Honey and Its Role in Relieving Multiple Facets of Atherosclerosis
Abstract
1. Introduction
2. Honey Composition and Antioxidant Activity
2.1. Honey Composition
2.2. Key Compositional Standards
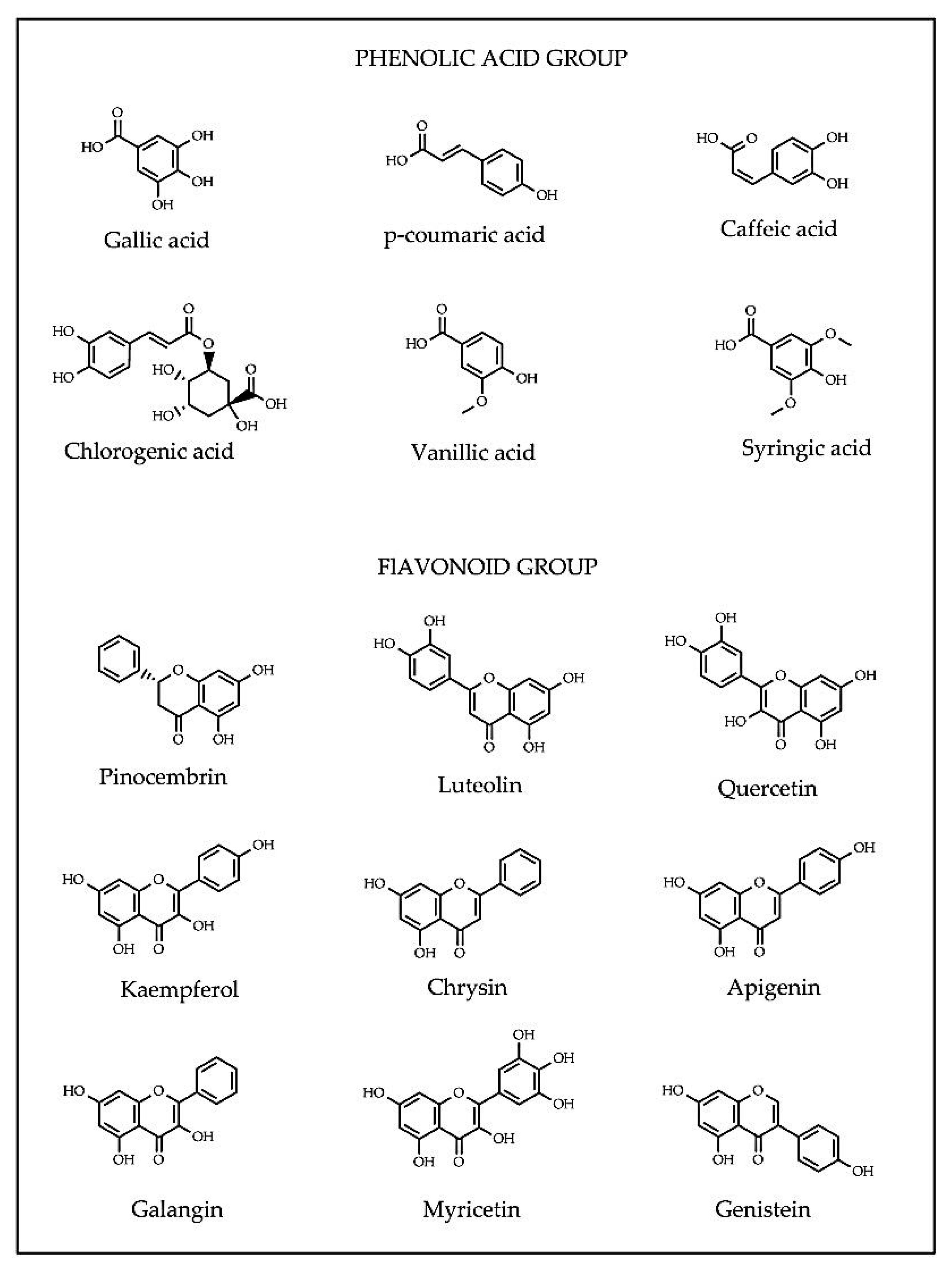
2.3. Antioxidant Capacity
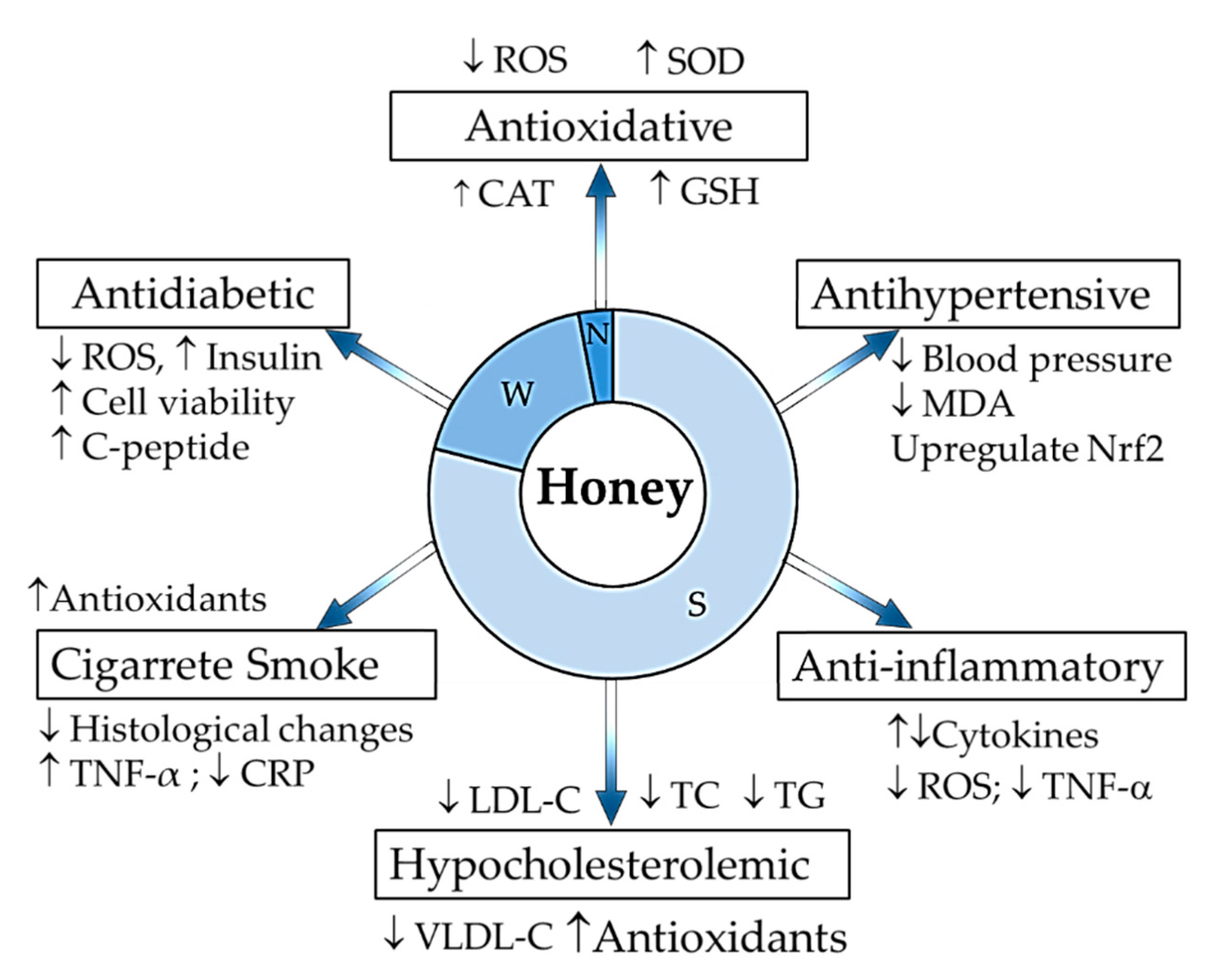
3. Honey in Relieving Multiple Facets of Atherosclerosis
3.1. Oxidative Damage
3.2. Inflammatory Responses
3.3. Hypercholesterolemia
3.4. Hypertension
3.5. Diabetes
3.6. Cigarette Smoking
4. Adverse Effects of Honey
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Toh, B.-H.; Kyaw, T.; Tipping, P.; Bobik, A. Chapter 71—Atherosclerosis. In The Autoimmune Diseases, 5th ed.; Rose, N.R., Mackay, I.R., Eds.; Academic Press: Boston, MA, USA, 2014; pp. 1049–1066. [Google Scholar]
- Torres, N.; Guevara-Cruz, M.; Velázquez-Villegas, L.A.; Tovar, A.R. Nutrition and atherosclerosis. Arch. Med. Res. 2015, 46, 408–426. [Google Scholar] [CrossRef] [PubMed]
- Falk, E. Pathogenesis of atherosclerosis. J. Am. Coll. Cardiol. 2006, 47, C7–C12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Cherubini, A.; Vigna, G.B.; Zuliani, G.; Ruggiero, C.; Senin, U.; Fellin, R. Role of antioxidants in atherosclerosis: Epidemiological and clinical update. Curr. Pharm. Des. 2005, 11, 2017–2032. [Google Scholar] [CrossRef] [PubMed]
- Poljsak, B. Strategies for Reducing or Preventing the Generation of Oxidative Stress. Oxid. Med. Cell. Longev. 2011, 2011, 194586. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, D. Healthy Heart: Strengthen Your Cardiovascular System Naturally; Storey Publishing: North Adams, MA, USA, 2017. [Google Scholar]
- Afrin, S.; Giampieri, F.; Gasparrini, M.; Forbes-Hernández, T.Y.; Cianciosi, D.; Reboredo-Rodriguez, P.; Amici, A.; Quiles, J.L.; Battino, M. The inhibitory effect of Manuka honey on human colon cancer HCT-116 and LoVo cell growth. Part 1: The suppression of cell proliferation, promotion of apoptosis and arrest of the cell cycle. Food Funct. 2018, 9, 2145–2157. [Google Scholar] [CrossRef]
- Ajibola, A.; Chamunorwa, J.P.; Erlwanger, K.H. Nutraceutical values of natural honey and its contribution to human health and wealth. Nutr. Metab. (Lond.) 2012, 9, 61. [Google Scholar] [CrossRef]
- Isidorov, V.A.; Czyżewska, U.; Jankowska, E.; Bakier, S. Determination of royal jelly acids in honey. Food Chem. 2011, 124, 387–391. [Google Scholar] [CrossRef]
- Juliano, C.; Magrini, G. Methylglyoxal, the Major Antibacterial Factor in Manuka Honey: An Alternative to Preserve Natural Cosmetics? Cosmetics 2019, 6, 1. [Google Scholar] [CrossRef]
- Afroz, R.; Tanvir, E.; Paul, S.; Bhoumik, N.C.; Gan, S.H.; Khalil, M. DNA damage inhibition properties of sundarban honey and its phenolic composition. J. Food Biochem. 2016, 40, 436–445. [Google Scholar] [CrossRef]
- Zumla, A.; Lulat, A. Honey: A remedy rediscovered. J. R. Soc. Med. 1989, 82, 384–385. [Google Scholar] [CrossRef] [PubMed]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S. Honey: A Novel Antioxidant. Molecules 2012, 17, 4400–4423. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Battino, M. Honey as a source of dietary antioxidants: Structures, bioavailability and evidence of protective effects against human chronic diseases. Curr. Med. Chem. 2013, 20, 621–638. [Google Scholar] [CrossRef] [PubMed]
- Escuredo, O.; Dobre, I.; Fernández-González, M.; Seijo, M.C. Contribution of botanical origin and sugar composition of honeys on the crystallization phenomenon. Food Chem. 2014, 149, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, P.M.; Gauche, C.; Gonzaga, L.V.; Costa, A.C.O.; Fett, R. Honey: Chemical composition, stability and authenticity. Food Chem. 2016, 196, 309–323. [Google Scholar] [CrossRef] [PubMed]
- Escuredo, O.; Miguez, M.; Fernandez-Gonzalez, M.; Carmen Seijo, M. Nutritional value and antioxidant activity of honeys produced in a European Atlantic area. Food Chem. 2013, 138, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.L.; Panyoyai, N.; Paramita, V.D.; Mantri, N.; Kasapis, S. Physicochemical and viscoelastic properties of honey from medicinal plants. Food Chem. 2018, 241, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Lampe, J.W. Health effects of vegetables and fruit: Assessing mechanisms of action in human experimental studies. Am. J. Clin. Nutr. 1999, 70, 475s–490s. [Google Scholar] [CrossRef]
- Liu, R.H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 2013, 4, 384S–392S. [Google Scholar] [CrossRef]
- Denisow, B.; Denisow-Pietrzyk, M. Biological and therapeutic properties of bee pollen: A review. J. Sci. Food Agric. 2016, 96, 4303–4309. [Google Scholar] [CrossRef]
