Sleep Duration and Excessive Daytime Sleepiness Are Associated with Obesity Independent of Diet and Physical Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sleep Measures
2.3. Anthropometric Assessment
2.4. Lifestyle Factors
2.5. Other Covariates
2.6. Statistical Analyses
3. Results
3.1. Study Population
3.2. Main Characteristics and Indices of Obesity by Sleep Duration
3.3. Main Characteristics and Indices of Obesity by Excessive Daytime Sleepiness
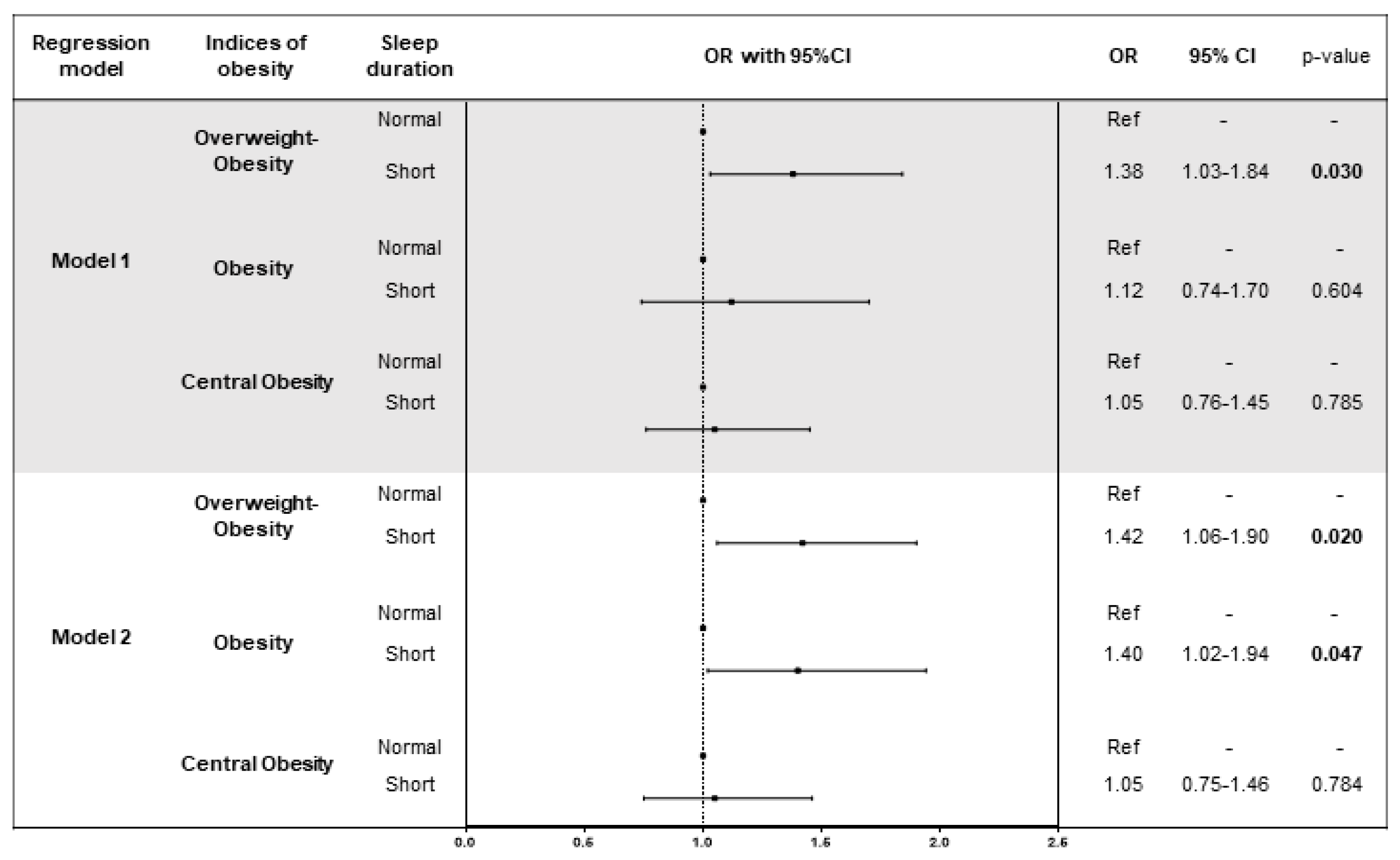
3.4. Association of Sleep Duration with Indices of Obesity
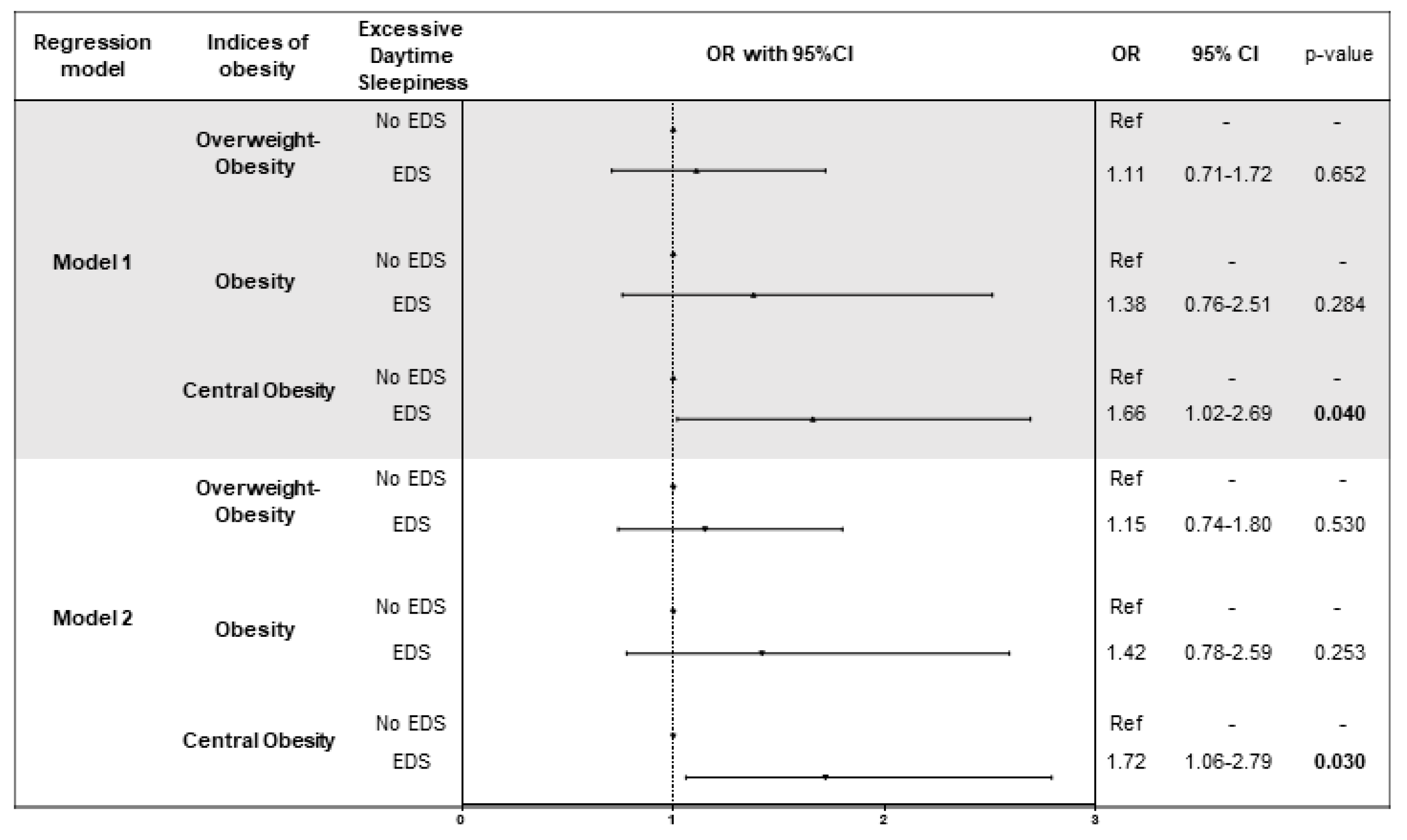
3.5. Association of Daytime Sleepiness with Indices of Obesity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Meir Kryger, T.R. William Dement. In Principles and Practice of Sleep Medicine, 5th ed.; Elsevier/Saunders: Amsterdam, The Netherlands, 2010; p. 1766. [Google Scholar]
- Medic, G.; Wille, M.; Hemels, M.E. Short- and long-term health consequences of sleep disruption. Nat. Sci. Sleep 2017, 9, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Prospective Studies, C.; Whitlock, G.; Lewington, S.; Sherliker, P.; Clarke, R.; Emberson, J.; Halsey, J.; Qizilbash, N.; Collins, R.; Peto, R. Body-mass index and cause-specific mortality in 900,000 adults: Collaborative analyses of 57 prospective studies. Lancet 2009, 373, 1083–1096. [Google Scholar] [CrossRef]
- Cappuccio, F.P.; D’Elia, L.; Strazzullo, P.; Miller, M.A. Sleep duration and all-cause mortality: A systematic review and meta-analysis of prospective studies. Sleep 2010, 33, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Seidell, J.C.; Halberstadt, J. The global burden of obesity and the challenges of prevention. Ann. Nutr. Metab. 2015, 66 (Suppl. 2), 7–12. [Google Scholar] [CrossRef] [PubMed]
