Bitter, Sweet, Salty, Sour and Umami Taste Perception Decreases with Age: Sex-Specific Analysis, Modulation by Genetic Variants and Taste-Preference Associations in 18 to 80 Year-Old Subjects
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Demographic, Anthropometric, Biochemical, Clinical and Lifestyle Variables
2.3. Taste Preference Assessment
2.4. Taste Perception Tests
2.5. DNA Genotyping
2.6. Statistical Analysis
3. Results
3.1. General Characteristics of the Population
3.2. Descriptive of the Taste Perception Tests
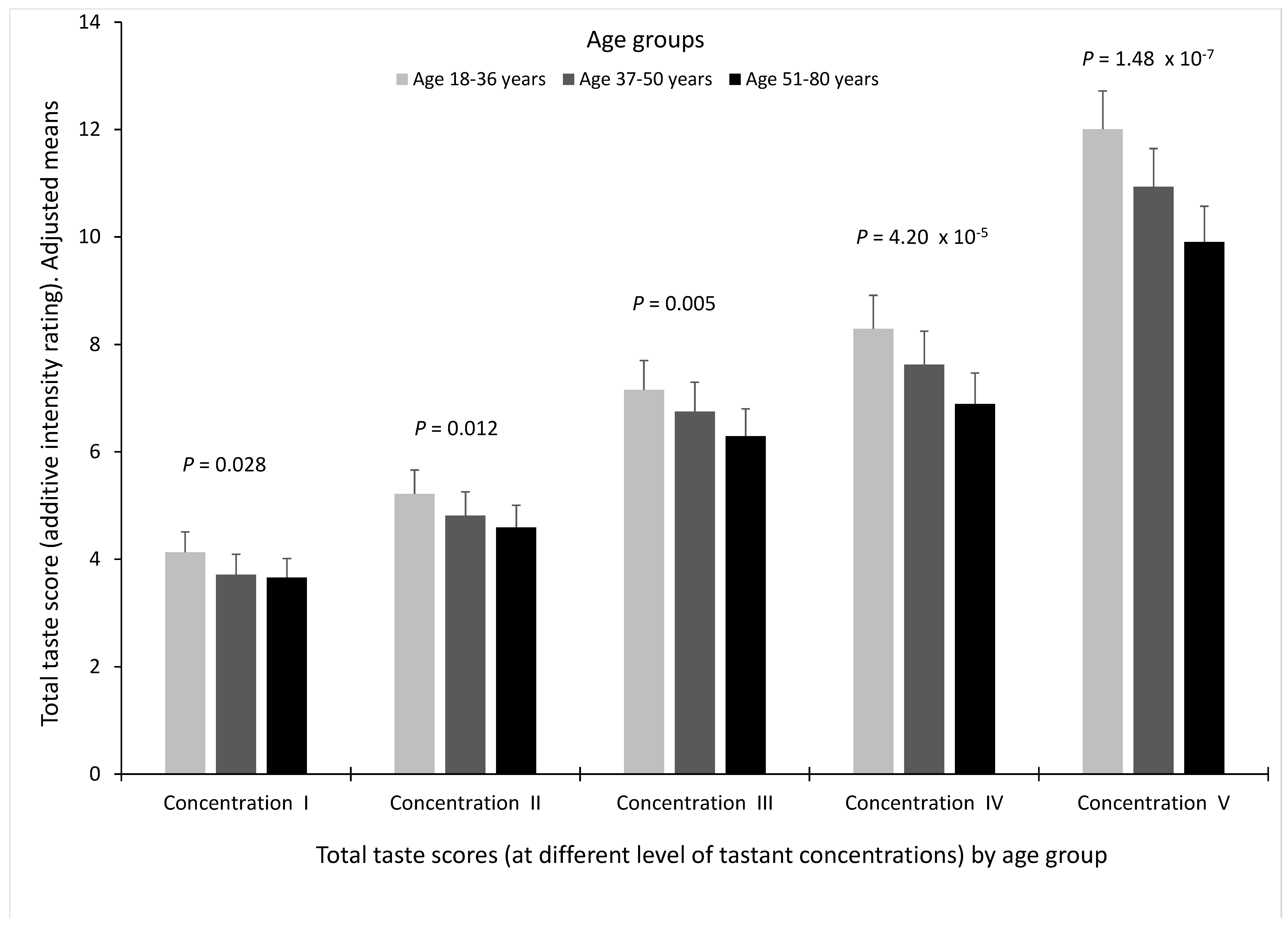
3.3. Associations between Age and Total Taste Score for the Different Concentrations of Tastants (I to V)
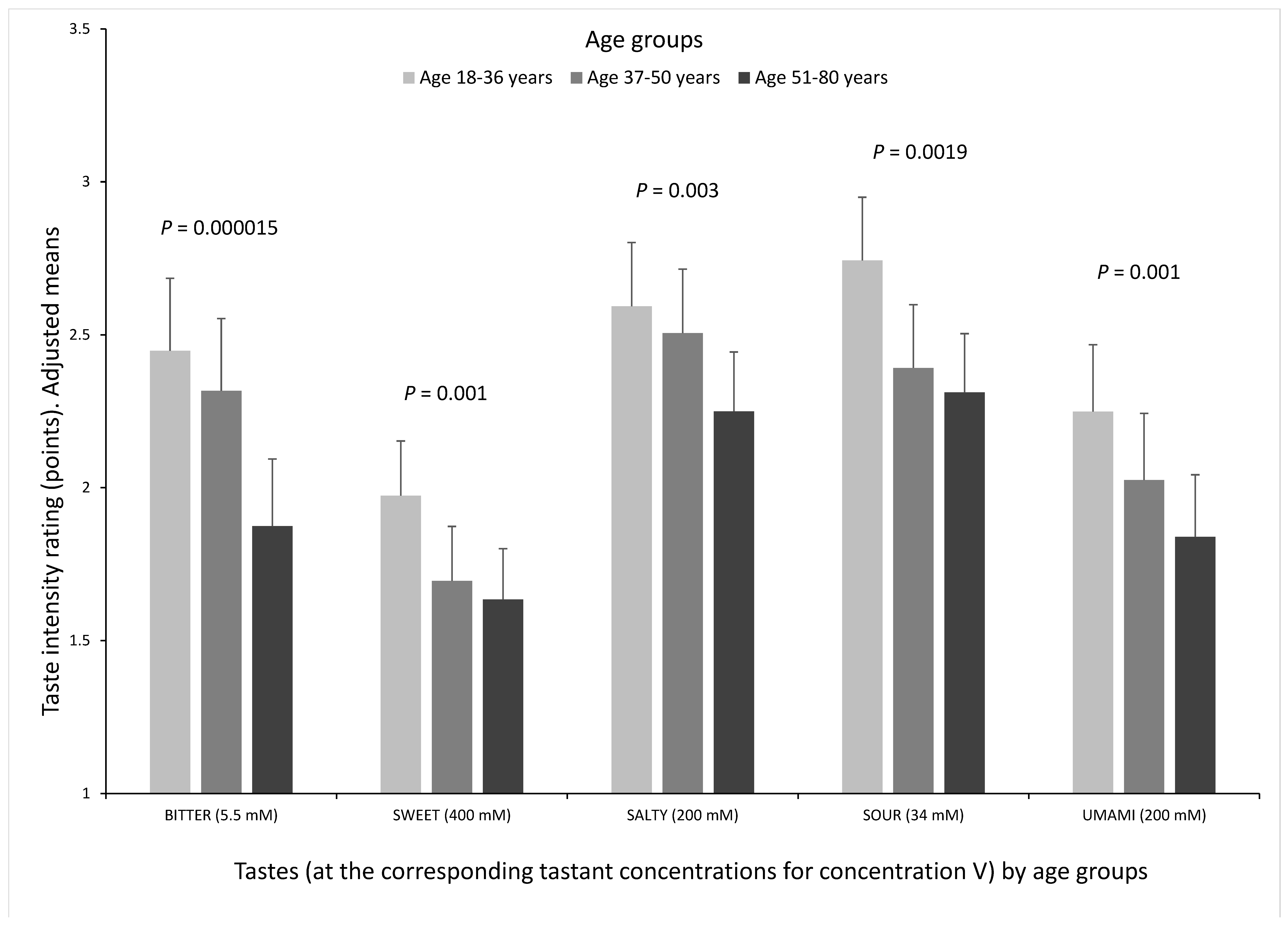
3.4. Correlation between Perception of the Different Tastes and Association between Age and the Five Tastes at Concentration V
3.5. Influence of Sex in Taste Perception
3.6. Taste Preferences and Association with Taste Perception
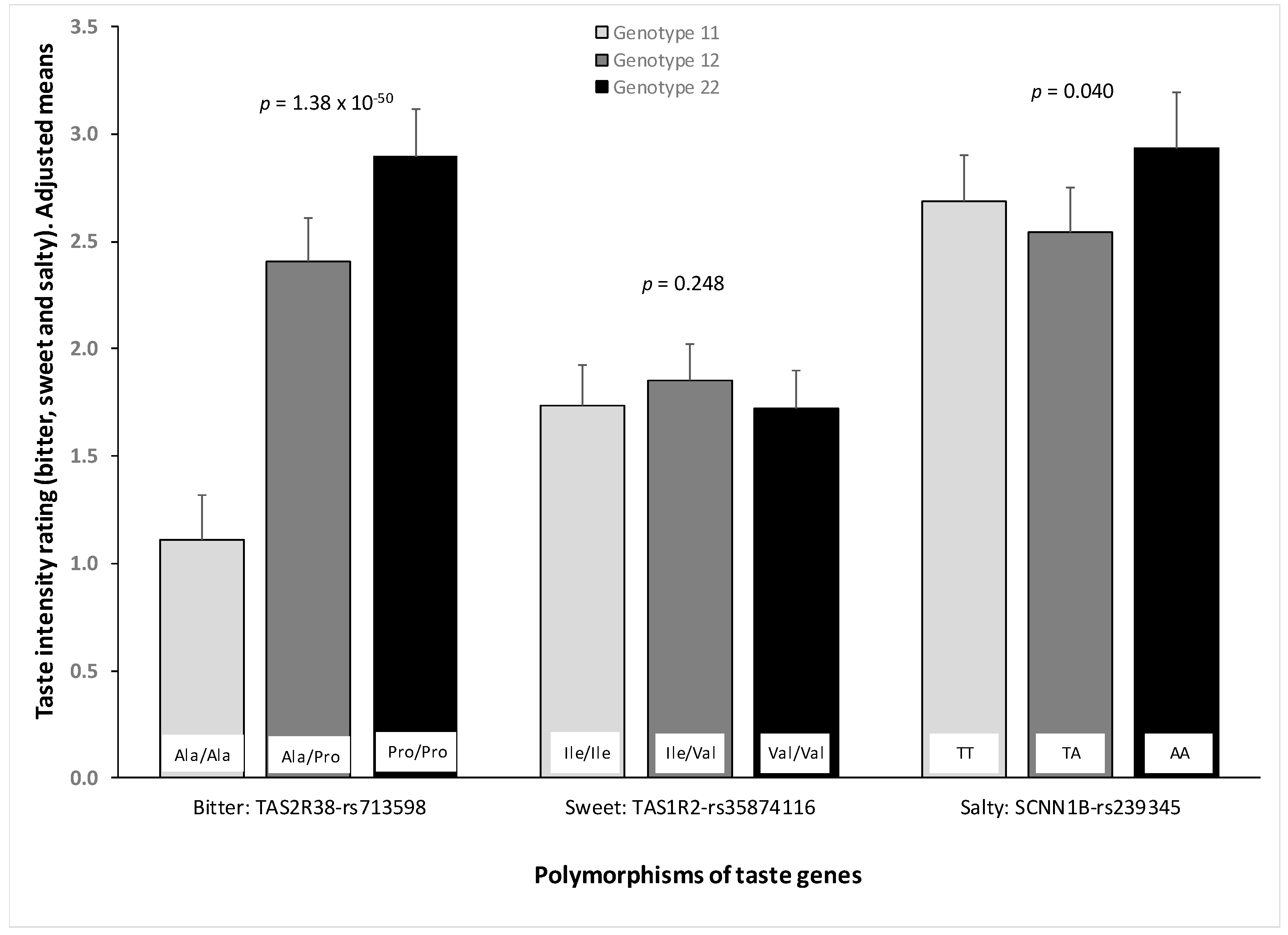
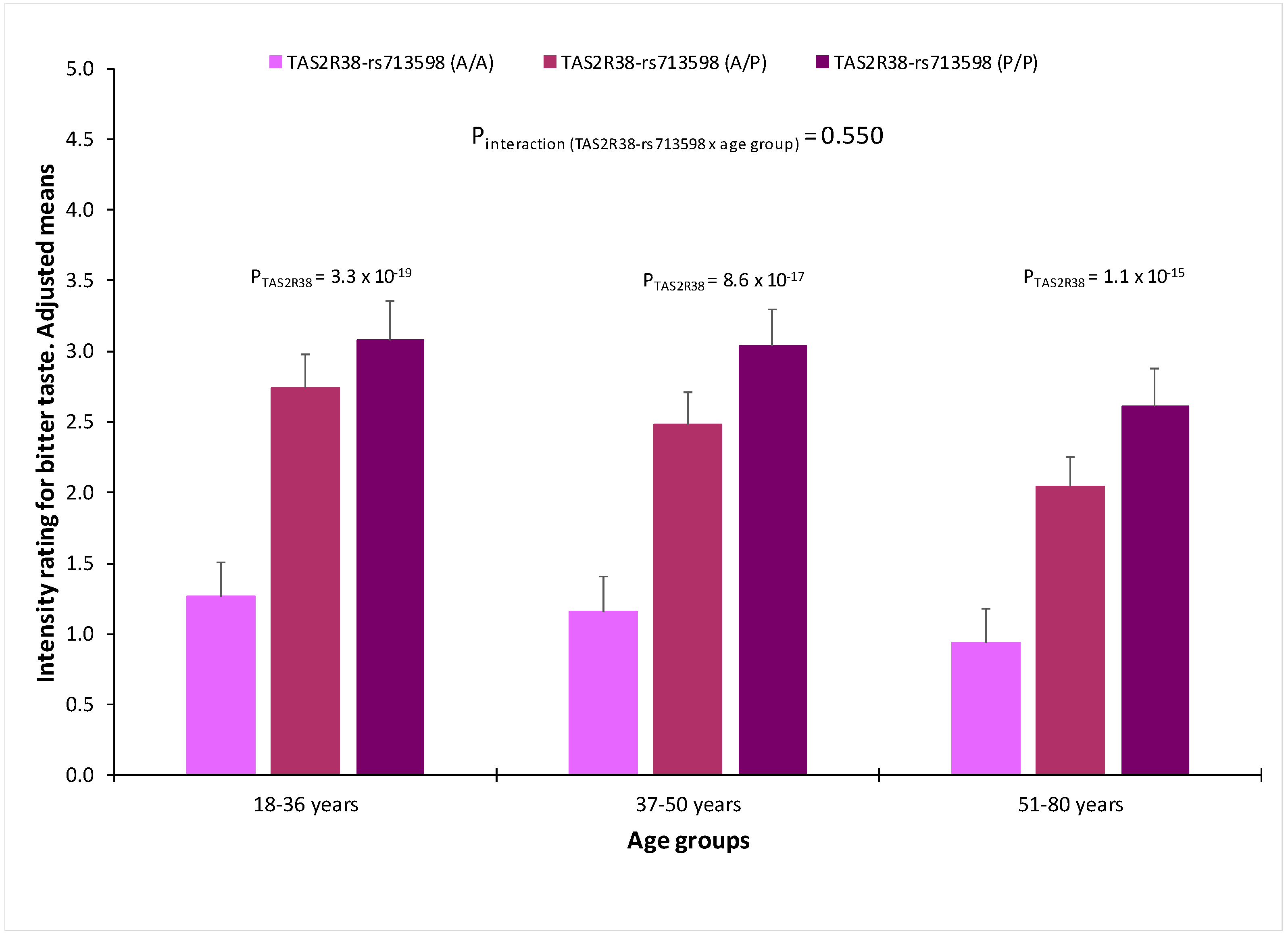
3.7. Association between Genetic Polymorphisms and Taste Perception
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Mathers, C.D.; Stevens, G.A.; Boerma, T.; White, R.A.; Tobias, M.I. Causes of international increases in older age life expectancy. Lancet 2015, 385, 540–548. [Google Scholar] [CrossRef]
- Johnson, N.B.; Hayes, L.D.; Brown, K.; Hoo, E.C.; Ethier, K.A.; Centers for Disease Control and Prevention (CDC). CDC National Health Report: Leading causes of morbidity and mortality and associated behavioral risk and protective factors—United States, 2005–2013. MMWR Suppl. 2014, 63, 3–27. [Google Scholar] [PubMed]
- The JA: EHLEIS Team; Robine, J.-M.; Cambois, E.; Nusselder, W.; Jeune, B.; Oyen, H.V.; Jagger, C. The joint action on healthy life years (JA: EHLEIS). Arch. Public Health 2013, 71, 2. [Google Scholar] [CrossRef] [PubMed]
- Alcañiz, M.; Solé-Auró, A. Feeling good in old age: Factors explaining health-related quality of life. Health Qual. Life Outcomes 2018, 16, 48. [Google Scholar] [CrossRef] [PubMed]
- Beard, J.R.; Officer, A.; de Carvalho, I.A.; Sadana, R.; Pot, A.M.; Michel, J.-P.; Lloyd-Sherlock, P.; Epping-Jordan, J.E.; Peeters, G.M.E.E.G.; Mahanani, W.R.; et al. The World report on ageing and health: A policy framework for healthy ageing. Lancet 2016, 387, 2145–2154. [Google Scholar] [CrossRef]
- Anisimov, V.N.; Sidorenko, A.V. It is necessary to establish an International Agency for Research on Aging. Aging 2018, 10, 863–867. [Google Scholar] [CrossRef] [PubMed]
- Sotos-Prieto, M.; Bhupathiraju, S.N.; Mattei, J.; Fung, T.T.; Li, Y.; Pan, A.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Association of Changes in Diet Quality with Total and Cause-Specific Mortality. N. Engl. J. Med. 2017, 377, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Govindaraju, T.; Sahle, B.; McCaffrey, T.; McNeil, J.; Owen, A. Dietary Patterns and Quality of Life in Older Adults: A Systematic Review. Nutrients 2018, 10, 971. [Google Scholar] [CrossRef] [PubMed]
- Gabrovec, B.; Veninšek, G.; Samaniego, L.L.; Carriazo, A.M.; Antoniadou, E.; Jelenc, M. The role of nutrition in ageing: A narrative review from the perspective of the European joint action on frailty—ADVANTAGE JA. Eur. J. Intern. Med. 2018, 56, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Aspry, K.E.; Van Horn, L.; Carson, J.A.S.; Wylie-Rosett, J.; Kushner, R.F.; Lichtenstein, A.H.; Devries, S.; Freeman, A.M.; Crawford, A.; Kris-Etherton, P. Medical Nutrition Education, Training, and Competencies to Advance Guideline-Based Diet Counseling by Physicians: A Science Advisory From the American Heart Association. Circulation 2018, 137, e821–e841. [Google Scholar] [CrossRef] [PubMed]
