Health Effects of Resveratrol: Results from Human Intervention Trials
Abstract
1. Introduction
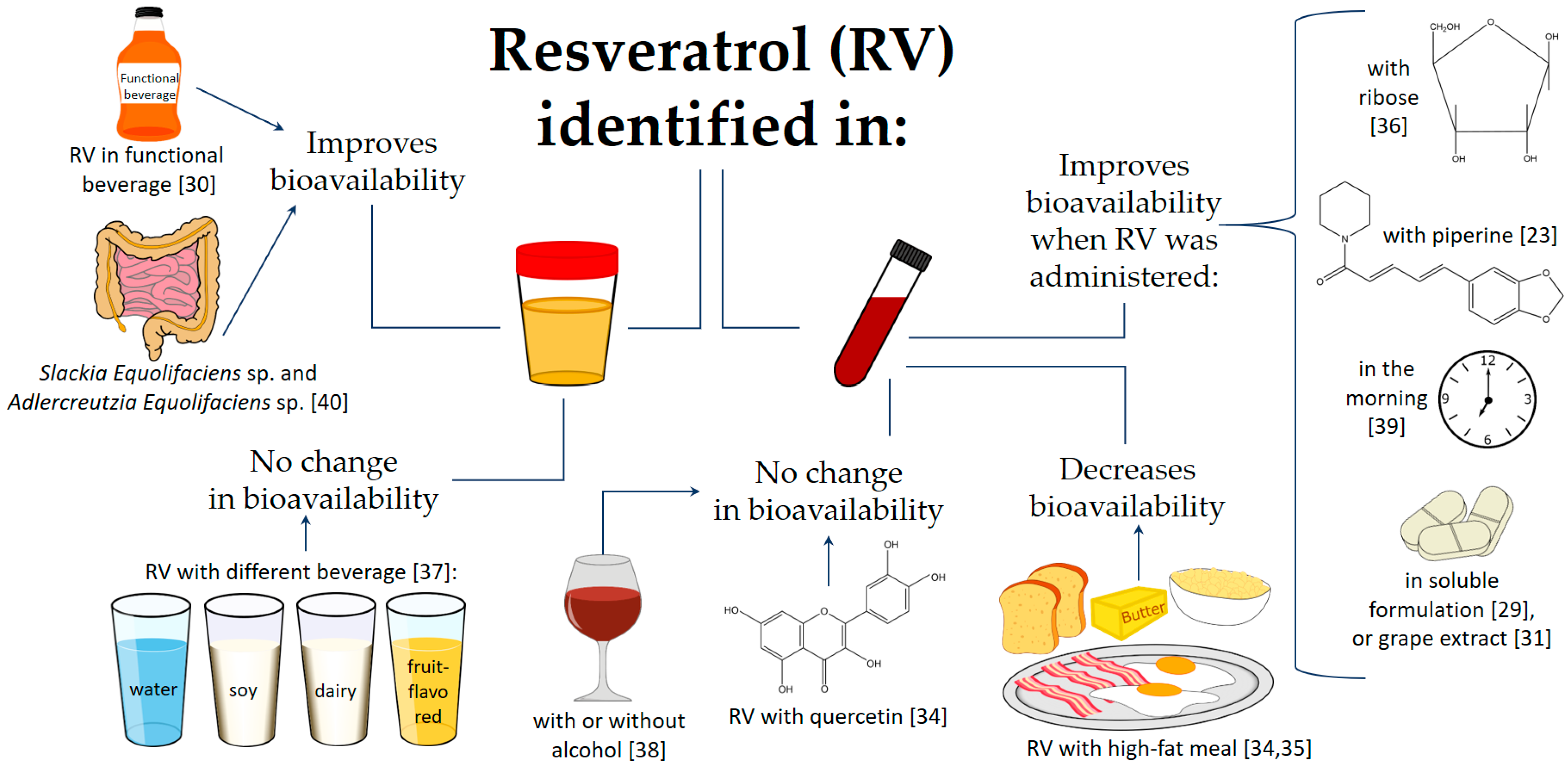
2. Bioavailability and Pharmacokinetics of Resveratrol
2.1. Effect of Pharmaceutical Formulation and Particle Size
2.2. Matrix Effect
2.3. Effects of Other Factors
2.4. Is It Safe to Consume Resveratrol?
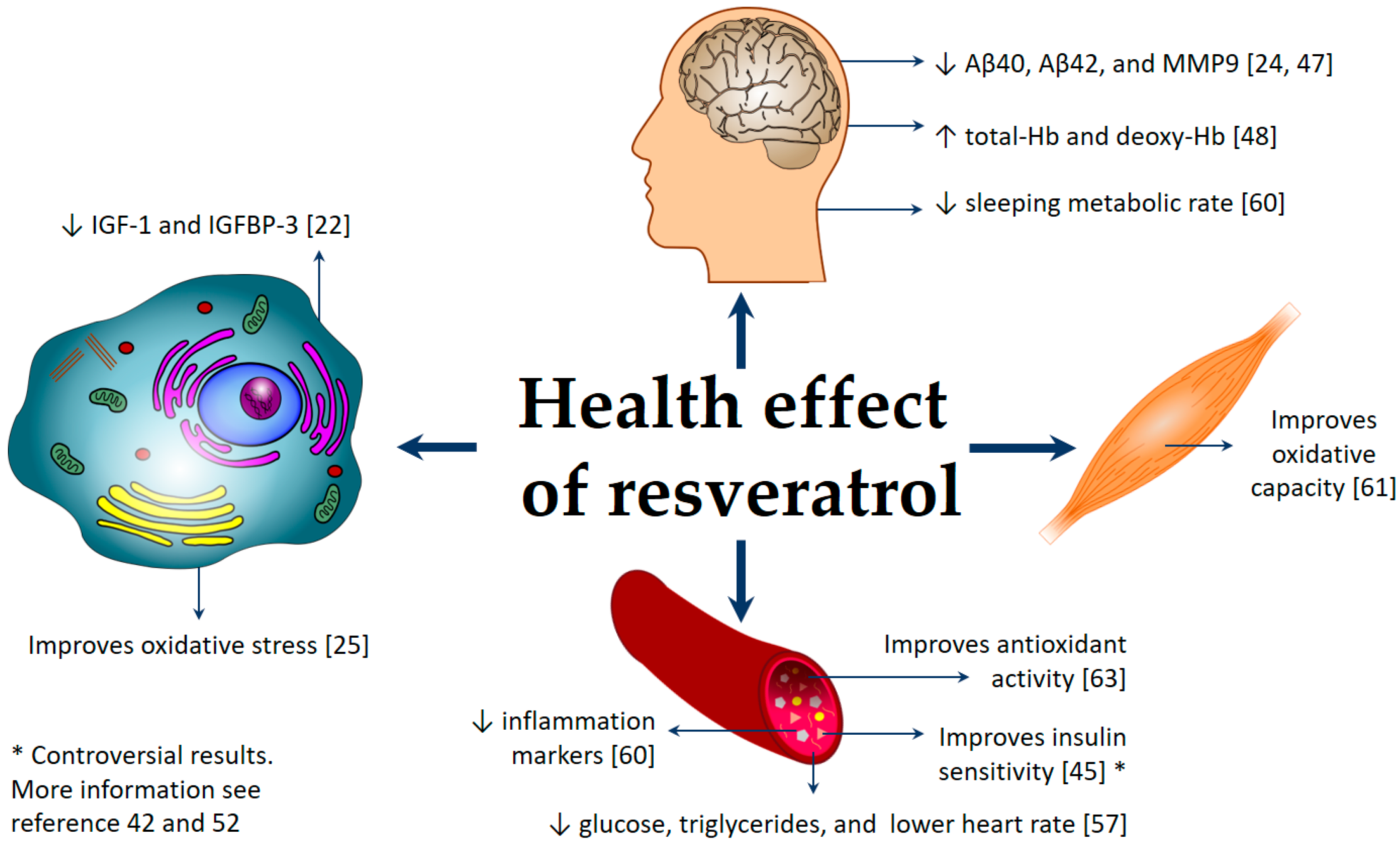
3. Different Health Effects of Resveratrol
3.1. Effects of Resveratrol on Neurological Diseases and Cognitive Performance
3.2. Effects of Resveratrol on Diabetes Mellitus
3.3. Effect of Resveratrol on Cancer
3.4. Effect of Resveratrol on Cardiovascular Diseases
3.5. Effect of Resveratrol on Obesity
3.6. Effect of Resveratrol on Other Health Conditions Associated with Oxidative Stress and Inflammation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1043. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (Poly)phenolics in Human Health: Structures, Bioavailability, and Evidence of Protective Effects Against Chronic Diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Stockley, C.; Teissedre, P.-L.; Boban, M.; Di Lorenzo, C.; Restani, P. Bioavailability of wine-derived phenolic compounds in humans: A review. Food Funct. 2012, 3, 995–1007. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Huélamo, M.; Vallverdú-Queralt, A.; Di Lecce, G.; Valderas-Martínez, P.; Tulipani, S.; Jáuregui, O.; Escribano-Ferrer, E.; Estruch, R.; Illan, M.; Lamuela-Raventós, R.M. Bioavailability of tomato polyphenols is enhanced by processing and fat addition: Evidence from a randomized feeding trial. Mol. Nutr. Food Res. 2016, 60, 1578–1589. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.A.; Harrison, D.E.; Astle, C.M.; Baur, J.A.; Boyd, A.R.; de Cabo, R.; Fernandez, E.; Flurkey, K.; Javors, M.A.; Nelson, J.F.; et al. Rapamycin, but not resveratrol or simvastatin, extends life span of genetically heterogeneous mice. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2011, 66, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Pearson, K.J.; Baur, J.A.; Lewis, K.N.; Peshkin, L.; Price, N.L.; Labinskyy, N.; Swindell, W.R.; Kamara, D.; Minor, R.K.; Perez, E.; et al. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending lifespan. Cell Metab. 2008, 8, 157–168. [Google Scholar] [CrossRef]
- Barger, J.L.; Kayo, T.; Vann, J.M.; Arias, E.B.; Wang, J.; Hacker, T.A.; Wang, Y.; Raederstorff, D.; Morrow, J.D.; Leeuwenburgh, C.; et al. A low dose of dietary resveratrol partially mimics caloric restriction and retards aging parameters in mice. PLoS ONE 2008, 3, e2264. [Google Scholar] [CrossRef]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef]
- Poulsen, M.M.; Vestergaard, P.F.; Clasen, B.F.; Radko, Y.; Christensen, L.P.; Stødkilde-Jørgensen, H.; Møller, N.; Jessen, N.; Pedersen, S.B.; Jørgensen, J.O.L. High-dose resveratrol supplementation in obese men: An investigator-initiated, randomized, placebo-controlled clinical trial of substrate metabolism, insulin sensitivity, and body composition. Diabetes 2013, 62, 1186–1195. [Google Scholar] [CrossRef]
- Ranney, A.; Petro, M.S. Resveratrol protects spatial learning in middle-aged C57BL/6 mice from effects of ethanol. Behav. Pharmacol. 2009, 20. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Dietary polyphenols: Good, bad, or indifferent for your health? Cardiovasc. Res. 2007, 73, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Lagouge, M.; Argmann, C.; Gerhart-hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol Improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1 a. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef] [PubMed]
- Zang, M.; Xu, S.; Maitland-Toolan, K.A.; Zuccollo, A.; Hou, X.; Jiang, B.; Wierzbicki, M.; Verbeuren, T.J.; Cohen, R.A. Polyphenols stimulate AMP-activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic LDL receptor–deficient mice. Diabetes 2006, 55, 2180. [Google Scholar] [CrossRef] [PubMed]
