The Glycaemic Index-Food-Frequency Questionnaire: Development and Validation of a Food Frequency Questionnaire Designed to Estimate the Dietary Intake of Glycaemic Index and Glycaemic Load: An Effort by the PREVIEW Consortium
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Population for Analyses
2.3. Glycaemic Index FFQ
2.4. Food-Frequency Questionnaire
2.5. Phone-Based 24 h-Recalls
2.6. Blood Sampling
2.7. Additional Measurements
2.8. Statistical Analysis
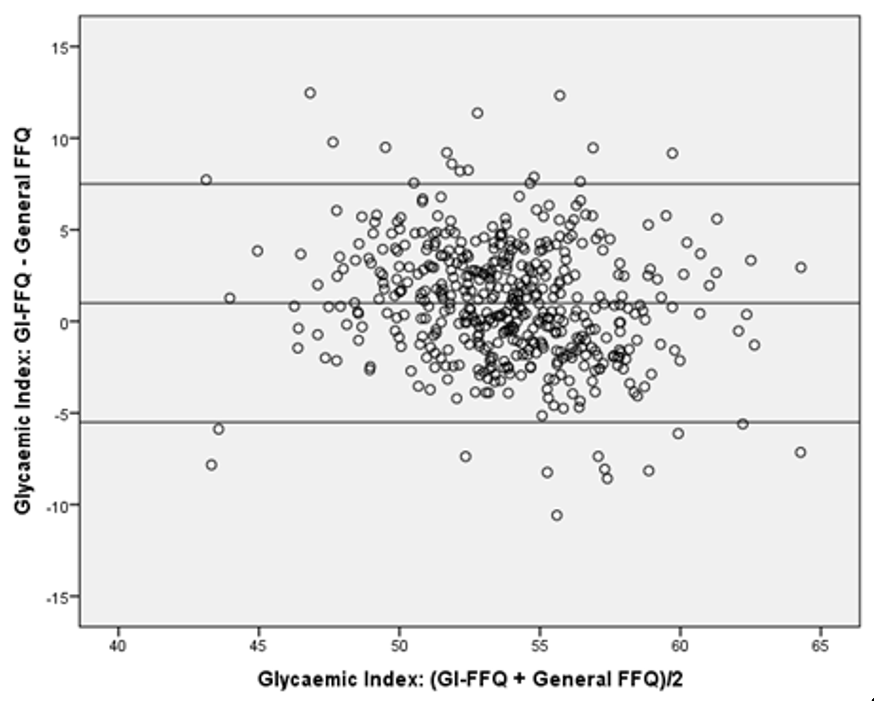
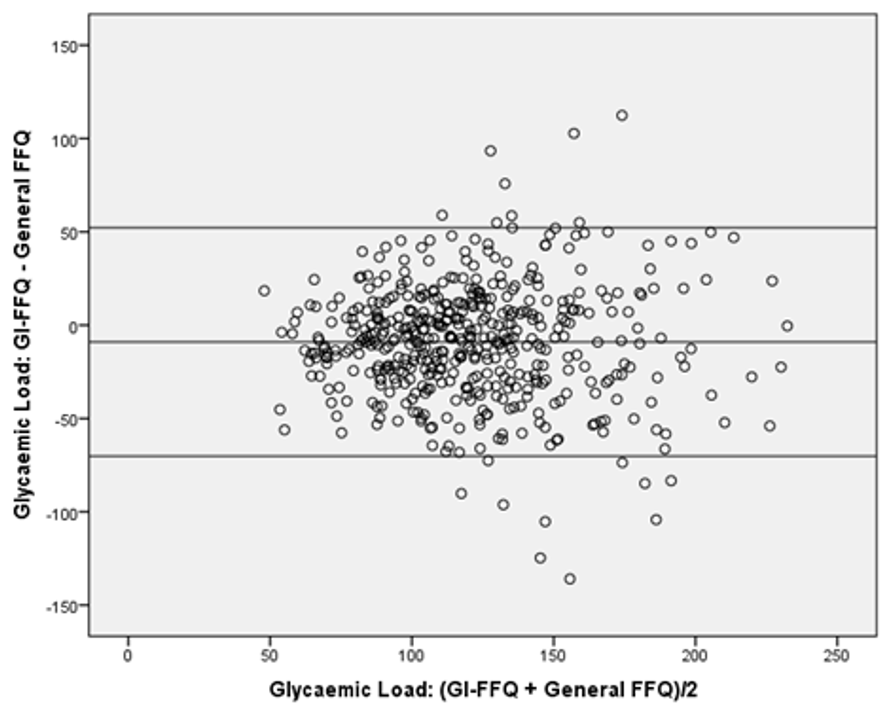
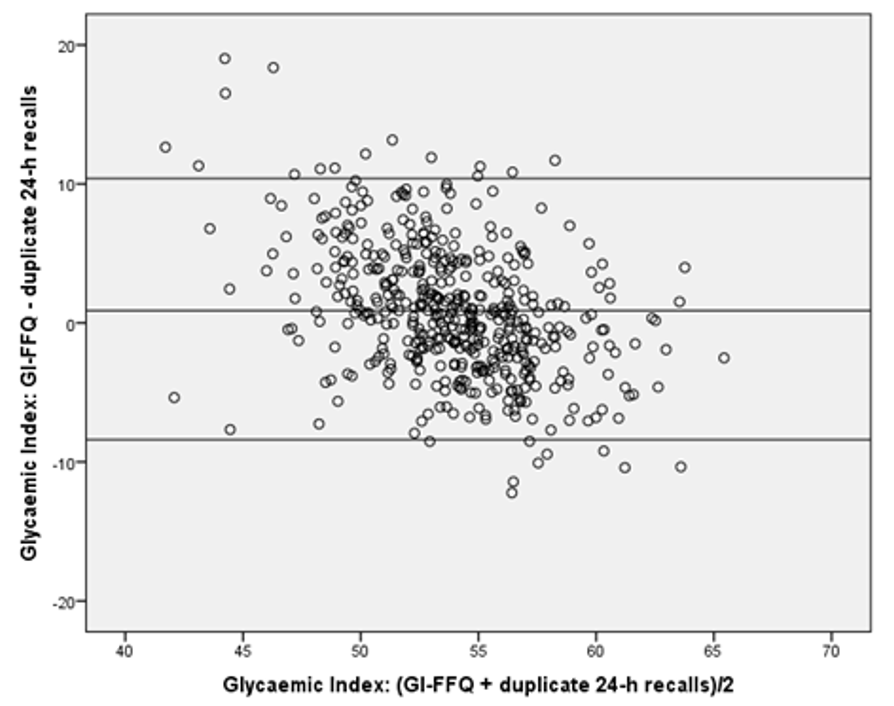
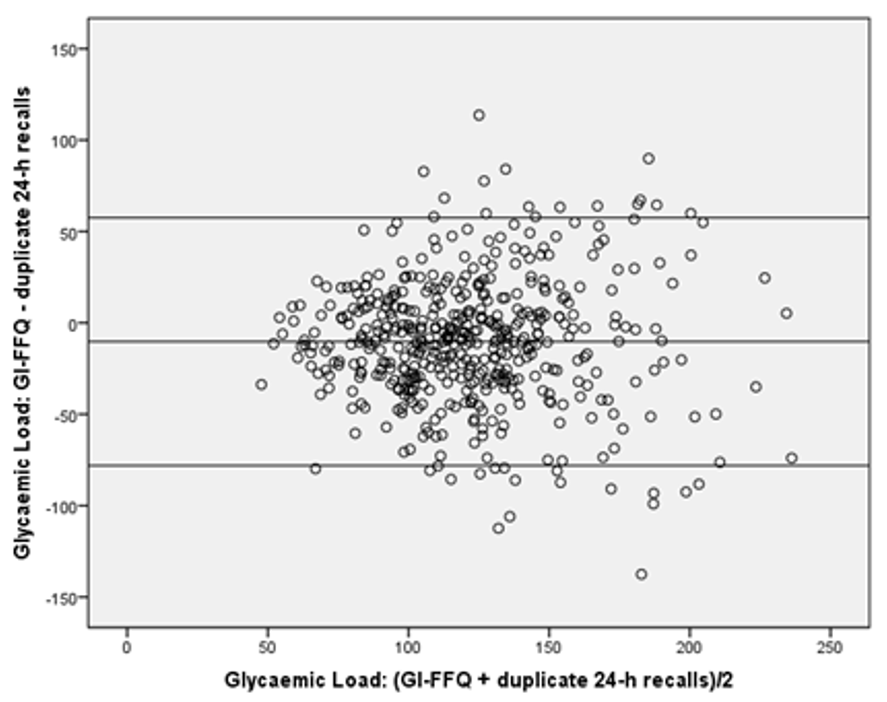
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barclay, A.W.; Petocz, P.; McMillan-Price, J.; Flood, V.M.; Prvan, T.; Mitchell, P.; Brand-Miller, J.C. Glycemic index, glycemic load, and chronic disease risk—A meta-analysis of observational studies. Am. J. Clin. Nutr. 2008, 87, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, D.C.; Threapleton, D.E.; Evans, C.E.; Cleghorn, C.L.; Nykjaer, C.; Woodhead, C.; Burley, V.J. Glycemic index, glycemic load, carbohydrates, and type 2 diabetes: Systematic review and dose-response meta-analysis of prospective studies. Diabetes Care 2013, 36, 4166–4171. [Google Scholar] [CrossRef] [PubMed]
- Rahimlou, M.; Morshedzadeh, N.; Karimi, S.; Jafarirad, S. Association between dietary glycemic index and glycemic load with depression: A systematic review. Eur. J. Nutr. 2018, 57, 2333–2340. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, S.; Chan, D.S.M.; Vingeliene, S.; Vieira, A.R.; Abar, L.; Polemiti, E.; Stevens, C.A.T.; Greenwood, D.C.; Aune, D.; Norat, T. Carbohydrates, glycemic index, glycemic load, and breast cancer risk: A systematic review and dose-response meta-analysis of prospective studies. Nutr. Rev. 2017, 75, 420–441. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Wu, Y.; Xu, J.; Ding, K.; Shan, X.; Xia, D. Association between dietary carbohydrate intake, glycemic index and glycemic load, and risk of gastric cancer. Eur. J. Nutr. 2017, 56, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Song, Y.; Wang, Y.; Hui, R.; Zhang, W. Dietary glycemic index, glycemic load, and risk of coronary heart disease, stroke, and stroke mortality: A systematic review with meta-analysis. PLoS ONE 2012, 7, e52182. [Google Scholar] [CrossRef] [PubMed]
- Gnagnarella, P.; Gandini, S.; La Vecchia, C.; Maisonneuve, P. Glycemic index, glycemic load, and cancer risk: A meta-analysis. Am. J. Clin. Nutr. 2008, 87, 1793–1801. [Google Scholar] [CrossRef]