- Sahakyan, K.R.; Somers, V.K.; Rodriguez-Escudero, J.P.; Hodge, D.O.; Carter, R.E.; Sochor, O.; Coutinho, T.; Jensen, M.D.; Roger, V.L.; Singh, P.; et al. Normal-weight central obesity: Implications for total and cardiovascular mortality. Ann. Intern. Med. 2015, 163, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Beccuti, G.; Pannain, S. Sleep and obesity. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Cappuccio, F.P.; Taggart, F.M.; Kandala, N.B.; Currie, A.; Peile, E.; Stranges, S.; Miller, M.A. Meta-analysis of short sleep duration and obesity in children and adults. Sleep 2008, 31, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhai, L.; Zhang, D. Sleep duration and obesity among adults: A meta-analysis of prospective studies. Sleep Med. 2014, 15, 1456–1462. [Google Scholar] [CrossRef] [PubMed]
- LaGrotte, C.; Fernandez-Mendoza, J.; Calhoun, S.L.; Liao, D.; Bixler, E.O.; Vgontzas, A.N. The relative association of obstructive sleep apnoea, obesity and excessive daytime sleepiness with incident depression: A longitudinal, population-based study. Int. J. Obes. 2016, 40, 1397–1404. [Google Scholar] [CrossRef] [PubMed]
- Cepeda, M.S.; Stang, P.; Blacketer, C.; Kent, J.M.; Wittenberg, G.M. Clinical relevance of sleep duration: Results from a cross-sectional analysis using nhanes. J. Clin. Sleep Med. 2016, 12, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Locke, A.E.; Kahali, B.; Berndt, S.I.; Justice, A.E.; Pers, T.H.; Day, F.R.; Powell, C.; Vedantam, S.; Buchkovich, M.L.; Yang, J.; et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 2015, 518, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Sayón-Orea, C.; Bes-Rastrollo, M.; Carlos, S.; Beunza, J.J.; Basterra-Gortari, F.J.; Martínez-González, M.A. Association