Quality of Life of Celiac Patients in Brazil: Questionnaire Translation, Cultural Adaptation and Validation
Abstract
1. Introduction
2. Materials and Methods
2.1. Questionnaire
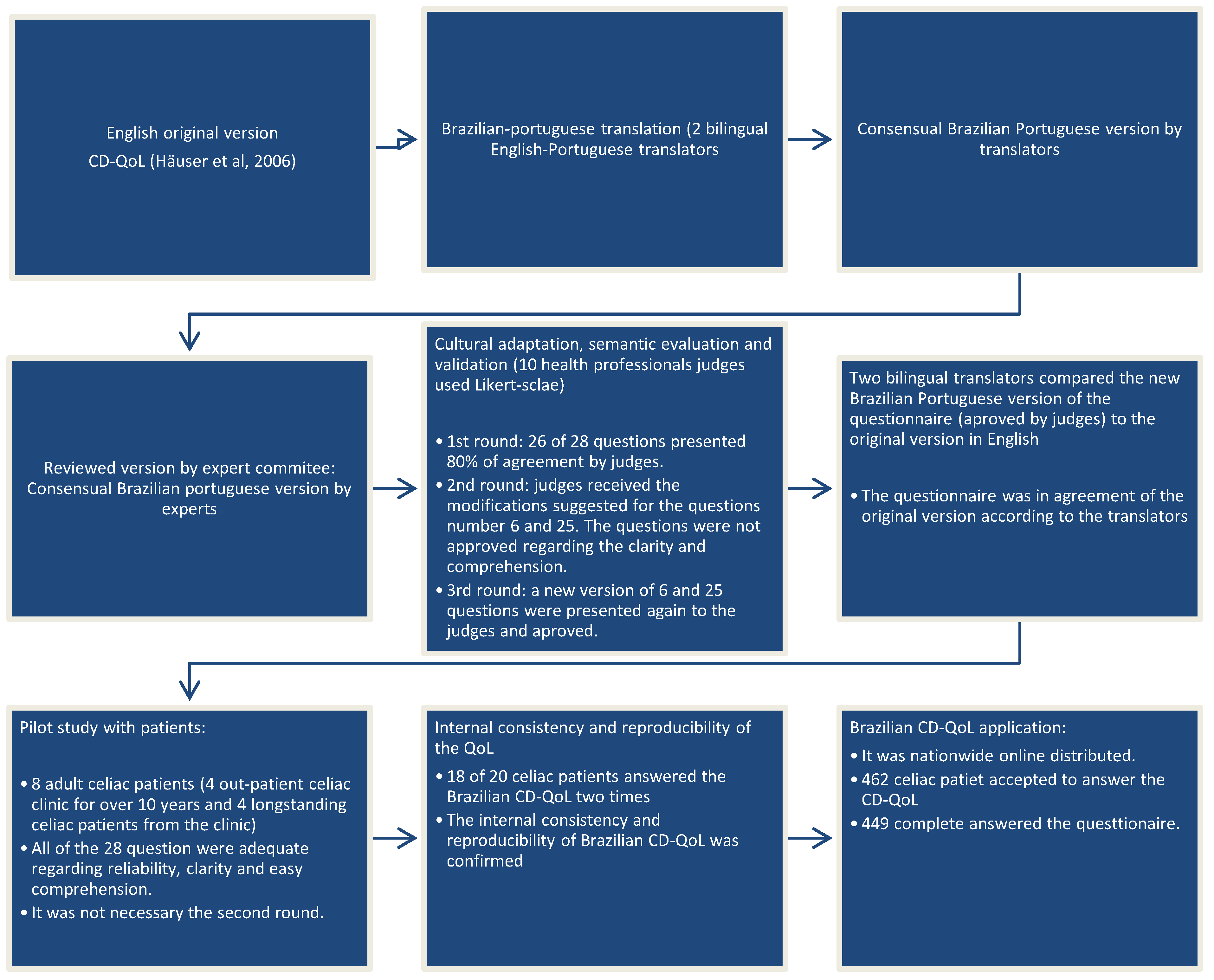
2.2. Translation, Cultural Adaptation and Validation
2.2.1. Translation and Retranslation
2.2.2. Cultural Adaptation, Semantic Evaluation and Validation (First Step)
2.2.3. Pilot Test (Second Step)
2.2.4. Internal Consistency and Reproducibility of the Brazilian CD-QoL
2.3. Brazilian CD-QoL Application
2.4. Psychometric Evaluation, Validation and Statistics
3. Results
3.1. Translation, Cultural Adaptation, Semantic Evaluation and Content Validation
Internal Consistency, Construct Validity and Reproducibility of the Brazilian CD-QoL
3.2. CD-QoL Application
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shamir, R.; Heyman, M.B.; Koning, F.; Wijimenga, C.; Gutierrez-Achury, J.; Catassi, C.; Gatti, S.; Fasano, A.; Discepolo, V.; Korponay-Szabó, I.R.; et al. Celiac Disease. J. Pediatr. Gastroenterol. Nutr. 2014, 59, S1. [Google Scholar] [CrossRef] [PubMed]
- Schuppan, D.; Junker, Y.; Barisani, D. Celiac Disease: From Pathogenesis to Novel Therapies. Gastroenterology 2009, 137, 1912–1933. [Google Scholar] [CrossRef] [PubMed]
- Barada, K.; Abu Daya, H.; Rostami, K.; Catassi, C. Celiac Disease in the Developing World. Gastrointest. Endosc. Clin. N. Am. 2012, 22, 773–796. [Google Scholar] [CrossRef] [PubMed]
- Barker, J.M.; Liu, E. Celiac disease: Pathophysiology, clinical manifestations and associated autoimmune conditions. Adv. Pediatr. 2008, 55, 349–365. [Google Scholar] [CrossRef] [PubMed]
- Kaukinen, K.; Lindfors, K.; Mäki, M. Advances in the treatment of coeliac disease: An immunopathogenic perspective. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Zingone, F.; Iavarone, A.; Tortora, R.; Imperatore, N.; Pellegrini, L.; Russo, T.; Dorn, S.D.; Ciacci, C. The Italian translation of the Celiac Disease-specific Quality of Life Scale in celiac patients on gluten free diet. Dig. Liver Dis. 2013, 45, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Häuser, W.; Gold, J.; Stein, J.; Caspary, W.F.; Stallmach, A. Health-related quality of life in adult coeliac disease in Germany: Results of a national survey. Eur. J. Gastroenterol. Hepatol. 2006, 18, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Klersy, C.; Biagi, F.; Balduzzi, D.; Bianchi, P.I.; Trotta, L.; Vattiato, C.; Zilli, A.; Rademacher, J.; Andrealli, A.; et al. Quality of life in coeliac patients: Italian validation of a coeliac questionnaire. Eur. J. Intern. Med. 2013, 24, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Aksan, A.; Mercanlıgil, S.M.; Häuser, W.; Karaismailoğlu, E. Validation of the Turkish version of the Celiac Disease Questionnaire (CDQ). Health Qual. Life Outcomes 2015, 13, 82. [Google Scholar] [CrossRef] [PubMed]
