Enhanced Omega-3 Polyunsaturated Fatty Acid Contents in Muscle and Edible Organs of Australian Prime Lambs Grazing Lucerne and Cocksfoot Pastures
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals, Diets, Experimental Design, and Feed Sample Collection
2.2. Slaughter Protocol and Fatty Acid Analysis
| (a) | FA% = (individual fatty acid area) × (100)/(sum total area of fatty acids) |
| (b) | FA mg/100 g = (Total lipid) × (LCF (0.916)) × ((%FA)/100) × 1000 |
2.3. Statistical Analysis
3. Results
3.1. FA Composition of Pastures and Supplementary Feeds
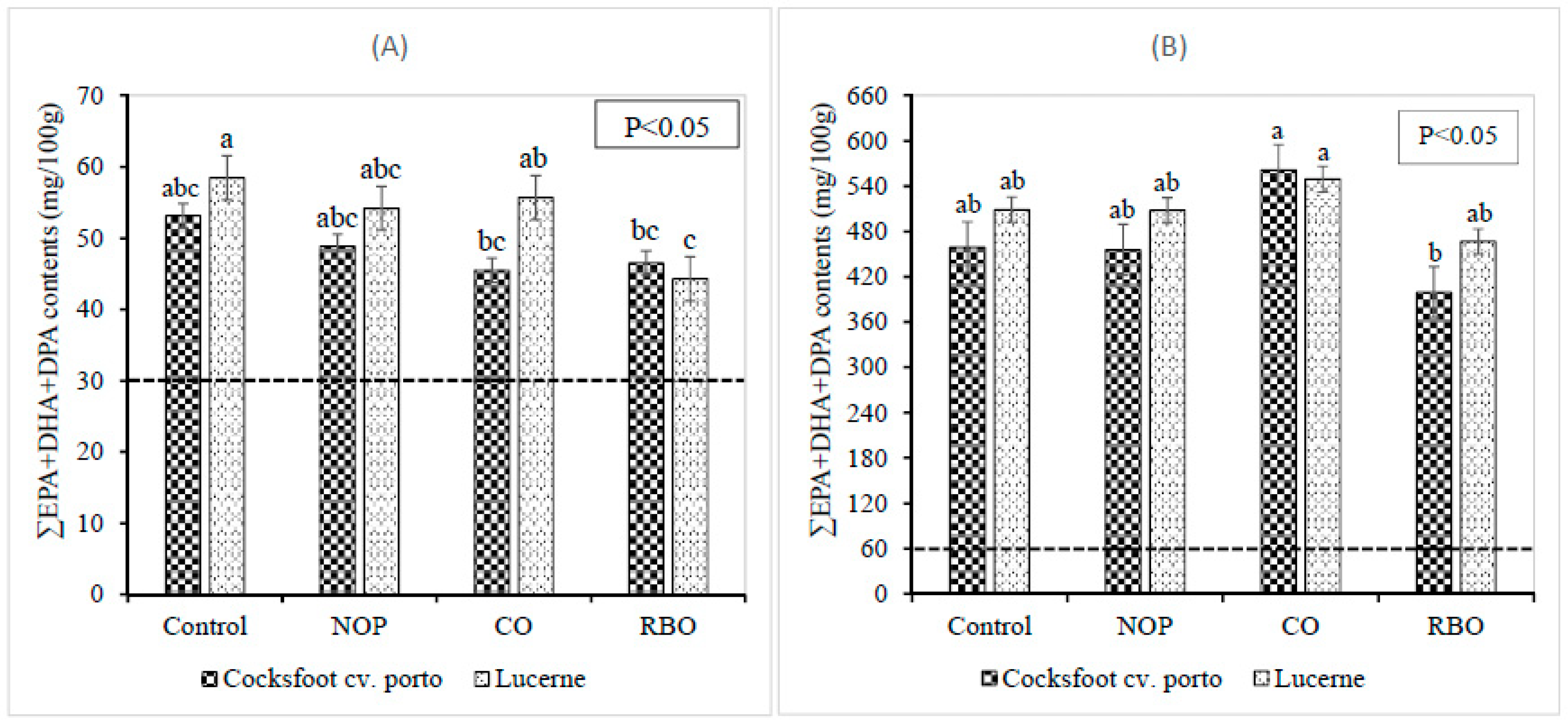
3.2. Effect of Pellet Supplements on the Fatty Acid Contents in Longissimus dorsi Muscle, Liver, Heart, and Kidney
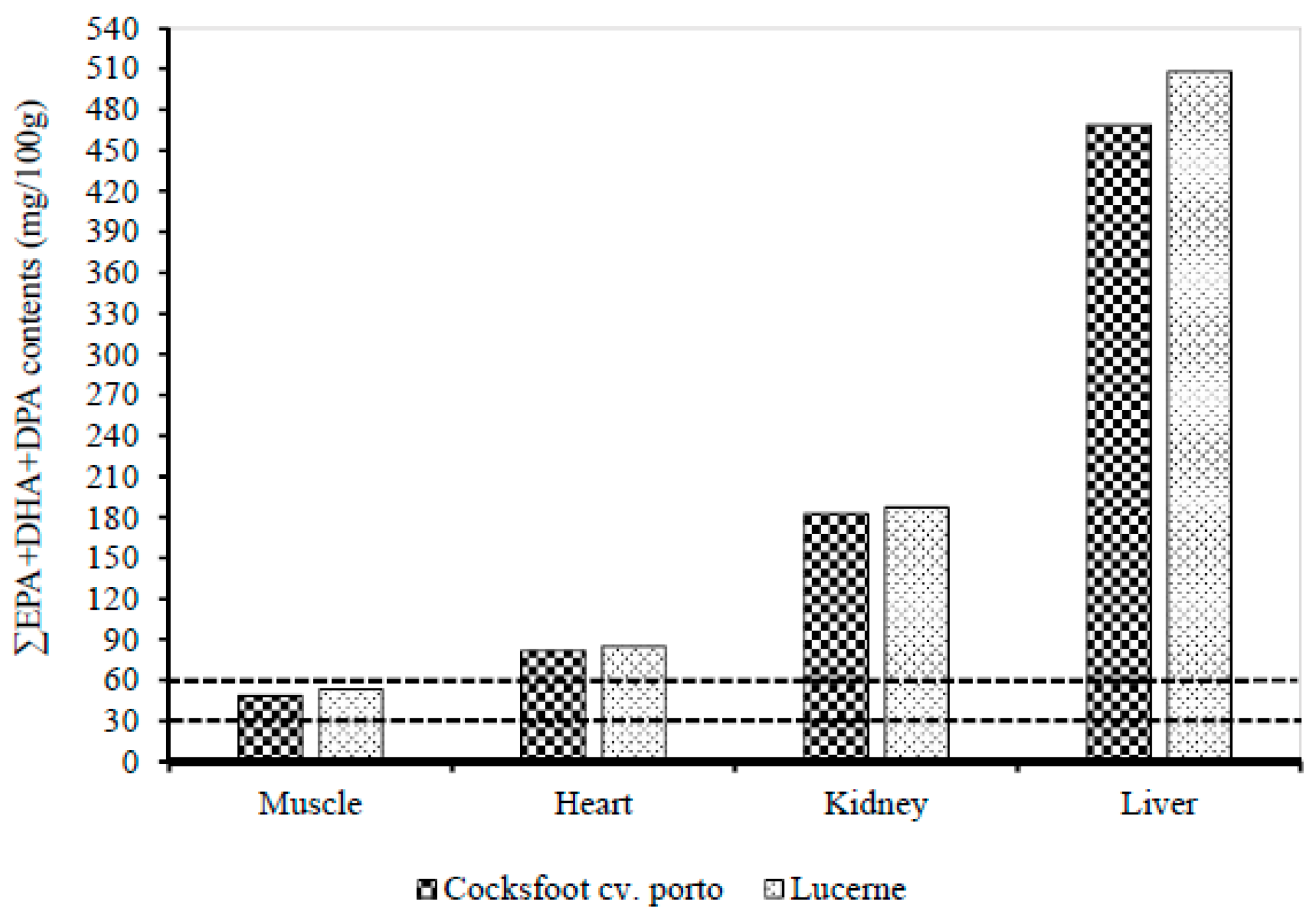
3.3. Effect of Pasture Types on the Fatty Acid Contents in Muscle, Liver, Heart, and Kidney
4. Discussion
4.1. FA of Pastures and Supplementary Feeds
4.2. Effect of Supplements on the Fatty Acid Contents in Longissimus dorsi Muscle, Liver, Heart and Kidney
4.3. Effect of Pasture Types on the Fatty Acid Contents in Muscle, Liver, Heart, and Kidney of Lambs
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Calder, P.C. Polyunsaturated fatty acids and inflammatory processes: New twists in an old tale. Biochimie 2009, 91, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Lu, L.; Liang, J.; Liu, M.; Li, X.C.; Sun, R.R.; Zheng, Y.; Zhang, P.Y. Omega-3 fatty acids and primary and secondary prevention of cardiovascular disease. Cell Biochem. Biophys. 2015, 72, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Leslie, M.A.; Cohen, D.J.A.; Liddle, D.M.; Robinson, L.E.; Ma, D.W.L. A review of the effect of omega-3 polyunsaturated fatty acids on blood triacylglycerol levels in normolipidemic and borderline hyperlipidemic individuals. Lipids Health Dis. 2015, 14, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Cabo-Garcia, L.; Achon-Tunon, M.; Gonzalez-Gonzalez, M.P. The influence of polyunsaturated fatty acids in the prevention and promotion of cancer. Nutr. Hosp. 2015, 32, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.Q.; Zheng, J.S.; Yang, B.; Li, D. Effect of individual omega-3 fatty acids on the risk of prostate cancer: A systematic review and dose-response meta-analysis of prospective cohort studies. J. Epidemiol. 2015, 25, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Manzi, L.; Costantini, L.; Molinari, R.; Merendino, N. Effect of dietary omega-3 polyunsaturated fatty acid DHA on glycolytic enzymes and Warburg phenotypes in cancer. Biomed. Res. Int. 2015, 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.F.; Bilotto, S.; Russo, G.L.; Orhan, I.E.; Habtemariam, S.; Daglia, M.; Devi, K.P.; Loizzo, M.R.; Tundis, R.; Nabavi, S.M. Omega-3 polyunsaturated fatty acids and cancer: Lessons learned from clinical trials. Cancer Metast. Rev. 2015, 34, 359–380. [Google Scholar] [CrossRef]
- Sheppard, K.W.; Cheatham, C.L. Omega-6/omega-3 fatty acid intake of children and older adults in the U.S.: Dietary intake in comparison to current dietary recommendations and the Healthy Eating Index. Lipids Health Dis. 2018, 17, 43. [Google Scholar] [CrossRef]
- Pittaway, J.; Chuang, L.; Ahuja, K.; Beckett, J.; Glew, R.; Ball, M. Omega-3 dietary fatty acid status of healthy older adults in Tasmania, Australia: An observational study. J. Nutr. Health Aging 2015, 19, 505–510. [Google Scholar] [CrossRef]
- Nichols, P.D.; Petrie, J.; Singh, S. Long-chain omega-3 oils–an update on sustainable sources. Nutrients 2010, 2, 572–585. [Google Scholar] [CrossRef]
- Howe, P.; Buckley, J.; Meyer, B. Long-chain omega-3 fatty acids in red meat. Nutr. Diet. 2007, 64, S135–S139. [Google Scholar] [CrossRef]
- ABARES Agricultural Commodities and Trade Data. Annual Commodity Statistics. Available online: http://www.agriculture.gov.au/abares/research-topics/agricultural-commodities/agricultural-commodities-trade-data#2017 (accessed on 11 September 2018).
