Coffee Consumption and Whole-Blood Gene Expression in the Norwegian Women and Cancer Post-Genome Cohort
Abstract
1. Introduction
2. Materials and Methods
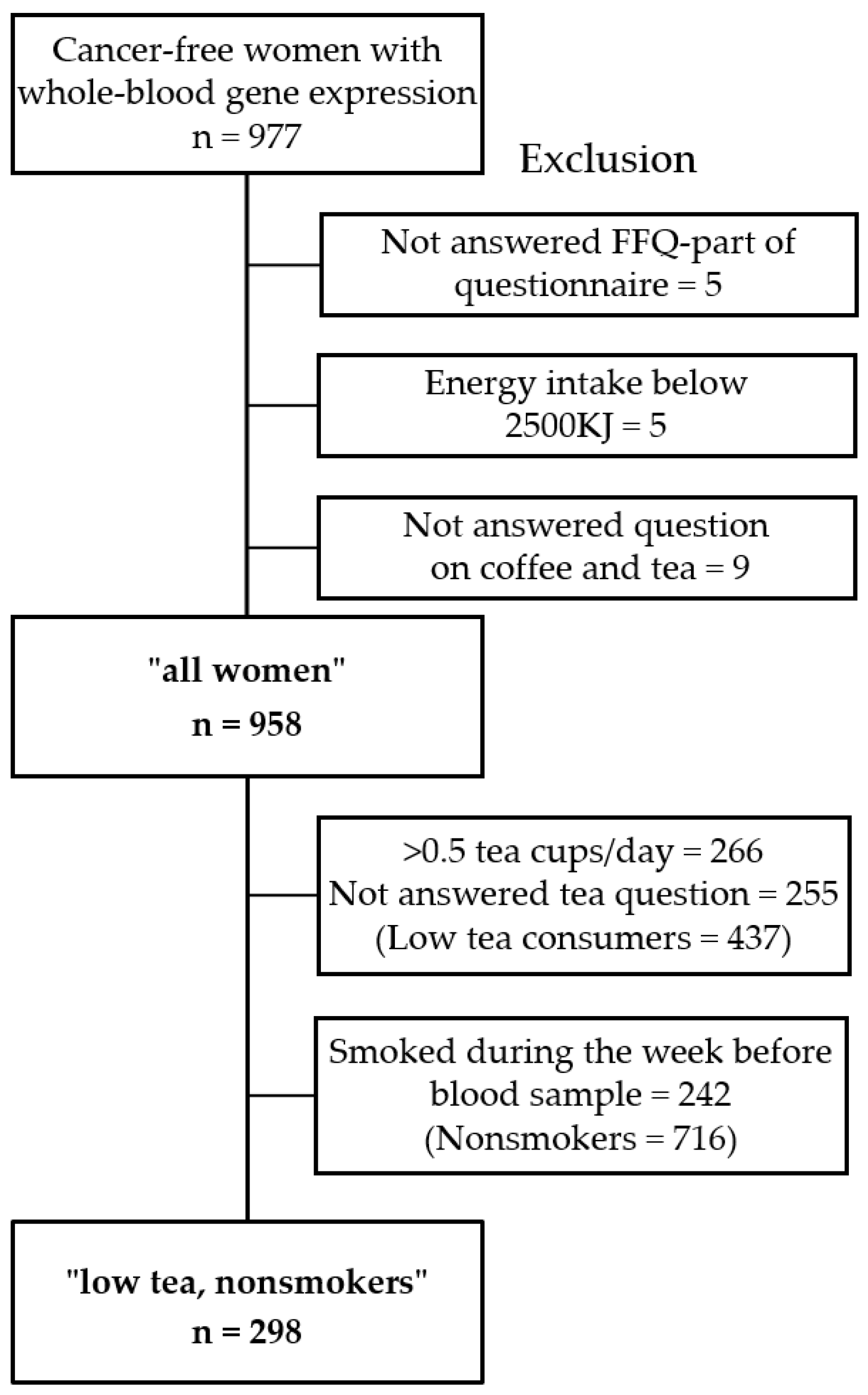
2.1. Study Population
2.2. Determination of Gene Expression Levels
2.3. Dietary Assessment and Descriptive Variables
2.4. Statistical Analysis
3. Results
3.1. Descriptors
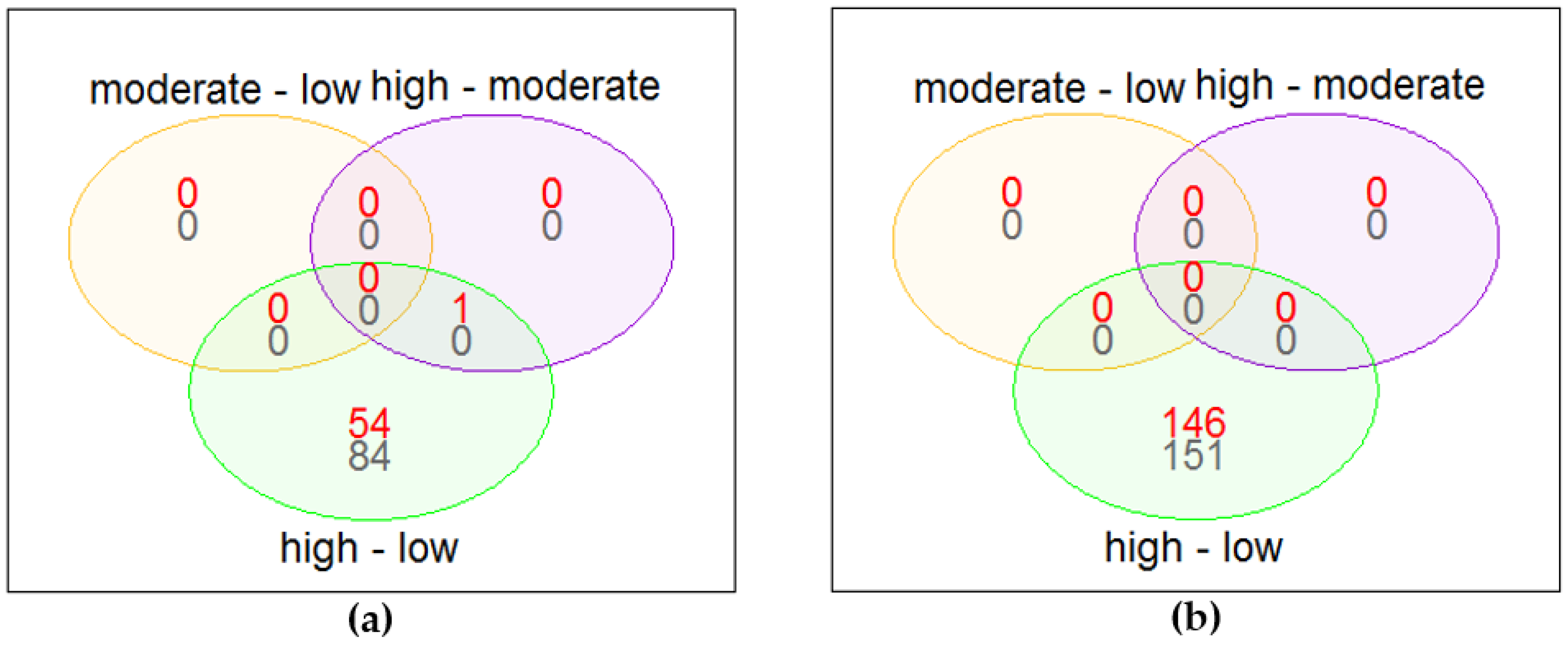
3.2. Differential Gene Expression
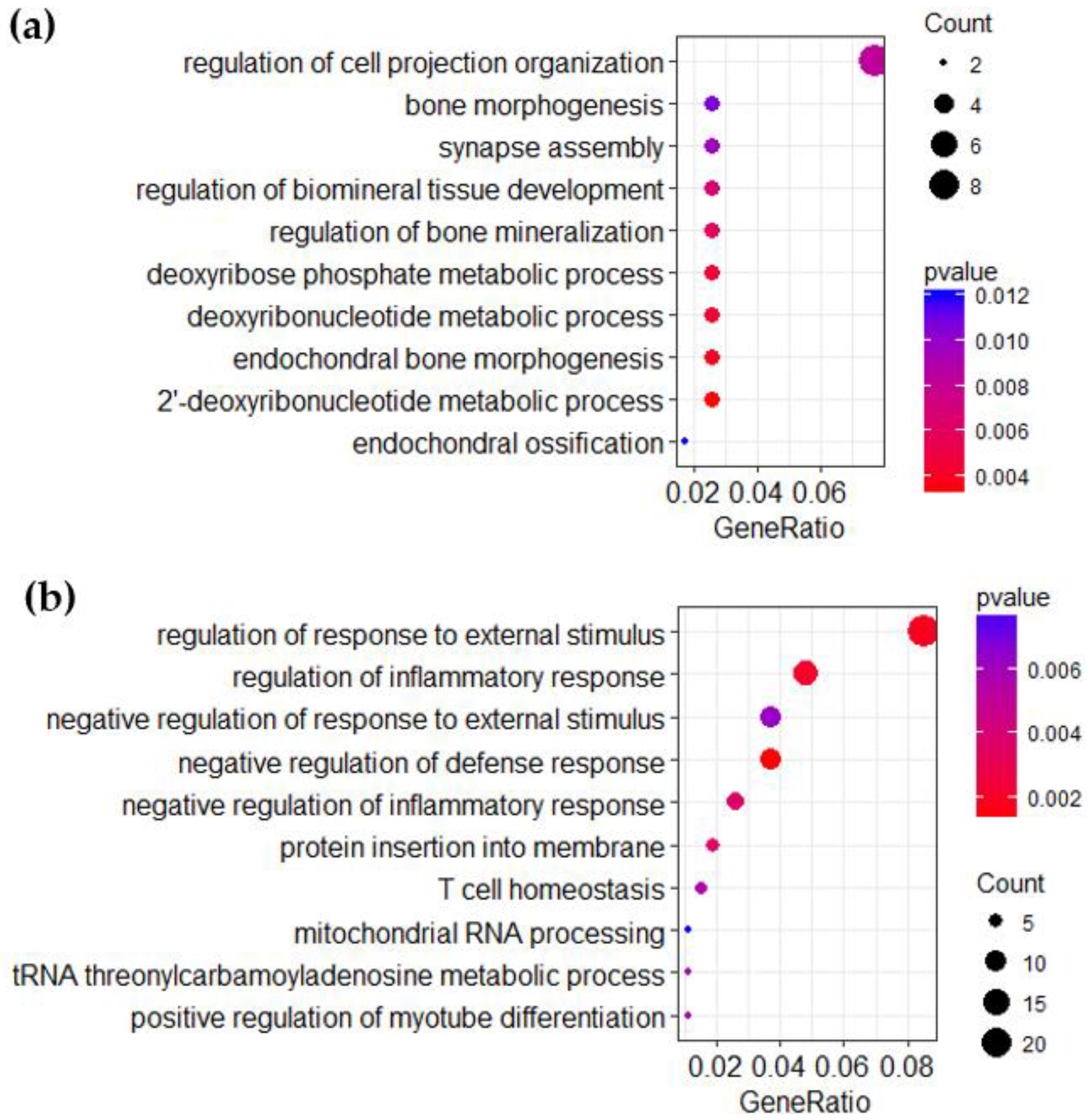
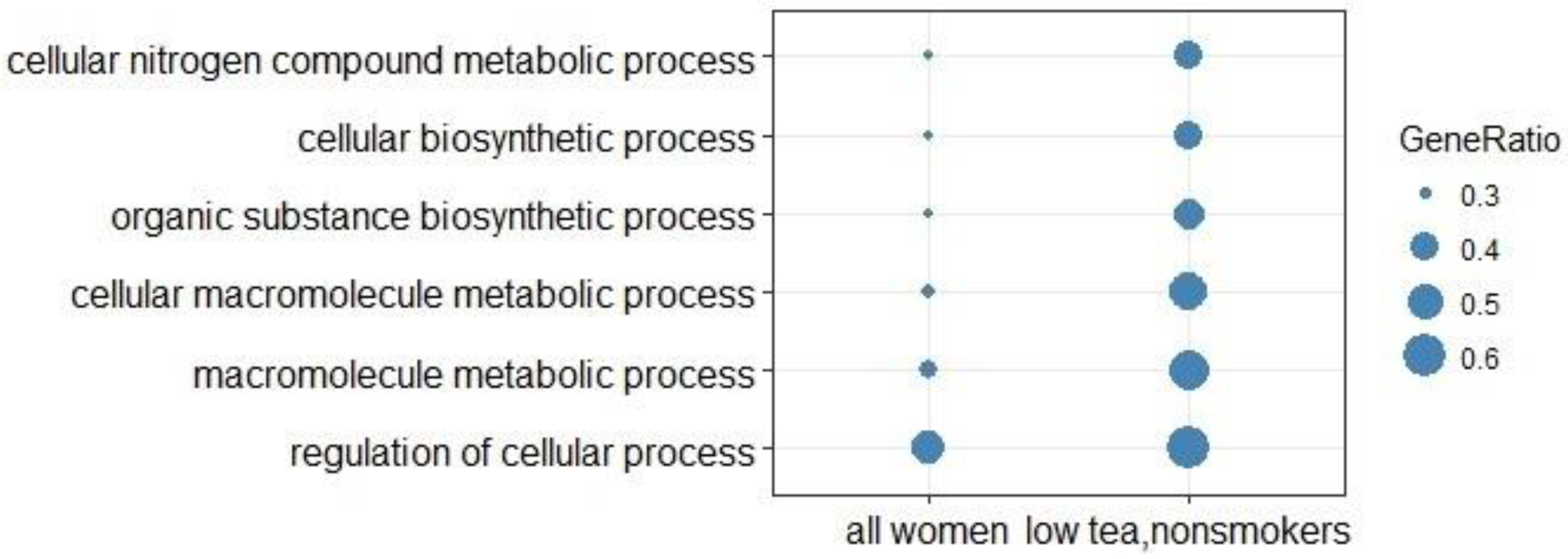
3.3. Over-Representation Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Coffee Council. Trends in Coffee Consumption in Selected Importing Countries; International Coffee Council: London, UK, 2012. [Google Scholar]
- The Norwegian Directorate of Health. Norkost 3. En Landsomfattende Kostholdsundersøkelse Blant Menn og Kvinner i Norge i Alderen 18-70 år, 2010-11; The Norwegian Directorate of Health: Oslo, Norway, 2012. (In Norwegian) [Google Scholar]
- Liu, Q.P.; Wu, Y.F.; Cheng, H.Y.; Xia, T.; Ding, H.; Wang, H.; Wang, Z.M.; Xu, Y. Habitual coffee consumption and risk of cognitive decline/dementia: A systematic review and meta-analysis of prospective cohort studies. Nutrition 2016, 32, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Li, S. Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of parkinson’s disease. Geriatr. Gerontol. Int. 2014, 14, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Hernan, M.A.; Takkouche, B.; Caamano-Isorna, F.; Gestal-Otero, J.J. A meta-analysis of coffee drinking, cigarette smoking, and the risk of parkinson’s disease. Ann. Neurol. 2002, 52, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.; Lunet, N.; Santos, C.; Santos, J.; Vaz-Carneiro, A. Caffeine exposure and the risk of parkinson’s disease: A systematic review and meta-analysis of observational studies. J. Alzheimers Dis. 2010, 20, 221–238. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Bhupathiraju, S.N.; Chen, M.; van Dam, R.M.; Hu, F.B. Caffeinated and decaffeinated coffee consumption and risk of type 2 diabetes: A systematic review and a dose-response meta-analysis. Diabetes Care 2014, 37, 569–586. [Google Scholar] [CrossRef] [PubMed]
- World Cancer Research Fund. American Institute for Cancer Research. In Continous Update Project Expert Report 2018. Non-Alcoholic Drinks and the Risk of Cancer.; World Cancer Research Fund: London, UK, 2018; pp. 34–39. [Google Scholar]