- Pouchot, J.; Despujol, C.; Malamut, G.; Ecosse, E.; Coste, J.; Cellier, C. Validation of a French version of the quality of life “Celiac Disease Questionnaire”. PLoS ONE 2014, 9, e96346. [Google Scholar] [CrossRef] [PubMed]
- Machado, J.; Gandolfi, L.; Coutinho De Almeida, F.; Malta Almeida, L.; Puppin Zandonadi, R.; Pratesi, R. Gluten-free dietary compliance in Brazilian celiac patients: Questionnaire versus serological test. Nutr. Clin. Diet. Hosp. 2013, 33, 46–49. [Google Scholar] [CrossRef]
- Farage, P.; Puppin Zandonadi, R.; Cortez Ginani, V.; Gandolfi, L.; Pratesi, R.; de Medeiros Nóbrega, Y.K. Content Validation and Semantic Evaluation of a Check-List Elaborated for the Prevention of Gluten Cross-Contamination in Food Services. Nutrients 2017, 9, 36. [Google Scholar] [CrossRef] [PubMed]
- Sverker, A.; Hensing, G.; Hallert, C. ‘Controlled by food’—Lived experiences of coeliac disease. J. Hum. Nutr. Diet. 2005, 18, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Bullinger, M.; Alonso, J.; Apolone, G.; Leplège, A.; Sullivan, M.; Wood-Dauphinee, S.; Gandek, B.; Wagner, A.; Aaronson, N.; Bech, P.; et al. Translating health status questionnaires and evaluating their quality: The IQOLA Project approach. International Quality of Life Assessment. J. Clin. Epidemiol. 1998, 51, 913–923. [Google Scholar] [CrossRef]
- Okoli, C.; Pawlowski, S.D. The Delphi method as a research tool: An example, design considerations and applications. Inf. Manag. 2004, 42, 15–29. [Google Scholar] [CrossRef]
- De Lima, T.C.; Gallani, M.C.B.J.; de Freitas, M.I.P. Content validation of an instrument to characterize people over 50 years of age living with human immunodeficiency virus/acquired immunodeficiency syndrome. Acta Paul. Enferm. 2012, 25, 4–10. [Google Scholar] [CrossRef]
- Conti, M.A.; Scagliusi, F.; Kawamura De Oliveira Queiroz, G.; Hearst, N.; Cordás, T.A. Cross-cultural adaptation: Translation and Portuguese language content validation of the Tripartite Influence Scale for body dissatisfaction. Cad. Saude Publ. 2010, 26, 503–513. [Google Scholar] [CrossRef]
- Polit, D.F.; Beck, C.T. Nursing Research: Principles and Methods; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2004; ISBN 0781737338. [Google Scholar]
- Kline, R.B. Principles and Practice of Structural Equation Modeling, 3rd ed.; The Guilford Press: New York, NY, USA, 2011; ISBN 978-1-60623-877-6. [Google Scholar]
- Hu, L.; Bentler, P. Cutoff criteria for fit indices in covariance structure analysis: Conventional criteria versus new alternatives. Struct. Equ. Model. 1999, 6, 1–55. [Google Scholar] [CrossRef]
- World Health Organization. The Third Ten Years of the World Health Organization, 1st ed.; WHO: Geneva, Switzerland, 2008; ISBN 978 92 4 156366 6. [Google Scholar]