- Wong, L.; Selvanathan, E.A.; Selvanathan, S. Modelling the meat consumption patterns in Australia. Econ. Model. 2015, 49, 1–10. [Google Scholar] [CrossRef]
- De Brito, G.F.; Ponnampalam, E.N.; Hopkins, D.L. The effect of extensive feeding systems on growth rate, carcass traits, and meat quality of finishing lambs. Compr. Rev. Food Sci. F. 2017, 16, 23–38. [Google Scholar] [CrossRef]
- Lolicato, S.; Rumball, W. Past and present improvement of cocksfoot (Dactylis glomerata L.) in Australia and New Zealand. N. Z. J. Agric. Res. 1994, 37, 379–390. [Google Scholar] [CrossRef]
- Clark, S.G.; Nie, Z.N.; Culvenor, R.A.; Harris, C.A.; Hayes, R.C.; Li, G.D.; Norton, M.R.; Partington, D.L. Field evaluation of cocksfoot, tall fescue and phalaris for dry marginal environments of south-eastern australia. 1. Establishment and herbage production. J. Agron. Crop Sci. 2016, 202, 96–114. [Google Scholar] [CrossRef]
- Clapham, W.M.; Foster, J.G.; Neel, J.P.; Fedders, J.M. Fatty acid composition of traditional and novel forages. J. Agric. Food Chem. 2005, 53, 10068–10073. [Google Scholar] [CrossRef] [PubMed]
- Casey, N.H.; van Niekerk, W.A.; Spreeth, E.B. Fatty acid composition of subcutaneous fat of sheep grazed on eight different pastures. Meat Sci. 1988, 23, 55–63. [Google Scholar] [CrossRef]
- Chikwanha, O.C.; Vahmani, P.; Muchenje, V.; Dugan, M.E.R.; Mapiye, C. Nutritional enhancement of sheep meat fatty acid profile for human health and wellbeing. Food Res. Int. 2018, 104, 25–38. [Google Scholar] [CrossRef]
- Ponnampalam, E.N.; Butler, K.L.; Jacob, R.H.; Pethick, D.W.; Ball, A.J.; Hocking Edwards, J.E.; Geesink, G.; Hopkins, D.L. Health beneficial long chain omega-3 fatty acid levels in Australian lamb managed under extensive finishing systems. Meat Sci. 2014, 96, 1104–1110. [Google Scholar] [CrossRef]
- Humphries, A.W. Future applications of lucerne for efficient livestock production in southern Australia. Crop Pasture Sci. 2012, 63, 909–917. [Google Scholar] [CrossRef]
- Ponnampalam, E.N.; Linden, N.P.; Mitchell, M.L.; Hopkins, D.L.; Jacobs, J.L. Production systems to deliver premium grade lambs to the growing international and Australian markets. Small Ruminant Res. 2017, 157, 32–39. [Google Scholar] [CrossRef]
- Fraser, M.D.; Speijers, M.H.M.; Theobald, V.J.; Fychan, R.; Jones, R. Production performance and meat quality of grazing lambs finished on red clover, lucerne or perennial ryegrass swards. Grass Forage Sci. 2004, 59, 345–356. [Google Scholar] [CrossRef]
- Boughalmi, A.; Araba, A. Effect of feeding management from grass to concentrate feed on growth, carcass characteristics, meat quality and fatty acid profile of Timahdite lamb breed. Small Ruminant Res. 2016, 144, 158–163. [Google Scholar] [CrossRef]
- Turner, K.E.; Belesky, D.P.; Cassida, K.A.; Zerby, H.N. Carcass merit and meat quality in Suffolk lambs, Katahdin lambs, and meat-goat kids finished on a grass–legume pasture with and without supplementation. Meat Sci. 2014, 98, 211–219. [Google Scholar] [CrossRef]
- Nguyen, D.V.; Flakemore, A.R.; Otto, J.R.; Ives, S.W.; Smith, R.W.; Nichols, P.D.; Malau-Aduli, A.E.O. Nutritional value and sensory characteristics of meat eating quality of Australian prime lambs supplemented with pelleted canola and flaxseed oils: Fatty acid profiles of muscle and adipose tissues. Int. Med. Rev. 2017, 3, 1–21. [Google Scholar]
- Goffman, F.D.; Pinson, S.; Bergman, C. Genetic diversity for lipid content and fatty acid profile in rice bran. J. Am. Oil Chem. Soc. 2003, 80, 485–490. [Google Scholar] [CrossRef]
- Lunsin, R.; Wanapat, M.; Yuangklang, C.; Rowlinson, P. Effect of rice bran oil supplementation on rumen fermentation, milk yield and milk composition in lactating dairy cows. Livest. Sci. 2012, 145, 167–173. [Google Scholar] [CrossRef]
- Bhatt, R.S.; Sahoo, A.; Karim, S.A.; Agrawal, A.R. Effects of calcium soap of rice bran oil fatty acids supplementation alone and with DL-α-tocopherol acetate in lamb diets on performance, digestibility, ruminal parameters and meat quality. J. Anim. Physiol. Anim. Nutr. 2016, 100, 578–589. [Google Scholar] [CrossRef]
- Umaraw, P.; Pathak, V.; Rajkumar, V.; Verma, A.K.; Singh, V.P.; Verma, A.K. Assessment of fatty acid and mineral profile of Barbari kid in longissimus lumborum muscle and edible byproducts. Small Ruminant Res. 2015, 132, 147–152. [Google Scholar] [CrossRef]
- Bester, M.; Schönfeldt, H.C.; Pretorius, B.; Hall, N. The nutrient content of selected South African lamb and mutton organ meats (offal). Food Chem. 2018, 238, 3–8. [Google Scholar] [CrossRef]
- Nguyen, D.V.; Le, V.H.; Nguyen, Q.V.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Omega–3 long-chain fatty acids in the heart, kidney, liver and plasma metabolite profiles of australian prime lambs supplemented with pelleted canola and flaxseed oils. Nutrients 2017, 9, 893. [Google Scholar] [CrossRef] [PubMed]
- Malau-Aduli, A.E.O.; Holman, B.W.B.; Kashani, A.; Nichols, P.D. Sire breed and sex effects on the fatty acid composition and content of heart, kidney, liver, adipose and muscle tissues of purebred and first-cross prime lambs. Anim. Prod. Sci. 2016, 56, 2122–2132. [Google Scholar] [CrossRef]
- Amaral, D.S.; Silva, F.A.P.; Bezerra, T.K.A.; Arcanjo, N.M.O.; Guerra, I.C.D.; Dalmás, P.S.; Madruga, M.S. Effect of storage time and packaging on the quality of lamb pâté prepared with ‘variety meat’. Food Packaging Shelf 2015, 3, 39–46. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Miller, M.R.; Nichols, P.D.; Barnes, J.; Davies, N.W.; Peacock, E.J.; Carter, C.G. Regiospecificity profiles of storage and membrane lipids from the gill and muscle tissue of Atlantic salmon (Salmo salar L.) grown at elevated temperature. Lipids 2006, 41, 865–876. [Google Scholar] [CrossRef] [PubMed]
- Flakemore, A.R.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Omega-3 fatty acids, nutrient retention values, and sensory meat eating quality in cooked and raw Australian lamb. Meat Sci. 2017, 123, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Clayton, E.H. Graham Centre Monograph No. 4: Long-Chain Omega-3 Polyunsaturated Fatty Acids in Ruminant Nutrition: Benefits to Animals and Humans; Nugent, T., Nicholls, C., Eds.; NSW Department of Primary Industries: Wagga Wagga, NSW, Australia, 2014.
- SAS. Statistical Analysis System. SAS/STAT User’s Guide: Statistics; Version 9.2; SAS Inc.: Cary, NC, USA, 2009.
