Monitoring Spatiotemporal Dynamics of Soil Moisture Under Water-Nitrogen Interactions in Arid Farmland Using UAV-Based Hyperspectral Sensing and Triple-Band Indices
Highlights
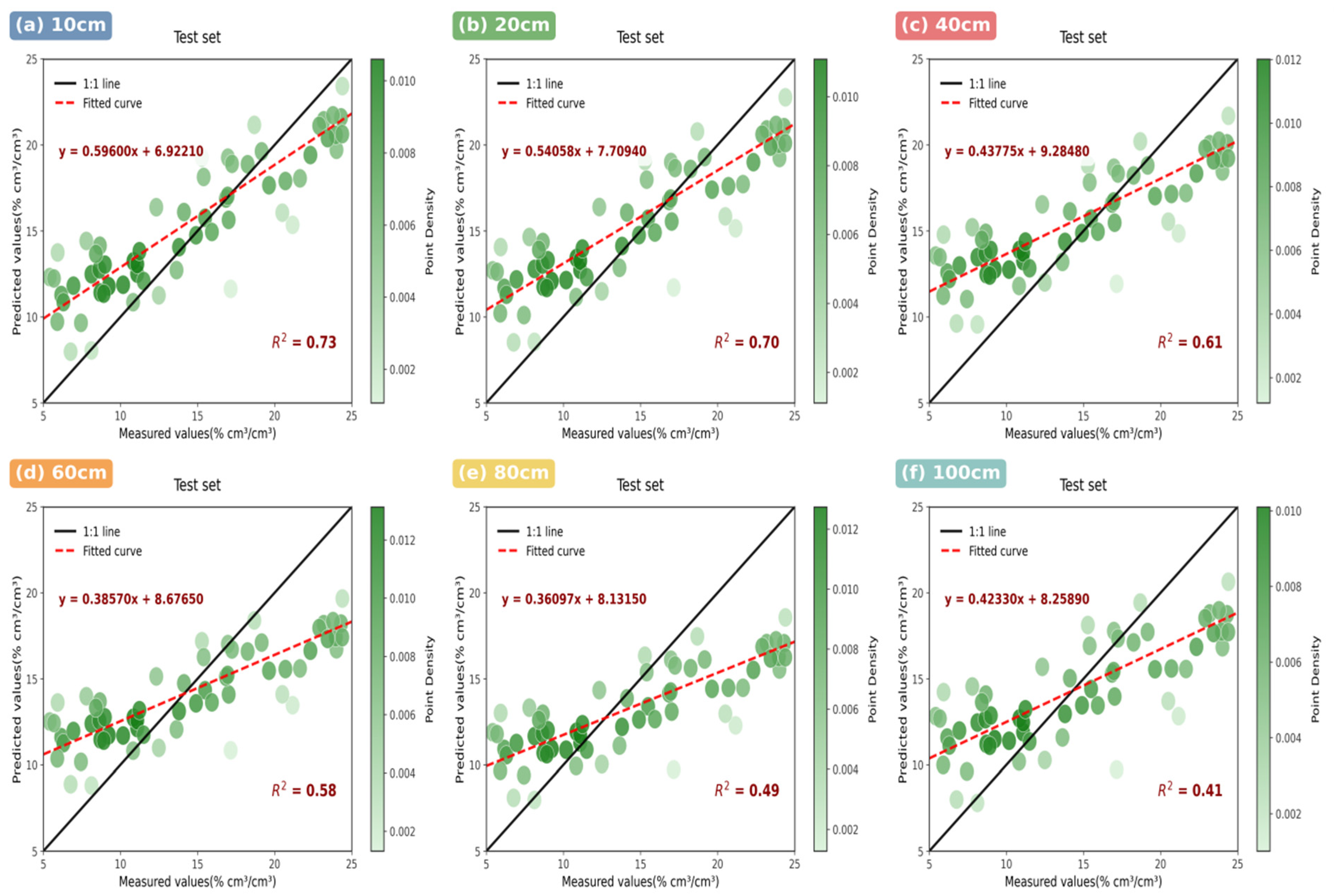
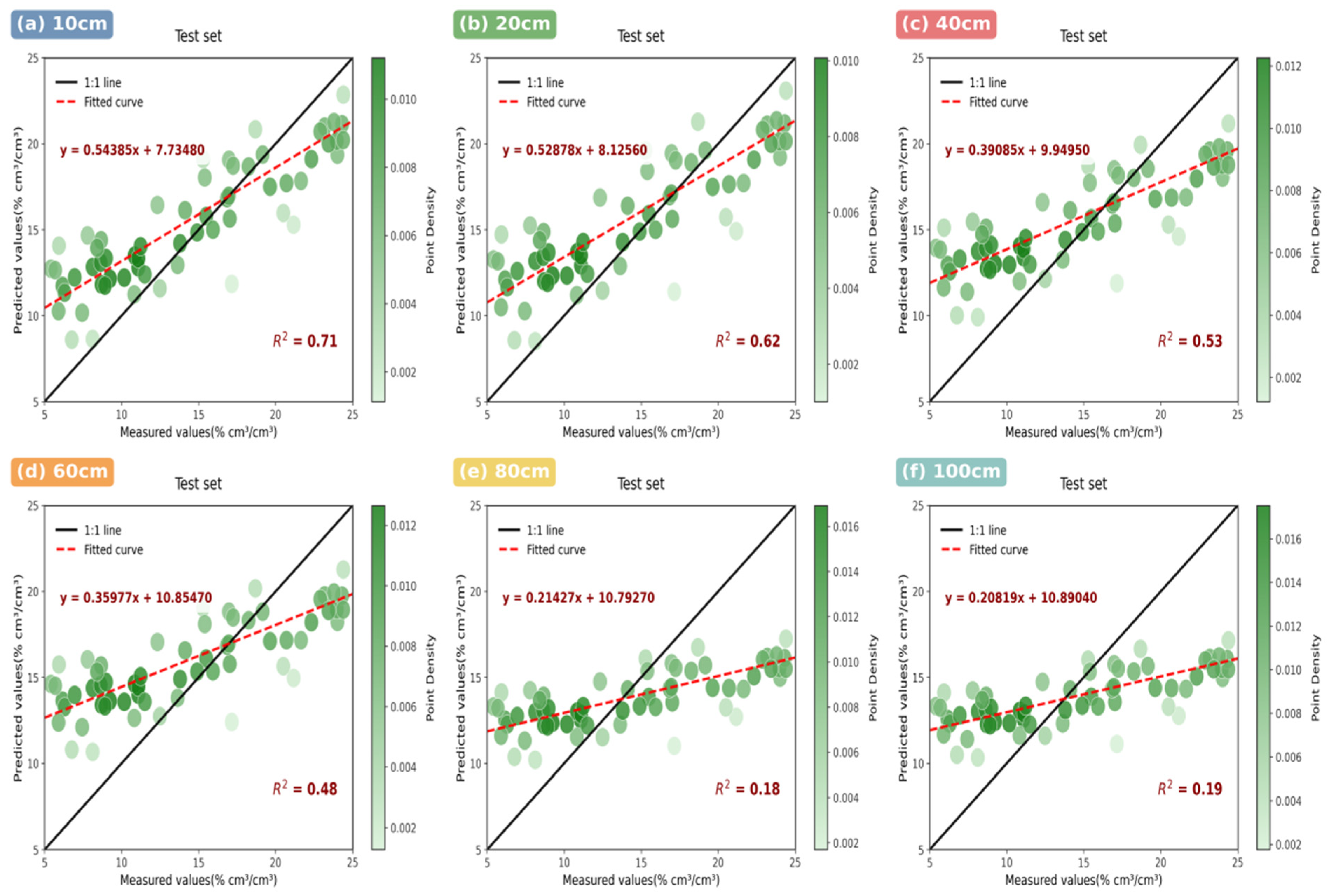
- UAV-based hyperspectral remote sensing with novel triple-band indices (MSR, RES) outperforms multispectral technology and traditional indices, achieving 18–32% higher correlation for soil moisture retrieval, especially in deep soil layers (>80 cm, R2 = 0.49 vs. 0.18 for multispectral).
- Irrigation intensity dominates the spatiotemporal dynamics of soil moisture, while nitrogen fertilization indirectly regulates water redistribution through root architectural adaptation rather than directly altering soil water-holding capacity.
- The identified optimal spectral region (450–760 nm) and developed inversion models provide a reliable technical solution for high-precision soil moisture monitoring in vegetated arid farmlands.
- The clarified water–nitrogen interaction mechanisms offer scientific guidance for integrated resource management, enabling 22 ± 4% water savings without yield loss in water-scarce agricultural systems.
