Possible Prevention of Diabetes with a Gluten-Free Diet
Abstract
1. Gluten
2. Gluten and T1D
2.1. The Role of Environmental Factors
2.2. GF Diet, Early Evidence and Timing
2.3. GF Diet and the Intestine
2.4. GF Ddiet and the Immune System
2.5. GF Diet, Risk of T1D and CD
2.6. Mucosal Tolerance Induction by Gliadin
2.7. GF Diet and the Beta Cell
3. Gluten and T2D
3.1. The Role of Environmental Factors
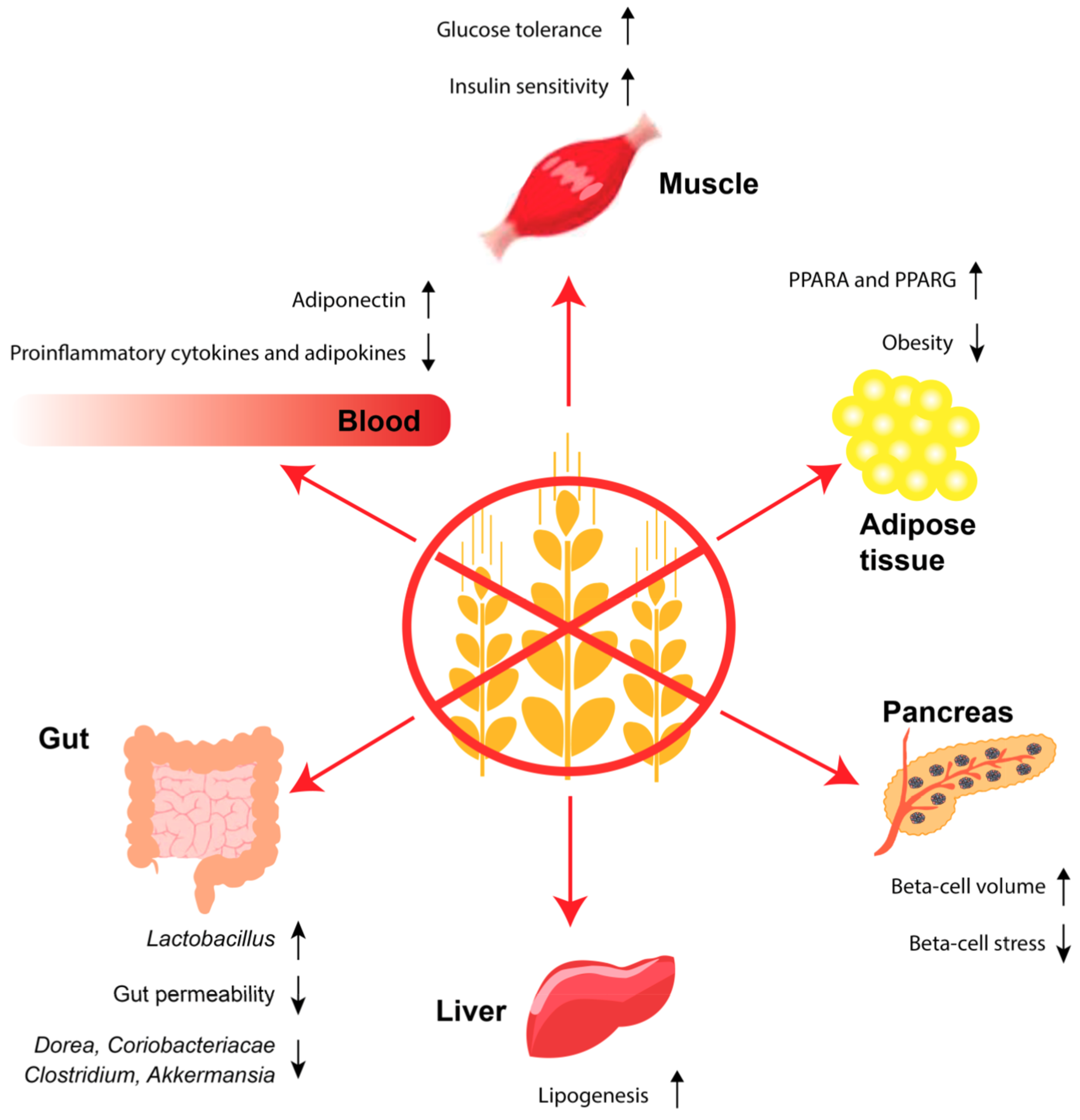
3.2. GF Diet, Leptin Resistance and the Link to Obesity
3.3. GF Diet and the Intestine
3.4. GF Diet, the Beta Cell and Adipose Tissue
3.5. GF Diet, the Immune System, Obesity and T2D
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABCC8 | ATP binding cassette subfamily C member 8 |
| Apoe | Apolipoprotein E |
| AUC | Area under the curve |
| BALB/c | Bagg albino |
| BB | Biobreeding |
| B6 | C57BL/6 |
| CCR7 | CC-chemokine receptor 7 |
| CD | Celiac disease |
| CTL | Cytotoxic T lymphocyte |
| DAISY | Diabetes autoimmunity study in the young |
| DC | Dendritic cell |
| DiViD | Diabetes virus detection study |
| FOXP3 | Forkhead box P3 |
| GALT | Gut-associated lymphoid tissue |
| gdTCR | Gamma delta T cell receptor |
| GF | Gluten-free |
| GF-HF | Gluten-free high-fat |
| GLUT-4 | Glucose transporter 4 |
| GSIS | Glucose-stimulated insulin secretion |
| HDL | High-density lipoprotein |
| HF | High-fat |
| ICAM-1 | Intercellular adhesion molecule 1 |
| IFNA | Interferon alpha |
| IFNG | Interferon gamma |
| IL | Interleukin |
| ILN | Inguinal lymph node |
| KC | Keratinocyte-derived cytokine |
| KCNJ11 | Potassium voltage-gated channel subfamily J member 11 |
| LPS | Lipopolysaccharide |
| MIP-2 | Macrophage inflammatory protein 2 |
| MLN | Mesenteric lymph node |
| MyD88 | Myeloid differentiation primary response 88 |
| NHANES | National health and nutrition examination survey |
| NK | Natural killer |
| NOD | Non-obese diabetic |
| PBMC | Peripheral blood mononuclear cell |
| PLN | Pancreatic lymph node |
| PPARA | Peroxisome proliferator activated receptor alpha |
| PPARG | Peroxisome proliferator activated receptor gamma |
| RORGT | Retinoid orphan receptor gamma t |
| SCFA | Short-chain fatty acid |
| STD | Standard |
| TH | T helper |
| TLR | Toll-like receptor |
| TNFA | Tumor necrosis factor alpha |
| Treg | Regulatory T cell |
| tTG | Tissue transglutaminase |
| T1D | Type 1 diabetes |
| T2D | Type 2 diabetes |
| V:C | Villus-to-crypt |
References
- Vajravelu, M.E.; Keren, R.; Weber, D.R.; Verma, R.; De Leon, D.D.; Denburg, M.R. Incidence and Risk of Celiac Disease after Type 1 Diabetes: A Population-Based Cohort Study Using the Health Improvement Network Database. Pediatr. Diabetes 2018. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A.; Berti, I.; Gerarduzzi, T.; Not, T.; Colletti, R.B.; Drago, S.; Elitsur, Y.; Green, P.H.; Guandalini, S.; Hill, I.D.; et al. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: A large multicenter study. Arch. Intern. Med. 2003, 163, 286–292. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Logan, R.F.; Hill, P.G.; Lloyd, A.; Lewis, S.; Hubbard, R.; Reader, R.; Holmes, G.K.; Khaw, K.T. Seroprevalence, correlates, and characteristics of undetected coeliac disease in England. Gut 2003, 52, 960–965. [Google Scholar] [CrossRef] [PubMed]
- Maki, M.; Mustalahti, K.; Kokkonen, J.; Kulmala, P.; Haapalahti, M.; Karttunen, T.; Ilonen, J.; Laurila, K.; Dahlbom, I.; Hansson, T.; et al. Prevalence of Celiac disease among children in Finland. N. Engl. J. Med. 2003, 348, 2517–2524. [Google Scholar] [CrossRef] [PubMed]
- Leonard, M.M.; Sapone, A.; Catassi, C.; Fasano, A. Celiac Disease and Nonceliac Gluten Sensitivity: A Review. JAMA 2017, 318, 647–656. [Google Scholar] [CrossRef] [PubMed]
- NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4.4 million participants. Lancet 2016, 387, 1513–1530. [Google Scholar] [CrossRef]
- Awika, J.M. Major cereal grains production and use around the world. Adv. Cereal Sci. Implic. Food Process. Health Promot. 2011, 1089, 1–13. [Google Scholar]
- Shewry, P.R.; Halford, N.G. Cereal seed storage proteins: Structures, properties and role in grain utilization. J. Exp. Bot. 2002, 53, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Stern, M.; Ciclitira, P.J.; Van Eckert, R.; Feighery, C.; Janssen, F.W.; Mendez, E.; Mothes, T.; Troncone, R.; Wieser, H. Analysis and clinical effects of gluten in coeliac disease. Eur. J. Gastroenterol. Hepatol. 2001, 13, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Piper, J.L.; Gray, G.M.; Khosla, C. Effect of prolyl endopeptidase on digestive-resistant gliadin peptides in vivo. J. Pharmacol. Exp. Ther. 2004, 311, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Hausch, F.; Shan, L.; Santiago, N.A.; Gray, G.M.; Khosla, C. Intestinal digestive resistance of immunodominant gliadin peptides. Am. J. Physiol. Gastrointest. Liver Physiol. 2002, 283. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A. Zonulin and its regulation of intestinal barrier function: The biological door to inflammation, autoimmunity, and cancer. Physiol. Rev. 2011, 91, 151–175. [Google Scholar] [CrossRef] [PubMed]
- Lammers, K.M.; Lu, R.; Brownley, J.; Lu, B.; Gerard, C.; Thomas, K.; Rallabhandi, P.; Shea-Donohue, T.; Tamiz, A.; Alkan, S.; et al. Gliadin induces an increase in intestinal permeability and zonulin release by binding to the chemokine receptor CXCR3. Gastroenterology 2008, 135, 194–204.e3. [Google Scholar] [CrossRef] [PubMed]