- Fagerdahl, A.-M.; Boström, L.; Ulfvarson, J.; Bergström, G.; Ottosson, C. Translation and validation of the wound-specific quality of life instrument Cardiff Wound Impact Schedule in a Swedish population. Scand. J. Caring Sci. 2014, 28, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.; Carlson, D.; Billington, R.; Orley, J. The WHO quality of life assessment instrument (WHOQOL-Bref): The importance of its items for cross-cultural research. Qual. Life Res. 2001, 10, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Guillemin, F.; Bombardier, C.; Beaton, D. Cross-cultural adaptation of health-related quality of life measures: Literature review and proposed guidelines. J. Clin. Epidemiol. 1993, 46, 1417–1432. [Google Scholar] [CrossRef]
- Beaton, D.E.; Bombardier, C.; Guillemin, F.; Ferraz, M.B. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine 2000, 25, 3186–3191. [Google Scholar] [CrossRef] [PubMed]
- Streiner, D.L. Starting at the Beginning: An Introduction to Coefficient Alpha and Internal Consistency Starting at the Beginning: An Introduction to Coefficient Alpha and Internal Consistency. J. Pers. Assess. 2003, 80, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Streiner, D.L.; Norman, G.R. Health Measurement Scales: A Practical Guide to Their Development and Use; Oxford University Press: Oxford, UK, 2008; ISBN 9780199231881. [Google Scholar]
- Mäki, M.; Collin, P. Coeliac disease. Lancet 1997, 349, 1755–1759. [Google Scholar] [CrossRef]
- Davidson, D.J.; Freudenburg, W.R. Gender and Environmental Risk Concerns. Environ. Behav. 1996, 28, 302–339. [Google Scholar] [CrossRef]
- Chen, M. Consumers’ health and taste attitude in Taiwan. Br. Food J. 2013, 115, 526–540. [Google Scholar] [CrossRef]
- Lee, A.R.; Wolf, R.; Contento, I.; Verdeli, H.; Green, P.H.R. Coeliac disease: The association between quality of life and social support network participation. J. Hum. Nutr. Diet. 2016, 29, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Castilhos, A.C.; Gonçalves, B.C.; Macedo e Silva, M.; Lanzoni, L.A.; Metzger, L.R.; Kotze, L.M.S.; Nisihara, R.M. Quality of life evaluation in celiac patients from Southern Brazil. Arq. Gastroenterol. 2015, 52, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Cervantes, K.L.; Remes-Troche, J.M.; del Pilar Milke-García, M.; Romero, V.; Uscanga, L.F. Characteristics and factors related to quality of life in Mexican Mestizo patients with celiac disease. BMC Gastroenterol. 2015, 15, 4. [Google Scholar] [CrossRef] [PubMed]
- Hallert, C.; Grännö, C.; Hultén, S.; Midhagen, G.; Ström, M.; Svensson, H.; Valdimarsson, T. Living with coeliac disease: Controlled study of the burden of illness. Scand. J. Gastroenterol. 2002, 37, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Hallert, C.; Sandlund, O.; Broqvist, M. Perceptions of health-related quality of life of men and women living with coeliac disease. Scand. J. Caring Sci. 2003, 17, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Zarkadas, M.; Cranney, A.; Case, S.; Molloy, M.; Switzer, C.; Graham, I.D.; Butzner, J.D.; Rashid, M.; Warren, R.E.; Burrows, V. The impact of a gluten-free diet on adults with coeliac disease: Results of a national survey. J. Hum. Nutr. Diet. 2006, 19, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Bellini, A.; Zanchi, C.; Martelossi, S.; Di Leo, G.; Not, T.; Ventura, A. Compliance with the Gluten-Free Diet: The Role of Locus of Control in Celiac Disease. J. Pediatr. 