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. In Situ Environmental Data Acquisition
- (1)
- Meteorological Data
- (2)
- Soil Environmental Indicators
2.3. UAV-Based Imagery Acquisition
2.4. Soil Moisture Inversion Methods
2.4.1. Spectral Indices Construction
Theoretical Derivation of the Triple-Band Form
Rationale for Form and Band Selection
- (1)
- Choice of Difference-Based Form
- (2)
- Definition of Scattering Reference Band Rk
- (3)
- Constraints for Target Bands (i,j)
Stability Evaluation of the Triple-Band Indices
2.4.2. Random Forest Modeling for Soil Moisture Inversion
Depth-Specific Modeling Strategy
Dataset Partitioning
Model Inputs and Preprocessing
Hyperparameter Optimization
2.5. Data Analysis and Model Evaluation Methods
3. Results
3.1. Hyperspectral Data Analysis and Sensitive Band Extraction
3.2. Soil Moisture Inversion and Validation
3.2.1. Hyperspectral Model Performance
3.2.2. Multispectral System Limitations
3.2.3. Comparative Sensor Analysis
3.3. Spatiotemporal Dynamics of Soil Moisture Under Water-Nitrogen Regulation
3.3.1. Depth-Dependent Variability in Moisture Content
3.3.2. Irrigation–Nitrogen Interaction Mechanisms
3.3.3. Agricultural Implementation Implications
4. Discussion
4.1. Technical Advantages and Modeling Rationale
4.2. Water–Nitrogen Interactions and Practical Value
4.3. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kang, S.; Hao, X.; Du, T.; Tong, L.; Su, X.; Lu, H.; Li, X.; Huo, Z.; Li, S.; Ding, R. Improving Agricultural Water Productivity to Ensure Food Security in China under Changing Environment: From Research to Practice. Agric. Water Manag. 2017, 179, 5–17. [Google Scholar] [CrossRef]
- Tsagkatakis, G.; Melebari, A.; Akbar, R.; Campbell, J.D.; Hodges, E.; Moghaddam, M. Deep Generative Regression Models for Soil Moisture Retrieval from GNSS-R Observations. In Proceedings of the 2023 International Conference on Electromagnetics in Advanced Applications (ICEAA), Venice, Italy, 9–13 October 2023; p. 291. [Google Scholar]
- Verma, P.; Kamble, A.; Barhate, A.; Tale, A.; Gudadhe, A. A Review on Soil Moisture Monitoring Methods Using Satellite Images. In Proceedings of the 2024 2nd DMIHER International Conference on Artificial Intelligence in Healthcare, Education and Industry (IDICAIEI), Wardha, India, 29–30 November 2024; pp. 1–6. [Google Scholar]
- Cui, Z.; Zhang, H.; Chen, X.; Zhang, C.; Ma, W.; Huang, C.; Zhang, W.; Mi, G.; Miao, Y.; Li, X.; et al. Pursuing Sustainable Productivity with Millions of Smallholder Farmers. Nature 2018, 555, 363–366. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Inanaga, S.; Li, Z.; Eneji, A.E. Optimizing Irrigation Scheduling for Winter Wheat in the North China Plain. Agric. Water Manag. 2005, 76, 8–23. [Google Scholar] [CrossRef]
- Hanjra, M.A.; Qureshi, M.E. Global Water Crisis and Future Food Security in an Era of Climate Change. Food Policy 2010, 35, 365–377. [Google Scholar] [CrossRef]
- Siebert, S.; Burke, J.; Faures, J.M.; Frenken, K.; Hoogeveen, J.; Doell, P.; Portmann, F.T. Groundwater Use for Irrigation—A Global Inventory. Hydrol. Earth Syst. Sci. 2010, 14, 1863–1880. [Google Scholar] [CrossRef]
- Blanka-Végi, V.; Tobak, Z.; Sipos, G.; Barta, K.; Szabó, B.; van Leeuwen, B. Estimation of the Spatiotemporal Variability of Surface Soil Moisture Using Machine Learning Methods Integrating Satellite and Ground-Based Soil Moisture and Environmental Data. Water Resour Manag. 2025, 39, 2317–2334. [Google Scholar] [CrossRef]
- Grant, S.; Harden, A.; Coggins, K.; Parker, D.; Tabei, F.; Askarian, B. A Comprehensive Comparison of IoT Soil Moisture Sensors for Agricultural Applications. In Proceedings of the 2024 IEEE 17th Dallas Circuits and Systems Conference (DCAS), Richardson, TX, USA, 19–21 April 2024; pp. 1–6. [Google Scholar]
