Effects of Adherence to a Higher Protein Diet on Weight Loss, Markers of Health, and Functional Capacity in Older Women Participating in a Resistance-Based Exercise Program
Abstract
1. Introduction
2. Methods
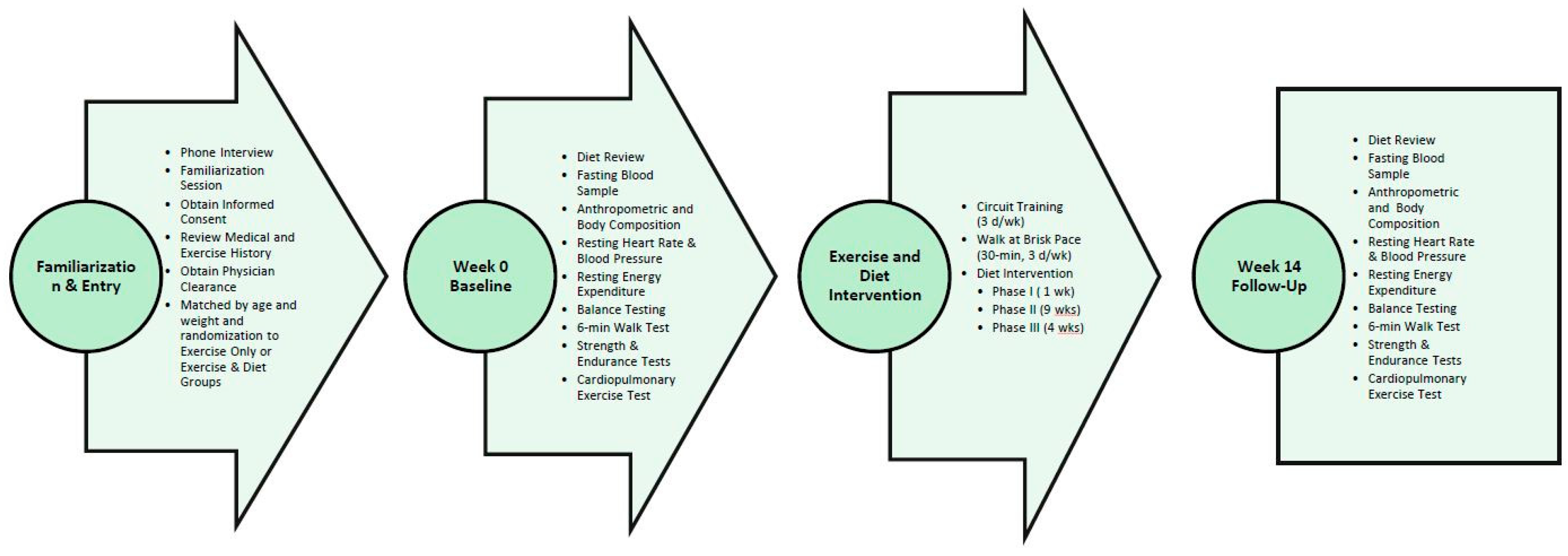
2.1. Experimental Design
2.2. Participants
2.3. Testing Sequence
2.4. Diet Intervention
2.5. Training Protocol
3. Procedures
3.1. Dietary Assessment
3.2. Resting Energy Expenditure and Metabolism
3.3. Anthropometric Measures and Body Composition
3.4. Exercise and Functional Capacity
3.5. Blood Collection and Analysis
3.6. Statistical Analysis
4. Results
4.1. Participant Demographics
4.2. Energy and Macronutrient Intake
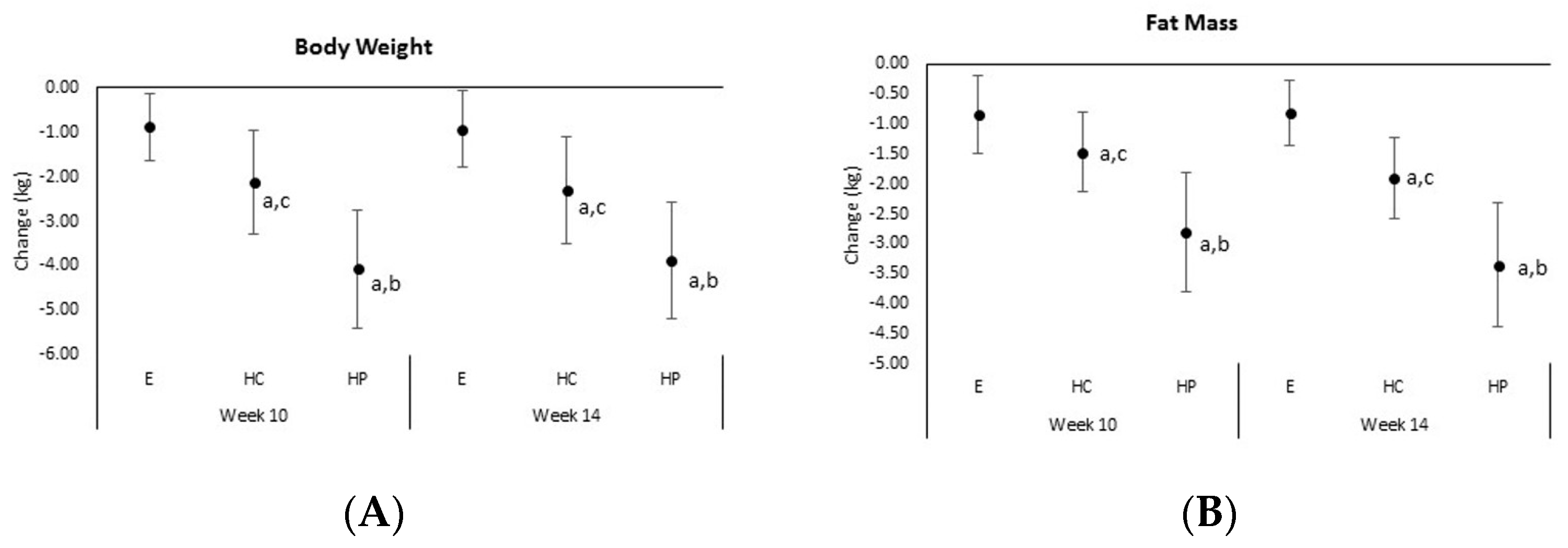
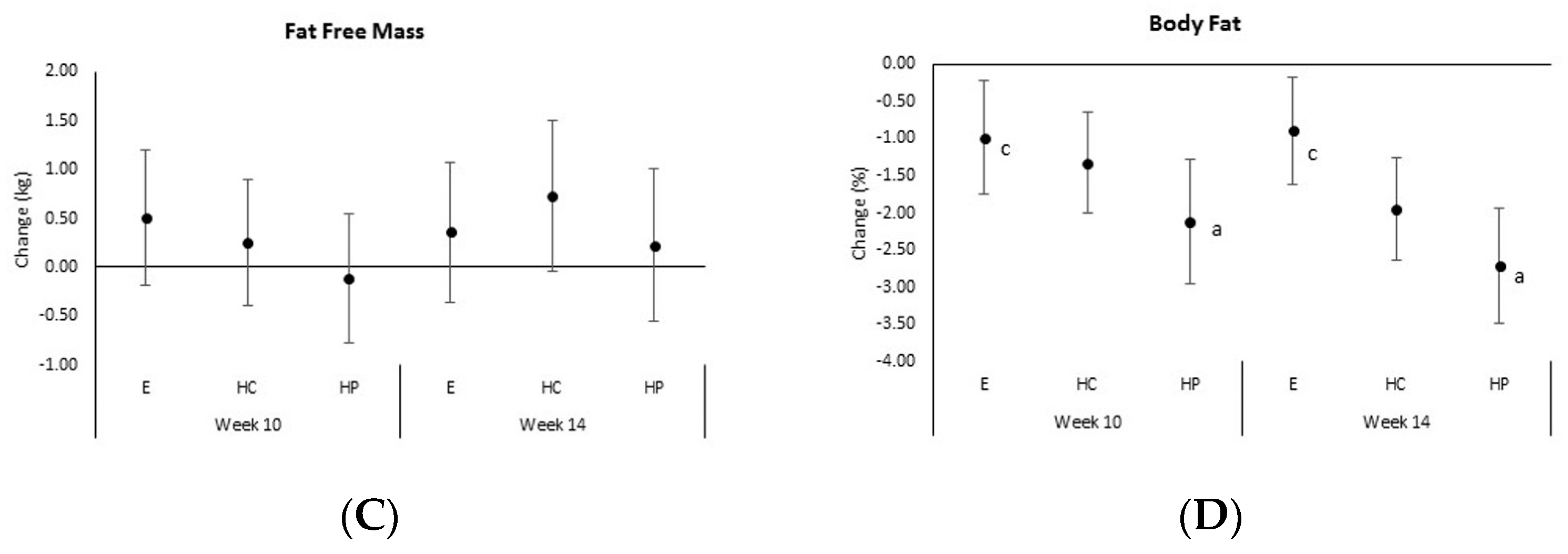
4.3. Anthropometrics, Body Composition, and Resting Energy Expenditure
4.4. Hematological Markers
4.5. Exercise and Functional Capacity
5. Discussion and Conclusions
5.1. Weight Loss
5.2. Markers of Health
5.3. Functional Capacity
5.4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Wilborn, C.; Beckham, J.; Campbell, B.; Harvey, T.; Galbreath, M.; La Bounty, P.; Nassar, E.; Wismann, J.; Kreider, R. Obesity: Prevalence, theories, medical consequences, management, and research directions. J. Int. Soc. Sports Nutr. 2005, 2, 4–31. [Google Scholar] [CrossRef] [PubMed]
- Buch, A.; Carmeli, E.; Boker, L.K.; Marcus, Y.; Shefer, G.; Kis, O.; Berner, Y.; Stern, N. Muscle function and fat content in relation to sarcopenia, obesity and frailty of old age—An overview. Exp. Gerontol. 2016, 76, 25–32. [Google Scholar] [CrossRef] [PubMed]
- McCullough, P.A.; Fallahzadeh, M.K.; Hegazi, R.M. Nutritional deficiencies and sarcopenia in heart failure: A therapeutic opportunity to reduce hospitalization and death. Rev. Cardiovasc. Med. 2016, 17 (Suppl. 1), S30–S39. [Google Scholar]
- Oh, C.; Jeon, B.H.; Reid Storm, S.N.; Jho, S.; No, J.K. The most effective factors to offset sarcopenia and obesity in the older korean: Physical activity, vitamin d, and protein intake. Nutrition 2017, 33, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Hamer, M.; O’Donovan, G. Sarcopenic obesity, weight loss, and mortality: The english longitudinal study of ageing. Am. J. Clin. Nutr. 2017, 106, 125–129. [Google Scholar] [CrossRef] [PubMed]
