Therapeutic Microbiology: The Role of Bifidobacterium breve as Food Supplement for the Prevention/Treatment of Paediatric Diseases
Abstract
1. Introduction
2. The Human Intestinal Microbiota
3. Probiotics with a Special Emphasis on Bifidobacterium breve
4. B. breve Effectiveness in Mice Models
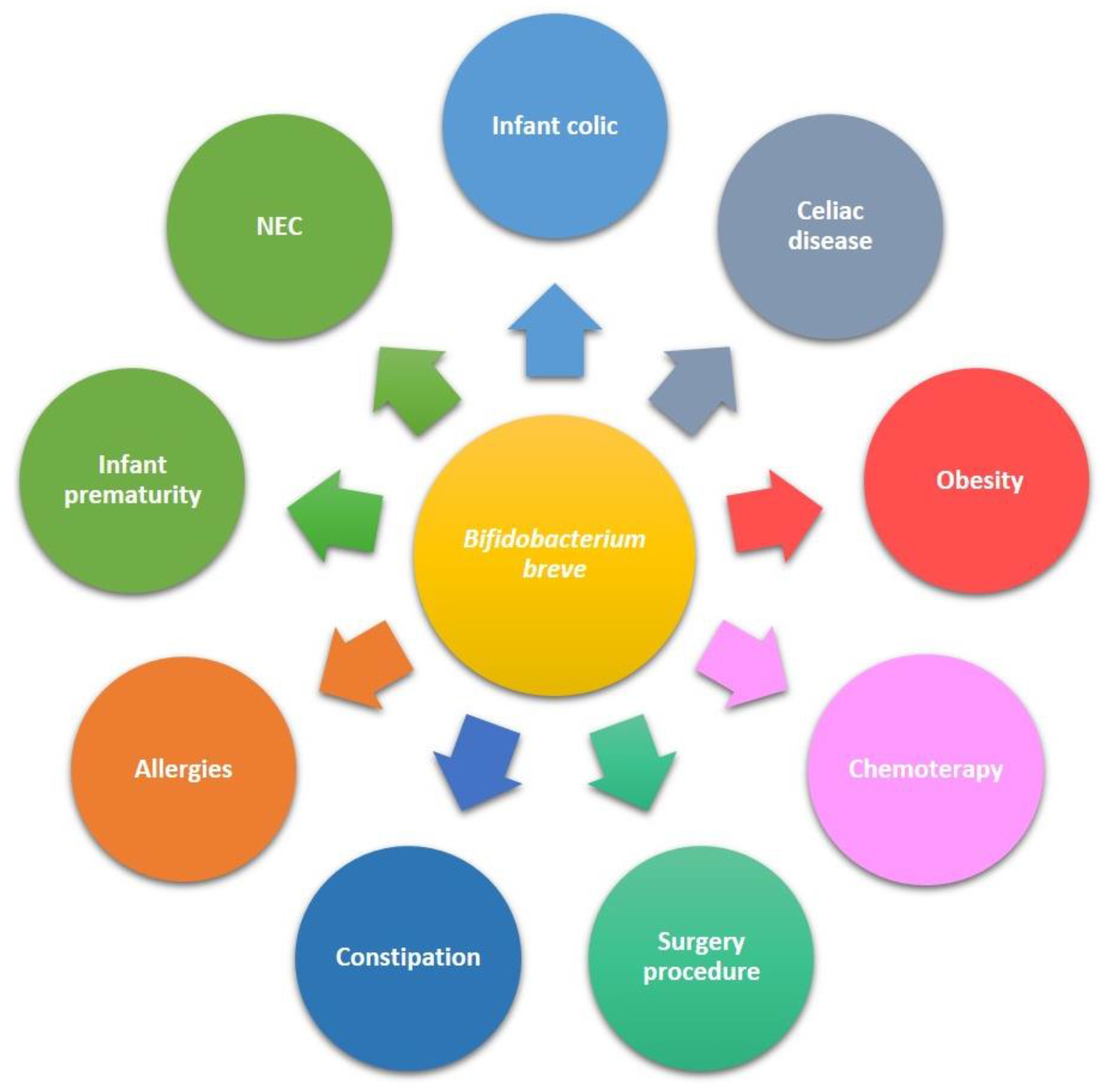
5. B. breve Application in Clinical Trials in Paediatrics
5.1. Preterm Infants and Necrotising Enterocolitis (NEC)
5.2. Gastrointestinal Disorders
5.3. Celiac Disease
5.4. Paediatric Obesity
5.5. Allergies
5.6. Surgical Procedures
5.7. Coadjuvant in Chemotherapic Treatment
6. B. breve Administration in Adults: A Short Outcome
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spinler, J.K.; Versalovic, J. Probiotics in Human Medicine: Overview. In Therapeutic Microbiology: Probiotics and Related Strategies, 1st ed.; Versalovic, J., Wilson, M., Eds.; ASM Press: Washington, DC, USA, 2008; pp. 225–229. [Google Scholar]
- Maassen, C.B.M.; Van Holten-Neelen, C.; Balk, F.; Heijne Den Bak-Glashouwer, M.J.; Leer, R.J.; Laman, J.D.; Boersma, W.J.A.; Claassen, E. Strain-dependent induction of cytokine profiles in the gut by orally administered Lactobacillus strains. Vaccine 2000, 18, 2613–2623. [Google Scholar] [CrossRef]
- O’Hara, A.M.; Shanahan, F. The gut flora as a forgotten organ. EMBO Rep. 2006, 7, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.N.; St. Amand, A.L.; Feldman, R.A.; Boedeker, E.C.; Harpaz, N.; Pace, N.R. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc. Natl. Acad. Sci. USA 2007, 104, 13780–13785. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, B.; Delgado, S.; Blanco-Míguez, A.; Lourenço, A.; Gueimonde, M.; Margolles, A. Probiotics, gut microbiota, and their influence on host health and disease. Mol. Nutr. Food Res. 2016, 61, 1600240. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; O’Mahony, S.M. The microbiome-gut-brain axis: From bowel to behavior. Neurogastroenterol. Motil. 2011, 23, 187–192. [Google Scholar] [CrossRef] [PubMed]
- De Theije, C.G.M.; Wu, J.; Da Silva, S.L.; Kamphuis, P.J.; Garssen, J.; Korte, S.M.; Kraneveld, A.D. Pathways underlying the gut-to-brain connection in autism spectrum disorders as future targets for disease management. Eur. J. Pharmacol. 2011, 668, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Berer, K.; Gerdes, L.A.; Cekanaviciute, E.; Jia, X.; Xiao, L.; Xia, Z.; Liu, C.; Klotz, L.; Stauffer, U.; Baranzini, S.E.; et al. Gut microbiota from multiple sclerosis patients enables spontaneous autoimmune encephalomyelitis in mice. Proc. Natl. Acad. Sci. USA 2017, 114, 10719–10724. [Google Scholar] [CrossRef] [PubMed]
- Scheperjans, F.; Aho, V.; Pereira, P.A.B.; Koskinen, K.; Paulin, L.; Pekkonen, E.; Haapaniemi, E.; Kaakkola, S.; Eerola-Rautio, J.; Pohja, M.; et al. Gut microbiota are related to Parkinson’s disease and clinical phenotype. Mov. Disord. 2015, 30, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Del Chierico, F.; Vernocchi, P.; Bonizzi, L.; Carsetti, R.; Castellazzi, A.M.; Dallapiccola, B.; de Vos, W.; Guerzoni, M.E.; Manco, M.; Marseglia, G.L.; et al. Early-life gut microbiota under physiological and pathological conditions: The central role of combined meta-omics-based approaches. J. Proteomics 2012, 75, 4580–4587. [Google Scholar] [CrossRef] [PubMed]
- Metagenomics of the Human Intestinal Tract. Available online: http://www.metahit.eu/ (accessed on 24 August 2018).
- NIH Human Microbiome Project. Available online: https://hmpdacc.org/ (accessed on 24 August 2018).
- My New Gut. Available online: http://www.mynewgut.eu/ (accessed on 24 August 2018).