2011, 158, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Wagner, G.; Berger, G.; Sinnreich, U.; Grylli, V.; Schober, E.; Huber, W.-D.; Karwautz, A. Quality of Life in Adolescents with Treated Coeliac Disease: Influence of Compliance and Age at Diagnosis. J. Pediatr. Gastroenterol. Nutr. 2008, 47, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Roma, E.; Roubani, A.; Kolia, E.; Panayiotou, J.; Zellos, A.; Syriopoulou, V.P. Dietary compliance and life style of children with coeliac disease. J. Hum. Nutr. Diet. 2010, 23, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Häuser, W.; Stallmach, A.; Caspary, W.F.; Stein, J. Predictors of reduced health-related quality of life in adults with coeliac disease. Aliment. Pharmacol. Ther. 2007, 25, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Jacobsson, L.R.; Friedrichsen, M.; Göransson, A.; Hallert, C. Impact of an Active Patient Education Program on Gastrointestinal Symptoms in Women With Celiac Disease Following a Gluten-Free Diet. Gastroenterol. Nurs. 2012, 35, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.; Dickson-Swift, V.; Anderson, K. Coeliac disease: The path to diagnosis and the reality of living with the disease. J. Hum. Nutr. Diet. 2013, 26, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Gazmararian, J.A.; Williams, M.V.; Peel, J.; Baker, D.W. Health literacy and knowledge of chronic disease. Patient Educ. Couns. 2003, 51, 267–275. [Google Scholar] [CrossRef]
- Everson, S.A.; Maty, S.C.; Lynch, J.W.; Kaplan, G.A. Epidemiologic evidence for the relation between socioeconomic status and depression, obesity and diabetes. J. Psychosom. Res. 2002, 53, 891–895. [Google Scholar] [CrossRef]
- Rojas-García, A.; Ruiz-Perez, I.; Rodríguez-Barranco, M.; Gonçalves Bradley, D.C.; Pastor-Moreno, G.; Ricci-Cabello, I. Healthcare interventions for depression in low socioeconomic status populations: A systematic review and meta-analysis. Clin. Psychol. Rev. 2015, 38, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Elovainio, M.; Pulkki-Råback, L.; Jokela, M.; Kivimäki, M.; Hintsanen, M.; Hintsa, T.; Viikari, J.; Raitakari, O.T.; Keltikangas-Järvinen, L. Socioeconomic status and the development of depressive symptoms from childhood to adulthood: A longitudinal analysis across 27 years of follow-up in the Young Finns study. Soc. Sci. Med. 2012, 74, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Mehra, S.; Leffler, D.A.; Pallav, K.; Tariq, S.; Shah, S.; Green, P.H.; Hansen, J.; Dennis, M.; Kelly, C.P. Socioeconomic Status Influences Celiac Disease Diagnosis. Gastroenterology 2011, 140, S-438. [Google Scholar] [CrossRef]
- IBGE. Pesquisas de Orçamentos Familiares; Brasília, Brazil, 2010. Available online: https://biblioteca.ibge.gov.br/visualizacao/livros/liv50063.pdf (accessed on 3 June 2018).