- Livesey, G.; Taylor, R.; Livesey, H.; Liu, S. Is there a dose-response relation of dietary glycemic load to risk of type 2 diabetes? Meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 2013, 97, 584–596. [Google Scholar] [CrossRef]
- Barclay, A.W.; Flood, V.M.; Brand-Miller, J.C.; Mitchell, P. Validity of carbohydrate, glycaemic index and glycaemic load data obtained using a semi-quantitative food-frequency questionnaire. Public Health Nutr. 2008, 11, 573–580. [Google Scholar] [CrossRef]
- Du, H.; van der, A.D.; van Bakel, M.M.; Verberne, L.D.; Ocke, M.; Feskens, E.J. Reproducibility and relative validity of dietary glycaemic index and glycaemic load assessed by the food-frequency questionnaire used in the dutch cohorts of the european prospective investigation into cancer and nutrition. Br. J. Nutr. 2009, 102, 601–604. [Google Scholar] [CrossRef]
- Kaartinen, N.E.; Tapanainen, H.; Valsta, L.M.; Simila, M.E.; Reinivuo, H.; Korhonen, T.; Harald, K.; Eriksson, J.G.; Peltonen, M.; Mannisto, S. Relative validity of a ffq in measuring carbohydrate fractions, dietary glycaemic index and load: Exploring the effects of subject characteristics. Br. J. Nutr. 2012, 107, 1367–1375. [Google Scholar] [CrossRef] [PubMed]
- Levitan, E.B.; Westgren, C.W.; Liu, S.; Wolk, A. Reproducibility and validity of dietary glycemic index, dietary glycemic load, and total carbohydrate intake in 141 swedish men. Am. J. Clin. Nutr. 2007, 85, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Sasaki, S.; Takahashi, Y.; Okubo, H.; Hirota, N.; Notsu, A.; Fukui, M.; Date, C. Reproducibility and relative validity of dietary glycaemic index and load assessed with a self-administered diet-history questionnaire in japanese adults. Br. J. Nutr. 2008, 99, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Van Bakel, M.M.; Slimani, N.; Feskens, E.J.; Du, H.; Beulens, J.W.; van der Schouw, Y.T.; Brighenti, F.; Halkjaer, J.; Cust, A.E.; Ferrari, P.; et al. Methodological challenges in the application of the glycemic index in epidemiological studies using data from the european prospective investigation into cancer and nutrition. J. Nutr. 2009, 139, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Brouwer-Brolsma, E.M.; Brennan, L.; Drevon, C.A.; van Kranen, H.; Manach, C.; Dragsted, L.O.; Roche, H.M.; Andres-Lacueva, C.; Bakker, S.J.L.; Bouwman, J.; et al. Combining traditional dietary assessment methods with novel metabolomics techniques: Present efforts by the food biomarker alliance. Proc. Nutr. Soc. 2017, 76, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Vega-Lopez, S.; Venn, B.J.; Slavin, J.L. Relevance of the glycemic index and glycemic load for body weight, diabetes, and cardiovascular disease. Nutrients 2018, 10, 1361. [Google Scholar] [CrossRef] [PubMed]
