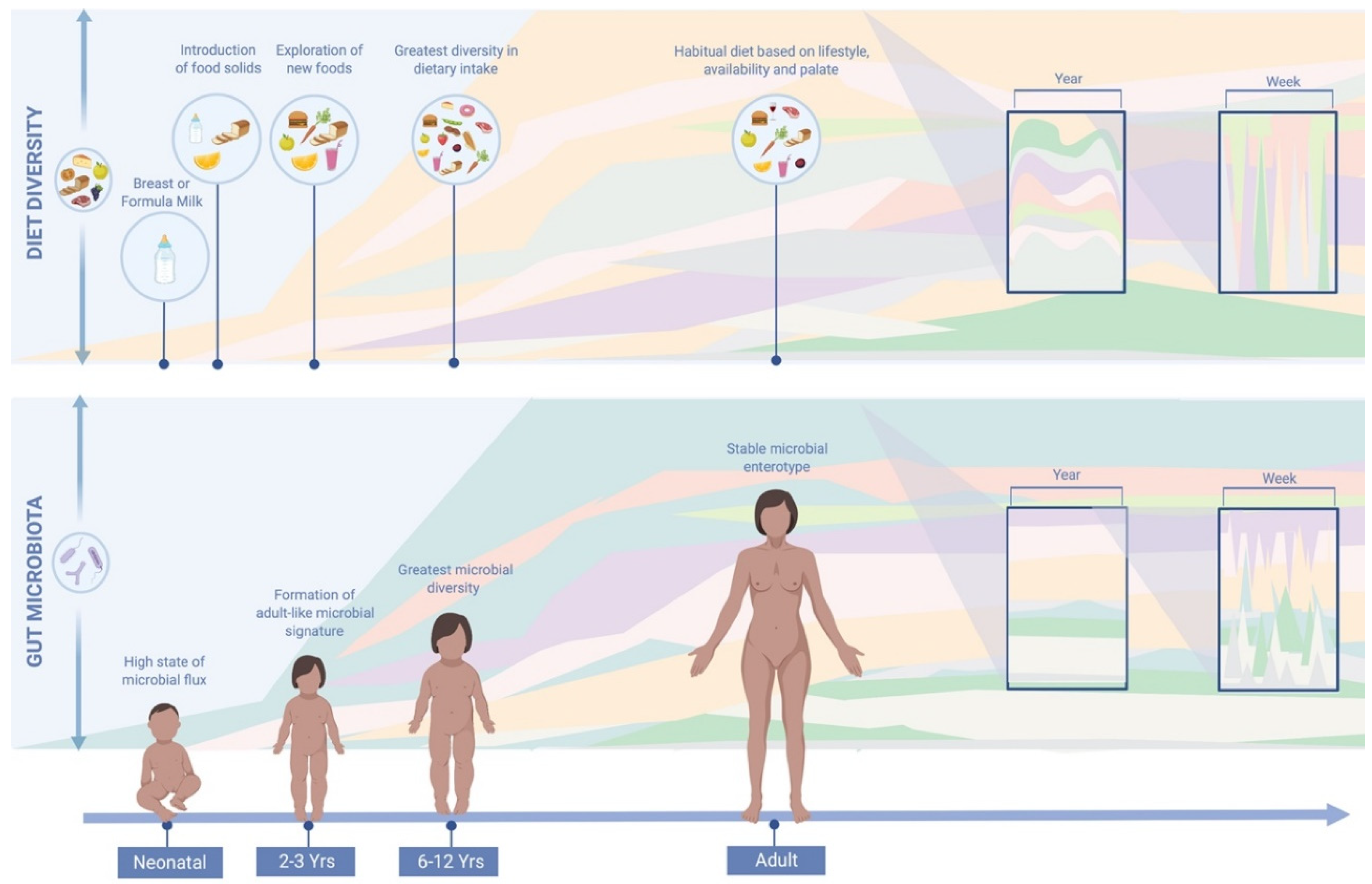
| Primary Author | Year | Organism | Participants | Design | Dietary Data | Dietary Investigation | Length | Faecal Sample | Post-Intervention Faecal Sample | Change to Microbiome |
|---|---|---|---|---|---|---|---|---|---|---|
| Short-term dietary interventions | ||||||||||
| C. Thiass [22] | 2014 | Mouse | 10 | Longitudinal | NA | Circadian rhythm: Ad libitum intake, for two light-dark cycles (12 h light, 12 h dark) | 2 days | Every 6 h for two days | No | Significant (p < 0.05) diurnal fluctuations in the abundance of more than 15% of all bacterial operational taxonomic units (OTUs). |
| D. Zeevi [63] | 2015 | Humans | 800 | Longitudinal | FFQ, daily 24-h records | 6898 habitual meals (total), and standardised meal per day | 1 week | once | NA | People eating identical meals presented variability in post-meal blood glucose response. |
| G. Wu [6] | 2011 | Humans | 10 | Randomised, controlled feeding study | NA | Two treatment groups: (1) high-fat/low-fibre diet; (2) low-fat/high-fibre diet | 10 days | 10 days | No | Microbiome composition changed within 24 h of initiating a high-fat/low-fibre or low-fat/high-fibre diet, but enterotype remained stable throughout. |
| A. Johnson [35] | 2019 | Humans | 34 | Longitudinal | daily 24-h food records | Habitual diet | 17 days | 17 days | NA | Dietary diversity associates with microbiome stability. Daily dietary intake and microbiome composition are highly variable and personalised. |
| M. Ukhanova [64] | 2014 | Humans | 18,16 | Two controlled feeding, randomized, crossover studies (almond or pistachios) | NA | Three treatment groups: (1) no nuts; (2) 1.5 servings/d either almonds or pistachios; (3) 3 servings/d of either almonds or pistachios. 18-day intervention period. Inbetween: 2-week washout period. | 18 days | 6× samples: first and last day of each treatment period | No | Pistachio consumption had a greater impact on gut microbiota composition than almond consumption, including an increase in the number of potentially beneficial butyrate-producing bacteria. Pistachio consumption was associated with a decrease in the number of lactic acid bacteria. |
| L. David [18] | 2013 | Humans | 11 (9 both diet arms) | Cross-over | Daily diet log with visual serving size portion guide, National Cancer Institute’s Diet History Questionnaire II (DHQ) | Two treatment groups: (1) Plant-based diet (5-days); (2) Animal-based diet (5-days). Prior to intervention: 4-days baseline habitual diet. Between: 6-days washout period. Post: 6-days washout period. | 20 days | One sample a day: three baseline days and 2 days on each experimental diet selected. | Collected for 6 day washout period post each diet study arm | Short-term consumption of diets composed entirely of animal or plant products alters microbial community structure and overwhelms interindividual differences in microbial gene expression. However, microbiota composition returned to baseline within 3 days post intervention. |
| J. Karl [65] | 2017 | Humans | 81 | Randomized, controlled, parallel, controlled feeding | The Three-Factor Eating Questionnaire (weeks 2, 8), visual analog scales for hunger, satiety, prospective consumption, and diet satisfaction (weekly) | Two treatment groups: (1) whole grain diet; (2) refined grain diet. Prior to intervention: 2 week run in | 6 weeks | All stools produced over 72 h during diet arm | No | Alpha-diversity and beta-diversity differed between groups at baseline (P < 0.05) but not after the intervention. Relative abundance of En- terobacteriaceae decreased and butyrate-producing Lachnospira and Roseburia increased in the WG compared with in the RG. |
