Evaluation of a High Concentrate Omega-3 for Correcting the Omega-3 Fatty Acid Nutritional Deficiency in Non-Alcoholic Fatty Liver Disease (CONDIN)
Abstract
1. Introduction
2. Materials and Methods
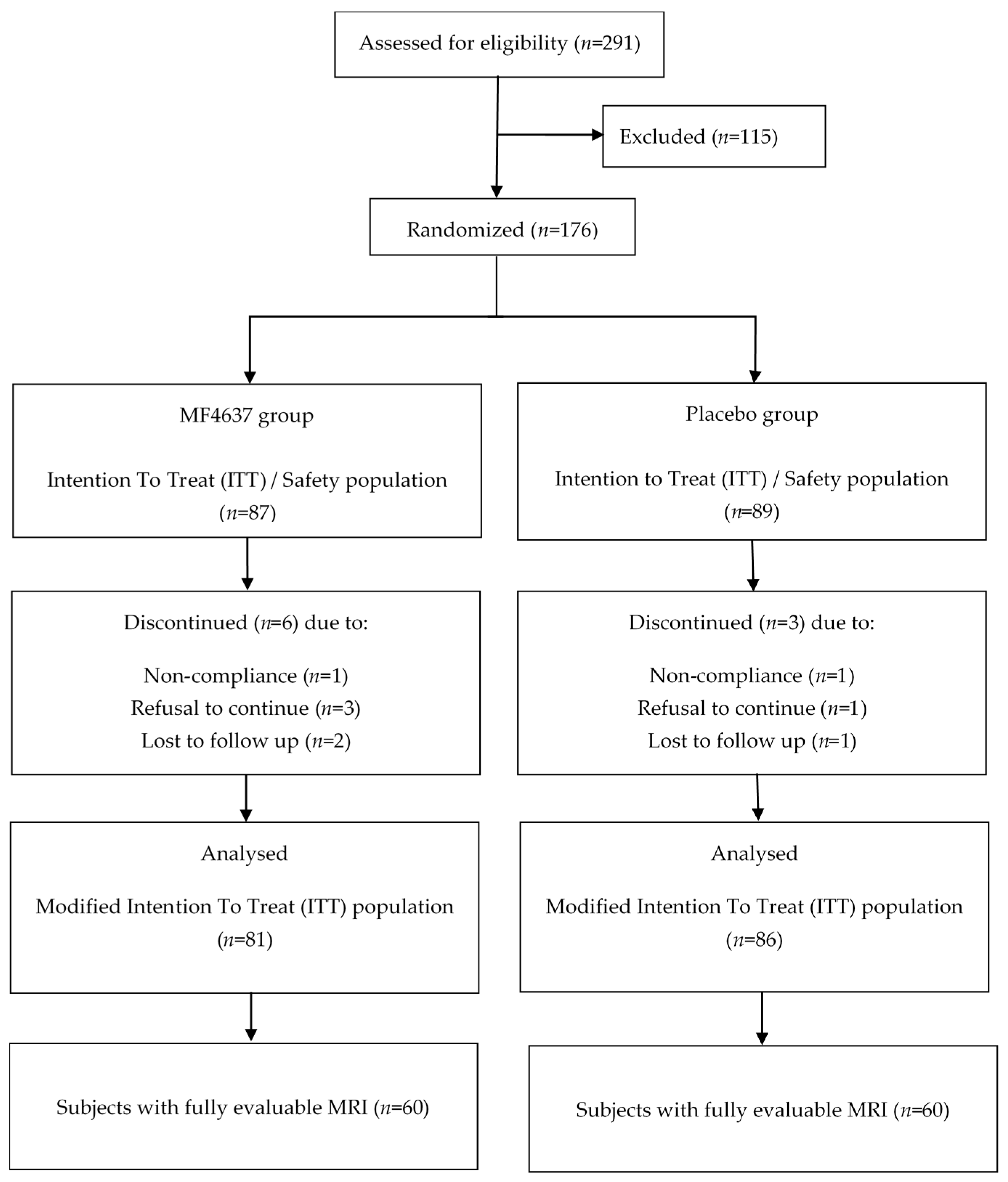
2.1. Study Design
2.2. Inclusion and Exclusion Criteria
2.3. Measurement of RBC EPA and DHA
2.3.1. Quantitative Measurement of RBC EPA and DHA
2.3.2. Qualitative Measurement of RBC EPA and DHA
2.4. Assessment of Change in Liver Fat
2.5. Calculation of Fatty Liver Index (FLI) Score
2.6. Sample Size Calculations
2.7. Statistical Analysis
3. Results
3.1. Effect of Intervention on Omega-3 Index
3.2. Effect of Intervention on RBC EPA + DHA, EPA and DHA Values
3.3. Effect of Intervention on RBC Omega-6: Omega-3 Ratio
3.4. Effect of Intervention on Liver Fat
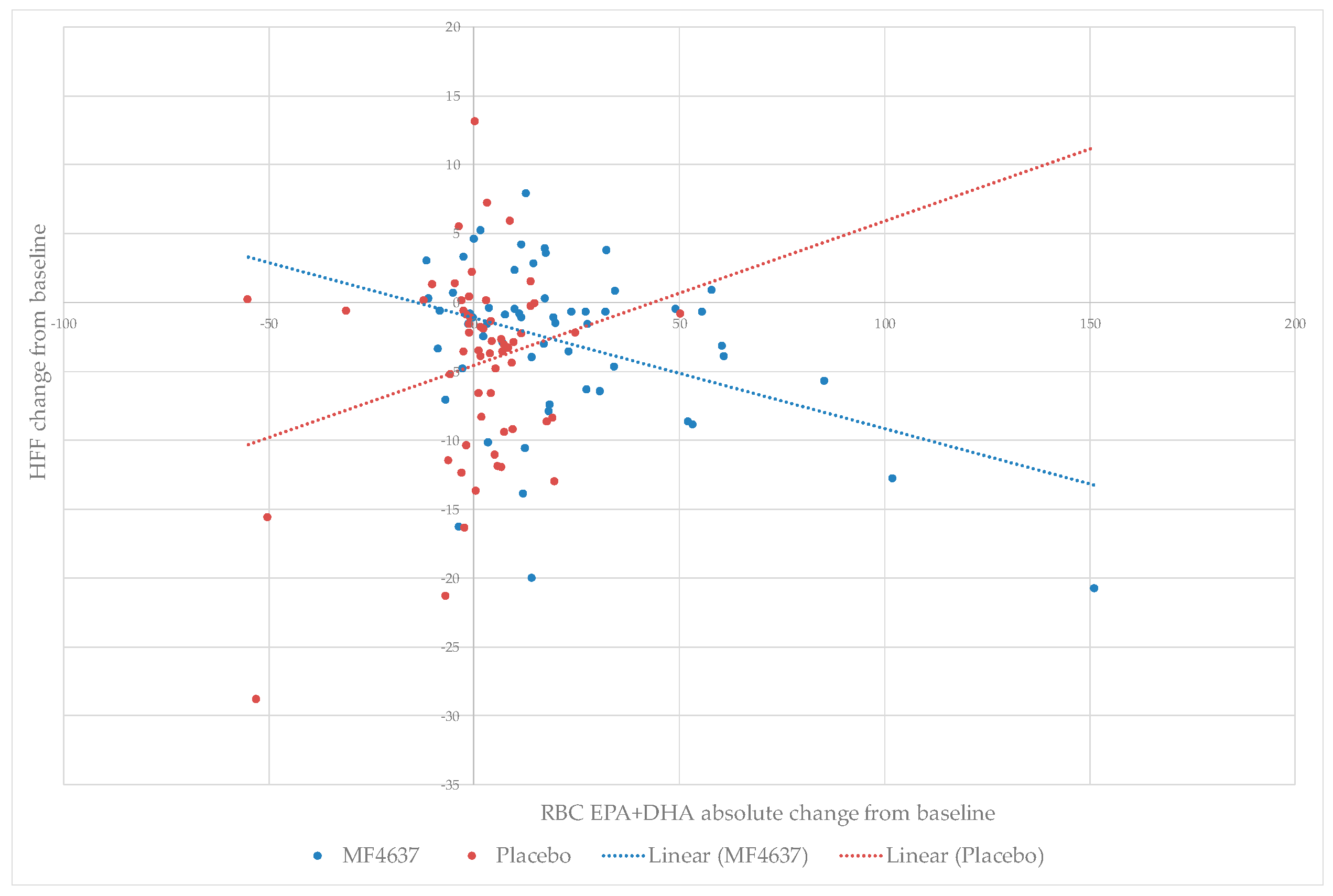
3.5. Relationship between RBC EPA + DHA Enrichment and Liver Fat Content
3.6. Relationship between Baseline Fatty Liver Index (FLI) and Change in Liver Fat Content
3.7. Relationship between RBC EPA + DHA Enrichment and Liver Enzymes AST and ALT
3.8. Effect of Intervention on Plasma Triglycerides
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The diagnosis and management of non-alcoholic fatty liver disease: Practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012, 55, 2005–2023. [Google Scholar] [CrossRef] [PubMed]
- Levene, A.P.; Goldin, R.D. The epidemiology, pathogenesis and histopathology of fatty liver disease. Histopathology 2012, 61, 141–152. [Google Scholar] [CrossRef] [PubMed]
- McCullough, A.J. The clinical features, diagnosis and natural history of nonalcoholic fatty liver disease. Clin. Liver Dis. 2004, 8, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Dowman, J.K.; Tomlinson, J.W.; Newsome, P.N. Pathogenesis of non-alcoholic fatty liver disease. Q. J. Med. 2010, 103, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Bedogni, G.; Miglioli, L.; Masutti, F.; Tiribelli, C.; Marchesini, G.; Bellentani, S. Prevalence of and risk factors for nonalcoholic fatty liver disease: The dionysos nutrition and liver study. Hepatology 2005, 42, 44–52. [Google Scholar] [CrossRef] [PubMed]