- Xu, D.; Wang, C.; Chen, J.; Shen, M.; Shen, B.; Yan, R.; Li, Z.; Karnieli, A.; Chen, J.; Yan, Y.; et al. The Superiority of the Normalized Difference Phenology Index (NDPI) for Estimating Grassland Aboveground Fresh Biomass. Remote Sens. Environ. 2021, 264, 112578. [Google Scholar] [CrossRef]
- Mohseni, F.; Mirmazloumi, S.M.; Mokhtarzade, M.; Jamali, S.; Homayouni, S. Global Evaluation of SMAP/Sentinel-1 Soil Moisture Products. Remote Sens. 2022, 14, 4624. [Google Scholar] [CrossRef]
- Yan, K.; Pu, J.; Park, T.; Xu, B.; Zeng, Y.; Yan, G.; Weiss, M.; Knyazikhin, Y.; Myneni, R.B. Performance Stability of the MODIS and VIIRS LAI Algorithms Inferred from Analysis of Long Time Series of Products. Remote Sens. Environ. 2021, 260, 112438. [Google Scholar] [CrossRef]
- Shi, J.; Yang, H.; Hou, X.; Zhang, H.; Tang, G.; Zhao, H.; Wang, F. Coupling SAR and Optical Remote Sensing Data for Soil Moisture Retrieval over Dense Vegetation Covered Areas. PLoS ONE 2025, 20, e0315971. [Google Scholar] [CrossRef]
- Zeng, Y.; Hao, D.; Park, T.; Zhu, P.; Huete, A.; Myneni, R.; Knyazikhin, Y.; Qi, J.; Nemani, R.R.; Li, F.; et al. Structural Complexity Biases Vegetation Greenness Measures. Nat. Ecol. Evol. 2023, 7, 1790–1798. [Google Scholar] [CrossRef]
- Kurunathan, H.; Huang, H.; Li, K.; Ni, W.; Hossain, E. Machine Learning-Aided Operations and Communications of Unmanned Aerial Vehicles: A Contemporary Survey. IEEE Commun. Surv. Tutor. 2024, 26, 496–533. [Google Scholar] [CrossRef]
- Li, Z.-L.; Leng, P.; Zhou, C.; Chen, K.-S.; Zhou, F.-C.; Shang, G.-F. Soil Moisture Retrieval from Remote Sensing Measurements: Current Knowledge and Directions for the Future. Earth-Sci. Rev. 2021, 218, 103673. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, Y.; Zhou, F.; Huang, Z.; Yao, Y. More Precise Monitoring of Soil Moisture Content in Agricultural Fields by Upscaling Conversion of Multispectral Image Data from Unmanned Aerial Vehicles. IEEE Access 2024, 12, 79892–79904. [Google Scholar] [CrossRef]
- Liu, H.; Chen, J.; Bian, J.; Li, Z.; Zhang, W.; Yang, N.; Du, R.; Qian, L.; Geng, H.; Chen, Y.; et al. Enhancing Field-Scale Soil Moisture Content Monitoring Using UAV Hyperspectral-Derived Multi-Dimensional Spectral Response Indices of Crop Comprehensive Phenotypic Traits. Comput. Electron. Agric. 2025, 235, 110399. [Google Scholar] [CrossRef]
- Tulczyjew, L.; Ruszczak, B.; Myller, M.; Wijata, A.M.; Boguszewska-Mańkowska, D.; Nalepa, J. Convolutional Neural Networks Estimate Root-Zone Soil Moisture from Hyperspectral Images. In Proceedings of the 2024 IEEE International Conference on Visual Communications and Image Processing (VCIP), Tokyo, Japan, 8–11 December 2024; pp. 1–5. [Google Scholar]
- Kim, K.-S.; Lee, J.; Park, J.; Hong, G.; Lee, K. Hyperspectral Remote Sensing and Artificial Intelligence for High-Resolution Soil Moisture Prediction. Water 2025, 17, 3069. [Google Scholar] [CrossRef]
- Zhang, X.; Ram, B.; Vullaganti, N.; Aderholdt, W.; Overby, P.; Sun, X. Soil Moisture Classification Using Hyperspectral Imaging and Deep Learning Models on Ground Robot Vehicles. Smart Agric. Technol. 2025, 12, 101413. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Relationships between Leaf Pigment Content and Spectral Reflectance across a Wide Range of Species, Leaf Structures and Developmental Stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Babaeian, E.; Sadeghi, M.; Franz, T.E.; Jones, S.; Tuller, M. Mapping Soil Moisture with the OPtical TRApezoid Model (OPTRAM) Based on Long-Term MODIS Observations. Remote Sens. Environ. 2018, 211, 425–440. [Google Scholar] [CrossRef]
