Sweet and Umami Taste Perception Differs with Habitual Exercise in Males
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Recruitment and Setting
2.4. Body Composition Measurement and Taste Perception Assessment Day Protocol
2.4.1. Taste Stimuli
2.4.2. Taste Perception Rating
2.4.3. Eating Behaviour and Habitual Diet
2.5. Statistical Analysis
3. Results
3.1. Subject Characteristics
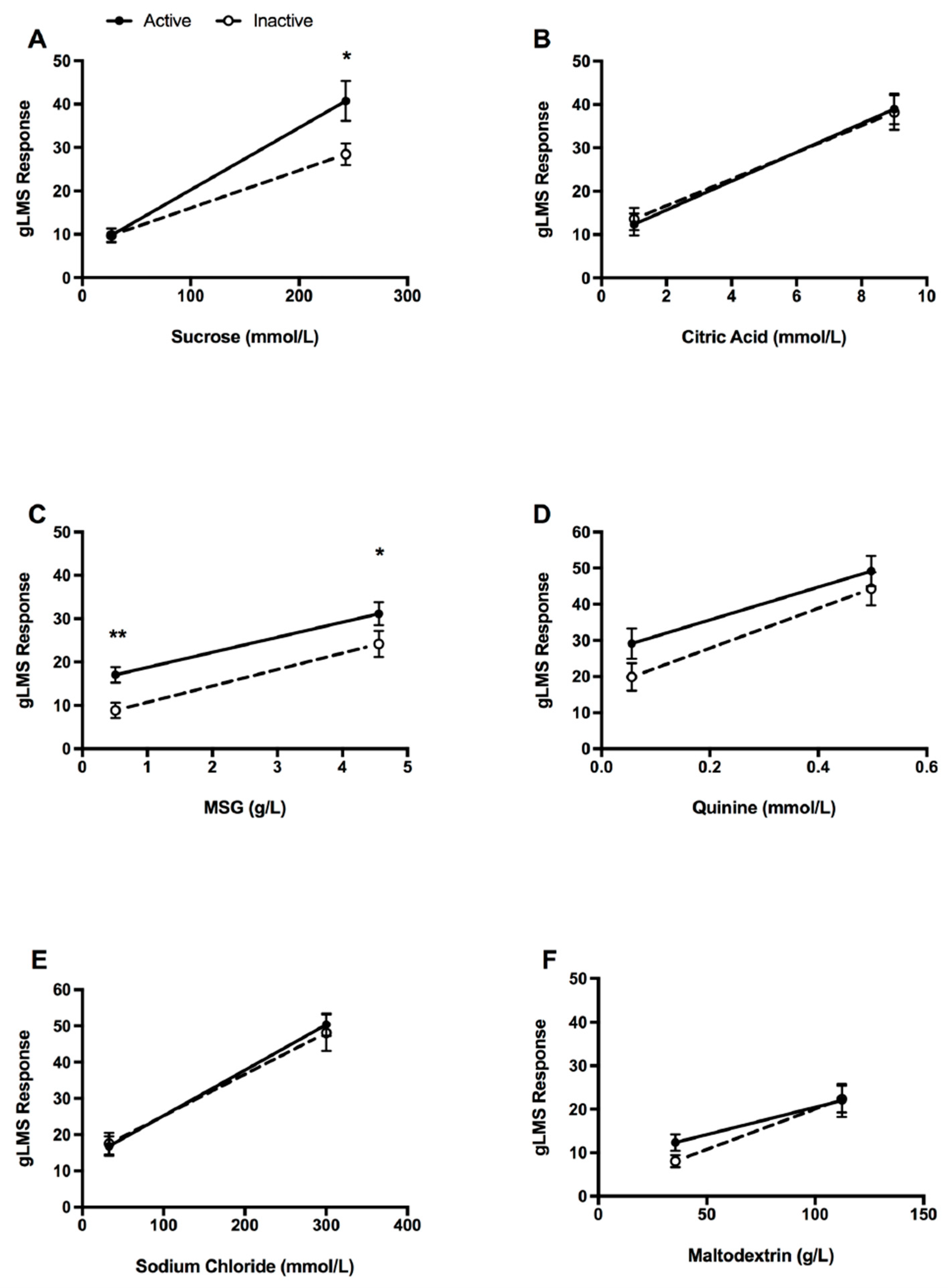
3.2. Taste Identification
3.3. Taste Intensity
3.4. Hedonic Response
3.5. Reproducibility of Taste Intensity Comparisons between Groups at Individual Test Days
3.6. Habitual Dietary Intake
3.7. Regression Analysis Including Age, BMI, and Body Composition
3.7.1. Taste Intensity
3.7.2. Liking
3.8. Regression Analysis with Eating Behaviour
3.8.1. Hunger
3.8.2. Disinhibition
3.8.3. Restraint
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhao, G.Q.; Zhang, Y.; Hoon, M.A.; Chandrashekar, J.; Erlenbach, I. The receptors for mammalian sweet and umami taste. Cell 2003, 115, 255–266. [Google Scholar] [CrossRef]
- Low, J.Y.; Lacy, K.E.; McBride, R.L.; Keast, R.S. Carbohydrate Taste Sensitivity Is Associated with Starch Intake and Waist Circumference in Adults. J. Nutr. 2017, 147, 2235–2242. [Google Scholar] [CrossRef] [PubMed]
- Feeney, E.; O’Brien, S.; Markey, A.; Scannell, A.; Gibney, E. Genetic variation in taste perception—Does it have a role in healthy eating? Proc. Nutr. Soc. 2011, 70, 135–143. [Google Scholar] [CrossRef]
- Hayes, J.E.; Feeney, E.L.; Allen, A.L. Do polymorphisms in chemosensory genes matter for human ingestive behavior? Food Qual. Prefer. 2013, 30, 202–216. [Google Scholar] [CrossRef]
- Chamoun, E.; Mutch, D.M.; Allen-Vercoe, E.; Buchholz, A.C.; Duncan, A.M.; Spriet, L.L.; Haines, J.; Ma, D.W.L.; Guelph Family Health Study. A review of the associations between single nucleotide polymorphisms in taste receptors, eating behaviors, and health. Crit. Rev. Food Sci. Nutr. 2018, 58, 194–207. [Google Scholar] [CrossRef]
- Fukunaga, A.; Uematsu, H.; Sugimoto, K. Influences of aging on taste perception and oral somatic sensation. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Lv, W.; Finlayson, G.; Dando, R. Sleep, food cravings and taste. Appetite 2018, 125, 210–216. [Google Scholar] [CrossRef]
- Bartoshuk, L.M.; Duffy, V.B.; Hayes, J.E.; Moskowitz, H.R.; Snyder, D.J. Psychophysics of sweet and fat perception in obesity: Problems, solutions and new perspectives. Phil. Trans. R. Soc. B Biol. Sci. 2006, 361, 1137–1148. [Google Scholar] [CrossRef]