- LaBrecque, D.; Abbas, Z.; Anania, F.; Ferenci, P.; Khan, A.; Goh, K.; Hamid, S.; Isakov, V.; Lizarzabal, M.; Penaranda, M.; et al. World Gastroenterology Organisation Global Guidelines: Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. J. Clin. Gastroenterol. 2014, 48, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Neuschwander-Tetri, B.A.; Caldwell, S.H. Nonalcoholic steatohepatitis: Summary of an AASLD single topic conference. Hepatology 2003, 37, 1202–1219. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.; Marques-Vidal, P.; Cortez-Pinto, H. Hepatic histology in obese patients undergoing bariatric surgery. J. Hepatol. 2006, 45, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Bertolini, L.; Padovani, R.; Rodella, S.; Tessari, R.; Zenari, L.; Day, C.; Arcaro, G. Prevalence of nonalcoholic fatty liver disease and its association with cardiovascular disease among Type 2 diabetic patients. Diabetes Care 2007, 30, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Bedogni, G.; Bellentani, S.; Miglioli, L.; Masutti, F.; Passalacqua, M.; Castiglione, A.; Tiribelli, C. The Fatty Liver Index: A simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kotronen, A.; Yki-Järvinen, H. Fatty liver: A novel component of the metabolic syndrome. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Vuppalanchi, R.; Chalasani, N. Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: Selected practical issues in their evaluation and management. Hepatology 2009, 49, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Ekstedt, M.; Franzén, L.E.; Mathiesen, U.L.; Thorelius, L.; Holmqvist, M.; Bodemar, G.; Kechagias, S. Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology 2006, 44, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Chitturi, S.; Abeygunasekera, S.; Farrell, G. NASH and insulin resistance: Insulin hypersecretion and specific association with the insulin resistance syndrome. Hepatology 2002, 2, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Allard, J.P.; Aghdassi, E.; Mohammed, S.; Raman, M.; Avand, G.; Arendt, B.M.; Jalali, P.; Kandasamy, T.; Prayitno, N.; Sherman, M.; et al. Nutritional assessment and hepatic fatty acid composition in non-alcoholic fatty liver disease (NAFLD): A cross-sectional study. J. Hepatol. 2008, 48, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Araya, J.; Rodrigo, R.; Videla, L.; Thielemann, L.; Orellana, M.; Pettinelli, P.; Poniachik, J. Increase in long-chain polyunsaturated fatty acid n-6/n-3 ratio in relation to hepatic steatosis in patients with non-alcoholic fatty liver disease. Clin. Sci. 2004, 106, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Pettinelli, P.; del Pozo, T.; Araya, J.; Rodrigo, R.; Araya, A.V.; Smok, G.; Csendes, A.; Gutierrez, L.; Rojas, J.; Korn, O.; et al. Enhancement in liver SREBP-1c/PPAR-α ratio and steatosis in obese patients: Correlations with insulin resistance and n-3 long-chain polyunsaturated fatty acid depletion. Biochim. Biophys. Acta-Mol. Basis. Dis. 2009, 1792, 1080–1086. [Google Scholar] [CrossRef] [PubMed]