- Jee, S.H.; He, J.; Appel, L.J.; Whelton, P.K.; Suh, I.; Klag, M.J. Coffee consumption and serum lipids: A meta-analysis of randomized controlled clinical trials. Am. J. Epidemiol. 2001, 153, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Ma, D.; Zhang, Y.; Liu, Z.; Wang, P. The effect of coffee consumption on serum lipids: A meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2012, 66, 872–877. [Google Scholar] [CrossRef] [PubMed]
- Grosso, L.M.; Bracken, M.B. Caffeine metabolism, genetics, and perinatal outcomes: A review of exposure assessment considerations during pregnancy. Ann. Epidemiol. 2005, 15, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, D.C.; Thatcher, N.J.; Ye, J.; Garrard, L.; Keogh, G.; King, L.G.; Cade, J.E. Caffeine intake during pregnancy and adverse birth outcomes: A systematic review and dose-response meta-analysis. Eur. J. Epidemiol. 2014, 29, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Wikoff, D.; Welsh, B.T.; Henderson, R.; Brorby, G.P.; Britt, J.; Myers, E.; Goldberger, J.; Lieberman, H.R.; O’Brien, C.; Peck, J.; et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem. Toxicol. 2017, 109, 585–648. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, I.A.; Clifford, M.N.; Lean, M.E.; Ashihara, H.; Crozier, A. Coffee: Biochemistry and potential impact on health. Food Funct. 2014, 5, 1695–1717. [Google Scholar] [CrossRef] [PubMed]
- Cano-Marquina, A.; Tarin, J.J.; Cano, A. The impact of coffee on health. Maturitas 2013, 75, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, M.C. Toward systems epidemiology of coffee and health. Curr. Opin. Lipidol. 2015, 26, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, M.C.; Monda, K.L.; Yu, K.; Paynter, N.; Azzato, E.M.; Bennett, S.N. Genome-wide meta-analysis identifies regions on 7p21 (ahr) and 15q24 (cyp1a2) as determinants of habitual caffeine consumption. PLoS Genet. 2011, 7, e1002033. [Google Scholar] [CrossRef] [PubMed]
- Sulem, P.; Gudbjartsson, D.F.; Geller, F.; Prokopenko, I.; Feenstra, B.; Aben, K.K. Sequence variants at cyp1a1-cyp1a2 and ahr associate with coffee consumption. Hum. Mol. Genet. 2011, 20, 2071–2077. [Google Scholar] [CrossRef] [PubMed]