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef] [PubMed]
- Graf, D.; Di Cagno, R.; Fåk, F.; Flint, H.J.; Nyman, M.; Saarela, M.; Watzl, B. Contribution of diet to the composition of the human gut microbiota. Microb. Ecol. Health Dis. 2015, 26, 26164. [Google Scholar] [CrossRef] [PubMed]
- Sokol, H.; Leducq, V.; Aschard, H.; Pham, H.-P.; Jegou, S.; Landman, C.; Cohen, D.; Liguori, G.; Bourrier, A.; Nion-Larmurier, I.; et al. Fungal microbiota dysbiosis in IBD. Gut 2017, 66, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Dridi, B.; Raoult, D.; Drancourt, M. Archaea as emerging organisms in complex human microbiomes. Anaerobe 2011, 17, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Ishaq, S.L.; Moses, P.L.; Wright, A.-D.G. The Pathology of Methanogenic Archaea in Human Gastrointestinal Tract Disease. In The Gut Microbiome; Mozsik, G., Ed.; IntechOpen: Rijeka, Croatia, 2016. [Google Scholar]
- Breitbart, M.; Hewson, I.; Felts, B.; Mahaffy, J.M.; Nulton, J.; Salamon, P.; Rohwer, F. Metagenomic Analyses of an Uncultured Viral Community from Human Feces. J. Bacteriol. 2003, 185, 6220–6223. [Google Scholar] [CrossRef] [PubMed]
- Breitbart, M.; Haynes, M.; Kelley, S.; Angly, F.; Edwards, R.A.; Felts, B.; Mahaffy, J.M.; Mueller, J.; Nulton, J.; Rayhawk, S.; et al. Viral diversity and dynamics in an infant gut. Res. Microbiol. 2008, 159, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Scarpellini, E.; Ianiro, G.; Attili, F.; Bassanelli, C.; De Santis, A.; Gasbarrini, A. The human gut microbiota and virome: Potential therapeutic implications. Dig. Liver Dis. 2015, 47, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
- Francino, M. Early Development of the Gut Microbiota and Immune Health. Pathogens 2014, 3, 769–790. [Google Scholar] [CrossRef] [PubMed]
- Solís, G.; de los Reyes-Gavilan, C.G.; Fernández, N.; Margolles, A.; Gueimonde, M. Establishment and development of lactic acid bacteria and bifidobacteria microbiota in breast-milk and the infant gut. Anaerobe 2010, 16, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Sharon, I.; Morowitz, M.J.; Thomas, B.C.; Costello, E.K.; Relman, D.A.; Banfield, J.F. Time series community genomics analysis reveals rapid shifts in bacterial species, strains, and phage during infant gut colonization. Genome Res. 2013, 23, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.B.; Konya, T.; Maughan, H.; Guttman, D.S.; Field, C.J.; Chari, R.S.; Sears, M.R.; Becker, A.B.; Scott, J.A.; Kozyrskyj, A.L. Gut microbiota of healthy Canadian infants: Profiles by mode of delivery and infant diet at 4 months. Can. Med. Assoc. J. 2013, 185, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.A.; Lim, J.Y.; Kim, B.S.; Cho, S.J.; Kim, N.Y.; Kim, O.B.; Kim, Y. Comparison of the gut microbiota profile in breast-fed and formula-fed Korean infants using pyrosequencing. Nutr. Res. Pract. 2015, 9, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Tham, C.S.C.; Peh, K.K.; Bhat, R.; Liong, M.T. Probiotic properties of bifidobacteria and lactobacilli isolated from local dairy products. Ann. Microbiol. 2012, 62, 1079–1087. [Google Scholar] [CrossRef]
- Liepke, C.; Adermann, K.; Raida, M.; Mägert, H.J.; Forssmann, W.G.; Zucht, H.D. Human milk provides peptides highly stimulating the growth of bifidobacteria. Eur. J. Biochem. 2002, 269, 712–718. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Huse, S.M.; Dethlefsen, L.; Huber, J.A.; Welch, D.M.; Relman, D.A.; Sogin, M.L. Exploring microbial diversity and taxonomy using SSU rRNA hypervariable tag sequencing. PLoS Genet. 2008, 4, 1000255. [Google Scholar] [CrossRef]
- Jernberg, C.; Löfmark, S.; Edlund, C.; Jansson, J.K. Long-term ecological impacts of antibiotic administration on the human intestinal microbiota. ISME J. 2007, 1, 56–66. [Google Scholar] [CrossRef] [PubMed]
- De La Cochetière, M.F.; Durand, T.; Lepage, P.; Bourreille, A.; Galmiche, J.P.; Doré, J. Resilience of the Dominant Human Fecal Microbiota upon Short-Course Antibiotic Challenge. J. Clin. Microbiol. 2005, 43, 5588–5592. [Google Scholar] [CrossRef]
- Arboleya, S.; Sánchez, B.; Solís, G.; Fernández, N.; Suárez, M.; Hernández-Barranco, A.M.; Milani, C.; Margolles, A.; De Los Reyes-Gavilán, C.G.; Ventura, M.; et al. Impact of prematurity and perinatal antibiotics on the developing intestinal microbiota: A functional inference study. Int. J. Mol. Sci. 2016, 17, 649. [Google Scholar] [CrossRef] [PubMed]
- Aloisio, I.; Quagliariello, A.; De Fanti, S.; Luiselli, D.; De Filippo, C.; Albanese, D.; Corvaglia, L.T.; Faldella, G.; Di Gioia, D. Evaluation of the effects of intrapartum antibiotic prophylaxis on newborn intestinal microbiota using a sequencing approach targeted to multi hypervariable 16S rDNA regions. Appl. Microbiol. Biotechnol. 2016, 100, 5537–5546. [Google Scholar] [CrossRef] [PubMed]
- Mazzola, G.; Murphy, K.; Ross, R.P.; Di Gioia, D.; Biavati, B.; Corvaglia, L.T.; Faldella, G.; Stanton, C. Early gut microbiota perturbations following intrapartum antibiotic prophylaxis to prevent group B streptococcal disease. PLoS ONE 2016, 11, e0157527. [Google Scholar] [CrossRef] [PubMed]
- Joint FAO/WHO. Expert consultation on evaluation of health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria. Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria: Cordoba, Argentina, 1–4 October 2001. Available online: http://isappscience.org/wp-content/uploads/2015/12/FAO-WHO-2001-Probiotics-Report.pdf (accessed on 28 August 2018).
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Di Mauro, A.; Riezzo, G.; Civardi, E.; Intini, C.; Corvaglia, L.; Ballardini, E.; Bisceglia, M.; Cinquetti, M.; Brazzoduro, E.; et al. Prophylactic use of a probiotic in the prevention of colic, regurgitation, and functional constipation a randomized clinical trial. JAMA Pediatr. 2014, 168, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H.; Gyrczuk, E.; Horvath, A. Lactobacillus reuteri DSM 17938 for the management of infantile colic in breastfed infants: A randomized, double-blind, placebo-controlled trial. J. Pediatr. 2013, 162, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Chau, K.; Lau, E.; Greenberg, S.; Jacobson, S.; Yazdani-Brojeni, P.; Verma, N.; Koren, G. Probiotics for infantile colic: A randomized, double-blind, placebo-controlled trial investigating Lactobacillus reuteri DSM 17938. J. Pediatr. 2015, 166, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Cordisco, L.; Tarasco, V.; Palumeri, E.; Calabrese, R.; Oggero, R.; Roos, S.; Matteuzzi, D. Lactobacillus reuteri DSM 17938 in Infantile Colic: A Randomized, Double-Blind, Placebo-Controlled Trial. Pediatrics 2010, 126, e526–e533. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Fornasero, S.; Ceratto, S.; De Marco, A.; Mandras, N.; Roana, J.; Tullio, V.; Amisano, G. Probiotics and gut health in infants: A preliminary case-control observational study about early treatment with Lactobacillus reuteri DSM 17938. Clin. Chim. Acta 2015, 451, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Oliva, S.; Di Nardo, G.; Ferrari, F.; Mallardo, S.; Rossi, P.; Patrizi, G.; Cucchiara, S.; Stronati, L. Randomised clinical trial: The effectiveness of Lactobacillus reuteri ATCC 55730 rectal enema in children with active distal ulcerative colitis. Aliment. Pharmacol. Ther. 2012, 35, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.J.; Martinez, E.G.; Gregorio, G.V.; Dans, L.F. Cochrane Review: Probiotics for treating acute infectious diarrhoea. Evid.-Based Child Health Cochrane Rev. J. 2011, 6, 1894–2021. [Google Scholar] [CrossRef]
