Arterial Stiffness, Sugar-Sweetened Beverages and Fruits Intake in a Rural Population Sample: Data from the Brisighella Heart Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Micha, R.; Peñalvo, J.L.; Cudhea, F.; Imamura, F.; Rehm, C.D.; Mozaffarian, D. Association Between Dietary Factors and Mortality from Heart Disease, Stroke, and Type 2 Diabetes in the United States. JAMA 2017, 317, 912–924. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.; Liu, J.; Zhu, M.; Zhao, G.; Bao, W.; Hu, F.B. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2014, 349, g4490. [Google Scholar] [CrossRef] [PubMed]
- Yip, C.S.C.; Chan, W.; Fielding, R. The Associations of Fruit and Vegetable Intakes with Burden of Diseases: A Systematic Review of Meta-Analyses. J. Acad. Nutr. Diet. 2019, 119, 464–481. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Mirmiran, P.; Tohidi, M.; Azizi, F. Longitudinal Associations of High-Fructose Diet with Cardiovascular Events and Potential Risk Factors: Tehran Lipid and Glucose Study. Nutrients 2017, 9, 872. [Google Scholar] [CrossRef]
- Di Nicolantonio, J.J.; O’Keefe, J.H.; Lucan, S.C. Added fructose: a principal driver of type 2 diabetes mellitus and its consequences. Mayo Clin. Proc. 2015, 90, 372–381. [Google Scholar] [CrossRef]
- Nemcsik, J.; Cseprekál, O.; Tislér, A. Measurement of Arterial Stiffness: A Novel Tool of Risk Stratification in Hypertension. Adv. Exp. Med. Biol. 2017, 956, 475–488. [Google Scholar] [CrossRef]
- Vasan, R.S.; Short, M.I.; Niiranen, T.J.; Xanthakis, V.; De Carli, C.; Cheng, S.; Seshadri, S.; Mitchell, G.F. Interrelations Between Arterial Stiffness, Target Organ Damage, and Cardiovascular Disease Outcomes. J. Am. Heart Assoc. 2019, 8, e012141. [Google Scholar] [CrossRef]