- Qi, G.; Yuzhen, W.; Chunhui, F. Remote Sensing Inversion of Desert Soil Moisture Based on Improved Spectral Indices. Remote Sens. Nat. Resour. 2022, 34, 142–150. [Google Scholar] [CrossRef]
- Li, Z.; Deng, C.; Zhao, B.; Tian, Y.; Huang, Y. Hyperspectral Inversion for Soil Moisture and Temperature Based on Gaussian Process Regression. In Proceedings of the 2019 IEEE International Conference on Signal, Information and Data Processing (ICSIDP), Chongquing, China, 11–13 December 2019; pp. 1–4. [Google Scholar]
- Wang, L.; Gao, Y. Soil Moisture Inversion Using Multi-Sensor Remote Sensing Data Based on Feature Selection Method and Adaptive Stacking Algorithm. Remote Sens. 2025, 17, 1569. [Google Scholar] [CrossRef]
- Hatfield, J.L. Spatial Patterns of Water and Nitrogen Response Within Corn Production Fields. In Agricultural Science; IntechOpen: London, UK, 2012; ISBN 978-953-51-0567-1. [Google Scholar][Green Version]
- Zhu, Q.; Castellano, M.J.; Yang, G. Coupling Soil Water Processes and the Nitrogen Cycle across Spatial Scales: Potentials, Bottlenecks and Solutions. Earth-Sci. Rev. 2018, 187, 248–258. [Google Scholar] [CrossRef]
- Wang, L.; Qu, J.J. NMDI: A Normalized Multi-Band Drought Index for Monitoring Soil and Vegetation Moisture with Satellite Remote Sensing. Geophys. Res. Lett. 2007, 34, L20405. [Google Scholar] [CrossRef]
- Rouse, J.W.; Haas, R.H.; Schell, J.A.; Deering, D.W. Monitoring Vegetation Systems in the Great Plains with ERTS. In Proceedings of the Third Earth Resources Technology Satellite-1 Symposium, Washington, DC, USA, 1 January 1974. [Google Scholar]
- Rondeaux, G.; Steven, M.; Baret, F. Optimization of Soil-Adjusted Vegetation Indices. Remote Sens. Environ. 1996, 55, 95–107. [Google Scholar] [CrossRef]
- Mishra, S.; Mishra, D.R. Normalized Difference Chlorophyll Index: A Novel Model for Remote Estimation of Chlorophyll-a Concentration in Turbid Productive Waters. Remote Sens. Environ. 2012, 117, 394–406. [Google Scholar] [CrossRef]
- Xue, L.; Cao, W.; Luo, W.; Dai, T.; Zhu, Y. Monitoring Leaf Nitrogen Status in Rice with Canopy Spectral Reflectance. Agron. J. 2004, 96, 135–142. [Google Scholar] [CrossRef]
- Huete, A.R. A Soil-Adjusted Vegetation Index (SAVI). Remote Sens. Environ. 1988, 25, 295–309. [Google Scholar] [CrossRef]
- Penuelas, J.; Baret, F.; Filella, I. Semi-Empirical Indices to Assess Carotenoids/Chlorophyll-a Ratio from Leaf Spectral Reflectance. Photosynthetica 1995, 31, 221–230. [Google Scholar]
- Broge, N.H.; Leblanc, E. Comparing Prediction Power and Stability of Broadband and Hyperspectral Vegetation Indices for Estimation of Green Leaf Area Index and Canopy Chlorophyll Density. Remote Sens. Environ. 2001, 76, 156–172. [Google Scholar] [CrossRef]
- Huete, A.; Didan, K.; Miura, T.; Rodriguez, E.P.; Gao, X.; Ferreira, L.G. Overview of the Radiometric and Biophysical Performance of the MODIS Vegetation Indices. Remote Sens. Environ. 2002, 83, 195–213. [Google Scholar] [CrossRef]
- Haboudane, D.; Miller, J.R.; Tremblay, N.; Zarco-Tejada, P.J.; Dextraze, L. Integrated Narrow-Band Vegetation Indices for Prediction of Crop Chlorophyll Content for Application to Precision Agriculture. Remote Sens. Environ. 2002, 81, 416–426. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Berjón, A.; López-Lozano, R.; Miller, J.R.; Martín, P.; Cachorro, V.; González, M.R.; de Frutos, A. Assessing Vineyard Condition with Hyperspectral Indices: Leaf and Canopy Reflectance Simulation in a Row-Structured Discontinuous Canopy. Remote Sens. Environ. 2005, 99, 271–287. [Google Scholar] [CrossRef]
- Qi, J.; Kerr, Y.H.; Moran, M.S.; Weltz, M.; Huete, A.R.; Sorooshian, S.; Bryant, R. Leaf Area Index Estimates Using Remotely Sensed Data and BRDF Models in a Semiarid Region. Remote Sens. Environ. 2000, 73, 18–30. [Google Scholar] [CrossRef]
