Abstract
This research used tomato waste as a substrate (fuel) in Single Chamber-Microbial Fuel Cells (scMFC) on a small scale. The electrochemical properties were monitored, the functional groups of the substrate were analyzed by Fourier Transform Infrared Spectrophotometry (FTIR) and a microbiological analysis was performed on the electrodes in order to identify the microorganisms responsible for the electrochemical process. The results show voltage peaks and an electrical current of 3.647 ± 0.157 mA and 0.957 ± 0.246 V. A pH of 5.32 ± 0.26 was measured in the substrate with an electrical current conductivity of 148,701 ± 5849 mS/cm and an internal resistance (Rint) of 77. 517 ± 8.541 Ω. The maximum power density (PD) displayed was 264.72 ± 3.54 mW/cm2 at a current density (CD) of 4.388 A/cm2. On the other hand, the FTIR spectrum showed a more intense decrease in its peaks, with the compound belonging to the phenolic groups being the most affected at 3361 cm−1. The micrographs show the formation of a porous biofilm where molecular identification allowed the identification of two bacteria (Proteus vulgaris and Proteus vulgaris) and a yeast (Yarrowia lipolytica) with 100% identity. The data found show the potential of this waste as a source of fuel for the generation of an electric current in a sustainable and environmentally friendly way, generating in the near future a mechanism for the reuse of waste in a beneficial way for farmers, communities and agro-industrial companies.
1. Introduction
The uncontrolled increase in the consumption of electrical energy from fossil sources has caused serious damage to the environment, resulting in significant changes in the climate and natural disasters that have affected many countries [1,2]. Humans use this type of energy daily for their activities, becoming a good and primary source worldwide (81%), divided into oil (32%), coal (27.1%), and gas (22%) [3,4]. Each year, more than 130 million tons are processed, and thus far, approximately eight million have already been processed [5]. In addition, recent statistics have shown that the consumption of electrical energy has increased from 13.72 billion KWH in 2000 to 24.57 billion KWH in 2018, which was even higher in the pandemic era [6,7]. On the flip side, the generation of fruits and vegetables has increased exponentially in recent years because human society has begun to consume healthier food, resulting in an economic boost for agro-industrial companies that export these products [8]. However, this increase in production has also generated large amounts of organic waste (agro-industrial waste), which has become a problem for governments because they do not have an organized method of collecting and managing this type of waste [9,10]. It has been reported that municipal waste in 2017 in Europe was 58 million tons, of which 46% was organic waste, which is estimated to increase by 32% by 2030 [11]. Among all the fruits produced by agro-industrial companies, tomato production (Solanum lycopersicum L.) is one of the most important; in 2019, its global production was estimated at approximately 37 million tons, and it has been estimated that of every 130 million tons produced, approximately 8 million tons represent the waste generated according to the World Processing Tomato Council (WPTC) [12,13,14]. This fruit contains natural bioactive compounds and antioxidants and has anti-inflammatory properties, which have beneficial health effects [15].
In the search to solve these two major problems that afflict the world, microbial fuel cell technology is presented as a novel solution because it uses any type of waste as fuel to generate electricity, which is due to the conversion of chemical energy into electricity through the reduction-oxidation process that occurs within cells [16,17]. The MFC design can have two chambers (anodic and cathodic) and a proton exchange membrane that joins them on the inside and an external electrical circuit [18]. A large number of designs and the use of various materials for their construction have been found in the literature, but one of the best-known is the single-chamber microbial fuel cell (scMFCs). The scMFCs is a low-cost cell. with its anode and cathode chambers in the same compartment, and because the anode is in direct contact with the environment (O2), high power density values are obtained, showing great potential for large-scale scaling [19,20,21].
In the literature, research has been conducted on the use of different types of organic waste as a substrate for the generation of bioelectricity; for example, Asefi et al. (2019) managed to generate 600 mV voltage spikes in their dual-chamber microbial fuel cells using food waste as fuel and carbon felt as electrodes [22]. Likewise, Mohamed et al. (2020) generated voltage peaks of approximately 585 mV and a maximum power density of 33 mW/m2 in microbial fuel cells using graphite as electrodes [23]. Yaqoob et al. (2022), using a mixture of rambutan, langsat, and mango residues, managed to generate peaks of 175 mV and a power density (PD) of 0.30 mW/m2 in their scMFCs with graphite electrodes [24]. In another study, Kondaveeti et al. (2019) used citrus peels in single-chamber microbial fuel cells and generated maximum peak voltages and power densities of 284 mV and 26.41 mW/m2, respectively, using graphite plates as electrodes [25]. Thus, it has also been observed that the values of electric current can increase with the use of electrodes of a metallic nature due to the intrinsic properties that these have to the passage of electrons for the generation of electricity, the current values have been compared to graphite electrodes, coal and its derivatives with equal amounts of substrate volume, microbial fuel cell design and with equal environmental parameters; managing to generate higher voltage, current and power density peaks [26,27,28].
