Foliar Application of Biochar-Based Suspensions: Effects on Composition and Sensory Properties of Malvazija istarska (Vitis vinifera L.) Must and Wine
Abstract
1. Introduction
2. Materials and Methods
2.1. Biochar Production from Grapevine Pruning Residues
2.2. Foliar Suspensions
2.3. Foliar Application of Aqueous Suspensions in Vineyard
2.4. Harvest and Vinification Process
2.5. Analysis of Key Compositional Parameters in Must and Wine
2.6. Determination of Wine Sensory Properties
2.7. Statistical Analysis
3. Results
3.1. Foliar Suspensions pH and Electrical Conductivity Values
3.2. Chemical Composition of Must
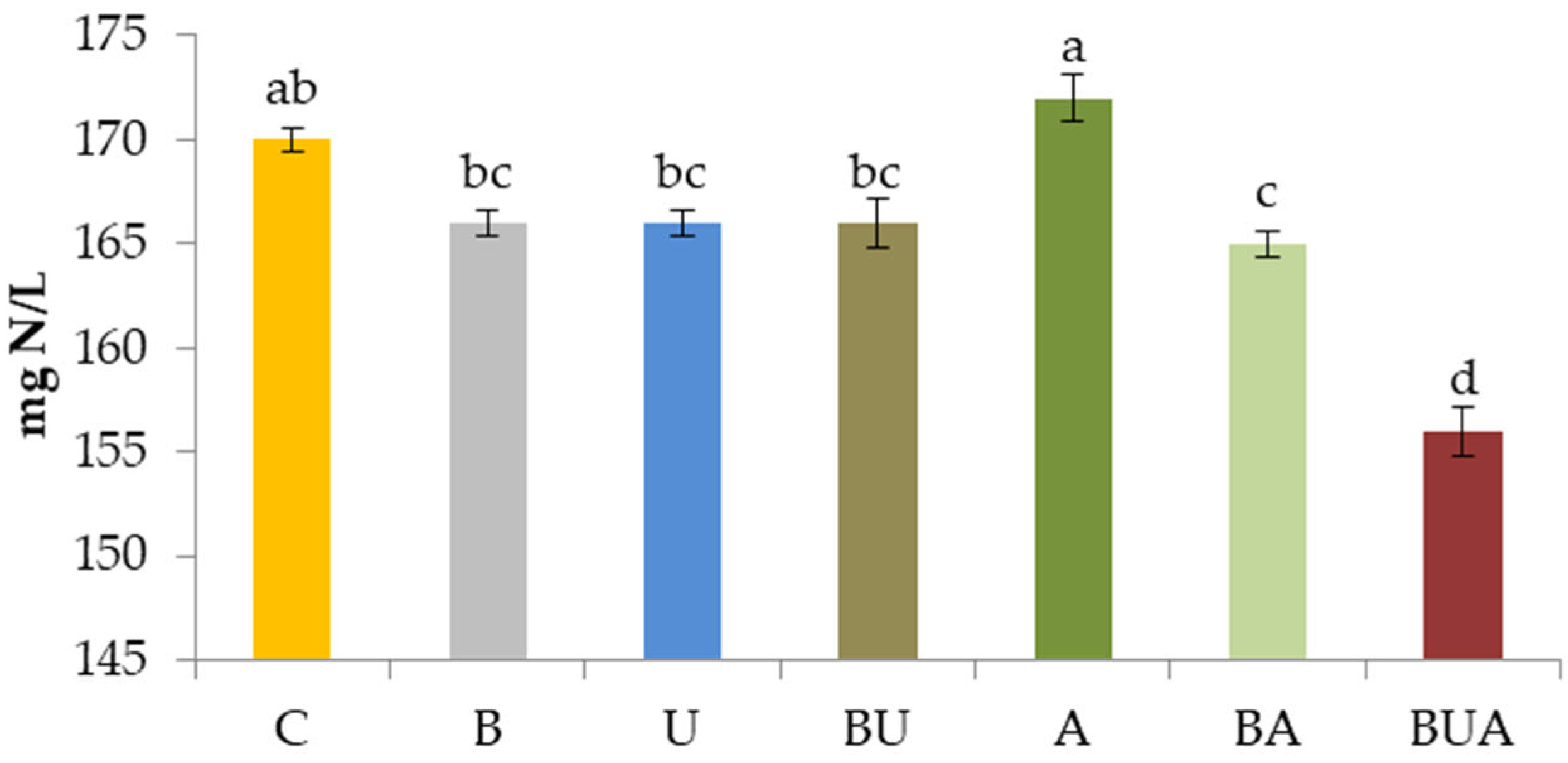
3.2.1. Nitrogen-Related Compounds in Must: Yeast Assimilable Nitrogen and Amino Acids
3.2.2. Carbon-Related Compounds in Must: Sugars and Organic Acids
3.3. Wine Characterization and Sensory Properties
3.3.1. Chemical Analysis of Wine
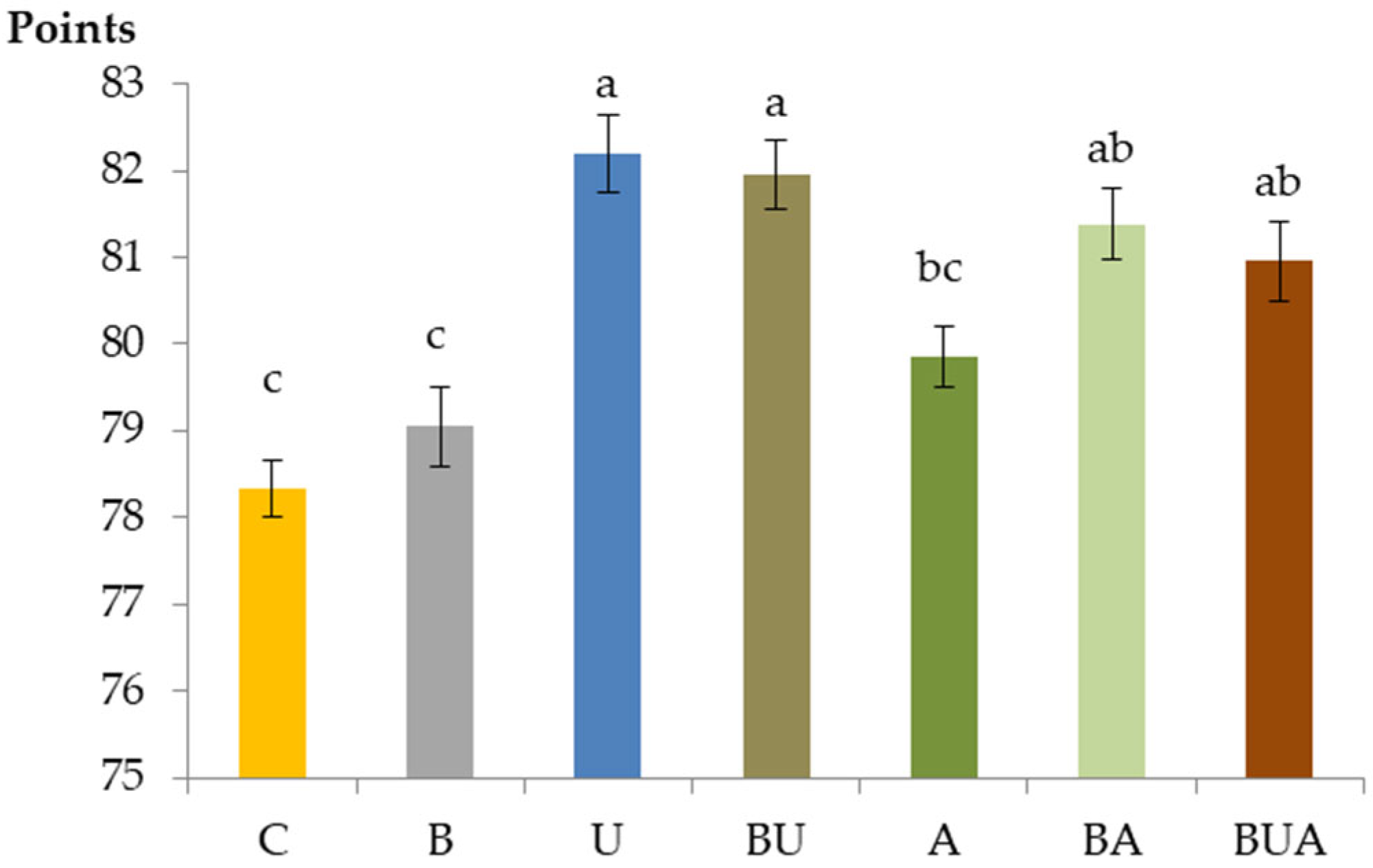
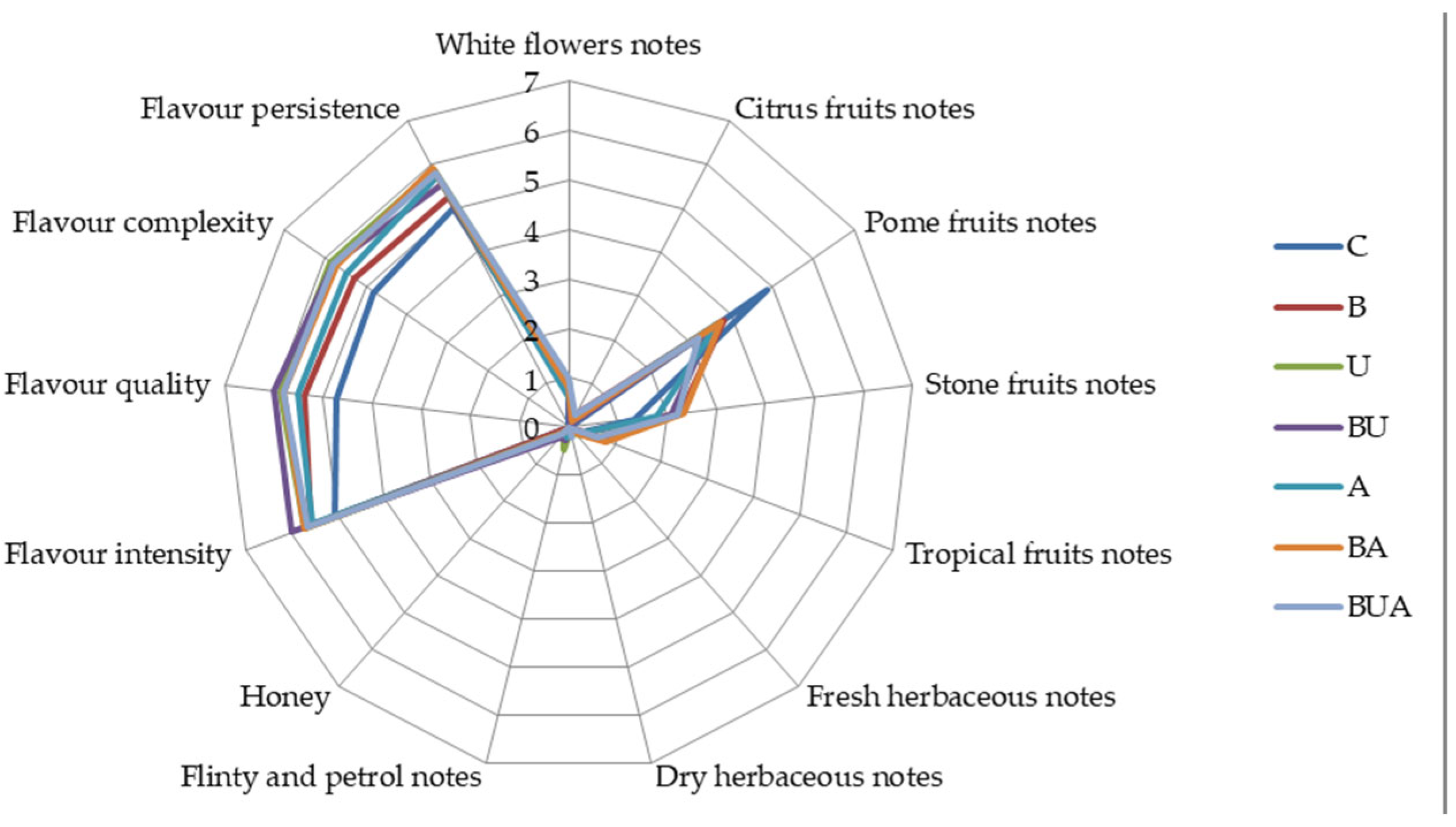
3.3.2. Sensory Evaluation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BC | Biochar |
| GPRs | Grapevine Pruning Residues |
| YAN | Yeast Assimilable Nitrogen |
| EC | Electrical Conductivity |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| LC-MS/MS | Liquid Chromatography Coupled with Tandem Mass Spectrometry |
| HPLC | High-Performance Liquid Chromatography |
| OIV | Organisation Internationale de la Vigne et du Vin |
| QDA | Quantitative Descriptive Analysis |
| ANOVA | Analysis of Variance |
| HSD | Honestly Significant Difference |
References
- Lippi, P.; Mattii, G.B.; Cataldo, E. Biochar, Properties and Skills with a Focus on Implications for Vineyard Land and Grapevine Performance. Phyton-Int. J. Exp. Bot. 2025, 94, 33–64. [Google Scholar] [CrossRef]
- Fu, X.; Chen, X.; Chen, Y.; Hui, Y.; Wang, R.; Wang, Y. Foliar Co-Applications of Nitrogen and Iron on Vines at Different Developmental Stages Impacts Wine Grape (Vitis vinifera L.) Composition. Plants 2024, 13, 2203. [Google Scholar] [CrossRef]
- Prasad, R. Fertilizer Urea, Food Security, Health and the Environment. Curr. Sci. 1998, 75, 677–683. [Google Scholar]
- Haytova, D. A Review of Foliar Fertilization of Some Vegetables Crops. Annu. Rev. Res. Biol. 2013, 3, 455–465. [Google Scholar]
- Dong, S.; Neilsen, D.; Neilsen, G.H.; Fuchigami, L.H. Foliar N Application Reduces Soil NO3−-N Leaching Loss in Apple Orchards. Plant Soil 2005, 268, 357–366. [Google Scholar] [CrossRef]
