Attenuating Pregnancy Weight Gain—What Works and Why: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Sources
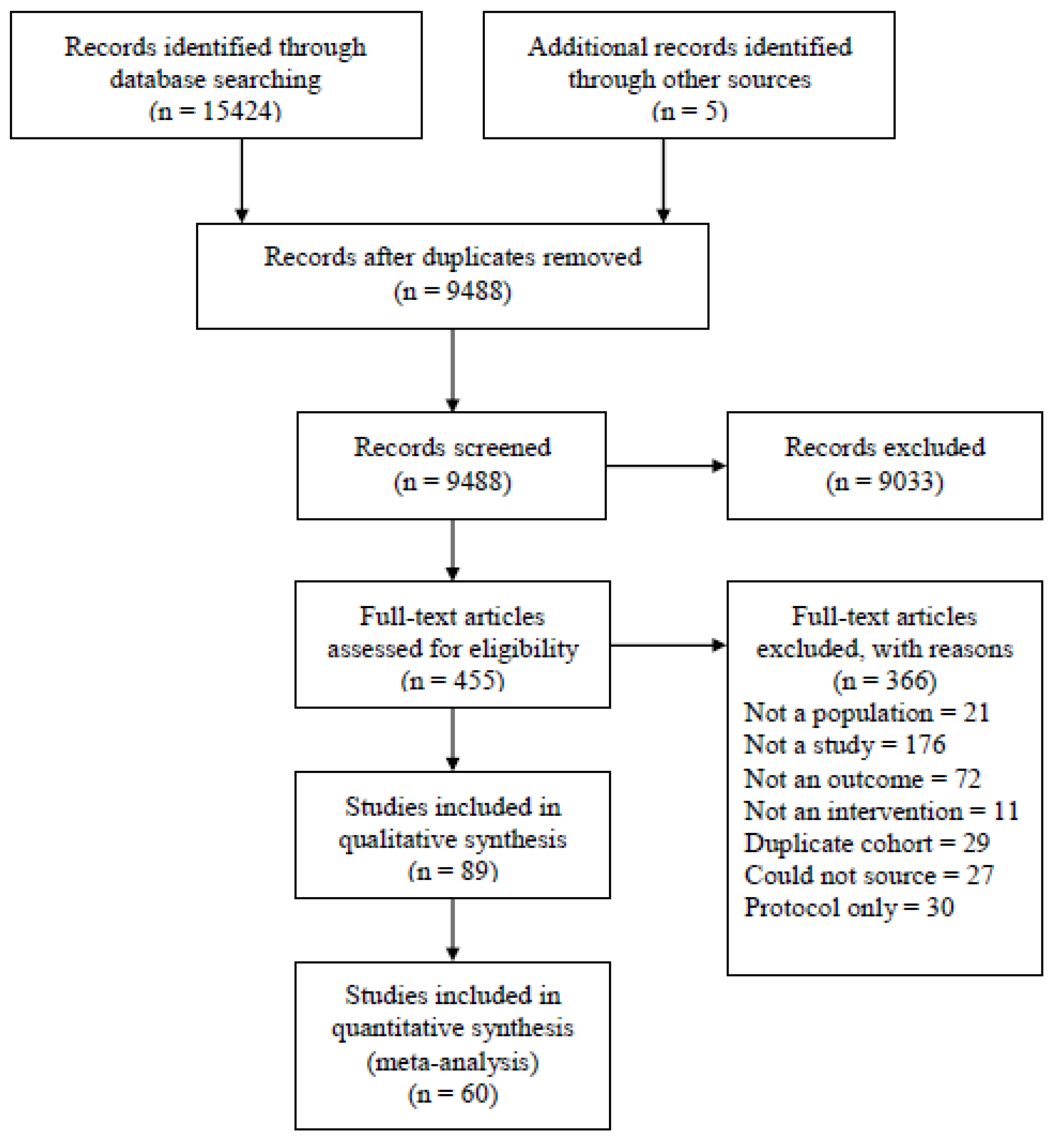
2.2. Study Selection
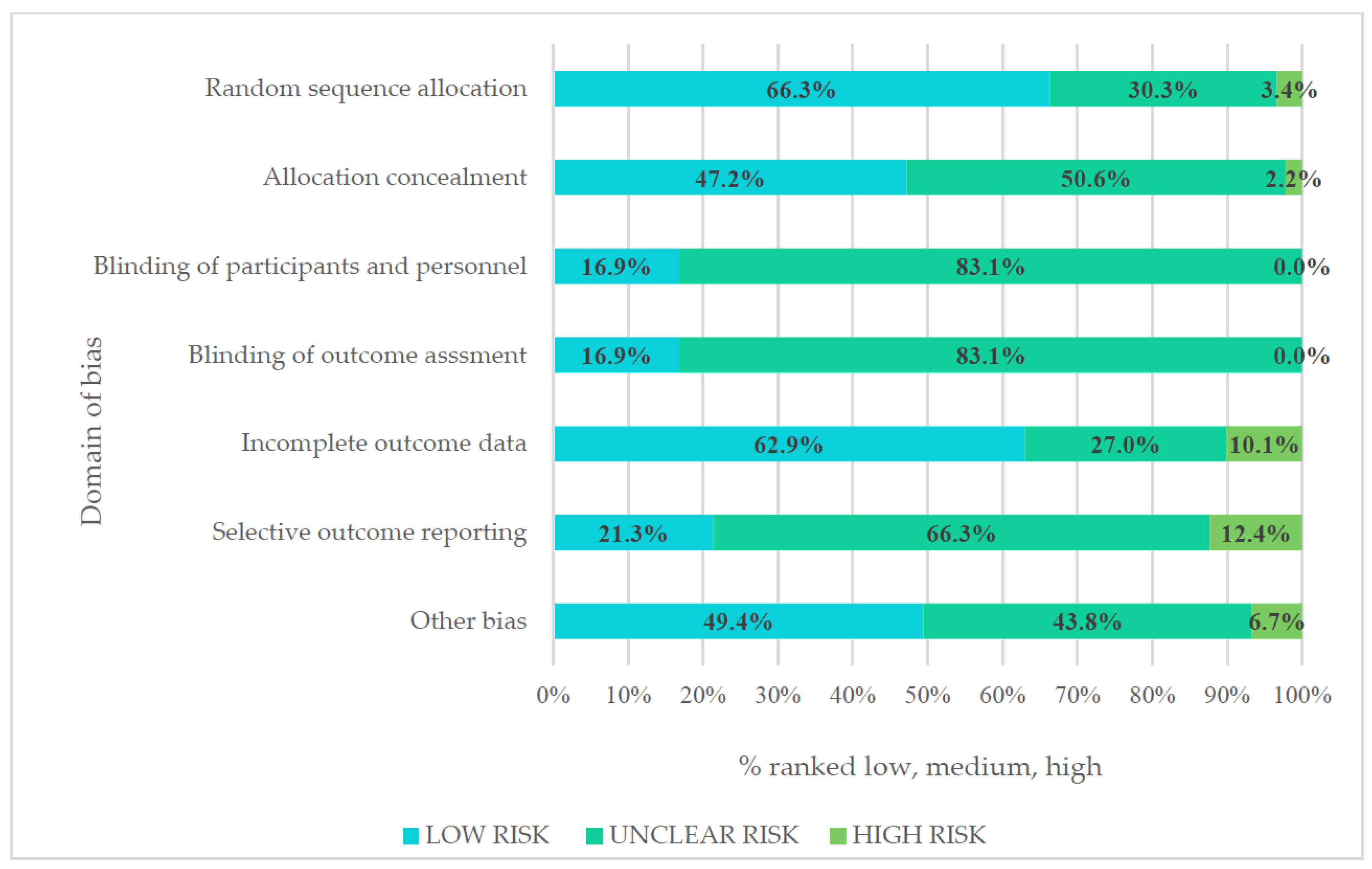
2.3. Assessment of Risk of Bias in Included Studies
2.4. Statistical Analyses
3. Results
3.1. Diet Interventions
3.2. Physical Activity
3.3. Lifestyle Interventions (Combining Both Diet and PA), Delivered Face-to-Face
3.4. eHealth Interventions
3.5. ‘Other’ Interventions
3.6. Effectiveness of Interventions According to Their Characteristics
3.7. Risk of Bias
3.8. Sensitivity Analyses
4. Discussion
4.1. Dietary Interventions
4.2. Individual, Group or Mixed Delivery?
4.3. Intensity and Frequency of Interventions
4.4. Trimester and Duration of Interventions
4.5. Future Directions
4.6. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Catalano, P.; De Mouzon, S. Maternal obesity and metabolic risk to the offspring: Why lifestyle interventions may have not achieved the desired outcomes. Int. J. Obes. 2015, 39, 642–649. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, K.; Abrams, B.; Bodnar, L.; Butte, N.; Catalano, P.; Siega-Riz, A.M. Recommendations for weight gain during pregnancy in the context of the obesity epidemic. Obstet. Gynecol. 2010, 116, 1191. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US); National Research Council (US). Committee reexamine IOM pregnancy weight guidelines. In Weight Gain during Pregnancy: Rexamining the Guidelines; Rasmussen, K.M., Yaktine, A.L., Eds.; National Acadamies Press (US): Washington, DC, USA, 2009. [Google Scholar] [CrossRef]
- De Jersey, S.; Nicholson, J.; Callaway, L.; Daniels, L. A prospective study of pregnancy weight gain in Australian women. Aust. N. Z. J. Obstet. Gynaecol. 2012, 52, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Simas, T.; Liao, X.; Garrison, A.; Sullivan, G.; Howard, A.; Hardy, J. Impact of updated Institute of Medicine guidelines on prepregnancy body mass index categorization, gestational weight gain recommendations, and needed counseling. J. Womens Health 2011, 20, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Truong, Y.; Yee, L.; Caughey, A.; Cheng, Y. Weight gain in pregnancy: Does the Institute of Medicine have it right? Am. J. Obstet. Gynecol. 2015, 212, 362. [Google Scholar] [CrossRef] [PubMed]
- Van Der Pligt, P.; Willcox, J.; Hesketh, K.; Ball, K.; Wilkinson, S.; Crawford, D.; Campbell, K. Systematic review of lifestyle interventions to limit postpartum weight retention: Implications for future opportunities to prevent maternal overweight and obesity following childbirth. Obes. Rev. 2013, 14, 792–805. [Google Scholar] [CrossRef] [PubMed]
- Paul, K.; Graham, M.; Olson, C. The web of risk factors for excessive gestational weight gain in low income women. Matern. Child Health J. 2013, 17, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Schack-Nielsen, L.; Michaelsen, K.; Gamborg, M.; Mortensen, E.; Sørensen, T. Gestational weight gain in relation to offspring body mass index and obesity from infancy through adulthood. Int. J. Obes. 2010, 34, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Siega-Riz, A.M.; Viswanathan, M.; Moos, M.; Deierlein, A.; Mumford, S.; Knaack, J.; Theida, P.; Lux, L.J.; Lohr, K.N. A systematic review of outcomes of maternal weight gain according to the Institute of Medicine recommendations: Birthweight, fetal growth, and postpartum weight retention. Am. J. Obstet. Gynecol. 2009, 201, 339. [Google Scholar] [CrossRef] [PubMed]
- Poston, L. Gestational weight gain: Influences on the long-term health of the child. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Hedderson, M.; Gunderson, E.; Ferrara, A. Gestational weight gain and risk of gestational diabetes mellitus. Obstet. Gynecol. 2010, 115, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Fortner, R.; Pekow, P.; Solomon, C.; Markenson, G.; Chasan-Taber, L. Prepregnancy body mass index, gestational weight gain, and risk of hypertensive pregnancy among Latina women. Am. J. Obstet. Gynecol. 2009, 200, 167. [Google Scholar] [CrossRef] [PubMed]
- Stotland, N.; Hopkins, L.; Caughey, A. Gestational weight gain, macrosomia, and risk of cesarean birth in nondiabetic nulliparas. Obstet. Gynecol. 2004, 104, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, R.; Abell, S.; Ranasinha, S.; Misso, M.; Boyle, J.; Black, M.; Li, N.; Hu, G.; Corrado, F.; Rode, L.; et al. Association of Gestational Weight Gain With Maternal and Infant Outcomes: A Systematic Review and Meta-analysis. JAMA 2017, 317, 2207–2225. [Google Scholar] [CrossRef] [PubMed]
- Olson, C.M. Achieving a healthy weight gain during pregnancy. Annu. Rev. Nutr. 2008, 28, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Langley-Evans, S. Nutrition in early life and the programming of adult disease: A review. J. Hum. Nutr. Diet. 2015, 28, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Brunner, S.; Stecher, L.; Ziebarth, S.; Nehring, I.; Rifas-Shiman, S.; Sommer, C.; Hauner, H.; von Kries, R. Excessive gestational weight gain prior to glucose screening and the risk of gestational diabetes: A meta-analysis. Diabetologia 2015, 58, 2229–2237. [Google Scholar] [CrossRef] [PubMed]
- Tanentsapf, I.; Heitmann, B.; Adegboye, A. Systematic review of clinical trials on dietary interventions to prevent excessive weight gain during pregnancy among normal weight, overweight and obese women. BMC Pregnancy Childbirth 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Muktabhant, B.; Lumbiganon, P.; Ngamjarus, C.; Dowswell, T. Interventions for preventing excessive weight gain during pregnancy. Cochrane Library 2012. [Google Scholar] [CrossRef]