- JafariNasabian, P.; Inglis, J.E.; Kelly, O.J.; Ilich, J.Z. Osteosarcopenic obesity in women: Impact, prevalence, and management challenges. Int. J. Womens Health 2017, 9, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Perna, S.; Peroni, G.; Faliva, M.A.; Bartolo, A.; Naso, M.; Miccono, A.; Rondanelli, M. Sarcopenia and sarcopenic obesity in comparison: Prevalence, metabolic profile, and key differences. A cross-sectional study in italian hospitalized elderly. Aging Clin. Exp. Res. 2017, 29, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Lou, N.; Chi, C.H.; Chen, X.D.; Zhou, C.J.; Wang, S.L.; Zhuang, C.L.; Shen, X. Sarcopenia in overweight and obese patients is a predictive factor for postoperative complication in gastric cancer: A prospective study. Eur. J. Surg. Oncol. 2017, 43, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Hwang, S.J.; McMahon, G.M.; Curhan, G.C.; McLean, R.R.; Murabito, J.M.; Fox, C.S. Mid-adulthood cardiometabolic risk factor profiles of sarcopenic obesity. Obesity (Silver Spring) 2016, 24, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Poggiogalle, E.; Lubrano, C.; Sergi, G.; Coin, A.; Gnessi, L.; Mariani, S.; Lenzi, A.; Donini, L.M. Sarcopenic obesity and metabolic syndrome in adult caucasian subjects. J. Nutr. Health Aging 2016, 20, 958–963. [Google Scholar] [CrossRef] [PubMed]
- Rier, H.N.; Jager, A.; Sleijfer, S.; van Rosmalen, J.; Kock, M.; Levin, M.D. Low muscle attenuation is a prognostic factor for survival in metastatic breast cancer patients treated with first line palliative chemotherapy. Breast 2017, 31, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Yadigar, S.; Yavuzer, H.; Yavuzer, S.; Cengiz, M.; Yuruyen, M.; Doventas, A.; Erdincler, D.S. Primary sarcopenia in older people with normal nutrition. J. Nutr. Health Aging 2016, 20, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Badley, E.M.; Canizares, M.; Perruccio, A.V.; Hogg-Johnson, S.; Gignac, M.A. Benefits gained, benefits lost: Comparing baby boomers to other generations in a longitudinal cohort study of self-rated health. Milbank Q. 2015, 93, 40–72. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, P.B. High and rising health care costs. Synth. Proj. Res. Synth. Rep. 2008, 16. [Google Scholar]
- Whelan, D.M.; Roy, R.C. Diseases of aging that emerge from the metabolic syndrome. Anesthesiol. Clin. 2006, 24, 599–619. [Google Scholar] [CrossRef] [PubMed]
- Fakhouri, T.H.; Ogden, C.L.; Carroll, M.D.; Kit, B.K.; Flegal, K.M. Prevalence of obesity among older adults in the united states, 2007–2010. NCHS Data Brief 2012, 106, 1–8. [Google Scholar]
- Osteoporosis—Prevention, Diagnosis and Treatment: A Systematic Review. Available online: https://www.ncbi.nlm.nih.gov/pubmed/28876760 (accessed on 11 August 2018).