- Zhong, Q.; Hu, M.J.; Cui, Y.J.; Liang, L.; Zhou, M.M.; Yang, Y.W.; Huang, F. Carotid-Femoral Pulse Wave Velocity in the Prediction of Cardiovascular Events and Mortality: An Updated Systematic Review and Meta-Analysis. Angiology 2018, 69, 617–629. [Google Scholar] [CrossRef]
- Cicero, A.F.; D’Addato, S.; Santi, F.; Ferroni, A.; Borghi, C.; Brisighella Heart Study Group. Leisure-time physical activity and cardiovascular disease mortality: the Brisighella Heart Study. J. Cardiovasc. Med. 2012, 13, 559–564. [Google Scholar] [CrossRef]
- Cicero, A.F.; Kuwabara, M.; Johnson, R.; Bove, M.; Fogacci, F.; Rosticci, M.; Giovannini, M.; D’Addato, S.; Borghi, C.; Brisighella Heart Study Group. LDL-oxidation, serum uric acid, kidney function and pulse-wave velocity: Data from the Brisighella Heart Study cohort. Int. J. Cardiol. 2018, 261, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Schröder, H.; Benitez Arciniega, A.; Soler, C.; Covas, M.I.; Baena-Díez, J.M.; Marrugat, J. Validity of two short screeners for diet quality in time-limited settings. Public Health Nutr. 2012, 15, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Ríos-Rodríguez, M.L.; García-Cerdán, M.R.; Calonge-Vallejo, A.R.; Tobella-Andreu, L.; Baena-Díez, J.M.; Schröder, H. Feasibility and results of the short Diet Quality Screener in Primary Care: EMAP study. Enferm. Clin. 2016, 26, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Fogacci, F.; Giovannini, M.; Grandi, E.; D’Addato, S.; Borghi, C.; Brisighella Heart Study Group. Interaction between low-density lipoprotein-cholesterolaemia, serum uric level and incident hypertension: Data from the Brisighella Heart Study. J. Hypertens. 2019, 37, 728–731. [Google Scholar] [CrossRef]
- Cicero, A.F.; Rosticci, M.; Tocci, G.; Bacchelli, S.; Urso, R.; D’Addato, S.; Borghi, C. Serum uric acid and other short-term predictors of electrocardiographic alterations in the Brisighella Heart Study cohort. Eur. J. Intern. Med. 2015, 26, 255–258. [Google Scholar] [CrossRef]