- Lammers, K.M.; Khandelwal, S.; Chaudhry, F.; Kryszak, D.; Puppa, E.L.; Casolaro, V.; Fasano, A. Identification of a novel immunomodulatory gliadin peptide that causes interleukin-8 release in a chemokine receptor CXCR3-dependent manner only in patients with coeliac disease. Immunology 2011, 132, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Maiuri, L.; Troncone, R.; Mayer, M.; Coletta, S.; Picarelli, A.; De Vincenzi, M.; Pavone, V.; Auricchio, S. In vitro activities of A-gliadin-related synthetic peptides: Damaging effect on the atrophic coeliac mucosa and activation of mucosal immune response in the treated coeliac mucosa. Scand. J. Gastroenterol. 1996, 31, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Sollid, L.M.; Qiao, S.W.; Anderson, R.P.; Gianfrani, C.; Koning, F. Nomenclature and listing of celiac disease relevant gluten T-cell epitopes restricted by HLA-DQ molecules. Immunogenetics 2012, 64, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Qiao, S.W.; Bergseng, E.; Molberg, O.; Xia, J.; Fleckenstein, B.; Khosla, C.; Sollid, L.M. Antigen presentation to celiac lesion-derived T cells of a 33-mer gliadin peptide naturally formed by gastrointestinal digestion. J. Immunol. 2004, 173, 1757–1762. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.; Molberg, O.; Parrot, I.; Hausch, F.; Filiz, F.; Gray, G.M.; Sollid, L.M.; Khosla, C. Structural basis for gluten intolerance in celiac sprue. Science 2002, 297, 2275–2279. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.; Qiao, S.W.; Arentz-Hansen, H.; Molberg, O.; Gray, G.M.; Sollid, L.M.; Khosla, C. Identification and analysis of multivalent proteolytically resistant peptides from gluten: Implications for celiac sprue. J. Proteome Res. 2005, 4, 1732–1741. [Google Scholar] [CrossRef] [PubMed]
- Schalk, K.; Lang, C.; Wieser, H.; Koehler, P.; Scherf, K.A. Quantitation of the immunodominant 33-mer peptide from alpha-gliadin in wheat flours by liquid chromatography tandem mass spectrometry. Sci. Rep. 2017, 7, 45092. [Google Scholar] [CrossRef] [PubMed]
- Clayton, D.G. Prediction and interaction in complex disease genetics: Experience in type 1 diabetes. PLOS Genet. 2009, 5, e1000540. [Google Scholar] [CrossRef] [PubMed]
- Pociot, F.; Lernmark, A. Genetic risk factors for type 1 diabetes. Lancet 2016, 387, 2331–2339. [Google Scholar] [CrossRef]
- Group, D.P. Incidence and trends of childhood Type 1 diabetes worldwide 1990–1999. Diabet. Med. 2006, 23, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Patterson, C.C.; Dahlquist, G.G.; Gyurus, E.; Green, A.; Soltesz, G.; Group, E.S. Incidence trends for childhood type 1 diabetes in Europe during 1989–2003 and predicted new cases 2005–20: A multicentre prospective registration study. Lancet 2009, 373, 2027–2033. [Google Scholar] [CrossRef]
- Gorelick, J.; Yarmolinsky, L.; Budovsky, A.; Khalfin, B.; Klein, J.D.; Pinchasov, Y.; Bushuev, M.A.; Rudchenko, T.; Ben-Shabat, S. The Impact of Diet Wheat Source on the Onset of Type 1 Diabetes Mellitus-Lessons Learned from the Non-Obese Diabetic (NOD) Mouse Model. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Bodansky, H.J.; Staines, A.; Stephenson, C.; Haigh, D.; Cartwright, R. Evidence for an environmental effect in the aetiology of insulin dependent diabetes in a transmigratory population. BMJ 1992, 304, 1020–1022. [Google Scholar] [CrossRef] [PubMed]
- Kondrashova, A.; Reunanen, A.; Romanov, A.; Karvonen, A.; Viskari, H.; Vesikari, T.; Ilonen, J.; Knip, M.; Hyoty, H. A six-fold gradient in the incidence of type 1 diabetes at the eastern border of Finland. Ann. Med. 2005, 37, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Kyvik, K.O.; Green, A.; Beck-Nielsen, H. Concordance rates of insulin dependent diabetes mellitus: A population based study of young Danish twins. BMJ 1995, 311, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Kaprio, J.; Tuomilehto, J.; Koskenvuo, M.; Romanov, K.; Reunanen, A.; Eriksson, J.; Stengard, J.; Kesaniemi, Y.A. Concordance for type 1 (insulin-dependent) and type 2 (non-insulin-dependent) diabetes mellitus in a population-based cohort of twins in Finland. Diabetologia 1992, 35, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Knip, M.; Veijola, R.; Virtanen, S.M.; Hyoty, H.; Vaarala, O.; Akerblom, H.K. Environmental triggers and determinants of type 1 diabetes. Diabetes 2005, 54, S125–S136. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, M.A.; Eisenbarth, G.S. Type 1 diabetes: New perspectives on disease pathogenesis and treatment. Lancet 2001, 358, 221–229. [Google Scholar] [CrossRef]
- Stene, L.C.; Oikarinen, S.; Hyoty, H.; Barriga, K.J.; Norris, J.M.; Klingensmith, G.; Hutton, J.C.; Erlich, H.A.; Eisenbarth, G.S.; Rewers, M. Enterovirus infection and progression from islet autoimmunity to type 1 diabetes: The Diabetes and Autoimmunity Study in the Young (DAISY). Diabetes 2010, 59, 3174–3180. [Google Scholar] [CrossRef] [PubMed]
- Oikarinen, S.; Martiskainen, M.; Tauriainen, S.; Huhtala, H.; Ilonen, J.; Veijola, R.; Simell, O.; Knip, M.; Hyoty, H. Enterovirus RNA in blood is linked to the development of type 1 diabetes. Diabetes 2011, 60, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Buschard, K.; Hastrup, N.; Rygaard, J. Virus-induced diabetes mellitus in mice and the thymus-dependent immune system. Diabetologia 1983, 24, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Dotta, F.; Censini, S.; van Halteren, A.G.; Marselli, L.; Masini, M.; Dionisi, S.; Mosca, F.; Boggi, U.; Muda, A.O.; Del Prato, S.; et al. Coxsackie B4 virus infection of beta cells and natural killer cell insulitis in recent-onset type 1 diabetic patients. Proc. Natl. Acad. Sci. USA 2007, 104, 5115–5120. [Google Scholar] [CrossRef] [PubMed]