- Streuling, I.; Beyerlein, A.; Von Kries, R. Can gestational weight gain be modified by increasing physical activity and diet counseling? A meta-analysis of interventional trials. Am. J. Clin. Nutr. 2010, 92, 678. [Google Scholar] [CrossRef] [PubMed]
- Thangaratinam, S.; Rogozińska, E.; Jolly, K.; Glinkowski, S.; Roseboom, T.; Tomlinson, J.; Kunz, R.; Mol, B.W.; Coomarasamy, A.; Khan, K.S. Effects of interventions in pregnancy on maternal weight and obstetric outcomes: Meta-analysis of randomised evidence. BMJ 2012, 344, e2088. [Google Scholar] [CrossRef] [PubMed]
- Dodd, J.; Grivell, R.; Crowther, C.; Robinson, J. Antenatal interventions for overweight or obese pregnant women: A systematic review of randomised trials. BJOG 2010, 117, 1316–1326. [Google Scholar] [CrossRef] [PubMed]
- Skouteris, H.; Hartley-Clark, L.; McCabe, M.; Milgrom, J.; Kent, B.; Herring, S.; Gale, J. Preventing excessive gestational weight gain: A systematic review of interventions. Obes. Rev. 2010, 11, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Campbell, F.; Johnson, M.; Messina, J.; Guillaume, L.; Goyder, E. Behavioural interventions for weight management in pregnancy: A systematic review of quantitative and qualitative data. BMC Public Health 2011, 11, 491. [Google Scholar] [CrossRef] [PubMed]
- Haby, K.; Berg, M.; Gyllensten, H.; Hanas, R.; Premberg, A. Mighty Mums—A lifestyle intervention at primary care level reduces gestational weight gain in women with obesity. BMC Obes. 2018, 5, 16. [Google Scholar] [CrossRef] [PubMed]
- Yeo, S.; Samuel-Hodge, C.D.; Smith, R.; Leeman, J.; Ferraro, A.M.; Asafu-Adjei, J.K. Challenges of integrating an evidence-based intervention in health departments to prevent excessive gestational weight gain among low-income women. Public Health Nurs. 2016, 33, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Eysenbach, G. What is e-health? J. Med. Internet Res. 2001, 3, e20. [Google Scholar] [CrossRef] [PubMed]
- Syngelaki, A.; Nicolaides, K.; Balani, J.; Hyer, S.; Akolekar, R.; Kotecha, R.; Pastides, A.; Shehata, H. Metformin versus Placebo in Obese Pregnant Women without Diabetes Mellitus. N. Engl. J. Med. 2016, 374, 434–443. [Google Scholar] [CrossRef] [PubMed]
- Brownfoot, F.; Davey, M.; Kornman, L.; Brownfoot, F. Routine weighing to reduce excessive antenatal weight gain: A randomised controlled trial. BJOG 2016, 123, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Rogozinska, E.; Marlin, N.; Betran, A.; Astruup, A.; Barakat, R.; Bogaerts, A.; Cecatti, J.C.; Devlieger, R.; Dodd, J.M.; El Beltagy, N. Effect of diet and physical activity based interventions in pregnancy on gestational weight gain and pregnancy outcomes: A meta-analysis of individual patient data from randomised trials. BMJ 2017, 358. [Google Scholar] [CrossRef]
- Bennett, C.; Walker, R.; Blumfield, M.; Gwini, S.; Ma, J.; Wang, F.; Wan, Y.; Truby, H. Interventions designed to reduce excessive gestational weight gain can reduce the incidence of gestational diabetes mellitus: A systematic review and meta-analysis of randomised controlled trials. Diabetes Res. Clin. Pract. 2018, 141, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Clapp, J.; Kim, H.; Burciu, B.; Schmidt, S.; Petry, K.; Lopez, B. Continuing regular exercise during pregnancy: Effect of exercise volume on fetoplacental growth. Am. J. Obstet. Gynecol. 2002, 186, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Nobles, C.; Marcus, B.; Stanek, E.; Braun, B.; Whitcomb, B.; Manson, J.; et al. The Effect of an Exercise Intervention on Gestational Weight Gain: The Behaviors Affecting Baby and You (B.A.B.Y.) Study: A Randomized Controlled Trial. Am. J. Health Promot. 2018, 32, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Markovic, T.; Muirhead, R.; Overs, S.; Ross, G.; Louie, J.; Kizirian, N.; et al. Randomized Controlled Trial Investigating the Effects of a Low-Glycemic Index Diet on Pregnancy Outcomes in Women at High Risk of Gestational Diabetes Mellitus: The GI Baby 3 Study. Diabetes Care 2016, 39, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Peccei, A.; Blake-Lamb, T.; Rahilly, D.; Hatoum, I.; Bryant, A. Intensive prenatal nutrition counseling in a community health setting: A randomized controlled trial. Obstet. Gynecol. 2017, 130, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, E.; Pawlak, D.; Takoudes, T.; Ebbeling, C.; Feldman, H.; Lovesky, M.; Cooke, E.A.; Leidig, M.M.; Ludwig, D.S. Effects of a low-glycemic load diet in overweight and obese pregnant women: A pilot randomized controlled trial. Am. J. Clin. Nutr. 2010, 92, 1306–1315. [Google Scholar] [CrossRef] [PubMed]
- Cahill, A.; Haire-Joshu, D.; Cade, W.; Stein, R.; Woolfolk, C.; Moley, K.; Mathur, A.; Schechtman, K.; Klein, S. Weight control program and gestational weight gain in disadvantaged women with overweight or obesity: A randomized clinical trial. Obesity 2018, 26, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Pawalia, A.; Kulandaivelan, S.; Savant, S.; Yadav, V. Exercise in pregnancy: Effect on obesity parameters in indian women—A randomized controlled trial. Rom. J. Diabetes Nutr. Metab. Dis. 2017, 24, 315–323. [Google Scholar] [CrossRef]
- Phelan, S.; Wing, R.; Brannen, A.; McHugh, A.; Hagobian, T.; Schaffner, A.; Jelalian, E.; Hart, C.N.; Scholl, T.O.; Munoz-Christian, K.; et al. Randomized controlled clinical trial of behavioral lifestyle intervention with partial meal replacement to reduce excessive gestational weight gain. Am. J. Clin. Nutr. 2018, 107, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Simmons, D.; Jelsma, J.; Galjaard, S.; Devlieger, R.; van Assche, A.; Jans, G.; Corcoy, R.; Adelantado, J.M.; Dunne, F.; Desoye, G.; et al. Results from a European multicenter randomized trial of physical activity and/or healthy eating to reduce the risk of gestational diabetes mellitus: The DALI lifestyle pilot. Diabetes Care 2015, 38, 1650–1656. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.; Tussing-Humphreys, L.; Goodman, M.; Olender, S. Gestational weight gain: Results from the delta healthy sprouts comparative impact trial. J. Pregnancy 2016, 2016, 12. [Google Scholar] [CrossRef] [PubMed]