- Anand, S.; Pang, E.; Livanos, G.; Mantri, N. Characterization of Physico-Chemical Properties and Antioxidant Capacities of Bioactive Honey Produced from Australian Grown Agastache rugosa and its Correlation with Colour and Poly-Phenol Content. Molecules 2018, 23, 108. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.; Gautam, S.; Sharma, A. Physical, biochemical and antioxidant properties of some Indian honeys. Food Chem. 2010, 118, 391–397. [Google Scholar] [CrossRef]
- Ahmed, S.; Sulaiman, S.A.; Baig, A.A.; Ibrahim, M.; Liaqat, S.; Fatima, S.; Jabeen, S.; Shamim, N.; Othman, N.H. Honey as a Potential Natural Antioxidant Medicine: An Insight into Its Molecular Mechanisms of Action. Oxid. Med. Cell. Longev. 2018, 2018, 8367846. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Gasparrini, M.; Forbes-Hernández, T.Y.; Mazzoni, L.; Giampieri, F. The composition and biological activity of honey: A focus on Manuka honey. Foods 2014, 3, 420–432. [Google Scholar] [CrossRef] [PubMed]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S. Effects of honey and its mechanisms of action on the development and progression of cancer. Molecules 2014, 19, 2497–2522. [Google Scholar] [CrossRef] [PubMed]
- Musa Özcan, M.; Al Juhaimi, F. Honey as source of natural antioxidants. J. Apic. Res. 2015, 54, 145–154. [Google Scholar] [CrossRef]
- Machado De-Melo, A.A.; Almeida-Muradian, L.B.D.; Sancho, M.T.; Pascual-Maté, A. Composition and properties of Apis mellifera honey: A review. J. Apic. Res. 2018, 57, 5–37. [Google Scholar] [CrossRef]
- Bogdanov, S.; Jurendic, T.; Sieber, R.; Gallmann, P. Honey for Nutrition and Health: A Review. J. Am. Coll. Nutr. 2013, 27, 677–689. [Google Scholar] [CrossRef]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, M.; Ataie-Jafari, A.; Hosseini, S.; Foruzanfar, M.H.; Rahmani, M.; Pajouhi, M. Effects of natural honey consumption in diabetic patients: An 8-week randomized clinical trial. Int. J. Food Sci. Nutr. 2009, 60, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Bantle, J.P. Dietary fructose and metabolic syndrome and diabetes. J. Nutr. 2009, 139, 1263S–1268S. [Google Scholar] [CrossRef] [PubMed]
- Deibert, P.; König, D.; Kloock, B.; Groenefeld, M.; Berg, A. Glycaemic and insulinaemic properties of some German honey varieties. Eur. J. Clin. Nutr. 2010, 64, 762. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, S.W.; Nepi, M.; Pacini, E. Nectaries and Nectar; Springer: Berlin, Germany, 2007; Volume 4. [Google Scholar]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Teixeira, J.; Gaspar, A.; Garrido, E.M.; Garrido, J.; Borges, F. Hydroxycinnamic acid antioxidants: An electrochemical overview. BioMed Res. Int. 2013, 2013, 251754. [Google Scholar] [CrossRef] [PubMed]
- Hollman, P.C.H.; Katan, M.B. Dietary flavonoids: Intake, health effects and bioavailability. Food Chem. Toxicol. 1999, 37, 937–942. [Google Scholar] [CrossRef]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.A.; Martos, I.; Ferreres, F.; Radovic, B.S.; Anklam, E. HPLC flavonoid profiles as markers for the botanical origin of European unifloral honeys. J. Sci. Food Agric. 2001, 81, 485–496. [Google Scholar] [CrossRef]
- Yao, L.; Jiang, Y.; Singanusong, R.; D’Arcy, B.; Datta, N.; Caffin, N.; Raymont, K. Flavonoids in Australian Melaleuca, Guioa, Lophostemon, Banksia and Helianthus honeys and their potential for floral authentication. Food Res. Int. 2004, 37, 166–174. [Google Scholar] [CrossRef]
- Anklam, E. A review of the analytical methods to determine the geographical and botanical origin of honey. Food Chem. 1998, 63, 549–562. [Google Scholar] [CrossRef]
- Yao, L.; Data, N.; Tomás-Barberán, F.A.; Ferreres, F.; Martos, I.; Singanusong, R. Flavonoids, phenolic acids and abscisic acid in Australian and New Zealand Leptospermum honeys. Food Chem. 2003, 81, 159–168. [Google Scholar] [CrossRef]
- Pisani, A.; Protano, G.; Riccobono, F. Minor and trace elements in different honey types produced in Siena County (Italy). Food Chem. 2008, 107, 1553–1560. [Google Scholar] [CrossRef]
- Chua, L.S.; Abdul-Rahaman, N.L.; Sarmidi, M.R.; Aziz, R. Multi-elemental composition and physical properties of honey samples from Malaysia. Food Chem. 2012, 135, 880–887. [Google Scholar] [CrossRef]
- EU. Council Directive 2001/110 relating to honey. Off. J. Eur. Communities 2001, L 10, 47–52. [Google Scholar]
- Commission, C.A. Codex Standard for Honey, CODEX STAN 12-1981; Food and Agriculture Organization of the United Nations: Rome, Italy; The World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Manzanares, A.B.; García, Z.H.; Galdón, B.R.; Rodríguez-Rodríguez, E.M.; Romero, C.D. Physicochemical characteristics and pollen spectrum of monofloral honeys from Tenerife, Spain. Food Chem. 2017, 228, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Mossel, B.; Bhandari, B.; D’Arcy, B.; Caffin, N. Determination of viscosity of some Australian honeys based on composition. Int. J. Food Prop. 2003, 6, 87–97. [Google Scholar] [CrossRef]