- Teixeira, W.F.; Fagan, E.B.; Soares, L.H.; Soares, J.N.; Reichardt, K.; Neto, D.D. Seed and Foliar Application of Amino Acids Improve Variables of Nitrogen Metabolism and Productivity in Soybean Crop. Front. Plant Sci. 2018, 9, 396. [Google Scholar] [CrossRef]
- Kumari, A.; Lakshmi, G.A.; Krishna, G.K.; Patni, B.; Prakash, S.; Bhattacharyya, M.; Singh, S.K.; Verma, K.K. Climate Change and Its Impact on Crops: A Comprehensive Investigation for Sustainable Agriculture. Agronomy 2022, 12, 3008. [Google Scholar] [CrossRef]
- Weber, K.; Quicker, P. Properties of Biochar. Fuel 2018, 217, 240–261. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, Modification and Environmental Application of Biochar: A Review. J. Clean. Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Kuppusamy, S.; Thavamani, P.; Megharaj, M.; Venkateswarlu, K.; Naidu, R. Agronomic and Remedial Benefits and Risks of Applying Biochar to Soil: Current Knowledge and Future Research Directions. Environ. Int. 2016, 87, 1–12. [Google Scholar] [CrossRef]
- Prelac, M.; Palcic, I.; Cvitan, D.; Andelini, D.; Repaji’c, M.; Curko, J.; Kovaˇcevi’c, T.K.; Goreta Ban, S.; Užila, Z.; Ban, D.; et al. Biochar from Grapevine Pruning Residues as an Efficient Adsorbent of Polyphenolic Compounds. Materials 2023, 16, 4716. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.; Ma, B.; Ai, L.; Tian, X.; Zhang, L. Foliar Spraying of Selenium Combined with Biochar Alleviates Cadmium Toxicity in Peanuts and Enriches Selenium in Peanut Grains. Int. J. Environ. Res. Public Health 2022, 19, 3542. [Google Scholar] [CrossRef]
- Kumar, A.; Joseph, S.; Graber, E.R.; Taherymoosavi, S.; Mitchell, D.R.G.; Munroe, P.; Tsechansky, L.; Lerdahl, O.; Aker, W.; Sæbø, M. Fertilizing Behavior of Extract of Organomineral-Activated Biochar: Low-Dose Foliar Application for Promoting Lettuce Growth. Chem. Biol. Technol. Agric. 2021, 8, 21. [Google Scholar] [CrossRef]
- Dayoub, E.B.; Tóth, Z.; Soós, G.; Anda, A. Chemical and Physical Properties of Selected Biochar Types and a Few Application Methods in Agriculture. Agronomy 2024, 14, 2540. [Google Scholar] [CrossRef]
- Palčić, I.; Anđelini, D.; Prelac, M.; Pasković, I.; Černe, M.; Major, N.; Goreta Ban, S.; Užila, Z.; Bubola, M.; Ban, D.; et al. Influence of Biochar Foliar Application on Malvazija Istarska Grapevine Physiology. Sustainability 2025, 17, 5947. [Google Scholar] [CrossRef]
- Anđelini, D.; Cvitan, D.; Prelac, M.; Pasković, I.; Černe, M.; Nemet, I.; Major, N.; Ban, S.G.; Užila, Z.; Zubin Ferri, T.; et al. Biochar from Grapevine-Pruning Residues Is Affected by Grapevine Rootstock and Pyrolysis Temperature. Sustainability 2023, 15, 4851. [Google Scholar] [CrossRef]