- Bordone, L.; Cohen, D.; Robinson, A.; Motta, M.C.; Van Veen, E.; Czopik, A.; Steele, A.D.; Crowe, H.; Marmor, S.; Luo, J.; et al. SIRT1 transgenic mice show phenotypes resembling calorie restriction. Aging Cell 2007, 6, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Buryanovskyy, L.; Fu, Y.; Boyd, M.; Ma, Y.; Hsieh, T.; Wu, J.M.; Zhang, Z. Crystal structure of quinone reductase 2 in complex with resveratrol. Biochemistry 2004, 43, 11417–11426. [Google Scholar] [CrossRef]
- Soleas, G.J.; Yan, J.; Goldberg, D.M. Ultrasensitive assay for three polyphenols (catechin, quercetin and resveratrol) and their conjugates in biological fluids utilizing gas chromatography with mass selective detection. J. Chromatogr. B Biomed. Sci. Appl. 2001, 757, 161–172. [Google Scholar] [CrossRef]
- Meng, X.; Maliakal, P.; Lu, H.; Lee, M.-J.; Yang, C.S. Urinary and plasma levels of resveratrol and quercetin in humans, mice, and rats after ingestion of pure compounds and grape juice. J. Agric. Food Chem. 2004, 52, 935–942. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Urpí-Sardà, M.; Lamuela-Raventós, R.M.; Estruch, R.; Vázquez-Agell, M.; Serrano-Martínez, M.; Jaeger, W.; Andres-Lacueva, C. Diagnostic performance of urinary resveratrol metabolites as a biomarker of moderate wine consumption. Clin. Chem. 2006, 52, 1373–1380. [Google Scholar] [CrossRef]
- Gambini, J.; Inglés, M.; Olaso, G.; Lopez-Grueso, R.; Bonet-Costa, V.; Gimeno-Mallench, L.; Mas-Bargues, C.; Abdelaziz, K.M.; Gomez-Cabrera, M.C.; Vina, J.; Borras, C. Properties of resveratrol: In vitro and in vivo studies about metabolism, bioavailability, and biological effects in animal models and humans. Oxid. Med. Cell. Longev. 2015, 2015, 1–13. [Google Scholar] [CrossRef]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E.J.; Walle, U.K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Brown, V.A.; Patel, K.R.; Viskaduraki, M.; Crowell, J.A.; Perloff, M.; Booth, T.D.; Vasilinin, G.; Sen, A.; Schinas, A.M.; Piccirilli, G.; et al. Repeat dose study of the cancer chemopreventive agent resveratrol in healthy volunteers: Safety, pharmacokinetics, and effect on the insulin-like growth factor axis. Cancer Res. 2010, 70, 9003–9011. [Google Scholar] [CrossRef] [PubMed]
- Wightman, E.L.; Reay, J.L.; Haskell, C.F.; Williamson, G.; Dew, T.P.; Kennedy, D.O. Effects of resveratrol alone or in combination with piperine on cerebral blood flow parameters and cognitive performance in human subjects: A randomised, double-blind, placebo-controlled, cross-over investigation. Br. J. Nutr. 2014, 112, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.S.; Thomas, R.G.; Craft, S.; van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S. A randomized, double-blind, placebo-controlled trial of resveratrol for Alzheimer disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Yiu, E.M.; Tai, G.; Peverill, R.E.; Lee, K.J.; Croft, K.D.; Mori, T.A.; Scheiber-Mojdehkar, B.; Sturm, B.; Praschberger, M.; Vogel, A.P.; et al. An open-label trial in Friedreich ataxia suggests clinical benefit with high-dose resveratrol, without effect on frataxin levels. J. Neurol. 2015, 262, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.X.; Howe, P.R.C.; Buckley, J.D.; Coates, A.M.; Kunz, I.; Berry, N.M. Acute resveratrol supplementation improves flow-mediated dilatation in overweight/obese individuals with mildly elevated blood pressure. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.X.; Raederstorff, D.; Howe, P.R.C. Acute resveratrol consumption improves neurovascular coupling capacity in adults with type 2 diabetes mellitus. Nutrients 2016, 8, 425. [Google Scholar] [CrossRef]
- Amri, A.; Chaumeil, J.C.; Sfar, S.; Charrueau, C. Administration of resveratrol: What formulation solutions to bioavailability limitations? J. Control. Release 2012, 158, 182–193. [Google Scholar] [CrossRef]
- Amiot, M.J.; Romier, B.; Dao, T.A.; Fanciullino, R.; Ciccolini, J.; Burcelin, R.; Pechere, L.; Emond, C.; Savouret, J.; Seree, E. Optimization of trans -Resveratrol bioavailability for human therapy. Biochimie 2013, 95, 1233–1238. [Google Scholar] [CrossRef]
- Rotches-Ribalta, M.; Urpi-Sarda, M.; Martí, M.M.; Reglero, G.; Andres-Lacueva, C. Resveratrol metabolic fingerprinting after acute and chronic intakes of a functional beverage in humans. Electrophoresis 2014, 35, 1637–1643. [Google Scholar] [CrossRef]