- Kędzierska-Matysek, M.; Florek, M.; Wolanciuk, A.; Skałecki, P.; Litwińczuk, A. Characterisation of viscosity, colour, 5-hydroxymethylfurfural content and diastase activity in raw rape honey (Brassica napus) at different temperatures. J. Food Sci. Technol. 2016, 53, 2092–2098. [Google Scholar] [CrossRef] [PubMed]
- Smanalieva, J.; Senge, B. Analytical and rheological investigations into selected unifloral German honey. Eur. Food Res. Technol. 2009, 229, 107–113. [Google Scholar] [CrossRef]
- Dobre, I.; Georgescu, L.A.; Alexe, P.; Escuredo, O.; Seijo, M.C. Rheological behavior of different honey types from Romania. Food Res. Int. 2012, 49, 126–132. [Google Scholar] [CrossRef]
- Bogdanov, S.; Martin, P.; Lullmann, C. Harmonised Methods of the International Honey Commission; Swiss Bee Research Centre: FAM, Liebefeld, Switzerland, 2002. [Google Scholar]
- Terrab, A.; Recamales, A.F.; Hernanz, D.; Heredia, F.J. Characterisation of Spanish thyme honeys by their physicochemical characteristics and mineral contents. Food Chem. 2004, 88, 537–542. [Google Scholar] [CrossRef]
- Mateo, R.; Bosch-Reig, F. Classification of Spanish unifloral honeys by discriminant analysis of electrical conductivity, color, water content, sugars, and pH. J. Agric. Food Chem. 1998, 46, 393–400. [Google Scholar] [CrossRef]
- Gomes, S.; Dias, L.G.; Moreira, L.L.; Rodrigues, P.; Estevinho, L. Physicochemical, microbiological and antimicrobial properties of commercial honeys from Portugal. Food Chem. Toxicol. 2010, 48, 544–548. [Google Scholar] [CrossRef]
- Kivrak, Ş.; Kivrak, I.; Karababa, E. Characterization of Turkish honeys regarding of physicochemical properties, and their adulteration analysis. Food Sci. Technol. (Campinas) 2017, 37, 80–89. [Google Scholar] [CrossRef]
- Estevinho, L.M.; Feás, X.; Seijas, J.A.; Vázquez-Tato, M.P. Organic honey from Trás-Os-Montes region (Portugal): Chemical, palynological, microbiological and bioactive compounds characterization. Food Chem. Toxicol. 2012, 50, 258–264. [Google Scholar] [CrossRef]
- Singh, N.; Bath, P.K. Quality evaluation of different types of Indian honey. Food Chem. 1997, 58, 129–133. [Google Scholar] [CrossRef]
- Del Campo, G.; Zuriarrain, J.; Zuriarrain, A.; Berregi, I. Quantitative determination of carboxylic acids, amino acids, carbohydrates, ethanol and hydroxymethylfurfural in honey by 1H NMR. Food Chem. 2016, 196, 1031–1039. [Google Scholar] [CrossRef]
- Silva, L.R.; Videira, R.; Monteiro, A.P.; Valentão, P.; Andrade, P.B. Honey from Luso region (Portugal): Physicochemical characteristics and mineral contents. Microchem. J. 2009, 93, 73–77. [Google Scholar] [CrossRef]
- Soares, S.; Pinto, D.; Rodrigues, F.; Alves, R.C.; Oliveira, M. Portuguese honeys from different geographical and botanical origins: A 4-year stability study regarding quality parameters and antioxidant activity. Molecules 2017, 22, 1338. [Google Scholar] [CrossRef]
- Kek, S.P.; Chin, N.L.; Yusof, Y.A.; Tan, S.W.; Chua, L.S. Classification of entomological origin of honey based on its physicochemical and antioxidant properties. Int. J. Food Prop. 2017, 20, S2723–S2738. [Google Scholar] [CrossRef]
- Gheldof, N.; Engeseth, N. Antioxidant capacity of honeys from various floral sources based on the determination of oxygen radical absorbance capacity and inhibition of in vitro lipoprotein oxidation in human serum samples. J. Agric. Food Chem. 2002, 50, 3050–3055. [Google Scholar] [CrossRef]
- Pontis, J.A.; Costa, L.A.M.A.D.; Silva, S.J.R.D.; Flach, A. Color, phenolic and flavonoid content, and antioxidant activity of honey from Roraima, Brazil. Food Sci. Technol. (Campinas) 2014, 34, 69–73. [Google Scholar] [CrossRef]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activiity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Giampieri, F.; Brenciani, A.; Mazzoni, L.; Gasparrini, M.; González-Paramás, A.M.; Santos-Buelga, C.; Morroni, G.; Simoni, S.; Forbes-Hernández, T.Y. Apis mellifera vs. Melipona beecheii Cuban polifloral honeys: A comparison based on their physicochemical parameters, chemical composition and biological properties. LWT Food Sci. Technol. 2018, 87, 272–279. [Google Scholar] [CrossRef]
- Rosa, A.; Tuberoso, C.I.G.; Atzeri, A.; Melis, M.P.; Bifulco, E.; Dessì, M.A. Antioxidant profile of strawberry tree honey and its marker homogentisic acid in several models of oxidative stress. Food Chem. 2011, 129, 1045–1053. [Google Scholar] [CrossRef]