| Mean (SD) | Median (IQR) | Range | Floor Effect (%) | Ceiling Effect (%) | Internal Consistency (Cronbach’s Alpha) | |
|---|---|---|---|---|---|---|
| Emotion | 26.70 (9.55) | 27 (20–34) | 7–49 | 1.3% | 0.2% | 0.927 |
| Social | 31.15 (8.18) | 32 (25–38) | 10–48 | 0% | 0% | 0.703 |
| Worries | 27.25 (9.85) | 27 (20–34) | 7–49 | 0.4% | 0.9% | 0.832 |
| Gastrointestinal | 34.87 (8.61) | 36 (29–41) | 10–49 | 0% | 2.4% | 0.793 |
| Total Score | 119.79 (30.16) | 120 (99–142) | 51–181 | 0% | 0% | 0.925 |
| Score | Pearson Correlation | Intraclass Correlation Coefficient | |||||
|---|---|---|---|---|---|---|---|
| Phase 1 Mean (SD) | Phase 2 Mean (SD) | p * | Correlation | p | ICC | p | |
| Emotion | 25.50 (10.16) | 27.06 (10.08) | 0.153 | 0.905 | <0.001 | 0.947 | <0.001 |
| Social | 33.00 (9.17) | 34.67 (7.08) | 0.224 | 0.769 | <0.001 | 0.850 | <0.001 |
| Worries | 30.06 (7.68) | 31.17 (8.54) | 0.291 | 0.863 | <0.001 | 0.923 | <0.001 |
| Gastrointestinal | 37.06 (6.91) | 35.17 (6.23) | 0.247 | 0.486 | 0.041 | 0.647 | 0.018 |
| Total | 125.61 (27.11) | 128.06 (27.08) | 0.531 | 0.821 | <0.001 | 0.905 | <0.001 |
| Emotion | Social | Worries | Gastrointestinal | Total | |
|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | |
| Gender * | |||||
| Women (n = 425) | 26.42 (9.51) | 30.08 (8.14) | 26.80 (9.69) | 34.75 (8.63) | 118.55 (29.98) |
| Men (n = 25) | 30.88 (9.03) | 36.40 (6.57) | 34.71 (9.96) | 36.84 (8.33) | 139.33 (25.95) |
| p | 0.023 | 0.000 | 0.000 | 0.238 | 0.001 |
| Age * | |||||
| 39 and under (n = 271) | 26.01 (9.13) | 30.28 (7.93) | 26.87 (9.64) | 34.28 (8.45) | 117.34 (28.98) |
| 40 and over (n = 179) | 27.74 (10.08) | 32.48 (8.40) | 27.82 (10.17) | 35.77 (8.79) | 123.52 (31.59) |
| p | 0.065 | 0.006 | 0.032 | 0.074 | 0.036 |
| Time of diagnosis * | |||||
| 29 and under (n = 170) | 25.27 (8.76) | 29.97 (8.14) | 26.75 (9.61) | 33.99 (8.51) | 115.72 (29.16) |
| 30 and over (n = 273) | 27.48 (9.92) | 31.80 (8.20) | 27.27 (9.93) | 35.34 (8.67) | 121.75 (30.58) |
| p | 0.015 | 0.024 | 0.588 | 0.109 | 0.043 |
| Educational level ** | |||||
| Elementary (n = 26) | 23.00 (10.92) a | 28.44 (6.68) a | 24.23 (10.78) a | 29.42 (8.68) a | 106.12 (30.32) a |
| High School (n = 116) | 24.10 (9.60) a,b | 29.29 (8.20) a,b | 25.69 (9.69) a,b | 32.69 (9.36) a,b | 111.57 (30.21) a,b |