- Sanders, M.E.; Akkermans, L.M.A.; Haller, D.; Hammerman, C.; Heimbach, J.T.; Hörmannsperger, G.; Huys, G. Safety assessment of probiotics for human use. Gut Microbes 2010, 1, 164–185. [Google Scholar] [CrossRef] [PubMed]
- Weizman, Z.; Asli, G.; Alsheikh, A. Effect of a Probiotic Infant Formula on Infections in Child Care Centers: Comparison of Two Probiotic Agents. Pediatrics 2005, 115, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Grandy, G.; Medina, M.; Soria, R.; Terán, C.G.; Araya, M. Probiotics in the treatment of acute rotavirus diarrhoea. A randomized, double-blind, controlled trial using two different probiotic preparations in Bolivian children. BMC Infect. Dis. 2010, 10, 253. [Google Scholar] [CrossRef] [PubMed]
- Vandenplas, Y.; De Hert, S.G. Randomised clinical trial: The synbiotic food supplement Probiotical vs. placebo for acute gastroenteritis in children. Aliment. Pharmacol. Ther. 2011, 34, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Hsu, C.; Chen, H.; Chung, M.; Hsu, J.; Lien, R.; Tsao, L.; Chen, C.; Su, B. Oral Probiotics Prevent Necrotizing Enterocolitis in Very Low Birth Weight Preterm Infants: A Multicenter, Randomized, Controlled Trial. Pediatrics 2008, 122, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Khailova, L.; Dvorak, K.; Arganbright, K.M.; Halpern, M.D.; Kinouchi, T.; Yajima, M.; Dvorak, B. Bifidobacterium bifidum improves intestinal integrity in a rat model of necrotizing enterocolitis. AJP Gastrointest. Liver Physiol. 2009, 1297, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Underwood, M.A.; Kananurak, A.; Coursodon, C.F.; Adkins-Reick, C.K.; Chu, H.; Bennett, S.H.; Wehkamp, J.; Castillo, P.A.; Leonard, B.C.; Tancredi, D.J.; et al. Bifidobacterium bifidum in a rat model of necrotizing enterocolitis: Antimicrobial peptide and protein responses. Pediatr. Res. 2012, 71, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Turroni, F.; Peano, C.; Pass, D.A.; Foroni, E.; Severgnini, M.; Claesson, M.J.; Kerr, C.; Hourihane, J.; Murray, D.; Fuligni, F.; et al. Diversity of bifidobacteria within the infant gut microbiota. PLoS ONE 2012, 7, e36957. [Google Scholar] [CrossRef] [PubMed]
- Reuter, G. Vergleichende Untersuchungen uber die Bifidus-flora im Sauglings-und Erwachsenenstuhl. Zentbl Bakteriol 1963, 191, 486–507. [Google Scholar]
- Rogosa, M. Genus III, Bifidobacterium Orla-Jensen. In Bergley’s Manual of Determinative Bacteriology, 8th ed.; Buchanan, R.E., Gibbons, N.E., Eds.; Williams and Wilkins Co.: Baltimore, MD, USA, 1974; pp. 669–676. [Google Scholar]
- Mara, D.D.; Oragui, J.I. Sorbitol-fermenting bifidobacteria as specific indicators of human faecal pollution. J. Appl. Bacteriol. 1983, 55, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Falk, P.; Hoskins, L.C.; Larson, G. Bacteria of the human intestinal microbiota produce glycosidases specific for lacto-series glycosphingolipids. J. Biochem. 1990, 108, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Asakuma, S.; Hatakeyama, E.; Urashima, T.; Yoshida, E.; Katayama, T.; Yamamoto, K.; Kumagai, H.; Ashida, H.; Hirose, J.; Kitaoka, M. Physiology of consumption of human milk oligosaccharides by infant gut-associated bifidobacteria. J. Biol. Chem. 2011, 286, 34583–34592. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on the maintenance of the list of QPS biological agents intentionally added to food and feed (2012 update). EFSA J. 2012, 11, 3449. [Google Scholar] [CrossRef]
- Fernández, L.; Langa, S.; Martín, V.; Maldonado, A.; Jiménez, E.; Martín, R.; Rodríguez, J.M. The human milk microbiota: Origin and potential roles in health and disease. Pharmacol. Res. 2013, 69, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, K.; Hosono, S.; Takahashi, E.; Ishizeki, S.; Takigawa, I.; Imura, S.; Yamauchi, K.; Yaeshima, T.; Hayasawa, H.; Shimamura, S. Effects of oral administration of Bifidobacterium breve on development of intestinal microflora in extremely premature infants. Acta Neonatol. Jpn. 1994, 30, 130–137. [Google Scholar]
- Aloisio, I.; Santini, C.; Biavati, B.; Dinelli, G.; Cencič, A.; Chingwaru, W.; Mogna, L.; Di Gioia, D. Characterization of Bifidobacterium spp. strains for the treatment of enteric disorders in newborns. Appl. Microbiol. Biotechnol. 2012, 96, 1561–1576. [Google Scholar] [CrossRef] [PubMed]
- Simone, M.; Gozzoli, C.; Quartieri, A.; Mazzola, G.; Di Gioia, D.; Amaretti, A.; Raimondi, S.; Rossi, M. The probiotic Bifidobacterium breve B632 inhibited the growth of enterobacteriaceae within colicky infant microbiota cultures. BioMed Res. Int. 2014. [Google Scholar] [CrossRef] [PubMed]
- Mogna, L.; Del Piano, M.; Deidda, F.; Nicola, S.; Soattini, L.; Debiaggi, R.; Sforza, F.; Strozzi, G.; Mogna, G. Assessment of the in vitro inhibitory activity of specific probiotic bacteria against different Escherichia coli strains. J. Clin. Gastroenterol. 2012, 46, S29–S32. [Google Scholar] [CrossRef] [PubMed]
- Mogna, L.; Del Piano, M.; Mogna, G. Capability of the two microorganisms Bifidobacterium breve B632 and Bifidobacterium breve BR03 to colonize the intestinal microbiota of children. J. Clin. Gastroenterol. 2014, 48, S37–S39. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, V.M.; Sleator, R.D.; Hill, C.; Fitzgerald, G.F. Improving gastric transit, gastrointestinal persistence and therapeutic efficacy of the probiotic strain Bifidobacterium breve UCC2003. Microbiology 2007, 153, 3563–3571. [Google Scholar] [CrossRef] [PubMed]
- Tojo, M.; Oikawa, T.; Morikawa, Y.; Yamashita, N.; Iwata, S.; Satoh, Y.; Hanada, J.; Tanaka, R. The Effects of Bifidobacterium breve Administration on Campylobacter Enteritis. Pediatr. Int. 1987, 29, 160–167. [Google Scholar] [CrossRef]
- Hotta, M.; Sato, Y.; Iwata, S.; Yamashita, N.; Sunakawa, K.; Oikawa, T.; Tanaka, R.; Takayama, H.; Yajima, M.; Watanabe, K.; et al. Clinical Effects of Bifidobacterium Preparations on Pediatric Intractable Diarrhea. Keio J. Med. 1987, 36, 298–314. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Shimizu, K.; Nomoto, K.; Hamabata, T.; Ozawa, A.; Takeda, Y. Probiotic Bifidobacteria Protect Mice from Lethal Infection with Shiga Toxin-Producing Escherichia coli O157:H7. Society 2004, 72, 2240–2247. [Google Scholar] [CrossRef]
- Kikuchi-Hayakawa, H.; Onodera, N.; Matsubara, S.; Yasuda, E.; Shimakawa, Y.; Ishikawa, F. Effects of soya milk and Bifidobacterium-fermented soya milk on plasma and liver lipids, and faecal steroids in hamsters fed on a cholesterol-free or cholesterol-enriched diet. Br. J. Nutr. 1998, 79, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Kano, M.; Ishikawa, F.; Matsubara, S.; Kikuchi-Hayakawa, H.; Shimakawa, Y. Soymilk products affect ethanol absorption and metabolism in rats during acute and chronic ethanol intake. J. Nutr. 2002, 132, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Ohta, T.; Nakatsugi, S.; Watanabe, K.; Kawamori, T.; Ishikawa, F.; Morotomi, M.; Sugie, S.; Toda, T.; Sugimura, T.; Wakabayashi, K. Inhibitory effects of Bifidobacterium-fermented soy milk on 2-amino-1-methyl-6-phenylimidazo[4,5-b] pyridine-induced rat mammary carcinogenesis, with a partial contribution of its component isoflavones. Carcinogenesis 2000, 21, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Chapman, T.M.; Plosker, G.L.; Figgitt, D.P. VSL# 3 probiotic mixture. Drugs 2006, 66, 1371–1387. [Google Scholar] [PubMed]
- Yasui, H.; Kiyoshima, J.; Hori, T.; Shida, K. Protection against influenza virus infection of mice fed Bifidobacterium breve YIT4064. Clin. Diagn. Lab. Immunol. 1999, 6, 186–192. [Google Scholar] [PubMed]