- Elizondo, A.; Araya, J.; Rodrigo, R.; Poniachik, J.; Csendes, A.; Maluenda, F.; Díaz, J.C.; Signorini, C.; Sgherri, C.; Comporti, M.; et al. A polyunsaturated fatty acid pattern in liver and erythrocyte phospholipids from obese patients. Obesity 2007, 15, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.; Veysey, M.; Lucock, M.; Niblett, S.; King, K.; Baines, S.; Garg, M.L. Association between erythrocyte omega-3 polyunsaturated fatty acid levels and fatty liver index in older people is sex dependent. J. Nutr. Intermed. Metab. 2016, 5, 78–85. [Google Scholar] [CrossRef]
- Toshimitsu, K.; Matsuura, B.; Ohkubo, I.; Niiya, T.; Furukawa, S.; Hiasa, Y.; Kawamura, M.; Ebihara, K.; Onji, M. Dietary habits and nutrient intake in non-alcoholic steatohepatitis. Nutrition 2007, 23, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Cortez-Pinto, H.; Jesus, L.; Barros, H.; Lopes, C.; Moura, M.C.; Camilo, M.E. How different is the dietary pattern in non-alcoholic steatohepatitis patients? Clin. Nutr. 2006, 25, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.D. Molecular mechanism for polyunsaturated fatty acid regulation of gene transcription. Am. J. Physiol. 2001, 281, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Bouzianas, D.G.; Bouziana, S.D.; Hatzitolios, A.I. Potential treatment of human nonalcoholic fatty liver disease with long-chain omega-3 polyunsaturated fatty acids. Nutr. Rev. 2013, 71, 753–771. [Google Scholar] [CrossRef] [PubMed]
- Hodson, L.; Bhatia, L.; Scorletti, E.; Smith, D.E.; Jackson, N.C.; Shojaee-Moradie, F.; Umpleby, M.; Calder, P.C.; Byrne, C.D. Docosahexaenoic acid enrichment in NAFLD is associated with improvements in hepatic metabolism and hepatic insulin sensitivity: A pilot study. Eur. J. Clin. Nutr. 2017, 71, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, Y.; Xu, Q.; Chen, S.-S. Omega-3 fatty acids as a treatment for non-alcoholic fatty liver disease in children: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. 2018, 37, 516–521. [Google Scholar] [CrossRef] [PubMed]