- Krogvold, L.; Edwin, B.; Buanes, T.; Frisk, G.; Skog, O.; Anagandula, M.; Korsgren, O.; Undlien, D.; Eike, M.C.; Richardson, S.J.; et al. Detection of a low-grade enteroviral infection in the islets of langerhans of living patients newly diagnosed with type 1 diabetes. Diabetes 2015, 64, 1682–1687. [Google Scholar] [CrossRef] [PubMed]
- Hober, D.; Sauter, P. Pathogenesis of type 1 diabetes mellitus: Interplay between enterovirus and host. Nat. Rev. Endocrinol. 2010, 6, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Hoorfar, J.; Buschard, K.; Dagnaes-Hansen, F. Prophylactic nutritional modification of the incidence of diabetes in autoimmune non-obese diabetic (NOD) mice. Br. J. Nutr. 1993, 69, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Scott, F.W. Food-induced type 1 diabetes in the BB rat. Diabetes Metab. Rev. 1996, 12, 341–359. [Google Scholar] [CrossRef]
- Funda, D.P.; Kaas, A.; Bock, T.; Tlaskalova-Hogenova, H.; Buschard, K. Gluten-free diet prevents diabetes in NOD mice. Diabetes Metab. Res. Rev. 1999, 15, 323–327. [Google Scholar] [CrossRef]
- Hansen, A.K.; Ling, F.; Kaas, A.; Funda, D.P.; Farlov, H.; Buschard, K. Diabetes preventive gluten-free diet decreases the number of caecal bacteria in non-obese diabetic mice. Diabetes Metab. Res. Rev. 2006, 22, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Marietta, E.V.; Gomez, A.M.; Yeoman, C.; Tilahun, A.Y.; Clark, C.R.; Luckey, D.H.; Murray, J.A.; White, B.A.; Kudva, Y.C.; Rajagopalan, G. Low incidence of spontaneous type 1 diabetes in non-obese diabetic mice raised on gluten-free diets is associated with changes in the intestinal microbiome. PLoS ONE 2013, 8, e78687. [Google Scholar] [CrossRef] [PubMed]
- Antvorskov, J.C.; Josefsen, K.; Haupt-Jorgensen, M.; Fundova, P.; Funda, D.P.; Buschard, K. Gluten-Free Diet Only during Pregnancy Efficiently Prevents Diabetes in NOD Mouse Offspring. J. Diabetes Res. 2016, 2016, 3047574. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.H.; Krych, L.; Buschard, K.; Metzdorff, S.B.; Nellemann, C.; Hansen, L.H.; Nielsen, D.S.; Frokiaer, H.; Skov, S.; Hansen, A.K. A maternal gluten-free diet reduces inflammation and diabetes incidence in the offspring of NOD mice. Diabetes 2014, 63, 2821–2832. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, A.G.; Schmid, S.; Huber, D.; Hummel, M.; Bonifacio, E. Early infant feeding and risk of developing type 1 diabetes-associated autoantibodies. JAMA 2003, 290, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Norris, J.M.; Barriga, K.; Klingensmith, G.; Hoffman, M.; Eisenbarth, G.S.; Erlich, H.A.; Rewers, M. Timing of initial cereal exposure in infancy and risk of islet autoimmunity. JAMA 2003, 290, 1713–1720. [Google Scholar] [CrossRef] [PubMed]
- Antvorskov, J.C.; Halldorsson, T.I.; Josefsen, K.; Svensson, J.; Granstrom, C.; Roep, B.O.; Olesen, T.H.; Hrolfsdottir, L.; Buschard, K.; Olsen, S.F. Association between maternal gluten intake and type 1 diabetes in offspring: National prospective cohort study in Denmark. BMJ 2018, 362, k3547. [Google Scholar] [CrossRef] [PubMed]
- Lamb, M.M.; Myers, M.A.; Barriga, K.; Zimmet, P.Z.; Rewers, M.; Norris, J.M. Maternal diet during pregnancy and islet autoimmunity in offspring. Pediatr. Diabetes 2008, 9, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, S.M.; Uusitalo, L.; Kenward, M.G.; Nevalainen, J.; Uusitalo, U.; Kronberg-Kippila, C.; Ovaskainen, M.L.; Arkkola, T.; Niinisto, S.; Hakulinen, T.; et al. Maternal food consumption during pregnancy and risk of advanced beta-cell autoimmunity in the offspring. Pediatr. Diabetes 2011, 12, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Sildorf, S.M.; Fredheim, S.; Svensson, J.; Buschard, K. Remission without insulin therapy on gluten-free diet in a 6-year old boy with type 1 diabetes mellitus. BMJ Case Rep. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Svensson, J.; Sildorf, S.M.; Pipper, C.B.; Kyvsgaard, J.N.; Bojstrup, J.; Pociot, F.M.; Mortensen, H.B.; Buschard, K. Potential beneficial effects of a gluten-free diet in newly diagnosed children with type 1 diabetes: A pilot study. Springerplus 2016, 5, 994. [Google Scholar] [CrossRef] [PubMed]
- Pastore, M.R.; Bazzigaluppi, E.; Belloni, C.; Arcovio, C.; Bonifacio, E.; Bosi, E. Six months of gluten-free diet do not influence autoantibody titers, but improve insulin secretion in subjects at high risk for type 1 diabetes. J. Clin. Endocrinol. Metab. 2003, 88, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Mooradian, A.D.; Morley, J.E.; Levine, A.S.; Prigge, W.F.; Gebhard, R.L. Abnormal intestinal permeability to sugars in diabetes mellitus. Diabetologia 1986, 29, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Bosi, E.; Molteni, L.; Radaelli, M.G.; Folini, L.; Fermo, I.; Bazzigaluppi, E.; Piemonti, L.; Pastore, M.R.; Paroni, R. Increased intestinal permeability precedes clinical onset of type 1 diabetes. Diabetologia 2006, 49, 2824–2827. [Google Scholar] [CrossRef] [PubMed]