- Heath, T.P.; Melichar, J.K.; Nutt, D.J.; Donaldson, L.F. Human taste thresholds are modulated by serotonin and noradrenaline. J. Neurosci. 2006, 26, 12664–12671. [Google Scholar] [CrossRef]
- Loper, H.B.; La Sala, M.; Dotson, C.; Steinle, N. Taste perception, associated hormonal modulation, and nutrient intake. Nutr. Rev. 2015, 73, 83–91. [Google Scholar] [CrossRef]
- Jayasinghe, S.N.; Kruger, R.; Walsh, D.C.I.; Cao, G.; Rivers, S.; Richter, M.; Breier, B.H. Is Sweet Taste Perception Associated with Sweet Food Liking and Intake? Nutrients 2017, 9, 750. [Google Scholar] [CrossRef] [PubMed]
- Elder, S.J.; Roberts, S.B. The effects of exercise on food intake and body fatness: A summary of published studies. Nutr. Rev. 2007, 65, 1–19. [Google Scholar] [CrossRef] [PubMed]
- King, N.A.; Horner, K.; Hills, A.P.; Byrne, N.M.; Wood, R.E.; Bryant, E.; Caudwell, P.; Finlayson, G.; Gibbons, C.; Hopkins, M.; et al. Exercise, appetite and weight management: Understanding the compensatory responses in eating behaviour and how they contribute to variability in exercise-induced weight loss. Br. J. Sports Med. 2012, 46, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Crystal, S.; Frye, C.A.; Kanarek, R.B. Taste preferences and sensory perceptions in female varsity swimmers. Appetite 1995, 24, 25–36. [Google Scholar] [CrossRef]
- Hopkins, M.; Blundell, J.E. Energy balance, body composition, sedentariness and appetite regulation: Pathways to obesity. Clin. Sci. (Lond.) 2016, 130, 1615–1628. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, K.; Hopkins, M.; Blundell, J.; Finlayson, G. Homeostatic and non-homeostatic appetite control along the spectrum of physical activity levels: An updated perspective. Physiol. Behav. 2018, 192, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Blundell, J.E. Physical activity and appetite control: Can we close the energy gap? Nutr. Bull. 2011, 36, 356–366. [Google Scholar] [CrossRef]
- Long, S.J.; Hart, K.; Morgan, L.M. The ability of habitual exercise to influence appetite and food intake in response to high- and low-energy preloads in man. Br. J. Nutr. 2002, 87, 517–523. [Google Scholar] [CrossRef]
- Beaulieu, K.; Hopkins, M.; Blundell, J.; Finlayson, G. Does Habitual Physical Activity Increase the Sensitivity of the Appetite Control System? A Systematic Review. Sports Med. 2016, 46, 1897–1919. [Google Scholar] [CrossRef]
- Horner, K.M.; Finlayson, G.; Byrne, N.M.; King, N.A. Food reward in active compared to inactive men: Roles for gastric emptying and body fat. Physiol. Behav. 2016, 160, 43–49. [Google Scholar] [CrossRef]