- De Castro, G.S.; Calder, P.C. Non-alcoholic fatty liver disease and its treatment with n-3 polyunsaturated fatty acids. Clin. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Musa-Veloso, K.; Venditti, C.; Lee, H.; Darch, M.; Floyd, S.; West, S.; Simon, R. Systematic review and meta-analysis of controlled intervention studies on the effectiveness of long chain omega-3 fatty acids in patients with non-alcoholic fatty liver disease. Nutr. Rev. 2018, 76, 581–602. [Google Scholar] [CrossRef] [PubMed]
- Katan, M.B.; Deslypere, J.P.; van Birgelen, A.P.; Penders, M.; Zegwaard, M. Kinetics of the incorporation of dietary fatty acids into serum cholesteryl esters, erythrocyte membranes, and adipose tissue: An 18-month controlled study. J. Lipid Res. 1997, 38, 2012–2022. [Google Scholar] [PubMed]
- Krauss, R.; Eckel, R.; Howard, B.; Appel, L.J.; Daniels, S.R.; Deckelbaum, R.J.; Erdman, J.W.; Kris-etherton, P.; Goldberg, I.J.; Kotchen, T.A.; et al. AHA dietary guidelines. Circulation 2000, 102, 2284–2299. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, M.; Ott, P.; Michaelsen, K.; Porsgaard, T.; Jensen, F.; Lanng, S. Long-chain PUFA in granulocytes, mononuclear cells, and RBC in patients with cystic fibrosis: Relation to liver disease. J. Pediatr. Gastroenterol. Nutr. 2012, 55, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Petit, J.M.; Guiu, B.; Duvillard, L.; Jooste, V.; Brindisi, M.C.; Athias, A.; Bouillet, B.; Habchi, M.; Cottet, V.; Gambert, P.; et al. Increased erythrocytes n-3 and n-6 polyunsaturated fatty acids is significantly associated with a lower prevalence of steatosis in patients with type 2 diabetes. Clin. Nutr. 2012, 31, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Arterburn, L.M.; Hall, E.B.; Oken, H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am. J. Clin. Nutr. 2006, 83, 1467S–1476S. [Google Scholar] [CrossRef] [PubMed]
- Tobin, D. BASF AS, Norway. CONDIN study. Unpublished work. 2018. [Google Scholar]
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.L.; Madden, J.; Gambell, J.M.; Young, S.; Wang, L.; Jebb, S.A.; Calder, P.C. Incorporation of eicosapentaenoic and docosahexaenoic acids into lipid pools when given as supplements providing doses equivalent to typical intakes of oily fish. Am. J. Clin. Nutr. 2012, 96, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Vuppalanchi, R.; Rinella, M.; Middleton, M.S.; Siddiqui, M.S.; Barritt, A.S.; Kolterman, O.; Flores, O.; Alonso, C.; Iruarrizaga-Lejarreta, M.; et al. Randomised clinical trial: A leucine-metformin-sildenafil combination (NS-0200) vs placebo in patients with non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2018, 47, 1639–1651. [Google Scholar] [CrossRef] [PubMed]