- Carratu, R.; Secondulfo, M.; de Magistris, L.; Iafusco, D.; Urio, A.; Carbone, M.G.; Pontoni, G.; Carteni, M.; Prisco, F. Altered intestinal permeability to mannitol in diabetes mellitus type I. J. Pediatr. Gastroenterol. Nutr. 1999, 28, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Sapone, A.; De Magistris, L.; Pietzak, M.; Clemente, M.G.; Tripathi, A.; Cucca, F.; Lampis, R.; Kryszak, D.; Carteni, M.; Generoso, M.; et al. Zonulin upregulation is associated with increased gut permeability in subjects with type 1 diabetes and their relatives. Diabetes 2006, 55, 1443–1449. [Google Scholar] [CrossRef] [PubMed]
- Murri, M.; Leiva, I.; Gomez-Zumaquero, J.M.; Tinahones, F.J.; Cardona, F.; Soriguer, F.; Queipo-Ortuno, M.I. Gut microbiota in children with type 1 diabetes differs from that in healthy children: A case-control study. BMC Med. 2013, 11, 46. [Google Scholar] [CrossRef] [PubMed]
- De Goffau, M.C.; Fuentes, S.; Dan den Bogert, B.; Honkanen, H.; De Vos, W.M.; Welling, G.W.; Hyoty, H.; Harmsen, H.J. Aberrant gut microbiota composition at the onset of type 1 diabetes in young children. Diabetologia 2014, 57, 1569–1577. [Google Scholar] [CrossRef] [PubMed]
- Vangay, P.; Ward, T.; Gerber, J.S.; Knights, D. Antibiotics, pediatric dysbiosis, and disease. Cell Host Microbe 2015, 17, 553–564. [Google Scholar] [CrossRef] [PubMed]
- Vaarala, O. Gut microbiota and type 1 diabetes. Rev. Diabet. Stud. 2012, 9, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.H.; Krych, L.; Nielsen, D.S.; Vogensen, F.K.; Hansen, L.H.; Sorensen, S.J.; Buschard, K.; Hansen, A.K. Early life treatment with vancomycin propagates Akkermansia muciniphila and reduces diabetes incidence in the NOD mouse. Diabetologia 2012, 55, 2285–2294. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Lin, S.; Vanhoutte, P.M.; Woo, C.W.; Xu, A. Akkermansia Muciniphila Protects Against Atherosclerosis by Preventing Metabolic Endotoxemia-Induced Inflammation in Apoe−/−Mice. Circulation 2016, 133, 2434–2446. [Google Scholar] [CrossRef] [PubMed]
- Endesfelder, D.; Engel, M.; Davis-Richardson, A.G.; Ardissone, A.N.; Achenbach, P.; Hummel, S.; Winkler, C.; Atkinson, M.; Schatz, D.; Triplett, E.; et al. Towards a functional hypothesis relating anti-islet cell autoimmunity to the dietary impact on microbial communities and butyrate production. Microbiome 2016, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Ohata, A.; Usami, M.; Miyoshi, M. Short-chain fatty acids alter tight junction permeability in intestinal monolayer cells via lipoxygenase activation. Nutrition 2005, 21, 838–847. [Google Scholar] [CrossRef] [PubMed]
- Marino, E.; Richards, J.L.; McLeod, K.H.; Stanley, D.; Yap, Y.A.; Knight, J.; McKenzie, C.; Kranich, J.; Oliveira, A.C.; Rossello, F.J.; et al. Gut microbial metabolites limit the frequency of autoimmune T cells and protect against type 1 diabetes. Nat. Immunol. 2017, 18, 552–562. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T. cells. Nature 2013, 504, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Haupt-Jorgensen, M.; Larsen, J.; Josefsen, K.; Jorgensen, T.Z.; Antvorskov, J.C.; Hansen, A.K.; Buschard, K. Gluten-free diet during pregnancy alleviates signs of diabetes and celiac disease in NOD mouse offspring. Diabetes Metab. Res. Rev. 2018, 34, e2987. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.; Dall, M.; Antvorskov, J.C.; Weile, C.; Engkilde, K.; Josefsen, K.; Buschard, K. Dietary gluten increases natural killer cell cytotoxicity and cytokine secretion. Eur. J. Immunol. 2014, 44, 3056–3067. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.; Weile, C.; Antvorskov, J.C.; Engkilde, K.; Nielsen, S.M.; Josefsen, K.; Buschard, K. Effect of dietary gluten on dendritic cells and innate immune subsets in BALB/c and NOD mice. PLoS ONE 2015, 10, e0118618. [Google Scholar] [CrossRef] [PubMed]
- Alam, C.; Valkonen, S.; Palagani, V.; Jalava, J.; Eerola, E.; Hanninen, A. Inflammatory tendencies and overproduction of IL-17 in the colon of young NOD mice are counteracted with diet change. Diabetes 2010, 59, 2237–2246. [Google Scholar] [CrossRef] [PubMed]
- Chakir, H.; Lefebvre, D.E.; Wang, H.; Caraher, E.; Scott, F.W. Wheat protein-induced proinflammatory T helper 1 bias in mesenteric lymph nodes of young diabetes-prone rats. Diabetologia 2005, 48, 1576–1584. [Google Scholar] [CrossRef] [PubMed]
- Antvorskov, J.C.; Fundova, P.; Buschard, K.; Funda, D.P. Impact of dietary gluten on regulatory T cells and Th17 cells in BALB/c mice. PLoS ONE 2012, 7, e33315. [Google Scholar] [CrossRef] [PubMed]
- Antvorskov, J.C.; Fundova, P.; Buschard, K.; Funda, D.P. Dietary gluten alters the balance of pro-inflammatory and anti-inflammatory cytokines in T cells of BALB/c mice. Immunology 2013, 138, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.E.; Sapone, A.; Fasano, A.; Vogel, S.N. Gliadin stimulation of murine macrophage inflammatory gene expression and intestinal permeability are MyD88-dependent: Role of the innate immune response in Celiac disease. J. Immunol. 2006, 176, 2512–2521. [Google Scholar] [CrossRef] [PubMed]
- Nikulina, M.; Habich, C.; Flohe, S.B.; Scott, F.W.; Kolb, H. Wheat gluten causes dendritic cell maturation and chemokine secretion. J. Immunol. 2004, 173, 1925–1933. [Google Scholar] [CrossRef] [PubMed]