- Rotches-Ribalta, M.; Andres-Lacueva, C.; Estruch, R.; Escribano, E.; Urpi-Sarda, M. Pharmacokinetics of resveratrol metabolic profile in healthy humans after moderate consumption of red wine and grape extract tablets. Pharmacol. Res. 2012, 66, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Hintz, R.; Johnson, K. The effect of particle size distribution on dissolution rate and oral absorption. Int. J. Pharm. 1989, 51, 9–17. [Google Scholar] [CrossRef]
- Howells, L.M.; Berry, D.P.; Elliott, P.J.; Jacobson, E.W.; Hoffmann, E.; Hegarty, B.; Brown, K.; Steward, W.P.; Gescher, A.J. Phase I randomized, double-blind pilot study of micronized resveratrol (SRT501) in patients with hepatic metastases—Safety, pharmacokinetics, and pharmacodynamics. Cancer Prev. Res. 2011, 4, 1419–1425. [Google Scholar] [CrossRef] [PubMed]
- La Porte, C.; Voduc, N.; Zhang, G.; Seguin, I.; Tardiff, D.; Singhal, N.; Cameron, D.W. Steady-State Pharmacokinetics and Tolerability of Trans-Resveratrol 2000 mg Twice Daily with Food, Quercetin and Alcohol (Ethanol) in Healthy Human Subjects. Clin. Pharmacokinet. 2010, 49, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Vaz-da-Silva, M.; Loureiro, A.I.; Falcao, A.; Nunes, T.; Rocha, J.-F.; Fernandes-Lopes, C.; Soares, E.; Wright, L.; Almeida, L.; Soares-da-Silva, P. Effect of food on the pharmacokinetic profile of trans-resveratrol. Int. J. Clin. Pharmacol. Ther. 2008, 46, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, O.L.; Friesenhahn, G.; Javors, M.A.; Smoliga, J.M. Development of a lozenge for oral transmucosal delivery of trans-resveratrol in humans: Proof of concept. PLoS ONE 2014, 9, e90131. [Google Scholar] [CrossRef] [PubMed]
- Draijer, R.; Van Dorsten, F.A.; Zebregs, Y.E.; Hollebrands, B.; Peters, S.; Duchateau, G.S.; Grün, C.H. Impact of proteins on the uptake, distribution, and excretion of phenolics in the human body. Nutrients 2016, 8, 814. [Google Scholar] [CrossRef]
- Rotches-Ribalta, M.; Urpi-Sarda, M.; Llorach, R.; Boto-Ordoñez, M.; Jauregui, O.; Chiva-Blanch, G.; Perez-Garcia, L.; Jaeger, W.; Guillen, M.; Corella, D.; et al. Gut and microbial resveratrol metabolite profiling after moderate long-term consumption of red wine versus dealcoholized red wine in humans by an optimized ultra-high-pressure liquid chromatography tandem mass spectrometry method. J. Chromatogr. A 2012, 1265, 105–113. [Google Scholar] [CrossRef]
- Almeida, L.; Vaz-da-Silva, M.; Falcão, A.; Soares, E.; Costa, R.; Loureiro, A.I.; Fernandes-Lopes, C.; Rocha, J.; Nunes, T.; Wright, L.; Soares-da-Silva, P. Pharmacokinetic and safety profile of trans-resveratrol in a rising multiple-dose study in healthy volunteers. Mol. Nutr. Food Res. 2009, 53, 7–15. [Google Scholar] [CrossRef]
- Bode, L.M.; Bunzel, D.; Huch, M.; Cho, G.; Ruhland, D.; Bunzel, M.; Bub, A.; Franz, C.M.A.P.; Kulling, S.E. In vivo and in vitro metabolism of trans -resveratrol by human gut. Am. J. Clin. Nutr. 2013, 97, 295–309. [Google Scholar] [CrossRef]
- Most, J.; Penders, J.; Lucchesi, M.; Goossens, G.H.; Blaak, E.E. Gut microbiota composition in relation to the metabolic response to 12-week combined polyphenol supplementation in overweight men and women. Eur. J. Clin. Nutr. 2017, 71, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Chachay, V.S.; Macdonald, G.A.; Martin, J.H.; Whitehead, J.P.; O’Moore–Sullivan, T.M.; Lee, P.; Franklin, M.; Klein, K.; Taylor, P.J.; Ferguson, M.; et al. Resveratrol Does Not Benefit Patients With Nonalcoholic Fatty Liver Disease. Clin. Gastroenterol. Hepatol. 2014, 12, 2092–2103.e6. [Google Scholar] [CrossRef] [PubMed]
- Boocock, D.J.; Faust, G.E.S.; Patel, K.R.; Schinas, A.M.; Brown, V.A.; Ducharme, M.P.; Booth, T.D.; Crowell, J.A.; Perloff, M.; Gescher, A.J.; et al. Phase i dose escalation pharmacokinetic study in healthy volunteers of resveratrol, a potential cancer chemopreventive agent. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1246–1253. [Google Scholar] [CrossRef]