- Baccaro, L.F.; Conde, D.M.; Costa-Paiva, L.; Pinto-Neto, A.M. The epidemiology and management of postmenopausal osteoporosis: A viewpoint from Brazil. Clin. Interv. Aging 2015, 10, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Borst, S.E. Interventions for sarcopenia and muscle weakness in older people. Age Ageing 2004, 33, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Hunter, G.R.; McCarthy, J.P.; Bamman, M.M. Effects of resistance training on older adults. Sports Med. 2004, 34, 329–348. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, E.; Calvani, R.; Tosato, M.; Cesari, M.; Di Bari, M.; Cherubini, A.; Broccatelli, M.; Savera, G.; D’Elia, M.; Pahor, M.; et al. Physical activity and exercise as countermeasures to physical frailty and sarcopenia. Aging Clin. Exp. Res. 2017, 29, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Montero-Fernandez, N.; Serra-Rexach, J.A. Role of exercise on sarcopenia in the elderly. Eur. J. Phys. Rehabil. Med. 2013, 49, 131–143. [Google Scholar] [PubMed]
- American College of Sports, M.; Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. American college of sports medicine position stand. Exercise and physical activity for older adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar]
- De Souto Barreto, P.; Morley, J.E.; Chodzko-Zajko, W.; Pitkala, K.H.; Weening-Djiksterhuis, E.; Rodriguez-Manas, L.; Barbagallo, M.; Rosendahl, E.; Sinclair, A.; Landi, F.; et al. Recommendations on physical activity and exercise for older adults living in long-term care facilities: A taskforce report. J. Am. Med. Dir. Assoc. 2016, 17, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, E.; Miller, M.; Yaxley, A.; Isenring, E. Malnutrition in the elderly: A narrative review. Maturitas 2013, 76, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Boirie, Y.; Morio, B.; Caumon, E.; Cano, N.J. Nutrition and protein energy homeostasis in elderly. Mech. Ageing Dev. 2014, 136–137, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Morais, J.A.; Chevalier, S.; Gougeon, R. Protein turnover and requirements in the healthy and frail elderly. J. Nutr. Health Aging 2006, 10, 272–283. [Google Scholar] [PubMed]
- Nowson, C.; O’Connell, S. Protein requirements and recommendations for older people: A review. Nutrients 2015, 7, 6874–6899. [Google Scholar] [CrossRef] [PubMed]
- Baetge, C.; Earnest, C.P.; Lockard, B.; Coletta, A.M.; Galvan, E.; Rasmussen, C.; Levers, K.; Simbo, S.Y.; Jung, Y.P.; Koozehchian, M.; et al. Efficacy of a randomized trial examining commercial weight loss programs and exercise on metabolic syndrome in overweight and obese women. Appl. Physiol. Nutr. Metab. 2017, 42, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Kerksick, C.; Thomas, A.; Campbell, B.; Taylor, L.; Wilborn, C.; Marcello, B.; Roberts, M.; Pfau, E.; Grimstvedt, M.; Opusunju, J.; et al. Effects of a popular exercise and weight loss program on weight loss, body composition, energy expenditure and health in obese women. Nutr. Metab. (Lond.) 2009, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Kerksick, C.M.; Wismann-Bunn, J.; Fogt, D.; Thomas, A.R.; Taylor, L.; Campbell, B.I.; Wilborn, C.D.; Harvey, T.; Roberts, M.D.; La Bounty, P.; et al. Changes in weight loss, body composition and cardiovascular disease risk after altering macronutrient distributions during a regular exercise program in obese women. Nutr. J. 2010, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Rasmussen, C.; Kerksick, C.M.; Wilborn, C.; Taylor, L.T.; Campbell, B.; Magrans-Courtney, T.; Fogt, D.; Ferreira, M.; Li, R.; et al. A carbohydrate-restricted diet during resistance training promotes more favorable changes in body composition and markers of health in obese women with and without insulin resistance. Phys. Sportsmed. 2011, 39, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Serra, M.; Beavers, K.M.; Moreillon, J.; Kresta, J.Y.; Byrd, M.; Oliver, J.M.; Gutierrez, J.; Hudson, G.; Deike, E.; et al. A structured diet and exercise program promotes favorable changes in weight loss, body composition, and weight maintenance. J. Am. Diet Assoc. 2011, 111, 828–843. [Google Scholar] [CrossRef] [PubMed]
- Lockard, B.; Earnest, C.P.; Oliver, J.; Goodenough, C.; Rasmussen, C.; Greenwood, M.; Kreider, R.B. Retrospective analysis of protein- and carbohydrate-focused diets combined with exercise on metabolic syndrome prevalence in overweight and obese women. Metab. Syndr. Relat. Disord. 2016, 14, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Magrans-Courtney, T.; Wilborn, C.; Rasmussen, C.; Ferreira, M.; Greenwood, L.; Campbell, B.; Kerksick, C.M.; Nassar, E.; Li, R.; Iosia, M.; et al. Effects of diet type and supplementation of glucosamine, chondroitin, and msm on body composition, functional status, and markers of health in women with knee osteoarthritis initiating a resistance-based exercise and weight loss program. J. Int. Soc. Sports Nutr. 2011, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Farris, G.; Wismann, J.; Farris, R.; Gandy, N.; Long, L.; Pfau, E.; Campbell, B.; La Bounty, P.; Rasmussen, C.; Wilson, R.; et al. Exercise intensity and energy expenditure analysis of women participating in the curves® exercise program. FASEB J. 2006, LB, 93–94. [Google Scholar]
- Kreider, R.; Parker, A.; Moreillon, J.; Rasmussen, C.; Greenwood, M. Energy expenditure analysis of women participating in a computerized hydraulic circuit training program. J. Strength Cond. Res. 2008, 22, A69–A70. [Google Scholar]
- La Bounty, P.; Wilborn, C.; Marcello, B.; Campbell, B.; Faries, M.; Shim, J.; Rasmussen, C.; Kreider, R. Analysis of exercise intensities of women using the curves® hydraulic training equipment. FASEB J. 2006, LB, 93. [Google Scholar]
- Matarese, L.E. Indirect calorimetry: Technical aspects. J. Am. Diet Assoc. 1997, 97, S154–S160. [Google Scholar] [CrossRef]
- Acsm’s Guidelines for Exercise Testing and Prescription, 6th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2000.