- Ordovas, J.M.; Ferguson, L.R.; Tai, E.S.; Mathers, J.C. Personalised nutrition and health. BMJ 2018. [Google Scholar] [CrossRef] [PubMed]
- Fuellen, G.; Schofield, P.; Flatt, T.; Schulz, R.-J.; Boege, F.; Kraft, K.; Rimbach, G.; Ibrahim, S.; Tietz, A.; Schmidt, C.; et al. Living Long and Well: Prospects for a Personalized Approach to the Medicine of Ageing. Gerontology 2016, 62, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Granic, A.; Mendonça, N.; Hill, T.; Jagger, C.; Stevenson, E.; Mathers, J.; Sayer, A. Nutrition in the Very Old. Nutrients 2018, 10, 269. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, Y.; Won, C.W.; Lee, K.E.; Chon, D. Nutritional Status and Frailty in Community-Dwelling Older Korean Adults: The Korean Frailty and Aging Cohort Study. J. Nutr. Health Aging 2018, 22, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Spirgienė, L.; Damulevičienė, G.; Tomkevičiūtė, J.; Riklikienė, O. Nutritional status of rural community-dwelling older people and changes after following nutritional recommendations: Malnutrition in community-dwelling older people. Int. J. Nurs. Pract. 2018, 31, e12688. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; McKeague, M. Dietary modulation of mitochondrial DNA damage: Implications in aging and associated diseases. J. Nutr. Biochem. 2019, 63, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Assmann, K.; Ruhunuhewa, I.; Adjibade, M.; Li, Z.; Varraso, R.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. The Mediating Role of Overweight and Obesity in the Prospective Association between Overall Dietary Quality and Healthy Aging. Nutrients 2018, 10, 515. [Google Scholar] [CrossRef] [PubMed]
- Leung, C.W.; Fung, T.T.; McEvoy, C.T.; Lin, J.; Epel, E.S. Diet Quality Indices and Leukocyte Telomere Length among Healthy US Adults: Data from the National Health and Nutrition Examination Survey, 1999–2002. Am. J. Epidemiol. 2018, 187, 2192–2201. [Google Scholar] [CrossRef] [PubMed]
- Maegawa, S.; Lu, Y.; Tahara, T.; Lee, J.T.; Madzo, J.; Liang, S.; Jelinek, J.; Colman, R.J.; Issa, J.-P.J. Caloric restriction delays age-related methylation drift. Nat. Commun. 2017, 8, 539. [Google Scholar] [CrossRef] [PubMed]
- Gibson, A.; Sainsbury, A. Strategies to Improve Adherence to Dietary Weight Loss Interventions in Research and Real-World Settings. Behav. Sci. 2017, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Stok, F.; Renner, B.; Clarys, P.; Lien, N.; Lakerveld, J.; Deliens, T. Understanding Eating Behavior during the Transition from Adolescence to Young Adulthood: A Literature Review and Perspective on Future Research Directions. Nutrients 2018, 10, 667. [Google Scholar] [CrossRef] [PubMed]
- Kabir, A.; Miah, S.; Islam, A. Factors influencing eating behavior and dietary intake among resident students in a public university in Bangladesh: A qualitative study. PLoS ONE 2018, 13, e0198801. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Shin, Y.; Kwon, O.; Kim, Y. Association of Sensory Liking for Fat with Dietary Intake and Metabolic Syndrome in Korean Adults. Nutrients 2018, 10, 877. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A. Ontogeny of taste preferences: Basic biology and implications for health. Am. J. Clin. Nutr. 2014, 99, 704S–711S. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.E. Taste and flavour: Their importance in food choice and acceptance. Proc. Nutr. Soc. 1998, 57, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A. Genetic and Environmental Determinants of Bitter Perception and Sweet Preferences. Pediatrics 2005, 115, e216–e222. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Bobowski, N.K. The sweetness and bitterness of childhood: Insights from basic research on taste preferences. Physiol. Behav. 2015, 152, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.E.; Cruickshanks, K.J.; Schubert, C.R.; Pinto, A.; Klein, B.E.K.; Klein, R.; Nieto, F.J.; Pankow, J.S.; Huang, G.-H.; Snyder, D.J. Taste intensity in the Beaver Dam Offspring Study: Taste Intensity-Beaver Dam Offspring Study. Laryngoscope 2013, 123, 1399–1404. [Google Scholar] [CrossRef] [PubMed]