- The Coffee and Caffeine Genetics Consortium; Cornelis, M.C.; Byrne, E.M.; Esko, T.; Nalls, M.A.; Ganna, A.; Paynter, N.; Monda, K.L.; Amin, N.; Fischer, K.; et al. Genome-wide meta-analysis identifies six novel loci associated with habitual coffee consumption. Mol. Psychiatry 2015, 20, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Lund, E.; Dumeaux, V.; Braaten, T.; Hjartaker, A.; Engeset, D.; Skeie, G.; Kumle, M. Cohort profile: The norwegian women and cancer study--nowac--kvinner og kreft. Int. J. Epidemiol. 2008, 37, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Dumeaux, V.; Borresen-Dale, A.L.; Frantzen, J.O.; Kumle, M.; Kristensen, V.N.; Lund, E. Gene expression analyses in breast cancer epidemiology: The norwegian women and cancer postgenome cohort study. Breast Cancer Res. 2008, 10, R13. [Google Scholar] [CrossRef] [PubMed]
- Günter, C.; Holden, M.; Holden, L. Preprocessing of Gene-Expression Data Related to Breast Cancer Diagnosis: Samba/35/14; Norsk Regnesentral: Oslo, Norway, 2014. [Google Scholar]
- Blaker, B.; Aarsland, M. Mål og vekt for Matvarer; Landsforeningen for Kosthold og Helse: Oslo, Norway, 1989. (In Norwegian) [Google Scholar]
- Norwegian Food Safety Authority; The Norwegian Directorate of Health; University of Oslo. Norwegian Food Composition Database. Available online: www.matvaretabellen.no (accessed on 1 July 2018).
- Parr, C.L.; Veierod, M.B.; Laake, P.; Lund, E.; Hjartaker, A. Test-retest reproducibility of a food frequency questionnaire (ffq) and estimated effects on disease risk in the norwegian women and cancer study (nowac). Nutr. J. 2006, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Hjartaker, A.; Andersen, L.F.; Lund, E. Comparison of diet measures from a food-frequency questionnaire with measures from repeated 24-hour dietary recalls. The norwegian women and cancer study. Public Health Nutr. 2007, 10, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Lukic, M.; Licaj, I.; Lund, E.; Skeie, G.; Weiderpass, E.; Braaten, T. Coffee consumption and the risk of cancer in the norwegian women and cancer (nowac) study. Eur. J. Epidemiol. 2016, 31, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Gavrilyuk, O.; Braaten, T.; Skeie, G.; Weiderpass, E.; Dumeaux, V.; Lund, E. High coffee consumption and different brewing methods in relation to postmenopausal endometrial cancer risk in the norwegian women and cancer study: A population-based prospective study. BMC Womens Health 2014, 14, 48. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical computing: Vienna, Austria, 2017. [Google Scholar]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma powers differential expression analyses for rna-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. Clusterprofiler: An r package for comparing biological themes among gene clusters. OMICS 2012, 16, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Schubert, M.M.; Irwin, C.; Seay, R.F.; Clarke, H.E.; Allegro, D.; Desbrow, B. Caffeine, coffee, and appetite control: A review. Int. J. Food Sci. Nutr. 2017, 68, 901–912. [Google Scholar] [CrossRef] [PubMed]
- Bjorngaard, J.H.; Nordestgaard, A.T.; Taylor, A.E.; Treur, J.L.; Gabrielsen, M.E.; Munafo, M.R.; Nordestgaard, B.G.; Asvold, B.O.; Romundstad, P.; Davey Smith, G. Heavier smoking increases coffee consumption: Findings from a mendelian randomization analysis. Int. J. Epidemiol. 2017, 46, 1958–1967. [Google Scholar] [CrossRef] [PubMed]
- Swanson, J.A.; Lee, J.W.; Hopp, J.W. Caffeine and nicotine—A review of their joint use and possible interactive effects in tobacco withdrawal. Addict. Behav. 1994, 19, 229–256. [Google Scholar] [CrossRef]
- Grela, A.; Kulza, M.; Piekoszewski, W.; Senczuk-Przybylowska, M.; Gomolka, E.; Florek, E. The effects of tobacco smoke exposure on caffeine metabolism. Ital. J. Food Sci. 2013, 25, 76–82. [Google Scholar]
- Huan, T.; Joehanes, R.; Schurmann, C.; Schramm, K.; Pilling, L.C.; Peters, M.J.; Magi, R.; DeMeo, D.; O’Connor, G.T.; Ferrucci, L.; et al. A whole-blood transcriptome meta-analysis identifies gene expression signatures of cigarette smoking. Hum. Mol. Genet. 2016, 25, 4611–4623. [Google Scholar] [CrossRef] [PubMed]
- Yi, B.; Yang, J.Y.; Yang, M. Past and future applications of cyp450-genetic polymorphisms for biomonitoring of environmental toxicants. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2007, 25, 353–377. [Google Scholar] [CrossRef] [PubMed]
- Spatzenegger, M.; Horsmans, Y.; Verbeeck, R.K. Cyp1a1 but not cyp1a2 proteins are expressed in human lymphocytes. Pharmacol. Toxicol. 2000, 86, 242–244. [Google Scholar] [CrossRef] [PubMed]