- Earley, A.; Miskulin, D.; Lamb, E.J.; Levey, A.S.; Uhlig, K. Estimating equations for glomerular filtration rate in the era of creatinine standardization: A systematic review. Ann. Intern. Med. 2012, 156, 785–795. [Google Scholar] [CrossRef]
- Hickson, S.S.; Butlin, M.; Broad, J.; Avolio, A.P.; Wilkinson, I.B.; McEniery, C.M. Validity and repeatability of the Vicorder apparatus: A comparison with the SphygmoCor device. Hypertens. Res. 2009, 32, 1079–1085. [Google Scholar] [CrossRef]
- Pucci, G.; Cheriyan, J.; Hubsch, A.; Hickson, S.S.; Gajendragadkar, P.R.; Watson, T.; O’Sullivan, M.; Woodcock-Smith, J.; Schillaci, G.; Wilkinson, I.B.; et al. Evaluation of the Vicorder, a novel cuff-based device for the noninvasive estimation of central blood pressure. J. Hypertens. 2013, 31, 77–85. [Google Scholar] [CrossRef]
- Parsons, T.J.; Sartini, C.; Ellins, E.A.; Halcox, J.P.; Smith, K.E.; Ash, S.; Lennon, L.T.; Wannamethee, S.G.; Lee, I.M.; Whincup, P.H.; et al. Objectively measured physical activity, sedentary time and subclinical vascular disease: Cross-sectional study in older British men. Prev. Med. 2016, 89, 194–199. [Google Scholar] [CrossRef]
- Müller, J.; Ewert, P.; Hager, A. Increased aortic blood pressure augmentation in patients with congenital heart defects—A cross-sectional study in 1125 patients and 322 controls. Int. J. Cardiol. 2015, 184, 225–229. [Google Scholar] [CrossRef]
- Ruscica, M.; Ferri, N.; Fogacci, F.; Rosticci, M.; Botta, M.; Marchiano, S.; Magni, P.; D’Addato, S.; Giovannini, M.; Borghi, C.; et al. Circulating Levels of Proprotein Convertase Subtilisin/Kexin Type 9 and Arterial Stiffness in a Large Population Sample: Data from the Brisighella Heart Study. J. Am. Heart Assoc. 2017, 6, e005764. [Google Scholar] [CrossRef]