- Shu-Fen, C.L.; Forde, C.G.; Tey, S.L.; Henry, C.J. Taste sensitivities and diet of Chinese and Indians in Singapore. Asia Pacif. J. Clin. Nutr. 2018, 27, 681–685. [Google Scholar]
- Chamoun, E.; Carroll, N.; Duizer, L.; Qi, W.; Feng, Z.; Darlington, G.; Duncan, A.; Haines, J.; Ma, D.; The Guelph Family Health Study. The Relationship between Single Nucleotide Polymorphisms in Taste Receptor Genes, Taste Function and Dietary Intake in Preschool-Aged Children and Adults in the Guelph Family Health Study. Nutrients 2018, 10, 990. [Google Scholar] [CrossRef] [PubMed]
- Mielby, L.; Wang, Q.; Jensen, S.; Bertelsen, A.; Kidmose, U.; Spence, C.; Byrne, D. See, Feel, Taste: The Influence of Receptacle Colour and Weight on the Evaluation of Flavoured Carbonated Beverages. Foods 2018, 7, 119. [Google Scholar] [CrossRef] [PubMed]
- Han, P.; Bagenna, B.; Fu, M. The sweet taste signalling pathways in the oral cavity and the gastrointestinal tract affect human appetite and food intake: A review. Int. J. Food Sci. Nutr. 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Garcia, J.C.; Alcaide, J.; Santiago-Fernandez, C.; Roca-Rodriguez, M.; Aguera, Z.; Baños, R.; Botella, C.; de la Torre, R.; Fernandez-Real, J.M.; Fruhbeck, G.; et al. An increase in visceral fat is associated with a decrease in the taste and olfactory capacity. PLoS ONE 2017, 12, e0171204. [Google Scholar]
- Kaufman, A.; Choo, E.; Koh, A.; Dando, R. Inflammation arising from obesity reduces taste bud abundance and inhibits renewal. PLoS Biol. 2018, 16, e2001959. [Google Scholar] [CrossRef] [PubMed]
- Rawal, S.; Huedo-Medina, T.B.; Hoffman, H.J.; Swede, H.; Duffy, V.B. Structural equation modeling of associations among taste-related risk factors, taste functioning, and adiposity: Taste Risk Factors, Taste Function, and Adiposity. Obesity 2017, 25, 781–787. [Google Scholar] [CrossRef] [PubMed]
- Schubert, C.R.; Cruickshanks, K.J.; Fischer, M.E.; Chen, Y.; Klein, B.E.K.; Klein, R.; Pinto, A.A. Sensory Impairments and Cognitive Function in Middle-Aged Adults. J. Gerontol. Ser. A 2017, 72, 1087–1090. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.M.; Kern, D.W.; Wroblewski, K.E.; Chen, R.C.; Schumm, L.P.; McClintock, M.K. Sensory Function: Insights From Wave 2 of the National Social Life, Health, and Aging Project. J. Gerontol. Ser. B 2014, 69, S144–S153. [Google Scholar] [CrossRef] [PubMed]
- Sergi, G.; Bano, G.; Pizzato, S.; Veronese, N.; Manzato, E. Taste loss in the elderly: Possible implications for dietary habits. Crit. Rev. Food Sci. Nutr. 2017, 57, 3684–3689. [Google Scholar] [CrossRef] [PubMed]
- Bachmanov, A.A.; Bosak, N.P.; Glendinning, J.I.; Inoue, M.; Li, X.; Manita, S.; McCaughey, S.A.; Murata, Y.; Reed, D.R.; Tordoff, M.G.; et al. Genetics of Amino Acid Taste and Appetite. Adv. Nutr. Int. Rev. J. 2016, 7, 806S–822S. [Google Scholar] [CrossRef] [PubMed]
- Chamoun, E.; Mutch, D.M.; Allen-Vercoe, E.; Buchholz, A.C.; Duncan, A.M.; Spriet, L.L.; Haines, J.; Ma, D.W.L.; on behalf of the Guelph Family Health Study. A review of the associations between single nucleotide polymorphisms in taste receptors, eating behaviors, and health. Crit. Rev. Food Sci. Nutr. 2018, 58, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Jaggupilli, A.; Howard, R.; Upadhyaya, J.D.; Bhullar, R.P.; Chelikani, P. Bitter taste receptors: Novel insights into the biochemistry and pharmacology. Int. J. Biochem. Cell Biol. 2016, 77, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Behrens, M.; Gunn, H.C.; Ramos, P.C.M.; Meyerhof, W.; Wooding, S.P. Genetic, Functional, and Phenotypic Diversity in TAS2R38-Mediated Bitter Taste Perception. Chem. Sens. 2013, 38, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Khataan, N.H.; Stewart, L.; Brenner, D.M.; Cornelis, M.C.; El-Sohemy, A. TAS2R38 Genotypes and Phenylthiocarbamide Bitter Taste Perception in a Population of Young Adults. J. Nutrigenet. Nutrigenom. 