- Yoshino, J.; Conte, C.; Fontana, L.; Mittendorfer, B.; Imai, S.I.; Schechtman, K.B.; Gu, C.; Kunz, I.; Fanelli, F.R.; Patterson, B.W.; et al. Resveratrol supplementation does not improve metabolic function in nonobese women with normal glucose tolerance. Cell Metab. 2012, 16, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Crandall, J.P.; Oram, V.; Trandafirescu, G.; Reid, M.; Kishore, P.; Hawkins, M.; Cohen, H.W.; Barzilai, N. Pilot study of resveratrol in older adults with impaired glucose tolerance. J. Gerontol. Ser. A 2012, 67, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Azachi, M.; Yatuv, R.; Katz, A.; Hagay, Y.; Danon, A. A novel red grape cells complex: Health effects and bioavailability of natural resveratrol. Int. J. Food Sci. Nutr. 2014, 65, 848–855. [Google Scholar] [CrossRef]
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R.S. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Wightman, E.L.; Reay, J.L.; Lietz, G.; Okello, E.J.; Wilde, A.; Haskell, C.F. Effects of resveratrol on cerebral blood flow variables and cognitive performance in humans: A double-blind, placebo-controlled, crossover. Am. J. Clin. Nutr. 2010, 91, 1590–1597. [Google Scholar] [CrossRef]
- Bakker, G.C.M.; Van Erk, M.J.; Pellis, L.; Wopereis, S.; Rubingh, C.M.; Cnubben, N.H.P.; Kooistra, T.; Van Ommen, B.; Hendriks, H.F.J. An antiinflammatory dietary mix modulates inflammation and oxidative and metabolic stress in overweight men: A nutrigenomics approach 1–4. Am. J. Clin. Nutr. 2010, 91, 1044–1059. [Google Scholar] [CrossRef] [PubMed]
- Brasnyó, P.; Molnár, G.A.; Mohás, M.; Markó, L.; Laczy, B.; Cseh, J.; Mikolás, E.; Szijártó, I.A.; Mérei, Á.; Halmai, R.; Mészáros, L.G.; et al. Resveratrol improves insulin sensitivity, reduces oxidative stress and activates the Akt pathway in type 2 diabetic patients. Br. J. Nutr. 2011, 106, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Dash, S.; Xiao, C.; Morgantini, C.; Szeto, L.; Lewis, G.F. High-dose resveratrol treatment for 2 weeks inhibits intestinal and hepatic lipoprotein production in overweight/obese men. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 2895–2901. [Google Scholar] [CrossRef] [PubMed]
- Timmers, S.; de Ligt, M.; Phielix, E.; van de Weijer, T.; Hansen, J.; Moonen-Kornips, E.; Schaart, G.; Kunz, I.; Hesselink, M.K.C.; Schrauwen-Hinderling, V.B.; et al. Resveratrol as add-on therapy in subjects with well-controlled type 2 diabetes: A randomized controlled trial. Diabetes Care 2016, 39, 2211–2217. [Google Scholar] [CrossRef] [PubMed]
- Knop, F.K.; Konings, E.; Timmers, S.; Schrauwen, P.; Holst, J.J.; Blaak, E.E. Thirty days of resveratrol supplementation does not affect postprandial incretin hormone responses, but suppresses postprandial glucagon in obese subjects. Diabet. Med. 2013, 30, 1214–1218. [Google Scholar] [CrossRef] [PubMed]
- Grifantini, K. Understanding pathways of calorie restriction: A way to prevent cancer? J. Natl. Cancer Inst. 2008, 100, 619–621. [Google Scholar] [CrossRef] [PubMed]
- Caramori, P.R.; Zago, A.J. Endothelial dysfunction and coronary artery disease. Arq. Bras. Cardiol. 2000, 75, 163–182. [Google Scholar] [CrossRef] [PubMed]
- Grassi, G.; Seravalle, G.; Scopelliti, F.; Dell’Oro, R.; Fattori, L.; Quarti-Trevano, F.; Brambilla, G.; Schiffrin, E.L.; Mancia, G. Structural and functional alterations of subcutaneous small resistance arteries in severe human obesity. Obesity 2010, 18, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Urpi-Sarda, M.; Lamuela-Raventós, R.M.; Martínez-González, M.Á.; Salas-Salvadó, J.; Arós, F.; Fitó, M.; Lapetra, J.; Estruch, R.; Andres-Lacueva, C. High urinary levels of resveratrol metabolites are associated with a reduction in the prevalence of cardiovascular risk factors in high-risk patients. Pharmacol. Res. 2012, 65, 615–620. [Google Scholar] [CrossRef]
- Van Der Made, S.M.; Plat, J.; Mensink, R.P. Resveratrol does not influence metabolic risk markers related to cardiovascular health in overweight and slightly obese subjects: A randomized, placebo-controlled crossover trial. PLoS ONE 2015, 10, 1–13. [Google Scholar] [CrossRef]