- Almada, A.L.; Kreider, R.B.; Ransom, J.; Rasmussen, C. Comparison of the reliability of repeated whole body dexa scans to repeated spine and hip scans. J. Bone. Miner. Res. 1999, 14, S369. [Google Scholar]
- Steffen, T.; Seney, M. Test-retest reliability and minimal detectable change on balance and ambulation tests, the 36-item short-form health survey, and the unified parkinson disease rating scale in people with parkinsonism. Phys. Ther. 2008, 88, 733–746. [Google Scholar] [CrossRef] [PubMed]
- Steffen, T.M.; Hacker, T.A.; Mollinger, L. Age- and gender-related test performance in community-dwelling elderly people: Six-minute walk test, berg balance scale, timed up & go test, and gait speeds. Phys. Ther. 2002, 82, 128–137. [Google Scholar] [PubMed]
- Alahmari, K.A.; Marchetti, G.F.; Sparto, P.J.; Furman, J.M.; Whitney, S.L. Estimating postural control with the balance rehabilitation unit: Measurement consistency, accuracy, validity, and comparison with dynamic posturography. Arch. Phys. Med. Rehabil. 2014, 95, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Carter, N.D.; Khan, K.M.; McKay, H.A.; Petit, M.A.; Waterman, C.; Heinonen, A.; Janssen, P.A.; Donaldson, M.G.; Mallinson, A.; Riddell, L.; et al. Community-based exercise program reduces risk factors for falls in 65- to 75-year-old women with osteoporosis: Randomized controlled trial. CMAJ 2002, 167, 997–1004. [Google Scholar] [PubMed]
- Cuka, S.; Dvornik, S.; Drazenovic, K.; Mihic, J. Evaluation of the dade behring dimension rxl clinical chemistry analyzer. Clin Lab 2001, 47, 35–40. [Google Scholar] [PubMed]
- McAuley, K.A.; Williams, S.M.; Mann, J.I.; Walker, R.J.; Lewis-Barned, N.J.; Temple, L.A.; Duncan, A.W. Diagnosing insulin resistance in the general population. Diabetes Care 2001, 24, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Page, P. Beyond statistical significance: Clinical interpretation of rehabilitation research literature. Int. J. Sports Phys. Ther. 2014, 9, 726–736. [Google Scholar] [PubMed]
- Mitchell, C.J.; Milan, A.M.; Mitchell, S.M.; Zeng, N.; Ramzan, F.; Sharma, P.; Knowles, S.O.; Roy, N.C.; Sjodin, A.; Wagner, K.H.; et al. The effects of dietary protein intake on appendicular lean mass and muscle function in elderly men: A 10-wk randomized controlled trial. Am. J. Clin. Nutr. 2017, 106, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Churchward-Venne, T.A.; Tieland, M.; Verdijk, L.B.; Leenders, M.; Dirks, M.L.; de Groot, L.C.; van Loon, L.J. There are no nonresponders to resistance-type exercise training in older men and women. J. Am. Med. Dir. Assoc. 2015, 16, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Jager, R.; Kerksick, C.M.; Campbell, B.I.; Cribb, P.J.; Wells, S.D.; Skwiat, T.M.; Purpura, M.; Ziegenfuss, T.N.; Ferrando, A.A.; Arent, S.M.; et al. International society of sports nutrition position stand: Protein and exercise. J. Int. Soc. Sports Nutr. 2017, 14, 20. [Google Scholar] [CrossRef] [PubMed]
- Pesta, D.H.; Samuel, V.T. A high-protein diet for reducing body fat: Mechanisms and possible caveats. Nutr. Metab. (Lond.) 2014, 11, 53. [Google Scholar] [CrossRef] [PubMed]
- Koopman, R. Dietary protein and exercise training in ageing. Proc. Nutr. Soc. 2011, 70, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Benton, M.J.; Whyte, M.D.; Dyal, B.W. Sarcopenic obesity: Strategies for management. Am. J. Nurs. 2011, 111, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Mateo-Gallego, R.; Marco-Benedi, V.; Perez-Calahorra, S.; Bea, A.M.; Baila-Rueda, L.; Lamiquiz-Moneo, I.; de Castro-Oros, I.; Cenarro, A.; Civeira, F. Energy-restricted, high-protein diets more effectively impact cardiometabolic profile in overweight and obese women than lower-protein diets. Clin. Nutr. 2017, 36, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Lee, J.; Bae, W.K.; Choi, J.K.; Kim, H.J.; Cho, B. Efficacy of low-calorie, partial meal replacement diet plans on weight and abdominal fat in obese subjects with metabolic syndrome: A double-blind, randomised controlled trial of two diet plans - one high in protein and one nutritionally balanced. Int. J. Clin. Pract. 