| Y. Sanz [66] | 2010 | Humans | 10 | Preliminary | NA | Gluten-free diet (GFD) | 30 days | Not reported | No | Numbers of beneficial bacteria decreased, while numbers of unhealthy bacteria increased parallel to reductions in the intake of polysaccharides after following the GFD. |
| F. De Filippis [67] | 2015 | Humans | 153 | Longitudinal | 7-day food record | Three habitual diet groups: (1) Omnivore (n = 51); (2) vegetarian (n = 51); (3) vegan (n = 51) | 3 weeks | 3 samples: 1 per week | NA | High-level consumption of plant foods are associated with beneficial microbiome-related metabolomic profiles. Significant associations were detected between consumption of vegetable-based diets and increased levels of faecal short-chain fatty acids, Prevotella and Firmicutes. |
| J. Kaczmarek [68] | 2019 | Humans | 18 | controlled feeding, randomized, crossover study | NA | Treatment group: 200 g broccoli and 20 g daikon radish per day. Control: traditional American diet excluding all brassicas. Treatment/control period: 18 days; between: 24-day washout | 60 days | 3× samples: baseline, end of each treatment period | No | Broccoli consumption decreased the relative abundance of Firmicutes by 9% compared to control (p < 0.05), increased the relative abundance of Bacteroidetes by 10% compared to control (p < 0.03) and increased Bacteroides by 8% relative to control (p < 0.02). |
| S. Duncan [69] | 2016 | Humans | 19 | Experimental | NA | Two treatment groups: (1) high protein/medium carbohydrate (4weeks); (2) high protein/low carbohydrate (4 weeks). Prior to intervention: maintenance diet (3 days) | 9 weeks | 3× samples: 3 post first maintenance period, during last 2 days on each of the main diets | No | No significant change was seen in the relative counts of the Bacteroides, clostridial cluster XIVa, cluster IX, or cluster IV groups. In contrast, the Roseburia spp. and Eubacterium rectale subgroup of cluster XIVa and bifidobacteria decreased as carbohydrate intake decreased. |
| W. Russell [70] | 2011 | Humans | 17 | Randomised cross-over | NA | Two treatment groups: (1) high-protein and moderate-carbohydrate diet (HPMC) (4 weeks); (2) high-protein and low carbohydrate diet (HPLC) (4 weeks). Prior to intervention: weight maintenance diet (7 days) | 9 weeks | 3× samples: end of maintenance, HPMC and HPLC diet periods | No | The HPLC diet decreased the proportion of butyrate in faecal short-chain fatty acid concentrations, which was concomitant with a reduction in the Roseburia/Eubacterium rectale group of bacteria, and greatly reduced concentrations of fibre-derived, antioxidant phenolic acids such as ferulate and its derivatives. |
| A. Walker [19] | 2010 | Humans | 14 | Randomised cross-over design | NA | Two cross-over treatment groups: (1) Diet high in resistant starch (RS) (3 weeks); (2) Diet high in non-starch polysaccharides (NSP) (3 weeks); Prior to intervention: Initial maintenance diet protein/carbohydrate/fat% 13:52:35 and 27.7 g NSP (1 week). Post intervention: High protein, reduced carbohydrate weight loss (WL) diet (3 weeks). | 10 weeks | Twice each week | No | Relatives of Ruminococcus bromii increased in most participants on the RS diet, accounting for a mean of 17% of total bacteria compared with 3.8% on the NSP diet, whereas the uncultured Oscillibacter group increased on the RS and WL diets. Relatives of Eubacterium rectale increased on RS (to mean 10.1%) but decreased, along with Collinsella aerofaciens, on WL. |