- Khan, T.A.; Sievenpiper, J.L. Controversies about sugars: Results from systematic reviews and meta-analyses on obesity, cardiometabolic disease and diabetes. Eur. J. Nutr. 2016, 255, 25–43. [Google Scholar] [CrossRef] [PubMed]
- Ares, G.R.; Ortiz, P.A. Direct renal effects of a fructose-enriched diet: Interaction with high salt intake. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R1078–R1081. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Rosticci, M.; Bove, M.; Fogacci, F.; Giovannini, M.; Urso, R.; D’Addato, S.; Borghi, C.; Brisighella Heart Study Group. Serum uric acid change and modification of blood pressure and fasting plasma glucose in an overall healthy population sample: Data from the Brisighella heart study. Ann. Med. 2017, 49, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Rosticci, M.; Fogacci, F.; Grandi, E.; D’Addato, S.; Borghi, C.; Brisighella Heart Study Group. High serum uric acid is associated to poorly controlled blood pressure and higher arterial stiffness in hypertensive subjects. Eur. J. Intern. Med. 2017, 37, 38–42. [Google Scholar] [CrossRef]
- Wang, D.D.; Sievenpiper, J.L.; de Souza, R.J.; Chiavaroli, L.; Ha, V.; Cozma, A.I.; Mirrahimi, A.; Yu, M.E.; Carleton, A.J.; Di Buono, M.; et al. The effects of fructose intake on serum uric acid vary among controlled dietary trials. J. Nutr. 2012, 142, 916–923. [Google Scholar] [CrossRef]
- Ebrahimpour-Koujan, S.; Saneei, P.; Larijani, B.; Esmaillzadeh, A. Consumption of sugar sweetened beverages and dietary fructose in relation to risk of gout and hyperuricemia: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 1–10. [Google Scholar] [CrossRef]
- Kaneko, C.; Ogura, J.; Sasaki, S.; Okamoto, K.; Kobayashi, M.; Kuwayama, K.; Narumi, K.; Iseki, K. Fructose suppresses uric acid excretion to the intestinal lumen as a result of the induction of oxidative stress by NADPH oxidase activation. Biochim. Biophys. Acta. Gen. Subj. 2017, 1861, 559–566. [Google Scholar] [CrossRef]