- Baltrušaitytė, V.; Venskutonis, P.R.; Čeksterytė, V. Radical scavenging activity of different floral origin honey and beebread phenolic extracts. Food Chem. 2007, 101, 502–514. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Dobersek, U.; Jamnik, M.; Golob, T. Evaluation of the phenolic content, antioxidant activity and colour of Slovenian honey. Food Chem. 2007, 105, 822–828. [Google Scholar] [CrossRef]
- Can, Z.; Yildiz, O.; Sahin, H.; Akyuz Turumtay, E.; Silici, S.; Kolayli, S. An investigation of Turkish honeys: Their physico-chemical properties, antioxidant capacities and phenolic profiles. Food Chem. 2015, 180, 133–141. [Google Scholar] [CrossRef]
- Socha, R.; Juszczak, L.; Pietrzyk, S.; Fortuna, T. Antioxidant activity and phenolic composition of herbhoneys. Food Chem. 2009, 113, 568–574. [Google Scholar] [CrossRef]
- Ulloa, P.A.; Maia, M.; Brigas, A.F. Physicochemical Parameters and Bioactive Compounds of Strawberry Tree (Arbutus unedo L.) Honey. J. Chem. 2015, 2015, 602792. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef]
- Apak, R.A.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant activity/capacity measurement. 2. Hydrogen atom transfer (HAT)-based, mixed-mode (electron transfer (ET)/HAT), and lipid peroxidation assays. J. Agric. Food Chem. 2016, 64, 1028–1045. [Google Scholar] [CrossRef]
- Li, Q.; Wang, X.; Chen, J.; Liu, C.; Li, T.; McClements, D.J.; Dai, T.; Liu, J. Antioxidant activity of proanthocyanidins-rich fractions from Choerospondias axillaris peels using a combination of chemical-based methods and cellular-based assay. Food Chem. 2016, 208, 309–317. [Google Scholar] [CrossRef]
- Finkel, T.; Holbrook, N. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74. [Google Scholar] [CrossRef]
- Hegazi, A.G.; Abd El-Hady, F.K. Influence of Honey on the Suppression of Human Low Density Lipoprotein (LDL) Peroxidation (In Vitro). Evid. Based Complement. Altern. Med. 2009, 6, 113–121. [Google Scholar] [CrossRef]
- Gheldof, N.; Wang, X.H.; Engeseth, N.J. Buckwheat honey increases serum antioxidant capacity in humans. J. Agric. Food Chem. 2003, 51, 1500–1505. [Google Scholar] [CrossRef]
- Pérez, E.; Rodríguez-Malaver, A.J.; Vit, P. Antioxidant capacity of Venezuelan honey in wistar rat homogenates. J. Med. Food 2006, 9, 510–516. [Google Scholar] [CrossRef]
- Alvarez Suarez, J.M.; Giampieri, F.; Damiani, E.; Astolfi, P.; Fattorini, D.; Regoli, F.; Quiles, J.L.; Battino, M. Radical-scavenging activity, protective effect against lipid peroxidation and mineral contents of monofloral Cuban honeys. Plant Foods Hum. Nutr. 2012, 67, 31–38. [Google Scholar] [CrossRef]
- Blasa, M.; Candiracci, M.; Accorsi, A.; Piacentini, M.P.; Piatti, E. Honey flavonoids as protection agents against oxidative damage to human red blood cells. Food Chem. 2007, 104, 1635–1640. [Google Scholar] [CrossRef]
- Fiorani, M.; Accorsi, A.; Blasa, M.; Diamantini, G.; Piatti, E. Flavonoids from Italian multifloral honeys reduce the extracellular ferricyanide in human red blood cells. J. Agric. Food Chem. 2006, 54, 8328–8334. [Google Scholar] [CrossRef]
- Cesquini, M.; Torsoni, M.; Stoppa, G.; Ogo, S.T. t-BOOH-induced oxidative damage in sickle red blood cells and the role of flavonoids. Biomed. Pharmacother. 2003, 57, 124–129. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Giampieri, F.; González-Paramás, A.M.; Damiani, E.; Astolfi, P.; Martinez-Sanchez, G.; Bompadre, S.; Quiles, J.L.; Santos-Buelga, C.; Battino, M. Phenolics from monofloral honeys protect human erythrocyte membranes against oxidative damage. Food Chem. Toxicol. 2012, 50, 1508–1516. [Google Scholar] [CrossRef]
- Beretta, G.; Orioli, M.; Facino, R.M. Antioxidant and radical scavenging activity of honey in endothelial cell cultures (EA. hy926). Planta Med. 2007, 73, 1182–1189. [Google Scholar] [CrossRef]
- Sahhugi, Z.; Hasenan, S.M.; Jubri, Z. Protective effects of gelam honey against oxidative damage in young and aged rats. Oxid. Med. Cell. Longev. 2014, 2014, 673628. [Google Scholar] [CrossRef]
- Busserolles, J.; Gueux, E.; Rock, E.; Mazur, A.; Rayssiguier, Y. Substituting honey for refined carbohydrates protects rats from hypertriglyceridemic and prooxidative effects of fructose. J. Nutr. 2002, 132, 3379–3382. [Google Scholar] [CrossRef]
- Schramm, D.D.; Karim, M.; Schrader, H.R.; Holt, R.R.; Cardetti, M.; Keen, C.L. Honey with high levels of antioxidants can provide protection to healthy human subjects. J. Agric. Food Chem. 2003, 51, 1732–1735. [Google Scholar] [CrossRef]