This research has as main objective to observe the potential to generate bioelectricity through MFCs at a laboratory scale using tomato residues as a substrate. To achieve this, pH, current, voltage, pH, and electrical conductivity were monitored. Likewise, the values of the power density, current density, and Rint. of the microbial fuel cells were measured, as well as the micrographs of the biofilm of the anodic electrode. Finally, the microorganisms present in the anode electrode at the end of the monitoring period were identified, and the FTIR spectra of the initial and final substrates were observed. This research will demonstrate the potential of tomato waste for use as fuel in single-chamber microbial fuel cells, using low-cost materials such as copper and zinc electrodes. Likewise, it will be possible to identify microorganisms that generate electrical energy for their subsequent use as biocatalysts, which will be able to repower microbial fuel cells. The replication of this technology on a large scale could benefit companies dedicated to the sale and purchase of these fruits in the near future.
2. Materials and Methods
2.1. Design of scMFCs
Three single-chamber microbial fuel cells (scMFCs) were manufactured for which copper (Cu) electrodes were used in the anode and zinc (Zn) in the cathode, a 500 mL polymethylmethacrylate tube was used and two 10 cm2 lids were used. At the ends of the tube, a 5 cm hole was made at one end for the cathode to have contact with the environment (O2), with an area of 60.12 cm2. The electrodes were joined by an external resistor joined with a copper wire (0.15 cm diameter) (see Figure 1).

Figure 1.
Schematic of the single-chamber microbial fuel cells prototype.
2.2. Collection and Preparation of Tomato Waste
Organic tomato waste was collected from the La Hermelinda plant in Trujillo, Peru, washed several times with distilled water to remove impurities (dust, insects or other impurities) and dried in an oven (Labtron, Camberley, UK, LDO-B10) for 12 h at 25 ± 1 °C. Obtain tomato juice (2 L, 500 mL per scMFC) using an extractor (Maqorito, Lima, Peru, 400 rpm).
2.3. Characterization of the scMFC
The monitoring time for the chemical–electrical parameters was 35 days. The voltage and current were measured using a multimeter (Prasek Premium PR-85) with an external resistance of 1000 Ω. The values of current density (CD) and power density (PD) were calculated according to the method of Segundo et al. (2023), with the same external resistance values [29]. Conductivity and pH were measured using a conductivity meter CD-4301 and a pH meter (Oakton Series 110), the transmittance values were measured by FTIR (Thermo Scientific IS50, Waltham, MA, USA). The electrochemical impedance spectroscopy (IES) measurements were evaluated with a Metrohm Autolab 302N potentiostat/galvanostat system on ensembled MFCs. The impedance spectra were obtained within the frequency range of 10 mHz and 100 kHz with a constant voltage of 10 mV and the Nyquist plot is shown in Figure.
2.4. Isolation and Molecular Identification of Anode Microorganisms
2.4.1. Isolation of Bacteria
From the anode (Copper-Cu plate), a sample was obtained by swabbing with a sterile swab. The sample was transferred to BHI agar, and the sample was extended with a bacteriological loop, making exhaustion streaks over the entire surface of the plate. The culture media were incubated in anaerobic jars at 36 ± 1 °C for 24 to 48 h. The colonies that grew in the culture medium were seeded on Nutrient Agar and MacConkey Agar. From the growth in these cultures, pure cultures were made on slant Nutrient Agar for later identification.
2.4.2. Yeast Isolation
Sampling was carried out in the same way as the previous procedure, the only variation was the culture medium and the incubation time. The medium used both for isolation and for pure culture was Sabouraud Agar with 4% dextrose plus antibiotic (chloramphenicol). While the incubation time was 30 ± 1 °C for 48 to 72 h.