- Moses, R.; Casey, S.; Quinn, E.; Cleary, J.; Tapsell, L.; Milosavljevic, M.; Petocz, P.; Brand-Miller, J.C. Pregnancy and Glycemic Index Outcomes study: Effects of low glycemic index compared with conventional dietary advice on selected pregnancy outcomes. Am. J. Clin. Nutr. 2014, 99, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Perales, M.; Cordero, Y.; Vargas, M.; Lucia, A.; Barakat, R. Exercise and depression in overweight and obese pregnant women: A randomised controlled trial. Arch. Med. Deporte 2015, 32, 156–163. [Google Scholar]
- Abdel-Aziz, S.; Hegazy, I.; Mohamed, D.; Abu El Kasem, M.; Hagag, S. Effect of dietary counseling on preventing excessive weight gain during pregnancy. Public Health 2018, 154, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Vítolo, M.; Bueno, M.; Gama, C. Impact of a dietary counseling program on the gain weight speed of pregnant women attended in a primary care service. Rev. Bras. Ginecol. Obstet. 2011, 33, 58–59. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Vélez, R.; Lobelo, F.; Aguilar-de Plata, A.; Izquierdo, M.; García-Hermoso, A. Exercise during pregnancy on maternal lipids: A secondary analysis of randomized controlled trial. BMC Pregnancy Childbirth 2017, 17, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Renault, K.; Nørgaard, K.; Nilas, L.; Carlsen, E.; Cortes, D.; Pryds, O.; Secher, N.J. The Treatment of Obese Pregnant Women (TOP) study: A randomized controlled trial of the effect of physical activity intervention assessed by pedometer with or without dietary intervention in obese pregnant women. Am. J. Obstet. Gynecol. 2014, 210, 134. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ma, J.; Yang, H. Lifestyle intervention for gestational diabetes mellitus prevention: A cluster-randomized controlled study. Chronic Dis. Transl. Med. 2015, 1, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Bosaeus, M.; Hussain, A.; Karlsson, T.; Andersson, L.; Hulthen, L.; Svelander, C.; Sandberg, A.S.; Larsson, I.; Ellegard, L.; Holmang, A. A randomized longitudinal dietary intervention study during pregnancy: Effects on fish intake, phospholipids, and body composition. Nutrition 2015, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Vinter, C.; Jensen, D.; Ovesen, P.; Beck-Nielsen, H.J.O. Lifestyle and pregnancy (LIP) study: The clinical effect of lifestyle intervention during pregnancy in obese women. Diabetes 2011, 60, 348–349. [Google Scholar] [CrossRef]
- Sterne, J. Meta-Analysis in Stata: An Updated Collection from the Stata Journal; CRC PRESS: Boca Raton, FL, USA, 2009. [Google Scholar]
- Egger, M.; Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, M. Introduction to meta-analysis; John Wiley and Sons: Chichester, UK, 2009. [Google Scholar]
- Racey, M.; O’Brien, C.; Douglas, S.; Marquez, O.; Hendrie, G.; Newton, G. Systematic review of school-based interventions to modify dietary behavior: Does intervention intensity impact effectiveness? J. Sch. Health 2016, 86, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Hendrie, G.A.; Brindal, E.; Baird, D.; Gardner, C. Improving children’s dairy food and calcium intake: Can intervention work? A systematic review of the literature. Public Health Nutr. 2013, 16, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Hingle, M.D.; O’Connor, T.M.; Dave, J.M.; Baranowski, T. Parental involvement in interventions to improve child dietary intake: A systematic review. Prev. Med. 2010, 51, 103–111. [Google Scholar] [CrossRef] [PubMed]
- StataCorp. Stata Statistical Software: Release 12; StataCorp LP: College Station, TX, USA, 2011. [Google Scholar]
- Korpi-Hyovalti, E.; Schwab, U.; Laaksonen, D.E.; Linjama, H.; Heinonen, S.; Niskanen, L. Effect of intensive counselling on the quality of dietary fats in pregnant women at high risk of gestational diabetes mellitus. Br. J. Nutr. 2012, 108, 910–917. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, E.; Walker, S.; Ugoni, A.; Lappas, M.; Leong, O.; Shub, A. Self-weighing and simple dietary advice for overweight and obese pregnant women to reduce obstetric complications without impact on quality of life: A randomised controlled trial. BJOG 2016, 123, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.; McGowan, C.; Mahony, R.; Foley, M.; McAuliffe, F. Low glycaemic index diet in pregnancy to prevent macrosomia (ROLO study): Randomised control trial. BMJ 2012, 345, e5605. [Google Scholar] [CrossRef] [PubMed]
- Thornton, Y.; Smarkola, C.; Kopacz, S.; Ishoof, S. Perinatal outcomes in nutritionally monitored obese pregnant women: A randomized clinical trial. J. Natl. Med. Assoc. 2009, 101, 569–577. [Google Scholar] [CrossRef]
- Deveer, R.; Deveer, M.; Akbaba, E.; Engin-Üstün, Y.; Aydoǧan, P.; Çelikkaya, H.; Danisman, N.; Mollamahmutoglu, L. The effect of diet on pregnancy outcomes among pregnants with abnormal glucose challenge test. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1258–1261. [Google Scholar] [PubMed]