- Yasui, H.; Ohwaki, M. Enhancement of Immune Response in Peyer’s Patch Cells Cultured with Bifidobacterium breve. J. Dairy Sci. 1991, 74, 1187–1195. [Google Scholar] [CrossRef]
- Fanning, S.; Hall, L.J.; van Sinderen, D. Bifidobacterium breve UCC2003 surface exopolysaccharide production is a beneficial trait mediating commensal-host interaction through immune modulation and pathogen protection. Gut Microbes 2012, 3, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Frederick, M.R.; Kuttler, C.; Hense, B.A.; Eberl, H.J. A mathematical model of quorum sensing regulated EPS production in biofilm communities. Theor. Biol. Med. Model. 2011, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Natividad, J.M.M.; Hayes, C.L.; Motta, J.P.; Jury, J.; Galipeau, H.J.; Philip, V.; Garcia-Rodenas, C.L.; Kiyama, H.; Bercik, P.; Verdú, E.F. Differential induction of antimicrobial REGIII by the intestinal microbiota and Bifidobacterium breve NCC2950. Appl. Environ. Microbiol. 2013, 79, 7745–7754. [Google Scholar] [CrossRef] [PubMed]
- Raftis, E.J.; Delday, M.I.; Cowie, P.; McCluskey, S.M.; Singh, M.D.; Ettorre, A.; Mulder, I.E. Bifidobacterium breve MRx0004 protects against airway inflammation in a severe asthma model by suppressing both neutrophil and eosinophil lung infiltration. Sci. Rep. 2018, 8, 12024. [Google Scholar] [CrossRef] [PubMed]
- Inoue, Y.; Iwabuchi, N.; Xiao, J.; Yaeshima, T.; Iwatsuki, K. Suppressive Effects of Bifidobacterium breve Strain M-16V on T-Helper Type 2 Immune Responses in a Murine Model. Biol. Pharm. Bull. 2009, 32, 760–763. [Google Scholar] [CrossRef] [PubMed]
- Schouten, B.; van Esch, B.C.A.M.; Hofman, G.A.; van Doorn, S.A.C.M.; Knol, J.; Nauta, A.J.; Garssen, J.; Willemsen, L.E.M.; Knippels, L.M.J. Cow Milk Allergy Symptoms Are Reduced in Mice Fed Dietary Synbiotics during Oral Sensitization with Whey. J. Nutr. 2009, 139, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Kostadinova, A.I.; Meulenbroek, L.A.; van Esch, B.C.; Hofman, G.A.; Garssen, J.; Willemsen, L.E.; Knippels, L.M. A Specific Mixture of Fructo-Oligosaccharides and Bifidobacterium breve M-16V Facilitates Partial Non-Responsiveness to Whey Protein in Mice Orally Exposed to β-Lactoglobulin-Derived Peptides. Front. Immunol. 2017, 7, 673. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.G.; Kayama, H.; Ueda, Y.; Takahashi, T.; Asahara, T.; Tsuji, H.; Tsuji, N.M.; Kiyono, H.; Ma, J.S.; Kusu, T.; et al. Probiotic Bifidobacterium breve induces IL-10-producing Tr1 cells in the colon. PLoS Pathog. 2012, 8, 1002714. [Google Scholar] [CrossRef] [PubMed]
- Kondo, S.; Xiao, J.; Satoh, T.; Odamaki, T.; TakaiashiI, S.; Sughara, H.; Yaeshima, T.; Iwatsuki, K.; Kamei, A.; Abe, K. Antiobesity Effects of Bifidobacterium breve Strain B-3 Supplementation in a Mouse Model with High-Fat Diet-Induced Obesity. Biosci. Biotechnol. Biochem. 2010, 74, 1656–1661. [Google Scholar] [CrossRef] [PubMed]
- Kondo, S.; Kamei, A.; Xiao, J.Z.; Iwatsuki, K.; Abe, K. Bifidobacterium breve B-3 exerts metabolic syndrome-suppressing effects in the liver of diet-induced obese mice: A DNA microarray analysis. Benef. Microbes 2013, 4, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.A.; McVey Neufeld, K.-A. Gut-brain axis: How the microbiome influences anxiety and depression. Trends Neurosci. 2013, 36, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Stanton, C.; Cryan, J.F. Psychobiotics: A Novel Class of Psychotropic. Biol. Psychiatry 2013, 74, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Savignac, H.M.; Kiely, B.; Dinan, T.G.; Cryan, J.F. Bifidobacteria exert strain-specific effects on stress-related behavior and physiology in BALB/c mice. Neurogastroenterol. Motil. 2014, 26, 1615–1627. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Sugahara, H.; Shimada, K.; Mitsuyama, E.; Kuhara, T.; Yasuoka, A.; Kondo, T.; Abe, K.; Xiao, J.Z. Therapeutic potential of Bifidobacterium breve strain A1 for preventing cognitive impairment in Alzheimer’s disease. Sci. Rep. 2017, 7, 13510. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glickman, J.N.; Garrett, W.S. The Microbial Metabolites, Short-Chain Fatty Acids, Regulate Colonic Treg Cell Homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Salem, N.; Wegher, B.; Mena, P.; Uauy, R. Arachidonic and docosahexaenoic acids are biosynthesized from their 18-carbon precursors in human infants. Proc. Natl. Acad. Sci. USA 1996, 93, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Coakley, M.; Ross, R.P.; Nordgren, M.; Fitzgerald, G.; Devery, R.; Stanton, C. Conjugated linoleic acid biosynthesis by human-derived Bifidobacterium species. J. Appl. Microbiol. 2002, 94, 138–145. [Google Scholar] [CrossRef]
- Kritchevsky, D.; Tepper, S.A.; Wright, S.; Tso, P.; Czarnecki, S.K. Influence of Conjugated Linoleic Acid (CLA) on Establishment and Progression of Atherosclerosis in Rabbits. J. Am. Coll. Nutr. 2000, 19, 472S–477S. [Google Scholar] [CrossRef] [PubMed]
- Bassaganya-Riera, J.; Hontecillas, R.; Beitz, D.C. Colonic anti-inflammatory mechanisms of conjugated linoleic acid. Clin. Nutr. 2002, 21, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Wall, R.; Ross, R.P.; Shanahan, F.; Mahony, L.O.; Mahony, C.O.; Coakley, M.; Hart, O.; Lawlor, P.; Quigley, E.M.; Kiely, B.; et al. Metabolic activity of the enteric microbiota in uences the fatty acid composition of murine and porcine liver and adipose tissues. Am. J. Clin. Nutr. 2009, 89, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Bolaños, C.A.; Nestler, E.J. Neurotrophic mechanisms in drug addiction. Neuromol. Med. 2004, 5, 69–83. [Google Scholar] [CrossRef]
- Nawa, H.; Takahashi, M.; Patterson, P.H. Cytokine and growth factor involvement in schizophrenia-support for the developmental model. Mol. Psychiatry 2000, 5, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Molteni, R.; Calabrese, F.; Cattaneo, A.; Mancini, M.; Gennarelli, M.; Racagni, G.; Riva, M.A. Acute Stress Responsiveness of the Neurotrophin BDNF in the Rat Hippocampus is Modulated by Chronic Treatment with the Antidepressant Duloxetine. Neuropsychopharmacology 2008, 34, 1523. [Google Scholar] [CrossRef] [PubMed]
- Chapman, C.M.C.; Gibson, G.R.; Rowland, I. Health benefits of probiotics: Are mixtures more effective than single strains? Eur. J. Nutr. 2011, 50, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Timmerman, H.M.; Koning, C.J.M.; Mulder, L.; Rombouts, F.M.; Beynen, A.C. Monostrain, multistrain and multispecies probiotics—A comparison of functionality and efficacy. Int. J. Food Microbiol. 2004, 96, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Stoll, B.J.; Hansen, N.; Fanaroff, A.A.; Wright, L.L.; Carlo, W.A.; Ehrenkranz, R.A.; Lemons, J.A.; Donovan, E.F.; Stark, A.R.; Tyson, J.E.; et al. Late-onset sepsis in very low birth weight neonates: The experience of the NICHD Neonatal Research Network. Pediatrics 2002, 110, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Pessoa-Silva, C.L.; Miyasaki, C.H.; de Almeida, M.F.; Kopelman, B.I.; Raggio, R.L.; Wey, S.B. Neonatal late-onset bloodstream infection: Attributable mortality, excess of length of stay and risk factors. Eur. J. Epidemiol. 2001, 17, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Panigrahi, P.; Gupta, S.; Gewolb, I.H.; Morris, J.G., Jr. Occurrence of Necrotizing Enterocolitis May Be Dependent on Patterns of Bacterial Adherence and Intestinal Colonization: Studies in Caco-2 Tissue Culture and Weanling Rabbit Models. Pediatr. Res. 1994, 36, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Dai, D.; Walker, W.A. Protective nutrients and bacterial colonization in the immature human gut. Adv. Pediatr. 1999, 46, 353–382. [Google Scholar] [PubMed]