between sleeping hours and siesta and the risk of obesity: The sun mediterranean cohort. Obes. Facts 2013, 6, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Stranges, S.; Cappuccio, F.P.; Kandala, N.B.; Miller, M.A.; Taggart, F.M.; Kumari, M.; Ferrie, J.E.; Shipley, M.J.; Brunner, E.J.; Marmot, M.G. Cross-sectional versus prospective associations of sleep duration with changes in relative weight and body fat distribution: The whitehall ii study. Am. J. Epidemiol. 2008, 167, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.R.; Malhotra, A.; White, D.P.; Gottlieb, D.J.; Hu, F.B. Association between reduced sleep and weight gain in women. Am. J. Epidemiol. 2006, 164, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kikuchi, H.; Tanaka, K.; Takahashi, M. Association of short sleep duration with weight gain and obesity at 1-year follow-up: A large-scale prospective study. Sleep 2010, 33, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Movsisyan, N.K.; Vinciguerra, M.; Lopez-Jimenez, F.; Kunzová, Š.; Homolka, M.; Jaresova, J.; Cífková, R.; Sochor, O. Kardiovize brno 2030, a prospective cardiovascular health study in central europe: Methods, baseline findings and future directions. Eur. J. Prev. Cardiol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Overweight and obesity—bmi statistics. Available online: http://ec.europa.eu/eurostat/statistics-explained/index.php/Overweight_and_obesity_-_BMI_statistics (accessed on 10 July 2018).
- Cardiovascular diseases statistics. Available online: http://ec.europa.eu/eurostat/statistics-explained/index.php/Cardiovascular_diseases_statistics (accessed on 10 July 2018).