- Flammer, A.J.; Anderson, T.; Celermajer, D.S.; Creager, M.A.; Deanfield, J.; Ganz, P.; Hamburg, N.M.; Lüscher, T.F.; Shechter, M.; Taddei, S. The assessment of endothelial function: From research into clinical practice. Circulation 2012, 126, 753–767. [Google Scholar] [CrossRef]
- Aird, W.C. Phenotypic heterogeneity of the endothelium: I. Structure, function, and mechanisms. Circ. Res. 2007, 100, 158–173. [Google Scholar] [CrossRef]
- Suwalsky, M.; Orellana, P.; Avello, M.; Villena, F. Protective effect of Ugni molinae Turcz against oxidative damage of human erythrocytes. Food Chem. Toxicol. 2007, 45, 130–135. [Google Scholar] [CrossRef]
- Rakha, M.K.; Nabil, Z.I.; Hussein, A.A. Cardioactive and vasoactive effects of natural wild honey against cardiac malperformance induced by hyperadrenergic activity. J. Med. Food 2008, 11, 91–98. [Google Scholar] [CrossRef]
- Gheldof, N.; Wang, X.-H.; Engeseth, N.J. Identification and quantification of antioxidant components of honeys from various floral sources. J. Agric. Food Chem. 2002, 50, 5870–5877. [Google Scholar] [CrossRef]
- Peake, J.M.; Suzuki, K.; Coombes, J.S. The influence of antioxidant supplementation on markers of inflammation and the relationship to oxidative stress after exercise. J. Nutr. Biochem. 2007, 18, 357–371. [Google Scholar] [CrossRef]
- Woo, K.J.; Jeong, Y.-J.; Inoue, H.; Park, J.-W.; Kwon, T.K. Chrysin suppresses lipopolysaccharide-induced cyclooxygenase-2 expression through the inhibition of nuclear factor for IL-6 (NF-IL6) DNA-binding activity. FEBS Lett. 2005, 579, 705–711. [Google Scholar] [CrossRef]
- Kleemann, R.; Verschuren, L.; Morrison, M.; Zadelaar, S.; van Erk, M.J.; Wielinga, P.Y.; Kooistra, T. Anti-inflammatory, anti-proliferative and anti-atherosclerotic effects of quercetin in human in vitro and in vivo models. Atherosclerosis 2011, 218, 44–52. [Google Scholar] [CrossRef]
- Kotanidou, A.; Xagorari, A.; Bagli, E.; Kitsanta, P.; Fotsis, T.; Papapetropoulos, A.; Roussos, C. Luteolin reduces lipopolysaccharide-induced lethal toxicity and expression of proinflammatory molecules in mice. Am. J. Respir. Crit. Care Med. 2002, 165, 818–823. [Google Scholar] [CrossRef]
- Kassim, M.; Yusoff, K.M.; Ong, G.; Sekaran, S.; Yusof, M.Y.B.M.; Mansor, M. Gelam honey inhibits lipopolysaccharide-induced endotoxemia in rats through the induction of heme oxygenase-1 and the inhibition of cytokines, nitric oxide, and high-mobility group protein B1. Fitoterapia 2012, 83, 1054–1059. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, R.A.; Mesaik, M.A. Anti inflammatory effect of natural honey on bovine thrombin-induced oxidative burst in phagocytes. Phytother. Res. 2009, 23, 801–808. [Google Scholar] [CrossRef]
- Bean, A. Investigating the Anti-Inflammatory Activity of Honey. Ph.D. Thesis, University of Waikato, Waikato, New Zealand, 2012. [Google Scholar]
- Kassim, M.; Achoui, M.; Mustafa, M.R.; Mohd, M.A.; Yusoff, K.M. Ellagic acid, phenolic acids, and flavonoids in Malaysian honey extracts demonstrate in vitro anti-inflammatory activity. Nutr. Res. 2010, 30, 650–659. [Google Scholar] [CrossRef]
- Van den Berg, A.; Van den Worm, E.; van Ufford, H.Q.; Halkes, S.; Hoekstra, M.; Beukelman, C. An in vitro examination of the antioxidant and anti-inflammatory properties of buckwheat honey. J. Wound Care 2008, 17, 172–179. [Google Scholar] [CrossRef]
- Gill, S.; Chow, R.; Brown, A. Sterol regulators of cholesterol homeostasis and beyond: The oxysterol hypothesis revisited and revised. Prog. Lipid Res. 2008, 47, 391–404. [Google Scholar] [CrossRef]
- Yung, L.; Leung, F.; Wong, W.; Tian, X.; Yung, L.; Chen, Z.; Yao, X.; Huang, Y. Tea polyphenols benefit vascular function. Inflammopharmacology 2008, 16, 230–234. [Google Scholar] [CrossRef]
- Chepulis, L.; Starkey, N. The Long-Term Effects of Feeding Honey Compared with Sucrose and a Sugar-Free Diet on Weight Gain, Lipid Profiles, and DEXA Measurements in Rats. J. Food Sci. 2008, 73, H1–H7. [Google Scholar] [CrossRef]
- Nemoseck, T.M.; Carmody, E.G.; Furchner-Evanson, A.; Gleason, M.; Li, A.; Potter, H.; Rezende, L.M.; Lane, K.J.; Kern, M. Honey promotes lower weight gain, adiposity, and triglycerides than sucrose in rats. Nutr. Res. 2011, 31, 55–60. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Nwobodo, N.N.; Akpan, J.L.; Okorie, U.A.; Ezeonu, C.T.; Ezeokpo, B.C.; Nwadike, K.I.; Erhiano, E.; Abdul Wahab, M.S.; Sulaiman, S.A. Nigerian Honey Ameliorates Hyperglycemia and Dyslipidemia in Alloxan-Induced Diabetic Rats. Nutrients 2016, 8, 95. [Google Scholar] [CrossRef]
- Alagwu, E.; Okwara, J.; Nneli, R.; Osim, E. Effect of honey intake on serum cholesterol, triglycerides and lipoprotein levels in albino rats and potential benefits on risks of coronary heart disease. Niger. J. Physiol. Sci. 2011, 26, 161–165. [Google Scholar]
