Maternal Choline and Betaine Supplementation Modifies the Placental Response to Hyperglycemia in Mice and Human Trophoblasts
Abstract
1. Introduction
2. Materials and Methods
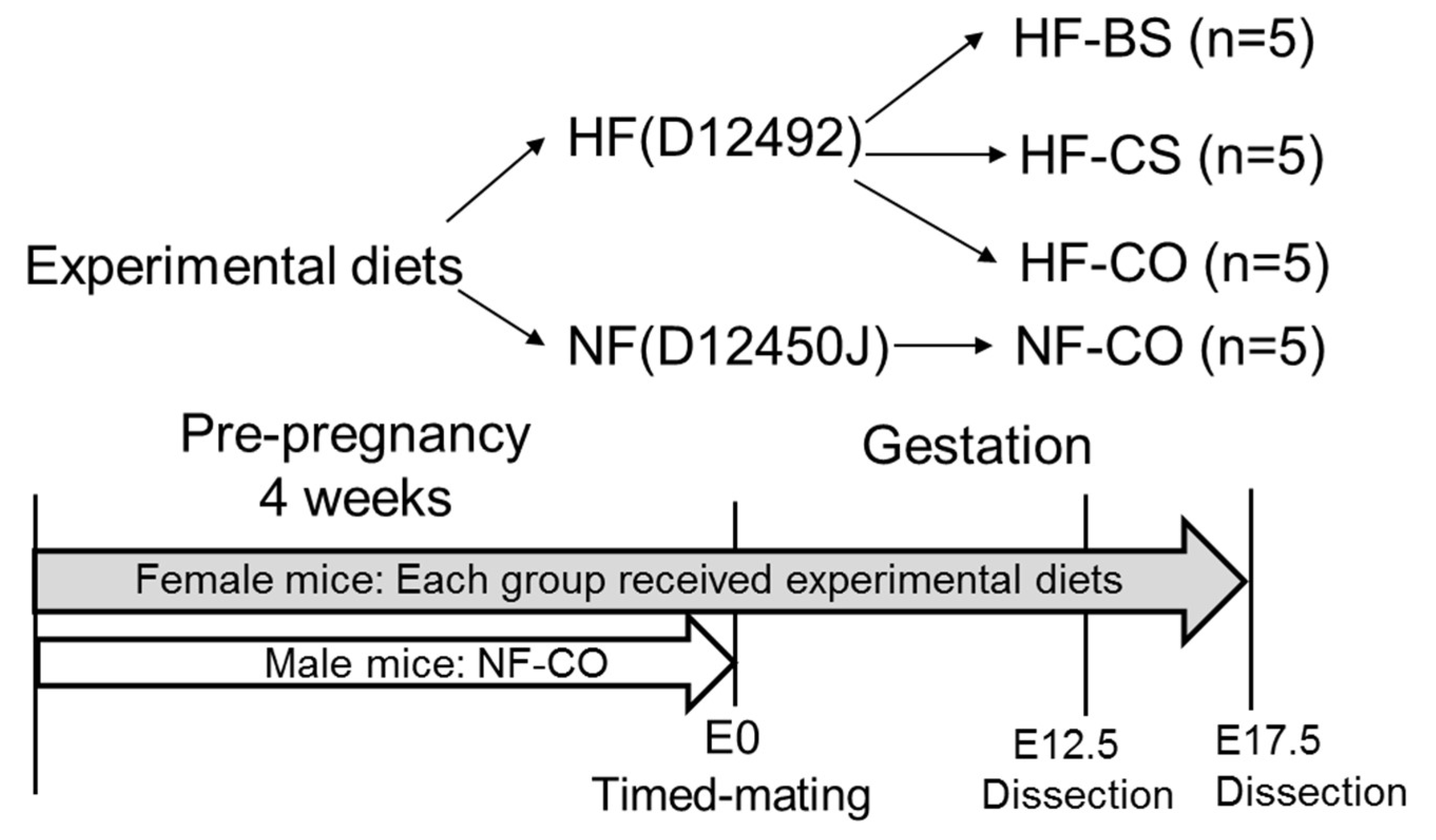
2.1. Animal Diet and Treatment
2.2. Sample Collection and Processing
2.3. Placental Histology
2.4. Cell Culture and Treatments
2.5. Accumulation of Glucose and Fatty Acid in BeWo Cells
2.6. RNA Extraction and Real-Time PCR
2.7. Statistical Analyses
3. Results
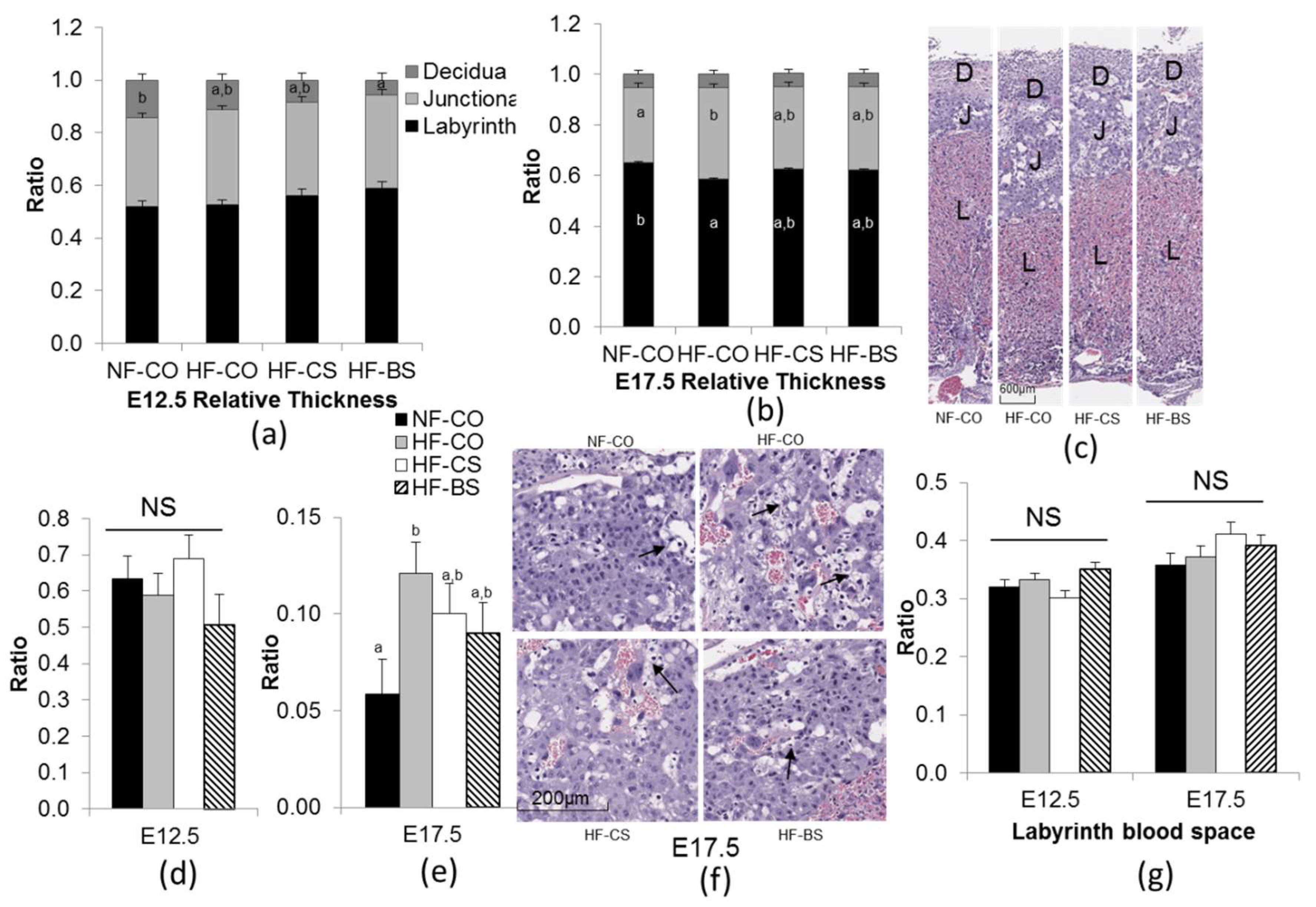
3.1. Choline and Betaine Supplementation Partially Normalized the Altered Placental Morphology of GDM Mice at E17.5
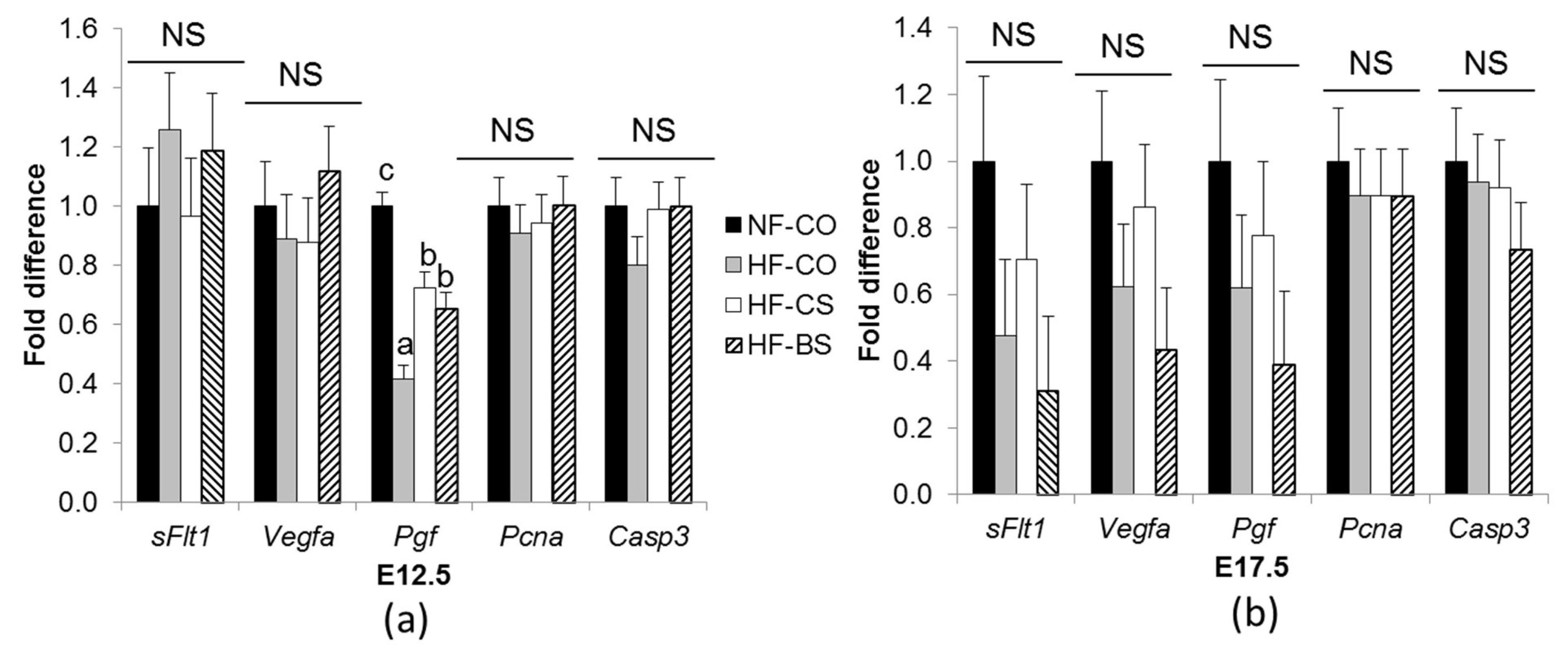
3.2. HF Feeding and Choline/Betaine Supplementation Had Minimal Effects on Placental Angiogenic Gene Expression in GDM Mice
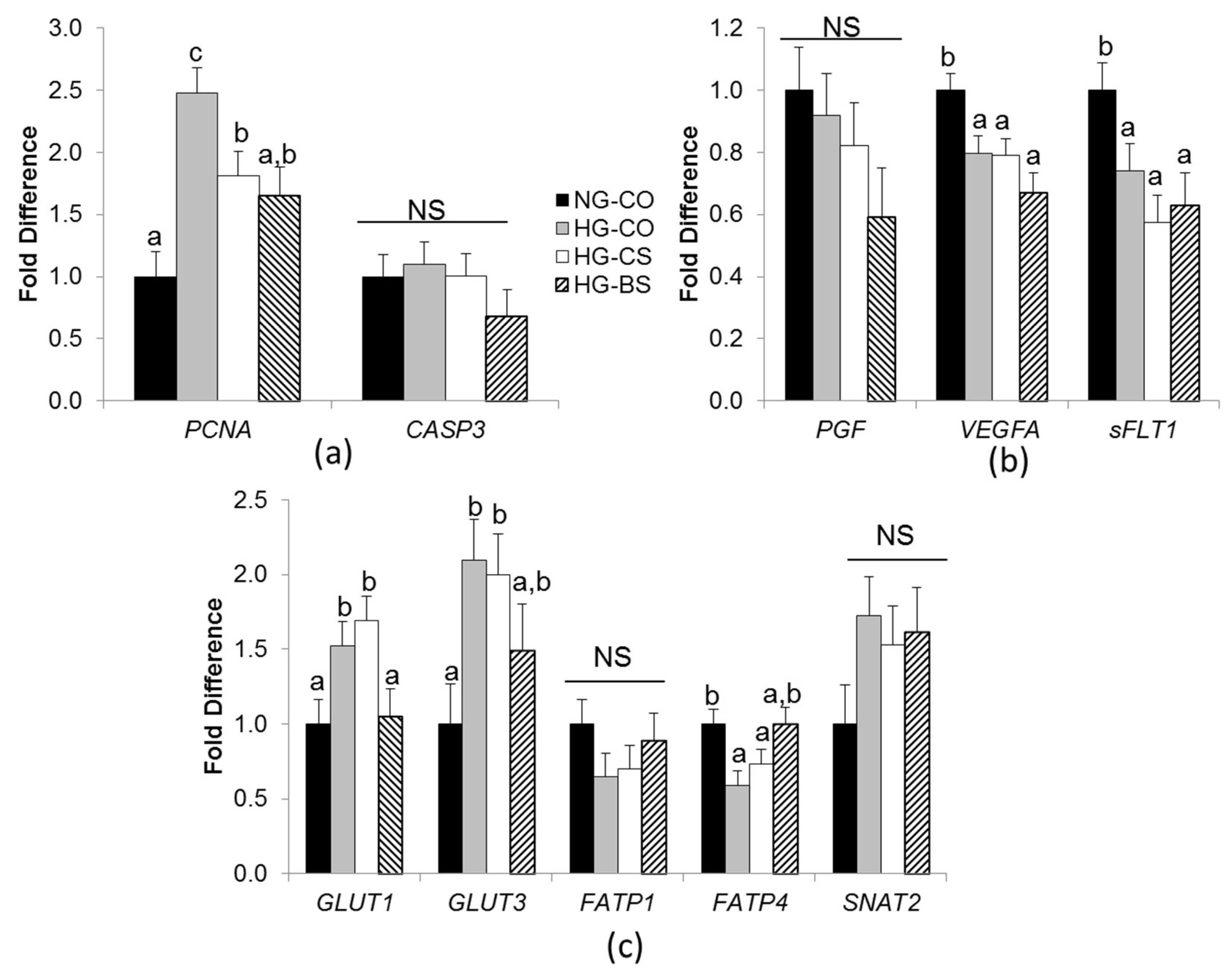
3.3. Choline/Betaine Supplementation Influenced Gene Expression in BeWo Cells under a Hyperglycemic Condition
3.4. Choline or Betaine Supplementation Lowers Glucose and Fatty Acid Accumulation in BeWo Cells
4. Discussion
4.1. Choline and Betaine Supplementation Mitigates the Alteration in Placental Layer Thickness in GDM Mice
4.2. Choline and Betaine Have Minimal Effects on the Vasculature of GDM Placentas
4.3. Both Betaine and Choline Reduce Glucose and Fatty Acid Accumulation in Trophoblasts
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chu, S.Y.; Callaghan, W.M.; Kim, S.Y.; Schmid, C.H.; Lau, J.; England, L.J.; Dietz, P.M. Maternal obesity and risk of gestational diabetes mellitus. Diabetes Care 2007, 30, 2070–2076. [Google Scholar] [CrossRef] [PubMed]
- Hunt, K.J.; Schuller, K.L. The increasing prevalence of diabetes in pregnancy. Obstet. Gynecol. Clin. N. Am. 2007, 34, 173–199. [Google Scholar] [CrossRef] [PubMed]