- Farvid, M.S.; Homayouni, F.; Shokoohi, M.; Fallah, A.; Farvid, M.S. Glycemic index, glycemic load and their association with glycemic control among patients with type 2 diabetes. Eur. J. Clin. Nutr. 2014, 68, 459–463. [Google Scholar] [CrossRef]
- Hosseinpour-Niazi, S.; Sohrab, G.; Asghari, G.; Mirmiran, P.; Moslehi, N.; Azizi, F. Dietary glycemic index, glycemic load, and cardiovascular disease risk factors: Tehran lipid and glucose study. Arch. Iran Med. 2013, 16, 401–407. [Google Scholar]
- Murakami, K.; Sasaki, S.; Takahashi, Y.; Okubo, H.; Hosoi, Y.; Horiguchi, H.; Oguma, E.; Kayama, F. Dietary glycemic index and load in relation to metabolic risk factors in japanese female farmers with traditional dietary habits. Am. J. Clin. Nutr. 2006, 83, 1161–1169. [Google Scholar] [CrossRef]
- Van Aerde, M.A.; Witte, D.R.; Jeppesen, C.; Soedamah-Muthu, S.S.; Bjerregaard, P.; Jorgensen, M.E. Glycemic index and glycemic load in relation to glucose intolerance among greenland’s inuit population. Diabetes Res. Clin. Pract. 2012, 97, 298–305. [Google Scholar] [CrossRef]
- Kohnert, K.D.; Heinke, P.; Vogt, L.; Salzsieder, E. Utility of different glycemic control metrics for optimizing management of diabetes. World J. Diabetes 2015, 6, 17–29. [Google Scholar] [CrossRef]
- Mayer-Davis, E.J.; Dhawan, A.; Liese, A.D.; Teff, K.; Schulz, M. Towards understanding of glycaemic index and glycaemic load in habitual diet: Associations with measures of glycaemia in the insulin resistance atherosclerosis study. Br. J. Nutr. 2006, 95, 397–405. [Google Scholar] [CrossRef] [PubMed]
- McKeown, N.M.; Meigs, J.B.; Liu, S.; Rogers, G.; Yoshida, M.; Saltzman, E.; Jacques, P.F. Dietary carbohydrates and cardiovascular disease risk factors in the framingham offspring cohort. J. Am. Coll. Nutr. 2009, 28, 150–158. [Google Scholar] [CrossRef]
- Shikany, J.M.; Tinker, L.F.; Neuhouser, M.L.; Ma, Y.; Patterson, R.E.; Phillips, L.S.; Liu, S.; Redden, D.T. Association of glycemic load with cardiovascular disease risk factors: The women’s health initiative observational study. Nutrition 2010, 26, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.L.; Gellar, L.; Nathanson, B.H.; Pbert, L.; Ma, Y.; Ockene, I.; Rosal, M.C. Decrease in glycemic index associated with improved glycemic control among latinos with type 2 diabetes. J. Acad. Nutr. Diet. 2015, 115, 898–906. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; Streppel, M.T.; van Lee, L.; Geelen, A.; Sluik, D.; van de Wiel, A.M.; de Vries, J.H.M.; van’t Veer, P.; Feskens, E.J.M. A national dietary assessment reference database (ndard) for the dutch population: Rationale behind the design. Nutrients 2017, 9, 1136. [Google Scholar] [CrossRef] [PubMed]