- Khalil, M.; Tanvir, E.; Afroz, R.; Sulaiman, S.A.; Gan, S.H. Cardioprotective effects of tualang honey: Amelioration of cholesterol and cardiac enzymes levels. BioMed Res. Int. 2015, 2015, 286051. [Google Scholar] [CrossRef]
- Mohamed, Z.B.H.; Alfarisi, H.A.H.; Abdullah, N.Z.; Harun, N.; Muhammad, N.; Rahim, R.A. Renoprotective Role of Tualang Honey against High Cholesterol Diet Induced Acute Kidney Diseases in an Animal Model. J. Appl. Pharm. Sci. 2017, 7, 97–101. [Google Scholar]
- Samat, S.; Kanyan Enchang, F.; Nor Hussein, F.; Ismail, W.; Iryani, W. Four-Week Consumption of Malaysian Honey Reduces Excess Weight Gain and Improves Obesity-Related Parameters in High Fat Diet Induced Obese Rats. Evid. Based Complement. Altern. Med. 2017, 2017, 1342150. [Google Scholar] [CrossRef]
- Bezerra, M.L.R.R.; de Souza, E.L.; Sousa, J.B.; Lima, M.; Alves, A.F.; das Graças Almeida, M.; Alves, R.C.C.; de Araújo, E.V.V.; Soares, N.L.; da Silva, G.D.A. Effects of honey from Mimosa quadrivalvis L.(malicia) produced by the Melipona subnitida D.(jandaira) stingless bee on lipid metabolism, antioxidant status and intestinal health of dyslipidaemic rats. Food Funct. 2018, 9, 4480–4492. [Google Scholar] [CrossRef]
- Al Waili, N.S. Natural honey lowers plasma glucose, C-reactive protein, homocysteine, and blood lipids in healthy, diabetic, and hyperlipidemic subjects: Comparison with dextrose and sucrose. J. Med. Food 2004, 7, 100–107. [Google Scholar] [CrossRef]
- Yaghoobi, N.; Al Waili, N.; Ghayour-Mobarhan, M.; Parizadeh, S.M.; Abasalti, Z.; Yaghoobi, Z.; Yaghoobi, F.; Esmaeili, H.; Kazemi-Bajestani, S.M.; Aghasizadeh, R.; et al. Natural honey and cardiovascular risk factors; effects on blood glucose, cholesterol, triacylglycerole, CRP, and body weight compared with sucrose. Sci. World J. 2008, 8, 463–469. [Google Scholar] [CrossRef]
- Whitfield, P.; Parry-Strong, A.; Walsh, E.; Weatherall, M.; Krebs, J.D. The effect of a cinnamon-, chromium- and magnesium-formulated honey on glycaemic control, weight loss and lipid parameters in type 2 diabetes: An open-label cross-over randomised controlled trial. Eur. J. Nutr. 2016, 55, 1123–1131. [Google Scholar] [CrossRef]
- Duarte, M.; Loro, V.; Rocha, J.; Leal, D.; Bem, A.; Dorneles, A.; Morsch, V.; Schetinger, M. Enzymes that hydrolyze adenine nucleotides of patients with hypercholesterolemia and inflammatory processes. FEBS J. 2007, 274, 2707–2714. [Google Scholar] [CrossRef]
- Dobrosielski, D.A.; Papandreou, C.; Patil, S.P.; Salas-Salvadó, J. Diet and exercise in the management of obstructive sleep apnoea and cardiovascular disease risk. Eur. Respir. J. 2017, 26, 160110. [Google Scholar] [CrossRef]
- Khalil, M.; Sulaiman, S. The potential role of honey and its polyphenols in preventing heart disease: A review. Afr. J. Tradit. Complement. Altern. Med. 2010, 7, 315–321. [Google Scholar] [CrossRef]
- Ademosun, A.O.; Oboh, G.; Passamonti, S.; Tramer, F.; Ziberna, L.; Boligon, A.A.; Athayde, M.L. Phenolics from grapefruit peels inhibit HMG-CoA reductase and angiotensin-I converting enzyme and show antioxidative properties in endothelial EA. Hy 926 cells. Food Sci. Hum. Wellness 2015, 4, 80–85. [Google Scholar] [CrossRef]
- Al-Naqeb, G.; Ismail, M.; Bagalkotkar, G.; Adamu, H.A. Vanillin rich fraction regulates LDLR and HMGCR gene expression in HepG2 cells. Food Res. Int. 2010, 43, 2437–2443. [Google Scholar] [CrossRef]
- Wong, T.Y.; Lin, S.-M.; Leung, L.K. The flavone luteolin suppresses SREBP-2 expression and post-translational activation in hepatic cells. PLoS ONE 2015, 10, e0135637. [Google Scholar] [CrossRef]
- Mbikay, M.; Sirois, F.; Simoes, S.; Mayne, J.; Chrétien, M. Quercetin-3-glucoside increases low-density lipoprotein receptor (LDLR) expression, attenuates proprotein convertase subtilisin/kexin 9 (PCSK9) secretion, and stimulates LDL uptake by Huh7 human hepatocytes in culture. FEBS Open Bio 2014, 4, 755–762. [Google Scholar] [CrossRef]
- Tul-Noor, Z.; Khan, T.A.; Mejia, S.B.; de Souza, R.; Sievenpiper, J.L. The Effect of Honey Intake on Lipid Risk Factors: A Systematic Review and Meta-Analysis of Controlled Trials. FASEB J. 2017, 31, 966.23. [Google Scholar]
- Panchal, S.K.; Poudyal, H.; Brown, L. Quercetin ameliorates cardiovascular, hepatic, and metabolic changes in diet-induced metabolic syndrome in rats. J. Nutr. 2012, 142, 1026–1032. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S.; Sirajudeen, K.N.S.; Salleh, S.; Gurtu, S. Honey Supplementation in Spontaneously Hypertensive Rats Elicits Antihypertensive Effect via Amelioration of Renal Oxidative Stress. Oxid. Med. Cell. Longev. 2012, 2012, 374037. [Google Scholar] [CrossRef]