- Meľuchová, B.; Blaško, J.; Kubinec, R.; Górová, R.; Dubravská, J.; Margetín, M.; Soják, L. Seasonal variations in fatty acid composition of pasture forage plants and CLA content in ewe milk fat. Small Ruminant Res. 2008, 78, 56–65. [Google Scholar] [CrossRef]
- Garcia, P.T.; Pordomingo, A.; Perez, C.D.; Rios, M.D.; Sancho, A.M.; Volpi Lagreca, G.; Casal, J.J. Influence of cultivar and cutting date on the fatty acid composition of forage crops for grazing beef production in Argentina. Grass Forage Sci. 2016, 71, 235–244. [Google Scholar] [CrossRef]
- Wiking, L.; Theil, P.K.; Nielsen, J.H.; Sorensen, M.T. Effect of grazing fresh legumes or feeding silage on fatty acids and enzymes involved in the synthesis of milk fat in dairy cows. J. Dairy Res. 2010, 77, 337–342. [Google Scholar] [CrossRef]
- Glasser, F.; Doreau, M.; Maxin, G.; Baumont, R. Fat and fatty acid content and composition of forages: A meta-analysis. Anim. Feed Sci. Tech. 2013, 185, 19–34. [Google Scholar] [CrossRef]
- Nguyen, D.V.; Malau-Aduli, B.S.; Cavalieri, J.; Nichols, P.D.; Malau-Aduli, A.E.O. Supplementation with plant-derived oils rich in omega-3 polyunsaturated fatty acids for lamb production. Veterinary Anim. Sci. 2018, 6, 29–40. [Google Scholar] [CrossRef]
- Ponnampalam, E.N.; Burnett, V.F.; Norng, S.; Warner, R.D.; Jacobs, J.L. Vitamin E and fatty acid content of lamb meat from perennial pasture or annual pasture systems with supplements. Anim. Prod. Sci. 2012, 52, 255–262. [Google Scholar] [CrossRef]
- Fruet, A.P.B.; Trombetta, F.; Stefanello, F.S.; Speroni, C.S.; Donadel, J.Z.; De Souza, A.N.M.; Rosado Júnior, A.; Tonetto, C.J.; Wagner, R.; De Mello, A.; et al. Effects of feeding legume-grass pasture and different concentrate levels on fatty acid profile, volatile compounds, and off-flavor of the M. longissimus thoracis. Meat Sci. 2018, 140, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Raes, K.; De Smet, S.; Demeyer, D. Effect of dietary fatty acids on incorporation of long chain polyunsaturated fatty acids and conjugated linoleic acid in lamb, beef and pork meat: A review. Anim. Feed Sci. Tech. 2004, 113, 199–221. [Google Scholar] [CrossRef]
- Ponnampalam, E.N.; Butler, K.L.; Pearce, K.M.; Mortimer, S.I.; Pethick, D.W.; Ball, A.J.; Hopkins, D.L. Sources of variation of health claimable long chain omega-3 fatty Australian lamb slaughtered at similar weights. Meat Sci. 2014, 96, 1095–1103. [Google Scholar] [CrossRef] [PubMed]
- Kashani, A.; Holman, B.W.B.; Nichols, P.D.; Malau-Aduli, A.E.O. Effect of level of Spirulina supplementation on the fatty acid compositions of adipose, muscle, heart, kidney and liver tissues in Australian dual-purpose lambs. Ann. Anim. Sci. 2015, 15, 945–960. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Nutrition Information User Guide to Standard 1.2.8—Nutrition Information Requirements Part B—Nutrition Claims. Available online: http://www.foodstandards.gov.au/code/userguide/Documents/Userguide_Nutrition%20Claims_PartB_March12.pdf (accessed on 1 November 2017).
- Johansen, M.; Lund, P.; Weisbjerg, M.R. Feed intake and milk production in dairy cows fed different grass and legume species: A meta-analysis. Animal 2017, 12, 66–75. [Google Scholar] [CrossRef]
- Kitessa, S.; Liu, S.; Briegel, J.; Pethick, D.; Gardner, G.; Ferguson, M.; Allingham, P.; Nattrass, G.; McDonagh, M.; Ponnampalam, E.; et al. Effects of intensive or pasture finishing in spring and linseed supplementation in autumn on the omega-3 content of lamb meat and its carcass distribution. Anim. Prod. Sci. 2010, 50, 130–137. [Google Scholar] [CrossRef]
- Bessa, R.J.B.; Alves, S.P.; Santos-Silva, J. Constraints and potentials for the nutritional modulation of the fatty acid composition of ruminant meat. Eur. J. Lipid Sci. Tech. 2015, 117, 1325–1344. [Google Scholar] [CrossRef]
| Chemical Composition (% DM) | Cocksfoot cv. Porto | Lucerne | NOP | CO | RBO |
|---|---|---|---|---|---|
| DM | 20.5 | 20.7 | 89.1 | 91.1 | 90.2 |
| CP | 13.3 | 18.6 | 15.7 | 15.3 | 14.7 |
| ADF | 26.7 | 25.6 | 6.8 | 7.4 | 8.0 |
| NDF | 43.8 | 35.9 | 18.3 | 19.9 | 18.7 |
| EE | 3.0 | 1.8 | 2.1 | 4.6 | 4.9 |
| Ash | 6.4 | 6.8 | 4.0 | 6.5 | 5.0 |
| %TDN | 62.3 | 63.2 | 77.2 | 76.8 | 76.4 |
| DE (Mcal/kg) | 2.7 | 2.8 | 3.4 | 3.4 | 3.4 |
| ME (MJ/kg) | 9.4 | 9.5 | 11.7 | 11.6 | 11.5 |
| % Lipid | Cocksfoot cv. Porto | Lucerne | NOP | CO | RBO |
|---|---|---|---|---|---|
| 14:0 | 0.6 | 1.2 | 0.2 | 0.2 | 0.3 |
| 15:0 | 0.2 | 0.4 | 0.1 | 0.1 | 0.1 |
| 16:1n-9c | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 |
| 16:1n-7c | 0.2 | 0.1 | 0.3 | 0.3 | 0.2 |
| 16:0 | 15.7 | 17.1 | 16.3 | 9.9 | 15.5 |
| 17:0 | 0.5 | 0.5 | 0.1 | 0.1 | 0.1 |
| 18:2n-6 LA | 14.8 | 17.1 | 50.3 | 32.9 | 42.2 |
| 18:3n-3 ALA | 57.6 | 51.9 | 3.5 | 5.7 | 2.7 |
| 18:1n-9c | 1.0 | 0.5 | 24.5 | 44.5 | 32.3 |
| 18:1n-7c | 0.2 | 0.1 | 1.0 | 2.6 | 1.2 |
| 18:1n-7t | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| 18:0 | 0.1 | 2.2 | 0.3 | 0.5 | 0.3 |
| 20:4n-6 ARA | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 |
| 20:5n-3 EPA | 0.0 | 0.0 | 0.1 | 0.1 | 0.1 |
| 20:3n-6 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| 20:4n-3 | 0.1 | 0.1 | 0.2 | 0.1 | 0.1 |