- Ciccocioppo, R.; Rossi, M.; Pesce, I.; Ricci, G.; Millimaggi, D.; Maurano, F.; Corazza, G.R. Effects of gliadin stimulation on bone marrow-derived dendritic cells from HLA-DQ8 transgenic MICE. Dig. Liver Dis. 2008, 40, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Healey, D.; Ozegbe, P.; Arden, S.; Chandler, P.; Hutton, J.; Cooke, A. In vivo activity and in vitro specificity of CD4+ Th1 and Th2 cells derived from the spleens of diabetic NOD mice. J. Clin. Investig. 1995, 95, 2979–2985. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zheng, S.G. Interleukin-22: A likely target for treatment of autoimmune diseases. Autoimmun. Rev. 2014, 13, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Guerrieri, A.; Bartolotta, E.; Coppa, G.V.; Giorgi, P.L. Antigliadin antibodies at onset of diabetes in children. Lancet 1987, 2, 158. [Google Scholar] [CrossRef]
- Troncone, R.; Franzese, A.; Mazzarella, G.; Paparo, F.; Auricchio, R.; Coto, I.; Mayer, M.; Greco, L. Gluten sensitivity in a subset of children with insulin dependent diabetes mellitus. Am. J. Gastroenterol. 2003, 98, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Mojibian, M.; Chakir, H.; Lefebvre, D.E.; Crookshank, J.A.; Sonier, B.; Keely, E.; Scott, F.W. Diabetes-specific HLA-DR-restricted proinflammatory T-cell response to wheat polypeptides in tissue transglutaminase antibody-negative patients with type 1 diabetes. Diabetes 2009, 58, 1789–1796. [Google Scholar] [CrossRef] [PubMed]
- Hamari, S.; Kirveskoski, T.; Glumoff, V.; Kulmala, P.; Simell, O.; Knip, M.; Ilonen, J.; Veijola, R. CD4(+) T-cell proliferation responses to wheat polypeptide stimulation in children at different stages of type 1 diabetes autoimmunity. Pediatr. Diabetes 2015, 16, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Palova-Jelinkova, L.; Rozkova, D.; Pecharova, B.; Bartova, J.; Sediva, A.; Tlaskalova-Hogenova, H.; Spisek, R.; Tuckova, L. Gliadin fragments induce phenotypic and functional maturation of human dendritic cells. J. Immunol. 2005, 175, 7038–7045. [Google Scholar] [CrossRef] [PubMed]
- Molberg, O.; McAdam, S.; Lundin, K.E.; Kristiansen, C.; Arentz-Hansen, H.; Kett, K.; Sollid, L.M. T cells from celiac disease lesions recognize gliadin epitopes deamidated in situ by endogenous tissue transglutaminase. Eur. J. Immunol. 2001, 31, 1317–1323. [Google Scholar] [CrossRef]
- Molberg, O.; McAdam, S.N.; Korner, R.; Quarsten, H.; Kristiansen, C.; Madsen, L.; Fugger, L.; Scott, H.; Noren, O.; Roepstorff, P.; et al. Tissue transglutaminase selectively modifies gliadin peptides that are recognized by gut-derived T cells in celiac disease. Nat. Med. 1998, 4, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Serena, G.; Camhi, S.; Sturgeon, C.; Yan, S.; Fasano, A. The Role of Gluten in Celiac Disease and Type 1 Diabetes. Nutrients 2015, 7, 7143–7162. [Google Scholar] [CrossRef] [PubMed]
- Cosnes, J.; Cellier, C.; Viola, S.; Colombel, J.F.; Michaud, L.; Sarles, J.; Hugot, J.P.; Ginies, J.L.; Dabadie, A.; Mouterde, O.; et al. Incidence of autoimmune diseases in celiac disease: Protective effect of the gluten-free diet. Clin. Gastroenterol. Hepatol. 2008, 6, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Barera, G.; Bonfanti, R.; Viscardi, M.; Bazzigaluppi, E.; Calori, G.; Meschi, F.; Bianchi, C.; Chiumello, G. Occurrence of celiac disease after onset of type 1 diabetes: A 6-year prospective longitudinal study. Pediatrics 2002, 109, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Hansen, D.; Bennedbaek, F.N.; Hansen, L.K.; Hoier-Madsen, M.; Hegedu, L.S.; Jacobsen, B.B.; Husby, S. High prevalence of coeliac disease in Danish children with type I. diabetes mellitus. Acta. Paediatr. 2001, 90, 1238–1243. [Google Scholar] [CrossRef] [PubMed]
- Aktay, A.N.; Lee, P.C.; Kumar, V.; Parton, E.; Wyatt, D.T.; Werlin, S.L. The prevalence and clinical characteristics of celiac disease in juvenile diabetes in Wisconsin. J. Pediatr. Gastroenterol. Nutr. 2001, 33, 462–465. [Google Scholar] [CrossRef] [PubMed]
- Not, T.; Tommasini, A.; Tonini, G.; Buratti, E.; Pocecco, M.; Tortul, C.; Valussi, M.; Crichiutti, G.; Berti, I.; Trevisiol, C.; et al. Undiagnosed coeliac disease and risk of autoimmune disorders in subjects with Type, I. diabetes mellitus. Diabetologia 2001, 44, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, A.K.; Axelsson, I.E.; Borulf, S.K.; Bredberg, A.C.; Lindberg, B.A.; Sjoberg, K.G.; Ivarsson, S.A. Prevalence of IgA-antiendomysium and IgA-antigliadin autoantibodies at diagnosis of insulin-dependent diabetes mellitus in Swedish children and adolescents. Pediatrics 1999, 103, 1248–1252. [Google Scholar] [CrossRef] [PubMed]
- Sblattero, D.; Maurano, F.; Mazzarella, G.; Rossi, M.; Auricchio, S.; Florian, F.; Ziberna, F.; Tommasini, A.; Not, T.; Ventura, A.; et al. Characterization of the anti-tissue transglutaminase antibody response in nonobese diabetic mice. J. Immunol. 2005, 174, 5830–5836. [Google Scholar] [CrossRef] [PubMed]
- Burbelo, P.D.; Lebovitz, E.E.; Bren, K.E.; Bayat, A.; Paviol, S.; Wenzlau, J.M.; Barriga, K.J.; Rewers, M.; Harlan, D.M.; Iadarola, M.J. Extrapancreatic autoantibody profiles in type I. diabetes. PLoS ONE 2012, 7, e45216. [Google Scholar] [CrossRef] [PubMed]
- Maurano, F.; Mazzarella, G.; Luongo, D.; Stefanile, R.; D’Arienzo, R.; Rossi, M.; Auricchio, S.; Troncone, R. Small intestinal enteropathy in non-obese diabetic mice fed a diet containing wheat. Diabetologia 2005, 48, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Auricchio, R.; Paparo, F.; Maglio, M.; Franzese, A.; Lombardi, F.; Valerio, G.; Nardone, G.; Percopo, S.; Greco, L.; Troncone, R. In vitro-deranged intestinal immune response to gliadin in type 1 diabetes. Diabetes 2004, 53, 1680–1683. [Google Scholar] [CrossRef] [PubMed]