- Butel, M.J.; Suau, A.; Campeotto, F.; Magne, F.; Aires, J.; Ferraris, L.; Kalach, N.; Leroux, B.; Dupont, C. Conditions of Bifidobacterial Colonization in Preterm Infants: A Prospective Analysis. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Jacquot, A.; Neveu, D.; Aujoulat, F.; Mercier, G.; Marchandin, H.; Jumas-Bilak, E.; Picaud, J.-C. Dynamics and Clinical Evolution of Bacterial Gut Microflora in Extremely Premature Patients. J. Pediatr. 2011, 158, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Arboleya, S.; Binetti, A.; Salazar, N.; Fernández, N.; Solís, G.; Hernández-Barranco, A.; Margolles, A.; de los Reyes-Gavilán, C.G.; Gueimonde, M. Establishment and development of intestinal microbiota in preterm neonates. FEMS Microbiol. Ecol. 2012, 79, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, H.; Sumida, Y.; Tanaka, R. Early administration of Bifidobacterium breve to preterm infants: Randomised controlled trial. Arch. Dis. Child. Fetal Neonatal Ed. 1997, 76, 101–107. [Google Scholar] [CrossRef]
- Li, Y.; Shimizu, T.; Hosaka, A.; Kaneko, N.; Ohtsuka, Y.; Yamashiro, Y. Effects of Bifidobacterium breve supplementation on intestinal flora of low birth weight infants. Pediatr. Int. 2004, 46, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Patole, S.; Keil, A.D.; Chang, A.; Nathan, E.; Doherty, D.; Simmer, K.; Esvaran, M.; Conway, P. Effect of Bifidobacterium breve M-16V supplementation on fecal bifidobacteria in preterm neonates—A randomised double blind placebo controlled trial. PLoS ONE 2014, 9, e89511. [Google Scholar] [CrossRef] [PubMed]
- Patole, S.K.; Rao, S.C.; Keil, A.D.; Nathan, E.A.; Doherty, D.A.; Simmer, K.N. Benefits of Bifidobacterium breve M-16V Supplementation in preterm neonates—A retrospective cohort study. PLoS ONE 2016, 11, e0150775. [Google Scholar] [CrossRef] [PubMed]
- Neu, J.; Walker, W.A. Necrotizing Enterocolitis. N. Engl. J. Med. 2011, 364, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.W.; Stoll, B.J. Necrotising enterocolitis. Lancet 2006, 368, 1271–1283. [Google Scholar] [CrossRef]
- Bell, M.J.; Ternberg, J.L.; Feigin, R.D.; Keating, J.P.; Marshall, R.; Barton, L.; Brotherton, T. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann. Surg. 1978, 187, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jain, L. Necrotizing Enterocolitis Prevention: Art or Science? Clin. Perinatol. 2013, 40, xiii–xv. [Google Scholar] [CrossRef] [PubMed]
- Mai, V.; Young, C.M.; Ukhanova, M.; Wang, X.; Sun, Y.; Casella, G.; Theriaque, D.; Li, N.; Sharma, R.; Hudak, M.; et al. Fecal Microbiota in Premature Infants Prior to Necrotizing Enterocolitis. PLoS ONE 2011, 6, e20647. [Google Scholar] [CrossRef] [PubMed]
- Satoh, Y.; Shinohara, K.; Umezaki, H.; Shoji, H.; Satoh, H.; Ohtsuka, Y.; Shiga, S.; Nagata, S.; Shimizu, T.; Yamashiro, Y. Bifidobacteria prevents necrotizing enterocolitic and infection in preterm infants. Int. J. Probiot. Prebiot. 2007, 2, 149–154. [Google Scholar]
- Lin, J. Too much short chain fatty acids cause neonatal necrotizing enterocolitis. Med. Hypotheses 2004, 62, 291–293. [Google Scholar] [CrossRef]
- Lin, J.; Nafday, S.M.; Chauvin, S.N.; Magid, M.S.; Pabbatireddy, S.; Holzman, I.R.; Babyatsky, M.W. Variable Effects of Short Chain Fatty Acids and Lactic Acid in Inducing Intestinal Mucosal Injury in Newborn Rats. J. Pediatr. Gastroenterol. Nutr. 2002, 35, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shoji, H.; Sato, H.; Nagata, S.; Ohtsuka, Y.; Shimizu, T.; Yamashiro, Y. Effects of oral administration of Bifidobacterium breve on fecal lactic acid and short-chain fatty acids in low birth weight infants. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Ohtsuka, Y.; Lee, T.; Kudo, T.; Shoji, H.; Sato, H.; Nagata, S.; Shimizu, T.; Yamashiro, Y. Bifidobacterium breve enhances transforming growth factor β1 signaling by regulating Smad7 expression in preterm infants. J. Pediatr. Gastroenterol. Nutr. 2006, 43, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuka, Y.; Sanderson, I.R. Transforming growth factor-β: An important cytokine in the mucosal immune response. Curr. Opin. Gastroenterol. 2000, 16, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Hikaru, U.; Koichi, S.; Yayoi, S.; Hiromichi, S.; Hiroaki, S.; Yoshikazu, O.; Seigo, S.; Nagata, S.; Toshiaki, S.; Yamashiro, Y. Bifidobacteria prevents preterm infants from developing infection and sepsis. Int. J. Probiot. Prebiot. 2010, 5, 33–36. [Google Scholar]
- Braga, T.D.; Alves, G.; Israel, P.; De Lira, C.; Lima, M.D.C. Efficacy of Bifidobacterium breve and Lactobacillus casei oral supplementation on necrotizing enterocolitis in very-low-birth-weight preterm infants: A double-blind, randomized, controlled trial 1–3. J. Clin. 2011, 93, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Pelle, E.; Palumeri, E.; Oggero, R.; Miniero, R. Lactobacillus reuteri (American Type Culture Collection Strain 55730) Versus Simethicone in the Treatment of Infantile Colic: A Prospective Randomized Study. Pediatrics 2007, 119, e124–e130. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Di Mauro, A.; Riezzo, G.; Cavallo, L.; Francavilla, R. Infantile colic, regurgitation, and constipation: An early traumatic insult in the development of functional gastrointestinal disorders in children? Eur. J. Pediatr. 2015, 174, 841–842. [Google Scholar] [CrossRef] [PubMed]
- Romanello, S.; Spiri, D.; Marcuzzi, E.; Zanin, A.; Boizeau, P.; Riviere, S.; Vizeneux, A.; Moretti, R.; Carbajal, R.; Mercier, J.C.; et al. Association between Childhood Migraine and History of Infantile Colic. JAMA 2013, 309, 1607–1612. [Google Scholar] [CrossRef] [PubMed]
- Giglione, E.; Prodam, F.; Bellone, S.; Monticone, S.; Beux, S.; Marolda, A.; Pagani, A.; Di Gioia, D.; Del Piano, M.; Mogna, G.; et al. The Association of Bifidobacterium breve BR03 and B632 is Effective to Prevent Colics in Bottle-fed Infants. J. Clin. Gastroenterol. 2016, 50, S164–S167. [Google Scholar] [CrossRef] [PubMed]
- Aloisio, I.; Prodam, F.; Giglione, E.; Bozzi Cionci, N.; Solito, A.; Bellone, S.; Baffoni, L.; Mogna, L.; Pane, M.; Bona, G.; et al. Three-Month Feeding Integration with Bifidobacterium Strains Prevents Gastrointestinal Symptoms in Healthy Newborns. Front. Nutr. 2018, 5, 39. [Google Scholar] [CrossRef] [PubMed]
- Magne, F.; Puchi Silva, A.; Carvajal, B.; Gotteland, M. The Elevated Rate of Cesarean Section and Its Contribution to Non-Communicable Chronic Diseases in Latin America: The Growing Involvement of the Microbiota. Front. Pediatr. 2017, 5, 192. [Google Scholar] [CrossRef] [PubMed]
- Kuhle, S.; Tong, O.S.; Woolcott, C.G. Association between caesarean section and childhood obesity: A systematic review and meta-analysis. Obes. Rev. 2015, 16, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, M.M.; Benninga, M.A.; Di Lorenzo, C. Epidemiology of Childhood Constipation: A Systematic Review. Am. J. Gastroenterol. 2006, 101, 2401–2409. [Google Scholar] [CrossRef] [PubMed]
- Bongers, M.E.J.; van Wijk, M.P.; Reitsma, J.B.; Benninga, M.A. Long-Term Prognosis for Childhood Constipation: Clinical Outcomes in Adulthood. Pediatrics 2010, 126, e156–e162. [Google Scholar] [CrossRef] [PubMed]
- Tabbers, M.M.; de Milliano, I.; Roseboom, M.G.; Benninga, M.A. Is Bifidobacterium breve effective in the treatment of childhood constipation? Results from a pilot study. Nutr. J. 2011, 10, 19. [Google Scholar] [CrossRef] [PubMed]