2009, 2, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Tepper, B.J.; Koelliker, Y.; Zhao, L.; Ullrich, N.V.; Lanzara, C.; D’Adamo, P.; Ferrara, A.; Ulivi, S.; Esposito, L.; Gasparini, P. Variation in the Bitter-taste Receptor Gene TAS2R38, and Adiposity in a Genetically Isolated Population in Southern Italy. Obesity 2008, 16, 2289–2295. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.L.; Adise, S. Variation in the Ability to Taste Bitter Thiourea Compounds: Implications for Food Acceptance, Dietary Intake, and Obesity Risk in Children. Annu. Rev. Nutr. 2016, 36, 157–182. [Google Scholar] [CrossRef] [PubMed]
- Habberstad, C.; Drake, I.; Sonestedt, E. Variation in the Sweet Taste Receptor Gene and Dietary Intake in a Swedish Middle-Aged Population. Front. Endocrinol. 2017, 8, 348. [Google Scholar] [CrossRef] [PubMed]
- Pilic, L.; Mavrommatis, Y. Genetic predisposition to salt-sensitive normotension and its effects on salt taste perception and intake. Br. J. Nutr. 2018, 120, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Bobowski, N.K.; Reed, D.R. The development of sweet taste: From biology to hedonics. Rev. Endocr. Metab. Disord. 2016, 17, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Gu, D.; He, J.; Rao, D.C.; Hixson, J.E.; Chen, J.; Li, J.; Huang, J.; Wu, X.; Rice, T.K.; et al. Resequencing Epithelial Sodium Channel Genes Identifies Rare Variants Associated With Blood Pressure Salt-Sensitivity: The GenSalt Study. Am. J. Hypertens. 2018, 31, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Lopez, O.; Panduro, A.; Martinez-Lopez, E.; Roman, S. Sweet Taste Receptor TAS1R2 Polymorphism (Val191Val) Is Associated with a Higher Carbohydrate Intake and Hypertriglyceridemia among the Population of West Mexico. Nutrients 2016, 8, 101. [Google Scholar] [CrossRef] [PubMed]
- Dias, A.G.; Eny, K.M.; Cockburn, M.; Chiu, W.; Nielsen, D.E.; Duizer, L.; El-Sohemy, A. Variation in the TAS1R2 Gene, Sweet Taste Perception and Intake of Sugars. J. Nutrigenet. Nutrigenom. 2015, 8, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Heid, I.M.; Huth, C.; Loos, R.J.F.; Kronenberg, F.; Adamkova, V.; Anand, S.S.; Ardlie, K.; Biebermann, H.; Bjerregaard, P.; Boeing, H.; et al. Meta-Analysis of the INSIG2 Association with Obesity Including 74,345 Individuals: Does Heterogeneity of Estimates Relate to Study Design? PLoS Genet. 2009, 5, e1000694. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Guillén, M.; Sáiz, C.; Portolés, O.; Sabater, A.; Folch, J.; Ordovas, J.M. Associations of LPL and APOC3 gene polymorphisms on plasma lipids in a Mediterranean population: Interaction with tobacco smoking and the APOE locus. J. Lipid Res. 2002, 43, 416–427. [Google Scholar] [PubMed]
- Zhao, L.; Kirkmeyer, S.V.; Tepper, B.J. A paper screening test to assess genetic taste sensitivity to 6-n-propylthiouracil. Physiol. Behav. 2003, 78, 625–633. [Google Scholar] [CrossRef]
- Bufe, B.; Breslin, P.A.S.; Kuhn, C.; Reed, D.R.; Tharp, C.D.; Slack, J.P.; Kim, U.-K.; Drayna, D.; Meyerhof, W. The Molecular Basis of Individual Differences in Phenylthiocarbamide and Propylthiouracil Bitterness Perception. Curr. Biol. 2005, 15, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.-Y.; Alarcon, S.; Tharp, A.; Ahmed, O.M.; Estrella, N.L.; Greene, T.A.; Rucker, J.; Breslin, P.A. Perceptual variation in umami taste and polymorphisms in TAS1R taste receptor genes. Am. J. Clin. Nutr. 2009, 90, 770S–779S. [Google Scholar] [CrossRef] [PubMed]