- Agodi, A.; Maugeri, A.; Kunzova, S.; Sochor, O.; Bauerova, H.; Kiacova, N.; Barchitta, M.; Vinciguerra, M. Association of dietary patterns with metabolic syndrome: Results from the kardiovize brno 2030 study. Nutrients 2018, 10, 898. [Google Scholar] [CrossRef] [PubMed]
- Hruskova, J.; Maugeri, A.; Podroužková, H.; Štípalová, T.; Jakubík, J.; Barchitta, M.; Medina-Inojosa, J.R.; Homolka, M.; Agodi, A.; Kunzova, S.; et al. Association of cardiovascular health with epicardial adipose tissue and intima media thickness: The kardiovize study. J. Clin. Med. 2018, 7, 113. [Google Scholar] [CrossRef] [PubMed]
- Maugeri, A.; Kunzova, S.; Medina-Inojosa, J.R.; Agodi, A.; Barchitta, M.; Homolka, M.; Kiacova, N.; Bauerova, H.; Sochor, O.; Lopez-Jimenez, F.; et al. Association between eating time interval and frequency with ideal cardiovascular health: Results from a random sample czech urban population. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (redcap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Watson, N.F.; Badr, M.S.; Belenky, G.; Bliwise, D.L.; Buxton, O.M.; Buysse, D.; Dinges, D.F.; Gangwisch, J.; Grandner, M.A.; Kushida, C.; et al. Recommended amount of sleep for a healthy adult: A joint consensus statement of the American academy of sleep medicine and sleep research society. J. Clin. Sleep Med. 2015, 11, 591–592. [Google Scholar] [PubMed]
- Dudysova, D.; Mala, I.; Mlada, K.; Saifutdinova, E.; Koprivova, J.; Sos, P. Structural and construct validity of the Czech version of the Pittsburgh sleep quality index in chronic insomnia. Neuro Endocrinol. Lett. 2017, 38, 67–73. [Google Scholar] [PubMed]
- Johns, M.; Hocking, B. Daytime sleepiness and sleep habits of Australian workers. Sleep 1997, 20, 844–849. [Google Scholar] [CrossRef] [PubMed]
- Hayley, A.C.; Williams, L.J.; Kennedy, G.A.; Berk, M.; Brennan, S.L.; Pasco, J.A. Excessive daytime sleepiness and body composition: A population-based study of adults. PLoS ONE 2014, 9, e112238. [Google Scholar] [CrossRef] [PubMed]
- Netzer, N.C.; Stoohs, R.A.; Netzer, C.M.; Clark, K.; Strohl, K.P. Using the berlin questionnaire to identify patients at risk for the sleep apnoea syndrome. Ann. Intern. Med. 1999, 131, 485–491. [Google Scholar] [CrossRef] [PubMed]
- WHO. Waist Circumference and Waist–Hip Ratio: Report of a Who Expert Consultation; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Jones, D.M.; Hong, Y.; Labarthe, D.; Mozaffarian, D.; Appel, L.J.; Van Horn, L.; Greenlund, K.; Daniels, S.; Nichol, G.; Tomaselli, G.F.; et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: The American heart association’s strategic impact goal through 2020 and beyond. Circulation 2010, 121, 586–613. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Jin, X.; Shan, Z.; Li, S.; Huang, H.; Li, P.; Peng, X.; Peng, Z.; Yu, K.; Bao, W.; et al. Relationship of sleep duration with all-cause mortality and cardiovascular events: A systematic review and dose-response meta-analysis of prospective cohort studies. J. Am. Heart Assoc. 2017, 6, e005947. [Google Scholar] [CrossRef] [PubMed]
- Bin, Y.S.; Marshall, N.S.; Glozier, N. Sleeping at the limits: The changing prevalence of short and long sleep durations in 10 countries. Am. J. Epidemiol. 2013, 177, 826–833. [Google Scholar] [CrossRef] [PubMed]