- Drewnowski, A. Taste preferences and food intake. Annu. Rev. Nutr. 1997, 17, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Feeney, E.L.; O’Brien, S.A.; Scannell, A.G.; Markey, A.; Gibney, E.R. Suprathreshold measures of taste perception in children—Association with dietary quality and body weight. Appetite 2017, 113, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Overberg, J.; Hummel, T.; Krude, H.; Wiegand, S. Differences in taste sensitivity between obese and non-obese children and adolescents. Arch. Dis. Child. 2012, 97, 1048–1052. [Google Scholar] [CrossRef] [PubMed]
- Noel, C.A.; Cassano, P.A.; Dando, R. College-Aged Males Experience Attenuated Sweet and Salty Taste with Modest Weight Gain. J. Nutr. 2017, 147, 1885–1891. [Google Scholar] [CrossRef] [PubMed]
- Birkenhead, K.L.; Slater, G. A Review of Factors Influencing Athletes’ Food Choices. Sports Med. 2015, 45, 1511–1522. [Google Scholar] [CrossRef]
- Beaulieu, K.; Hopkins, M.; Blundell, J.; Finlayson, G. Impact of physical activity level and dietary fat content on passive overconsumption of energy in non-obese adults. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 14. [Google Scholar] [CrossRef]
- Horner, K.M.; Byrne, N.M.; Cleghorn, G.J.; King, N.A. Influence of habitual physical activity on gastric emptying in healthy males and relationships with body composition and energy expenditure. Br. J. Nutr. 2015, 114, 489–496. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Low, J.Y.; Lacy, K.E.; McBride, R.; Keast, R.S. The Association between Sweet Taste Function, Anthropometry, and Dietary Intake in Adults. Nutrients 2016, 8, 241. [Google Scholar] [CrossRef]
- Hayes, J.E.; Allen, A.L.; Bennett, S.M. Direct comparison of the generalized visual analog scale (gVAS) and general labeled magnitude scale (gLMS). Food Qual. Prefer. 2013, 28, 36–44. [Google Scholar] [CrossRef]
- Green, B.G.; Shaffer, G.S.; Gilmore, M.M. Derivation and Evaluation of a Semantic Scale of Oral Sensation Magnitude with Apparent Ratio Properties. Chem. Senses 1993, 18, 683–702. [Google Scholar] [CrossRef]
- Green, B.G.; Dalton, P.; Cowart, B.; Shaffer, G.; Rankin, K. Evaluating the Labeled Magnitude Scale for measuring sensations of taste and smell. Chem. Senses 1996, 21, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.; Urban, L.; Green, B.G. Measures of Individual Differences in Taste and Creaminess Perception. Chem. Senses 2008, 33, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Stunkard, A.J.; Messick, S. The three-factor eating questionnaire to measure dietary restraint, disinhibition and hunger. J. Psychosom. Res. 1985, 29, 71–83. [Google Scholar] [CrossRef]