- Van der Made, S.M.; Plat, J.; Mensink, R.P. Trans-resveratrol supplementation and endothelial function during the fasting and postprandial phase: A randomized placebo-controlled trial in overweight and slightly obese participants. Nutrients 2017, 9, 596. [Google Scholar] [CrossRef]
- Timmers, S.; Konings, E.; Bilet, L.; Houtkooper, R.H.; van de Weijer, T.; Goossens, G.H.; Hoeks, J.; van der Krieken, S.; Ryu, D.; Kersten, S.; et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011, 14, 612–622. [Google Scholar] [CrossRef]
- Most, J.; Timmers, S.; Warnke, I.; Jocken, J.W.; van Boekschoten, M.; de Groot, P.; Bendik, I.; Schrauwen, P.; Goossens, G.H.; Blaak, E.E. Combined epigallocatechin-3-gallate and resveratrol supplementation for 12 wk increases mitochondrial capacity and fat oxidation, but not insulin sensitivity, in obese humans: A randomized controlled trial. Am. J. Clin. Nutr. 2016, 104, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Gualdoni, G.A.; Kovarik, J.J.; Hofer, J.; Dose, F.; Doberer, D.; Steinberger, P.; Wolzt, M.; Zlabinger, G.J. Resveratrol enhances TNF-α production in human monocytes upon bacterial stimulation. Biochim. Biophys. Acta Gen. Subj. 2014, 1840, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Espinoza, J.L.; Trung, L.Q.; Inaoka, P.T.; Yamada, K.; An, D.T.; Mizuno, S.; Nakao, S.; Takami, A. The Repeated Administration of Resveratrol Has Measurable Effects on Circulating T-Cell Subsets in Humans. Oxid. Med. Cell. Longev. 2017, 2017, 6781872. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Sun, X.; Lin, A. Supplementation with high-dose trans-resveratrol improves ultrafiltration in peritoneal dialysis patients: A prospective, randomized, double-blind study. Ren. Fail. 2016, 38, 214–221. [Google Scholar] [CrossRef] [PubMed]
| Metabolite (form of RV) | Sample | Type of Study | (n) | Dose | Participants’ Health Status | Effect | Ref. |
|---|---|---|---|---|---|---|---|
| RV and six metabolites: two monosulfates, one disulfate, two monoglucuronides, and one glucuronide-sulfate | Urine and feces | Clinical trial | 40 | After a five-day washout, 10 subjects received a 0.5 g dose, which was escalated sequentially to 1, 2.5, and 5 g | Healthy | An intake of up to one dose of 5 g of RV was safe, with minor adverse events in some cases; 77% of urinary excretion of RV and its metabolites occurred within four hours after the lowest dose. RV underwent enterohepatic recirculation. | [43] |
| tRV | Plasma | Randomized, crossover, open-label, and single-dose | 24 | Two treatments with a single dose of 400 mg tRV after a high-fat meal or eight hours without breakfast, separated by a washout of seven days or more. | Healthy | The rate of absorption of tRV was reduced by the presence of a meal | [35] |
| tRV | Plasma | Phase I, randomized, double-blind, placebo-controlled, and single-center | 40 | 25, 50, 100, or 150 mg, administered at four hours intervals (six times/day) for 48 h (13 doses in total) | Healthy | High daily doses of tRV were well tolerated but produced low plasma tRV levels; tRV bioavailability was higher when it was administered in the morning. | [39] |
| tRV | Plasma | Open-label and single-arm | 8 | tRV 2000 mg twice daily for seven days and tRV 2000 mg with quercetin 500 mg twice daily for seven days, with a two-week washout period | Healthy | tRV 2000 g twice daily had adequate exposure and was well tolerated by subjects. Moreover, combined intake with quercetin did not influence its exposure | [34] |
| RV glucuronide and sulfate conjugates, RV glucoside, piceid glucuronides, sulfates, DHR, glucuronide, and sulfate conjugates | Plasma and urine | Randomized and crossover | 10 | After a three-day washout period, three people were chosen for the pilot study in which they consumed 15 grape extract tablets (total RV 4.72 ± 0.07 mg) with 400 mL of water within 10 min. In parallel, seven people were selected randomly to drink 375 mL of red wine (total RV 6.30 ± 0.09 mg) with 400 mL of water consumed within 10 min. | Healthy | Statistically significant differences between grape extract tablets and red wine treatments were obtained for some metabolites, mainly due to the different composition of RV and piceid from both sources. The grape extract tablets delayed RV absorption compared to the red wine treatment. | [31] |