- Denden, S.; Bouden, B.; Haj Khelil, A.; Ben Chibani, J.; Hamdaoui, M.H. Gender and ethnicity modify the association between the cyp1a2 rs762551 polymorphism and habitual coffee intake: Evidence from a meta-analysis. Genet. Mol. Res. 2016, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- McGraw, J.; Waller, D. Cytochrome p450 variations in different ethnic populations. Expert Opin. Drug Metab. Toxicol. 2012, 8, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Kashiwakura, J.; Suzuki, N.; Nagafuchi, H.; Takeno, M.; Takeba, Y.; Shimoyama, Y.; Sakane, T. Txk, a nonreceptor tyrosine kinase of the tec family, is expressed in t helper type 1 cells and regulates interferon gamma production in human t lymphocytes. J. Exp. Med. 1999, 190, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Mullen, A.C.; Hutchins, A.S.; High, F.A.; Lee, H.W.; Sykes, K.J.; Chodosh, L.A.; Reiner, S.L. Hlx is induced by and genetically interacts with t-bet to promote heritable t(h)1 gene induction. Nat. Immunol. 2002, 3, 652–658. [Google Scholar] [CrossRef] [PubMed]
- De Santa, F.; Totaro, M.G.; Prosperini, E.; Notarbartolo, S.; Testa, G.; Natoli, G. The histone h3 lysine-27 demethylase jmjd3 links inflammation to inhibition of polycomb-mediated gene silencing. Cell 2007, 130, 1083–1094. [Google Scholar] [CrossRef] [PubMed]
- Schlicher, L.; Brauns-Schubert, P.; Schubert, F.; Maurer, U. Spata2: More than a missing link. Cell Death Differ. 2017, 24, 1142–1147. [Google Scholar] [CrossRef] [PubMed]
- Dhavan, R.; Tsai, L.H. A decade of cdk5. Nat. Rev. Mol. Cell Biol. 2001, 2, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Reiter, V.; Matschkal, D.M.S.; Wagner, M.; Globisch, D.; Kneuttinger, A.C.; Muller, M.; Carell, T. The cdk5 repressor cdk5rap1 is a methylthiotransferase acting on nuclear and mitochondrial rna. Nucleic Acids Res. 2012, 40, 6235–6240. [Google Scholar] [CrossRef] [PubMed]
- De Mello, V.D.F.; Kolehmanien, M.; Schwab, U.; Pulkkinen, L.; Uusitupa, M. Gene expression of peripheral blood mononuclear cells as a tool in dietary intervention studies: What do we know so far? Mol. Nutr. Food Res. 2012, 56, 1160–1172. [Google Scholar] [CrossRef] [PubMed]
- Olsen, K.S.; Skeie, G.; Lund, E. Whole-blood gene expression profiles in large-scale epidemiological studies: What do they tell? Curr. Nutr. Rep. 2015, 4, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.F.; Jacobs, D.R.; Carlsen, M.H.; Blomhoff, R. Consumption of coffee is associated with reduced risk of death attributed to inflammatory and cardiovascular diseases in the iowa women’s health study. Am. J. Clin. Nutr. 