- Di Carlo, C.; Iannotti, G.; Sparice, S.; Chiacchio, M.; Greco, E.; Tommaselli, G.; Nappi, C. The role of a personalized dietary intervention in managing gestational weight gain: A prospective, controlled study in a low-risk antenatal population. Arch. Gynecol. Obstet. 2014, 289, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Bonomo, M.; Corica, D.; Mion, E.; Goncalves, D.; Motta, G.; Merati, R.; Ragusa, A.; Morabito, A. Evaluating the therapeutic approach in pregnancies complicated by borderline glucose intolerance: A randomized clinical trial. Diabet. Med. 2005, 22, 1536–1541. [Google Scholar] [CrossRef] [PubMed]
- Wolff, S.; Legarth, J.; Vangsgaard, K.; Toubro, S.; Astrup, A. A randomized trial of the effects of dietary counseling on gestational weight gain and glucose metabolism in obese pregnant women. Int. J. Obes. 2008, 32, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Ilmonen, J.; Isolauri, E.; Poussa, T.; Laitinen, K. Impact of dietary counselling and probiotic intervention on maternal anthropometric measurements during and after pregnancy: A randomized placebo-controlled trial. Clin. Nutr. 2011, 30, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Cordero, Y.; Coteron, J.; Luaces, M.; Montejo, R. Exercise during pregnancy improves maternal glucose screen at 24–28 weeks: A randomised controlled trial. Br. J. Sports Med. 2012, 46, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Lucia, A.; Ruiz, J. Resistance exercise training during pregnancy and newborn’s birth size: A randomised controlled trial. Int. J. Obes. 2009, 33, 9. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Pelaez, M.; Cordero, Y.; Perales, M.; Lopez, C.; Coteron, J.; Mottola, M.F. Exercise during pregnancy protects against hypertension and macrosomia: Randomized clinical trial. Am. J. Obstet. Gynecol. 2016, 214, 649-e1. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Pelaez, M.; Lopez, C.; Lucia, A.; Ruiz, J.R. Exercise during pregnancy and gestational diabetes-related adverse effects: A randomised controlled trial. Br. J. Sports Med. 2013, 47, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Pelaez, M.; Montejo, R.; Luaces, M.; Zakynthinaki, M. Exercise during pregnancy improves maternal health perception: A randomized controlled trial. Am. J. Obstet. Gynecol. 2011, 204, 402. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Pelaez, M.; Montejo, R.; Refoyo, I.; Coteron, J. Exercise throughout pregnancy does not cause preterm delivery: A randomized, controlled trial. J. Phys. Act. Health 2014, 11, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.; Perales, M.; Pelaez, M.; Lopez, C.; Lucia, A.; Barakat, R. Supervised exercise-based intervention to prevent excessive gestational weight gain: A randomized controlled trial. Mayo Clin. Proc. 2013, 88, 1388–1397. [Google Scholar] [CrossRef] [PubMed]
- Clapp, J.; Kim, H.; Burciu, B.; Lopez, B. Beginning regular exercise in early pregnancy: Effect on fetoplacental growth. Am. J. Obstet. Gynecol. 2000, 183, 1484–1488. [Google Scholar] [CrossRef] [PubMed]
- Garshasbi, A.; Faghih Zadeh, S. The effect of exercise on the intensity of low back pain in pregnant women. Int. J. Gynecol. Obstet. 2005, 88, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, S.; Cecatti, J.; Pereira, R.; Baciuk, E.; Bernardo, A.; Silveira, C. Water aerobics II: Maternal body composition and perinatal outcomes after a program for low risk pregnant women. Reprod. Health 2009, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Haakstad, L.; Bø, K. Effect of regular exercise on prevention of excessive weight gain in pregnancy: A randomised controlled trial. Eur. J. Contracept. Reprod. Health Care 2011, 16, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, S.; Surita, F.; Parpinelli, M.; Siani, S.; Pinto e Silva, J. The effect of an antenatal physical exercise programme on maternal/perinatal outcomes and quality of life in overweight and obese pregnant women: A randomised clinical trial. BJOG 2011, 118, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Kong, K.; Campbell, C.; Foster, R.; Peterson, A.; Lanningham-Foster, L. A pilot walking program promotes moderate-intensity physical activity during pregnancy. Med. Sci. Sports Exerc. 2014, 46, 462–471. [Google Scholar] [CrossRef] [PubMed]
- Bisson, M.; Almeras, N.; Dufresne, S.; Robitaille, J.; Rheaume, C.; Bujold, E.; Frenette, J.; Tremblay, A.; Marc, I. A 12-week exercise program for pregnant women with obesity to improve physical activity levels: An open randomised preliminary study. PLoS ONE 2015, 10, e0137742. [Google Scholar] [CrossRef] [PubMed]
- Ronnberg, A.; Ostlund, I.; Fadl, H.; Gottvall, T.; Nilsson, K. Intervention during pregnancy to reduce excessive gestational weight gain-a randomised controlled trial. BJOG 2015, 122, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Dekker Nitert, M.; Barrett, H.; Denny, K.; McIntyre, H.; Callaway, L. Exercise in pregnancy does not alter gestational weight gain, MCP-1 or leptin in obese women. Aust. N. Z. J. Obstet. Gynaecol. 2015, 55, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Oostdam, N.; Van Poppel, M.; Wouters, M.; Eekhoff, E.; Bekedam, D.; Kuchenbecker, W.; Quartero, H.; Heres, M.; van Mechelen, W. No effect of the FitFor2 exercise programme on blood glucose, insulin sensitivity, and birthweight in pregnant women who were overweight and at risk for gestational diabetes: Results of a randomised controlled trial. BJOG 2012, 119, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Bacchi, M.; Mottola, M.; Perales, M.; Refoyo, I.; Barakat, R. Aquatic activities during pregnancy prevent excessive maternal weight gain and preserve birth weight: A randomized clinical trial. Am. J. Health Promot. 2018, 32, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Franco, E.; Perales, M.; Lopez, C.; Mottola, M. Exercise during pregnancy is associated with shorter duration of labor: A randomized clinical trial. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 224, 33–40. [Google Scholar] [CrossRef] [PubMed]