| Free RV and conjugated RV (monosulfate, disulfate, and glucoronide) | Plasma | Clinical trial | 15 | Single dose (40 mg) of tRV in soluble formulation or dry powder | Healthy | Bioavailability was higher with soluble formulation compared to dry powder. | [29] |
| tRV, DHR, 3,4′-dihydroxy-trans-stilbene, and 3,4′-dihydroxybibenzyl (lunularin). | 24 h urine and feces | Controlled intervention | 12 | Following a washout period, all the subjects received a single oral dose of 0.5 mg tRV/kg body weight in the form of a grapevine-shoot supplement (7.7% tRV as well as other stilbene mono- and oligomers [14.6%-ε-viniferin, 3.4% ampelopsin A, 1.8% hopeaphenol, 0.6% trans-piceatannol, 1.6% r-2-viniferin (vitisin A), 2.5% miyabenol C, 2.5% r-viniferin (vitisin B), and 2.4% iso-trans-ε-viniferin]. | Healthy | The human gut microbiota produced pronounced interindividual differences in tRV. Slackia Equolifaciens sp. and Adlercreutzia Equolifaciens sp. were identified as DHR producers, but the bacteria that produce dehydroxylated metabolites were not determined. | [40] |
| tRV | Plasma | Pilot study | 2 | 146 +/− 5.5 mg tRV per 2000 mg of lozenge mass, containing about 46% ribose, 46% (fructose/sucrose mixture), and 8% tRV | Healthy | A mixture of ribose and RV oral transmucosal administration achieved a much higher and quicker RV release compared to the reported traditional free RV capsules. | [36] |
| Free and conjugated RV | Plasma | Randomized and three-way crossover | 15 | Oral doses equivalent to 50 mg or 150 mg of tRV or plant-derived RV (150 mg) on three occasions separated by seven-day washout periods. | Healthy | 150 mg dose of tRV showed higher total and free levels than 50 mg dose | [46] |
| tRV, RV, 3-O-sulphate, RV 4′-O-glucuronide, and RV 3-O-glucuronide | Plasma | Randomized, double-blind, and placebo-controlled | 23 | 250 mg of tRV or 250 mg of tRV with 20 mg of piperine on separate days at least a week apart. | Healthy | Piperine co-supplementation with 250 mg of tRV or 250 mg of tRV; piperine enhanced the absorption of the polyphenol leading to an increase in cerebral blood flow. | [23] |
| tR4G, cR4G, tR3G, cR3G, tR4S, cR4S, tR3S, cR3S, tR34dS, RV-SG, tpiceid, cpiceid, Pic-G, Pic-S1, Pic-S2, DHR, DHR-G1, DHR-G2, DHR-S1, DHR-S2, and DHR-SG | 24 h urine | Randomized, double-blind, placebo-controlled, crossover, and intervention study | 26 | Consumed twice a day (with breakfast and dinner) for 15 days (per each phase) 187 mL of: a control placebo and a functional beverage (4280 g/L of hydroxycinnamic acids, 16 mg/L of anthocyanins, 96 mg/L of flavanols, 83 mg/L of hydroxybenzoic acids, and 5.7 mg/L of stilbenes) | Healthy | The whole profile of the 21 RV metabolites increased after acute and chronic consumption of the functional beverage with respect to the control-placebo beverage and to the baseline. | [30] |
| Phenolic acids including, 3-hydroxyphenylacetic acid, 3-hydroxyhippuric acid, 4-hydroxyhippuric acid, and Hippuric Acid, | 24 h urine | Randomized, placebo-controlled, and crossover | 35 | Six placebo gelatin capsules consumed with 200 mL of water (control) Six capsules containing 800 mg polyphenols (141 mg anthocyanins, 24 mg flavan-3-ols, 16 mg procyanidins, 10 mg phenolic acids, 9 mg flavonols, and 1 mg stilbenes) derived from red wine and grape extracts, or the same dose of polyphenols incorporated into one of the following: 200 mL of water (positive control), 200 g of dairy drink, 200 g of soy drink, 200 g fruit-flavored drink, or protein-free drink. | Healthy | Bioavailability of polyphenols and the excretion of their phenolic metabolites were not significantly affected when polyphenols were consumed in protein-rich soy or dairy drinks. | [37] |
| Total RV | Plasma | Randomized and double-blind | 9 | 5 g/day of SRT501 for approximately 14 days | IV colorectal cancer and hepatic metastasis subjects scheduled to undergo hepatectomy. | RV treatment was well tolerated by the patients. The peak plasma after ingestion of SRT501 was 1.942 ng/m, higher than that of an equivalent dose of non-micronized RV supplementation. | [33] |