- He, X.-X.; Wu, X.-L.; Chen, R.-P.; Chen, C.; Liu, X.-G.; Wu, B.-J.; Huang, Z.-M. Effectiveness of omega-3 polyunsaturated fatty acids in non-alcoholic fatty liver disease: A meta-analysis of randomized controlled trials. PLoS One 2016, 11, e0162368. [Google Scholar] [CrossRef] [PubMed]
- Parker, H.M.; Johnson, N.A.; Burdon, C.A.; Cohn, J.S.; Connor, H.T.O.; George, J. Omega-3 supplementation and non-alcoholic fatty liver disease: A systematic review and meta-analysis. J. Hepatol. 2012, 56, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Capanni, M.; Calella, F.; Biagini, M.R.; Genise, S.; Raimondi, L.; Bedogni, G.; Svegliati-Baroni, G.; Sofi, F.; Milani, S.; Abbate, R.; et al. Prolonged n-3 polyunsaturated fatty acid supplementation ameliorates hepatic steatosis in patients with non-alcoholic fatty liver disease: A. pilot study. Aliment. Pharmacol. Ther. 2006, 23, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Zhou, Y.; Chen, S.H.; Zhao, X.L.; Ran, L.; Zeng, X.L.; Wu, Y.; Chen, J.L.; Kang, C.; Shu, F.R.; et al. Fish oil supplements lower serum lipids and glucose in correlation with a reduction in plasma fibroblast growth factor 21 and prostaglandin E2 in nonalcoholic fatty liver disease associated with hyperlipidemia: A randomized clinical trial. PLoS ONE 2015, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Scorletti, E.; Bhatia, L.; Mccormick, K.G.; Clough, G.F.; Nash, K.; Hodson, L.; Moyses, H.E.; Calder, P.C.; Byrne, C.D. Effects of purified eicosapentaenoic and docosahexaenoic acids in nonalcoholic fatty liver disease: Results from the WELCOME study. Hepatology 2014, 60, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Vega, G.; Chandalia, M.; Szczepaniak, L.; Grundy, S. Effects of N-3 fatty acids on hepatic triglyceride content in humans. J. Investig. Med. 2008, 56, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Dulai, P.; Sirlin, C.; Loomba, R. MRI and MRE for non-invasive quantitative assessment of hepatic steatosis and fibrosis in NAFLD and NASH: Clinical trials to clinical practice. J. Hepatol. 2016, 65, 1006–1016. [Google Scholar] [CrossRef] [PubMed]
| Variables | Placebo 1 | MF4637 2 | p-Value * |
|---|---|---|---|