- Scott, F.W.; Rowsell, P.; Wang, G.S.; Burghardt, K.; Kolb, H.; Flohe, S. Oral exposure to diabetes-promoting food or immunomodulators in neonates alters gut cytokines and diabetes. Diabetes 2002, 51, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Funda, D.P.; Kaas, A.; Tlaskalova-Hogenova, H.; Buschard, K. Gluten-free but also gluten-enriched (gluten+) diet prevent diabetes in NOD mice; the gluten enigma in type 1 diabetes. Diabetes Metab. Res. Rev. 2008, 24, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Labeta, M.O.; Durieux, J.J.; Spagnoli, G.; Fernandez, N.; Wijdenes, J.; Herrmann, R. CD14 and tolerance to lipopolysaccharide: Biochemical and functional analysis. Immunology 1993, 80, 415–423. [Google Scholar] [PubMed]
- Funda, D.P.; Fundova, P.; Hansen, A.K.; Buschard, K. Prevention or early cure of type 1 diabetes by intranasal administration of gliadin in NOD mice. PLoS ONE 2014, 9, e94530. [Google Scholar] [CrossRef] [PubMed]
- Locke, N.R.; Stankovic, S.; Funda, D.P.; Harrison, L.C. TCR gamma delta intraepithelial lymphocytes are required for self-tolerance. J. Immunol. 2006, 176, 6553–6559. [Google Scholar] [CrossRef] [PubMed]
- Lindley, S.; Dayan, C.M.; Bishop, A.; Roep, B.O.; Peakman, M.; Tree, T.I. Defective suppressor function in CD4(+) CD25(+) T-cells from patients with type 1 diabetes. Diabetes 2005, 54, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Haupt-Jorgensen, M.; Nielsen, E.; Engkilde, K.; Lerche, M.; Larsen, J.; Buschard, K. Occupation with grain crops is associated with lower type 1 diabetes incidence: Registry-based case-control study. PLoS ONE 2017, 12, e0181143. [Google Scholar] [CrossRef] [PubMed]
- Bruun, S.W.; Josefsen, K.; Tanassi, J.T.; Marek, A.; Pedersen, M.H.; Sidenius, U.; Haupt-Jorgensen, M.; Antvorskov, J.C.; Larsen, J.; Heegaard, N.H.; et al. Large Gliadin Peptides Detected in the Pancreas of NOD and Healthy Mice following Oral Administration. J. Diabetes Res. 2016, 2016, 2424306. [Google Scholar] [CrossRef] [PubMed]
- Freire, R.H.; Fernandes, L.R.; Silva, R.B.; Coelho, B.S.; de Araujo, L.P.; Ribeiro, L.S.; Andrade, J.M.; Lima, P.M.; Araujo, R.S.; Santos, S.H.; et al. Wheat gluten intake increases weight gain and adiposity associated with reduced thermogenesis and energy expenditure in an animal model of obesity. Int. J. Obes. 2016, 40, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Chirdo, F.G.; Rumbo, M.; Anon, M.C.; Fossati, C.A. Presence of high levels of non-degraded gliadin in breast milk from healthy mothers. Scand. J. Gastroenterol. 1998, 33, 1186–1192. [Google Scholar] [PubMed]
- In’t Veld, P.; Marichal, M. Microscopic anatomy of the human islet of Langerhans. Adv. Exp. Med. Biol. 2010, 654, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Dall, M.; Calloe, K.; Haupt-Jorgensen, M.; Larsen, J.; Schmitt, N.; Josefsen, K.; Buschard, K. Gliadin fragments and a specific gliadin 33-mer peptide close KATP channels and induce insulin secretion in INS-1E cells and rat islets of langerhans. PLoS ONE 2013, 8, e66474. [Google Scholar] [CrossRef] [PubMed]
- Aaen, K.; Rygaard, J.; Josefsen, K.; Petersen, H.; Brogren, C.H.; Horn, T.; Buschard, K. Dependence of antigen expression on functional state of beta-cells. Diabetes 1990, 39, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Ientile, R.; Caccamo, D.; Griffin, M. Tissue transglutaminase and the stress response. Amino Acids 2007, 33, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Van Lummel, M.; Duinkerken, G.; van Veelen, P.A.; De Ru, A.; Cordfunke, R.; Zaldumbide, A.; Gomez-Tourino, I.; Arif, S.; Peakman, M.; Drijfhout, J.W.; et al. Posttranslational modification of HLA-DQ binding islet autoantigens in type 1 diabetes. Diabetes 2014, 63, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.E.; Sicree, R.A.; Zimmet, P.Z. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res. Clin. Pract. 2010, 87, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Leahy, J.L. Natural history of beta-cell dysfunction in NIDDM. Diabetes Care 1990, 13, 992–1010. [Google Scholar] [CrossRef] [PubMed]
- Perley, M.; Kipnis, D.M. Plasma insulin responses to glucose and tolbutamide of normal weight and obese diabetic and nondiabetic subjects. Diabetes 1966, 15, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Kloppel, G.; Lohr, M.; Habich, K.; Oberholzer, M.; Heitz, P.U. Islet pathology and the pathogenesis of type 1 and type 2 diabetes mellitus revisited. Surv. Synth. Pathol. Res. 1985, 4, 110–125. [Google Scholar] [PubMed]
- Kahn, S.E. Clinical review 135: The importance of beta-cell failure in the development and progression of type 2 diabetes. J. Clin. Endocrinol. Metab. 2001, 86, 4047–4058. [Google Scholar] [CrossRef] [PubMed]
- Florez, J.C.; Hirschhorn, J.; Altshuler, D. The inherited basis of diabetes mellitus: Implications for the genetic analysis of complex traits. Annu. Rev. Genomics Hum. Genet. 2003, 4, 257–291. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, P.; Kyvik, K.O.; Vaag, A.; Beck-Nielsen, H. Heritability of type II (non-insulin-dependent) diabetes mellitus and abnormal glucose tolerance--a population-based twin study. Diabetologia 1999, 42, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Diabetes mellitus in twins: A cooperative study in Japan. Committee on Diabetic Twins, Japan Diabetes Society. Diabetes Res. Clin. Pract. 1988, 5, 271–280. [CrossRef]
- Romao, I.; Roth, J. Genetic and environmental interactions in obesity and type 2 diabetes. J. Am. Diet. Assoc. 2008, 108, S24–S28. [Google Scholar] [CrossRef] [PubMed]