2006, 83, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Schulze, M.B.; Hoffmann, K.; Manson, J.E.; Willett, W.C.; Meigs, J.B.; Weikert, C.; Heidemann, C.; Colditz, G.A.; Hu, F.B. Dietary pattern, inflammation, and incidence of type 2 diabetes in women. Am. J. Clin. Nutr. 2005, 82, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Zampelas, A.; Panagiotakos, D.B.; Pitsavos, C.; Chrysohoou, C.; Stefanadis, C. Associations between coffee consumption and inflammatory markers in healthy persons: The attica study. Am. J. Clin. Nutr. 2004, 80, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Svilaas, A.; Sakhi, A.K.; Andersen, L.F.; Svilaas, T.; Strom, E.C.; Jacobs, D.R.J.; Ose, L.; Blomhoff, R. Intakes of antioxidants in coffee, wine, and vegetables are correlated with plasma carotenoids in humans. J. Nutr. 2004, 134, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Halvorsen, B.; Ranheim, T.; Nenseter, M.S.; Huggett, A.C.; Drevon, C.A. Effect of a coffee lipid (cafestol) on cholesterol metabolism in human skin fibroblasts. J. Lipid Res. 1998, 39, 901–912. [Google Scholar] [PubMed]
- Hurtubise, J.; McLellan, K.; Durr, K.; Onasanya, O.; Nwabuko, D.; Ndisang, J.F. The different facets of dyslipidemia and hypertension in atherosclerosis. Curr. Atheroscler Rep. 2016, 18, 82. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.; Dieminger, N.; Beusch, A.; Lee, Y.M.; Dunkel, A.; Suess, B.; Skurk, T.; Wahl, A.; Hauner, H.; Hofmann, T. Bioappearance and pharmacokinetics of bioactives upon coffee consumption. Anal. Bioanal. Chem. 2013, 405, 8487–8503. [Google Scholar] [CrossRef] [PubMed]
- Mohr, S.; Liew, C.C. The peripheral-blood transcriptome: New insights into disease and risk assessment. Trends Mol. Med. 2007, 13, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Whitney, A.R.; Diehn, M.; Popper, S.J.; Alizadeh, A.A.; Boldrick, J.C.; Relman, D.A.; Brown, P.O. Individuality and variation in gene expression patterns in human blood. Proc. Natl. Acad. Sci. USA 2003, 100, 1896–1901. [Google Scholar] [CrossRef] [PubMed]
| All Women (n = 958) | Low Coffee Consumers (n = 132, 13.8%) | Moderate Coffee Consumers (n = 422, 44.1%) | High Coffee Consumers (n = 404, 42.2%) | |
|---|---|---|---|---|
| Age, years **,a | 55 (48–61, 958) | 53 (47–60, 132) | 55 (48–61, 422) | 54 (48–60, 404) |