- Bain, E.; Crane, M.; Tieu, J.; Han, S.; Crowther, C.A.; Middleton, P. Diet and exercise interventions for preventing gestational diabetes mellitus. Cochrane Database Syst. Rev. 2015, 4, CD010443. [Google Scholar] [CrossRef] [PubMed]
- Hermann, G.M.; Dallas, L.M.; Haskell, S.E.; Roghair, R.D. Neonatal macrosomia is an independent risk factor for adult metabolic syndrome. Neonatology 2010, 98, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Kc, K.; Shakya, S.; Zhang, H. Gestational diabetes mellitus and macrosomia: A literature review. Ann. Nutr. Metab. 2015, 66, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Brett, K.E.; Ferraro, Z.M.; Yockell-Lelievre, J.; Gruslin, A.; Adamo, K.B. Maternal-fetal nutrient transport in pregnancy pathologies: the role of the placenta. Int. J. Mol. Sci. 2014, 15, 16153–16185. [Google Scholar] [CrossRef] [PubMed]
- Desoye, G.; Gauster, M.; Wadsack, C. Placental transport in pregnancy pathologies. Am. J. Clin. Nutr. 2011, 94, S1896–S1902. [Google Scholar] [CrossRef] [PubMed]
- Rosario, F.J.; Powell, T.L.; Jansson, T. Activation of placental insulin and mTOR signaling in a mouse model of maternal obesity associated with fetal overgrowth. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 310, R87–R93. [Google Scholar] [CrossRef] [PubMed]
- Bell, A.W.; Ehrhardt, R.A. Regulation of placental nutrient transport and implications for fetal growth. Nutr. Res. Rev. 2002, 15, 211–230. [Google Scholar] [CrossRef] [PubMed]
- Malassiné, A.; Frendo, J.L.; Evain-Brion, D. A comparison of placental development and endocrine functions between the human and mouse model. Hum. Reprod. Update 2003, 9, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Aires, M.B.; Dos Santos, A.C. Effects of maternal diabetes on trophoblast cells. World J. Diabetes 2015, 6, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Taricco, E.; Radaelli, T.; Nobile de Santis, M.S.; Cetin, I. Foetal