- Erejuwa, O.; Sulaiman, S.; Suhaimi, M.; Sirajudeen, K.; Salleh, S.; Gurtu, S. Impaired Nrf2-ARE pathway contributes to increased oxidative damage in kidney of spontaneously hypertensive rats: Effect of antioxidant (honey). Int. J. Cardiol. 2011, 152, S45. [Google Scholar] [CrossRef]
- Zhang, Y.-J.; Gan, R.-Y.; Li, S.; Zhou, Y.; Li, A.-N.; Xu, D.-P.; Li, H.-B. Antioxidant phytochemicals for the prevention and treatment of chronic diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef]
- Batumalaie, K.; Qvist, R.; Yusof, K.M.; Ismail, I.S.; Sekaran, S.D. The antioxidant effect of the Malaysian Gelam honey on pancreatic hamster cells cultured under hyperglycemic conditions. Clin. Exp. Med. 2014, 14, 185–195. [Google Scholar] [CrossRef]
- Gholami, M.; Hemmati, M.; Taheri-Ghahfarokhi, A.; Hoshyar, R.; Moossavi, M. Expression of glucokinase, glucose 6-phosphatase, and stress protein in streptozotocin-induced diabetic rats treated with natural honey. Int. J. Diabetes Dev. Ctries. 2016, 36, 125–131. [Google Scholar] [CrossRef]
- Abdulrhman, M.; El-Hefnawy, M.; Hussein, R.; El-Goud, A.A. The glycemic and peak incremental indices of honey, sucrose and glucose in patients with type 1 diabetes mellitus: Effects on C-peptide level—A pilot study. Acta Diabetol. 2011, 48, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, J.A.; Barua, R.S. The pathophysiology of cigarette smoking and cardiovascular disease: An update. J. Am. Coll. Cardiol. 2004, 43, 1731–1737. [Google Scholar] [CrossRef]
- Mohamed, M.; Sulaiman, S.A.; Jaafar, H.; Sirajudeen, K.N.S. Antioxidant protective effect of honey in cigarette smoke-induced testicular damage in rats. Int. J. Mol. Sci. 2011, 12, 5508–5521. [Google Scholar] [CrossRef] [PubMed]
- Ghazali, W.S.W.; Romli, A.C.; Mohamed, M. Effects of honey supplementation on inflammatory markers among chronic smokers: A randomized controlled trial. BMC Complement. Altern. Med. 2017, 17, 175. [Google Scholar] [CrossRef]
- Wahdan, H.A.L. Causes of the antimicrobial activity of honey. Infection 1998, 26, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Kwakman, P.H.; te Velde, A.A.; de Boer, L.; Speijer, D.; Vandenbroucke-Grauls, C.M.; Zaat, S.A. How honey kills bacteria. FASEB J. 2010, 24, 2576–2582. [Google Scholar] [CrossRef] [PubMed]
- Nevas, M.; Hielm, S.; Lindström, M.; Horn, H.; Koivulehto, K.; Korkeala, H. High prevalence of Clostridium botulinum types A and B in honey samples detected by polymerase chain reaction. Int. J. Food Microbiol. 2002, 72, 45–52. [Google Scholar] [CrossRef]
- Al-Waili, N.; Salom, K.; Al-Ghamdi, A.; Ansari, M.J. Antibiotic, pesticide, and microbial contaminants of honey: Human health hazards. Sci. World J. 2012, 2012, 930849. [Google Scholar] [CrossRef] [PubMed]
- Schocken-Iturrino, R.P.; Carneiro, M.C.; Kato, E.; Sorbara, J.O.; Rossi, O.D.; Gerbasi, L.E. Study of the presence of the spores of Clostridium botulinum in honey in Brazil. FEMS Immunol. Med. Microbiol. 1999, 24, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Satorres, S.; Alcaraz, L.; Centorbi, H.; Fernández, R. Detection of Clostridium botulinum spores in honey. Rev. Argent. Microbiol. 1997, 29, 147–151. [Google Scholar]
- Nakano, H.; Sakagucki, G. An unusually heavy contamination of honey products by Clostridium botulinum type F and Bacillus alvei. FEMS Microbiol. Lett. 1991, 79, 171–178. [Google Scholar] [CrossRef]
- Ahmed, A.K.J.; Hoekstra, M.J.; Hage, J.J.; Karim, R.B. Honey-medicated dressing: Transformation of an ancient remedy into modern therapy. Ann. Plast. Surg. 2003, 50, 143–148. [Google Scholar] [CrossRef]
- Okuyan, E.; Uslu, A.; Ozan Levent, M. Cardiac effects of “mad honey”: A case series. Clin. Toxicol. 2010, 48, 528–532. [Google Scholar] [CrossRef]
| Proximates (g) | Minerals (mg) | Vitamins (mg) | |||
|---|---|---|---|---|---|
| Fructose | 38.2 | Calcium | 3–31 | Ascorbic acid | 2.2–2.5 |
| Glucose | 31.3 | Potassium | 40.0–3500.0 | Thiamin | 0.0–0.01 |
| Sucrose | 0.7 | Copper | 0.02–0.60 | Riboflavin | 0.01–0.02 |
| Other disaccharides | 5.0 | Iron | 0.03–4.00 | Niacin | 0.1–0.2 |
| Water | 17.1 | Magnesium | 0.7–13.0 | Pantothenic acid | 0.02–0.11 |
| Organic acids | 0.5 | Manganese | 0.02–2.0 | Pyridoxine (B6) | 0.01–0.32 |
| Proteins, amino acids | 0.3 | Phosphorus | 2.0–15.0 | ||
| Sodium | 1.6–17.0 | ||||
| Zinc | 0.05–2.00 | ||||
| Se | 0.001–0.003 | ||||
| Criteria | Values |
|---|---|
| Moisture content (%) | ≤20.0 |
| Fructose and glucose (Sum, g/100 g) | ≥60 |
| Sucrose (g/100 g) | ≤5.0 |
| Water-insoluble content (g/100 g) | <0.1 |
| Electrical conductivity (mS/cm) | ≤0.8 |
| Free acid (meq/kg) | ≤50.0 |
| Diastase activity (Schade scale) | ≥8.0 |
| Hydroxymethylfurfural (HMF, mg/kg) | ≤40.0 |