| College (n = 152) | 27.32 (9.13) b,c | 31.34 (8.06) a,b | 27.29 (9.54) a,b | 35.43 (8.22) b,c | 121.01 (29.36) b,c |
| Graduate & Post-grad (n = 156) | 28.65 (9.14) c | 32.79 (8.16) b | 28.88 (9.90) b | 36.86 (7.67) c | 127.01 (28.86) c |
| p | 0.000 | 0.002 | 0.022 | 0.000 | 0.000 |
| Marital status * | |||||
| With partners (n = 275) | 27.51 (9.68) | 31.69 (8.25) | 27.30 (9.68) | 35.36 (8.39) | 121.78 (29.85) |
| Without partners (n = 175) | 25.43 (9.22) | 30.31 (8.02) | 27.18 (10.14) | 34.11 (8.91) | 116.68 (30.47) |
| p | 0.025 | 0.085 | 0.908 | 0.136 | 0.084 |
| Gluten-free diet compliance * | |||||
| No (n = 51) | 20.61 (9.37) | 28.10 (7.43) | 25.16 (10.97) | 29.73 (8.71) | 104.20 (30.00) |
| Yes (n = 399) | 27.48 (9.30) | 31.53 (8.20) | 27.52 (9.68) | 35.53 (8.38) | 121.76 (29.64) |
| p | 0.000 | 0.005 | 0.108 | 0.838 | 0.000 |
| Antidepressant medicines * | |||||
| No (n = 371) | 27.67 (9.37) | 31.67 (7.98) | 27.76 (9.78) | 35.50 (8.49) | 122.45 (29.63) |
| Yes (n = 79) | 22.15 (9.09) | 28.79 (8.72) | 24.85 (9.90) | 31.95 (8.63) | 107.73 (29.81) |
| p | 0.000 | 0.004 | 0.017 | 0.001 | 0.000 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pratesi, C.B.; Häuser, W.; Uenishi, R.H.; Selleski, N.; Nakano, E.Y.; Gandolfi, L.; Pratesi, R.; Zandonadi, R.P. Quality of Life of Celiac Patients in Brazil: Questionnaire Translation, Cultural Adaptation and Validation. Nutrients 2018, 10, 1167. https://doi.org/10.3390/nu10091167
Pratesi CB, Häuser W, Uenishi RH, Selleski N, Nakano EY, Gandolfi L, Pratesi R, Zandonadi RP. Quality of Life of Celiac Patients in Brazil: Questionnaire Translation, Cultural Adaptation and Validation. Nutrients. 2018; 10(9):1167. https://doi.org/10.3390/nu10091167
Chicago/Turabian StylePratesi, Claudia B., Winfred Häuser, Rosa Harumi Uenishi, Nicole Selleski, Eduardo Yoshio Nakano, Lenora Gandolfi, Riccardo Pratesi, and Renata Puppin Zandonadi. 2018. "Quality of Life of Celiac Patients in Brazil: Questionnaire Translation, Cultural Adaptation and Validation" Nutrients 10, no. 9: 1167. https://doi.org/10.3390/nu10091167
APA StylePratesi, C. B., Häuser, W., Uenishi, R. H., Selleski, N., Nakano, E. Y., Gandolfi, L., Pratesi, R., & Zandonadi, R. P. (2018). Quality of Life of Celiac Patients in Brazil: Questionnaire Translation, Cultural Adaptation and Validation. Nutrients, 10(9), 1167. https://doi.org/10.3390/nu10091167