| 20:2n-6 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| 20:0 | 1.6 | 1.2 | 0.5 | 0.6 | 0.6 |
| 22:5n-6 DPA-6 | 0.0 | 0.0 | 0.1 | 0.1 | 0.1 |
| 22:6n-3 DHA | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| 22:5n-3 DPA-3 | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 |
| 22:0 | 1.0 | 1.3 | 0.6 | 0.3 | 0.4 |
| 23:0 | 0.3 | 0.5 | 0.1 | 0.0 | 0.1 |
| 24:0 | 0.9 | 1.4 | 0.3 | 0.2 | 0.3 |
| ∑SFA | 20.9 | 25.8 | 18.5 | 11.8 | 17.6 |
| ∑MUFA | 4.9 | 3.9 | 26.9 | 48.7 | 34.6 |
| ∑PUFA | 73.1 | 69.7 | 54.5 | 39.2 | 45.5 |
| ∑n-3 LC-PUFA | 0.1 | 0.1 | 0.3 | 0.3 | 0.3 |
| ∑n-3 PUFA | 58.0 | 52.2 | 3.8 | 6.0 | 3.0 |
| ∑n-6 PUFA | 15.2 | 17.5 | 50.6 | 33.1 | 42.5 |
| ∑other FA | 1.0 | 0.4 | 0.1 | 0.2 | 2.3 |
| n-6/n-3 | 0.3 | 0.3 | 13.3 | 5.5 | 14.2 |
| Items | Control | NOP | CO | RBO | ||||
|---|---|---|---|---|---|---|---|---|
| CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | |
| IMF percentage | 3.5 ± 0.5 | 3.4 ± 0.5 | 2.5 ± 0.5 | 2.5 ± 0.5 | 3.1 ± 0.5 | 2.7 ± 0.5 | 2.6 ± 0.5 | 3.6 ± 0.5 |
| 14:0 | 68.9 ± 13.9 | 54.5 ± 13.9 | 43.7 ± 13.9 | 65.7 ± 13.9 | 52.5 ± 13.9 | 39.5 ± 13.9 | 30.3 ± 13.9 | 49.6 ± 13.9 |
| 15:0 | 10.1 ± 1.7 a | 7.4 ± 1.7 ab | 5.5 ± 1.7 ab | 8.9 ± 1.7 a | 6.4 ± 1.7 ab | 5.9 ± 1.7 ab | 3.9 ± 1.7 b | 7.9 ± 1.7 ab |
| 16:1n-9c | 9.0 ± 1.3 a | 6.2 ± 1.3 ab | 5.3 ± 1.3 ab | 8.4 ± 1.3 a | 5.6 ± 1.3 ab | 5.6 ± 1.3 ab | 4 ± 1.3 b | 7.3 ± 1.3 ab |
| 16:1n-7c | 37.1 ± 7.7 | 31.8 ± 7.7 | 27.0 ± 7.7 | 41.9 ± 7.7 | 32.1 ± 7.7 | 30.7 ± 7.7 | 22.4 ± 7.7 | 35.0 ± 7.7 |
| 16:0 | 712.9 ± 113.8 ab | 541.3 ± 113.8 ab | 433.5 ± 113.8 ab | 753.2 ± 113.8 a | 545.9 ± 113.8 ab | 592.1 ± 113.8 ab | 416.6 ± 113.8 b | 663.9 ± 113.8 ab |
| 17:0 | 32.6 ± 5.5 ab | 24.9 ± 5.5 ab | 19 ± 5.5 bc | 36.3 ± 5.5 a | 21.2 ± 5.5 ab | 25.7 ± 5.5 ab | 16.6 ± 5.5 c | 32.9 ± 5.5 ab |
| 18:2n-6 LA | 119.3 ± 8.7 bc | 122.8 ± 8.7 bc | 123.4 ± 8.7 bc | 156.8 ± 8.7 a | 110.6 ± 8.7 c | 144.2 ± 8.7 ab | 119.8 ± 8.7 bc | 157.2 ± 8.7 a |
| 18:3n-3 ALA | 67.1 ± 5.5 ab | 68.1 ± 5.5 a | 33.9 ± 5.5 d | 57.3 ± 5.5 ab | 35.8 ± 5.5 cd | 51.6 ± 5.5 bc | 35.5 ± 5.5 cd | 39.5 ± 5.5 cd |
| 18:1n-9c | 1169.0 ± 197.0 ab | 902.9 ± 197.0 ab | 767.5 ± 197.0 b | 1351.9 ± 197.0 a | 925.8 ± 197.0 ab | 1024.5 ± 197.0 ab | 759.6 ± 197.0 b | 1153.8 ± 197.0 ab |
| 18:1n-7c | 41.2 ± 7.5 ab | 33.4 ± 7.5 b | 38.4 ± 7.5 ab | 54.1 ± 7.5 ab | 43.2 ± 7.5 ab | 50.3 ± 7.5 ab | 34.2 ± 7.5 ab | 55.5 ± 7.5 a |
| 18:1n-7t | 83.6 ± 18.3 ab | 57 ± 18.3 b | 56 ± 18.3 b | 102.5 ± 18.3 ab | 67.5 ± 18.3 b | 82.9 ± 18.3 ab | 57.2 ± 18.3 b | 122.5 ± 18.3 a |
| 18:0 | 592.4 ± 75.4 a | 364.8 ± 75.4 bc | 328.4 ± 75.4 bc | 533.4 ± 75.4 ab | 362.5 ± 75.4 bc | 402.1 ± 75.4 abc | 307.3 ± 75.4 c | 515.2 ± 75.4 abc |
| 20:4n-6 ARA | 33.7 ± 2.9 | 35.7 ± 2.9 | 38.2 ± 2.9 | 36 ± 2.9 | 34.8 ± 2.9 | 37.2 ± 2.9 | 34.5 ± 2.9 | 36.9 ± 2.9 |
| 20:5n-3 EPA | 24.7 ± 1.9 abc | 26.9 ± 1.9 a | 22.1 ± 1.9 abc | 24.2 ± 1.9 abc | 19.8 ± 1.9bc | 25 ± 1.9 ab | 20.2 ± 1.9 bc | 19.1 ± 1.9 c |
| 20:3n-6 | 6.1 ± 0.5 c | 6.7 ± 0.5 bc | 6.6 ± 0.5 bc | 8.0 ± 0.5 a | 5.8 ± 0.5 c | 7.7 ± 0.5 ab | 6.4 ± 0.5 bc | 7.5 ± 0.5 ab |
| 20:4n-3 | 2.0 ± 0.3 | 1.9 ± 0.3 | 2.2 ± 0.3 | 1.9 ± 0.3 | 1.7 ± 0.3 | 2.1 ± 0.3 | 1.9 ± 0.3 | 2.1 ± 0.3 |
| 20:2n-6 | 1.4 ± 0.2 bc | 1.1 ± 0.2 c | 1.5 ± 0.2 bc | 2.5 ± 0.2 a | 1.2 ± 0.2c | 1.7 ± 0.2 bc | 1.9 ± 0.2 abc | 2.1 ± 0.2 ab |
| 20:0 | 4.4 ± 0.7 | 3 ± 0.7 | 3.3 ± 0.7 | 4.4 ± 0.7 | 3.9 ± 0.7 | 3.4 ± 0.7 | 2.7 ± 0.7 | 4.2 ± 0.7 |
| 22:5n-6 DPA-6 | 1.2 ± 0.2 | 1 ± 0.2 | 1.2 ± 0.2 | 1.4 ± 0.2 | 1.6 ± 0.2 | 1.2 ± 0.2 | 1.3 ± 0.2 | 1.2 ± 0.2 |
| 22:6n-3 DHA | 6.7 ± 0.8 | 7.1 ± 0.8 | 5 ± 0.8 | 7.1 ± 0.8 | 7 ± 0.8 | 7.1 ± 0.8 | 6.1 ± 0.8 | 5.7 ± 0.8 |
| 22:5n-3 DPA-3 | 21.8 ± 1.1 abc | 24.5 ± 1.1 a | 21.7 ± 1.1 abc | 22.9 ± 1.1 ab | 18.8 ± 1.1c | 23.7 ± 1.1 a | 20.2 ± 1.1 bc | 19.5 ± 1.1 c |
| 22:0 | 1.5 ± 0.1 | 1.6 ± 0.1 | 1.4 ± 0.1 | 1.5 ± 0.1 | 1.3 ± 0.1 | 1.4 ± 0.1 | 1.3 ± 0.1 | 1.5 ± 0.1 |
| 23:0 | 2.1 ± 0.1 a | 2 ± 0.1 a | 1.3 ± 0.1 c | 1.8 ± 0.1 ab | 1.4 ± 0.1 bc | 1.8 ± 0.1 ab | 1.5 ± 0.1 bc | 1.7 ± 0.1 abc |
| 24:0 | 2.3 ± 0.1 a | 2.2 ± 0.1 a | 1.8 ± 0.1 b | 2.1 ± 0.1 ab | 1.9 ± 0.1 ab | 2.1 ± 0.1 ab | 1.9 ± 0.1 ab | 2.1 ± 0.1 ab |
| Total FA | 3291.8 ± 467.8 ab | 2514.6 ± 467.8 ab | 2162.5 ± 467.8 ab | 3513.2 ± 467.8 a | 2496 ± 467.8 ab | 2762 ± 467.8 ab | 2070.8 ± 467.8 b | 3161.8 ± 467.8 ab |
| ∑SFA | 1427.0 ± 208.0 a | 1001.5 ± 208.0 ab | 837.6 ± 208.0 ab | 1407.3 ± 208.0 a | 997 ± 208.0 ab | 1073.8 ± 208.0 ab | 782 ± 208.0 b | 1278.9 ± 208.0 ab |
| ∑MUFA | 1469.1 ± 242.9 ab | 1138.6 ± 242.9 ab | 977.0 ± 242.9 ab | 1685.7 ± 242.9 a | 1162.8 ± 242.9 ab | 1297.6 ± 242.9 ab | 952.2 ± 242.9 b | 1485.8 ± 242.9 ab |
| ∑PUFA | 294.7 ± 17.3 abcd | 306.2 ± 17.3 abc | 268.3 ± 17.3 bcd | 329.8 ± 17.3 a | 247.4 ± 17.3 d | 312.0 ± 17.3 ab | 257.4 ± 17.3 cd | 301.6 ± 17.3 abc |
| ∑n-3 LC-PUFA | 55.2 ± 3.6 abc | 60.4 ± 3.6 a | 51.0 ± 3.6 abc | 56.0 ± 3.6 abc | 47.2 ± 3.6 c | 57.8 ± 3.6 ab | 48.3 ± 3.6 bc | 46.4 ± 3.6 c |
| ∑n-3 PUFA | 123.2 ± 7.7 a | 129.8 ± 7.7 a | 85.5 ± 7.7 b | 114.4 ± 7.7 a | 83.1 ± 7.7 b | 110.2 ± 7.7 a | 84.1 ± 7.7 b | 86.1 ± 7.7 b |
| ∑n-6 PUFA | 164.9 ± 10.8 bc | 170.9 ± 10.8 bc | 175 ± 10.8 bc | 208.8 ± 10.8 a | 157.4 ± 10.8 c | 196 ± 10.8 ab | 167.4 ± 10.8 bc | 209.2 ± 10.8 a |