- Feunekes, G.I.; Van Staveren, W.A.; De Vries, J.H.; Burema, J.; Hautvast, J.G. Relative and biomarker-based validity of a food-frequency questionnaire estimating intake of fats and cholesterol. Am. J. Clin. Nutr. 1993, 58, 489–496. [Google Scholar] [CrossRef]
- Siebelink, E.; Geelen, A.; de Vries, J.H. Self-reported energy intake by ffq compared with actual energy intake to maintain body weight in 516 adults. Br. J. Nutr. 2011, 106, 274–281. [Google Scholar] [CrossRef]
- Streppel, M.T.; de Vries, J.H.; Meijboom, S.; Beekman, M.; de Craen, A.J.; Slagboom, P.E.; Feskens, E.J. Relative validity of the food frequency questionnaire used to assess dietary intake in the leiden longevity study. Nutr. J. 2013, 12, 75. [Google Scholar] [CrossRef]
- Wolever, T.M.; Mehling, C. Long-term effect of varying the source or amount of dietary carbohydrate on postprandial plasma glucose, insulin, triacylglycerol, and free fatty acid concentrations in subjects with impaired glucose tolerance. Am. J. Clin. Nutr. 2003, 77, 612–621. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; van Lee, L.; Streppel, M.T.; Sluik, D.; van de Wiel, A.M.; de Vries, J.H.M.; Geelen, A.; Feskens, E.J.M. Nutrition questionnaires plus (nqplus) study, a prospective study on dietary determinants and cardiometabolic health in dutch adults. BMJ Open 2018, 8, e020228. [Google Scholar] [CrossRef] [PubMed]
- Rhee, J.J.; Sampson, L.; Cho, E.; Hughes, M.D.; Hu, F.B.; Willett, W.C. Comparison of methods to account for implausible reporting of energy intake in epidemiologic studies. Am. J. Epidemiol. 2015, 181, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Sluik, D.; Atkinson, F.S.; Brand-Miller, J.C.; Fogelholm, M.; Raben, A.; Feskens, E.J. Contributors to dietary glycaemic index and glycaemic load in the netherlands: The role of beer. Br. J. Nutr. 2016, 115, 1218–1225. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; van der, A.D.; van Bakel, M.M.; van der Kallen, C.J.; Blaak, E.E.; van Greevenbroek, M.M.; Jansen, E.H.; Nijpels, G.; Stehouwer, C.D.; Dekker, J.M.; et al. Glycemic index and glycemic load in relation to food and nutrient intake and metabolic risk factors in a dutch population. Am. J. Clin. Nutr. 2008, 87, 655–661. [Google Scholar] [CrossRef] [PubMed]
- The Dutch Nutrition Centre. Zo eet Nederland: Resultaten van de Voedselconsumptiepeiling 1997–1998 (Results of the Dutch Food Consumption Survey 1997/1998); Voedingscentrum: Den Haag, The Netherlands, 1998. (In Dutch) [Google Scholar]