- DIN ISO 10390; 2021 Soil Quality—Determination of PH. Beuth Verlag: Berlin, Germany, 2021.
- Wang, M.; Sun, X.; Zhong, N.; Cai, D.; Wu, Z. Promising Approach for Improving Adhesion Capacity of Foliar Nitrogen Fertilizer. ACS Sustain. Chem. Eng. 2015, 3, 499–506. [Google Scholar] [CrossRef]
- COOMBE, B.G. Growth Stages of the Grapevine: Adoption of a System for Identifying Grapevine Growth Stages. Aust. J. Grape Wine Res. 1995, 1, 104–110. [Google Scholar] [CrossRef]
- Plavša, T.; Bubola, M.; Jeromel, A.; Tomaz, I.; Krapac, M. Exploring the Potential of Indigenous Grape Varieties for Sparkling Wine Production in the Hrvatska Istra Subregion (Croatia). Beverages 2025, 11, 78. [Google Scholar] [CrossRef]
- Ding, Y.; Wei, R.; Wang, L.; Wang, W.; Wang, H.; Li, H. Exploring the Ecological Characteristics of Natural Microbial Communities along the Continuum from Grape Berries to Winemaking. Food Res. Int. 2023, 167, 112718. [Google Scholar] [CrossRef]
- International Organization of Vine and Wine. OIV Standard for International Wine and Spirituous Beverages of Vitivinicultural Origin Competitions; OIV: Dijon, France, 2021. [Google Scholar]
- Petoumenou, D.G.; Liava, V. Sustainable Foliar Applications to Improve Grapevine Responses to Drought, High Temperatures, and Salinity: Impacts on Physiology, Yields, and Berry Quality. Plants 2025, 14, 2157. [Google Scholar] [CrossRef] [PubMed]
- Samarakoon, U.; Palmer, J.; Ling, P.; Altland, J. Effects of Electrical Conductivity, PH, and Foliar Application of Calcium Chloride on Yield and Tipburn of Lactuca Sativa Grown Using the Nutrient-Film Technique. HortScience 2020, 55, 1265–1271. [Google Scholar] [CrossRef]
- Tsouvaltzis, P.; Kasampali, D.S.; Aktsoglou, D.C.; Barbayiannis, N.; Siomos, A.S. Effect of Reduced Nitrogen and Supplemented Amino Acids Nutrient Solution on the Nutritional Quality of Baby Green and Red Lettuce Grown in a Floating System. Agronomy 2020, 10, 922. [Google Scholar] [CrossRef]
- Valero, E.; Millan, C.; Orteg, J.M.; Mauricio, J.C. Concentration of Amino Acids in Wine after the End of Fermentation by Saccharomyces Cerevisiae Strains. J. Sci. Food Agric. 2003, 83, 830–835. [Google Scholar] [CrossRef]
- Bouloumpasi, E.; Skendi, A.; Soufleros, E.H. Survey on Yeast Assimilable Nitrogen Status of Musts from Native and International Grape Varieties: Effect of Variety and Climate. Fermentation 2023, 9, 830–835. [Google Scholar] [CrossRef]