| BMI | 25.1 (20.2–33.4, 941) | 25.0 (19.8–33.7, 130) | 25.0 (20.3–32.8, 418) | 25.3 (20.2–33.8, 393) |
| Education (years in school) **,a,b,c | 12 (8–18, 916) | 13 (8–19, 128) | 12 (8–19, 398) | 11 (8–18, 390) |
| Smoking week before blood sample **,a,c | ||||
| Yes % (n) | 25.1% (240) | 14.4% (19) | 17.3% (73) | 36.8% (148) |
| No % (n) | 74.9% (716) | 85.6% (113) | 82.7% (349) | 63.2% (254) |
| Physical activity level | 6 (3–9, 894) | 6 (3–9, 122) | 6 (3–9, 394) | 6 (2–8, 378) |
| Energy intake (kJ/d) | 7106 (4274–10409, 958) | 7052 (4087–10705, 132) | 7057 (4504–10094, 422) | 7172 (4050–10817, 404) |
| Alcohol consumption (g/d) | 2 (0–12, 958) | 2 (0–13, 132) | 2 (0–12, 422) | 2 (0–12, 404) |
| Dairy intake (g/d) **,a,b | 169 (15–635, 958) | 128 (6–614, 132) | 175 (20–634, 422) | 179 (16–641, 404) |
| Black and green tea consumption (g/d) **,a,b,c | 135 (0–945, 703) | 405 (0–1512, 99) | 135 (0–675, 309) | 0 (0–675, 295) |
| Red and processed meat intake (g/d) **,c | 89 (15–176, 958) | 86 (10–174, 132) | 86 (18–161, 422) | 93 (23–185, 404) |
| Low Tea, Nonsmoking Women (n = 298) | Low Coffee Consumers (n = 25, 8.4%) | Moderate Coffee Consumers (n = 139, 46.6%) | High Coffee Consumers (n = 134, 45.0%) | |
|---|---|---|---|---|
| Age, years | 55 (47–61, 298) | 55 (47–61, 25) | 55 (47–61, 139) | 55 (47–61, 134) |
| BMI | 25.7 (20.4–33.0, 293) | 25.8 (19.5–34.4, 24) | 25.4 (20.4–33.0, 138) | 26.2 (20.7–32.7, 131) |
| Education (years in school) | 12 (8–18, 288) | 14 (8–19, 24) | 12 (8–18, 133) | 12 (8–18, 131) |
| Physical activity level | 6 (3–9, 282) | 7 (2–9, 24) | 6 (3–9, 131) | 6 (3–8, 127) |
| Energy intake (kJ/d) **,b,c | 6911 (4251–10455, 298) | 6450(3742–9486, 25) | 6625 (4440–9458, 139) | 7188 (4309–10995, 134) |
| Alcohol consumption (g/d) | 2 (0–12, 298) | 3(0–13, 25) | 2 (0–13, 139) | 2 (0–9, 134) |
| Dairy intake (g/d) | 188 (24–617, 298) | 110 (18–491, 25) | 191 (24–618, 139) | 199 (28–623, 134) |
| Red and processed meat intake (g/d) | 90 (12–169, 298) | 80 (2–170, 25) | 87 (14–148, 139) | 95 (14–186, 134) |
| Gene Symbol | Gene Name | Log Fold Change | Average Expression | T | p-Value |
|---|---|---|---|---|---|
| TLE3 | Transducin like enhancer of split 3 | −0.318 | 6.907 | −4.682 | 0.000004 |
| HLX | H2.0 like homeobox | −0.299 | 7.276 | −4.577 | 0.000007 |
| DDX18 | DEAD-box helicase 18 | 0.282 | 8.330 | 4.536 | 0.000008 |