- The Dutch National Institute for Public Health and the Environment (RIVM). Nevo-Tabel. Nederlands Voedingsstoffenbestand 2011; Voedingscentrum: Den Haag, The Netherlands, 2011. [Google Scholar]
- Blanton, C.A.; Moshfegh, A.J.; Baer, D.J.; Kretsch, M.J. The usda automated multiple-pass method accurately estimates group total energy and nutrient intake. J. Nutr. 2006, 136, 2594–2599. [Google Scholar] [CrossRef] [PubMed]
- Conway, J.M.; Ingwersen, L.A.; Moshfegh, A.J. Accuracy of dietary recall using the usda five-step multiple-pass method in men: An observational validation study. J. Am. Diet. Assoc. 2004, 104, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Conway, J.M.; Ingwersen, L.A.; Vinyard, B.T.; Moshfegh, A.J. Effectiveness of the us department of agriculture 5-step multiple-pass method in assessing food intake in obese and nonobese women. Am. J. Clin. Nutr. 2003, 77, 1171–1178. [Google Scholar] [CrossRef]
- Moshfegh, A.J.; Rhodes, D.G.; Baer, D.J.; Murayi, T.; Clemens, J.C.; Rumpler, W.V.; Paul, D.R.; Sebastian, R.S.; Kuczynski, K.J.; Ingwersen, L.A.; et al. The us department of agriculture automated multiple-pass method reduces bias in the collection of energy intakes. Am. J. Clin. Nutr. 2008, 88, 324–332. [Google Scholar] [CrossRef]
- Scholtens, S.; Smidt, N.; Swertz, M.A.; Bakker, S.J.; Dotinga, A.; Vonk, J.M.; van Dijk, F.; van Zon, S.K.; Wijmenga, C.; Wolffenbuttel, B.H.; et al. Cohort profile: Lifelines, a three-generation cohort study and biobank. Int. J. Epidemiol. 2015, 44, 1172–1180. [Google Scholar] [CrossRef]
- Willett, W.C. Nutritional Epidemiology, 3rd ed.; Oxford University Press: New York, NY, USA, 2013. [Google Scholar]
- Lombard, M.J.; Steyn, N.P.; Charlton, K.E.; Senekal, M. Application and interpretation of multiple statistical tests to evaluate validity of dietary intake assessment methods. Nutr. J. 2015, 14, 40. [Google Scholar] [CrossRef]
- Sluik, D.; Geelen, A.; de Vries, J.H.; Eussen, S.J.; Brants, H.A.; Meijboom, S.; van Dongen, M.C.; Bueno-de-Mesquita, H.B.; Wijckmans-Duysens, N.E.; van’t Veer, P.; et al. A national ffq for the netherlands (the ffq-nl 1.0): Validation of a comprehensive ffq for adults. Br. J. Nutr. 2016, 116, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.; Elliott, E.J. Low glycaemic index, or low glycaemic load, diets for diabetes mellitus. Cochrane Database Syst. Rev. 2009, Cd006296. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.; Moller, B.K.; Raben, A.; Tetens, I.; Holst, J.J.; Astrup, A. The use of glycaemic index tables to predict glycaemic index of breakfast meals. Br. J. Nutr. 2005, 94, 135–136. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.; Yang, M.; Zeng, X.Y.; Atkinson, F.; Brand-Miller, J.C. Food glycemic index, as given in glycemic index tables, is a significant determinant of glycemic responses elicited by composite breakfast meals. Am. J. Clin. Nutr. 2006, 83, 1306–1312. [Google Scholar] [CrossRef] [PubMed]
- Cade, J.; Thompson, R.; Burley, V.; Warm, D. Development, validation and utilisation of food-frequency questionnaires—A review. Public Health Nutr. 2002, 5, 567–587. [Google Scholar] [CrossRef] [PubMed]
| All (n = 475) | Men (n = 263) | Women (n = 212) | |
|---|---|---|---|
| Age, years | 55 ± 11 | 57 ± 10 | 53 ± 11 |
| Men, n (%) | 263 (55) | 263 (100) | 0 (0) |
| BMI, kg/m2 | 25.5 ± 3.7 | 25.9 ± 3.4 | 25.2 ± 4.0 |
| BMI-category | |||
| <25 kg/m2 | 231 (49) | 114 (43) | 117 (55) |
| ≥25 kg/m2 | 244 (51) | 149 (57) | 95 (45) |
| Waist circumference, cm | 90.7 ± 11.9 | 95.1 ± 10.4 | 85.2 ± 11.3 |
| Education, n (%) | |||
| Low | 28 (6) | 16 (6) | 12 (6) |
| Medium | 132 (28) | 65 (25) | 67 (31) |
| High | 314 (66) | 182 (69) | 132 (63) |
| Smoking status, n (%) | |||
| Never | 231 (52) | 114 (46) | 117 (60) |
| Former | 179 (40) | 111 (45) | 68 (35) |
| Current | 35 (8) | 24 (9) | 11 (5) |
| Diabetes, n (%) | 12 (3) | 8 (3) | 4 (2) |
| Diet during past month, n (%) | 31 (7) | 14 (5) | 17 (8) |
| GI-FFQ | General-FFQ | 2-day 24 h-Recall | |||||
|---|---|---|---|---|---|---|---|
| GI 1 | Mean | SD | Mean | SD | Mean | SD | |
| Energy, kJ/day | - | 5854 | 1725 | 9054 | 2113 | 9084 | 2028 |
| Carbohydrates, g/day | - | 214 | 65 | 234 | 63 | 237 | 64 |
| Mono/disaccharides, g/day | - | 100 | 39 | 103 | 33 | 107 | 38 |
| Polysaccharides, g/day | - | 114 | 36 | 132 | 41 | 130 | 39 |
| Fibres, g/day | - | 20 | 6 | 25 | 7 | 24 | 7 |
| Bread, g/day | 63 | 127 | 60 | 137 | 58 | 149 | 69 |
| Breakfast cereals, g/day | 54 | 8 | 14 | 14 | 35 | 11 | 19 |
| Potatoes, g/day | 85 | 62 | 39 | 82 | 57 | 103 | 98 |
| Pasta, g/day | 46 | 27 | 22 | 26 | 24 | 49 | 99 |
| Rice, g/day | 72 | 24 | 22 | 31 | 29 | 36 | 79 |
| Vegetables, g/day | 20 | 110 | 71 | 156 | 83 | 166 | 104 |
| Fruit, g/day | 47 | 207 | 126 | 193 | 126 | 203 | 136 |
| Dairy, g/day | 33 | 331 | 210 | 352 | 182 | 354 | 186 |
| Soup, g/day | 42 | 44 | 43 | 44 | 53 | 98 | 150 |
| Coffee, g/day | 62 | 486 | 287 | 466 | 312 | 465 | 293 |
| SSB, g/day | 62 | 52 | 115 | 25 | 61 | 89 | 178 |
| ASB, g/day | 11 | 25 | 97 | 22 | 67 | 43 | 134 |
| Fruit juices, g/day | 54 | 89 | 121 | 61 | 75 | 106 | 160 |
| Alcoholic beverages, g/day | 89 | 75 | 145 | 154 | 196 | 194 | 247 |