2009, 63, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Maltais, M.L.; Perreault, K.; Courchesne-Loyer, A.; Lagace, J.C.; Barsalani, R.; Dionne, I.J. Effect of resistance training and various sources of protein supplementation on body fat mass and metabolic profile in sarcopenic overweight older adult men: A pilot study. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Muller, M.J.; Enderle, J.; Pourhassan, M.; Braun, W.; Eggeling, B.; Lagerpusch, M.; Gluer, C.C.; Kehayias, J.J.; Kiosz, D.; Bosy-Westphal, A. Metabolic adaptation to caloric restriction and subsequent refeeding: The minnesota starvation experiment revisited. Am. J. Clin. Nutr. 2015, 102, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Trepanowski, J.F.; Kroeger, C.M.; Barnosky, A.; Klempel, M.C.; Bhutani, S.; Hoddy, K.K.; Gabel, K.; Freels, S.; Rigdon, J.; Rood, J.; et al. Effect of alternate-day fasting on weight loss, weight maintenance, and cardioprotection among metabolically healthy obese adults: A randomized clinical trial. JAMA Int. Med. 2017, 177, 930–938. [Google Scholar] [CrossRef] [PubMed]
- Malin, S.K.; Niemi, N.; Solomon, T.P.; Haus, J.M.; Kelly, K.R.; Filion, J.; Rocco, M.; Kashyap, S.R.; Barkoukis, H.; Kirwan, J.P. Exercise training with weight loss and either a high- or low-glycemic index diet reduces metabolic syndrome severity in older adults. Ann. Nutr. Metab. 2012, 61, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M.; Keogh, J.B.; Noakes, M. Long-term effects of a high-protein weight-loss diet. Am. J. Clin. Nutr. 2008, 87, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, M.F.; Ritz, C.; Blaak, E.E.; Saris, W.H.; Langin, D.; Poulsen, S.K.; Larsen, T.M.; Sorensen, T.I.; Zohar, Y.; Astrup, A. Pretreatment fasting plasma glucose and insulin modify dietary weight loss success: Results from 3 randomized clinical trials. Am. J. Clin. Nutr. 2017, 106, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Ott, B.; Skurk, T.; Hastreiter, L.; Lagkouvardos, I.; Fischer, S.; Buttner, J.; Kellerer, T.; Clavel, T.; Rychlik, M.; Haller, D.; et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci. Rep. 2017, 7, 11955. [Google Scholar] [CrossRef] [PubMed]
- Van Gemert, W.A.; May, A.M.; Schuit, A.J.; Oosterhof, B.Y.; Peeters, P.H.; Monninkhof, E.M. Effect of weight loss with or without exercise on inflammatory markers and adipokines in postmenopausal women: The shape-2 trial, a randomized controlled trial. Cancer Epidemiol. Biomark. Prev. 2016, 25, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Daly, R.M.; Gianoudis, J.; Prosser, M.; Kidgell, D.; Ellis, K.A.; O’Connell, S.; Nowson, C.A. The effects of a protein enriched diet with lean red meat combined with a multi-modal exercise program on muscle and cognitive health and function in older adults: Study protocol for a randomised controlled trial. Trials 2015, 16, 339. [Google Scholar] [CrossRef] [PubMed]
- Burton, L.A.; Sumukadas, D. Optimal management of sarcopenia. Clin. Interv. Aging 2010, 5, 217–228. [Google Scholar] [PubMed]
- Winett, R.A.; Davy, B.M.; Marinik, E.; Savla, J.; Winett, S.G.; Phillips, S.M.; Lutes, L.D. Developing a new treatment paradigm for disease prevention and healthy aging. Transl. Behav. Med. 2014, 4, 117–123. [Google Scholar] [CrossRef] [PubMed]
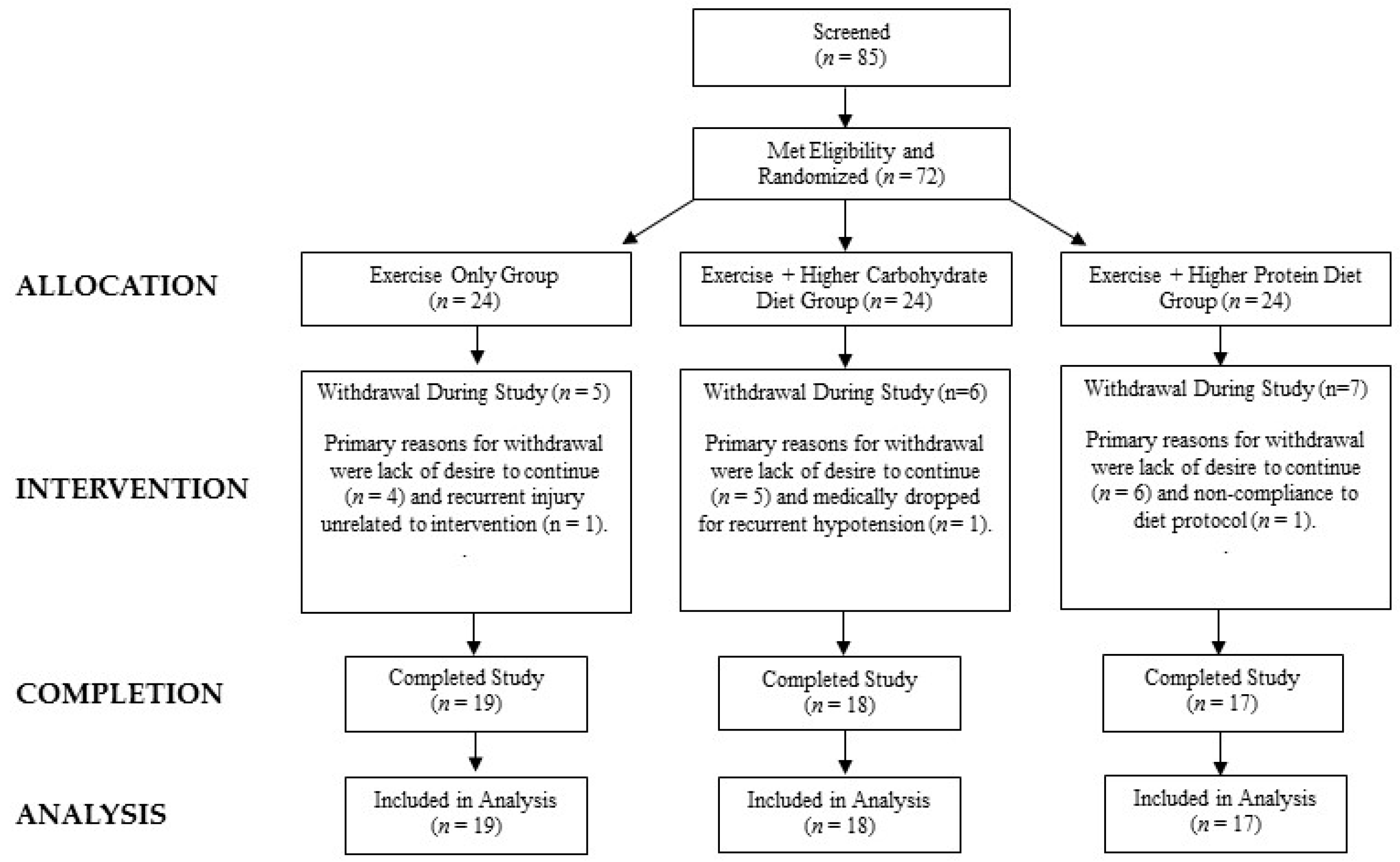
| Variable | E | HC | HP | p-Value |
|---|---|---|---|---|
| Age (years) | 66.0 ± 4.3 | 63.3 ± 4.8 | 65.5 ± 5.2 | 0.87 |
| Weight (kg) | 76.0 ± 9.5 | 78.6 ± 10.7 | 81.6 ± 11.4 | 0.29 |
| Height (cm) | 159.6 ± 3.7 | 161.3 ± 4.9 | 161.7 ± 6.8 | 0.41 |
| BMI (kg/m2) | 29.9 ± 4.1 | 30.3 ± 4.0 | 31.3 ± 4.2 | 0.61 |
| Body fat (%) | 42.7 ± 3.7 | 44.2 ± 3.5 | 43.6 ± 3.7 | 0.46 |
| Variable | Group | Week | Group (SEM) | Source | p-Value | ||
|---|---|---|---|---|---|---|---|
| 0 | 10 | 14 | |||||
| Energy intake | E | 1540 ± 397 | 1580 ± 399 | 1490 ± 485 | 1537 ± 70 | Group | 0.004 |
| (kcal/d) | HC | 1339 ± 321 | 1253 ± 269 | 1260 ± 308 | 1283 ± 70 c | Time | 0.854 |
| HP | 1598 ± 415 | 1567 ± 292 | 1711 ± 352 | 1626 ± 74 b | G × T | 0.322 | |