- Aschonitis, V.G.; Lekakis, E.; Tziachris, P.; Doulgeris, C.; Papadopoulos, F.; Papadopoulos, A.; Papamichail, D. A Ranking System for Comparing Models’ Performance Combining Multiple Statistical Criteria and Scenarios: The Case of Reference Evapotranspiration Models. Environ. Model. Softw. 2019, 114, 98–111. [Google Scholar] [CrossRef]
- Yu, K.; Belwalkar, A.; Wang, W.; Hu, Y.; Hunegnaw, A.; Nurunnabi, A.; Ruf, T.; Li, F.; Jia, L.; Kooistra, L.; et al. UAV Hyperspectral Remote Sensing for Crop Nitrogen Monitoring: Progress, Challenges, and Perspectives. Smart Agric. Technol. 2025, 12, 101507. [Google Scholar] [CrossRef]
- Verrelst, J.; Schaepman, M.E.; Koetz, B.; Kneubühler, M. Angular Sensitivity Analysis of Vegetation Indices Derived from CHRIS/PROBA Data. Remote Sens. Environ. 2008, 112, 2341–2353. [Google Scholar] [CrossRef]
- Yuan, J.; Liu, Y.; Yan, C.; Hu, C.; Xu, J. Construction of Soil Moisture Three-Band Indices with Vis-NIR Spectroscopy Based on the Kubelka-Munk and Hapke Model. Measurement 2025, 249, 116979. [Google Scholar] [CrossRef]
- Zhang, M.; Lang, F.; Zheng, N. Soil Moisture Retrieval during the Wheat Growth Cycle Using SAR and Optical Satellite Data. Water 2021, 13, 135. [Google Scholar] [CrossRef]
- Haboudane, D.; Miller, J.R.; Pattey, E.; Zarco-Tejada, P.J.; Strachan, I.B. Hyperspectral Vegetation Indices and Novel Algorithms for Predicting Green LAI of Crop Canopies: Modeling and Validation in the Context of Precision Agriculture. Remote Sens. Environ. 2004, 90, 337–352. [Google Scholar] [CrossRef]
- Babaeian, E.; Paheding, S.; Siddique, N.; Devabhaktuni, V.K.; Tuller, M. Estimation of Root Zone Soil Moisture from Ground and Remotely Sensed Soil Information with Multisensor Data Fusion and Automated Machine Learning. Remote Sens. Environ. 2021, 260, 112434. [Google Scholar] [CrossRef]
- Peng, J.; Shen, H.; He, S.W.; Wu, J.S. Soil Moisture Retrieving Using Hyperspectral Data with the Application of Wavelet Analysis. Env. Earth Sci. 2013, 69, 279–288. [Google Scholar] [CrossRef]
- Li, J.; Yang, J. Research Advances in the Effects of Water, Nitrogen and Their Interaction on the Yield, Water and Nitrogen Use Efficiencies of Rice. Chin. J. Rice Sci. 2017, 31, 327. [Google Scholar] [CrossRef]
- Li, J.; Cheng, T.; Ma, X.; Hou, X.; Wu, H.; Yang, L. Effect of Nitrogen Functional Groups on Competitive Adsorption between Toluene and Water Vapor onto Nitrogen-Doped Spherical Resorcinol-Formaldehyde Resin-Based Activated Carbon. Environ. Sci. Pollut. Res. 2022, 29, 85257–85270. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Bruning, B.; Garnett, T.; Berger, B. The Performances of Hyperspectral Sensors for Proximal Sensing of Nitrogen Levels in Wheat. Sensors 2020, 20, 4550. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, X.; Tian, F. Study on Soil Moisture Status of Soybean and Corn across the Whole Growth Period Based on UAV Multimodal Remote Sensing. Remote Sens. 2024, 16, 3166. [Google Scholar] [CrossRef]
- Quemada, M.; Gabriel, J.L. Approaches for Increasing Nitrogen and Water Use Efficiency Simultaneously. Glob. Food Secur. 2016, 9, 29–35. [Google Scholar] [CrossRef]
- Barker, J.B.; Bhatti, S.; Heeren, D.M.; Neale, C.M.U.; Rudnick, D.R. Variable Rate Irrigation of Maize and Soybean in West-Central Nebraska Under Full and Deficit Irrigation. Front. Big Data 2019, 2, 34. [Google Scholar] [CrossRef]
- Zhao, D.; Shen, J.; Lang, K.; Liu, Q.; Li, Q. Effects of Irrigation and Wide-Precision Planting on Water Use, Radiation Interception, and Grain Yield of Winter Wheat in the North China Plain. Agric. Water Manage. 2013, 118, 87–92. [Google Scholar] [CrossRef]