- Christofi, S.; Papanikolaou, S.; Dimopoulou, M.; Terpou, A.; Cioroiu, I.B.; Cotea, V.; Kallithraka, S. Effect of Yeast Assimilable Nitrogen Content on Fermentation Kinetics, Wine Chemical Composition and Sensory Character in the Production of Assyrtiko Wines. Appl. Sci. 2022, 12, 1405. [Google Scholar] [CrossRef]
- Su, Y.; Heras, J.M.; Gamero, A.; Querol, A.; Guillamón, J.M. Impact of Nitrogen Addition on Wine Fermentation by S. Cerevisiae Strains with Different Nitrogen Requirements. J. Agric. Food Chem. 2021, 69, 6022–6031. [Google Scholar] [CrossRef]
- Mataffo, A.; Scognamiglio, P.; Dente, A.; Strollo, D.; Colla, G.; Rouphael, Y.; Basile, B. Foliar Application of an Amino Acid-Enriched Urea Fertilizer on ‘Greco’ Grapevines at Full Veraison Increases Berry Yeast-Assimilable Nitrogen Content. Plants 2020, 9, 619. [Google Scholar] [CrossRef]
- Yao, J.; Wang, Z.; Liu, M.; Bai, B.; Zhang, C. Nitrate-Nitrogen Adsorption Characteristics and Mechanisms of Various Garden Waste Biochars. Materials 2023, 16, 5726. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, H.; Liu, J.; Wang, X.; Li, J.; Shi, E.; Wang, C.; Yang, J.; Zhang, Z. A Study on and Adsorption Mechanism of Ammonium Nitrogen by Modified Corn Straw Biochar. R. Soc. Open Sci. 2023, 10, 221535. [Google Scholar] [CrossRef]
- Dey, D.; Mavi, M.S. Biochar and Urea Co-Application Regulates Nitrogen Availability in Soil. Environ. Monit. Assess. 2021, 193, 326. [Google Scholar] [CrossRef]
- Tahim, C.M.; Mansfield, A.K. Yeast Assimilable Nitrogen Optimization for Cool-Climate Riesling. Am. J. Enol. Vitic. 2019, 70, 127–138. [Google Scholar] [CrossRef]
- Nisbet, M.A.; Martinson, T.E.; Mansfield, A.K. Accumulation and Prediction of Yeast Assimilable Nitrogen in New York Winegrape Cultivars. Am. J. Enol. Vitic. 2014, 65, 325–332. [Google Scholar] [CrossRef]
- Gutiérrez, A.; Chiva, R.; Sancho, M.; Beltran, G.; Arroyo-López, F.N.; Guillamon, J.M. Nitrogen Requirements of Commercial Wine Yeast Strains during Fermentation of a Synthetic Grape Must. Food Microbiol. 2012, 31, 25–32. [Google Scholar] [CrossRef]
- Fairbairn, S.; McKinnon, A.; Musarurwa, H.T.; Ferreira, A.C.; Bauer, F.F. The Impact of Single Amino Acids on Growth and Volatile Aroma Production by Saccharomyces Cerevisiae Strains. Front. Microbiol. 2017, 8, 2554. [Google Scholar] [CrossRef]
- Núñez, L.; Serratosa, M.P.; Godoy, A.; Fariña, L.; Dellacassa, E.; Moyano, L. Comparison of Physicochemical Properties, Amino Acids, Mineral Elements, Total Phenolic Compounds, and Antioxidant Capacity of Cuban Fruit and Rice Wines. Food Sci. Nutr. 2021, 9, 3673–3682. [Google Scholar] [CrossRef] [PubMed]