| E. Bellikci-koyu [71] | 2019 | Humans | 22 | Randomised parallel, controlled trial | 24-h food recall × 2 (week 0, week 12) | Two treatment groups: (1) 180 mL/day kefir (12); (2) unfermented milk (10) control | 12 weeks | 2× samples: baseline, end of intervention | No | Kefir was associated with a significant increase in the relative abundance of Actinobacteria (p = 0.023) only. No significant change in the relative abundance of Bacteroidetes, Proteobacteria or Verrucomicrobia was obtained. |
| A. Cotillard [72] | 2013 | Humans | 49 | Control | 7-day food record with interview by a dietitian | Intervention: energy-restricted high-protein diet (6 weeks). Control: weight-maintenance diet (6 weeks) | 12 weeks | 3× samples: baseline, 6 and 12 week | No | A significant increase of abundance of most gene clusters on energy restricted diet, however on weight-maintenance diet the abundance of 14 species decreased. |
| A. Salonen [73] | 2014 | Humans | 14 | Randomised cross-over design for RS and NSP interventions | NA | Two cross-over treatment groups: (1) Diet high in resistant starch (RS) (3 weeks); (2) Diet high in non-starch polysaccharides (NSP) (3 weeks); Prior to intervention: Initial maintenance diet protein/carbohydrate/fat% 13:52:35 and 27.7 g NSP (1 week). Post intervention: High protein, reduced carbohydrate weight loss (WL) diet (3 weeks). | 10 weeks | 4× samples: at end of each diet regimen | No | Multiple Ruminococcaceae phylotypes increased on the RS diet, whereas mostly Lachnospiraceae phylotypes increased on the NSP diet. Bifidobacteria decreased significantly on the WL diet. The RS diet decreased the diversity of the microbiota significantly. The dietary responsiveness of the individual’s microbiota varied substantially and associated inversely with its diversity. |
| M. Dao [74] | 2016 | Humans | 49 | Control | 3× 7-day food records (prior to baseline, week 6 and week 12) | Intervention: calorie restricted diet (CR) enriched with fibre and protein (6 weeks). Control: weight stabilisation diet (6 weeks) | 12 weeks | 3× samples: baseline, week 6, week 12 | No | Individuals with higher baseline A. muciniphila displayed greater improvement in insulin sensitivity markers and other clinical parameters after CR. |
| Long-term dietary studies | ||||||||||
| H. Roager [75] | 2014 | Humans | 62 | Parallel randomised control trial | NA | Intervention: ad libitum New Nordic Diet (NND) (n = 36) (24–28 weeks). Control: ad libitum Average Danish Diet (ADD) (n = 26) (24–28 weeks). Prior to intervention: ADD (7–10 days) | 6 months | 2× samples: baseline, end of intervention | No | Negative association between Prevotella spp. and Bacteroides spp. did not reveal significant changes in 35 selected bacterial taxa resulting from the dietary interventions. |
| L. David [5] | 2014 | Humans | 2 | Longitudinal | daily 24-h food records | Habitual diet | 1 year | Subject A: day 0–364. Subject B: day 0–252 | NA | Human gut microbial landscapes are generally stable, but they can be quickly and profoundly altered. |
| S. Smits [76] | 2017 | Humans | 188 | Longitudinal | NA | Habitual hunter-gatherer diet of the Hadza tribe, Tanzania | >1year | 350 samples | NA | Annual cyclic reconfiguration of the microbiome; some taxa became undetectable only to reappear in a subsequent season. Comparison of the Hadza data set with data collected from 18 populations in 16 countries reveals that gut community membership corresponds to modernization. |