- Alberti, G.; Zimmet, P.; Shaw, J.; Bloomgarden, Z.; Kaufman, F.; Silink, M. Type 2 diabetes in the young: The evolving epidemic: The international diabetes federation consensus workshop. Diabetes Care 2004, 27, 1798–1811. [Google Scholar] [CrossRef] [PubMed]
- Neel, J.V. Diabetes mellitus: A ‘thrifty’ genotype rendered detrimental by ’progress’? Am. J. Hum. Genet. 1962, 14, 353–362. [Google Scholar] [PubMed]
- Tinggaard, J.; Wohlfahrt-Veje, C.; Husby, S.; Christiansen, L.; Skakkebaek, N.E.; Jensen, T.K.; Grandjean, P.; Main, K.M.; Andersen, H.R. Prenatal pesticide exposure and PON1 genotype associated with adolescent body fat distribution evaluated by dual X-ray absorptiometry (DXA). Andrology 2016, 4, 735–744. [Google Scholar] [CrossRef] [PubMed]
- Hales, C.N.; Barker, D.J. Type 2 (non-insulin-dependent) diabetes mellitus: The thrifty phenotype hypothesis. Diabetologia 1992, 35, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, T.; Memon, A.A.; Sundquist, K.; Sundquist, J.; Olsson, S.; Nalla, A.; Bauer, M.; Linse, S. Digested wheat gluten inhibits binding between leptin and its receptor. BMC Biochem. 2015, 16, 3. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Guo, J.; Su, Z. Advances in understanding the interrelations between leptin resistance and obesity. Physiol. Behav. 2014, 130, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, T.; Olsson, S.; Ahren, B.; Bog-Hansen, T.C.; Dole, A.; Lindeberg, S. Agrarian diet and diseases of affluence--do evolutionary novel dietary lectins cause leptin resistance? BMC Endocr. Disord. 2005, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, T.; Ahren, B.; Pacini, G.; Sundler, F.; Wierup, N.; Steen, S.; Sjoberg, T.; Ugander, M.; Frostegard, J.; Goransson, L.; et al. A Paleolithic diet confers higher insulin sensitivity, lower C-reactive protein and lower blood pressure than a cereal-based diet in domestic pigs. Nutr. Metab. 2006, 3, 39. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Microbiota and diabetes: An evolving relationship. Gut 2014, 63, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, F.H.; Tremaroli, V.; Nookaew, I.; Bergstrom, G.; Behre, C.J.; Fagerberg, B.; Nielsen, J.; Backhed, F. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature 2013, 498, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Fan, C.; Li, P.; Lu, Y.; Chang, X.; Qi, K. Short Chain Fatty Acids Prevent High-fat-diet-induced Obesity in Mice by Regulating G Protein-coupled Receptors and Gut Microbiota. Sci. Rep. 2016, 6, 37589. [Google Scholar] [CrossRef] [PubMed]
- Horton, F.; Wright, J.; Smith, L.; Hinton, P.J.; Robertson, M.D. Increased intestinal permeability to oral chromium (51 Cr) -EDTA in human Type 2 diabetes. Diabet. Med. 2014, 31, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.J.; Zhang, P.; Bowden, D.W.; Devereaux, B.; Davoren, P.M.; Cripps, A.W.; West, N.P. Increased intestinal permeability as a risk factor for type 2 diabetes. Diabetes Metab. 2017, 43, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Andersen, D.; Roager, H.M.; Bahl, M.I.; Hansen, C.H.; Danneskiold-Samsoe, N.B.; Kristiansen, K.; Radulescu, I.D.; Sina, C.; Frandsen, H.L.; et al. Effects of Gliadin consumption on the Intestinal Microbiota and Metabolic Homeostasis in Mice Fed a High-fat Diet. Sci. Rep. 2017, 7, 44613. [Google Scholar] [CrossRef] [PubMed]
- Rajilic-Stojanovic, M.; De Vos, W.M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 2014, 38, 996–1047. [Google Scholar] [CrossRef] [PubMed]
- Clavel, T.; Desmarchelier, C.; Haller, D.; Gerard, P.; Rohn, S.; Lepage, P.; Daniel, H. Intestinal microbiota in metabolic diseases: From bacterial community structure and functions to species of pathophysiological relevance. Gut Microbes 2014, 5, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Saulnier, D.M.; Riehle, K.; Mistretta, T.A.; Diaz, M.A.; Mandal, D.; Raza, S.; Weidler, E.M.; Qin, X.; Coarfa, C.; Milosavljevic, A.; et al. Gastrointestinal microbiome signatures of pediatric patients with irritable bowel syndrome. Gastroenterology 2011, 141, 1782–1791. [Google Scholar] [CrossRef] [PubMed]
- Rajilic-Stojanovic, M.; Biagi, E.; Heilig, H.G.; Kajander, K.; Kekkonen, R.A.; Tims, S.; de Vos, W.M. Global and deep molecular analysis of microbiota signatures in fecal samples from patients with irritable bowel syndrome. Gastroenterology 2011, 141, 1792–1801. [Google Scholar] [CrossRef] [PubMed]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shen, D.; Fang, Z.; Jie, Z.; Qiu, X.; Zhang, C.; Chen, Y.; Ji, L. Human gut microbiota changes reveal the progression of glucose intolerance. PLoS ONE 2013, 8, e71108. [Google Scholar] [CrossRef] [PubMed]
- Amar, J.; Chabo, C.; Waget, A.; Klopp, P.; Vachoux, C.; Bermudez-Humaran, L.G.; Smirnova, N.; Berge, M.; Sulpice, T.; Lahtinen, S.; et al. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes: Molecular mechanisms and probiotic treatment. EMBO Mol. Med. 2011, 3, 559–572. [Google Scholar] [CrossRef] [PubMed]
- Fagerberg, L.; Hallstrom, B.M.; Oksvold, P.; Kampf, C.; Djureinovic, D.; Odeberg, J.; Habuka, M.; Tahmasebpoor, S.; Danielsson, A.; Edlund, K.; et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol. Cell. Proteom. 2014, 13, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Kokoeva, M.V.; Inouye, K.; Tzameli, I.; Yin, H.; Flier, J.S. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Investig. 2006, 116, 3015–3025. [Google Scholar] [CrossRef] [PubMed]