- da Silva, S.; Hallal, P.; Domingues, M.; Bertoldi, A.; Silveira, M.; Bassani, D.; da Silva, I.C.; da Sliva, B.C.; Coll, C.D.; Evenson, K. A randomized controlled trial of exercise during pregnancy on maternal and neonatal outcomes: Results from the PAMELA study. Int. J. Behav. Nutr. Phys. Act. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Daly, N.; Farren, M.; McKeating, A.; O’Kelly, R.; Stapleton, M.; Turner, M.; et al. A medically supervised pregnancy exercise intervention in obese women: A randomized controlled trial. Obstet. Gynecol. 2017, 130, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Garnæs, K.; Mørkved, S.; Salvesen, Ø.; Moholdt, T. Exercise training and weight gain in obese pregnant women: A randomized controlled trial (ETIP Trial). PLoS Med. 2016, 13, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wei, Y.; Zhang, X.; Zhang, Y.; Xu, Q.; Sun, Y.; Su, S.; Zhang, L.; Liu, C.; Feng, Y.; et al. A randomized clinical trial of exercise during pregnancy to prevent gestational diabetes mellitus and improve pregnancy outcome in overweight and obese pregnant women. Am. J. Obstet. Gynecol. 2017, 216, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Blanque, R.; Carlos Sánchez-García, J.; Manuel Sánchez-López, A.; Mur-Villar, N.; Fernández-Castillo, R.; José Aguilar-Cordero, M. Influence of physical exercise during pregnancy on birthweight: A randomized clinical trial. Nutr. Hosp. 2017, 34, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Petrella, E.; Malavolti, M.; Bertarini, V.; Pignatti, L.; Neri, I.; Battistini, N.C.; Facchinetti, F. Gestational weight gain in overweight and obese women enrolled in a healthy lifestyle and eating habits program. J. Matern. Neonatal Med. 2014, 27, 1348–1352. [Google Scholar] [CrossRef] [PubMed]
- Phelan, S.; Phipps, M.; Abrams, B.; Darroch, F.; Schaffner, A.; Wing, R. Randomized trial of a behavioral intervention to prevent excessive gestational weight gain: The Fit for Delivery Study. Am. J. Clin. Nutr. 2011, 93, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Polley, B.; Wing, R.; Sims, C. Randomized controlled trial to prevent excessive weight gain in pregnant women. Int. J. Obes. 2002, 26, 1494–1502. [Google Scholar] [CrossRef] [PubMed]
- Poston, L.; Bell, R.; Croker, H.; Flynn, A.; Godfrey, K.; Goff, L.; Hayes, L.; Khazaezadeh, N.; Nelson, S.M.; Oteng-Ntim, E.; et al. Effect of a behavioural intervention in obese pregnant women (the UPBEAT study): A multicentre, randomised controlled trial. Lancet Diabetes Endocrinol. 2015, 3, 767–777. [Google Scholar] [CrossRef]
- Asbee, S.; Jenkins, T.; Butler, J.; White, J.; Elliot, M.; Rutledge, A. Preventing excessive weight gain during pregnancy through dietary and lifestyle counseling: A randomized controlled trial. Obstet. Gynecol. 2009, 113, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Aşcı, Ö.; Rathfisch, G. Effect of lifestyle interventions of pregnant women on their dietary habits, lifestyle behaviors, and weight gain: A randomized controlled trial. J. Health Popul. Nutr. 2016, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Guelinckx, I.; Devlieger, R.; Mullie, P.; Vansant, G. Effect of lifestyle intervention on dietary habits, physical activity, and gestational weight gain in obese pregnant women: A randomized controlled trial. Am. J. Clin. Nutr. 2010, 91, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Yeh, C.; Tsai, Y. A diet and physical activity intervention for preventing weight retention among Taiwanese childbearing women: A randomised controlled trial. Midwifery 2011, 27, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Hui, A.; Back, L.; Ludwig, S.; Gardiner, P.; Sevenhuysen, G.; Dean, H.; Sellers, E.; McGavock, J.; Morris, M.; Bruce, S.; et al. Lifestyle intervention on diet and exercise reduced excessive gestational weight gain in pregnant women under a randomized controlled trial. Obstet. Gynecol. Surv. 2012, 67, 263–264. [Google Scholar] [CrossRef]
- Hui, A.; Back, L.; Ludwig, S.; Gardiner, P.; Sevenhuysen, G.; Dean, H.; Sellers, E.; McGavock, J.; Morris, M.; Jiang, D.; et al. Effects of lifestyle intervention on dietary intake, physical activity level, and gestational weight gain in pregnant women with different pre-pregnancy Body Mass Index in a randomized control trial. BMC Pregnancy Childbirth 2014, 14, 331. [Google Scholar] [CrossRef] [PubMed]
- Ruchat, S.; Davenport, M.; Giroux, I.; Hillier, M.; Batada, A.; Sopper, M.; Hammond, J.M.; Mottola, M.F. Nutrition and exercise reduce excessive weight gain in normal-weight pregnant women. Med. Sci. Sports Exerc. 2012, 44, 1419–1426. [Google Scholar] [CrossRef] [PubMed]
- Bogaerts, A.; Devlieger, R.; Nuyts, E.; Witters, I.; Gyselaers, W.; Van den Bergh, B. Effects of lifestyle intervention in obese pregnant women on gestational weight gain and mental health: A randomized controlled trial. Int. J. Obes. 2013, 37, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Harrison, C.; Lombard, C.; Strauss, B.; Teede, H. Optimizing healthy gestational weight gain in women at high risk of gestational diabetes: A randomized controlled trial. Obesity 2013, 21, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, M.; Hosker, M.; Marcus, B.; Rosal, M.; Braun, B.; Stanek, E.; Markenson, G.; Chasan-Taber, L. A pregnancy lifestyle intervention to prevent gestational diabetes risk factors in overweight Hispanic women: A feasibility randomized controlled trial. Diabet. Med. 2015, 32, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Rauh, K.; Gabriel, E.; Kerschbaum, E.; Schuster, T.; von Kries, R.; Amann-Gassner, U.; Hauner, H. Safety and efficacy of a lifestyle intervention for pregnant women to prevent excessive maternal weight gain: A cluster-randomized controlled trial. BMC Pregnancy Childbirth 2013, 13, 151. [Google Scholar] [CrossRef] [PubMed]