- Giannetti, E.; Maglione, M.; Alessandrella, A.; Strisciuglio, C.; De Giovanni, D.; Campanozzi, A.; Miele, E.; Staiano, A. A Mixture of 3 Bifidobacteria Decreases Abdominal Pain and Improves the Quality of Life in Children with Irritable Bowel Syndrome. J. Clin. Gastroenterol. 2017, 51, e5–e10. [Google Scholar] [CrossRef] [PubMed]
- Saez-Lara, M.J.; Gomez-Llorente, C.; Plaza-Diaz, J.; Gil, A. The Role of Probiotic Lactic Acid Bacteria and Bifidobacteria in the Prevention and Treatment of Inflammatory Bowel Disease and Other Related Diseasea: A Systematic Review of Randomized Human Clinical Trials. BioMed Res. Int. 2015, 2015, 505878. [Google Scholar] [CrossRef] [PubMed]
- Guandalini, S.; Magazzù, G.; Chiaro, A.; La Balestra, V.; Di Nardo, G.; Gopalan, S.; Sibal, A.; Romano, C.; Canani, R.B.; Lionetti, P.; et al. VSL#3 improves symptoms in children with irritable bowel Syndrome: A multicenter, randomized, placebo-controlled, double-blind, crossover study. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Miele, E.; Pascarella, F.; Giannetti, E.; Quaglietta, L.; Baldassano, R.N.; Staiano, A. Effect of a probiotic preparation (VSL#3) on induction and maintenance of remission in children with ulcerative colitis. Am. J. Gastroenterol. 2009, 104, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Heyman, M.B.; Kirschner, B.S.; Gold, B.D.; Ferry, G.; Baldassano, R.; Cohen, S.A.; Winter, H.S.; Fain, P.; King, C.; Smith, T.; et al. Children with early-onset inflammatory bowel disease (IBD): Analysis of a pediatric IBD consortium registry. J. Pediatr. 2005, 146, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Walsh, C.M.; Benchimol, E.I.; Mann, E.H.; Thomas, K.E.; Chow, C.; McLernon, R.A.; Walters, T.D.; Swales, J.; Steinhart, A.H.; et al. Severe paediatric ulcerative colitis: Incidence, outcomes and optimal timing for second-line therapy. Gut 2008, 57, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Dubey, P.A.; Rajeshwari, K.; Chakravarty, A.; Famularo, G. Use of VSL # 3 in the Treatment of Rotavirus Diarrhea in Children: Preliminary results. J. Clin. Gastroenterol. 2008, 42, 126–129. [Google Scholar]
- Sinha, A.; Gupta, S.S.; Chellani, H.; Maliye, C.; Kumari, V.; Arya, S.; Garg, B.S.; Gaur, S.D.; Gaind, R.; Deotale, V.; et al. Role of probiotics VSL#3 in prevention of suspected sepsis in low birthweight infants in India: A randomised controlled trial. BMJ Open 2015, 5, e006564. [Google Scholar] [CrossRef] [PubMed]
- Thibault, H.; Aubert-Jacquin, C.; Goulet, O. Effects of Long-term Consumption of a Fermented Infant Formula (with Bifidobacterium breve c50 and Streptococcus thermophilus 065) on Acute Diarrhea in Healthy Infants. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Klemenak, M.; Dolinšek, J.; Langerholc, T.; Di Gioia, D.; Mičetić-Turk, D. Administration of Bifidobacterium breve Decreases the Production of TNF-α in Children with Celiac Disease. Dig. Dis. Sci. 2015, 60, 3386–3392. [Google Scholar] [CrossRef] [PubMed]
- Quagliariello, A.; Aloisio, I.; Bozzi Cionci, N.; Luiselli, D.; D’Auria, G.; Martinez-Priego, L.; Pérez-Villarroya, D.; Langerholc, T.; Primec, M.; Mičetić-Turk, D.; et al. Effect of Bifidobacterium breve on the Intestinal Microbiota of Coeliac Children on a Gluten Free Diet: A Pilot Study. Nutrients 2016, 8, 660. [Google Scholar] [CrossRef] [PubMed]
- Primec, M.; Klemenak, M.; Di Gioia, D.; Aloisio, I.; Bozzi Cionci, N.; Quagliariello, A.; Gorenjak, M.; Mičetić-Turk, D.; Langerholc, T. Clinical intervention using Bifidobacterium strains in celiac disease children reveals novel microbial modulators of TNF-α and short-chain fatty acids. Clin. Nutr. 2018, 4, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Horz, H.-P.; Citron, D.M.; Warren, Y.A.; Goldstein, E.J.C.; Conrads, G. Synergistes Group Organisms of Human Origin. J. Clin. Microbiol. 2006, 44, 2914–2920. [Google Scholar] [CrossRef] [PubMed]
- Rajilić-Stojanović, M.; de Vos, W.M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 2014, 38, 996–1047. [Google Scholar] [CrossRef] [PubMed]
- Schenk, S.; Saberi, M.; Olefsky, J.M. Insulin sensitivity: Modulation by nutrients and inflammation. J. Clin. Investig. 2008, 118, 2992–3002. [Google Scholar] [CrossRef] [PubMed]
- Rocha, V.Z.; Folco, E.J. Inflammatory Concepts of Obesity. Int. J. Inflam. 2011, 2011, 529061. [Google Scholar] [CrossRef] [PubMed]
- Angelakis, E.; Armougom, F.; Million, M.; Raoult, D. The relationship between gut microbiota and weight gain in humans. Future Microbiol. 2012, 7, 91–109. [Google Scholar] [CrossRef] [PubMed]
- Kalliomäki, M.; Carmen Collado, M.; Salminen, S.; Isolauri, E. Early differences in fecal microbiota composition in children may predict overweight. Am. J. Clin. Nutr. 2008, 87, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.N.; Yu, Q.F.; Fu, N.; Liu, X.W.; Lu, F.G. Effects of four Bifidobacteria on obesity in high-fat diet induced rats. World J. Gastroenterol. 2010, 16, 3394–3401. [Google Scholar] [CrossRef] [PubMed]
- Prodam, F.; Archero, F.; Aloisio, I.; Solito, A.; Ricotti, R.; Giglione, E.; Bozzi Cionci, N.; Bellone, S.; Di Gioia, D.; Bona, G. Efficacy of the treatment with Bifidobacterium breve B632 and Bifidobacterium breve BR03 on endocrine response to the oral glucose tolerance test in pediatric obesity: A cross-over double blind randomized controlled trial. In Proceedings of the 39° Congresso Nazionale Società Italiana di Endocrinologia (Endocrinologia 2.0), Rome, Italy, 21–24 June 2017. [Google Scholar]
- Watanabe, S.; Narisawa, Y.; Arase, S.; Okamatsu, H.; Ikenaga, T.; Tajiri, Y.; Kumemura, M. Differences in fecal microflora between patients with atopic dermatitis and healthy control subjects. J. Allergy Clin. Immunol. 2003, 111, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Taniuchi, S.; Hattori, K.; Yamamoto, A.; Sasai, M.; Hatano, Y.; Kojima, T.; Kobayashi, Y.; Iwamoto, H.; Yaeshima, T. Administration of Bifidobacterium to infants with atopic dermatitis: Changes in fecal microflora and clinical symptoms. J. Appl. Res. 2005, 5, 387–396. [Google Scholar]
- Van Der Aa, L.B.; Heymans, H.S.; Van Aalderen, W.M.; Sillevis Smitt, J.H.; Knol, J.; Ben Amor, K.; Goossens, D.A.; Sprikkelman, A.B. Effect of a new synbiotic mixture on atopic dermatitis in infants: A randomized-controlled trial. Clin. Exp. Allergy 2010, 40, 795–804. [Google Scholar] [CrossRef] [PubMed]
- Knol, J.; Scholtens, P.; Kafka, C.; Steenbakkers, J.; Gro, S.; Helm, K.; Klarczyk, M.; Schöpfer, H.; Böckler, H.-M.; Wells, J. Colon Microflora in Infants Fed Formula with Galacto- and Fructo-Oligosaccharides: More Like Breast-Fed Infants. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Van Der Aa, L.B.; Van Aalderen, W.M.C.; Heymans, H.S.A.; Henk Sillevis Smitt, J.; Nauta, A.J.; Knippels, L.M.J.; Ben Amor, K.; Sprikkelman, A.B. Synbiotics prevent asthma-like symptoms in infants with atopic dermatitis. Allergy Eur. J. Allergy Clin. Immunol. 2011, 66, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, T.; Sowa, M.; Nishimori, K.; Shimazu, S.; Yoshida, A.; Yamada, K.; Furukawa, F.; Nakagawa, T.; Yanagisawa, N.; Iwabuchi, N.; et al. Effects of Bifidobacterial Supplementation to Pregnant Women and Infants in the Prevention of Allergy Development in Infants and on Fecal Microbiota. Allergol. Int. 2014, 63, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, T.; Yokoyama, Y.; Nishio, H.; Ebata, T.; Sugawara, G.; Asahara, T.; Nomoto, K.; Nagino, M. Intraoperative Bacterial Translocation Detected by Bacterium-Specific Ribosomal RNA-Targeted Reverse-Transcriptase Polymerase Chain Reaction for the Mesenteric Lymph Node Strongly Predicts Postoperative Infectious Complications After Major Hepatectomy for. Ann. Surg. 2010, 252, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, T.; Asahara, T.; Yamataka, A.; Ogasawara, Y.; Lane, G.J.; Nomoto, K.; Nagata, S.; Yamashiro, Y. Intestinal microbiota in pediatric surgical cases administered Bifidobacterium breve: A randomized controlled trial. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Umenai, T.; Shime, N.; Asahara, T.; Nomoto, K.; Itoi, T. A pilot study of Bifidobacterium breve in neonates undergoing surgery for congenital heart disease. J. Intensive Care 2014, 2, 36. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, Y.; Hashizume, K.; Sugiyama, M.; Morotomi, M.; Yuki, N. Combination therapy with Bifidobacterium breve, Lactobacillus casei, and galactooligosaccharides dramatically improved the intestinal function in a girl with short bowel syndrome: A novel synbiotics therapy for intestinal failure. Dig. Dis. Sci. 2001, 46, 2010–2016. [Google Scholar] [CrossRef] [PubMed]