- Ladefoged, M.; Buschard, K.; Hansen, A.M. Increased expression of toll-like receptor 4 and inflammatory cytokines, interleukin-6 in particular, in islets from a mouse model of obesity and type 2 diabetes. APMIS 2013, 121, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Palova-Jelinkova, L.; Danova, K.; Drasarova, H.; Dvorak, M.; Funda, D.P.; Fundova, P.; Kotrbova-Kozak, A.; Cerna, M.; Kamanova, J.; Martin, S.F.; et al. Pepsin digest of wheat gliadin fraction increases production of IL-1beta via TLR4/MyD88/TRIF/MAPK/NF-kappaB signaling pathway and an NLRP3 inflammasome activation. PLoS ONE 2013, 8, e62426. [Google Scholar] [CrossRef] [PubMed]
- Mokady, S.; Einav, P. Effect of dietary wheat gluten in lipid metabolism in growing rats. Nutr. Metab. 1978, 22, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Soares, F.L.; de Oliveira Matoso, R.; Teixeira, L.G.; Menezes, Z.; Pereira, S.S.; Alves, A.C.; Batista, N.V.; De Faria, A.M.; Cara, D.C.; Ferreira, A.V.; et al. Gluten-free diet reduces adiposity, inflammation and insulin resistance associated with the induction of PPAR-alpha and PPAR-gamma expression. J. Nutr. Biochem. 2013, 24, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Haupt-Jorgensen, M.; Buschard, K.; Hansen, A.K.; Josefsen, K.; Antvorskov, J.C. Gluten-free diet increases beta-cell volume and improves glucose tolerance in an animal model of type 2 diabetes. Diabetes Metab. Res. Rev. 2016, 32, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Rune, I.; Rolin, B.; Larsen, C.; Nielsen, D.S.; Kanter, J.E.; Bornfeldt, K.E.; Lykkesfeldt, J.; Buschard, K.; Kirk, R.K.; Christoffersen, B.; et al. Modulating the Gut Microbiota Improves Glucose Tolerance, Lipoprotein Profile and Atherosclerotic Plaque Development in ApoE-Deficient Mice. PLoS ONE 2016, 11, e0146439. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, L.S.; Hartvigsen, M.L.; Brader, L.J.; Astrup, A.; Schrezenmeir, J.; Holst, J.J.; Thomsen, C.; Hermansen, K. Differential effects of protein quality on postprandial lipemia in response to a fat-rich meal in type 2 diabetes: Comparison of whey, casein, gluten, and cod protein. Am. J. Clin. Nutr. 2009, 90, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Demyen, M.F.; Mathew, J.; Kothari, N.; Feurdean, M.; Ahlawat, S.K. Obesity, Metabolic Syndrome, and Cardiovascular Risk in Gluten-Free Followers Without Celiac Disease in the United States: Results from the National Health and Nutrition Examination Survey 2009–2014. Dig. Dis. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Mari-Bauset, S.; Llopis-Gonzalez, A.; Zazpe, I.; Mari-Sanchis, A.; Suarez-Varela, M.M. Nutritional Impact of a Gluten-Free Casein-Free Diet in Children with Autism Spectrum Disorder. J. Autism. Dev. Disord. 2016, 46, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Zong, G.; Lebwohl, B.; Hu, F.B.; Sampson, L.; Dougherty, L.W.; Willett, W.C.; Chan, A.T.; Sun, Q. Gluten intake and risk of type 2 diabetes in three large prospective cohort studies of US men and women. Diabetologia 2018, 61, 2164–2173. [Google Scholar] [CrossRef] [PubMed]
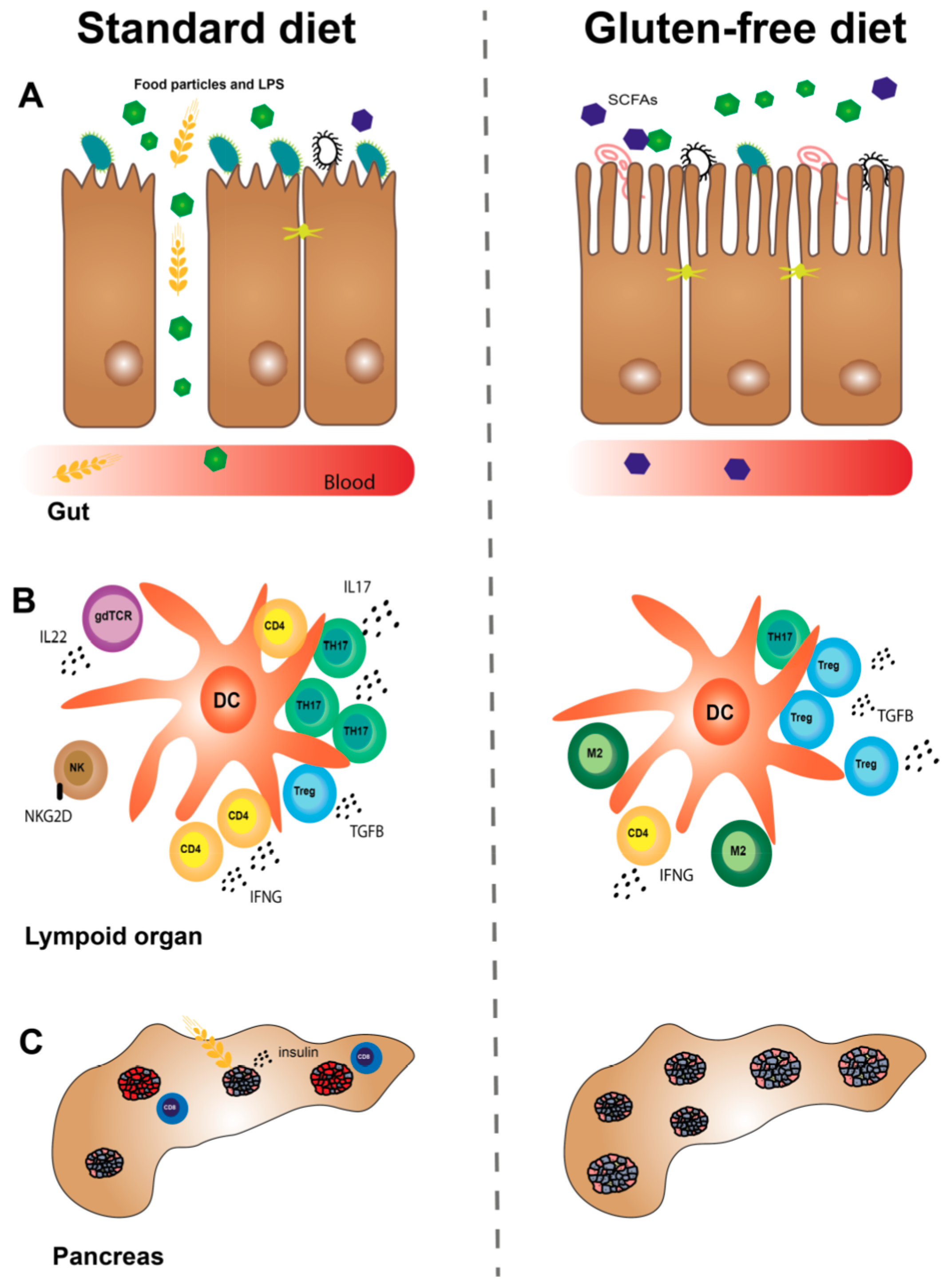
| Immunological Effects of a GF versus a STD Diet in Utero in NOD Mice | References |
| ↑M2 macrophage gene expression in intestine. | [44] |
| ↓DC (CD11b+CD11c+) numbers in PLN, MLN and spleen. | [44] |
| ↓TH1 cell (IFNG+CD4+) numbers in spleen. | [67] |
| ↓TH17 (RORGT) gene expression in colon. | [43] |
| ↓gdTCR cell (IL22+gdTCR+) numbers in spleen. | [67] |
| ↑Treg cell (FOXP3+CD4+) numbers in PLN. | [44] |
| ↑T cell (α4β7+CD4+/CD8+) numbers in PLN. | [44] |
| ↓proinflammatory cytokine gene expression in intestine. | [44] |
| ↓T cells (gdTCR+, CD4+, CD8+ and FOXP3+) inflammatory cytokine profile in spleen, PLN, MLN and ILN. | [67] |
| Immunological Effects of a GF versus a STD Diet Postnatally in Animal Models of T1D | References |
| ↓NK cell (activated) (NKG2D+NKp46+/DX5+) numbers in PLN (BALB/c mice) and spleen (NOD mice). | [68,69] |
| ↓DC (CD11b+CD11c+) numbers in PLN and MLN (BALB/c mice). | [69] |
| ↓DC (CD11b+CD11c+) numbers in colon (NOD mice). | [70] |
| ↓DC (activated) (CD40+/CCR7+/MHCII+ on CD11c+) numbers in different lymphoid organs (BALB/c mice). | [69] |
| ↑DC (tolerogenic) (CD103+CD11b+) numbers in PLN (BALB/c mice). | [69] |
| ↓TH cell (CD4+) numbers in colon (NOD mice). | [70] |
| ↓TH1 cell (IFNG+CD4+) numbers in MLN (BB rats). | [71] |
| ↓TH17 cell (IL17+CD4+) numbers in PLN (BALB/c mice). | [72] |
| ↓TH17 cell (IL17+CD4+) numbers in colon (NOD mice). | [70] |
| ↓T cell (CD3+) proinflammatory profile (BALB/c mice). | [73] |
| ↓CTL (activated) (NKG2D+CD8+) numbers in PLN (NOD mice). | [69] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haupt-Jorgensen, M.; Holm, L.J.; Josefsen, K.; Buschard, K. Possible Prevention of Diabetes with a Gluten-Free Diet. Nutrients 2018, 10, 1746. https://doi.org/10.3390/nu10111746
Haupt-Jorgensen M, Holm LJ, Josefsen K, Buschard K. Possible Prevention of Diabetes with a Gluten-Free Diet. Nutrients. 2018; 10(11):1746. https://doi.org/10.3390/nu10111746
Chicago/Turabian StyleHaupt-Jorgensen, Martin, Laurits J. Holm, Knud Josefsen, and Karsten Buschard. 2018. "Possible Prevention of Diabetes with a Gluten-Free Diet" Nutrients 10, no. 11: 1746. https://doi.org/10.3390/nu10111746
APA StyleHaupt-Jorgensen, M., Holm, L. J., Josefsen, K., & Buschard, K. (2018). Possible Prevention of Diabetes with a Gluten-Free Diet. Nutrients, 10(11), 1746. https://doi.org/10.3390/nu10111746