- Kovačević Ganić, K.; Peršurić, Đ.; Gluhić, D.; Banović, M.; Komes, D.; Gracin, L.; Lovrić, T. Aroma Precursors of Malvasia Istriana Wine. Riv. Vitic. Enol. 2005, 2–4, 99–108. [Google Scholar]
- Palčić, I.; Korenika, A.J.; Jakobović, S.; Pasković, I.; Major, N.; Ban, D.; Ban, S.G.; Karoglan, M.; Ćustić, M.H.; Jeromel, A.; et al. Soil Type Affects Grape Juice Free Amino Acids Profile during Ripening of Cv. Malvasia Istriana (Vitis vinifera L.). N. Zealand J. Crop Hortic. Sci. 2020, 48, 22–33. [Google Scholar] [CrossRef]
- Figué, A.; Violleau, F.; Gosset, M.; Durner, D. Wine Amino Acids of Four Autochthonous Grape Varieties from Southwest France: Influencing Factors and Role in Taste Perception. OENO One 2025, 59. [Google Scholar] [CrossRef]
- Zhou, W.; Fang, R.; Chen, Q. Effect of Gallic and Protocatechuic Acids on the Metabolism of Ethyl Carbamate in Chinese Yellow Rice Wine Brewing. Food Chem. 2017, 233, 174–181. [Google Scholar] [CrossRef]
- Alberto, M.R.; de Nadra, M.C.M.; Arena, M.E. Influence of Phenolic Compounds on the Growth and Arginine Deiminase System in a Wine Lactic Acid Bacterium. Braz. J. Microbiol. 2012, 43, 167–176. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khalafyan, A.; Temerdashev, Z.; Abakumov, A.; Yakuba, Y.; Sheludko, O.; Kaunova, A. Multidimensional Analysis of the Interaction of Volatile Compounds and Amino Acids in the Formation of Sensory Properties of Natural Wine. Heliyon 2023, 9, e12814. [Google Scholar] [CrossRef]
- Stines, A.P.; Grubb, J.; Gockowiak, H.; Henschke, P.A.; Høj, P.B.; Van Heeswijck, R. Proline and Arginine Accumulation in Developing Berries of Vitis vinifera L. in Australian Vineyards: Influence of Vine Cultivar, Berry Maturity and Tissue Type. Aust. J. Grape Wine Res. 2000, 6, 150–158. [Google Scholar] [CrossRef]
- Petrovic, G.; Buica, A. Grape Must Profiling And Cultivar Discrimination Based On Amino Acid Composition And General Discriminant Analysis With Best Subset. S. Afr. J. Enol. Vitic. 2019, 40. [Google Scholar] [CrossRef]
- Gutiérrez-Gamboa, G.; Romanazzi, G.; Garde-Cerdán, T.; Pérez-Álvarez, E.P. A Review of the Use of Biostimulants in the Vineyard for Improved Grape and Wine Quality: Effects on Prevention of Grapevine Diseases. J. Sci. Food Agric. 2019, 99, 1001–1009. [Google Scholar] [CrossRef]
- Raspor, P.I.; Zagorc, T.I.; Povhe Jemec, K.R.; Čadež, N.J. Yeast Biodiversity in Slovenian Wine Regions: Case Amino Acids in Spontaneous and Induced Fermentations of Malvasia. Proc. Nat. Sci. Matica Srp. Novi Sad 2009, 117, 97–110. [Google Scholar] [CrossRef]