- Sishodia, R.P.; Ray, R.L.; Singh, S.K. Applications of Remote Sensing in Precision Agriculture: A Review. Remote Sens. 2020, 12, 3136. [Google Scholar] [CrossRef]
- Weiss, M.; Jacob, F.; Duveiller, G. Remote Sensing for Agricultural Applications: A Meta-Review. Remote Sens. Environ. 2020, 236, 111402. [Google Scholar] [CrossRef]
- Qiu, J.; Crow, W.T.; Wagner, W.; Zhao, T. Effect of Vegetation Index Choice on Soil Moisture Retrievals via the Synergistic Use of Synthetic Aperture Radar and Optical Remote Sensing. Int. J. Appl. Earth Obs. Geoinf. 2019, 80, 47–57. [Google Scholar] [CrossRef]
- Patil, N.G.; Singh, S.K. Pedotransfer Functions for Estimating Soil Hydraulic Properties: A Review. Pedosphere 2016, 26, 417–430. [Google Scholar] [CrossRef]









| Date | 5.4 | 6.3 | 7.7 | 7.16 | 7.29 | 8.12 | 8.31 | Total (mm) |
|---|---|---|---|---|---|---|---|---|
| W1N1 | 80 | 38.49 | 21.86 | 68.27 | 75.69 | 101.46 | 90.95 | 476.71 |
| W1N2 | 80 | 33.53 | 23.34 | 65.2 | 66.59 | 92.07 | 99.52 | 460.23 |
| W1N3 | 80 | 34.1 | 30.39 | 59.92 | 68.8 | 90.65 | 87.59 | 451.44 |
| W2N1 | 80 | 29.6 | 19.64 | 50.46 | 55.47 | 88.15 | 82.23 | 405.56 |
| W2N2 | 80 | 27.1 | 18.99 | 50.61 | 55.51 | 80 | 74.29 | 386.49 |
| W2N3 | 80 | 27.9 | 24.87 | 53.78 | 62.31 | 91.04 | 82.61 | 422.52 |
| W3N1 | 80 | 19.6 | 16.19 | 32.1 | 40.33 | 60.91 | 54.76 | 303.9 |
| W3N2 | 80 | 15.41 | 22.48 | 31.86 | 37.6 | 58.68 | 52.22 | 298.24 |
| W3N3 | 80 | 16.86 | 15.05 | 33.65 | 41.57 | 58.69 | 53.32 | 299.14 |
| CK | 80 | 14.94 | 16.72 | 33.65 | 37.6 | 57.82 | 53.47 | 294.19 |
| Reproductive Stage | Seedling Stage | Jointing Phase | Heading Period | Grout Period | Maturity |
|---|---|---|---|---|---|
| fertilization ratio | 20% | 30% | 30% | 20% | 0% |
| Soil Layer | Dry Bulk Density (g/cm3) | Field Capacity (cm3/cm3) |
|---|---|---|
| 0–20 cm | 1.61 | 32.18% |
| 20–40 cm | 1.57 | |
| 40–60 cm | 1.66 | |
| 60–80 cm | 1.55 | |
| 80–100 cm | 1.52 |
| Sensor | Spectral | Formulation |
|---|---|---|
| Multispectral | Normalized difference vegetation index (NDVI) [30] | NDVI = (NIR − R)/(NIR + R) |
| Optimized soil-adjusted vegetation index (OSAVI) [31] | OSAVI = 1.16(NIR − R)/(NIR + R + 0.16) | |
| Ratio vegetation index (RVI) [32] | RVI = NIR/R | |
| Ratio vegetation index 2 (RVI2) [33] | RVI2 = NIR/G | |
| Soil-adjusted vegetation index (SAVI) [34] | SAVI = 1.5(NIR − R)(NIR + R + 0.5) | |
| Structure insensitive pigment index (SIPI) [35] | SIPI = (NIR − B)/(NIR + B) | |
| Triangular vegetation index (TVI) [36] | TVI = 60(NIR − G) − 100(R − G) | |
| Enhanced vegetation index (EVI) [37] | EVI = 2.5(NIR − R)/(NIR + 6R − 7.5B + 1) | |
| Modified chlorophyll absorption in reflectance index (MCARI) [38] | MCARI = [(RE − R) − 0.2(RE − G)](RE/R) | |
| Transformed chlorophyll absorption in reflectance index (TCARI) [38] | TCARI = 3[(RE − R) − 0.2(RE − G)(RE/R)] | |
| Green index (GI) [39] | GI = G/R | |
| Green normalized difference vegetation index (GNDVI) [40] | GNDVI = (NIR − G)/(NIR + G) | |
| Simple ratio pigment index (SRPI) [41] | SRPI = B/R | |
| Normalized pigment chlorophyll index (NPCI) [42] | NPCI = (R − B)/(R + B) | |
| Normalized difference vegetation index 2 (NDVIgb) [43] | NDVIgb = (G − B)/(G + B) |
| Soil Layer | NDI | R | RI | R | DI | R | MSR | R | RES | R | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Jointing stage | 10 cm | (774 nm, 810 nm) | 0.56 | (774 nm, 810 nm) | 0.56 | (902 nm, 950 nm) | 0.63 | (510 nm, 514 nm, 666 nm) | 0.67 | (454 nm, 510 nm, 666 nm) | 0.66 |
| 20 cm | (786 nm, 842 nm) | 0.74 | (786 nm, 842 nm) | 0.74 | (790 nm, 842 nm) | 0.74 | (786 nm, 842 nm, 910 nm) | 0.74 | (786 nm, 842 nm, 910 nm) | 0.74 | |