- Prather, A.A.; Vogelzangs, N.; Penninx, B.W. Sleep duration, insomnia and markers of systemic inflammation: Results from The Netherlands study of depression and anxiety (Nesda). J. Psychiatr. Res. 2015, 60, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Irish, L.A.; Kline, C.E.; Gunn, H.E.; Buysse, D.J.; Hall, M.H. The role of sleep hygiene in promoting public health: A review of empirical evidence. Sleep Med. Rev. 2015, 22, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Shockey, T.M.; Wheaton, A.G. Short sleep duration by occupation group—29 states, 2013–2014. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Conomos, M.P.; Manor, O.; Rohwer, J.E.; Magis, A.T.; Lovejoy, J.C. Habitual sleep duration and sleep duration variation are independently associated with body mass index. Int. J. Obes. 2017, 42, 794–800. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P.; Grandner, M.A.; Brown, D.; Conroy, M.B.; Jean-Louis, G.; Coons, M.; Bhatt, D.L.; American Heart Association Obesity, Behavior Change, Diabetes; Nutrition Committees of the Council on Lifestyle; Cardiometabolic Health; et al. Sleep duration and quality: Impact on lifestyle behaviours and cardiometabolic health: A scientific statement from the American heart association. Circulation 2016, 134, e367–e386. [Google Scholar] [CrossRef] [PubMed]
- Potter, G.D.M.; Cade, J.E.; Hardie, L.J. Longer sleep is associated with lower BMI and favorable metabolic profiles in UK adults: Findings from the national diet and nutrition survey. PLoS ONE 2017, 12, e0182195. [Google Scholar] [CrossRef] [PubMed]
- Ferranti, R.; Marventano, S.; Castellano, S.; Giogianni, G.; Nolfo, F.; Rametta, S.; Matalone, M.; Mistretta, A. Sleep quality and duration is related with diet and obesity in young adolescent living in Sicily, southern Italy. Sleep Sci. 2016, 9, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Bixler, E.O.; Vgontzas, A.N.; Lin, H.M.; Calhoun, S.L.; Vela-Bueno, A.; Kales, A. Excessive daytime sleepiness in a general population sample: The role of sleep apnoea, age, obesity, diabetes and depression. J. Clin. Endocrinol. Metab. 2005, 90, 4510–4515. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Papanicolaou, D.A.; Bixler, E.O.; Kales, A.; Tyson, K.; Chrousos, G.P. Elevation of plasma cytokines in disorders of excessive daytime sleepiness: Role of sleep disturbance and obesity. J. Clin. Endocrinol. Metab. 1997, 82, 1313–1316. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Bixler, E.O.; Tan, T.L.; Kantner, D.; Martin, L.F.; Kales, A. Obesity without sleep apnoea is associated with daytime sleepiness. Arch. Intern. Med. 1998, 158, 1333–1337. [Google Scholar] [CrossRef] [PubMed]
- Resta, O.; Foschino Barbaro, M.P.; Bonfitto, P.; Giliberti, T.; Depalo, A.; Pannacciulli, N.; De Pergola, G. Low sleep quality and daytime sleepiness in obese patients without obstructive sleep apnoea syndrome. J. Intern. Med. 2003, 253, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Fernandez-Mendoza, J.; Miksiewicz, T.; Kritikou, I.; Shaffer, M.L.; Liao, D.; Basta, M.; Bixler, E.O. Unveiling the longitudinal association between short sleep duration and the incidence of obesity: The penn state cohort. Int. J. Obes. 2014, 38, 825–832. [Google Scholar] [CrossRef] [PubMed]
- Pillar, G.; Shehadeh, N. Abdominal fat and sleep apnea: The chicken or the egg? Diabetes Care 2008, 31, S303–S309. [Google Scholar] [CrossRef] [PubMed]
- Dixon, J.B.; Dixon, M.E.; Anderson, M.L.; Schachter, L.; O’Brien, P.E. Daytime sleepiness in the obese: Not as simple as obstructive sleep apnea. Obesity 2007, 15, 2504–2511. [Google Scholar] [CrossRef] [PubMed]