- Da Conceicao Neta, E.R.; Johanningsmeier, S.D.; McFeeters, R.F. The Chemistry and Physiology of Sour Taste? A Review. J. Food Sci. 2007, 72, R33–R38. [Google Scholar] [CrossRef] [PubMed]
- Fikentscher, R.; Roseburg, B.; Spinar, H.; BruchmüLler, W. Loss of taste in the elderly: Sex differences. Clin. Otolaryngol. 1977, 2, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Guido, D.; Perna, S.; Carrai, M.; Barale, R.; Grassi, M.; Rondanelli, M. Multidimensional evaluation of endogenous and health factors affecting food preferences, taste and smell perception. J. Nutr. Health Aging 2016, 20, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Yoshinaka, M.; Ikebe, K.; Uota, M.; Ogawa, T.; Okada, T.; Inomata, C.; Takeshita, H.; Mihara, Y.; Gondo, Y.; Masui, Y.; et al. Age and sex differences in the taste sensitivity of young adult, young-old and old-old Japanese: Age and sex differences in taste sense. Geriatr. Gerontol. Int. 2016, 16, 1281–1288. [Google Scholar] [CrossRef] [PubMed]
- Uota, M.; Ogawa, T.; Ikebe, K.; Arai, Y.; Kamide, K.; Gondo, Y.; Masui, Y.; Ishizaki, T.; Inomata, C.; Takeshita, H.; et al. Factors related to taste sensitivity in elderly: Cross-sectional findings from SONIC study. J. Oral Rehabil. 2016, 43, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Tepper, B.; Melis, M.; Koelliker, Y.; Gasparini, P.; Ahijevych, K.; Tomassini Barbarossa, I. Factors Influencing the Phenotypic Characterization of the Oral Marker, PROP. Nutrients 2017, 9, 1275. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Uota, M.; Ikebe, K.; Arai, Y.; Kamide, K.; Gondo, Y.; Masui, Y.; Ishizaki, T.; Inomata, C.; Takeshita, H.; et al. Longitudinal study of factors affecting taste sense decline in old-old individuals. J. Oral Rehabil. 2017, 44, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Cavazzana, A.; Röhrborn, A.; Garthus-Niegel, S.; Larsson, M.; Hummel, T.; Croy, I. Sensory-specific impairment among older people. An investigation using both sensory thresholds and subjective measures across the five senses. PLoS ONE 2018, 13, e0202969. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Reed, D.R.; Roberts, K.M.; Mathew, P.S.; Mansfield, C.J. Age-Related Differences in Bitter Taste and Efficacy of Bitter Blockers. PLoS ONE 2014, 9, e103107. [Google Scholar] [CrossRef] [PubMed]
- Mojet, J. Taste Perception with Age: Generic or Specific Losses in Threshold Sensitivity to the Five Basic Tastes? Chem. Sens. 2001, 26, 845–860. [Google Scholar] [CrossRef]
- Hansen, J.L.; Reed, D.R.; Wright, M.J.; Martin, N.G.; Breslin, P.A.S. Heritability and genetic covariation of sensitivity to PROP, SOA, quinine HCl, and caffeine. Chem. Sens. 2006, 31, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Cordero, E.; Malacara-Hernandez, J.M.; Martinez-Cordero, C. Taste perception in normal and overweight Mexican adults. Appetite 2015, 89, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.A.; Bartoshuk, L.M.; Fillingim, R.B.; Dotson, C.D. Exploring Ethnic Differences in Taste Perception. Chem. Sens. 2016, 41, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Ledda, M.; Kutalik, Z.; Souza Destito, M.C.; Souza, M.M.; Cirillo, C.A.; Zamboni, A.; Martin, N.; Morya, E.; Sameshima, K.; Beckmann, J.S.; et al. GWAS of human bitter taste perception identifies new loci and reveals additional complexity of bitter taste genetics. Hum. Mol. Genet. 