| 40 cm | (790 nm, 810 nm) | 0.54 | (790 nm, 810 nm) | 0.54 | (870 nm, 910 nm) | 0.56 | (510 nm, 530 nm, 666 nm) | 0.65 | (506 nm, 510 nm, 666 nm) | 0.66 | |
| 60 cm | (774 nm, 778 nm) | 0.54 | (774 nm,778 nm) | 0.54 | (774 nm, 778 nm) | 0.49 | (526 nm, 570 nm, 706 nm) | 0.55 | (526 nm, 570 nm, 706 nm) | 0.54 | |
| 80 cm | (754 nm, 758 nm) | 0.53 | (754 nm, 758 nm) | 0.53 | (782 nm, 830 nm) | 0.49 | (734 nm, 754 nm, 758 nm) | 0.59 | (734 nm, 754 nm, 758 nm) | 0.59 | |
| 100 cm | (478 nm, 482 nm) | 0.41 | (478 nm, 482 nm) | 0.41 | (686 nm, 690 nm) | 0.43 | (638 nm, 670 nm, 902 nm) | 0.46 | (638 nm, 670 nm, 902 nm) | 0.46 | |
| Tasseling stage | 10 cm | (586 nm, 590 nm) | 0.85 | (586 nm, 590 nm) | 0.85 | (786 nm, 926 nm) | 0.81 | (626 nm, 682 nm, 706 nm) | 0.9 | (626 nm, 682 nm, 706 nm) | 0.9 |
| 20 cm | (550 nm, 586 nm) | 0.82 | (550 nm, 586 nm) | 0.82 | (790 nm, 950 nm) | 0.74 | (562 nm, 622 nm, 682 nm) | 0.86 | (558 nm, 642 nm, 678 nm) | 0.87 | |
| 40 cm | (566 nm, 590 nm) | 0.68 | (566 nm,590 nm) | 0.68 | (794 nm,918 nm) | 0.64 | (566 nm, 610 nm, 678 nm) | 0.75 | (566 nm, 610 nm, 678 nm) | 0.76 | |
| 60 cm | (538 nm, 586 nm) | 0.68 | (538 nm, 586 nm) | 0.68 | (530 nm, 582 nm) | 0.59 | (562 nm, 586 nm, 666 nm) | 0.72 | (562 nm, 586 nm, 682 nm) | 0.73 | |
| 80 cm | (570 nm, 578 nm) | 0.51 | (570 nm,578 nm) | 0.51 | (538 nm,554 nm) | 0.47 | (570 nm, 598 nm, 686 nm) | 0.62 | (570 nm, 598 nm, 686 nm) | 0.62 | |
| 100 cm | (454 nm, 490 nm) | 0.64 | (454 nm, 490 nm) | 0.65 | (454 nm, 490 nm) | 0.61 | (610 nm, 614 nm, 694 nm) | 0.82 | (838 nm, 858 nm, 866 nm) | 0.82 | |
| Grain filling stage | 10 cm | (542 nm, 702 nm) | 0.79 | (542 nm, 702 nm) | 0.79 | (542 nm, 702 nm) | 0.79 | (542 nm, 634 nm, 662 nm) | 0.82 | (542 nm, 634 nm, 662 nm) | 0.84 |
| 20 cm | (538 nm, 578 nm) | 0.77 | (538 nm, 578 nm) | 0.77 | (538 nm, 602 nm) | 0.75 | (526 nm, 538 nm, 578 nm) | 0.81 | (538 nm, 602 nm, 626 nm) | 0.81 | |
| 40 cm | (538 nm, 586 nm) | 0.7 | (538 nm, 586 nm) | 0.71 | (538 nm, 586 nm) | 0.7 | (538 nm, 550 nm, 682 nm) | 0.78 | (538 nm, 550 nm, 682 nm) | 0.78 | |
| 60 cm | (466 nm, 602 nm) | 0.55 | (466 nm, 602 nm) | 0.56 | (466 nm, 510 nm) | 0.52 | (542 nm, 546 nm, 566 nm) | 0.66 | (770 nm, 830 nm, 838 nm) | 0.69 | |
| 80 cm | (462 nm, 494 nm) | 0.47 | (462 nm, 494 nm) | 0.47 | (462 nm, 494 nm) | 0.45 | (866 nm, 878 nm, 918 nm) | 0.46 | (554 nm, 666 nm, 674 nm) | 0.56 | |
| 100 cm | (542 nm, 578 nm) | 0.56 | (542 nm, 578 nm) | 0.56 | (542 nm, 578 nm) | 0.54 | (514 nm, 526 nm, 674 nm) | 0.66 | (542 nm, 578 nm, 598 nm) | 0.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sun, M.; Su, K.; Tian, F. Monitoring Spatiotemporal Dynamics of Soil Moisture Under Water-Nitrogen Interactions in Arid Farmland Using UAV-Based Hyperspectral Sensing and Triple-Band Indices. Remote Sens. 2026, 18, 726. https://doi.org/10.3390/rs18050726
Sun M, Su K, Tian F. Monitoring Spatiotemporal Dynamics of Soil Moisture Under Water-Nitrogen Interactions in Arid Farmland Using UAV-Based Hyperspectral Sensing and Triple-Band Indices. Remote Sensing. 2026; 18(5):726. https://doi.org/10.3390/rs18050726
Chicago/Turabian StyleSun, Minghui, Kaikai Su, and Fei Tian. 2026. "Monitoring Spatiotemporal Dynamics of Soil Moisture Under Water-Nitrogen Interactions in Arid Farmland Using UAV-Based Hyperspectral Sensing and Triple-Band Indices" Remote Sensing 18, no. 5: 726. https://doi.org/10.3390/rs18050726
APA StyleSun, M., Su, K., & Tian, F. (2026). Monitoring Spatiotemporal Dynamics of Soil Moisture Under Water-Nitrogen Interactions in Arid Farmland Using UAV-Based Hyperspectral Sensing and Triple-Band Indices. Remote Sensing, 18(5), 726. https://doi.org/10.3390/rs18050726