and placental weights in relation to maternal characteristics in gestational diabetes. Placenta 2003, 24, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Akison, L.K.; Nitert, M.D.; Clifton, V.L.; Moritz, K.M.; Simmons, D.G. Review: Alterations in placental glycogen deposition in complicated pregnancies: Current preclinical and clinical evidence. Placenta 2017, 54, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Shafrir, E.; Barash, V. Placental glycogen metabolism in diabetic pregnancy. Isr. J. Med. Sci. 1991, 27, 449–461. [Google Scholar] [PubMed]
- Cvitic, S.; Desoye, G.; Hiden, U. Glucose, insulin, and oxygen interplay in placental hypervascularisation in diabetes mellitus. BioMed Res. Int. 2014, 2014, 145846. [Google Scholar] [CrossRef] [PubMed]
- Gaither, K.; Quraishi, A.N.; Illsley, N.P. Diabetes alters the expression and activity of the human placental GLUT1 glucose transporter. J. Clin. Endocrinol. Metab. 1999, 84, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.N.; Woollett, L.A.; Barbour, N.; Prasad, P.D.; Powell, T.L.; Jansson, T. High-fat diet before and during pregnancy causes marked up-regulation of placental nutrient transport and fetal overgrowth in C57/BL6 mice. FASEB J. 2009, 23, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Castrejon, M.; Powell, T.L. Placental Nutrient Transport in Gestational Diabetic Pregnancies. Front. Endocrinol. (Lausanne) 2017, 8, 306. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthy, M.V.; Lodhi, I.J.; Yin, L.; Malapaka, R.R.; Xu, H.E.; Turk, J.; Semenkovich, C.F. Identification of a physiologically relevant endogenous ligand for PPARalpha in liver. Cell 2009, 138, 476–488. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Kang, L.; Jiang, Y. Effect of dietary betaine supplementation on lipogenesis gene expression and CpG methylation of lipoprotein lipase gene in broilers. Mol. Biol. Rep. 2011, 38, 1975–1981. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Shen, L.; Tan, Z.; Zhang, P.; Zhao, X.; Xu, Y.; Gan, M.; Yang, Q.; Ma, J.; Jiang, A.; et al. Betaine Supplementation Enhances Lipid Metabolism and Improves Insulin Resistance in Mice Fed a High-Fat Diet. Nutrients 2018, 10, 131. [Google Scholar] [CrossRef] [PubMed]
- Lee, I. Betaine is a positive regulator of mitochondrial respiration. Biochem. Biophys. Res. Commun. 2015, 456, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Jack-Roberts, C.; Joselit, Y.; Nanobashvili, K.; Bretter, R.; Malysheva, O.V.; Caudill, M.A.; Saxena, A.; Axen, K.; Gomaa, A.; Jiang, X. Choline Supplementation Normalizes Fetal Adiposity and Reduces Lipogenic Gene Expression in a Mouse Model of Maternal Obesity. Nutrients 2017, 9, 899. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.; Greenwald, E.; Jack-Roberts, C.; Ajeeb, T.T.; Malysheva, O.V.; Caudill, M.A.; Axen, K.; Saxena, A.; Semernina, E.; Nanobashvili, K.; et al. Choline prevents fetal overgrowth and normalizes placental fatty acid and glucose metabolism in a mouse model of maternal obesity. J. Nutr. Biochem. 2017, 49, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Joselit, Y.; Nanobashvili, K.; Jack-Roberts, C.; Greenwald, E.; Malysheva, O.V.; Caudill, M.A.; Saxena, A.; Jiang, X. Maternal betaine supplementation affects fetal growth and lipid metabolism of high-fat fed mice in a temporal-specific manner. Nutr. Diabetes 2018, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Jones, S.; Andrew, B.Y.; Ganti, A.; Malysheva, O.V.; Giallourou, N.; Brannon, P.M.; Roberson, M.S.; Caudill, M.A. Choline Inadequacy Impairs Trophoblast Function and Vascularization in Cultured Human Placental Trophoblasts. J. Cell. Physiol. 2014, 229, 1016–1027. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Bar, H.Y.; Yan, J.; Jones, S.; Brannon, P.M.; West, A.A.; Perry, C.A.; Ganti, A.; Pressman, E.; Devapatla, S.; et al. A higher maternal choline intake among third-trimester pregnant women lowers placental and circulating concentrations of the antiangiogenic factor fms-like tyrosine kinase-1 (sFLT1). FASEB J. 2013, 27, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Yan, J.; West, A.A.; Perry, C.A.; Malysheva, O.V.; Devapatla, S.; Pressman, E.; Vermeylen, F.; Caudill, M.A. Maternal choline intake alters the epigenetic state of fetal cortisol-regulating genes in humans. FASEB J. 2012, 26, 3563–3574. [Google Scholar] [CrossRef] [PubMed]
- Kwan, S.T.C.; King, J.H.; Yan, J.; Wang, Z.; Jiang, X.; Hutzler, J.S.; Klein, H.R.; Brenna, J.T.; Roberson, M.S.; Caudill, M.A. Maternal Choline Supplementation Modulates Placental Nutrient Transport and Metabolism in Late Gestation of Mouse Pregnancy. J. Nutr. 2017, 147, 2083–2092. [Google Scholar] [CrossRef] [PubMed]
- Kwan, S.T.C.; King, J.H.; Grenier, J.K.; Yan, J.; Jiang, X.; Roberson, M.S.; Caudill, M.A. Maternal Choline Supplementation during Normal Murine Pregnancy Alters the Placental Epigenome: Results of an Exploratory Study. Nutrients 2018, 10, 417. [Google Scholar] [CrossRef] [PubMed]
- Mellott, T.J.; Williams, C.L.; Meck, W.H.; Blusztajn, J.K. Prenatal choline supplementation advances hippocampal development and enhances MAPK and CREB activation. FASEB J. 2004, 18, 545–547. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yao, T.; Pini, M.; Zhou, Z.; Fantuzzi, G.; Song, Z. Betaine improved adipose tissue function in mice fed a high-fat diet: a mechanism for hepatoprotective effect of betaine in nonalcoholic fatty liver disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G634–G642. [Google Scholar] [CrossRef] [PubMed]