2014, 23, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Perna, S.; Riva, A.; Nicosanti, G.; Carrai, M.; Barale, R.; Vigo, B.; Allegrini, P.; Rondanelli, M. Association of the bitter taste receptor gene TAS2R38 (polymorphism RS713598) with sensory responsiveness, food preferences, biochemical parameters and body-composition markers. A cross-sectional study in Italy. Int. J. Food Sci. Nutr. 2018, 69, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Henderson, S.A.; Levine, A.; Hann, C. Taste and food preferences as predictors of dietary practices in young women. Public Health Nutr. 1999, 2, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Jayasinghe, S.; Kruger, R.; Walsh, D.; Cao, G.; Rivers, S.; Richter, M.; Breier, B. Is Sweet Taste Perception Associated with Sweet Food Liking and Intake? Nutrients 2017, 9, 750. [Google Scholar] [CrossRef] [PubMed]
- Bobowski, N.; Mennella, J.A. Personal Variation in Preference for Sweetness: Effects of Age and Obesity. Childhood Obes. 2017, 13, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Leong, C.S.-F.; Forde, C.G.; Tey, S.L.; Henry, C.J. Taste perception and diet in people of Chinese ancestry. Asia Pacif. J. Clin. Nutr. 2018, 27, 478–486. [Google Scholar]
- Mennella, J.A.; Finkbeiner, S.; Lipchock, S.V.; Hwang, L.-D.; Reed, D.R. Preferences for Salty and Sweet Tastes Are Elevated and Related to Each Other during Childhood. PLoS ONE 2014, 9, e92201. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.C.; Salgado, R.V.; Dresler, C.; Faller, R.W.; Bartlett, C. Flavour preferences in youth versus adults: A review. Tobacco Control 2016, 25, ii32–ii39. [Google Scholar] [CrossRef] [PubMed]
- Grunberg, N.E.; Straub, R.O. The role of gender and taste class in the effects of stress on eating. Health Psychol. 1992, 11, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A. Taste preferences and food intake. Annu. Rev. Nutr. 1997, 17, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Deglaire, A.; Méjean, C.; Castetbon, K.; Kesse-Guyot, E.; Hercberg, S.; Schlich, P. Associations between weight status and liking scores for sweet, salt and fat according to the gender in adults (The Nutrinet-Santé study). Eur. J. Clin. Nutr. 2015, 69, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Lampuré, A.; Castetbon, K.; Deglaire, A.; Schlich, P.; Péneau, S.; Hercberg, S.; Méjean, C. Associations between liking for fat, sweet or salt and obesity risk in French adults: A prospective cohort study. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 74. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, M.C.; Tordoff, M.G.; El-Sohemy, A.; van Dam, R.M. Recalled taste intensity, liking and habitual intake of commonly consumed foods. Appetite 2017, 109, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bailo, B.; Toguri, C.; Eny, K.M.; El-Sohemy, A. Genetic Variation in Taste and Its Influence on Food Selection. OMICS 2009, 13, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Negri, R.; Di Feola, M.; Di Domenico, S.; Scala, M.G.; Artesi, G.; Valente, S.; Smarrazzo, A.; Turco, F.; Morini, G.; Greco, L. Taste Perception and Food Choices. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.-G.; Chen, C.-B.; Wang, Z.-X.; Liu, Y.-P.; Wen, X.-Y.; Zhang, S.-F.; Sun, T.-W. A case-control study on the relationship between salt intake and salty taste and risk of gastric cancer. World J. Gastroenterol. 2011, 17, 2049–2053. [Google Scholar] [CrossRef] [PubMed]
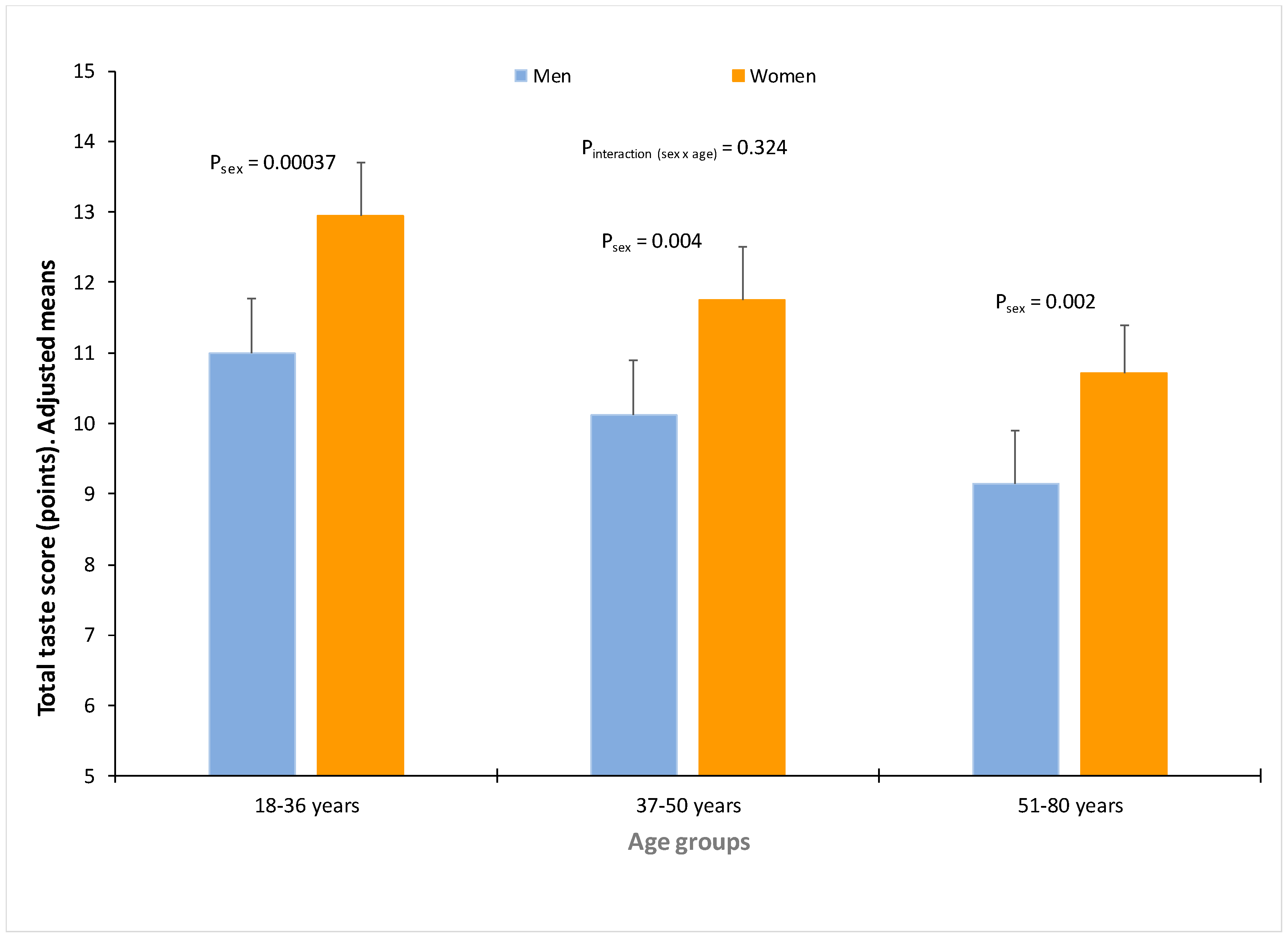
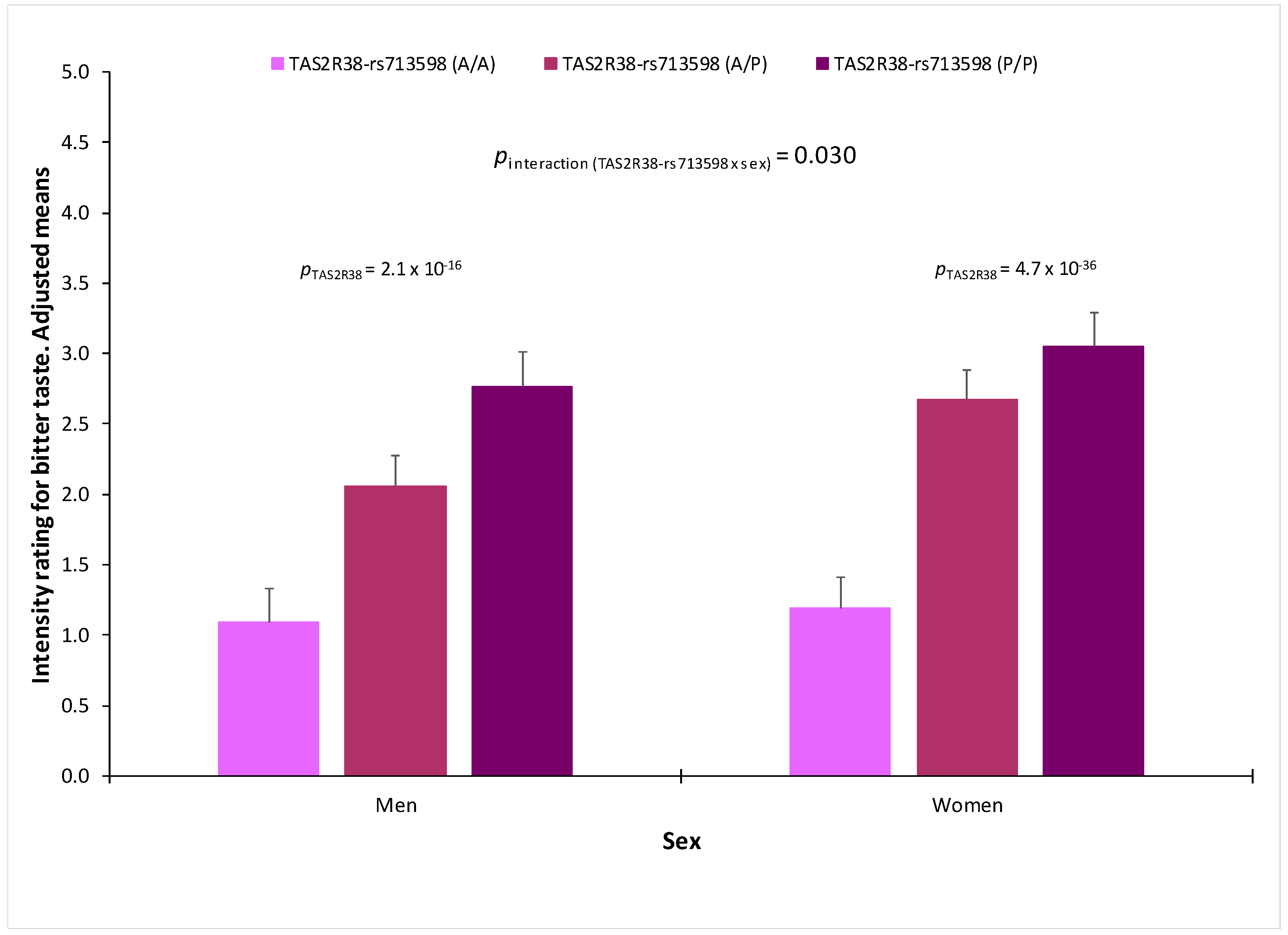
| Characteristics | Total (n = 1020) | Men (n = 365) | Women (n = 655) | p |
|---|---|---|---|---|
| Age (years) | 43.2 ± 14.3 | 42.2 ± 14.5 | 43.8 ± 14.1 | 0.095 |
| BMI (Kg/m2) | 27.1 ± 5.3 | 28.0 ± 5.2 | 26.6 ± 5.3 | <0.001 |
| Obesity cases: n, % | 281 (27.5) | 118 (32.3) | 163 (24.1) | 0.011 |
| Type 2 diabetes: n, % | 46 (4.5) | 19 (5.2) | 27 (4.1) | 0.422 |
| Current smokers: n, % | 206 (20.2) | 70 (19.2) | 136 (20.8) | 0.704 |
| SBP (mmHg) | 124.6 ± 17.4 | 131.4 ± 15.9 | 120.7 ± 17.1 | <0.001 |