| Age, years | 55.1 (10.9) | 55.3 (13.3) | 0.93 |
| Sex, M/F | 44/42 | 36/45 | 0.39 |
| Weight, kg | 90.1 (18.8) | 88.4 (18.4) | 0.55 |
| Waist circumference, cm | 105.9 (13.1) | 106.3 (13.2) | 0.87 |
| Hip circumference, cm | 110.5 (12.1) | 110.9 (11.8) | 0.85 |
| Waist to hip ratio | 0.96 (0.09) | 0.96 (0.08) | 0.95 |
| BMI, kg/m2 | 32.4 (5.0) | 32.1 (4.8) | 0.59 |
| Systolic blood pressure, mm Hg | 127.0 (10.7) | 128.0 (11.9) | 0.64 |
| Diastolic blood pressure, mm Hg | 80.3 (7.2) | 79.8 (7.6) | 0.33 |
| Heart rate, beats/min | 74.7 (8.7) | 73.2 (9.1) | 0.25 |
| Statin use, % | 34.9 | 30.9 | 0.58 |
| Diabetes, % | 39.5 | 35.0 | 0.97 |
| Fasting glucose, mg/dL | 120.1 (48.5) 2 | 119.4 (38.1) 3 | 0.97 |
| Fasting insulin, µIU/mL | 30.2 (41.3) | 20.8 (18.2) 4 | 0.04 |
| HbA1c, % | 6.5 (1.5) 5 | 6.3 (1.4) 6 | 0.20 |
| Triglycerides, mg/dL | 199.1 (123.0) 7 | 192.0 (125.1) 4 | 0.70 |
| BUN, mg/dL | 14.7 (4.9) 7 | 15.4 (5.0) 4 | 0.24 |
| Creatinine, mg/dL | 0.8 (0.2) 7 | 0.3 (0.2) 4 | 0.83 |
| TSH µIU/mL | 1.9 (1.0) 7 | 1.7 (0.9) 8 | 0.17 |
| Hs-CRP, mg/L | 6.4 (9.2) 7 | 8.1 (17.5) | 0.61 |
| Albumin, g/dL | 4.29 (0.3) 7 | 4.29 (0.3) 4 | 0.91 |
| ALT, IU/L | 35.6 (24.0) | 37.5 (39.0) | 0.40 |
| AST, IU/L | 25.8 (12.2) | 27.1 (20.3) | 0.79 |
| ALP, IU/L | 81.5 (31.0) | 85.5 (45.3) | 0.62 |
| GGT, IU/L | 47.1 (49.0) | 62.2 (151.4) | 0.78 |
| Bilirubin, mg/dL | 0.5 (0.2) | 0.5 (0.3) | 0.91 |
| Variables | Placebo 1 | MF4637 2 | Two-Sample Test for Change from Baseline p-Value # | ||||
|---|---|---|---|---|---|---|---|
| Baseline | End of Study | p-Value * | Baseline | End of Study | p-Value * | ||
| Age, years | 55.1 (10.9) | 55.3 (13.3) | |||||
| Sex, M/F | 44/42 | 36/45 | |||||
| Weight, kg | 90.1 (18.8) | 89.5 (18.5) | 0.84 | 88.4 (18.4) | 89.0 (18.8) | 0.082 | 0.23 |
| Waist circumference, cm | 105.9 (13.1) | 104.7 (13.9) | 0.005 | 106.3 (13.2) | 105.5 (12.2) | 0.72 | 0.18 |
| Hip circumference, cm | 110.5 (12.1) | 110.0 (11.7) | 0.31 | 110.9 (11.8) | 110.4 (11.9) | 0.81 | 0.42 |
| Waist to hip ratio | 0.96 (0.1) | 0.95 (0.1) | 0.081 | 0.96 (0.08) | 0.96 (0.1) | 0.66 | 0.58 |
| BMI, kg/m2 | 32.4 (5.0) | 32.3 (4.8) | 0.74 | 32.1 (4.8) | 32.3 (5.0) | 0.078 | 0.18 |
| SBP, mm Hg | 127.0 (10.7) | 128.8 (15.0) | 0.23 | 128.0 (11.9) | 126.6 (10.6) | 0.29 | 0.11 |
| DBP, mm Hg | 80.3 (7.2) | 79.9 (9.9) | 0.25 | 79.8 (7.6) | 77.8 (8.4) | 0.032 | 0.53 |
| Heart rate, beats/min | 74.7 (8.7) | 74.1 (8.2) | 0.59 | 73.2 (9.1) | 74.2 (9.8) | 0.17 | 0.17 |