- Rescic, J.; Mikulic-Petkovsek, M.; Rusjan, D. The Impact of Canopy Managements on Grape and Wine Composition of Cv. ‘Istrian Malvasia’ (Vitis vinifera L.). J. Sci. Food Agric. 2016, 96, 4724–4735. [Google Scholar] [CrossRef]
- Bubola, M.; Peršurić, Đ.; Kovačević Ganić, K.; Karoglan, M.; Kozina, B. Effects of Fruit Zone Leaf Removal on the Concentrations of Phenolic and Organic Acids in Istrian Malvasia Grape Juice and Wine. Food Technol. Biotechnol. 2012, 50, 159–166. [Google Scholar]
- Paramithiotis, S.; Stasinou, V.; Tzamourani, A.; Kotseridis, Y.; Dimopoulou, M. Malolactic Fermentation—Theoretical Advances and Practical Considerations. Fermentation 2022, 8, 521. [Google Scholar] [CrossRef]
- Radeka, S.; Bestulić, E.; Rossi, S.; Orbanić, F.; Bubola, M.; Plavša, T.; Lukić, I.; Jeromel, A. Effect of Different Vinification Techniques on the Concentration of Volatile Aroma Compounds and Sensory Profile of Malvazija Istarska Wines. Fermentation 2023, 9, 676. [Google Scholar] [CrossRef]
- Marinov, L.; Jeromel, A.; Tomaz, I.; Preiner, D.; Korenika, A.J. Učinak Sekvencijalne Fermentacije s Kvascima Lachancea Thermotelerans i Torulaspora Delbrueckii Na Kemijski Sastav Vina ’Malvazija Istarska’. Glas. Zaštite Bilja 2021, 4, 56–66. [Google Scholar] [CrossRef]
- Crespo, J.; Romero, V.; García, M.; Arroyo, T.; Cabellos, J.M. Influence of Skin-Contact Treatment on Aroma Profile of Malvasia Aromatica Wines in D.O. “Vinos de Madrid”. In Grapes and Wine; Morata, A., Loira, I., González, C., Eds.; IntechOpen: London, UK, 2021. [Google Scholar]


| Parameter | Unit | Value |
|---|---|---|
| pH | – | 9.79 ± 0.05 |
| EC | μS/cm | 792 ± 65.9 |
| Ash | % | 8.36 ± 0.01 |
| Total carbon | % | 73.1 ± 0.43 |
| Specific surface area | m2/g | 2.07 ± 0.14 |
| N | % | 1.06 ± 0.01 |
| P | g/kg | 27.2 ± 0.21 |
| K | g/kg | 22.8 ± 0.78 |
| Mg | g/kg | 27.5 ± 1.67 |
| S | g/kg | 12.4 ± 0.30 |
| Ca | g/kg | 187 ± 9.61 |
| Cu | mg/kg | 4.65 ± 0.25 |
| Mn | mg/kg | 6.56 ± 1.22 |
| Mo | mg/kg | 0.11 ± 0.00 |
| Zn | mg/kg | 2.69 ± 0.01 |
| Foliar Suspension | Formulation |
|---|---|
| C | ultrapure water |
| B | ultrapure water + 300 mg biochar/L |
| U | ultrapure water + 400 mg urea/L |
| BU | ultrapure water + 300 mg biochar/L+ 400 mg urea/L |
| A | ultrapure water + 2 mL amino acid mixture |
| BA | ultrapure water + 300 mg biochar/L + 2 mL amino acid mixture |
| BUA | ultrapure water + 300 mg biochar/L + 400 mg urea/L 2 mL + amino acid mixture |
| Foliar Suspension | pH | EC (µS/cm) |
|---|---|---|
| C | 6.15 ± 0.45 c | 1.80 ± 0.30 c |