| Mean | 1489 ± 388 | 1462 ± 355 | 1478 ± 425 | ||||
| Protein | E | 68.3 ± 13.9 | 73.7 ± 11.0 bc | 68.5 ± 17.0 c | 70.2 ± 2.5 | Group | <0.001 |
| (g/d) | HC | 57.1 ± 13.5 | 55.3 ± 9.5 ac | 61.6 ± 16.1 a | 58.0 ± 2.5 c | Time | 0.003 |
| HP | 66.1 ± 13.0 | 94.9 ± 31.4 †ab | 86.2 ± 18.3 †ab | 82.4 ± 2.6 b | G × T | 0.002 | |
| Mean | 63.7 ± 14.1 | 73.9 ± 24.9 † | 71.6 ± 19.6 † | ||||
| Carbohydrate | E | 196 ± 70 | 182 ± 61 c | 173 ± 62 | 187 ± 11 | Group | 0.290 |
| (g/d) | HC | 166 ± 52 c | 171 ± 56 | 168 ± 51 | 163 ± 11 | Time | 0.022 |
| HP | 203 ± 56 b | 146 ± 44 †a | 193 ± 67 | 181 ± 12 | G × T | 0.017 | |
| Mean | 188 ± 61 | 173 ± 54 † | 182 ± 60 | ||||
| Total fat | E | 58.6 ± 20.1 | 65.7 ± 19.4 b | 67.4 ± 22.5 b | 63.9 ± 3.7 b | Group | 0.002 |
| (g/d) | HC | 48.9 ± 12.5 | 43.6 ± 9.9 ac | 43.4 ± 14.9 ac | 45.3 ± 3.5 ac | Time | 0.415 |
| HP | 54.9 ± 15.4 | 63.0 ± 21.9 b | 58.7 ± 23.4 b | 58.8 ± 3.5 b | G × T | 0.101 | |
| Mean | 53.9 ± 16.3 | 57.1 ± 20.1 | 56.1 ± 22.5 | ||||
| Calories | E | 20.1 ± 6.0 | 21.5 ± 5.5 | 20.4 ± 7.0 | 20.9 ± 1.06 | Group | 0.015 |
| (kcal/kg/d) | HC | 17.3 ± 4.5 a | 16.8 ± 4.4 ac | 17.0 ± 5.2 c | 17.0 ± 1.06 ac | Time | 0.550 |
| HP | 19.8 ± 4.4 | 20.8 ± 4.6 | 22.7 ± 5.4 | 21.1 ± 1.12 | G × T | 0.234 | |
| Mean | 19.3 ± 5.2 | 19.6 ± 5.2 | 19.9 ± 6.3 | ||||
| Protein | E | 0.93 ± 0.24 | 1.01 ± 0.22 | 0.94 ± 0.27 | 0.96 ± 0.05 | Group | <0.001 |
| (g/kg/d) | HC | 0.74 ± 0.19 a | 0.73 ± 0.15 | 0.83 ± 0.26 | 0.77 ± 0.04 | Time | <0.001 |
| HP | 0.83 ± 0.16 | 1.28 ± 0.5 ab | 1.15 ± 0.30 b | 1.09 ± 0.05 ab | G × T | 0.001 | |
| Mean | 0.83 ± 0.21 | 1.00 ± 0.40 † | 0.97 ± 0.30 † | ||||
| Carbohydrate | E | 2.66 ± 1.1 | 2.56 ± 0.9 c | 2.47 ± 1.0 | 2.56 ± 0.16 | Group | 0.233 |
| (g/kg/d) | HC | 2.14 ± 07 | 2.21 ± 0.8 | 2.14 ± 0.7 | 2.16 ± 0.16 | Time | 0.127 |
| HP | 2.50 ± 0.5 | 1.92 ± 0.5 a | 2.54 ± 0.9 | 2.32 ± 0.17 | G × T | 0.029 | |
| Mean | 2.43 ± 0.80 | 2.24 ± 0.8 | 2.38 ± 0.9 | ||||
| Total fat | E | 0.74 ± 0.23 | 0.85 ± 0.27 | 0.80 ± 0.32 | 0.79 ± 0.05 | Group | 0.004 |
| (g/kg/d) | HC | 0.63 ± 0.18 | 0.58 ± 0.14 | 0.59 ± 0.23 | 0.60 ± 0.05 ac | Time | 0.100 |
| HP | 0.73 ± 0.24 | 0.87 ± 0.30 | 0.90 ± 0.34 | 0.83 ± 0.05 | G × T | 0.072 | |
| Mean | 0.70 ± 0.22 | 0.76 ± 0.28 | 0.76 ± 0.32 | ||||
| Variable | Group | Weeks | Group (SEM) | Source | p-Value | ||
|---|---|---|---|---|---|---|---|
| 0 | 10 | 14 | |||||
| Body weight | E | 75.99 ± 9.5 | 75.11 ± 9.7 | 75.12 ± 9.7 | 75.39 ± 2.4 | Group | 0.606 |
| (kg) | HC | 78.63 ± 10.7 | 75.50 ± 11.1 † | 76.50 ± 11.1 † | 77.15 ± 2.5 | Time | <0.001 |
| HP | 81.59 ± 11.4 | 77.63 ± 10.8 † | 77.71 ± 10.8 † | 78.94 ± 2.6 | G × T | <0.001 | |
| Mean | 78.63 ± 10.6 | 76.34 ± 10.4 † | 78.34 ± 10.4 | ||||
| Fat mass | E | 29.89 ± 6.2 | 29.05 ± 6.0 † | 29.07 ± 6.1 † | 29.34 ± 1.4 | Group | 0.772 |
| (kg) | HC | 31.89 ± 6.7 | 30.43 ± 6.6 † | 29.99 ± 6.5 † | 30.77 ± 1.4 | Time | <0.001 |
| HP | 32.78 ± 6.0 | 29.98 ± 5.7 † | 29.42 ± 5.6 † | 30.73 ± 1.5 | G × T | <0.001 | |
| Mean | 31.46 ± 6.3 | 29.80 ± 6.0 † | 29.49 ± 6.0 † | ||||
| Fat free mass | E | 39.66 ± 4.1 | 40.1 ± 4.2 | 40.01 ± 4.3 | 39.95 ± 1.2 | Group | 0.351 |
| (kg) | HC | 39.86 ± 4.6 | 40.1 ± 4.9 | 40.59 ± 5.4 | 40.19 ± 1.2 | Time | 0.064 |
| HP | 42.40 ± 5.8 | 42.09 ± 6.1 | 42.42 ± 6.6 | 42.23 ± 1.3 | G × T | 0.427 | |
| Mean | 40.53 ± 4.9 | 40.75 ± 5.1 | 40.96 ± 5.5 | ||||
| Body fat | E | 42.68 ± 3.7 | 41.69 ± 3.7 † | 41.78 ± 3.5 † | 42.05 ± 0.8 | Group | 0.584 |
| (%) | HC | 44.17 ± 3.5 | 42.85 ± 3.5 † | 42.23 ± 3.3 † | 43.08 ± 0.8 | Time | <0.001 |
| HP | 43.57 ± 3.8 | 41.45 ± 3.8 † | 40.86 ± 4.3 † | 41.96 ± 0.9 | G × T | 0.005 | |
| Mean | 43.46 ± 3.6 | 42.00 ± 3.6 † | 41.63 ± 3.7 † | ||||
| Bone mineral | E | 1531 ± 183 | 1527 ± 179 | 1535 ± 182 | 1531 ± 68 a | Group | 0.045 |
| content (g) | HC | 1613 ± 214 | 1615 ± 212 | 1608 ± 227 | 1612 ± 70 | Time | 0.349 |
| HP | 1789 ± 427 | 1767 ± 454 | 1788 ± 439 | 1781 ± 72 c | G × T | 0.365 | |
| Mean | 1640 ± 305 | 1632 ± 312 | 1639 ± 312 | ||||
| Waist | E | 88.4 ± 7.9 | 88.3 ± 7.3 | 87.5 ± 7.6 | 88.1 ± 2.2 | Group | 0.978 |