| ∑other FA | 99.6 ± 12.8 | 67.0 ± 12.8 | 78.3 ± 12.8 | 89.4 ± 12.8 | 87.6 ± 12.8 | 78.1 ± 12.8 | 78.8 ± 12.8 | 94.4 ± 12.8 |
| n-6/n-3 | 1.4 ± 0.1 c | 1.3 ± 0.1 c | 2.1 ± 0.1 b | 1.9 ± 0.1 b | 1.9 ± 0.1 b | 1.8 ± 0.1 b | 2.0 ± 0.1 b | 2.4 ± 0.1 a |
| Items | Control | NOP | CO | RBO | ||||
|---|---|---|---|---|---|---|---|---|
| CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | |
| Lipid percentage | 6.8 ± 0.4 | 6.9 ± 0.4 | 6.3 ± 0.4 | 7.0 ± 0.4 | 6.7 ± 0.4 | 6.7 ± 0.4 | 6.5 ± 0.4 | 6.5 ± 0.4 |
| 14:0 | 23.8 ± 5.0 ab | 21.4 ± 5.0 b | 38.4 ± 5.0 a | 22.7 ± 5.0 b | 28.4 ± 5.0 ab | 26.3 ± 5.0 ab | 22.6 ± 5.0 b | 17.7 ± 5.0 b |
| 15:0 | 12.9 ± 1.2 bc | 12.2 ± 1.2 c | 18.3 ± 1.2 a | 13.4 ± 1.2 bc | 15.9 ± 1.2 ab | 14.1 ± 1.2 bc | 13.4 ± 1.2 bc | 13.0 ± 1.2 bc |
| 16:1n-9c | 22.2 ± 4.0 | 21.8 ± 4.0 | 31.1 ± 4.0 | 23.1 ± 4.0 | 24.8 ± 4.0 | 21.5 ± 4.0 | 21.2 ± 4.0 | 19.4 ± 4.0 |
| 16:1n-7c | 33.9 ± 10.4 ab | 32.2 ± 10.4 ab | 60.2 ± 10.4 a | 29.6 ± 10.4 b | 36.5 ± 10.4 ab | 32.5 ± 10.4 ab | 28.0 ± 10.4 b | 26.4 ± 10.4 b |
| 16:0 | 723.7 ± 83.3 | 686.9 ± 83.3 | 879.1 ± 83.3 | 703.5 ± 83.3 | 786.6 ± 83.3 | 835.2 ± 83.3 | 660.6 ± 83.3 | 665.8 ± 83.3 |
| 17:0 | 51.3 ± 3.8 ab | 55.8 ± 3.8 ab | 57.4 ± 3.8 ab | 57.4 ± 3.8 ab | 56.5 ± 3.8 ab | 61.7 ± 3.8 a | 47.2 ± 3.8 b | 58.4 ± 3.8 ab |
| 18:2n-6 LA | 370.2 ± 48.4 | 384.9 ± 48.4 | 394.2 ± 48.4 | 396.2 ± 48.4 | 419.6 ± 48.4 | 412.1 ± 48.4 | 327.0 ± 48.4 | 402.4 ± 48.4 |
| 18:3n-3 ALA | 108.0 ± 22.8 b | 167.5 ± 22.8 ab | 180.0 ± 22.8 a | 162.1 ± 22.8 ab | 164.1 ± 22.8 ab | 184.8 ± 22.8 a | 106.2 ± 22.8 b | 109.0 ± 22.8 b |
| 18:1n-9c | 928.1 ± 148.5 | 850.4 ± 148.5 | 1174.3 ± 148.5 | 800.9 ± 148.5 | 977.1 ± 148.5 | 950.8 ± 148.5 | 864.2 ± 148.5 | 807.2 ± 148.5 |
| 18:1n-7c | 75.4 ± 15.0 | 67.7 ± 15.0 | 90.9 ± 15.0 | 66.3 ± 15.0 | 72.1 ± 15.0 | 73.0 ± 15.0 | 69.8 ± 15.0 | 71.8 ± 15.0 |
| 18:1n-7t | 230.7 ± 43.5 | 237.5 ± 43.5 | 238.4 ± 43.5 | 213.8 ± 43.5 | 254.0 ± 43.5 | 240.4 ± 43.5 | 228.4 ± 43.5 | 217.2 ± 43.5 |
| 18:0 | 876.1 ± 65.8 ab | 859.9 ± 65.8 ab | 923.4 ± 65.8 ab | 808.2 ± 65.8 b | 918.5 ± 65.8 ab | 1023.0 ± 65.8 a | 786.0 ± 65.8 b | 869.3 ± 65.8 ab |
| 20:4n-6 ARA | 260.6 ± 23.1 | 203.5 ± 23.1 | 211.9 ± 23.1 | 204.4 ± 23.1 | 240.2 ± 23.1 | 238.7 ± 23.1 | 200.1 ± 23.1 | 249.0 ± 23.1 |
| 20:5n-3 EPA | 75.4 ± 14.4 bc | 117.2 ± 14.4 ab | 105.1 ± 14.4 abc | 108.8 ± 14.4 abc | 114.7 ± 14.4 ab | 117.7 ± 14.4 a | 68.1 ± 14.4 c | 82.2 ± 14.4 abc |
| 20:3n-6 | 31.6 ± 3.3 abc | 37.7 ± 3.3 abc | 30.3 ± 3.3 bc | 36.8 ± 3.3 abc | 39.4 ± 3.3 abc | 39.7 ± 3.3 ab | 29.7 ± 3.3 c | 40.4 ± 3.3 a |
| 20:4n-3 | 9.4 ± 1.3 b | 9.6 ± 1.3 b | 9.8 ± 1.3 b | 10.8 ± 1.3 ab | 11.4 ± 1.3 ab | 14.0 ± 1.3 a | 9.4 ± 1.3 b | 11.7 ± 1.3 ab |
| 20:2n-6 | 5.5 ± 0.8 b | 6.3 ± 0.8 ab | 5.0 ± 0.8 b | 5.8 ± 0.8 b | 8.1 ± 0.8 a | 7.1 ± 0.8 ab | 5.2 ± 0.8 b | 6.0 ± 0.8 ab |
| 20:0 | 6.0 ± 0.7 ab | 5.5 ± 0.7 ab | 6.0 ± 0.7 ab | 4.6 ± 0.7 b | 7.0 ± 0.7 a | 6.6 ± 0.7 ab | 5.4 ± 0.7 ab | 4.8 ± 0.7 b |
| 22:5n-6 DPA-6 | 9.2 ± 2.2 | 4.8 ± 2.2 | 9.3 ± 2.2 | 3.9 ± 2.2 | 5.1 ± 2.2 | 8.6 ± 2.2 | 9.7 ± 2.2 | 4.8 ± 2.2 |
| 22:6n-3 DHA | 173.1 ± 26.2 | 132.8 ± 26.2 | 149.5 ± 26.2 | 156.8 ± 26.2 | 196.5 ± 26.2 | 207.8 ± 26.2 | 159.8 ± 26.2 | 165.9 ± 26.2 |
| 22:5n-3 DPA-3 | 210.5 ± 20.2 ab | 258.9 ± 20.2 a | 201.1 ± 20.2 ab | 242.5 ± 20.2 a | 249.9 ± 20.2 a | 223.8 ± 20.2 ab | 171.5 ± 20.2 b | 218.5 ± 20.2 ab |
| 22:0 | 8.5 ± 0.4 ab | 7.9 ± 0.4 b | 8.4 ± 0.4 b | 7.4 ± 0.4 b | 9.8 ± 0.4 a | 8.2 ± 0.4 b | 7.6 ± 0.4 b | 8.3 ± 0.4 b |
| 23:0 | 16.4 ± 2.2 | 18.3 ± 2.2 | 20.2 ± 2.2 | 21.3 ± 2.2 | 20.3 ± 2.2 | 22.1 ± 2.2 | 18.5 ± 2.2 | 22.2 ± 2.2 |
| 24:0 | 15.6 ± 0.9 ab | 15.1 ± 0.9 ab | 16.5 ± 0.9 ab | 14.4 ± 0.9 b | 17.7 ± 0.9 a | 16.2 ± 0.9 ab | 14.6 ± 0.9 b | 16.0 ± 0.9 ab |
| Total FA | 4662.0 ± 446.0 | 4580.7 ± 446.0 | 5334.2 ± 446.0 | 4476.4 ± 446.0 | 5075.9 ± 446.0 | 5183.5 ± 446.0 | 4246.8 ± 446.0 | 4480.4 ± 446.0 |
| ∑SFA | 1734.2 ± 143.1 ab | 1682.7 ± 143.1 ab | 1967.7 ± 143.1 ab | 1652.8 ± 143.1 ab | 1860.5 ± 143.1 ab | 2013.2 ± 143.1 a | 1575.7 ± 143.1 b | 1675.3 ± 143.1 ab |
| ∑MUFA | 1490.4 ± 231.0 | 1406.3 ± 231.0 | 1844.3 ± 231.0 | 1343.8 ± 231.0 | 1578.8 ± 231.0 | 1527.9 ± 231.0 | 1417.2 ± 231.0 | 1344.9 ± 231.0 |
| ∑PUFA | 1318.3 ± 96.5 ab | 1376.1 ± 96.5 ab | 1369.8 ± 96.5 ab | 1376.9 ± 96.5 ab | 1509.0 ± 96.5 a | 1510.4 ± 96.5 a | 1137.4 ± 96.5 b | 1356.4 ± 96.5 ab |
| ∑n-3 LC-PUFA | 468.4 ± 49.6 ab | 518.4 ± 49.6 ab | 465.4 ± 49.6 ab | 518.8 ± 49.6 ab | 572.6 ± 49.6 a | 563.3 ± 49.6 a | 408.7 ± 49.6 b | 478.3 ± 49.6 ab |
| ∑n-3 PUFA | 582.6 ± 68.2 ab | 691.3 ± 68.2 ab | 658.0 ± 68.2 ab | 687.3 ± 68.2 ab | 744.4 ± 68.2 a | 753.2 ± 68.2 a | 520.5 ± 68.2 a | 593.5 ± 68.2 ab |
| ∑n-6 PUFA | 708.9 ± 76.1 | 662.8 ± 76.1 | 685.7 ± 76.1 | 673.4 ± 76.1 | 741.1 ± 76.1 | 733.8 ± 76.1 | 597.5 ± 76.1 | 740.5 ± 76.1 |
| ∑other FA | 118.5 ± 16.3 | 114.8 ± 16.3 | 150.2 ± 16.3 | 102.6 ± 16.3 | 126.7 ± 16.3 | 131.3 ± 16.3 | 115.0 ± 16.3 | 103.6 ± 16.3 |
| n-6/n-3 | 1.2 ± 0.2 | 1.0 ± 0.2 | 1.1 ± 0.2 | 1.0 ± 0.2 | 1.0 ± 0.2 | 1.1 ± 0.2 | 1.2 ± 0.2 | 1.3 ± 0.2 |
| Items | Control | NOP | CO | RBO | ||||
|---|---|---|---|---|---|---|---|---|
| CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | |
| Lipid percentage | 2.3 ± 0.1 | 2.3 ± 0.1 | 2.4 ± 0.1 | 2.3 ± 0.1 | 2.4 ± 0.1 | 2.3 ± 0.1 | 2.3 ± 0.1 | 2.4 ± 0.1 |
| 14:0 | 6.0 ± 2.1 | 5.7 ± 2.1 | 9.5 ± 2.1 | 5.7 ± 2.1 | 3.9 ± 2.1 | 6.7 ± 2.1 | 7.7 ± 2.1 | 5.9 ± 2.1 |
| 15:0 | 2.6 ± 0.5 | 2.6 ± 0.5 | 3.2 ± 0.5 | 3.0 ± 0.5 | 2.5 ± 0.5 | 3.1 ± 0.5 | 3.3 ± 0.5 | 2.7 ± 0.5 |
| 16:1n-9c | 2.7 ± 0.4 | 2.8 ± 0.4 | 3.1 ± 0.4 | 2.7 ± 0.4 | 2.4 ± 0.4 | 2.6 ± 0.4 | 3.4 ± 0.4 | 2.4 ± 0.4 |
| 16:1n-7c | 5.2 ± 0.9 | 5.1 ± 0.9 | 6.4 ± 0.9 | 5.0 ± 0.9 | 4.2 ± 0.9 | 4.8 ± 0.9 | 6.7 ± 0.9 | 4.9 ± 0.9 |
| 16:0 | 184.4 ± 13.4 | 197.6 ± 13.4 | 193.9 ± 13.4 | 196.5 ± 13.4 | 168.5 ± 13.4 | 187.7 ± 13.4 | 202.1 ± 13.4 | 194.9 ± 13.4 |
| 17:0 | 13.3 ± 1.6 | 15.6 ± 1.6 | 14.6 ± 1.6 | 15.4 ± 1.6 | 12.2 ± 1.6 | 15.3 ± 1.6 | 15.9 ± 1.6 | 14.0 ± 1.6 |
| 18:2n-6 LA | 390.0 ± 24.4 | 416.4 ± 24.4 | 357.8 ± 24.4 | 415.7 ± 24.4 | 392.6 ± 24.4 | 401.2 ± 24.4 | 371.0 ± 24.4 | 400.8 ± 24.4 |
| 18:3n-3 ALA | 33.0 ± 10.2 | 55.2 ± 10.2 | 53.9 ± 10.2 | 53.8 ± 10.2 | 33.5 ± 10.2 | 35.0 ± 10.2 | 54.4 ± 10.2 | 52.6 ± 10.2 |
| 18:1n-9c | 208.2 ± 34.6 | 205.4 ± 34.6 | 239.7 ± 34.6 | 192.0 ± 34.6 | 200.8 ± 34.6 | 195.4 ± 34.6 | 257.4 ± 34.6 | 187.3 ± 34.6 |
| 18:1n-7c | 40.6 ± 3.4 | 37.3 ± 3.4 | 31.8 ± 3.4 | 38.8 ± 3.4 | 36.8 ± 3.4 | 36.4 ± 3.4 | 38.0 ± 3.4 | 33.0 ± 3.4 |
| 18:1n-7t | 44.3 ± 6.3 | 46.2 ± 6.3 | 41.8 ± 6.3 | 46.7 ± 6.3 | 51.8 ± 6.3 | 49.4 ± 6.3 | 43.5 ± 6.3 | 43.7 ± 6.3 |
| 18:0 | 278.7 ± 26.1 | 267.3 ± 26.1 | 312.6 ± 26.1 | 277.4 ± 26.1 | 269.5 ± 26.1 | 284.4 ± 26.1 | 303.2 ± 26.1 | 268.2 ± 26.1 |
| 20:4n-6 ARA | 124.4 ± 8.1 a | 94.8 ± 8.1 b | 90.1 ± 8.1 b | 105.7 ± 8.1 ab | 100.9 ± 8.1 b | 111.2 ± 8.1 ab | 105.9 ± 8.1 ab | 99.0 ± 8.1 b |
| 20:5n-3 EPA | 33.7 ± 4.5 | 37.2 ± 4.5 | 31.2 ± 4.5 | 38.4 ± 4.5 | 27.0 ± 4.5 | 28.3 ± 4.5 | 38.1 ± 4.5 | 36.9 ± 4.5 |
| 20:3n-6 | 11.5 ± 0.5 a | 11.5 ± 0.5 a | 9.8 ± 0.5 b | 11.5 ± 0.5 a | 10.9 ± 0.5 ab | 11.7 ± 0.5 a | 10.9 ± 0.5 ab | 11.2 ± 0.5 ab |
| 20:4n-3 | 2.1 ± 0.3 | 2.0 ± 0.3 | 2.2 ± 0.3 | 1.9 ± 0.3 | 2.1 ± 0.3 | 1.5 ± 0.3 | 1.9 ± 0.3 | 2.1 ± 0.3 |
| 20:2n-6 | 2.0 ± 0.2 ab | 2.3 ± 0.2 a | 1.7 ± 0.2 b | 1.9 ± 0.2 ab | 2.0 ± 0.2 ab | 2.0 ± 0.2 ab | 1.8 ± 0.2 b | 1.9 ± 0.2 ab |
| 20:0 | 3.5 ± 0.3 ab | 3.4 ± 0.3 ab | 3.6 ± 0.3 ab | 3.1 ± 0.3 b | 3.3 ± 0.3 ab | 3.4 ± 0.3 ab | 4.0 ± 0.3 a | 3.2 ± 0.3 b |
| 22:5n-6 DPA-6 | 0.9 ± 0.2 ab | 1.1 ± 0.2 ab | 0.7 ± 0.2 b | 1.1 ± 0.2 ab | 1.4 ± 0.2 a | 1.1 ± 0.2 ab | 1.2 ± 0.2 ab | 0.9 ± 0.2 ab |
| 22:6n-3 DHA | 18.7 ± 1.9 a | 13.1 ± 1.9 b | 12.7 ± 1.9 b | 16.7 ± 1.9 ab | 17.3 ± 1.9 ab | 16.0 ± 1.9 ab | 17.8 ± 1.9 ab | 17.9 ± 1.9 ab |
| 22:5n-3 DPA-3 | 37.5 ± 2.3 a | 36.2 ± 2.3 abc | 26.8 ± 2.3 d | 36.8 ± 2.3 ab | 29.8 ± 2.3 cd | 32.1 ± 2.3 abcd | 36.8 ± 2.3 ab | 30.3 ± 2.3 bcd |
| 22:0 | 5.7 ± 0.3 | 5.6 ± 0.3 | 5.4 ± 0.3 | 5.7 ± 0.3 | 5.5 ± 0.3 | 5.7 ± 0.3 | 6.0 ± 0.3 | 5.7 ± 0.3 |
| 23:0 | 7.4 ± 0.8 b | 8.2 ± 0.8 ab | 8.5 ± 0.8 ab | 9.8 ± 0.8 a | 9.2 ± 0.8 ab | 9.4 ± 0.8 ab | 8.5 ± 0.8 ab | 9.6 ± 0.8 ab |
| 24:0 | 5.4 ± 0.3 | 5.9 ± 0.3 | 5.6 ± 0.3 | 6.1 ± 0.3 | 6.0 ± 0.3 | 6.2 ± 0.3 | 5.8 ± 0.3 | 6.2 ± 0.3 |
| Total FA | 1743.8 ± 87.7 | 1768.3 ± 87.7 | 1719.2 ± 87.7 | 1777.5 ± 87.7 | 1655.1 ± 87.7 | 1721.1 ± 87.7 | 1823.7 ± 87.7 | 1682.4 ± 87.7 |
| ∑SFA | 506.8 ± 42.9 | 511.9 ± 42.9 | 557.0 ± 42.9 | 522.7 ± 42.9 | 480.4 ± 42.9 | 521.8 ± 42.9 | 556.4 ± 42.9 | 510.2 ± 42.9 |
| ∑MUFA | 387.9 ± 41.3 | 374.7 ± 41.3 | 405.1 ± 41.3 | 366.7 ± 41.3 | 387.0 ± 41.3 | 375.6 ± 41.3 | 435.7 ± 41.3 | 361.7 ± 41.3 |
| ∑PUFA | 671.6 ± 22.4 a | 685.2 ± 22.4 a | 602.0 ± 22.4 b | 697.5 ± 22.4 a | 634.7 ± 22.4 ab | 656.1 ± 22.4 ab | 654.3 ± 22.4 ab | 667.2 ± 22.4 ab |
| ∑n-3 LC-PUFA | 92.0 ± 7.7 | 88.4 ± 7.7 | 72.8 ± 7.7 | 93.8 ± 7.7 | 76.2 ± 7.7 | 77.9 ± 7.7 | 94.7 ± 7.7 | 87.2 ± 7.7 |
| ∑n-3 PUFA | 125.2 ± 17.0 | 143.8 ± 17.0 | 127.2 ± 17.0 | 147.6 ± 17.0 | 110.1 ± 17.0 | 113.1 ± 17.0 | 149.2 ± 17.0 | 140.2 ± 17.0 |
| ∑n-6 PUFA | 534.1 ± 29.3 | 530.8 ± 29.3 | 464.4 ± 29.3 | 541.1 ± 29.3 | 512.2 ± 29.3 | 532.7 ± 29.3 | 495.2 ± 29.3 | 518.4 ± 29.3 |
| ∑other FA | 177.5 ± 18.4 | 196.5 ± 18.4 | 155.1 ± 18.4 | 190.4 ± 18.4 | 153.0 ± 18.4 | 167.5 ± 18.4 | 177.3 ± 18.4 | 143.3 ± 18.4 |
| n-6/n-3 | 4.4 ± 0.7 | 3.9 ± 0.7 | 4.3 ± 0.7 | 3.8 ± 0.7 | 4.7 ± 0.7 | 4.8 ± 0.7 | 3.6 ± 0.7 | 4.3 ± 0.7 |
| Items | Control | NOP | CO | RBO | ||||
|---|---|---|---|---|---|---|---|---|
| CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | |
| Lipid percentage | 2.8 ± 0.1 | 2.9 ± 0.1 | 2.8 ± 0.1 | 3.0 ± 0.1 | 2.9 ± 0.1 | 3.0 ± 0.1 | 2.9 ± 0.1 | 3.1 ± 0.1 |
| 14:0 | 6.4 ± 1.3 | 3.7 ± 1.3 | 5.9 ± 1.3 | 3.9 ± 1.3 | 5.0 ± 1.3 | 4.0 ± 1.3 | 7.1 ± 1.3 | 5.2 ± 1.3 |
| 15:0 | 4.5 ± 0.4 ab | 3.7 ± 0.4 b | 4.6 ± 0.4 ab | 4.1 ± 0.4 ab | 4.3 ± 0.4 ab | 4.4 ± 0.4 ab | 4.8 ± 0.4 a | 4.3 ± 0.4 ab |
| 16:1n-9c | 4.9 ± 0.7 | 3.2 ± 0.7 | 4.4 ± 0.7 | 3.5 ± 0.7 | 4.2 ± 0.7 | 3.8 ± 0.7 | 4.4 ± 0.7 | 3.7 ± 0.7 |
| 16:1n-7c | 6.4 ± 0.8 | 4.7 ± 0.8 | 6.2 ± 0.8 | 4.4 ± 0.8 | 4.8 ± 0.8 | 4.9 ± 0.8 | 6.3 ± 0.8 | 5.1 ± 0.8 |
| 16:0 | 318.9 ± 14.6 a | 272.2 ± 14.6 b | 304.4 ± 14.6 ab | 305.8 ± 14.6 ab | 286.8 ± 14.6 ab | 291.2 ± 14.6 ab | 305.2 ± 14.6 ab | 311.7 ± 14.6 ab |