| YRDC | YrdC N6-threonylcarbamoyltransferase domain containing | 0.159 | 7.034 | 4.415 | 0.000014 |
| KDM6B | Lysine demethylase 6B | −0.294 | 7.003 | −4.401 | 0.000015 |
| CANT1 | Calcium activated nucleotidase 1 | −0.264 | 8.524 | −4.397 | 0.000015 |
| WDR61 | WD repeat domain 61 | 0.213 | 7.608 | 4.387 | 0.000016 |
| MTSS1 | MTSS1, I-BAR domain containing | 0.260 | 7.383 | 4.375 | 0.000017 |
| MACF1 | Microtubule-actin crosslinking factor 1 | 0.248 | 7.815 | 4.336 | 0.000020 |
| PPP3CC | Protein phosphatase 3 catalytic subunit gamma | 0.241 | 7.482 | 4.284 | 0.000025 |
| FAM36A | Cytochrome c oxidase assembly factor | 0.190 | 6.781 | 4.274 | 0.000026 |
| TXK | TXK tyrosine kinase | 0.265 | 7.023 | 4.261 | 0.000027 |
| TFE3 | Transcription factor binding to IGHM enhancer 3 | −0.198 | 7.043 | −4.230 | 0.000031 |
| SPATA2L | Spermatogenesis associated 2 like | −0.220 | 7.134 | −4.204 | 0.000035 |
| DYSF | Dysferlin | −0.529 | 9.697 | −4.197 | 0.000036 |
| TTC13 | Tetratricopeptide repeat domain 13 | 0.219 | 7.541 | 4.193 | 0.000036 |
| LOC642684 | - | −0.136 | 6.339 | −4.188 | 0.000037 |
| CDK5RAP1 | CDK5 regulatory subunit associated protein 1 | 0.166 | 7.422 | 4.178 | 0.000039 |
| LOC441124 | - | −0.261 | 7.219 | −4.175 | 0.000039 |
| PHOSPHO1 | Phosphoethanolamine/phosphocholine phosphatase | −0.400 | 7.532 | −4.169 | 0.000040 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
B. Barnung, R.; H. Nøst, T.; Ulven, S.M.; Skeie, G.; S. Olsen, K. Coffee Consumption and Whole-Blood Gene Expression in the Norwegian Women and Cancer Post-Genome Cohort. Nutrients 2018, 10, 1047. https://doi.org/10.3390/nu10081047
B. Barnung R, H. Nøst T, Ulven SM, Skeie G, S. Olsen K. Coffee Consumption and Whole-Blood Gene Expression in the Norwegian Women and Cancer Post-Genome Cohort. Nutrients. 2018; 10(8):1047. https://doi.org/10.3390/nu10081047
Chicago/Turabian StyleB. Barnung, Runa, Therese H. Nøst, Stine M. Ulven, Guri Skeie, and Karina S. Olsen. 2018. "Coffee Consumption and Whole-Blood Gene Expression in the Norwegian Women and Cancer Post-Genome Cohort" Nutrients 10, no. 8: 1047. https://doi.org/10.3390/nu10081047
APA StyleB. Barnung, R., H. Nøst, T., Ulven, S. M., Skeie, G., & S. Olsen, K. (2018). Coffee Consumption and Whole-Blood Gene Expression in the Norwegian Women and Cancer Post-Genome Cohort. Nutrients, 10(8), 1047. https://doi.org/10.3390/nu10081047