| Statin use, % | 34.9 | 30.9 | |||||
| Diabetes, % | 39.5 | 35.0 | |||||
| Fasting glucose, mg/dL | 120.1 (48.5) 3 | 125.4 (56.8) 3 | 0.39 | 119.4 (38.1) 3 | 127.5 (55.2) 3 | 0.0616 | 0.38 |
| Fasting insulin, µIU/mL | 30.2 (41.3) 4 | 30.1 (35.8) 4 | 0.51 | 20.8 (18.2) 5 | 24.0 (24.3) 5 | 0.21 | 0.63 |
| HbA1c, % | 6.5 (1.5) 6 | 6.6 (1.7) 6 | 0.67 | 6.3 (1.4) 7 | 6.3 (1.4) 7 | 0.83 | 0.87 |
| Triglycerides, mg/dL | 199.1 (123.0) 2 | 185.7 (118.0) 2 | 0.52 | 192.0 (125.1) 5 | 157.8 (84.2) 5 | 0.0008 | 0.053 |
| BUN, mg/dL | 14.7 (4.9) 2 | 15.0 (4.5) 2 | 0.88 | 15.4 (5.0) 5 | 16.2 (4.9) 5 | 0.25 | 0.46 |
| Creatinine, mg/dL | 0.8 (0.2) 2 | 0.82 (0.2) 2 | 0.51 | 0.3 (0.2) 5 | 0.84 (0.2) 5 | 0.26 | 0.19 |
| TSH µIU/mL | 1.9 (1.0) 2 | 2.5 (2.7) 2 | 0.025 | 1.7 (0.9) 7 | 1.8 (0.9) 7 | 0.043 | 0.86 |
| Hs-CRP, mg/L | 6.4 (9.2) 2 | 5.5 (5.8) 2 | 0.46 | 8.1 (17.5) 8 | 6.7 (10.9) 8 | 0.75 | 0.82 |
| Albumin, g/dL | 4.29 (0.3) 2 | 4.3 (0.3) 2 | 0.69 | 4.29 (0.3) 5 | 4.3 (0.3) 5 | 0.62 | 0.91 |
| ALT, IU/L | 35.6 (24.0) | 29.8 (21.2) | 0.005 | 37.5 (39.0) | 38.1 (37.7) | 0.48 | 0.015 |
| AST, IU/L | 25.8 (12.2) | 23.9 (13.5) | 0.036 | 27.1 (20.3) | 28.7 (24.6) | 0.37 | 0.036 |
| ALP, IU/L | 81.5 (31.0) | 76.3 (21.5) | 0.01 | 85.5 (45.3) | 83.2 (40.3) | 0.09 | 0.73 |
| GGT, IU/L | 47.1 (49.0) | 37.1 (32.2) | <.0001 | 62.2 (151.4) | 57.1 (108.7) | 0.37 | 0.058 |
| Bilirubin, mg/dL | 0.5 (0.2) | 0.47 (0.2) | 0.70 | 0.5 (0.3) | 0.5 (0.2) | 0.88 | 0.72 |
| Placebo (n = 86) | MF4637 (n = 81) | p-Value 1 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | T = 12 weeks | T = 24 weeks | Change from Baseline | Baseline | T = 12 weeks | T = 24 weeks | Change from Baseline | ||
| RBC omega-3 index, % | 4.9 (1.2) | 5.8 (1.3) | 5.3 (1.1) | 0.4 (1.0) | 4.8 (1.1) | 8.7 (2.3) | 8.0 (2.6) | 3.2 (2.7) | <0.0001 |
| RBC EPA + DHA, µg/mL | 32.3 (26.4) | 34.9 (21.0) | 33.1 (20.5) | 1.2 (14.9) | 29.6 (17.5) | 51.5 (38.9) | 52.9 (40.7) | 21.2 (28.7) | <0.0001 |
| RBC EPA, % | 0.54 (0.3) | 0.54 (0.2) | 0.54 (0.2) | 0.002 (0.3) | 0.53 (0.2) | 1.6 (0.9) | 1.4 (0.9) | 0.9 (1.0) | <0.0001 |
| RBC EPA, µg/mL | 3.8 (6.6) | 3.7 (2.6) | 4.1 (5.7) | 0.4 (7.0) | 3.0 (2.6) | 10.4 (10.7) | 10.6 (12.0) | 7.1 (10.5) | <0.0001 |
| RBC DHA, % | 4.3 (1.1) | 5.2 (1.2) | 4.8 (1.0) | 0.4 (0.9) | 4.3 (1.0) | 7.1 (1.5) | 6.6 (1.8) | 2.3 (1.9) | <0.0001 |