| 17:0 | 18.4 ± 0.8 abc | 18.1 ± 0.8 abc | 18.0 ± 0.8 bc | 20.4 ± 0.8 a | 17.3 ± 0.8 c | 19.5 ± 0.8 abc | 18.9 ± 0.8 abc | 19.7 ± 0.8 ab |
| 18:2n-6 LA | 228.0 ± 14.2 | 215.3 ± 14.2 | 221.0 ± 14.2 | 250.0 ± 14.2 | 240.5 ± 14.2 | 250.3 ± 14.2 | 229.2 ± 14.2 | 228.1 ± 14.2 |
| 18:3n-3 ALA | 23.8 ± 4.7 | 29.4 ± 4.7 | 33.0 ± 4.7 | 28.0 ± 4.7 | 22.2 ± 4.7 | 23.0 ± 4.7 | 34.7 ± 4.7 | 31.7 ± 4.7 |
| 18:1n-9c | 240.8 ± 15.5 | 200.1 ± 15.5 | 226.3 ± 15.5 | 213.8 ± 15.5 | 226.3 ± 15.5 | 219.5 ± 15.5 | 232.9 ± 15.5 | 211.9 ± 15.5 |
| 18:1n-7c | 34.0 ± 2.6 | 27.8 ± 2.6 | 27.5 ± 2.6 | 30.1 ± 2.6 | 30.1 ± 2.6 | 30.5 ± 2.6 | 27.5 ± 2.6 | 27.2 ± 2.6 |
| 18:1n-7t | 37.1 ± 6.6 | 34.3 ± 6.6 | 31.6 ± 6.6 | 36.5 ± 6.6 | 39.6 ± 6.6 | 37.9 ± 6.6 | 39.2 ± 6.6 | 36.6 ± 6.6 |
| 18:0 | 319.0 ± 16.0 | 291.3 ± 16.0 | 308.4 ± 16.0 | 336.3 ± 16.0 | 310.7 ± 16.0 | 301.9 ± 16.0 | 324.5 ± 16.0 | 318.6 ± 16.0 |
| 20:4n-6 ARA | 236.9 ± 16.9 a | 184.8 ± 16.9 b | 202.9 ± 16.9 ab | 221.7 ± 16.9 ab | 214.6 ± 16.9 ab | 218.0 ± 16.9 ab | 210.7 ± 16.9 ab | 218.6 ± 16.9 ab |
| 20:5n-3 EPA | 57.2 ± 11.7 | 70.2 ± 11.7 | 70.1 ± 11.7 | 76.2 ± 11.7 | 49.9 ± 11.7 | 49.8 ± 11.7 | 75.5 ± 11.7 | 68.9 ± 11.7 |
| 20:3n-6 | 15.5 ± 1.4 b | 16.2 ± 1.4 b | 15.2 ± 1.4b | 20.4 ± 1.4 a | 15.1 ± 1.4 b | 16.2 ± 1.4 b | 15.5 ± 1.4 b | 18.3 ± 1.4 ab |
| 20:4n-3 | 2.7 ± 0.6 | 3.2 ± 0.6 | 4.2 ± 0.6 | 2.8 ± 0.6 | 2.5 ± 0.6 | 3.0 ± 0.6 | 3.8 ± 0.6 | 4.3 ± 0.6 |
| 20:2n-6 | 4.1 ± 0.6 | 4.6 ± 0.6 | 4.9 ± 0.6 | 5.4 ± 0.6 | 4.6 ± 0.6 | 5.8 ± 0.6 | 4.2 ± 0.6 | 5.8 ± 0.6 |
| 20:0 | 5.4 ± 0.4 | 5.0 ± 0.4 | 5.5 ± 0.4 | 5.5 ± 0.4 | 4.9 ± 0.4 | 5.8 ± 0.4 | 5.4 ± 0.4 | 5.6 ± 0.4 |
| 22:5n-6 DPA-6 | 1.5 ± 0.1 a | 0.9 ± 0.1 b | 1.1 ± 0.1 ab | 1.0 ± 0.1 b | 1.5 ± 0.1 a | 1.0 ± 0.1 b | 1.1 ± 0.1 ab | 1.0 ± 0.1 b |
| 22:6n-3 DHA | 51.4 ± 5.4 ab | 37.7 ± 5.4 b | 47.3 ± 5.4 ab | 49.9 ± 5.4 ab | 55.6 ± 5.4 a | 45.8 ± 5.4 ab | 53.0 ± 5.4 ab | 53.7 ± 5.4 ab |
| 22:5n-3 DPA-3 | 66.1 ± 5.8 b | 72.4 ± 5.8 ab | 71.6 ± 5.8 ab | 84.4 ± 5.8 a | 63.7 ± 5.8 b | 64.5 ± 5.8 b | 68.5 ± 5.8 ab | 72.1 ± 5.8 ab |
| 22:0 | 29.5 ± 1.9 ab | 26.2 ± 1.9 b | 30.1 ± 1.9 ab | 30.7 ± 1.9 ab | 28.2 ± 1.9 ab | 31.8 ± 1.9 a | 29.2 ± 1.9 ab | 29.8 ± 1.9 ab |
| 23:0 | 9.0 ± 0.7 ab | 8.5 ± 0.7 b | 8.9 ± 0.7 ab | 10.3 ± 0.7 ab | 9.3 ± 0.7 ab | 10.4 ± 0.7 a | 9.6 ± 0.7 ab | 10.0 ± 0.7 ab |
| 24:0 | 31.5 ± 2.6 | 30.1 ± 2.6 | 32.4 ± 2.6 | 33.8 ± 2.6 | 32.8 ± 2.6 | 32.1 ± 2.6 | 30.9 ± 2.6 | 33.0 ± 2.6 |
| Total FA | 1924.5 ± 75.8 ab | 1710.2 ± 75.8 b | 1852.6 ± 75.8 ab | 1953.3 ± 75.8 a | 1840.2 ± 75.8 ab | 1850.3 ± 75.8 ab | 1905.0 ± 75.8 ab | 1904.2 ± 75.8 ab |
| ∑SFA | 742.6 ± 32.2 | 658.6 ± 32.2 | 718.1 ± 32.2 | 750.9 ± 32.2 | 699.3 ± 32.2 | 701.2 ± 32.2 | 735.6 ± 32.2 | 737.8 ± 32.2 |
| ∑MUFA | 413.2 ± 25.2 | 347.6 ± 25.2 | 379.0 ± 25.2 | 383.5 ± 25.2 | 392.6 ± 25.2 | 385.1 ± 25.2 | 393.0 ± 25.2 | 372.4 ± 25.2 |
| ∑PUFA | 706.0 ± 26.3 ab | 650.1 ± 26.3 b | 688.5 ± 26.3 ab | 755.2 ± 26.3 a | 685.8 ± 26.3 ab | 696.6 ± 26.3 ab | 712.2 ± 26.3 ab | 722.4 ± 26.3 ab |
| ∑n-3 LC-PUFA | 177.3 ± 19.6 | 183.6 ± 19.6 | 193.1 ± 19.6 | 213.2 ± 19.6 | 171.6 ± 19.6 | 163.2 ± 19.6 | 200.7 ± 19.6 | 202.2 ± 19.6 |
| ∑n-3 PUFA | 201.1 ± 23.2 | 213.0 ± 23.2 | 226.2 ± 23.2 | 241.3 ± 23.2 | 193.8 ± 23.2 | 186.2 ± 23.2 | 235.5 ± 23.2 | 234.0 ± 23.2 |
| ∑n-6 PUFA | 493.2 ± 26.7 ab | 427.2 ± 26.7 b | 451.9 ± 26.7 ab | 505.4 ± 26.7 a | 482.6 ± 26.7 ab | 500.4 ± 26.7 ab | 466.4 ± 26.7 ab | 479.0 ± 26.7 ab |
| ∑other FA | 62.7 ± 4.3 ab | 53.9 ± 4.3 b | 67.0 ± 4.3 a | 63.7 ± 4.3 ab | 62.5 ± 4.3 ab | 67.5 ± 4.3 a | 64.2 ± 4.3 ab | 71.6 ± 4.3 a |
| n-6/n-3 | 2.5 ± 0.3 | 2.1 ± 0.3 | 2.2 ± 0.3 | 2.2 ± 0.3 | 2.5 ± 0.3 | 2.7 ± 0.3 | 2.0 ± 0.3 | 2.2 ± 0.3 |
| Items | Liver | p | Kidney | p | Heart | p | Muscle | p | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | CFP | Lucerne | |||||
| Lipid percentage | 6.6 ± 0.2 | 6.8 ± 0.2 | 0.460 | 2.9 ± 0.1 | 3.0 ± 0.1 | 0.055 | 2.4 ± 0.1 | 2.3 ± 0.1 | 0.865 | 2.9 ± 0.3 | 3.1 ± 0.3 | 0.697 |
| 14:0 | 28.3 ± 2.5 | 22.0 ± 2.5 | 0.089 | 6.1 ± 0.7 a | 4.2 ± 0.7 b | 0.047 | 6.8 ± 1.1 | 6.0 ± 1.1 | 0.608 | 48.8 ± 6.9 | 52.3 ± 6.9 | 0.727 |
| 15:0 | 15.1 ± 0.6 | 13.2 ± 0.6 | 0.029 | 4.6 ± 0.2 | 4.1 ± 0.2 | 0.097 | 2.9 ± 0.2 | 2.9 ± 0.2 | 0.971 | 6.5 ± 0.8 | 7.5 ± 0.8 | 0.381 |
| 16:1n-9c | 24.8 ± 2.0 | 21.4 ± 2.0 | 0.245 | 4.5 ± 0.4 | 3.5 ± 0.4 | 0.088 | 2.9 ± 0.2 | 2.6 ± 0.2 | 0.374 | 6.0 ± 0.7 | 6.8 ± 0.7 | 0.361 |
| 16:1n-7c | 39.6 ± 5.2 | 30.2 ± 5.2 | 0.209 | 5.9 ± 0.4 a | 4.7 ± 0.4 b | 0.049 | 5.6 ± 0.4 | 4.9 ± 0.4 | 0.260 | 29.6 ± 3.8 | 34.9 ± 3.8 | 0.347 |
| 16:0 | 762.5 ± 41.7 | 722.8 ± 41.7 | 0.508 | 303.8 ± 7.3 | 295.2 ± 7.3 | 0.413 | 187.2 ± 6.7 | 194.2 ± 6.7 | 0.470 | 527.2 ± 56.9 | 637.6 ± 56.9 | 0.183 |
| 17:0 | 53.1 ± 1.9 | 58.3 ± 1.9 | 0.063 | 18.1 ± 0.4 b | 19.4 ± 0.4 a | 0.031 | 14.0 ± 0.8 | 15.1 ± 0.8 | 0.341 | 22.4 ± 2.8 | 29.9 ± 2.8 | 0.064 |
| 18:2n-6 LA | 377.7 ± 24.2 | 398.9 ± 24.2 | 0.542 | 229.7 ± 7.1 | 235.9 ± 7.1 | 0.540 | 377.8 ± 12.2 | 408.5 ± 12.2 | 0.088 | 118.3 ± 4.4 b | 145.3 ± 4.4 a | 0.000 |
| 18:3n-3 ALA | 139.6 ± 11.4 | 155.8 ± 11.4 | 0.324 | 28.4 ± 2.3 | 28.0 ± 2.3 | 0.912 | 43.7 ± 5.1 | 49.1 ± 5.1 | 0.460 | 43.1 ± 2.8 b | 54.1 ± 2.8 a | 0.009 |