| B | 9.21 ± 0.26 a | 36.0 ± 1.04 c |
| U | 7.16 ± 0.33 a | 4.83 ± 0.66 c |
| BU | 9.10 ± 0.24 bc | 36.6 ± 1.32 c |
| A | 6.77 ± 0.03 bc | 460 ± 15.2 a |
| BA | 7.18 ± 0.03 bc | 493 ± 26.7 a |
| BUA | 7.40 ± 0.06 b | 287 ± 10.3 b |
| p value | *** | *** |
| Compound (mg/L) | Treatment | p | ||||||
|---|---|---|---|---|---|---|---|---|
| C | B | U | BU | A | BA | BUA | ||
| Serine | 15.8 ± 1.72 b | 15.0 ± 0.86 b | 32.7 ± 1.21 a | 15.5 ± 1.82 b | 13.9 ± 0.69 b | 13.4 ± 4.90 b | 15.3 ± 5.56 b | ** |
| Aspartic acid | 18.7 ± 0.65 ab | 20.0 ± 0.61 ab | 24.5 ± 0.69 a | 15.0 ± 0.35 b | 19.4 ± 0.03 ab | 18.1 ± 2.30 b | 17.0 ± 2.21 b | ** |
| Glycine | 1.43 ± 0.13 ab | 1.15 ± 0.04 ab | 1.46 ± 0.07 a | 1.19 ± 0.04 ab | 1.15 ± 0.03 ab | 1.04 ± 0.05 b | 1.14 ± 0.16 ab | * |
| Threonine | 15.7 ± 0.46 | 15.9 ± 0.29 | 18.9 ± 0.58 | 16.9 ± 0.48 | 15.8 ± 0.49 | 16.8 ± 0.35 | 16.7 ± 1.56 | n.s. |
| 4-hydroxyproline | 1.56 ± 0.05 | 1.81 ± 0.04 | 1.68 ± 0.05 | 1.69 ± 0.06 | 1.74 ± 0.05 | 1.74 ± 0.07 | 1.65 ± 0.10 | n.s. |
| Glutamine | 1794 ± 115 | 1748 ± 85.7 | 1827 ± 71.1 | 1967 ± 137 | 1601 ± 21.8 | 1961 ± 285 | 2083 ± 216 | n.s. |
| Alanine | 15.4 ± 2.42 a–c | 8.68 ± 1.64 cd | 21.2 ± 0.86 a | 10.7 ± 3.67 b-d | 19.3 ± 1.62 ab | 5.49 ± 0.65 d | 12.2 ± 1.98 a-d | *** |
| Glutamic acid | 36.0 ± 1.35 ab | 27.1 ± 0.79 b | 40.6 ± 2.84 a | 34.8 ± 1.47 ab | 36.6 ± 1.91 ab | 31.5 ± 2.34 ab | 32.3 ± 2.95 ab | ** |
| Proline | 7.23 ± 0.36 b | 9.38 ± 0.35 ab | 7.76 ± 1.13 ab | 11.4 ± 0.57 a | 7.04 ± 0.36 b | 10.7 ± 1.13 ab | 9.68 ± 1.45 ab | ** |
| Asparagine | 0.58 ± 0.04 | 0.54 ± 0.05 | 0.75 ± 0.08 | 0.49 ± 0.08 | 0.46 ± 0.01 | 0.50 ± 0.08 | 0.48 ± 0.07 | n.s. |
| Treatment | Glucose (g/L) | Fructose (g/L) | Total Sugars (g/L) |
|---|---|---|---|
| C | 85.1 ± 2.49 ab | 89.7 ± 2.67 ab | 197 ± 5.86 ab |
| B | 61.9 ± 0.71 c | 65.8 ± 0.75 c | 145 ± 1.61 c |
| U | 76.1 ± 7.04 bc | 79.9 ± 7.48 bc | 175 ± 15.90 bc |
| BU | 90.5 ± 1.50 a | 95.6 ± 1.57 a | 208 ± 3.34 a |
| A | 89.4 ± 3.62 ab | 94.9 ± 3.82 ab | 207 ± 8.19 ab |
| BA | 97.5 ± 2.42 a | 103 ± 2.53 a | 223 ± 5.22 a |
| BUA | 90.5 ± 1.56 a | 95.8 ± 1.75 a | 209 ± 3.70 a |
| p-value | *** | *** | *** |
| Treatment | Tartaric Acid (mg/L) | Malic Acid (mg/L) | Lactic Acid (mg/L) | Citric Acid (mg/L) |
|---|---|---|---|---|
| C | 7561 ± 33.7 | 1624 ± 89.5 ab | 30.0 ± 1.75 a | 292 ± 9.92 ab |
| B | 8902 ± 308 | 1626 ± 27.6 ab | 27.3 ± 1.61 a | 326 ± 8.14 ab |
| U | 7372 ± 213 | 1786 ± 101 a | 29.7 ± 3.45 a | 336 ± 4.75 a |
| BU | 8270 ± 288 | 1516 ± 61.2 ab | 24.2 ± 1.76 ab | 300 ± 5.03 ab |