| RBC DHA, µg/mL | 28.5 (21.0) | 31.2 (18.6) | 29.0 (16.2) | 0.7 (10.7) | 26.6 (15.3) | 41.0 (28.8) | 42.4 (29.8) | 14.1 (19.5) | <0.0001 |
| RBC omega-6: omega-3 | 4.9 (1.1) | 4.5 (0.9) | 4.7 (0.8) | −0.2 (0.7) | 4.9 (1.2) | 3.0 (0.9) | 3.3 (1.5) | −1.6 (1.8) | <0.0001 |
| Placebo 1 | MF4637 1 | p-Value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| T = 0 weeks | T = 24 weeks | Change from Baseline | T = 0 weeks | T = 24 weeks | Change from Baseline | ||||
| Absolute | Relative | Absolute | Relative | ||||||
| Liver fat, % | 17.4 (10.4) | 12.6 (8.0) | −4.4 (6.9) | −27.6 | 14.4 (10.1) | 10.7 (7.6) | −2.8 (5.8) | −25.7 | 0.1838 |
| Baseline HFF Mean (SD) | Study End HFF Mean (SD) | Change in MRI-PDFF Liver Fat Percentage (%) | |||||
|---|---|---|---|---|---|---|---|
| Absolute Change | p-Value 1 | Relative Change | p-Value 1 | ||||
| FLI <30 2 | Placebo | 15.6 (11.2) | 11.2 (7.8) | 2.25 (1.39) | 0.11 | 2.7 (11.3) | 0.81 |
| Omega-3 | 12.5 (8.6) | 10.3 (7.7) | |||||
| FLI ≥30 3 | Placebo | 20.6 (9.4) | 16.0 (7.9) | −2.47 (2.53) | 0.34 | −9.3 (15.3) | 0.55 |
| Omega-3 | 19.3 (11.1) | 13.5 (7.0) | |||||
| FLI ≥40 4 | Placebo | 20.2 (9.5) | 16.3 (8.4) | −7.45 (2.81) | 0.02 | −44.1 (14.6) | 0.009 |
| Omega-3 | 20.7 (10.4) | 10.6 (5.2) | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tobin, D.; Brevik-Andersen, M.; Qin, Y.; Innes, J.K.; Calder, P.C. Evaluation of a High Concentrate Omega-3 for Correcting the Omega-3 Fatty Acid Nutritional Deficiency in Non-Alcoholic Fatty Liver Disease (CONDIN). Nutrients 2018, 10, 1126. https://doi.org/10.3390/nu10081126
Tobin D, Brevik-Andersen M, Qin Y, Innes JK, Calder PC. Evaluation of a High Concentrate Omega-3 for Correcting the Omega-3 Fatty Acid Nutritional Deficiency in Non-Alcoholic Fatty Liver Disease (CONDIN). Nutrients. 2018; 10(8):1126. https://doi.org/10.3390/nu10081126
Chicago/Turabian StyleTobin, Derek, Merethe Brevik-Andersen, Yan Qin, Jacqueline K. Innes, and Philip C. Calder. 2018. "Evaluation of a High Concentrate Omega-3 for Correcting the Omega-3 Fatty Acid Nutritional Deficiency in Non-Alcoholic Fatty Liver Disease (CONDIN)" Nutrients 10, no. 8: 1126. https://doi.org/10.3390/nu10081126
APA StyleTobin, D., Brevik-Andersen, M., Qin, Y., Innes, J. K., & Calder, P. C. (2018). Evaluation of a High Concentrate Omega-3 for Correcting the Omega-3 Fatty Acid Nutritional Deficiency in Non-Alcoholic Fatty Liver Disease (CONDIN). Nutrients, 10(8), 1126. https://doi.org/10.3390/nu10081126