| 18:1n-9c | 985.9 ± 74.2 | 852.3 ± 74.2 | 0.215 | 231.6 ± 7.7 | 211.3 ± 7.7 | 0.076 | 226.5 ± 17.3 | 195.0 ± 17.3 | 0.211 | 905.4 ± 98.5 | 1108.3 ± 98.5 | 0.158 |
| 18:1n-7c | 77.0 ± 7.5 | 69.7 ± 7.5 | 0.495 | 29.8 ± 1.3 | 28.9 ± 1.3 | 0.632 | 36.8 ± 1.7 | 36.4 ± 1.7 | 0.864 | 39.3 ± 3.8 | 48.3 ± 3.8 | 0.103 |
| 18:1n-7t | 237.9 ± 21.7 | 227.2 ± 21.7 | 0.732 | 36.9 ± 3.3 | 36.3 ± 3.3 | 0.906 | 45.4 ± 3.1 | 46.5 ± 3.1 | 0.800 | 66.1 ± 9.2 | 91.2 ± 9.2 | 0.064 |
| 18:0 | 876.0 ± 32.9 | 890.1 ± 32.9 | 0.764 | 315.6 ± 8.0 | 312.0 ± 8.0 | 0.752 | 291.0 ± 13.1 | 274.3 ± 13.1 | 0.376 | 397.6 ± 37.7 | 453.9 ± 37.7 | 0.302 |
| 20:4n-6 ARA | 228.2 ± 11.6 | 223.9 ± 11.6 | 0.794 | 216.2 ± 8.4 | 210.7 ± 8.4 | 0.650 | 105.3 ± 4.0 | 102.7 ± 4.0 | 0.646 | 35.3 ± 1.5 | 36.4 ± 1.5 | 0.582 |
| 20:5n-3 EPA | 90.8 ± 7.2 | 106.5 ± 7.2 | 0.139 | 63.2 ± 5.8 | 66.3 ± 5.8 | 0.709 | 32.5 ± 2.2 | 35.2 ± 2.2 | 0.409 | 21.7 ± 1.0 | 23.8 ± 1.0 | 0.140 |
| 20:3n-6 | 32.7 ± 1.7 | 38.7 ± 1.7 | 0.019 | 15.3 ± 0.7 b | 17.7 ± 0.7 a | 0.024 | 10.7 ± 0.3 | 11.5 ± 0.3 | 0.055 | 6.2 ± 0.2 b | 7.4 ± 0.2 a | 0.001 |
| 20:4n-3 | 10.0 ± 0.7 | 11.5 ± 0.7 | 0.112 | 3.3 ± 0.3 | 3.3 ± 0.3 | 0.968 | 2.1 ± 0.2 | 1.9 ± 0.2 | 0.400 | 1.9 ± 0.1 | 2.0 ± 0.1 | 0.802 |
| 20:2n-6 | 5.9 ± 0.4 | 6.3 ± 0.4 | 0.513 | 4.4 ± 0.3 b | 5.4 ± 0.3 a | 0.043 | 1.9 ± 0.1 | 2.1 ± 0.1 | 0.170 | 1.5 ± 0.1 | 1.8 ± 0.1 | 0.073 |
| 20:0 | 6.1 ± 0.4 | 5.3 ± 0.4 | 0.143 | 5.3 ± 0.2 | 5.5 ± 0.2 | 0.500 | 3.6 ± 0.1 | 3.3 ± 0.1 | 0.114 | 3.6 ± 0.3 | 3.7 ± 0.3 | 0.713 |
| 22:5n-6 DPA-6 | 8.3 ± 1.1 | 5.5 ± 1.1 | 0.081 | 1.3 ± 0.1 a | 1.0 ± 0.1 b | 0.003 | 1.0 ± 0.1 | 1.0 ± 0.1 | 0.922 | 1.3 ± 0.1 | 1.2 ± 0.1 | 0.454 |
| 22:6n-3 DHA | 169.7 ± 13.1 | 165.8 ± 13.1 | 0.833 | 51.8 ± 2.7 | 47.6 ± 2.7 | 0.279 | 16.6 ± 1.0 | 15.9 ± 1.0 | 0.607 | 6.2 ± 0.4 | 6.7 ± 0.4 | 0.363 |
| 22:5n-3 DPA-3 | 208.2 ± 10.1 | 235.9 ± 10.1 | 0.064 | 67.4 ± 2.9 | 73.3 ± 2.9 | 0.166 | 32.7 ± 1.2 | 33.8 ± 1.2 | 0.496 | 20.6 ± 0.5 b | 22.6 ± 0.5 a | 0.015 |
| 22:0 | 8.6 ± 0.2 | 7.9 ± 0.2 | 0.057 | 29.2 ± 0.9 | 29.6 ± 0.9 | 0.775 | 5.6 ± 0.1 | 5.7 ± 0.1 | 0.863 | 1.4 ± 0.1 | 1.5 ± 0.1 | 0.208 |
| 23:0 | 18.8 ± 1.1 | 21.0 ± 1.1 | 0.181 | 9.2 ± 0.3 | 9.8 ± 0.3 | 0.203 | 8.4 ± 0.4 | 9.2 ± 0.4 | 0.138 | 1.6 ± 0.1 b | 1.8 ± 0.1 a | 0.022 |
| 24:0 | 16.1 ± 0.5 | 15.4 ± 0.5 | 0.288 | 31.9 ± 1.3 | 32.2 ± 1.3 | 0.867 | 5.7 ± 0.1 | 6.1 ± 0.1 | 0.067 | 2.0 ± 0.1 | 2.1 ± 0.1 | 0.089 |
| Total FA | 4829.7 ± 223.0 | 4680.3 ± 223.0 | 0.640 | 1880.6 ± 37.9 | 1854.5 ± 37.9 | 0.631 | 1735.4 ± 43.8 | 1737.3 ± 43.8 | 0.976 | 2505.3 ± 233.9 | 2987.9 ± 233.9 | 0.158 |
| ∑SFA | 1784.5 ± 71.5 | 1756.0 ± 71.5 | 0.781 | 723.9 ± 16.1 | 712.1 ± 16.1 | 0.610 | 525.1 ± 21.5 | 516.6 ± 21.5 | 0.782 | 1010.9 ± 104.0 | 1190.4 ± 104.0 | 0.234 |
| ∑MUFA | 1582.7 ± 115.5 | 1405.7 ± 115.5 | 0.290 | 394.4 ± 12.6 | 372.2 ± 12.6 | 0.223 | 403.9 ± 20.7 | 369.7 ± 20.7 | 0.254 | 1140.3 ± 121.4 | 1401.9 ± 121.4 | 0.141 |
| ∑PUFA | 1333.6 ± 48.3 | 1404.9 ± 48.3 | 0.306 | 698.1 ± 13.2 | 706.1 ± 13.2 | 0.673 | 640.6 ± 11.2 b | 676.5 ± 11.2 a | 0.033 | 266.9 ± 8.6 b | 312.4 ± 8.6 a | 0.001 |
| ∑n-3 LC-PUFA | 478.8 ± 24.8 | 519.7 ± 24.8 | 0.255 | 185.7 ± 9.8 | 190.5 ± 9.8 | 0.729 | 83.9 ± 3.9 | 86.8 ± 3.9 | 0.603 | 50.4 ± 1.8 | 55.1 ± 1.8 | 0.074 |
| ∑n-3 PUFA | 626.4 ± 34.1 | 681.3 ± 34.1 | 0.266 | 214.1 ± 11.6 | 218.6 ± 11.6 | 0.788 | 127.9 ± 8.5 | 136.2 ± 8.5 | 0.500 | 94.0 ± 3.8 b | 110.1 ± 3.8 a | 0.007 |
| ∑n-6 PUFA | 683.3 ± 38.0 | 702.6 ± 38.0 | 0.722 | 473.5 ± 13.4 | 478.0 ± 13.4 | 0.816 | 501.5 ± 14.6 | 530.8 ± 14.6 | 0.170 | 166.1 ± 5.4 b | 196.2 ± 5.4 a | 0.001 |
| ∑other FA | 127.6 ± 8.1 | 113.1 ± 8.1 | 0.220 | 64.1 ± 2.1 | 64.2 ± 2.1 | 0.979 | 165.7 ± 9.2 | 174.4 ± 9.2 | 0.510 | 86.1 ± 6.4 | 82.2 ± 6.4 | 0.674 |
| n-6/n-3 | 1.1 ± 0.1 | 1.1 ± 0.1 | 0.957 | 2.3 ± 0.1 | 2.3 ± 0.1 | 0.928 | 4.2 ± 0.3 | 4.2 ± 0.3 | 0.929 | 1.8 ± 0.0 | 1.9 ± 0.0 | 0.788 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le, H.V.; Nguyen, Q.V.; Nguyen, D.V.; Otto, J.R.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Enhanced Omega-3 Polyunsaturated Fatty Acid Contents in Muscle and Edible Organs of Australian Prime Lambs Grazing Lucerne and Cocksfoot Pastures. Nutrients 2018, 10, 1985. https://doi.org/10.3390/nu10121985
Le HV, Nguyen QV, Nguyen DV, Otto JR, Malau-Aduli BS, Nichols PD, Malau-Aduli AEO. Enhanced Omega-3 Polyunsaturated Fatty Acid Contents in Muscle and Edible Organs of Australian Prime Lambs Grazing Lucerne and Cocksfoot Pastures. Nutrients. 2018; 10(12):1985. https://doi.org/10.3390/nu10121985
Chicago/Turabian StyleLe, Hung V., Quang V. Nguyen, Don V. Nguyen, John R. Otto, Bunmi S. Malau-Aduli, Peter D. Nichols, and Aduli E. O. Malau-Aduli. 2018. "Enhanced Omega-3 Polyunsaturated Fatty Acid Contents in Muscle and Edible Organs of Australian Prime Lambs Grazing Lucerne and Cocksfoot Pastures" Nutrients 10, no. 12: 1985. https://doi.org/10.3390/nu10121985
APA StyleLe, H. V., Nguyen, Q. V., Nguyen, D. V., Otto, J. R., Malau-Aduli, B. S., Nichols, P. D., & Malau-Aduli, A. E. O. (2018). Enhanced Omega-3 Polyunsaturated Fatty Acid Contents in Muscle and Edible Organs of Australian Prime Lambs Grazing Lucerne and Cocksfoot Pastures. Nutrients, 10(12), 1985. https://doi.org/10.3390/nu10121985