- Gesell, S.; Katula, J.; Strickland, C.; Vitolins, M. Feasibility and initial efficacy evaluation of a community-based cognitive-behavioral lifestyle intervention to prevent excessive weight gain during pregnancy in Latina women. Matern. Child Health J. 2015, 19, 1842–1852. [Google Scholar] [CrossRef] [PubMed]
- Skouteris, H.; McPhie, S.; Hill, B.; McCabe, M.; Milgrom, J.; Kent, B.; Bruce, L.; Herring, S.; Gale, J.; Mihalopoulos, C.; et al. Health coaching to prevent excessive gestational weight gain: A randomized-controlled trial. Br. J. Health Psychol. 2016, 21, 31–51. [Google Scholar] [CrossRef] [PubMed]
- Sagedal, L.; Øverby, N.; Bere, E.; Torstveit, M.; Lohne-Seiler, H.; Småstuen, M.; Hillesund, E.R.; Henriksen, T.; Vistad, I. Lifestyle intervention to limit gestational weight gain: The Norwegian Fit for Delivery randomised controlled trial. BJOG 2016, 124, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Althuizen, E.; van der Wijden, C.; van Mechelen, W.; Seidell, J.; van Poppel, M. The effect of a counselling intervention on weight changes during and after pregnancy: A randomised trial. BJOG 2013, 120, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Dodd, J.; Turnbull, D.; McPhee, A.; Deussen, A.; Grivell, R.; Yelland, L.; Crowther, C.A.; Wittert, G.; Owens, J.A.; Robinson, J.S. Antenatal lifestyle advice for women who are overweight or obese: LIMIT randomised trial. BMJ 2014, 348, 1285. [Google Scholar] [CrossRef] [PubMed]
- Vesco, K.; Karanja, N.; King, J.; Gillman, M.; Leo, M.; Perrin, N.; McEvoy, C.T.; Eckhardt, C.L.; Smith, K.S.; Stevens, C.L. Efficacy of a group-based dietary intervention for limiting gestational weight gain among obese women: A randomized trial. Obesity 2014, 22, 1989–1996. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Huang, Y.; Liu, X.; Luo, B.; Yang, Y.; Liao, S. The effect of a personalized intervention on weight gain and physical activity among pregnant women in China. Int. J. Gynaecol. Obstet. 2015, 129, 138–141. [Google Scholar] [CrossRef] [PubMed]
- Luoto, R.; Kinnunen, T.; Aittasalo, M.; Ojala, K.; Mansikkamaki, K.; Toropainen, E.; Kolu, P.; Vasankari, T. Prevention of gestational diabetes: Design of a cluster-randomized controlled trial and one-year follow-up. BMC Pregnancy Childbirth 2010, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Bruno, R.; Petrella, E.; Bertarini, V.; Pedrielli, G.; Neri, I.; Facchinetti, F. Adherence to a lifestyle programme in overweight/obese pregnant women and effect on gestational diabetes mellitus: A randomized controlled trial. Matern. Child Nutr. 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Herring Cruice, J.; Bennett, G.; Rose, M.; Davey, A.; Foster, G. Preventing excessive gestational weight gain among African American women: A randomized clinical trial. Obesity 2016, 24, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Willcox, J.; Wilkinson, S.; Lappas, M.; Ball, K.; Crawford, D.; McCarthy, E.; Fjeldsoe, B.; Whittaker, R.; Maddison, R.; Campbell, K. A mobile health intervention promoting healthy gestational weight gain for women entering pregnancy at a high body mass index: The txt4two pilot randomised controlled trial. BJOG 2017, 124, 1718–1728. [Google Scholar] [CrossRef] [PubMed]
- Olsen, C.; Groth, S.; Graham, M.; Reschke, J.; Strawderman, M.; Fernandez, D. The effectiveness of an online intervention in preventing excessive gestational weight gain: The e-moms roc randomized controlled trial. BMC Pregnancy Childbirth 2018, 18, 148. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.; Lanningham-Foster, L.; Welch, A.; Campbell, C. Web-Based Behavioral Intervention Increases Maternal Exercise but Does Not Prevent Excessive Gestational Weight Gain in Previously Sedentary Women. J. Phys. Act. Health 2016, 13, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Jackson, R.; Stotland, N.; Caughey, A.; Gerbert, B. Improving diet and exercise in pregnancy with Video Doctor counseling: A randomized trial. Patient Educ. Couns. 2011, 83, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Daley, A.; Jolly, K.; Jebb, S.; Lewis, A.; Clifford, S.; Roalfe, A.; Kenyon, S.; Aveyard, P. Feasibility and acceptability of regular weighing, setting weight gain limits and providing feedback by community midwives to prevent excess weight gain during pregnancy: Randomised controlled trial and qualitative study. BMC Obes. 2015, 2, 35. [Google Scholar] [CrossRef] [PubMed]
- Jeffries, K.; Shub, A.; Walker, S.; Hiscock, R.; Permezel, M. Reducing excessive weight gain in pregnancy: A randomised controlled trial. Med. J. Aust. 2009, 191, 429–433. [Google Scholar] [PubMed]
- Quinlivan, J.; Lam, L.; Fisher, J. A randomised trial of a four-step multidisciplinary approach to the antenatal care of obese pregnant women. Aust. N. Z. J. Obstet. Gynaecol. 2011, 51, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, A.; Di Benedetto, A.; Petrella, E.; Pintaudi, B.; Corrado, F.; D’Anna, R.; Neri, I.; Facchinetti, F. Myo-inositol may prevent gestational diabetes onset in overweight women: A randomized, controlled trial. J. Matern. Fetal Neonatal Med. 2016, 29, 3234–3237. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Perdigon, J.; Hopkins, E.; Marcalle, M.; Brooten, D.; Youngblut, J.; Lizardo, M. Weight gain in high-risk pregnant women: Comparison by primary diagnosis and type of care. Clin. Excell. Nurse Pract. 2005, 9, 195–201. [Google Scholar] [PubMed]