| DBP (mmHg) | 77.9 ± 10.4 | 80.6 ± 11.0 | 76.5 ± 9.8 | <0.001 |
| Total cholesterol (mg/dL) 1 | 207.7 ± 39.3 | 201.9 ± 38.3 | 210.8 ± 39.5 | 0.001 |
| HDL-C (mg/dL) | 60.2 ± 14.3 | 52.2 ± 10.8 | 64.6 ± 14.1 | <0.001 |
| LDL-C (mg/dL) | 133.6 ± 31.5 | 133.6 ± 32.1 | 133.5 ± 31.1 | 0.947 |
| Triglycerides (mg/dL) | 105.5 ± 56.3 | 118.6 ± 65.5 | 98.3 ± 49.1 | 0.138 |
| Fasting glucose (mg/dL) | 93.8 ± 19.3 | 96.6 ± 22.4 | 92.3 ± 17.2 | 0.001 |
| Medications: n, % | ||||
| Antihypertensive drugs | 132 (12.9) | 67 (18.4) | 65 (9.9) | <0.001 |
| Hypolipidaemic drugs | 123 (12.1) | 52 (14.2) | 71 (10.8) | 0.098 |
| TAS2R38-rs713598: n, % 2 | 0.329 | |||
| Ala/Ala | 304 (32.2) | 116 (33.9) | 188 (31.2) | |
| Ala/Pro | 464 (49.1) | 157 (45.9) | 307 (50.9) | |
| Pro/Pro | 177 (18.7) | 69 (20.2) | 108 (17.9) | |
| TAS1R2-rs35874116: n, % | 0.853 | |||
| Ile/Ile | 345 (36.4) | 122 (35.9) | 223 (36.6) | |
| Ile/Val | 447 (47.1) | 164 (48.2) | 283 (46.5) | |
| Val/Val | 157 (16.5) | 54 (15.9) | 103 (16.9) | |
| SCNN1B-rs239345: n, % | 0.534 | |||
| TT | 481 (51.9) | 169 (51.2) | 312 (52.3) | |
| TA | 371 (40.0) | 139 (41.8) | 233 (39.0) | |
| AA | 75 (8.1) | 23 (7.0) | 52 (8.7) |
| Taste (Tastant) | Mean (SD) 3 | Coefficients | Bitter | Sweet | Salty | Sour | Umami |
|---|---|---|---|---|---|---|---|
| Bitter | 2.07 (1.54) | r | 1 | ||||
| (PROP 5.5 mM) | p 4 | ||||||
| Sweet | 1.86 (1.14) | r | 0.162 | 1 | |||
| (Sucrose 400 mM) | p | <0.001 | |||||
| Salty | 2.57 (1.35) | r | 0.255 | 0.526 | 1 | ||
| (NaCl 200 mM) | p | <0.001 | <0.001 | ||||
| Sour | 2.73 (1.35) | r | 0.221 | 0.433 | 0.569 | 1 | |
| (Citric acid 34 mM) | p | <0.001 | <0.001 | <0.001 | |||
| Umami | 1.98 (1.39) | r | 0.230 | 0.359 | 0.352 | 0.348 | 1 |
| (MPG 200 mM) | p | <0.001 | <0.001 | <0.001 | <0.001 | ||
| Total taste score 5 | 11.22 (4.68) | r | 0.576 | 0.664 | 0.770 | 0.731 | 0.658 |
| p | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| Taste | B 1 (SE) | p1 | B 2 (SE) | p2 | B 3 (SE) | p3 |
|---|---|---|---|---|---|---|
| Bitter (PROP 5.5 mM) | −0.021 (0.003) | 3.7 × 10−10 | −0.020 (0.007) | 9.6 × 10−9 | −0.019 (0.004) | 7.8 × 10−7 |
| Sweet (Sucrose 400 mM) | −0.012 (0.002) | 2.0 × 10−6 | −0.011 (0.003) | 7.0 × 10−5 | −0.011 (0.003) | 1.5 × 10−4 |
| Salty (NaCl 200 mM) | −0.013 (0.003) | 7.0 × 10−6 | −0.013 (0.003) | 5.8 × 10−5 | −0.011 (0.003) | 1.0 × 10−3 |
| Sour (Citric acid 34 mM) | −0.018 (0.003) | 1.2 × 10−9 | −0.016 (0.003) | 1.4 × 10−7 | −0.015 (0.003) | 6.0 × 10−6 |
| Umami (MPG 200 mM) | −0.013 (0.003) | 1.5 × 10−5 | −0.012 (0.003) | 3.0 × 10−4 | −0.013 (0.004) | 2.5 × 10−4 |
| Total taste score 4 | −0.077 (0.010) | 2.9 × 10−14 | −0.071 (0.011) | 2.1 × 10−11 | −0.070 (0.029) | 2.4 × 10−9 |
| Taste | B (SE) | p1 | p2 Interaction Term (Sex × Age) |
|---|---|---|---|
| Bitter (PROP 5.5 mM) | 0.472 (0.100) | 1.3 × 10−6 | 0.965 |
| Sweet (Sucrose 400 mM) | 0.166 (0.076) | 0.026 | 0.172 |
| Salty (NaCl 200 mM) | 0.417 (0.089) | 1.3 × 10−6 | 0.410 |
| Sour (Citric acid 34 mM) | 0.498 (0.087) | 1.6 × 10−8 | 0.703 |
| Umami (MPG 200 mM) | 0.169 (0.093) | 0.069 | 0.107 |
| Total taste score 3 | 1.720 (0.301) | 1.4 × 10−8 | 0.324 |
| Taste | Mean (SD) 3 | Coefficients | Bitter Taste Preference | Sweet Taste Preference | Salty Taste Preference | Sour Taste Preference |
|---|---|---|---|---|---|---|
| Whole population | ||||||
| Bitter taste | 0.60 (0.80) | r | 1 | |||
| preference | p4 | |||||
| Sweet taste | 2.43 (0.80) | r | −0.175 | 1 | ||
| preference | p | <0.001 | ||||
| Salty taste | 2.22 (0.91) | r | 0.048 | −0.053 | 1 | |
| preference | p | 0.139 | 0.100 | |||
| Sour taste | 0.63 (0.84) | r | 0.346 | −0.125 | 0.131 | 1 |
| preference | p | <0.001 | <0.001 | <0.001 | ||
| Men | ||||||