- McClive, P.J.; Sinclair, A.H. Rapid DNA extraction and PCR-sexing of mouse embryos. Mol. Reprod. Dev. 2001, 60, 225–226. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.W.; Young, S.L.; Grattan, D.R.; Jasoni, C.L. Obesity during pregnancy disrupts placental morphology, cell proliferation, and inflammation in a sex-specific manner across gestation in the mouse. Biol. Reprod. 2014, 90, 130. [Google Scholar] [CrossRef] [PubMed]
- Coan, P.M.; Conroy, N.; Burton, G.J.; Ferguson-Smith, A.C. Origin and characteristics of glycogen cells in the developing murine placenta. Dev. Dyn. 2006, 235, 3280–3294. [Google Scholar] [CrossRef] [PubMed]
- Cuffe, J.S.; Walton, S.L.; Singh, R.R.; Spiers, J.G.; Bielefeldt-Ohmann, H.; Wilkinson, L.; Little, M.H.; Moritz, K.M. Mid- to late term hypoxia in the mouse alters placental morphology, glucocorticoid regulatory pathways and nutrient transporters in a sex-specific manner. J. Physiol. 2014, 592, 3127–3141. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Greenwald, E.; Jack-Roberts, C. Effects of Choline on DNA Methylation and Macronutrient Metabolic Gene Expression in In Vitro Models of Hyperglycemia. Nutr. Metab. Insights 2016, 9, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Desoye, G.; Hauguel-de Mouzon, S. The human placenta in gestational diabetes mellitus. The insulin and cytokine network. Diabetes Care 2007, 30, S120–S126. [Google Scholar] [CrossRef] [PubMed]
- Schneider, H.; Reiber, W.; Sager, R.; Malek, A. Asymmetrical transport of glucose across the in vitro perfused human placenta. Placenta 2003, 24, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Taricco, E.; Radaelli, T.; Rossi, G.; Nobile de Santis, M.S.; Bulfamante, G.P.; Avagliano, L.; Cetin, I. Effects of gestational diabetes on fetal oxygen and glucose levels in vivo. BJOG 2009, 116, 1729–1735. [Google Scholar] [CrossRef] [PubMed]