| tR4G, cR4G, tR3G, cR3G, tR4S, cR4S, tR3S, cR3S, tR34dS, RV-SG, tpiceid, cpiceid, Pic-G, Pic-S1, Pic-S2, DHR, DHR-G1, DHR-G2, DHR-S1, DHR-S2, and DHR-SG | 24 h urine | Randomized, crossover, and controlled clinical trial | 59 | 15-day run-in period in which they consumed neither grape-derived products nor alcoholic beverages. Afterwards, they consumed every day for four weeks: 272 mL of RW (red wine) with 30 g ethanol/day or 272 mL of DRW (dealcoholized red wine), following the same background diet. | High cardiovascular risk | The whole profile of the 21 RV metabolites increased after RW and DRW consumption, and no differences between them were presented | [38] |
| Metabolite (form of RV) | Sample | Type of Study | (n) | Dose | Participants’ Health Status | Effect | Ref. |
|---|---|---|---|---|---|---|---|
| RV 3-O-glucuronidated-RV, 4-O-glucuronidated-RV, and 3-sulfated-RV | Plasma | Phase II, randomized, double-blind, placebo-controlled, and multi-center | 119 | 500 mg/day RV with 500 mg increments every 13 weeks up to 52 weeks, ending with 1000 mg twice daily | Alzheimer | RV was safe and well tolerated, decreased Aβ40 and MMP9 in CSF, modulated neuroinflammation, and induced adaptive immunity. | [24,47] |
| RV, RV-3-glucuronide, RV-4′-glucuronide, and RV-3-sulfate RV-4′-sulfate | Plasma | Non-randomized, and open-label | 24 | Low-dose RV (1 g daily) or high-dose RV (5 g daily) over a 12-week period | Friedreich ataxia | PBMC frataxin protein levels were not affected. High-dose RV treatment showed a beneficial effect on both oxidative stress and some clinical outcome measures. | [25] |
| Total tRV | Plasma | Randomized, double-blind, and placebo-controlled | 36 | 0, 75, 150, and 300 mg at weekly intervals | T2D | A 75 mg dose of RV correlated with an increase in plasma RV concentration, enhanced the cerebrovascular responsiveness to selected stimuli in T2DM adults. | [27] |
| RV and dehydro RV (aglycones and glucuronide conjugates) | Plasma | Randomized, double-blind, and crossover | 17 | 150 mg/day of resVida (RV) for 30 days | T2D | Intrahepatic lipid content correlated negatively with the plasma RV content. RV plasma levels might be affected by metformin treatment; RV did not improve insulin sensitivity. | [52] |
| TRM: tR3G, cR4G, cR3G, tR4S, tR3S, cR4S, and cR3S | Urine | Randomized, parallel-group, multi-center, and controlled clinical trial | 1000 | Exploratory study of the baseline data of PREDIMED study | T2D or at less three major cardiovascular risk factors | Total urinary RV metabolites were directly associated with lower concentrations of fasting blood glucose and triglycerides, and also with lower heart rate. No significant associations were observed between TRM and total cholesterol, HDL, and LDL concentrations, or blood pressure. Therefore, RV may help to decrease cardiovascular risk. | [57] |
| Total RV | Plasma | Randomized | 10 | 1, 1.5, and 2 g/day RV, taken in divided doses for four weeks | Overweight or obese and insulin resistant. | Fasting glucose was unchanged, but postprandial glucose and three-hour glucose area under the curve decreased significantly. Insulin sensitivity (using the Matsuda index) improved. Fasting lipid profile, CRP, and adiponectin were unchanged. | [45] |
| Total RV | Plasma | Randomized, double-blind, and placebo-controlled | 20 | 3000 mg/day RV for eight weeks | Overweight or obese with non-alcoholic fatty liver disease | RV did not improve insulin sensitivity, plasma lipids, antioxidant activity, and IGF-1, but it increased ALT and AST, liver enzymes that indicate hepatic stress. | [42] |
| Total RV | Plasma | Randomized, double-blind, placebo-controlled, and crossover | 19 | A single dose of RV (30, 90, and 270 mg) administered at one-week intervals over four weeks | Overweight and obese individuals or postmenopausal women with untreated borderline hypertension | Significant linear relationship between RV dose intake and plasma RV concentration. Higher plasma RV concentration was associated with acute flow-mediated dilatation response. | [26] |