- Sherman, P.; Lichtman, S. Small Bowel Bacterial Overgrowth Syndrome. Dig. Dis. 1987, 5, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Bongaerts, G.P.A.; Tolboom, J.J.M.; Naber, A.H.J.; Sperl, W.J.K.; Severijnen, R.S.V.M.; Bakkeren, J.A.J.M.; Willems, J.L. Role of bacteria in the pathogenesis of short bowel syndrome-associated d-lactic acidemia. Microb. Pathog. 1997, 22, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, Y.; Sugiyama, M.; Hashizume, K.; Yuki, N.; Morotomi, M.; Tanaka, R. Experience of long-term synbiotic therapy in seven short bowel patients with refractory enterocolitis. J. Pediatr. Surg. 2004, 39, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, Y.; Iwanaka, T.; Sugiyama, M.; Komura, M.; Takahashi, T.; Yuki, N.; Morotomi, M.; Tanaka, R. Early use of probiotics is important therapy in infants with severe congenital anomaly. Pediatr. Int. 2010, 52, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Ding, H.; Wang, T.; Hooper, L.V.; Koh, G.Y.; Nagy, A.; Semenkovich, C.F.; Gordon, J.I. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl. Acad. Sci. USA 2004, 101, 15718–15723. [Google Scholar] [CrossRef] [PubMed]
- Klastersky, J. A review of chemoprophylaxis and therapy of bacterial infections in neutropenic patients. Diagn. Microbiol. Infect. Dis. 1989, 12, 201–207. [Google Scholar] [CrossRef]
- Resta-Lenert, S.; Barrett, K.E. Live probiotics protect intestinal epithelial cells from the effects of infection with enteroinvasive Escherichia coli (EIEC). Gut 2003, 52, 988–997. [Google Scholar] [CrossRef] [PubMed]
- Wada, M.; Nagata, S.; Saito, M.; Shimizu, T.; Yamashiro, Y.; Matsuki, T.; Asahara, T.; Nomoto, K. Effects of the enteral administration of Bifidobacterium breve on patients undergoing chemotherapy for pediatric malignancies. Support. Care Cancer 2010, 18, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Minami, J.I.; Kondo, S.; Yanagisawa, N.; Odamaki, T.; Xiao, J.Z.; Abe, F.; Nakajima, S.; Hamamoto, Y.; Saitoh, S.; Shimoda, T. Oral administration of Bifidobacterium breve B-3 modifies metabolic functions in adults with obese tendencies in a randomised controlled trial. J. Nutr. Sci. 2015, 4, e17. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Matsumoto, S.; Ohashi, Y.; Imaoka, A.; Setoyama, H.; Umesaki, Y.; Tanaka, R.; Otani, T. Beneficial effects of probiotic Bifidobacterium and galacto-oligosaccharide in patients with ulcerative colitis: A randomized controlled study. Digestion 2011, 84, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Matts, S.G. The value of rectal biopsy in the diagnosis of ulcerative colitis. Q. J. Med. 1961, 30, 393–407. [Google Scholar] [PubMed]
- Carlson, M.; Raab, Y.; Sevéus, L.; Xu, S.; Hällgren, R.; Venge, P. Human neutrophil lipocalin is a unique marker of neutrophil inflammation in ulcerative colitis and proctitis. Gut 2002, 50, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Kano, M.; Masuoka, N.; Kaga, C.; Sugimoto, S.; Iizuka, R.; Manabe, K.; Sone, T.; Oeda, K.; Nonaka, C.; Miazaki, K.; et al. Consecutive Intake of Fermented Milk Containing Bifidobacterium breve Strain Yakult and Galacto-oligosaccharides Benefits Skin Condition in Healthy Adult Women. Biosci. Microbiota Food Health 2013, 21, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Hitomi, K.; van Vlijmen-Willems, I.M.J.J.; de Jongh, G.J.; Yamamoto, K.; Nishi, K.; Watts, C.; Reinheckel, T.; Schalkwijk, J.; Zeeuwen, P.L.J.M. Cystatin M/E Is a High Affinity Inhibitor of Cathepsin V and Cathepsin L by a Reactive Site That Is Distinct from the Legumain-binding Site: A novel clue for the role of cystatin M/E in epidermal cornification. J. Biol. Chem. 2006, 281, 15893–15899. [Google Scholar] [CrossRef] [PubMed]
- Brigidi, P.; Vitali, B.; Swennen, E.; Bazzocchi, G.; Matteuzzi, D. Effects of probiotic administration upon the composition and enzymatic activity of human fecal microbiota in patients with irritable bowel syndrome or functional diarrhea. Res. Microbiol. 2001, 152, 735–741. [Google Scholar] [CrossRef]
- Mobley, H.L.; Hausinger, R.P. Microbial ureases: Significance, regulation, and molecular characterization. Microbiol. Rev. 1989, 53, 85–108. [Google Scholar] [PubMed]
- Pronio, A.; Montesani, C.; Butteroni, C.; Vecchione, S.; Mumolo, G.; Vestri, A.; Vitolo, D.; Boirivant, M. Probiotic administration in patients with ileal pouch-anal anastomosis for ulcerative colitis is associated with expansion of mucosal regulatory cells. Inflamm. Bowel Dis. 2008, 14, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Kühbacher, T.; Ott, S.J.; Helwig, U.; Mimura, T.; Rizzello, F.; Kleessen, B.; Gionchetti, P.; Blaut, M.; Campieri, M.; Fölsch, U.R.; et al. Bacterial and fungal microbiota in relation to probiotic therapy (VSL#3) in pouchitis. Gut 2006, 55, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Bibiloni, R.; Fedorak, R.N.; Tannock, G.W.; Madsen, K.L.; Gionchetti, P.; Campieri, M.; De Simone, C.; Sartor, R.B. VSL#3 probiotic-mixture induces remission in patients with active ulcerative colitis. Am. J. Gastroenterol. 2005, 100, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Venturi, A.; Gionchetti, P.; Rizzello, F.; Johansson, R.; Zucconi, E.; Brigidi, P.; Matteuzzi, D.; Campieri, M. Impact on the composition of the faecal flora by a new probiotic preparation: Preliminary data on maintenance treatment of patients with ulcerative colitis. Aliment. Pharmacol. Ther. 1999, 13, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Brandimarte, G.; Giorgetti, G.M.; Forti, G.; Modeo, M.E.; Gigliobianco, A. Low-dose balsalazide plus a high-potency probiotic preparation is more effective than balsalazide alone or mesalazine in the treatment of acute mild-to-moderate ulcerative colitis. Med. Sci. Monit. 2004, 10, PI126–PI131. [Google Scholar] [PubMed]
- Tursi, A.; Brandimarte, G.; Papa, A.; Giglio, A.; Elisei, W.; Giorgetti, G.M.; Forti, G.; Morini, S.; Hassan, C.; Pistoia, M.A.; et al. Treatment of relapsing mild-to-moderate ulcerative colitis with the probiotic VSL3 as adjunctive to a standard pharmaceutical treatment: A double-blind, randomized, placebo-controlled study. Am. J. Gastroenterol. 2010, 105, 2218–2227. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.K.; Yang, C.; Song, G.H.; Wong, J.; Ho, K.Y. Melatonin Regulation as a Possible Mechanism for Probiotic (VSL#3) in Irritable Bowel Syndrome: A Randomized Double-Blinded Placebo Study. Dig. Dis. Sci. 2014, 60, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Camilleri, M.; Mckinzie, S. A Randomized Controlled Trial of a Probiotic, VSL#3, on Gut Transit and Symptoms in Diarrhoea-Predominant Irritable Bowel Syndrome. Aliment. Pharmacol. Ther. 2003, 17, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Michail, S.; Kenche, H. Gut Microbiota is Not Modified by Randomized, Double-Blind, Placebo-Controlled Trial of VSL#3 in Diarrhea-Predominant Irritable Bowel Syndrome. Probiot. Antimicrob. Proteins 2011, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