- Jirkovská, M.; Kubínová, L.; Janácek, J.; Moravcová, M.; Krejcí, V.; Karen, P. Topological properties and spatial organization of villous capillaries in normal and diabetic placentas. J. Vasc. Res. 2002, 39, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, F. Histomorphometry of the placenta of the diabetic women: class a diabetes mellitus. Placenta 1981, 2, 241–251. [Google Scholar] [CrossRef]
- Pietro, L.; Daher, S.; Rudge, M.V.; Calderon, I.M.; Damasceno, D.C.; Sinzato, Y.K.; Bandeira, C.; Bevilacqua, E. Vascular endothelial growth factor (VEGF) and VEGF-receptor expression in placenta of hyperglycemic pregnant women. Placenta 2010, 31, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Nadarajah, V.D.; Min, R.G.; Judson, J.P.; Jegasothy, R.; Ling, E.H. Maternal plasma soluble fms-like tyrosine kinase-1 and placental growth factor levels as biochemical markers of gestational hypertension for Malaysian mothers. J. Obstet. Gynaecol. Res. 2009, 35, 855–863. [Google Scholar] [CrossRef] [PubMed]
- El-Tarhouny, S.A.; Almasry, S.M.; Elfayomy, A.K.; Baghdadi, H.; Habib, F.A. Placental growth factor and soluble Fms-like tyrosine kinase 1 in diabetic pregnancy: A possible relation to distal villous immaturity. Histol. Histopathol. 2014, 29, 259–272. [Google Scholar] [PubMed]
- Pereira, R.D.; De Long, N.E.; Wang, R.C.; Yazdi, F.T.; Holloway, A.C.; Raha, S. Angiogenesis in the placenta: the role of reactive oxygen species signaling. BioMed Res. Int. 2015, 2015, 814543. [Google Scholar] [CrossRef] [PubMed]
- Olsen, J.M.; Sato, M.; Dallner, O.S.; Sandström, A.L.; Pisani, D.F.; Chambard, J.C.; Amri, E.Z.; Hutchinson, D.S.; Bengtsson, T. Glucose uptake in brown fat cells is dependent on mTOR complex 2-promoted GLUT1 translocation. J. Cell Biol. 2014, 207, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Larqué, E.; Demmelmair, H.; Gil-Sánchez, A.; Prieto-Sánchez, M.T.; Blanco, J.E.; Pagán, A.; Faber, F.L.; Zamora, S.; Parrilla, J.J.; Koletzko, B. Placental transfer of fatty acids and fetal implications. Am. J. Clin. Nutr. 2011, 94, S1908–S1913. [Google Scholar] [CrossRef] [PubMed]