- Caliceti, C.; Calabria, D.; Roda, A.; Cicero, A.F.G. Fructose Intake, Serum Uric Acid, and cardiometabolic Disorders: A Critical Review. Nutrients 2017, 9, 395. [Google Scholar] [CrossRef]
- Softic, S.; Cohen, D.E.; Kahn, C.R. Role of Dietary Fructose and Hepatic De Novo Lipogenesis in Fatty Liver Disease. Dig. Dis. Sci. 2016, 61, 1282–1293. [Google Scholar] [CrossRef]
- Nakagawa, T.; Lanaspa, M.A.; Johnson, R.J. The effects of fruit consumption in patients with hyperuricaemia or gout. Rheumatology 2019, 58, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Ayoub-Charette, S.; Liu, Q.; Khan, T.A.; Au-Yeung, F.; Blanco Mejia, S.; de Souza, R.J.; Wolever, T.M.; Leiter, L.A.; Kendall, C.; Sievenpiper, J.L. Important food sources of fructose-containing sugars and incident gout: A systematic review and meta-analysis of prospective cohort studies. BMJ Open 2019, 9, e024171. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; He, J.; Li, S.; Chen, W.; Bazzano, L.; Sun, X.; Shen, L.; Liang, L.; Shen, Y.; Gu, X.; et al. Novel Metabolites Are Associated with Augmentation Index and Pulse Wave Velocity: Findings from the Bogalusa Heart Study. Am. J. Hypertens. 2019, 32, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Gaddi, A.; Cicero, A.F.; Wani, F.O.; Dormi, A.; Pasquarelli, V.; D’Addato, S. The realization of a project aimed at reducing the plasmatic lipid level in a large Italian population improves the mean calcium daily intake: The Brisighella Study. Eur. J. Clin. Nutr. 2001, 55, 97–106. [Google Scholar] [CrossRef][Green Version]
- Funtikova, A.; Baena-Díez, J.M.; Koebnick, C.; Gomez, S.F.; Covas, M.I.; Goday, A.; Schröder, H. Validity of a short diet-quality index to predict changes in anthropometric and cardiovascular risk factors: A simulation study. Eur. J. Clin. Nutr. 2012, 66, 1369–1371. [Google Scholar] [CrossRef]