| A | 8864 ± 477 | 1263 ± 66.4 b | 28.9 ± 5.48 a | 313 ± 21.4 ab |
| BA | 9043 ± 467 | 1461 ± 112 ab | 17.9 ± 1.66 b | 276 ± 5.18 b |
| BUA | 9099 ± 874 | 1532 ± 108 ab | 17.3 ± 1.75 b | 311 ± 16.6 ab |
| p-value | n.s. | * | * | * |
| Treatment | Alcohol Concentration (% v/v) | Titratable Acidity (g/L) | Volatile Acidity (g/L) | pH | Reducing Sugars (g/L) |
|---|---|---|---|---|---|
| C | 9.52 ± 0.01 d | 8.08 ± 0.01 ab | 0.02 ± 0.01 | 2.96 ± 0.00 b | 2.57 ± 0.07 ab |
| B | 9.58 ± 0.01 c | 8.21 ± 0.22 a | n.d. | 2.93 ± 0.01 c | 2.87 ± 0.09 a |
| U | 10.1 ± 0.00 b | 7.61 ± 0.00 c | 0.01 ± 0.01 | 3.01 ± 0.00 a | 2.57 ± 0.19 ab |
| BU | 10.2 ± 0.01 b | 8.00 ± 0.01 a-c | 0.02 ± 0.02 | 2.92 ± 0.00 c | 2.30 ± 0.10 bc |
| A | 9.52 ± 0.01 d | 8.15 ± 0.00 ab | n.d. | 2.90 ± 0.00 d | 2.53 ± 0.07 ab |
| BA | 10.4 ± 0.01 a | 7.78 ± 0.00 bc | 0.03 ± 0.01 | 2.97 ± 0.00 b | 2.00 ± 0.12 c |
| BUA | 10.1 ± 0.01 b | 8.05 ± 0.01 ab | n.d. | 2.89 ± 0.00 d | 2.23 ± 0.09 bc |
| p-value | *** | ** | n.s. | *** | ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Prelac, M.; Anđelini, D.; Cvitan, D.; Užila, Z.; Major, N.; Kovačević, T.K.; Goreta Ban, S.; Ban, D.; Plavša, T.; Damijanić, K.; et al. Foliar Application of Biochar-Based Suspensions: Effects on Composition and Sensory Properties of Malvazija istarska (Vitis vinifera L.) Must and Wine. Sustainability 2026, 18, 364. https://doi.org/10.3390/su18010364
Prelac M, Anđelini D, Cvitan D, Užila Z, Major N, Kovačević TK, Goreta Ban S, Ban D, Plavša T, Damijanić K, et al. Foliar Application of Biochar-Based Suspensions: Effects on Composition and Sensory Properties of Malvazija istarska (Vitis vinifera L.) Must and Wine. Sustainability. 2026; 18(1):364. https://doi.org/10.3390/su18010364
Chicago/Turabian StylePrelac, Melissa, Dominik Anđelini, Danko Cvitan, Zoran Užila, Nikola Major, Tvrtko Karlo Kovačević, Smiljana Goreta Ban, Dean Ban, Tomislav Plavša, Kristijan Damijanić, and et al. 2026. "Foliar Application of Biochar-Based Suspensions: Effects on Composition and Sensory Properties of Malvazija istarska (Vitis vinifera L.) Must and Wine" Sustainability 18, no. 1: 364. https://doi.org/10.3390/su18010364
APA StylePrelac, M., Anđelini, D., Cvitan, D., Užila, Z., Major, N., Kovačević, T. K., Goreta Ban, S., Ban, D., Plavša, T., Damijanić, K., & Palčić, I. (2026). Foliar Application of Biochar-Based Suspensions: Effects on Composition and Sensory Properties of Malvazija istarska (Vitis vinifera L.) Must and Wine. Sustainability, 18(1), 364. https://doi.org/10.3390/su18010364