- Pagán, A.; Prieto-Sánchez, M.T.; Blanco-Carnero, J.E.; Gil-Sánchez, A.; Parrilla, J.J.; Demmelmair, H.; Koletzko, B.; Larqué, E. Materno-fetal transfer of docosahexaenoic acid is impaired by gestational diabetes mellitus. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E826–E833. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nanobashvili, K.; Jack-Roberts, C.; Bretter, R.; Jones, N.; Axen, K.; Saxena, A.; Blain, K.; Jiang, X. Maternal Choline and Betaine Supplementation Modifies the Placental Response to Hyperglycemia in Mice and Human Trophoblasts. Nutrients 2018, 10, 1507. https://doi.org/10.3390/nu10101507
Nanobashvili K, Jack-Roberts C, Bretter R, Jones N, Axen K, Saxena A, Blain K, Jiang X. Maternal Choline and Betaine Supplementation Modifies the Placental Response to Hyperglycemia in Mice and Human Trophoblasts. Nutrients. 2018; 10(10):1507. https://doi.org/10.3390/nu10101507
Chicago/Turabian StyleNanobashvili, Khatia, Chauntelle Jack-Roberts, Rachel Bretter, Naudia Jones, Kathleen Axen, Anjana Saxena, Kali Blain, and Xinyin Jiang. 2018. "Maternal Choline and Betaine Supplementation Modifies the Placental Response to Hyperglycemia in Mice and Human Trophoblasts" Nutrients 10, no. 10: 1507. https://doi.org/10.3390/nu10101507
APA StyleNanobashvili, K., Jack-Roberts, C., Bretter, R., Jones, N., Axen, K., Saxena, A., Blain, K., & Jiang, X. (2018). Maternal Choline and Betaine Supplementation Modifies the Placental Response to Hyperglycemia in Mice and Human Trophoblasts. Nutrients, 10(10), 1507. https://doi.org/10.3390/nu10101507