| Total conjugated, unconjugated RV, and DHR | Plasma | Randomized, double-blind, and crossover | 11 | 150 mg/day RV for 30 days | Obese | RV supplementation modestly mimicked the beneficial effects of calorie restriction. It reduced sleeping metabolic rate, affected the AMPK–SIRT1–PGC1α axis, decreased hepatic lipid accumulation. and reduced inflammation markers. | [60] |
| Epigallocatechin-3-gallate, RV, and DHR | Plasma | Randomized, double-blind, placebo-controlled, and parallel intervention | 38 | Epigallocatechin-3-gallate + RV 282 and 80 mg/day, respectively for 12 weeks | Overweight and obese | The supplementation improved skeletal muscle oxidative capacity, preserved fasting and postprandial fat oxidation, and prevented an increase in triacylglycerol concentrations. | [61] |
| Total RV and DHR (both free and conjugated) | Plasma | Randomized, placebo-controlled, and crossover | 45 | 150 mg/day RV capsule for four weeks, with a four-week wash-out period | Overweight and slightly obese | RV did not have an effect on cardiovascular risk metabolic markers, endothelial function, or inflammation. | [58,59] |
| Total RV and DHR (free and conjugated forms) | Plasma | Randomized, double-blind, and placebo-controlled | 45 | 75 mg/day (99% pure tRV), for 12 weeks. | Lean and overweight, postmenopausal | RV supplementation did not change plasma substrates and hormones (glucose, plasma lipids, and insulin), adiponectin, leptin, CRP, and IL-6. | [44] |
| Total RV, RV glucuronide, and RV sulfate | Plasma | Randomized, double-blind, placebo-controlled, and crossover | 22 | 250 and 500 mg tRV on separate days. On three visits, the participants received two single-dose capsules. The capsules were combined to give the following treatments: 1) inert placebo, 2) 250 mg tRV, and 3) 500 mg tRV. | Healthy | RV intake increased total-Hb and deoxy-Hb concentration, variables related to cerebral blood flow. | [48] |
| tR4G, tRDS, tR3G, tR4S, and tR3S | Plasma | Pilot study, randomized, open-label, single-dose, and parallel-group | 10 | Single 5 g dose | Healthy | RV increased TNF-α level 24 h after supplementation, by an average of 3.5 pg/mL, compared with placebo. High levels of sulfo- and glucuronide-conjugated RV compounds. | [62] |
| RV | Plasma | Phase I and randomized | 9 | 1000 mg/day RV for 28 days | Healthy | RV was associated with an increase in the number of circulating γδ T cells and regulatory T cells and higher plasma antioxidant activity. | [63] |
| RV-3-O-sulfate, RV-4′-O-glucuronide, RV-3-O-glucuronide, | Plasma | Clinical trial | 40 | 0.5, 1.0, 2.5, or 5.0 g/day RV for 29 days | Healthy | Treatment with 2.5 g RV decreased IGF-1 and IGFBP-3 levels in all volunteers; RV might contribute to cancer chemoprevention. | [22] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Muñoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdú-Queralt, A.; Lamuela-Raventós, R.M. Health Effects of Resveratrol: Results from Human Intervention Trials. Nutrients 2018, 10, 1892. https://doi.org/10.3390/nu10121892
Ramírez-Garza SL, Laveriano-Santos EP, Marhuenda-Muñoz M, Storniolo CE, Tresserra-Rimbau A, Vallverdú-Queralt A, Lamuela-Raventós RM. Health Effects of Resveratrol: Results from Human Intervention Trials. Nutrients. 2018; 10(12):1892. https://doi.org/10.3390/nu10121892
Chicago/Turabian StyleRamírez-Garza, Sonia L., Emily P. Laveriano-Santos, María Marhuenda-Muñoz, Carolina E. Storniolo, Anna Tresserra-Rimbau, Anna Vallverdú-Queralt, and Rosa M. Lamuela-Raventós. 2018. "Health Effects of Resveratrol: Results from Human Intervention Trials" Nutrients 10, no. 12: 1892. https://doi.org/10.3390/nu10121892
APA StyleRamírez-Garza, S. L., Laveriano-Santos, E. P., Marhuenda-Muñoz, M., Storniolo, C. E., Tresserra-Rimbau, A., Vallverdú-Queralt, A., & Lamuela-Raventós, R. M. (2018). Health Effects of Resveratrol: Results from Human Intervention Trials. Nutrients, 10(12), 1892. https://doi.org/10.3390/nu10121892