| circumference | HC | 89.7 ± 12.9 | 88.9 ± 12.0 | 88.2 ± 12.1 | 89.9 ± 2.2 | Time | <0.001 |
| (cm) | HP | 91.2 ± 8.2 | 89.1 ± 7.5 † | 87.6± 7.6 † | 89.3 ± 2.3 | G × T | 0.016 |
| Mean | 89.7 ± 9.0 | 88.7 ± 9.0 † | 87.8 ± 9.2 † | ||||
| Hip | E | 107.6 ± 8.1 | 107.0 ± 8.1 | 106.0 ± 7.4 | 106.9 ± 1.7 | Group | <0.001 |
| circumference | HC | 109.9 ± 5.5 | 108.0± 6.3 | 107.9 ± 7.6 † | 108.6 ± 1.7 | Time | <0.001 |
| (cm) | HP | 112.7 ± 8.8 | 109.4 ± 8.2 † | 108.8 ± 7.9 † | 110.3 ± 18 | G × T | 0.019 |
| Mean | 110.0± 7.5 | 108.1± 7.5 † | 107.5 ± 7.6 † | ||||
| Waist:hip | E | 0.822 ± 0.05 | 0.826 ± 0.08 | 0.827 ± 0.06 | 0.825 ± 0.01 | Group | 0.684 |
| ratio | HC | 0.815 ± 0.09 | 0.821 ± 0.08 | 0.815 ± 0.07 | 0.817 ± 0.02 | Time | 0.180 |
| HP | 0.810 ± 0.05 | 0.815 ± 0.05 | 0.805 ± 0.05 | 0.810 ± 0.02 | G × T | 0.616 | |
| Mean | 0.816 ± 0.07 | 0.821 ± 0.06 | 0.816 ± 0.06 | ||||
| Resting energy expenditure (kcal/d) | E | 1317 ± 184 | 1341 ± 198 | 1345 ± 168 | 1334 ± 39 | Group | 0.923 |
| HC | 1313 ± 199 | 1358 ± 211 | 1356 ± 181 | 1342 ± 37 | Time | 0.065 | |
| HP | 1311 ± 151 | 1361 ±130 | 1287 ± 176 | 1320 ± 43 | G × T | 0.356 | |
| Mean | 1314 ± 178 | 1353 ± 183 | 1332 ± 174 | ||||
| Variable | Group | Weeks | Group (SEM) | Source | p-Value | ||
|---|---|---|---|---|---|---|---|
| 0 | 10 | 14 | |||||
| Glucose | E | 109.0 ± 21 | 108.3 ± 23 | 106.5 ± 21 | 107.9 ± 5 | Group | 0.400 |
| (mg/dL) | HC | 96.8 ± 14 | 98.4 ± 9 | 100.2 ± 10 | 98.5 ± 5 | Time | 0.335 |
| HP | 114.5 ± 61 | 105.3 ± 26 | 99.5 ± 19 | 106.4 ± 5 | G × T | 0.295 | |
| Mean | 103.7 ± 37 | 104.1 ± 21 | 102.2 ± 18 | ||||
| Insulin | E | 11.1 ± 6.5 | 14.0 ± 8.0 | 17.1 ± 9.5 | 14.1 ± 1.9 | Group | 0.459 |
| (µU/mL) | HC | 10.1 ± 6.5 | 9.7 ± 5.9 | 16.1 ± 7.7 | 12.0 ± 1.9 | Time | <0.001 |
| HP | 12.7 ± 12.8 | 15.3 ± 10.9 | 18.1 ± 12.3 | 15.4 ± 2.0 | G × T | 0.486 | |
| Mean | 11.3 ± 8.8 | 13.0 ± 8.0 | 17.1 ± 9.8 † | ||||
| Insulin sensitivity | E | 3.17 ± 2.4 | 3.80 ± 4.5 | 4.61 ± 2.8 | 3.86 ± 0.6 | Group | 0.308 |
| (HOMAIR) | HC | 2.47 ± 1.6 | 2.39 ± 1.5 | 3.96 ± 1.9 | 2.94 ± 0.6 | Time | <0.001 |
| HP | 3.67 ± 3.8 | 4.46 ± 4.4 | 4.86 ± 4.3 | 4.33 ± 0.7 | G × T | 0.527 | |
| Mean | 3.10 ± 2.7 | 3.53 ± 3.1 | 4.47 ± 3.1 † | ||||
| Adiponectin | E | 11.5 ± 5.7 | 10.3 ± 3.9 | 16.6 ± 14.2 | 12.8 ± 1.8 | Group | 0.844 |
| (µg/mL) | HC | 13.7 ± 6.1 | 13.1 ± 4.8 | 9.5 ± 5.1 a | 12.1 ± 1.8 | Time | 0.007 |
| HP | 14.0 ± 7.7 | 6.5 ± 2.6 † | 23.1 ± 25.1 †b | 14.6 ± 1.9 | G × T | 0.002 | |
| Mean | 13.1 ± 6.5 | 10.1 ± 4.7 † | 16.3 ± 17.3 † | ||||
| Leptin | E | 36.7 ± 13.4 c | 31.3 ± 23.2 | 35.2 ± 17.5 b | 34.4 ± 3.5 | Group | 0.218 |
| (ng/mL) | HC | 36.7 ± 16.4 c | 32.8 ± 19.5 | 50.4 ± 24.6 †ac | 39.9 ± 3.6 | Time | <0.001 |
| HP | 61.5 ± 18.6 ab | 37.9 ± 14.5 † | 38.4 ± 19.7 †b | 45.9 ± 3.7 | G × T | <0.001 | |
| Mean | 44.5 ± 19.7 | 33.9 ± 19.4 † | 41.3 ± 21.4 | ||||
| Triglycerides | E | 141 ± 95 | 172 ± 125 †c | 138 ± 89 bc | 150 ± 19 | Group | 0.898 |
| (mg/dL) | HC | 145 ± 65 | 160 ± 78 † | 158 ± 66 c | 154 ± 19 | Time | 0.186 |
| HP | 159 ± 81 | 149 ± 67 a | 157 ± 84 a | 155 ± 20 | G × T | 0.046 | |
| Mean | 148 ± 80 | 161 ± 79 | 151 ± 79 | ||||
| Total cholesterol | E | 200 ± 36 | 215 ± 36 | 191 ± 31 | 202 ± 7 | Group | 0.343 |
| (mg/dL) | HC | 199 ± 64 | 201 ± 27 | 199 ± 31 | 199 ± 7 | Time | 0.062 |
| HP | 214 ± 32 | 216 ± 28 | 204 ± 24 | 211 ± 7 | G × T | 0.470 | |
| Mean | 204 ± 46 | 210 ± 31 | 198 ± 29 | ||||
| Low density | E | 115 ± 35 | 127 ± 28 | 115 ± 32 | 119 ± 6 | Group | 0.455 |
| lipoprotein | HC | 114 ± 43 | 110 ± 21 | 109 ± 24 | 111 ± 6 | Time | 0.828 |
| (mg/dL) | HP | 122 ± 25 | 123 ± 24 | 120 ± 25 | 121 ± 6 | G × T | 0.594 |
| Mean | 117 ± 35 | 120 ± 25 | 115 ± 27 | ||||
| High density | E | 54.1 ± 15.4 | 55.6 ± 16.2 | 55.3 ± 15.4 | 55.0 ± 3.0 | Group | 0.876 |
| lipoprotein | HC | 55.06 ± 10.4 | 55.1 ± 11.2 | 57.3 ± 13.1 | 55.8 ± 3.1 | Time | 0.239 |
| (mg/dL) | HP | 57.8 ± 18.9 | 55.5 ± 13.8 | 55.6 ± 12.9 | 56.3 ± 3.2 | G × T | 0.216 |
| Mean | 55.6 ± 15.0 | 55.4 ± 13.7 | 56.1 ± 13.7 | ||||
| Variable | Group | Weeks | Group (SEM) | Source | p-Value | |