- Cicero, A.F.; Reggi, A.; Tartagni, E.; Grandi, E.; D’Addato, S.; Borghi, C.; Brisighella Heart Study Group. Dietary determinants of oxidized-low-density lipoprotein antibodies in a sample of pharmacologically untreated non-smoker subjects: data from the Brisighella heart study. Adv. Clin. Exp. Med. 2013, 22, 69–76. [Google Scholar]
- Fogacci, F.; Cicero, A.F.G.; D’Addato, S.; Giovannini, M.; Borghi, C.; Brisighella Heart Study Group. Effect of spontaneous changes in dietary components and lipoprotein(a) levels: Data from the Brisighella Heart Study. Atherosclerosis 2017, 262, 202–204. [Google Scholar] [CrossRef]
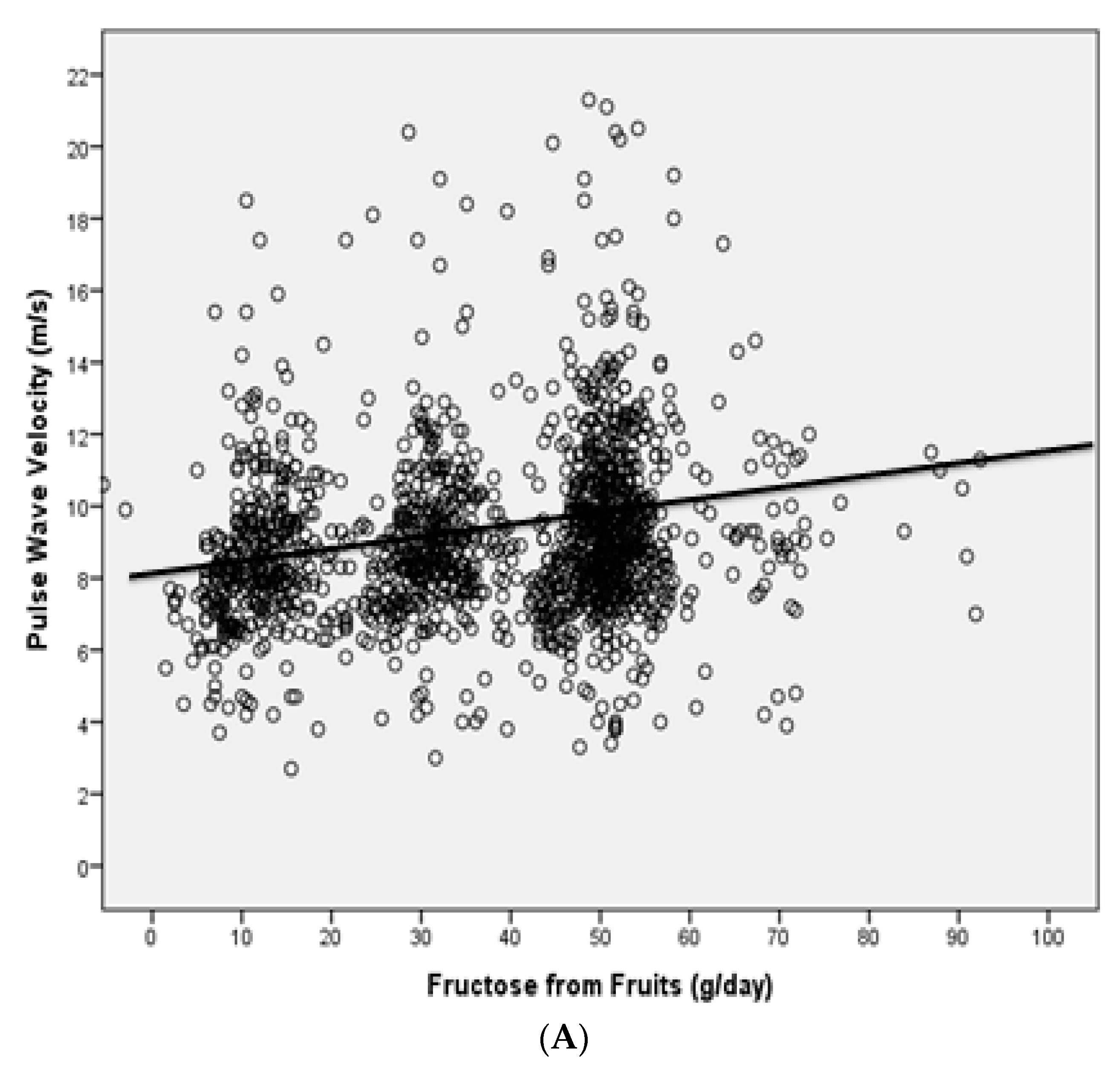
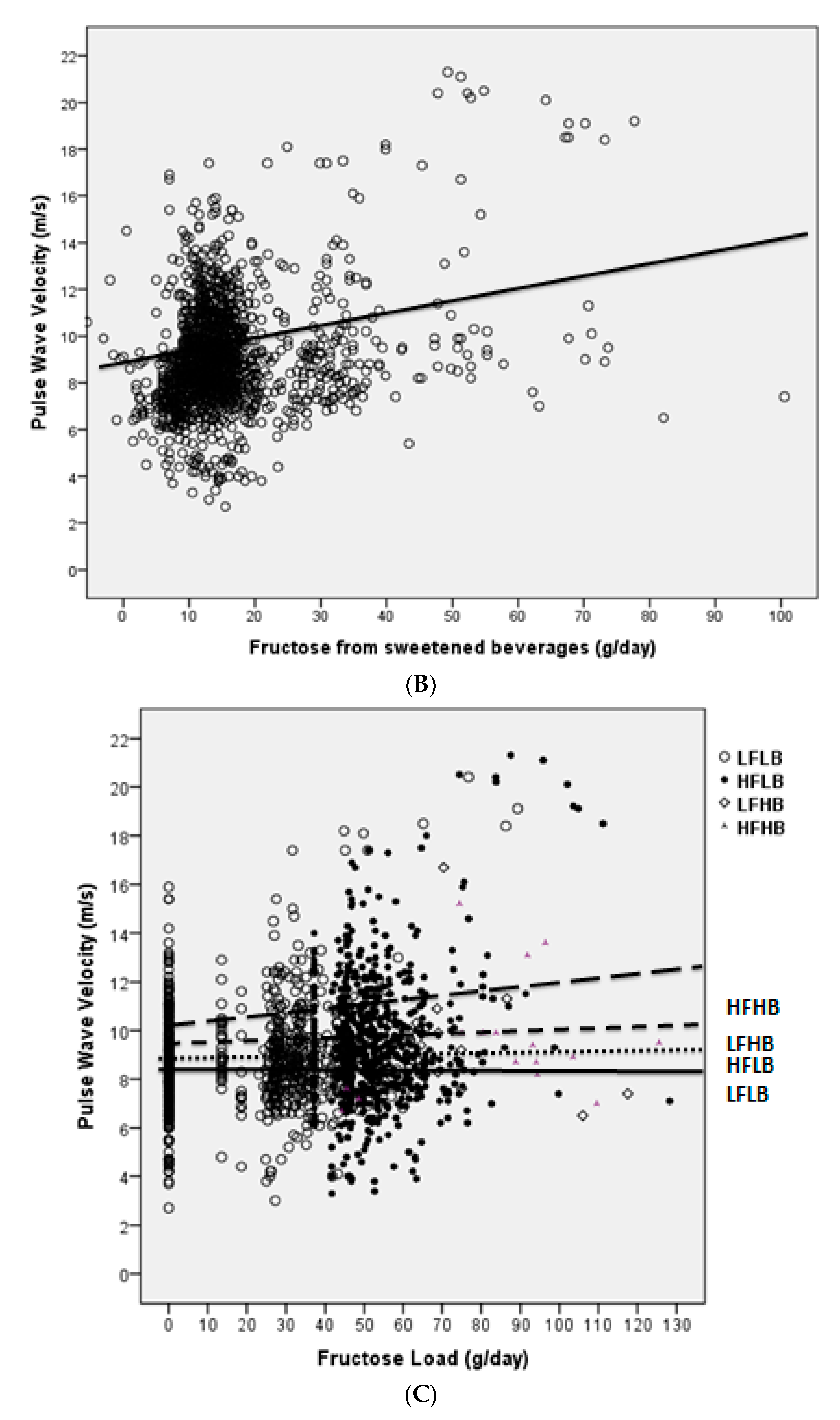
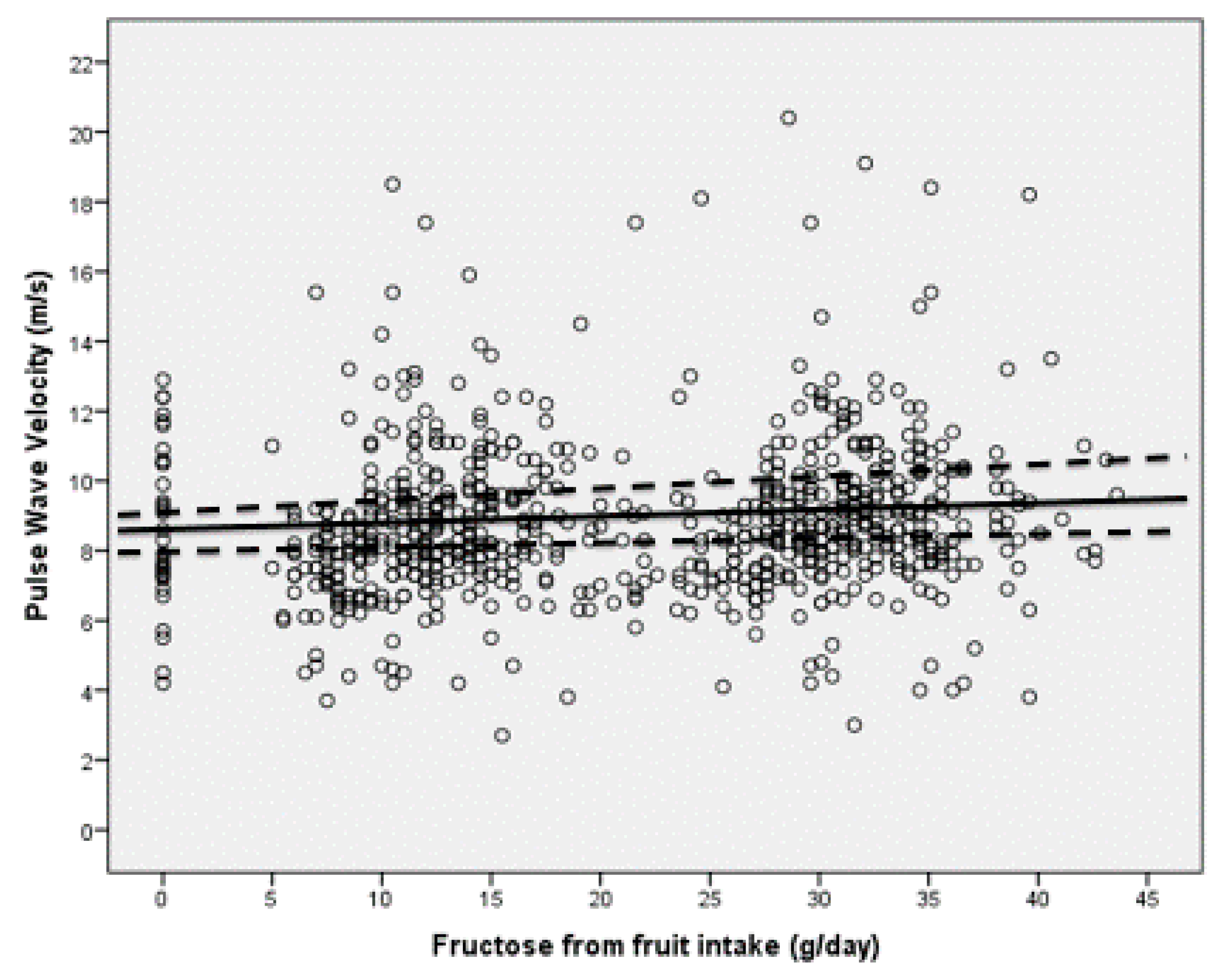
| LFLB (N. 437) | HFLB (N. 419) | LFHB (N. 133) | HFHB (N. 116) | |
|---|---|---|---|---|
| Gender | ||||
| - Males | 48% | 55% | 57% | 57% |
| - Females | 52% | 45% | 43% | 43% |
| Smoking habit | ||||
| - Current | 21% | 19% | 22% | 21% |
| - Former | 18% | 17% | 16% | 15% |
| - Never | 61% | 64% | 62% | 64% |
| Physical activity | ||||
| - No/Mild | 20% | 21% | 24% | 22% |
| - Moderate | 63% | 61% | 61% | 60% |
| - Intense | 17% | 18% | 15% | 18% |
| LFLB (N. 437) | HFLB (N. 419) | LFHB (N. 133) | HFHB (N. 116) | |
|---|---|---|---|---|
| Age (years) | 59.6 ± 15.6 | 61.9 ± 14.7 | 59.1 ± 16.8 | 58.7 ± 16.8 |
| Dietary Quality Index score | 39.8 ± 4.6 | 41.4 ± 4.4 | 38.9 ± 4.9 | 39.0 ± 5.0 |
| Fructose intake from fruits (g/day) | 6.6 ± 3.5 | 38.7 ± 9.7 * | 8.5 ± 3.8 | 38.4 ± 11.5 *# |
| Fructose intake from sugar sweetened beverages (g/day) | 7.1 ± 3.7 | 2.5 ± 2.6 * | 35.3 ± 12.2 * | 34.2 ± 12.9 *° |
| Body Mass Index (kg/m2) | 26.4 ± 4.5 | 26.7 ± 4.7 | 26.8 ± 4.0 | 27.1 ± 4.6 * |
| Total Cholesterol (mg/dL) | 216.8 ± 16.1 | 224.8 ± 15.3 | 226.3 ± 20.1 | 229.1 ± 20.7 |