- Kroke, A.; Klipstein-Grobusch, K.; Voss, S.; Moseneder, J.; Thielecke, F.; Noack, R.; Boeing, H. Validation of a self-administered food-frequency questionnaire administered in the European Prospective Investigation into Cancer and Nutrition (EPIC) Study: Comparison of energy, protein, and macronutrient intakes estimated with the doubly labeled water, urinary nitrogen, and repeated 24-h dietary recall methods. Am. J. Clin. Nutr. 1999, 70, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Westerterp-Plantenga, M.S.; Verwegen, C.R.; Ijedema, M.J.; Wijckmans, N.E.; Saris, W.H. Acute effects of exercise or sauna on appetite in obese and nonobese men. Physiol. Behav. 1997, 62, 1345–1354. [Google Scholar] [CrossRef]
- Nakagawa, M.; Mizuma, K.; Inui, T. Changes in taste perception following mental or physical stress. Chem. Senses 1996, 21, 195–200. [Google Scholar] [CrossRef]
- Horio, T.; Kawamura, Y. Influence of physical exercise on human preferences for various taste solutions. Chem. Senses 1998, 23, 417–421. [Google Scholar] [CrossRef]
- Cicerale, S.; Riddell, L.J.; Keast, R.S. The association between perceived sweetness intensity and dietary intake in young adults. J. Food Sci. 2012, 77, H31–H35. [Google Scholar] [CrossRef]
- Tepper, B.J.; Ullrich, N.V. Influence of genetic taste sensitivity to 6-n-propylthiouracil (PROP), dietary restraint and disinhibition on body mass index in middle-aged women. Physiol. Behav. 2002, 75, 305–312. [Google Scholar] [CrossRef]
- Li, X.; Staszewski, L.; Xu, H.; Durick, K.; Zoller, M.; Adler, E. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 2002, 99, 4692–4696. [Google Scholar] [CrossRef]
- Martin, B.; Dotson, C.D.; Shin, Y.K.; Ji, S.; Drucker, D.J.; Maudsley, S.; Munger, S.D. Modulation of taste sensitivity by GLP-1 signaling in taste buds. Ann. N. Y. Acad. Sci. 2009, 1170, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.K.; Martin, B.; Golden, E.; Dotson, C.D.; Maudsley, S.; Kim, W.; Jang, H.J.; Mattson, M.P.; Drucker, D.J.; Egan, J.M.; et al. Modulation of taste sensitivity by GLP-1 signaling. J. Neurochem. 2008, 106, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Sanematsu, K.; Ohta, R.; Shirosaki, S.; Koyano, K.; Nonaka, K.; Shigemura, N.; Ninomiya, Y. Diurnal variation of human sweet taste recognition thresholds is correlated with plasma leptin levels. Diabetes 2008, 57, 2661–2665. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, R.; Ohkuri, T.; Jyotaki, M.; Yasuo, T.; Horio, N.; Yasumatsu, K.; Sanematsu, K.; Shigemura, N.; Yamamoto, T.; Margolskee, R.F. Endocannabinoids selectively enhance sweet taste. Proc. Natl. Acad. Sci. USA 2010, 107, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.; Costa, G.; Cordeiro, C.; Pinheiro, C.; Amado, F.; Lamy, E. Salivary proteome and glucose levels are related with sweet taste sensitivity in young adults. Food Nutr. Res. 2017, 61. [Google Scholar] [CrossRef]
- Tantimonaco, M.; Ceci, R.; Sabatini, S.; Catani, M.V.; Rossi, A.; Gasperi, V.; Maccarrone, M. Physical activity and the endocannabinoid system: An overview. Cell. Mol. Life Sci. 2014, 71, 2681–2698. [Google Scholar] [CrossRef]
- Yeomans, M.R. Taste, palatability and the control of appetite. Proc. Nutr. Soc. 1998, 57, 609–615. [Google Scholar] [CrossRef]
- Perry, D.M.; Hayes, J.E. Effects of Matrix Composition on Detection Threshold Estimates for Methyl Anthranilate and 2-Aminoacetophenone. Foods 2016, 5, 35. [Google Scholar] [CrossRef]
- Sartor, F.; Donaldson, L.F.; Markland, D.A.; Loveday, H.; Jackson, M.J.; Kubis, H.P. Taste perception and implicit attitude toward sweet related to body mass index and soft drink supplementation. Appetite 2011, 57, 237–246. [Google Scholar] [CrossRef]
- Fernandez-Garcia, J.C.; Alcaide, J.; Santiago-Fernandez, C.; Roca-Rodriguez, M.M.; Aguera, Z.; Banos, R.; Botella, C.; de la Torre, R.; Fernandez-Real, J.M.; Fruhbeck, G.; et al. An increase in visceral fat is associated with a decrease in the taste and olfactory capacity. PLoS ONE 2017, 12, e0171204. [Google Scholar] [CrossRef]
- Pietrobelli, A.; Rubiano, F.; St-Onge, M.P.; Heymsfield, S.B. New bioimpedance analysis system: Improved phenotyping with whole-body analysis. Eur. J. Clin. Nutr. 2004, 58, 1479–1484. [Google Scholar] [CrossRef] [PubMed]
- Volgyi, E.; Tylavsky, F.A.; Lyytikainen, A.; Suominen, H.; Alen, M.; Cheng, S. Assessing body composition with DXA and bioimpedance: Effects of obesity, physical activity, and age. Obesity (Silver Spring) 2008, 16, 700–705. [Google Scholar] [CrossRef] [PubMed]
| Active | Inactive | p-Value | Effect Size (d) | |
|---|---|---|---|---|
| Age (years) | ||||
| Median (IQR) | 21 (21.0–22.5) | 21 (20.5–25.5) | 0.79 | r = 0.05 1 |
| Height (cm) | ||||
| Mean (SD) | 179.83 (4.91) | 181.09 (5.12) | 0.50 | 0.25 |
| Weight (kg) | ||||
| Mean (SD) | 79.03 (8.02) | 81.49 (10.96) | 0.49 | 0.26 |
| BMI (kg/m2) | ||||
| Mean (SD) | 24.41 (1.93) | 24.80 (3.14) | 0.68 | 0.15 |
| Body Fat (%) | ||||
| Mean (SD) | 12.33 (4.30) | 18.87 (6.45) | <0.01 | 1.19 |
| Active Mean (SD) | Inactive Mean (SD) | p-Value | Effect Size (d) | |
|---|---|---|---|---|