- Jaussent, I.; Morin, C.M.; Ivers, H.; Dauvilliers, Y. Incidence, worsening and risk factors of daytime sleepiness in a population-based 5-year longitudinal study. Sci. Rep. 2017, 7, 1372. [Google Scholar] [CrossRef] [PubMed]
- St-Onge, M.P. Sleep-obesity relation: Underlying mechanisms and consequences for treatment. Obes. Rev. 2017, 18, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Broussard, J.L.; Chapotot, F.; Abraham, V.; Day, A.; Delebecque, F.; Whitmore, H.R.; Tasali, E. Sleep restriction increases free fatty acids in healthy men. Diabetologia 2015, 58, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Bosy-Westphal, A.; Hinrichs, S.; Jauch-Chara, K.; Hitze, B.; Later, W.; Wilms, B.; Settler, U.; Peters, A.; Kiosz, D.; Muller, M.J. Influence of partial sleep deprivation on energy balance and insulin sensitivity in healthy women. Obes. Facts 2008, 1, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Markwald, R.R.; Melanson, E.L.; Smith, M.R.; Higgins, J.; Perreault, L.; Eckel, R.H.; Wright, K.P., Jr. Impact of insufficient sleep on total daily energy expenditure, food intake and weight gain. Proc. Natl. Acad. Sci. USA 2013, 110, 5695–5700. [Google Scholar] [CrossRef] [PubMed]
- Spaeth, A.M.; Dinges, D.F.; Goel, N. Effects of experimental sleep restriction on weight gain, caloric intake and meal timing in healthy adults. Sleep 2013, 36, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Cowan, D.C.; Livingston, E. Obstructive sleep apnoea syndrome and weight loss: Review. Sleep Disord. 2012, 2012, 163296. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.P.; Foster, R.G.; Peirson, S.N. The circadian control of sleep. Handb. Exp. Pharmacol. 2013, 157–183. [Google Scholar]
- Mazzoccoli, G.; Pazienza, V.; Vinciguerra, M. Clock genes and clock-controlled genes in the regulation of metabolic rhythms. Chronobiol. Int. 2012, 29, 227–251. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Sleep Duration | p-Value b | ||
|---|---|---|---|---|
| Short (n = 457) | Normal (n = 1011) | Long (n = 14) | ||
| Age, years | 48.0 (22.0) | 44.5 (20.0) | 29.0 (20.0) | <0.001 |
| Sex (% male) | 48.4% | 51.7% | 28.6% | 0.126 |
| Marital status (% married) | 65.4% | 63.9% | 40.0% | 0.247 |
| Employment (% workers) | 80.7% | 83.1% | 77.8% | 0.567 |
| Smoking status (% current) | 21.4% | 19.9% | 20.0% | 0.838 |
| Diet | ||||
| 0–1 components | 11.4% | 14.3% | 11.3% | 0.131 |
| 2–3 components | 84.2% | 82.4% | 80.3% | |
| 4–5 components | 4.4% | 3.4% | 8.5% | |
| Total energy intake, Kcal a | 2077 (966) | 2063 (968) | 1925 (755) | 0.311 |
| Physical activity, MET-min/week a | 3876 (5400) | 3035 (4352) | 1896 (3774) | <0.001 |
| Physical activity categories | ||||
| Low | 11.8% | 14.2% | 7.1% | 0.219 |
| Moderate | 29.5% | 32.0% | 50.0% | |
| High | 58.6% | 53.7% | 42.9% | |
| Diabetes | 9.8% | 6.8% | 0% | 0.073 |
| Use of antidiabetics | 5.2% | 2.5% | 0.0% | 0.051 |
| Hypertension | 42.0% | 35.6% | 28.6% | 0.052 |
| Use of antihypertensives | 21.9% | 16.1% | 20.0% | 0.063 |
| Hyperlipidaemia | 64.7% | 68.1% | 78.6% | 0.289 |
| Use of hypolipidaemics | 8.9% | 6.2% | 10.0% | 0.235 |
| ESS a | 6.0 (6.0) | 5.0 (4.0) | 3.0 (4.0) | <0.001 |
| High risk of sleep apnoea | 27.1% | 17.4% | 14.3% | <0.001 |
| Characteristics | Sleep Duration | p-Value b | ||
|---|---|---|---|---|
| Short (n = 457) | Normal (n = 1011) | Long (n = 14) | ||
| BMI, Kg/m2 a | 26.7 (6.3) | 24.7 (5.4) | 23.7 (8.3) | lt;0.001 |