| B. breve Strains | Reported Effect(s) | References |
|---|---|---|
| B. breve B632 | Strong antimicrobial activity against pathogens, stimulation of mitochondrial dehydrogenase activity of macrophages, stimulation of proinflammatory cytokines production in in vitro study | [62] |
| B. breve BR03 | Inhibition of the growth of 4 E. coli biotypes in in vitro study | [64] |
| B. breve B632 + B. breve BR03 | Reduction of total faecal coliforms in healthy children | [65] |
| Reduction of pro-inflammatory TNF-α in blood samples of celiac children | [145] | |
| Reduction of minutes of daily crying in healthy infants | [129] | |
| Restoration of the healthy percentage of main gut microbial components in celiac children | [146] | |
| Improvement of glucose metabolism and weight management in obese children | [155] | |
| Reduction of daily vomit frequency, daily evacuation, improved stool consistency, protection against developing metabolic disturbance in healthy infants | [130] | |
| Modulation of faecal SCFAs profile in celiac children | [147] | |
| B. breve Yakult (BBG-01) | Anti-infective activity against Shiga-toxin-producing E. coli in mice model | [69] |
| Reduction of febrile episodes and use of intravenous antibiotics in cancer paediatric subjects | [173] | |
| Improvement of composition and metabolic activity of gut microbiota and reduction of incidence of bacteria in blood in paediatric surgery subjects | [163] | |
| Increased defecation frequency, improvement of stool consistency, frequency episodes of faecal incontinence and abdominal pain in constipated children | [133] | |
| Stimulation of anti-inflammatory IL-10-producing CD4+T cells in mice model | [83] | |
| Improvement of composition and metabolic activity of gut microbiota in paediatric surgery infants with congenital heart disease | [164] | |
| B. breve YIT4064 | Stimulation of anti-influenza virus hemagglutinin IgA production by Peyer’s patch cells in mice model | [75] |
| Stimulation of antigen-specific IgG production against pathogenic antigens in mice model | [74] | |
| B. breve UCC2003 | Reduction of Citrobacter rodentium gut colonization in mice model | [76] |
| B. breve NCC2950 | Induction of REGIII-γ expression in mice model and REGIII-α in in vitro study | [78] |
| B. breve MRx0004 | Reduction of pro-inflammatory cytokines and lung neutrophil and eosinophil infiltration in severe asthma mice model | [79] |
| B. breve M-16V | Improvement of allergic symptoms associated to cow’s milk hypersensitivity in infants | [157] |
| Immunomodulation activity by increasing TGF-β1 in preterm infants | [122] | |
| Reduction of infections and mortality for NEC in extremely and very low birth weight infants | [118] | |
| Reduction of faecal butyric acid in extremely and very low birth weight infants | [121] | |
| Reduction of total IgE, OVA-specific IgE and OVA-specific IgG in mice model | [80] | |
| Protection against developing of whey allergy symptoms in model mice | [81] | |
| Reduction of infections and sepsis incidence in extremely and very low birth weight infants | [124] | |
| Improvement of composition and metabolic activity of gut microbiota in infants with atopic dermatitis | [158] | |
| Reduction of asthma-like symptoms prevalence and asthma medication use prevalence in infants with atopic dermatitis | [160] | |
| Shifted gut microbiota towards a healthy profile in preterm infants | [111] | |
| Low incidence of NEC (≥stage II) in very low birth weight infants | [112] | |
| Partially protection against developing skin reaction due to cow’s milk allergy, increased cecal content of butyrate and propionate and increased antimicrobial IL-22 expression in mice model | [82] | |
| B. breve B-3 | Suppression of epididymal fat and body weight gain in mice model with diet-induced obesity | [84,85] |
| B. breve 1205 | Amelioration of anxiety condition and general metabolism in mice model | [88] |
| B. breve A1 | Prevention of cognitive decline in Alzheimer disease and reduction of neural inflammation in mice model | [89] |
| B. breve NCIMB 702258 | Increased CLA isomer (c9, t11), EPA and DHA in adipose tissue and reduced proinflammatory cytokines in mice model | [96] |
| B. breve YIT4010 | Reduced abdominal symptoms and improved weight gain in preterm infants | [109] |
| Establishment of beneficial gut microbiota and prevention of infections in preterm infant | [110] |
| B. breve Strains | Probiotic Mixture | Reported Effect(s) | References |
|---|---|---|---|
| B. breve M-16V | B. breve M-16V B. longum BB536 | Reduction of developing eczema and atopic dermatitis in infants | [161] |
| B. breve M-16V B. infantis M-63 B. longum BB536 | Reduction of abdominal pain prevalence and frequency, improvement of quality of life in IBS children | [134] | |
| B. breve Yakult (BBG-01) | B. breve Yakult L. casei Shirota | Improvement of composition and metabolic activity of gut microbiota and of overall health status in infants with short bowel syndrome | [165,168] |
| Prevention of enterocolitis, improvement of body weight and clinical course in infants with congenital disorders | [169] | ||
| B. breve Yakult L. casei | Reduction of NEC incidence and improvement of intestinal motility in infants | [125] | |
| B. breve C50 | B. breve C50 S. thermophilus 065 | Reduction of number of dehydration cases and medical consultation cases in children exposed to risk of developing acute diarrhoea | [144] |
| B. breve DSM 24732 | VSL#3 | Reduction of stool frequency and improving of stool consistency in children with acute rotavirus diarrhoea | [142] |
| Manifestation of high rate of remission and low incidence of relapse in UC children | [139] | ||
| Improvement of symptoms, severity and frequency of abdominal pain and bloating and family assessment of life disruption in IBS children | [138] | ||
| Reduction of the risk of suspected sepsis in most vulnerable very low birth weight infants | [144] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bozzi Cionci, N.; Baffoni, L.; Gaggìa, F.; Di Gioia, D. Therapeutic Microbiology: The Role of Bifidobacterium breve as Food Supplement for the Prevention/Treatment of Paediatric Diseases. Nutrients 2018, 10, 1723. https://doi.org/10.3390/nu10111723
Bozzi Cionci N, Baffoni L, Gaggìa F, Di Gioia D. Therapeutic Microbiology: The Role of Bifidobacterium breve as Food Supplement for the Prevention/Treatment of Paediatric Diseases. Nutrients. 2018; 10(11):1723. https://doi.org/10.3390/nu10111723
Chicago/Turabian StyleBozzi Cionci, Nicole, Loredana Baffoni, Francesca Gaggìa, and Diana Di Gioia. 2018. "Therapeutic Microbiology: The Role of Bifidobacterium breve as Food Supplement for the Prevention/Treatment of Paediatric Diseases" Nutrients 10, no. 11: 1723. https://doi.org/10.3390/nu10111723
APA StyleBozzi Cionci, N., Baffoni, L., Gaggìa, F., & Di Gioia, D. (2018). Therapeutic Microbiology: The Role of Bifidobacterium breve as Food Supplement for the Prevention/Treatment of Paediatric Diseases. Nutrients, 10(11), 1723. https://doi.org/10.3390/nu10111723