| Savoury snacks, g/day | 57 | 44 | 41 | 39 | 37 | 22 | 41 |
| Cake/cookies, g/day | 61 | 38 | 28 | 36 | 27 | 52 | 43 |
| Sweets, g/day | 52 | 33 | 28 | 32 | 24 | 33 | 25 |
| GI | - | 54 | 3 | 53 | 4 | 53 | 5 |
| GL | - | 117 | 37 | 126 | 38 | 127 | 37 |
| GI-FFQ with General-FFQ | GI-FFQ with 2-day 24 h-Recalls | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Similar Q or T | Crude | Energy-Adjusted | Similar Q or T | Crude | ICC | De-Attenuated | Energy-Adjusted | Energy Adjusted De-Attenuated | |
| % | r | r | % | r | r | r | r | ||
| Energy, kJ/day | 46 | 0.58 | - | 39 | 0.43 | 0.52 | 0.60 | - | |
| Carbohydrates, g/day | 48 | 0.64 | 0.34 | 40 | 0.53 | 0.63 | 0.67 | 0.27 | 0.34 |
| Mono/disaccharides, g/day | 45 | 0.60 | 0.45 | 41 | 0.48 | 0.63 | 0.60 | 0.41 | 0.52 |
| Polysaccharides, g/day | 48 | 0.61 | 0.31 | 44 | 0.57 | 0.58 | 0.75 | 0.35 | 0.46 |
| Fibres, g/day | 44 | 0.57 | 0.60 | 41 | 0.52 | 0.67 | 0.64 | 0.48 | 0.59 |
| Bread, g/day | 54 | 0.71 | 0.61 | 46 | 0.61 | 0.67 | 0.75 | 0.49 | 0.60 |
| Breakfast cereals, g/day | 62 | 0.41 | 0.46 | 62 | 0.43 | 0.70 | 0.51 | 0.42 | 0.50 |
| Potatoes, g/day | 43 | 0.55 | 0.49 | 33 | 0.27 | 0.29 | 0.50 | 0.25 | 0.46 |
| Pasta, g/day | 43 | 0.56 | 0.51 | - | 0.15 | 0.10 | 0.47 | 0.11 | 0.35 |
| Rice, g/day | 45 | 0.45 | 0.45 | - | 0.14 | 0.09 | 0.47 | 0.16 | 0.53 |
| Vegetables, g/day | 41 | 0.47 | 0.47 | 32 | 0.20 | 0.38 | 0.32 | 0.19 | 0.31 |
| Fruit, g/day | 52 | 0.67 | 0.67 | 40 | 0.48 | 0.65 | 0.60 | 0.49 | 0.61 |
| Dairy, g/day | 45 | 0.58 | 0.58 | 45 | 0.58 | 0.66 | 0.71 | 0.56 | 0.69 |
| Soup, g/day | 37 | 0.46 | 0.45 | 48 | 0.24 | 0.10 | 0.76 | 0.20 | 0.63 |
| Coffee, g/day | 49 | 0.79 | 0.76 | 55 | 0.76 | 0.82 | 0.84 | 0.75 | 0.83 |
| SSB, g/day | 58 | 0.50 | 0.41 | 51 | 0.32 | 0.48 | 0.46 | 0.31 | 0.45 |
| ASB, g/day | - | 0.44 | 0.51 | - | 0.28 | 0.66 | 0.34 | 0.33 | 0.41 |
| Fruit juices, g/day | 51 | 0.59 | 0.56 | 46 | 0.40 | 0.51 | 0.56 | 0.33 | 0.46 |
| Alcoholic beverages, g/day | 53 | 0.67 | 0.65 | 42 | 0.47 | 0.64 | 0.59 | 0.41 | 0.51 |
| Savoury snacks, g/day | 39 | 0.44 | 0.37 | 40 | 0.22 | 0.12 | 0.64 | 0.16 | 0.46 |
| Cake/cookies, g/day | 47 | 0.62 | 0.55 | 36 | 0.42 | 0.30 | 0.77 | 0.41 | 0.75 |
| Sweets, g/day | 42 | 0.45 | 0.44 | 37 | 0.37 | 0.48 | 0.53 | 0.44 | 0.64 |
| GI | 43 | 0.58 | 0.53 | 35 | 0.40 | 0.39 | 0.64 | 0.38 | 0.61 |
| GL | 48 | 0.65 | 0.30 | 44 | 0.57 | 0.59 | 0.74 | 0.27 | 0.35 |
| GI-FFQ with HbA1c | General-FFQ with HbA1c | 24 h-Recalls with HbA1c | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crude Spearman | Same Quartile | Adjacent Quartile | Crude Spearman r | Same Quartile | Adjacent Quartile | Crude Spearman r | De-Attenuated | Same Quartile | Adjacent Quartile | |||||