| BMI categories | ||||
| Underweight | 1.8% | 2.4% | 7.1% | 0.002 |
| Normal weight | 39.4% | 49.4% | 57.1% | |
| Overweight | 37.9% | 34.1% | 21.4% | |
| Obesity | 21.0% | 14.1% | 14.3% | |
| WHR a | 0.89 (0.16) | 0.86 (0.14) | 0.78 (0.20) | 0.018 |
| Central obesity | 45.2% | 38.4% | 28.6% | 0.032 |
| Body fat mass, Kg a | 20.0 (13.6) | 17.3 (11.6) | 17.9 (9.6) | 0.004 |
| Characteristics | Excessive Daytime Sleepiness | p-Value b | |
|---|---|---|---|
| No EDS (n = 1319) | EDS (n = 163) | ||
| Age, years | 44.0 (20.0) | 48.5 (22.0) | 0.711 |
| Sex (% male) | 49.6% | 58.3% | 0.036 |
| Marital status (% married) | 63.5% | 72.3% | 0.058 |
| Employment (% workers) | 81.9% | 87.4% | 0.136 |
| Smoking status (% current) | 21.1% | 14.3% | 0.081 |
| Diet | |||
| 0–1 components | 13.2% | 13.5% | 0.836 |
| 2–3 components | 82.7% | 83.4% | |
| 4–5 components | 4.0% | 3.1% | |
| Total energy intake, Kcal a | 2049 (948) | 2136 (888) | 0.002 |
| Physical activity, MET-min/week a | 3074 (4164) | 2253 (5773) | 0.406 |
| Physical activity categories | |||
| Low | 13.5% | 13.5% | 0.832 |
| Moderate | 31.7% | 29.4% | |
| High | 54.8% | 57.1% | |
| Diabetes | 7.8% | 6.7% | 0.625 |
| Use of antidiabetics | 3.3% | 3.4% | 0.957 |
| Hypertension | 37.0% | 41.7% | 0.239 |
| Use of antihypertensives | 18.1% | 16.0% | 0.569 |
| Hyperlipidaemia | 66.9% | 69.3% | 0.538 |
| Use of hypolipidaemics | 7.3% | 5.0% | 0.361 |
| Short sleep duration | 28.6% | 46.6% | <0.001 |
| High risk of sleep apnoea | 19.5% | 27.0% | 0.026 |
| Characteristics | Excessive Daytime Sleepiness | p-Value b | |
|---|---|---|---|
| No EDS (n = 1319) | EDS (n = 163) | ||
| BMI, Kg/m2 a | 25.0 (6.3) | 28.6 (7.3) | 0.020 |
| BMI categories | |||
| Underweight | 2.4% | 1.2% | 0.150 |
| Normal weight | 47.1% | 39.9% | |
| Overweight | 34.8% | 38.0% | |
| Obesity | 15.7% | 20.9% | |
| WHR a | 0.87 (0.14) | 0.93 (0.12) | 0.013 |
| Central obesity | 38.9% | 52.1% | 0.001 |
| Body fat mass, Kg a | 17.9 (12.2) | 20.0 (11.9) | 0.772 |
| BMI, Kg/m2 a | 38.0 (7.0) | 39.0 (5.0) | 0.005 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maugeri, A.; Medina-Inojosa, J.R.; Kunzova, S.; Agodi, A.; Barchitta, M.; Sochor, O.; Lopez-Jimenez, F.; Geda, Y.E.; Vinciguerra, M. Sleep Duration and Excessive Daytime Sleepiness Are Associated with Obesity Independent of Diet and Physical Activity. Nutrients 2018, 10, 1219. https://doi.org/10.3390/nu10091219
Maugeri A, Medina-Inojosa JR, Kunzova S, Agodi A, Barchitta M, Sochor O, Lopez-Jimenez F, Geda YE, Vinciguerra M. Sleep Duration and Excessive Daytime Sleepiness Are Associated with Obesity Independent of Diet and Physical Activity. Nutrients. 2018; 10(9):1219. https://doi.org/10.3390/nu10091219
Chicago/Turabian StyleMaugeri, Andrea, Jose R. Medina-Inojosa, Sarka Kunzova, Antonella Agodi, Martina Barchitta, Ondrej Sochor, Francisco Lopez-Jimenez, Yonas E. Geda, and Manlio Vinciguerra. 2018. "Sleep Duration and Excessive Daytime Sleepiness Are Associated with Obesity Independent of Diet and Physical Activity" Nutrients 10, no. 9: 1219. https://doi.org/10.3390/nu10091219
APA StyleMaugeri, A., Medina-Inojosa, J. R., Kunzova, S., Agodi, A., Barchitta, M., Sochor, O., Lopez-Jimenez, F., Geda, Y. E., & Vinciguerra, M. (2018). Sleep Duration and Excessive Daytime Sleepiness Are Associated with Obesity Independent of Diet and Physical Activity. Nutrients, 10(9), 1219. https://doi.org/10.3390/nu10091219