|---|---|---|---|---|---|---|
| 0 | 14 | |||||
| Resting heart rate | E | 77.4 ± 12.6 | 72.0 ± 9.3 | 74.7 ± 2.4 | Group | 0.283 |
| (bpm) | HC | 74.4 ± 12.8 | 73.9 ± 11.8 | 74.2 ± 2.4 | Time | 0.074 |
| HP | 74.1 ± 11.4 | 71.4 ± 11.6 | 72.7 ± 2.6 | G × T | 0.455 | |
| Time | 75.4 ± 12.6 | 72.4 ± 10.8 | ||||
| Resting systolic | E | 125.7 ± 10.5 | 122.8 ± 13.2 | 124.3 ± 2.3 | Group | 0.038 |
| blood pressure | HC | 124.6 ± 12.5 | 123.7 ± 14.1 | 124.18 ± 23 | Time | 0.238 |
| (mmHg) | HP | 126.5 ± 16.3 | 121.9 ± 11.3 | 124.2 ± 2.4 | G × T | 0.814 |
| Time | 125.6 ± 13.0 | 122.8 ± 12.8 | ||||
| Resting diastolic | E | 77.8± 9.80 | 71.9 ± 7.8 | 74.8 ± 1.6 | Group | 0.330 |
| blood pressure | HC | 73.3 ± 10.4 | 72.8 ± 9.4 | 73.1 ± 1.7 | Time | 0.032 |
| (mmHg) | HP | 75.1 ± 9.5 | 70.9 ± 7.5 | 73.0 ± 1.7 | G × T | 0.379 |
| Time | 75.4 ± 9.9 | 71.9 ± 8.1 † | ||||
| Peak VO2 | E | 17.70 ± 3.3 | 18.39 ± 3.2 | 18.05 ± 0.6 | Group | 0.110 |
| (mL/kg/min) | HC | 15.53 ± 2.2 | 17.14 ± 2.8 | 16.34 ± 0.7 | Time | <0.001 |
| HP | 17.13 ± 3.7 | 19.29 ± 3.1 | 18.21 ± 0.7 | G × T | 0.227 | |
| Time | 16.82 ± 3.2 | 18.26 ± 3.2 † | ||||
| Walk test (6-min) | E | 496 ± 67 | 534 ± 59 | 516 ± 13 | Group | 0.218 |
| (m) | HC | 517 ± 47 | 551 ± 40 | 533 ± 14 | Time | <0.001 |
| HP | 538 ± 70 | 570 ± 77 | 554 ± 14 | G × T | 0.860 | |
| Time | 516 ± 63 | 551 ± 61 † | ||||
| Bench press (1RM) | E | 21.4 ± 5.0 | 26.2 ± 3.3 | 23.8 ± 1.2 | Group | 0.536 |
| (kg) | HC | 20.6 ± 5.6 | 23.4 ± 5.7 | 22.0 ± 1.2 | Time | <0.001 |
| HP | 20.6 ± 6.2 | 24.5 ± 4.5 | 22.5 ± 1.3 | G × T | 0.245 | |
| Time | 20.9 ± 5.5 | 24.7 ± 4.7 † | ||||
| Bench press | E | 127.8 ± 62 | 143.4 ± 43 | 135.6 ± 10 | Group | 0.237 |
| endurance volume | HC | 98.5 ± 53 | 135.7 ± 58 | 117.1 ± 10 | Time | <0.001 |
| (kg) | HP | 132.9 ± 43 | 148.0 ± 55 | 140.4 ± 11 | G × T | 0.568 |
| Time | 119.0 ± 55 | 142.1 ± 52 † | ||||
| Leg press 1RM | E | 89.4 ± 32 | 110.0 ± 34 | 99.7 ± 8 | Group | 0.781 |
| (kg) | HC | 82.7 ± 33 | 105.5± 37 | 94.1 ± 8 | Time | 0.025 |
| HP | 78.1 ± 32 | 106.2 ± 28 | 92.2± 8 | G × T | 0.636 | |
| Time | 83.5 ± 32 | 107.2 ± 33† | ||||
| Leg press | E | 936 ± 623 | 1182 ± 959 | 1059 ± 147 | Group | 0.792 |
| endurance volume | HC | 839 ± 451 | 1114 ± 708 | 977 ± 143 | Time | 0.122 |
| (kg) | HP | 1161 ± 732 | 1076 ± 375 | 1118 ± 152 | G × T | 0.229 |
| Time | 972 ± 609 | 1125 ± 713 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galbreath, M.; Campbell, B.; La Bounty, P.; Bunn, J.; Dove, J.; Harvey, T.; Hudson, G.; Gutierrez, J.L.; Levers, K.; Galvan, E.; et al. Effects of Adherence to a Higher Protein Diet on Weight Loss, Markers of Health, and Functional Capacity in Older Women Participating in a Resistance-Based Exercise Program. Nutrients 2018, 10, 1070. https://doi.org/10.3390/nu10081070
Galbreath M, Campbell B, La Bounty P, Bunn J, Dove J, Harvey T, Hudson G, Gutierrez JL, Levers K, Galvan E, et al. Effects of Adherence to a Higher Protein Diet on Weight Loss, Markers of Health, and Functional Capacity in Older Women Participating in a Resistance-Based Exercise Program. Nutrients. 2018; 10(8):1070. https://doi.org/10.3390/nu10081070
Chicago/Turabian StyleGalbreath, Melyn, Bill Campbell, Paul La Bounty, Jennifer Bunn, Jacqueline Dove, Travis Harvey, Geoffrey Hudson, Jean L. Gutierrez, Kyle Levers, Elfego Galvan, and et al. 2018. "Effects of Adherence to a Higher Protein Diet on Weight Loss, Markers of Health, and Functional Capacity in Older Women Participating in a Resistance-Based Exercise Program" Nutrients 10, no. 8: 1070. https://doi.org/10.3390/nu10081070
APA StyleGalbreath, M., Campbell, B., La Bounty, P., Bunn, J., Dove, J., Harvey, T., Hudson, G., Gutierrez, J. L., Levers, K., Galvan, E., Jagim, A., Greenwood, L., Cooke, M. B., Greenwood, M., Rasmussen, C., & Kreider, R. B. (2018). Effects of Adherence to a Higher Protein Diet on Weight Loss, Markers of Health, and Functional Capacity in Older Women Participating in a Resistance-Based Exercise Program. Nutrients, 10(8), 1070. https://doi.org/10.3390/nu10081070