| r | P | % | % | r | P | % | % | r | P | ICC | r | % | % | |
| Carbohydrates | 0.00 | 0.97 | 26 | 36 | −0.03 | 0.56 | 28 | 35 | −0.04 | 0.40 | 0.63 | −0.05 | 23 | 38 |
| Mono- and disaccharides | 0.02 | 0.62 | 24 | 41 | 0.00 | 0.99 | 22 | 41 | −0.05 | 0.28 | 0.63 | −0.06 | 23 | 38 |
| Polysaccharides | −0.02 | 0.72 | 26 | 36 | −0.05 | 0.26 | 28 | 34 | 0.00 | 0.94 | 0.58 | −0.01 | 25 | 37 |
| Fibres | −0.02 | 0.70 | 27 | 35 | 0.02 | 0.72 | 27 | 39 | −0.02 | 0.72 | 0.67 | −0.02 | 25 | 35 |
| GI | −0.09 | 0.04 | 23 | 36 | −0.04 | 0.34 | 25 | 36 | 0.04 | 0.35 | 0.39 | 0.07 | 26 | 40 |
| GL | −0.02 | 0.74 | 28 | 34 | −0.03 | 0.49 | 26 | 37 | −0.02 | 0.61 | 0.59 | −0.03 | 25 | 36 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brouwer-Brolsma, E.M.; Berendsen, A.A.M.; Sluik, D.; Van de Wiel, A.M.; Raben, A.; De Vries, J.H.M.; Brand-Miller, J.; Feskens, E.J.M. The Glycaemic Index-Food-Frequency Questionnaire: Development and Validation of a Food Frequency Questionnaire Designed to Estimate the Dietary Intake of Glycaemic Index and Glycaemic Load: An Effort by the PREVIEW Consortium. Nutrients 2019, 11, 13. https://doi.org/10.3390/nu11010013
Brouwer-Brolsma EM, Berendsen AAM, Sluik D, Van de Wiel AM, Raben A, De Vries JHM, Brand-Miller J, Feskens EJM. The Glycaemic Index-Food-Frequency Questionnaire: Development and Validation of a Food Frequency Questionnaire Designed to Estimate the Dietary Intake of Glycaemic Index and Glycaemic Load: An Effort by the PREVIEW Consortium. Nutrients. 2019; 11(1):13. https://doi.org/10.3390/nu11010013
Chicago/Turabian StyleBrouwer-Brolsma, Elske M., Agnes A.M. Berendsen, Diewertje Sluik, Anne M. Van de Wiel, Anne Raben, Jeanne H.M. De Vries, Jennie Brand-Miller, and Edith J.M. Feskens. 2019. "The Glycaemic Index-Food-Frequency Questionnaire: Development and Validation of a Food Frequency Questionnaire Designed to Estimate the Dietary Intake of Glycaemic Index and Glycaemic Load: An Effort by the PREVIEW Consortium" Nutrients 11, no. 1: 13. https://doi.org/10.3390/nu11010013
APA StyleBrouwer-Brolsma, E. M., Berendsen, A. A. M., Sluik, D., Van de Wiel, A. M., Raben, A., De Vries, J. H. M., Brand-Miller, J., & Feskens, E. J. M. (2019). The Glycaemic Index-Food-Frequency Questionnaire: Development and Validation of a Food Frequency Questionnaire Designed to Estimate the Dietary Intake of Glycaemic Index and Glycaemic Load: An Effort by the PREVIEW Consortium. Nutrients, 11(1), 13. https://doi.org/10.3390/nu11010013