- Bechtel-Blackwell, D. Computer-assisted self-interview and nutrition education in pregnant teens. Clin. Nurs. Res. 2002, 11, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.; Sutton, A.; Ioannidis, J.; Terrin, N.; Jones, D.; Lau, J.; Carpenter, J.; Rucker, G.; Harbord, R.M.; Schmid, C.H.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011, 343, 4002. [Google Scholar] [CrossRef] [PubMed]
- Willimas, R.; Wood, L.; Collins, C.; Callister, R. Effectiveness of weight loss interventions—Is there a difference between men and women: A systematic review. Obes. Rev. 2015, 16, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Knight, M.; Foster, C. Diet and exercise in pregnancy. BMJ 2017, 358. [Google Scholar] [CrossRef] [PubMed]
- Di Cesare, M.; Bentham, J.; Stevens, G.; Zhou, B.; Danaei, G.; Lu, Y.; Bixby, H.; Cowan, M.J.; Riley, L.M.; Hajifathalian, K.; et al. Trends in adult body-mass-index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 2016, 387, 1377–1396. [Google Scholar] [CrossRef]
- Heslehurst, N.; Rankin, J.; Wilkinson, J.; Summerbell, C. A nationally representative study of maternal obesity in England, UK: Trends in incidence and demographic inequalities in 619,323 births, 1989–2007. Int. J. Obes. 2010, 34, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Hebden, L.; Chey, T.; Allman-Farinelli, M. Lifestyle intervention for preventing weight gain in young adults: A systematic review and meta-analysis of RCTs. Obes. Rev. 2012, 13, 692–710. [Google Scholar] [CrossRef] [PubMed]
- Chi, Y.; Sha, F.; Yip, P.; Chen, J.; Chen, Y. Randomized comparison of group versus individual educational interventions for pregnant women to reduce their secondhand smoke exposure. Medicine 2016, 95, 5072. [Google Scholar] [CrossRef] [PubMed]
- Lombard, C.; Deeks, A.; Jolley, D.; Ball, K.; Teede, H. A low intensity, community based lifestyle programme to prevent weight gain in women with young children: Cluster randomised controlled trial. BMJ 2010, 341, 3215. [Google Scholar] [CrossRef] [PubMed]
- Lombard, C.; Harrison, C.; Kozica, S.; Zoungas, S.; Keating, C.; Teede, H. Effectiveness and implementation of an obesity prevention intervention: The HeLP-her Rural cluster randomised controlled trial. BMC Public Health 2014, 14, 608. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, L.; Klempel-Donchenko, M.; Redman, L. Pregnancy as a window to future health: Excessive gestational weight gain and obesity. Semin. Perinatol. 2015, 39, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Tomedi, L.; Simhan, H.; Chang, C.; McTigue, K.; Bodnar, L. Gestational weight gain, early pregnancy maternal adiposity distribution, and maternal hyperglycemia. Matern. Child Health J. 2014, 18, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.; Kumar, A.; Blumfield, M.; Truby, H. Maternal nutrition and weight management in pregnancy: A nudge in the right direction. Nutr. Bull. 2018, 43, 69–78. [Google Scholar] [CrossRef]
- Stephenson, J.; Heslehurst, N.; Hall, J.; Schoenaker, D.; Hutchison, J.; Cade, J.; Poston, L.; Barrett, G.; Crozier, S.R.; Barker, M.; et al. Before the beginning: Nutrition and lifestyle in the preconception period and its importance for future health. Lancet 2018, 391, 1831–1834. [Google Scholar] [CrossRef]
- Centers of Disease Control and Prevention (CDC). Antropometric Reference Data for Children and Adults: United States, 2007–2010. CDC. Available online: https://www.cdc.gov/nchs/data/series/sr_11/sr11_252.pdf (accessed on 6 June 2018).
- Steer, P. Routine weighing of women during pregnancy is of limited value and should be abandoned: FOR: Routine weighing does not solve the problem of obesity in pregnancy. BJOG 2015, 122, 1101. [Google Scholar] [CrossRef] [PubMed]
- Dawes, M.; Grudzinskas, J. Repeated measurement of maternal weight during pregnancy. Is this a useful practice? BJOG 1991, 98, 189–194. [Google Scholar] [CrossRef]
- Allen-Walker, V.; Woodside, J.; Holmes, V.; Young, I.; Cupples, M.E.; Hunter, A.; McKinley, M.C. Routine weighing of women during pregnancy-is it time to change current practice? BJOG 2016, 123, 871–874. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Avery, A. Healthy weight management during pregnancy: What advice and information is being provided. J. Hum. Nutr. Dietetics 2012, 25, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Schmied, V.; Duff, M.; Dahlen, H.; Mills, A.; Kolt, G. ‘Not waving but drowning’: A study of the experiences and concerns of midwives and other health professionals caring for obese childbearing women. Midwifery 2011, 27, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Stotland, N.E.; Gilbert, P.; Bogetz, A.; Harper, C.C.; Abrams, B.; Gerbert, B. Preventing excessive weight gain in pregnancy: How do prenatal care providers approach counseling? J. Womens Health 2010, 19, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.; Mazza, D.; Blumfield, M.; Bennett, C.; Truby, H. Maternal gestational weight gain during pregnancy: Prioritising the conversation. Aust. J. Prim. Health 2017, 24, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Herring, S. Do mHealth interventions prevent excessive gestational weight gain? BJOG 2017, 124, 1728. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). Dietary Interventions and Advice for Adults. NICE. Available online: https://pathways.nice.org.uk/pathways/diet/dietary-interventions-and-advice-for-adults#coentent=view-node%3Anodes-advice-for-all-women (accessed on 16 July 2018).
- National Health and Medical Research Council (NHMRC). Clinical Practice Guidelines for the Management of Overweight and Obesity in Adults, Adolescents and Children in Australia. NHMRC. Available online: https://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/n57_obesity_guidelines_140630.pdf (accessed on 16 July 2018).