| Honey Type | Research Model | Main Findings on Honey Effects | Reference(s) |
|---|---|---|---|
| Local honey | Rat kidney, brain, liver and lung homogenates | ↓ Lipid hydroperoxides and malondialdehyde (MDA) value | [81] |
| Christmas vine, Morning glory, black mangrove, linen vine singing bean honey | Rat liver homogenates | Highest radical scavenging capacity in linen vine honey ↓ Lipid peroxidation | [82] |
| Fireweed, tupelo, Hawaiian Christmas berry clover, acacia, buckwheat, soybean honey | Human blood serum | AOC is different among honeys, ↓ Lipoprotein oxidation (LPO) Correlation of ORAC value and LPO inhibition. | [64] |
| Acacia, coriander, sider and palm honey | Human LDL | High antioxidant activity in xanthine-xanthine oxidase system and LDL oxidation | [79] |
| Buckwheat honey | Human blood serum | ↑ Serum antioxidant capacity | [80] |
| Multifloral honey | Human red blood cells (RBC) | ↓ Lipid peroxidation | [83] |
| Multifloral honey | RBC | ↓ Extracellular ferricyanide level | [84] |
| Christmas vine, linen vine honey | RBC | Protection of human erythrocyte membranes from oxidative damage ↑ Defence responses and ↑ cell functions | [82,85,86] |
| Native multifloral honey | Endothelial cell (EA.hy926) | Protection of EA.hy926 from hydrogen peroxide and peroxyl radical Synergistic effect of phenolic antioxidants in honey | [87] |
| Gelam honey | Rat blood sample | ↑ Antioxidant enzyme activities | [88] |
| Multifloral honey | Rat plasma and heart tissue | ↓ Hypertriglyceridemia and pro-oxidative effects ↑ Plasma α-tocopherol and α-tocopherol/triglycerides, ↓ plasma NOx, ↓ peroxidation | [89] |
| Buckwheat honey | Human blood plasma | ↑ Plasma antioxidant activity, ↑ defences against oxidative stress | [90] |
| Honey Type | Research Model | Main Findings of Honey Effect | Reference(s) |
|---|---|---|---|
| Honeydew honey | Rat blood serum | Similar weight gain and body fat in honey and control group; ↓ HbA1c, ↑ HDL-C | [107] |
| Clover honey | Rat blood serum | ↓ Weight gain and adiposity, ↓ TGs but ↑ non-HDL-C levels | [108] |
| Native honey | Rat blood samples | ↓ glucose and lipids no deteriorated effects on hyperglycaemia and dyslipidaemia | [109] |
| Local honey | Rat blood serum | ↑ Plasma TG, HDL-C and VLDL-C but ↓ plasma LDL-C and TC | [110] |
| Tualang honey | Rat heart tissue | ↑ Antioxidant enzyme levels in heart tissue and ↓ lipoprotein oxidation (LPO) | [111] |
| Tualang honey | Rat blood serum, kidneys | ↓ TC and TG compared to the control at 7 days; ↓ Serum creatinine level than no honey group after 48 h; No structural effect histologically in the HCD-fed rats | [112] |
| Gelam, Acacia honey | Rat blood serum, internal organs | ↓ Excess weight gain and adiposity index; ↓ plasma glucose, TGs, TG and obesity at similar levels to orlistat drug group | [113] |
| Malícia honey | Rat blood serum, liver | ↓ Food consumption, ↑ glucose tolerance and SOD activity; ↓ TC, LDL and AST levels; ↑ beneficial bacteria and organic acids; Colon and liver was protected | [114] |
| Natural local honey | Healthy, diabetic and hyperlipidaemic human subjects, blood samples | ↓ Blood lipids, homocysteine and C-reactive protein (CRP) in normal and hyperlipidaemic subjects; ↓ plasma glucose elevation in diabetics | [115] |
| Natural honey | Human plasma | ↓ TC (3.3%), LDL-C (4.3%), TGs (19%) and CRP (3.3%) in elevated variable subjects; No increased body weight in overweight or obese participants | [116] |
| Natural unprocessed honey | Type 2 diabetes human subjects, weight and blood samples | ↓ Body weight, TC, LDL-C, TGs ↑ HDL-C and HbA1C levels | [32] |
| Kanuka honey, formulated with cinnamon, chromium and magnesium | Type 2 diabetes human subject, weight and blood samples | ↓ Weight Improve blood lipid profile | [117] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, H.T.L.; Panyoyai, N.; Kasapis, S.; Pang, E.; Mantri, N. Honey and Its Role in Relieving Multiple Facets of Atherosclerosis. Nutrients 2019, 11, 167. https://doi.org/10.3390/nu11010167
Nguyen HTL, Panyoyai N, Kasapis S, Pang E, Mantri N. Honey and Its Role in Relieving Multiple Facets of Atherosclerosis. Nutrients. 2019; 11(1):167. https://doi.org/10.3390/nu11010167
Chicago/Turabian StyleNguyen, Huong Thi Lan, Naksit Panyoyai, Stefan Kasapis, Edwin Pang, and Nitin Mantri. 2019. "Honey and Its Role in Relieving Multiple Facets of Atherosclerosis" Nutrients 11, no. 1: 167. https://doi.org/10.3390/nu11010167
APA StyleNguyen, H. T. L., Panyoyai, N., Kasapis, S., Pang, E., & Mantri, N. (2019). Honey and Its Role in Relieving Multiple Facets of Atherosclerosis. Nutrients, 11(1), 167. https://doi.org/10.3390/nu11010167