| Sucrose | ||||
| High | 38.91 (27.68) | 34.75 (28.66) | 0.69 | 0.15 |
| Low | 1.44 (10.98) | 5.18 (21.85) | 0.42 | 0.22 |
| MSG2 | ||||
| High | −17.06 (31.66) | −16.79 (29.36) | 0.98 | <0.01 |
| Low | −13.09 (20.31) | 2.82 (11.50) | <0.01 | 0.96 |
| Citric acid | ||||
| High | −3.72 (34.10) | 0.36 (33.31) | 0.74 | 0.12 |
| Low | −5.34 (18.19) | 2.07 (22.59) | 0.33 | 0.36 |
| Quinine | ||||
| High | −57.50 (20.97) | −38.79 (28.06) | 0.05 | 0.76 |
| Low | −34.00 (22.86) | −17.89 (29.02) | 0.08 | 0.62 |
| Sodium chloride | ||||
| High | −33.16 (43.22) | −29.57 (35.17) | 0.50 | 0.09 |
| Low | −18.72 (16.21) | −11.96 (23.87) | 0.73 | 0.33 |
| Maltodextrin | ||||
| High | −7.16 (25.5) | −3.32 (22.31) | 0.67 | 0.16 |
| Low | −12.28 (12.76) | 0.21 (14.29) | <0.01 | 0.93 |
| Active (n = 13) 1 Mean (SD) | Inactive (n = 13) 1 Mean (SD) | p-Value (2-Tailed) | |
|---|---|---|---|
| Energy Intake kcal/day | 2290.7 (841.6) | 2018.7 (826.7) | 0.41 |
| Macronutrient Intake, % of energy | |||
| Fat | 36.4 (3.7) | 37.7 (6.6) | 0.53 |
| Protein | 21.5 (3.6) | 19.9 (2.8) | 0.22 |
| Carbohydrate | 43.8 (4.6) | 42.0 (8.8) | 0.50 |
| Sugar | 18.1 (3.5) | 18.7 (4.6) | 0.73 |
| Sucrose | 6.5 (2.3) | 6.7 (1.9) | 0.78 |
| Fructose | 2.5 (1.0) | 3.4 (1.2) | 0.04 |
| Galactose | 0.1 (0.1) | 0.1 (0.1) | 0.59 |
| Maltose | 0.5 (0.1) | 0.5 (0.3) | 0.77 |
| Lactose | 5.6 (2.0) | 4.1 (2.7) | 0.11 |
| Starch | 25.0 (3.8) | 22.5 (6.9) | 0.25 |
| Fibre | 2.9 (0.8) | 3.6 (1.0) | 0.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feeney, E.L.; Leacy, L.; O’Kelly, M.; Leacy, N.; Phelan, A.; Crowley, L.; Stynes, E.; de Casanove, A.; Horner, K. Sweet and Umami Taste Perception Differs with Habitual Exercise in Males. Nutrients 2019, 11, 155. https://doi.org/10.3390/nu11010155
Feeney EL, Leacy L, O’Kelly M, Leacy N, Phelan A, Crowley L, Stynes E, de Casanove A, Horner K. Sweet and Umami Taste Perception Differs with Habitual Exercise in Males. Nutrients. 2019; 11(1):155. https://doi.org/10.3390/nu11010155
Chicago/Turabian StyleFeeney, Emma L., Laura Leacy, Mark O’Kelly, Niamh Leacy, Abbie Phelan, Leah Crowley, Emily Stynes, Aude de Casanove, and Katy Horner. 2019. "Sweet and Umami Taste Perception Differs with Habitual Exercise in Males" Nutrients 11, no. 1: 155. https://doi.org/10.3390/nu11010155
APA StyleFeeney, E. L., Leacy, L., O’Kelly, M., Leacy, N., Phelan, A., Crowley, L., Stynes, E., de Casanove, A., & Horner, K. (2019). Sweet and Umami Taste Perception Differs with Habitual Exercise in Males. Nutrients, 11(1), 155. https://doi.org/10.3390/nu11010155