| Triglycerides (mg/dL) | 140.44 ± 45.0 | 157.07 ± 46.0 * | 167.24 ± 42.0 *° | 183.19 ± 48.6 *°# |
| HDL- Cholesterol (mg/dL) | 52.4 ± 5.9 | 51.2 ± 4.5 | 46.2 ± 3.9 *° | 44.6 ± 4.8 *°# |
| LDL- Cholesterol (mg/dL) | 137.8 ± 16.2 | 143.3 ± 17.7 | 147.6 ± 23.3 * | 146.8 ± 20.5 * |
| Apolipoprotein AI (mg/dL) | 154.6 ± 18.9 | 153.1 ± 17.2 | 144.5 ± 16.1 | 144.6 ± 29.2 |
| Apolipoprotein B (mg/dL) | 90.82 ± 7.4 | 93.7 ± 7.1 | 94.5 ± 6.5 | 93.3 ± 6.9 |
| Fasting Plasma Glucose (mg/dL) | 91.5 ± 9.7 | 92.7 ± 9.7 | 94.7 ± 10.0 | 95.9 ± 14.2 * |
| AST (U/L) | 23.5 ± 4.9 | 23.5 ± 4.9 | 21.1 ± 4.1 | 23.2 ± 4.9 |
| ALT (U/L) | 25.8 ± 4.6 | 23.4 ± 4.4 | 22.8 ± 4.0 | 28.0 ± 5.0 |
| gamma-GT (U/L) | 27.7 ± 5.0 | 26.3 ± 8.1 | 27.5 ± 9.4 | 40.7 ± 9.0 *°# |
| Serum Uric Acid (mg/dL) | 5.3 ± 1.3 | 5.1 ± 1.2 | 5.7 ± 1.4 *° | 5.9 ± 1.5 *° |
| Creatinine (mg/dL) | 1.04 ± 0.17 | 1.03 ± 0.22 | 1.06 ± 0.19 | 1.03 ± 0.16 |
| Estimated GFR (mL/min) | 73.4 ± 5.2 | 74.0 ± 5.4 | 73.7 ± 6.1 | 74.5 ± 5.5 |
| LFLB (N. 437) | HFLB (N. 419) | LFHB (N. 133) | HFHB (N. 116) | |
|---|---|---|---|---|
| SBP (mmHg) | 139.3 ± 11.5 | 138.1 ± 12.2 | 142.5 ± 11.3 *° | 142.0 ± 15.5 *° |
| DBP (mmHg) | 734 ± 5.8 | 73.6 ± 5.7 | 74.5 ± 5.7 | 72.3 ± 6.3 |
| Heart Rate (bpm) | 64.0 ± 12.9 | 63.8 ± 10.4 | 66.4 ± 12.2 | 63.0 ± 10.1 |
| cfPWV (m/s) | 8.8 ± 2.1 | 8.4 ± 2.5 | 9.5 ± 2.4 *° | 9.8 ± 2.2 *° |
| 95%CI | ||||
|---|---|---|---|---|
| Parameter | Beta Coefficient | Lower Limit | Upper Limit | Sig, |
| Model 1 (Including fruit-derived fructose) | ||||
| Fasting Plasma Glucose(mg/dL) | 0.018 | 0.006 | 0.025 | 0.003 |
| Triglycerides(mg/dL) | 0.132 | 0.079 | 0.272 | 0.021 |
| Fruit-derived fructose | 0.184 | 0.091 | 0.302 | 0.009 |
| Model 2 (Including sugar-sweetened beverages) | ||||
| Estimated Glomerular Filtration Rate(mL/min) | −0.055 | −0.074 | −0.031 | <0.001 |
| Fasting Plasma Glucose(mg/dL) | 0.019 | 0.007 | 0.028 | 0.001 |