| Bitter taste | 0.75 (0.87) 5 | r | 1 | |||
| preference | p | |||||
| Sweet taste | 2.45 (0.74) | r | −0.181 | 1 | ||
| preference | p | <0.001 | ||||
| Salty taste | 2.23 (0.86) | r | 0.068 | −0.027 | 1 | |
| preference | p | 0.216 | 0.622 | |||
| Sour taste | 0.70 (0.81) 6 | r | 0.430 | −0.039 | 0.199 | 1 |
| preference | p | <0.001 | 0.471 | <0.001 | ||
| Women | ||||||
| Bitter taste | 0.51 (0.75) 5 | r | 1 | |||
| preference | p | |||||
| Sweet taste | 2.42 (0.83) | r | −0.173 | 1 | ||
| preference | p | <0.001 | ||||
| Salty taste | 2.22 (0.94) | r | 0.040 | −0.067 | 1 | |
| preference | p | 0.318 | 0.096 | |||
| Sour taste | 0.60 (0.86) 6 | r | 0.286 | −0.167 | 0.100 | 1 |
| preference | p | <0.001 | <0.001 | 0.013 | ||
| Taste (Tastant) | Sex | Coeffi-Cients | Bitter Taste Preference | Sweet Taste Preference | Salty Taste Preference | Sour Taste Preference |
|---|---|---|---|---|---|---|
| Men | ||||||
| Bitter | r | 0.010 | −0.040 | −0.020 | 0.035 | |
| (PROP 5.5 mM) | p3 | 0.858 | 0.461 | 0.712 | 0.521 | |
| Sweet | r | 0.111 | −0.043 | 0.044 | 0.126 | |
| (Sucrose 400 mM) | p | 0.042 | 0.428 | 0.415 | 0.021 | |
| Salty | r | 0.114 | 0.016 | 0.011 | 0.090 | |
| (NaCl 200 mM) | p | 0.036 | 0.774 | 0.839 | 0.098 | |
| Sour | r | 0.090 | −0.030 | 0.018 | 0.066 | |
| (Citric acid 34 mM) | p | 0.099 | 0.582 | 0.742 | 0.225 | |
| Umami | r | 0.178 | −0.060 | −0.050 | 0.135 | |
| (MPG 200 mM) | p | 0.001 | 0.269 | 0.362 | 0.013 | |
| Women | ||||||
| Bitter | r | 0.026 | 0.059 | −0.011 | 0.002 | |
| (PROP 5.5 mM) | P3 | 0.515 | 0.140 | 0.789 | 0.951 | |
| Sweet | r | 0.110 | 0.038 | −0.011 | 0.101 | |
| (Sucrose 400 mM) | p | 0.006 | 0.352 | 0.783 | 0.012 | |
| Salty | r | 0.037 | 0.062 | −0.103 | 0.040 | |
| (NaCl 200 mM) | p | 0.359 | 0.126 | 0.011 | 0.325 | |
| Sour | r | 0.080 | 0.015 | −0.039 | 0.085 | |
| (Citric acid 34 mM) | p | 0.046 | 0.706 | 0.335 | 0.036 | |
| Umami | r | 0.098 | 0.008 | −0.066 | 0.015 | |
| (MPG 200 mM) | p | 0.015 | 0.846 | 0.103 | 0.702 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barragán, R.; Coltell, O.; Portolés, O.; Asensio, E.M.; Sorlí, J.V.; Ortega-Azorín, C.; González, J.I.; Sáiz, C.; Fernández-Carrión, R.; Ordovas, J.M.; et al. Bitter, Sweet, Salty, Sour and Umami Taste Perception Decreases with Age: Sex-Specific Analysis, Modulation by Genetic Variants and Taste-Preference Associations in 18 to 80 Year-Old Subjects. Nutrients 2018, 10, 1539. https://doi.org/10.3390/nu10101539
Barragán R, Coltell O, Portolés O, Asensio EM, Sorlí JV, Ortega-Azorín C, González JI, Sáiz C, Fernández-Carrión R, Ordovas JM, et al. Bitter, Sweet, Salty, Sour and Umami Taste Perception Decreases with Age: Sex-Specific Analysis, Modulation by Genetic Variants and Taste-Preference Associations in 18 to 80 Year-Old Subjects. Nutrients. 2018; 10(10):1539. https://doi.org/10.3390/nu10101539
Chicago/Turabian StyleBarragán, Rocio, Oscar Coltell, Olga Portolés, Eva M. Asensio, José V. Sorlí, Carolina Ortega-Azorín, José I. González, Carmen Sáiz, Rebeca Fernández-Carrión, Jose M. Ordovas, and et al. 2018. "Bitter, Sweet, Salty, Sour and Umami Taste Perception Decreases with Age: Sex-Specific Analysis, Modulation by Genetic Variants and Taste-Preference Associations in 18 to 80 Year-Old Subjects" Nutrients 10, no. 10: 1539. https://doi.org/10.3390/nu10101539
APA StyleBarragán, R., Coltell, O., Portolés, O., Asensio, E. M., Sorlí, J. V., Ortega-Azorín, C., González, J. I., Sáiz, C., Fernández-Carrión, R., Ordovas, J. M., & Corella, D. (2018). Bitter, Sweet, Salty, Sour and Umami Taste Perception Decreases with Age: Sex-Specific Analysis, Modulation by Genetic Variants and Taste-Preference Associations in 18 to 80 Year-Old Subjects. Nutrients, 10(10), 1539. https://doi.org/10.3390/nu10101539