- Higgins, J.; Thompson, G.; Deeks, J.; Altman, D. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557. [Google Scholar] [CrossRef] [PubMed]
- Krukowski, R.; West, D.; Dicarlo, M.; Shankar, K.; Cleves, M.; Saylors, M.; Andrea, A. Are early first trimester weights valid proxies for preconception weight? BMC Pregnancy Childbirth 2016, 16, 557. [Google Scholar] [CrossRef] [PubMed]
- Jaacks, L.; Kavle, J.; Perry, A.; Myaku, A. Programming maternal and child overweight and obesity in the context of undernutrition: Current evidence and key considerations for low and middle-income countries. Public Health Nutr. 2017, 20, 1286–1296. [Google Scholar] [CrossRef] [PubMed]
| Sub-Group and Overall | Studies a | Participants | Overall Effect Size WMD (95% CI) | p Value | I2 | |
|---|---|---|---|---|---|---|
| Diet | Overall: Measurement GWG > 24 weeks | 9 | 2049 | −3.27 (−4.96, −1.58) | <0.00 | 92.8% |
| Physical activity | Measurement GWG 12–24 weeks | 3 | 250 | −0.83 (−3.55, 1.90) | 0.55 | 83.3% |
| Measurement GWG > 24 weeks | 21 | 4651 | −1.02 (−1.56, −0.47) | <0.00 | 81.8% | |
| Overall | 24 | 4901 | −1.02 (−1.56, −0.49) | <0.00 | 81.9% | |
| Lifestyle | Measurement GWG 12–24 weeks | 5 | 2908 | −0.67 (−1.58, 0.23) | 0.14 | 80.1% |
| Measurement GWG > 24 weeks | 19 | 4471 | −0.92 (−1.48, −0.36) | <0.00 | 69.8% | |
| Overall | 24 | 7379 | −0.84 (−1.29, −0.39) | <0.00 | 71.0% | |
| eHealth | Overall: Measurement GWG > 24 weeks | 3 | 192 | −1.06 (−4.13, 2.00) | 0.50 | 73.6% |
| Diet (10 Observations) | PA (25 Observations) I2 = 82.0% | Lifestyle (27 Observations) I2 = 71.0% | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Characteristic | n= | ß | 95% CI | p | n= | ß | 95% CI | p | n= | ß | 95% CI | p |
| Trimester | 9 | 25 | 27 | |||||||||
| 1 | 3 | Ref | 12 | Ref | 6 | Ref | ||||||
| 2 | 6 | 0.35 | −4.61, 5.32 | 0.87 | 13 | 0.81 | −0.13, 1.75 | 0.09 | 21 | −0.16 | −1.50, 1.17 | 0.81 |
| 3 | 0 | NA | NA | NA | 0 | NA | NA | NA | 0 | NA | NA | NA |
| Intensity | 10 | 25 | 23 | |||||||||
| 1–4 h | 5 | Ref | 5 | Ref | 8 | Ref | ||||||
| >4–8 h | 2 | −1.08 | −7.43, 5.28 | 0.70 | 1 | 1.50 | −2.77, 5.77 | 0.47 | 7 | 0.00 | −1.46, 1.46 | 1.00 |
| ≥8 times | 3 | 0.71 | −5.04, 6.46 | 0.78 | 19 | 0.05 | −1.37, 1.48 | 0.94 | 8 | −1.32 | −2.71, 0.08 | 0.06 |
| Frequency | 10 | 25 | 27 | |||||||||
| 1–3 times | 5 | Ref | 3 | Ref | 3 | Ref | ||||||
| 4–7 times | 1 | −1.92 | −10.18, 6.33 | 0.60 | 2 | 0.55 | −2.71, 3.81 | 0.73 | 10 | 0.18 | −1.79, 2.14 | 0.86 |
| ≥8 times | 4 | 0.46 | −4.73, 5.65 | 0.84 | 20 | 0.02 | −1.94, 1.98 | 0.99 | 17 | −0.04 | −1.95, 1.88 | 0.97 |
| Setting | 10 | 20 | 27 | |||||||||
| Individual | 9 | Ref | 7 | Ref | 19 | Ref | ||||||
| Group | 0 | NA | NA | NA | 11 | −0.62 | −1.56, 0.31 | 0.18 | 8 | −1.09 | −2.28, 0.09 | 0.07 |
| Mixed | 1 | 2.01 | −5.17, 9.20 | 0.54 | 2 | −1.99 | −3.45, −0.52 | 0.01 | 0 | NA | NA | NA |
| Duration | 9 | 25 | 27 | |||||||||
| Reference | Ref | Ref | Ref | |||||||||
| −0.65 | −1.81, 0.52 | 0.23 | 0.01 | −0.09, 0.10 | 0.90 | −0.04 | −0.12, 0.04 | 0.34 | ||||
| Type | 10 | |||||||||||
| General | 3 | Ref | ||||||||||
| Low-GI | 1 | −0.06 | −8.59, 8.48 | 0.99 | ||||||||
| kJ restriction | 5 | −3.20 | −8.81, 2.42 | 0.21 | ||||||||
| Probiotic | 1 | −2.56 | −11.32, 6.21 | 0.50 | ||||||||
| Effective, p < 0.05 (n = 32) | Ineffective, p > 0.05 (n = 41) | Total a (n = 73) | |||
|---|---|---|---|---|---|
| n (%) | n (%) | n (%) | p= | ||
| Commenced | |||||
| (Trimester) | |||||
| 1 | 12 (41.4) | 17 (58.6) | 29 | 0.73 | |
| 2 | 18 (45.0) | 22 (55.0) | 40 | 0.83 | |
| 3 | 0 (0.0) | 0 (0.0) | 0 | - | |
| Unsure | 2 (50.0) | 2 (50.0) | 4 | 0.80 | |
| Duration | |||||
| (Weeks) | |||||
| <12 | 2 (66.7) | 1 (33.3) | 3 | 0.42 | |
| 12–29 | 21 (40.4) | 31 (59.6) | 52 | 0.35 | |
| ≥30 | 7 (50.0) | 7 (50.0) | 14 | 0.61 | |
| Unsure | 2 (50.0) | 2 (50.0) | 4 | 0.80 | |
| Intensity | |||||
| (Hours) | |||||
| <4 | 6 (33.3) | 12 (66.7) | 18 | 0.30 | |
| 4–8 | 3 (37.5) | 5 (62.5) | 8 | 0.70 | |
| >8 | 14 (43.8) | 18 (56.3) | 32 | 0.99 | |
| Unsure | 9 (60.0) | 6 (40.0) | 15 | 0.16 | |
| Frequency | |||||
| (Contact) | |||||
| ≤3 | 4 (33.3) | 8 (66.7) | 12 | 0.42 | |
| 4–7 | 4 (30.8) | 9 (69.2) | 13 | 0.30 | |
| ≥8 | 19 (47.5) | 21 (52.5) | 40 | 0.49 | |
| Unsure | 5 (62.5) | 3 (37.5) | 8 | 0.26 | |
| Delivery method | |||||
| Individual | 14 (33.3) | 28 (66.7) | 42 | 0.04 | |
| Group b | 15 (62.5) | 9 (37.5) | 24 | 0.02 | |
| Unsure | 3 (42.9) | 4 (51.7) | 7 | 0.96 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walker, R.; Bennett, C.; Blumfield, M.; Gwini, S.; Ma, J.; Wang, F.; Wan, Y.; Truby, H. Attenuating Pregnancy Weight Gain—What Works and Why: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 944. https://doi.org/10.3390/nu10070944
Walker R, Bennett C, Blumfield M, Gwini S, Ma J, Wang F, Wan Y, Truby H. Attenuating Pregnancy Weight Gain—What Works and Why: A Systematic Review and Meta-Analysis. Nutrients. 2018; 10(7):944. https://doi.org/10.3390/nu10070944
Chicago/Turabian StyleWalker, Ruth, Christie Bennett, Michelle Blumfield, Stella Gwini, Jianhua Ma, Fenglei Wang, Yi Wan, and Helen Truby. 2018. "Attenuating Pregnancy Weight Gain—What Works and Why: A Systematic Review and Meta-Analysis" Nutrients 10, no. 7: 944. https://doi.org/10.3390/nu10070944
APA StyleWalker, R., Bennett, C., Blumfield, M., Gwini, S., Ma, J., Wang, F., Wan, Y., & Truby, H. (2018). Attenuating Pregnancy Weight Gain—What Works and Why: A Systematic Review and Meta-Analysis. Nutrients, 10(7), 944. https://doi.org/10.3390/nu10070944