| Serum Uric Acid(mg/dL) | 0.389 | 0.229 | 0.492 | <0.001 |
| Triglycerides(mg/dL) | 0.233 | 0.115 | 0.394 | 0.012 |
| Sugar-sweetened beverages | 0.295 | 0.096 | 0.535 | 0.003 |
| Model 3 (Including fructose load from both fruits and sugar-sweetened beverages) | ||||
| Estimated Glomerular Filtration Rate(mL/min) | −0.043 | −0.052 | −0.035 | <0.001 |
| Fasting Plasma Glucose(mg/dL) | 0.015 | 0.008 | 0.022 | <0.001 |
| Serum Uric Acid(mg/dL) | 0.391 | 0.321 | 0.486 | 0.001 |
| Triglycerides(mg/dL) | 0.228 | 0.117 | 0.389 | 0.018 |
| Fructose Load | 0.310 | 0.099 | 0.522 | 0.004 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cicero, A.F.G.; Fogacci, F.; Desideri, G.; Grandi, E.; Rizzoli, E.; D’Addato, S.; Borghi, C. Arterial Stiffness, Sugar-Sweetened Beverages and Fruits Intake in a Rural Population Sample: Data from the Brisighella Heart Study. Nutrients 2019, 11, 2674. https://doi.org/10.3390/nu11112674
Cicero AFG, Fogacci F, Desideri G, Grandi E, Rizzoli E, D’Addato S, Borghi C. Arterial Stiffness, Sugar-Sweetened Beverages and Fruits Intake in a Rural Population Sample: Data from the Brisighella Heart Study. Nutrients. 2019; 11(11):2674. https://doi.org/10.3390/nu11112674
Chicago/Turabian StyleCicero, Arrigo F.G., Federica Fogacci, Giovambattista Desideri, Elisa Grandi, Elisabetta Rizzoli, Sergio D’Addato, and Claudio Borghi. 2019. "Arterial Stiffness, Sugar-Sweetened Beverages and Fruits Intake in a Rural Population Sample: Data from the Brisighella Heart Study" Nutrients 11, no. 11: 2674. https://doi.org/10.3390/nu11112674
APA StyleCicero, A. F. G., Fogacci, F., Desideri, G., Grandi, E., Rizzoli, E., D’Addato, S., & Borghi, C. (2019). Arterial Stiffness, Sugar-Sweetened Beverages and Fruits Intake in a Rural Population Sample: Data from the Brisighella Heart Study. Nutrients, 11(11), 2674. https://doi.org/10.3390/nu11112674