| Cross-sectional dietary studies | ||||||||||
| N. Griffin [77] | 2016 | Humans | 170 (34 CRON, 198 AMER) | Cross-sectional | Food journals | Habitual dietary patterns (DP): (1) chronic calorie restriction with optimized intake of nutrients (CRON); (2) without prescribed or self-imposed dietary restrictions (AMER). | NA | 1 sample | NA | AMER displayed less diverse faecal microbiota than those of individuals adhering to CRON. |
| D. McDonald [78] | 2018 | Humans | >10,000 | Cross-sectional | FFQ, primary diet survey | Habitual diet | NA | 1 sample | NA | The diversity of plants consumed are associated with microbial diversity with improved explanatory power vs. categorical variables (such as veganism). |
| C. Le Roy [79] | 2019 | Humans | 916 | Cross-sectional | FFQ | Beer, cider, red wine, white wine, spirits and total alcohol (sum) | NA | 1 sample | NA | Red wine consumption was positively associated in a frequency dependent manner with alpha-diversity with even rare consumption displaying an effect |
| J. Shikany [80] | 2019 | Humans | 517 | Cross-sectional | FFQ | Habitual dietary patterns (DP) based on factor analysis: (Factor 1) ‘Western’ pattern (processed meats, refined grains, potatoes, eggs, sweets and salty snacks); (2) (Factor 2) ’prudent’ pattern (fruits, vegetables, nuts, fish, chicken and turkey without skin) | NA | 1 sample | NA | Greater adherence to the Western pattern was positively associated with families Mogibacteriaceae and Veillonellaceae and genera Alistipes, Anaerotruncus, CC-115, Collinsella, Coprobacillus, Desulfovibrio, Dorea, Eubacterium, and Ruminococcus, while greater adherence to the prudent pattern was positively associated with order Streptophyta, family Victivallaceae, and genera Cetobacterium, Clostridium, Faecalibacterium, Lachnospira, Paraprevotella, and Veillonella |
| M. Claesson [81] | 2012 | Humans | 178 | Cross-sectional | FFQ | Habitual dietary patterns (DP): (1) low fat/high fibre; (2) moderate fat/high fibre; (3) moderate fat/low fibre; (4) high fat/low fibre. Residential location was also considered (community vs. long-term care homes) | Na | 1 sample | NA | The healthy food diversity index (HFD23) positively correlated with three microbiota diversity indices and all four indices showed significant differences between community and long-stay subjects indicating that a healthy, diverse diet promotes a more diverse gut microbiota. |
| G. Wu [6] | 2011 | Humans | 98 | Cross-sectional | FFQ × 1, 24-h food record × 3 | Habitual diet | NA | 1 sample | NA | 72 and 97 microbiome-associated nutrients were identified in 24-h recall and FFQ. Long-term diet was correlated with enterotype clustering. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leeming, E.R.; Johnson, A.J.; Spector, T.D.; Le Roy, C.I. Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration. Nutrients 2019, 11, 2862. https://doi.org/10.3390/nu11122862
Leeming ER, Johnson AJ, Spector TD, Le Roy CI. Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration. Nutrients. 2019; 11(12):2862. https://doi.org/10.3390/nu11122862
Chicago/Turabian StyleLeeming, Emily R, Abigail J Johnson, Tim D Spector, and Caroline I Le Roy. 2019. "Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration" Nutrients 11, no. 12: 2862. https://doi.org/10.3390/nu11122862
APA StyleLeeming, E. R., Johnson, A. J., Spector, T. D., & Le Roy, C. I. (2019). Effect of Diet on the Gut Microbiota: Rethinking Intervention Duration. Nutrients, 11(12), 2862. https://doi.org/10.3390/nu11122862