2.4.3. Molecular Identification
The identification was carried out by the Analysis and Research Center of the labora-tory “Biodes Laboratorios”. To accomplish this, axenic cultures of bacteria and yeasts were sent to extract their genetic material (DNA) using the CTAB method. Amplification was carried out by the PCR technique, and then sequenced in the Macrogen Laboratory (USA). The sequences obtained were analyzed using the bioinformatics software Molecular Evolutionary Genetics Analysis (MEGA). Alignment and comparison with other sequences were carried out using the BLAST program (https://blast.ncbi.nlm.nih.gov/Blast.cgi (accessed on 2 April 2023)), through which percentages of identity were obtained for both bacteria and yeast. Finally, dendrograms of the species identified using MEGA X were constructed.
3. Results and Analysis
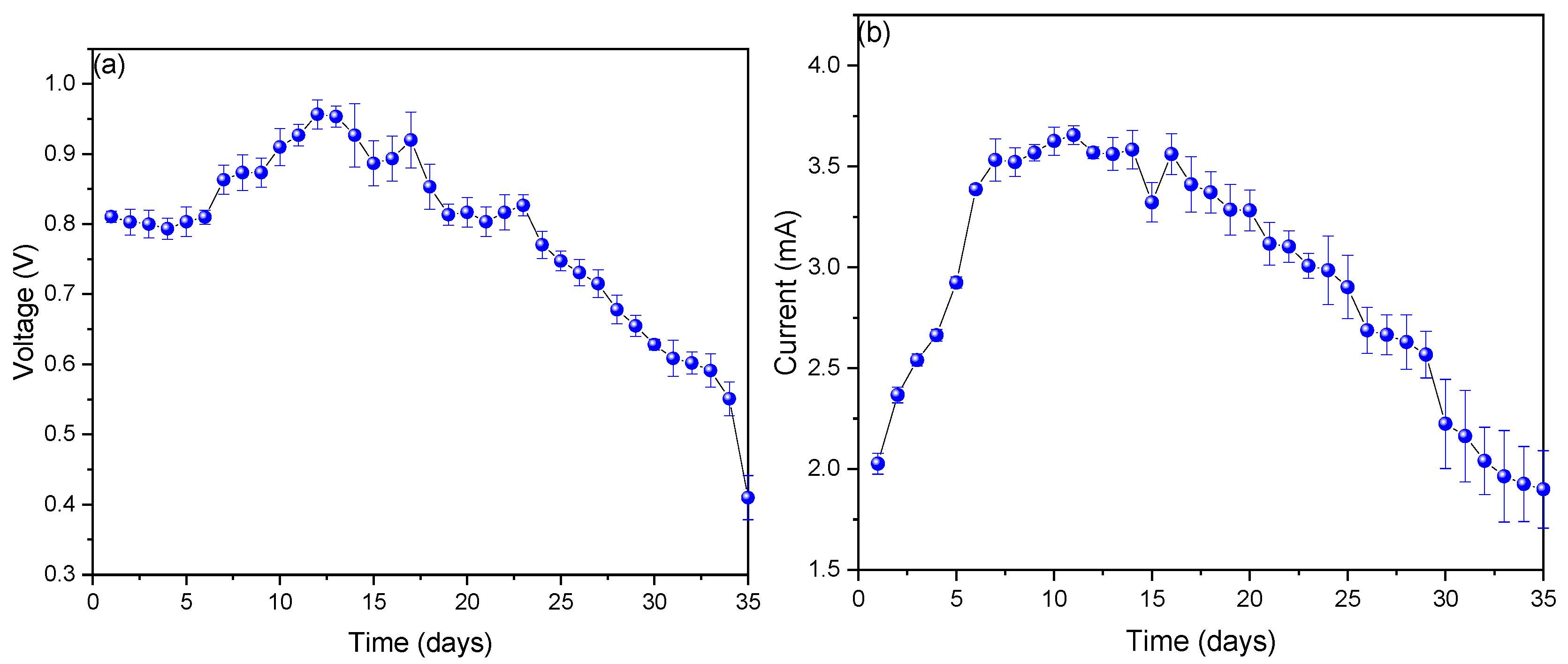
Figure 2a shows the voltage values observed during the monitoring of the scMFCs, which showed a voltage increase from day 1 (0.801 ± 0.014 V) to day 12 (0.957 ± 0.246 V) and later a slow decrease until day 35 (0.414 ± 0.297 V). Studies have shown that the highest voltage values in MFCs are directly proportional to the oxidation reactions that occur within these cells, while the decrease in the final monitoring stage is due to the sedimentation of the used substrate [30,31]. In this sense, Aiyer, K. (2020) in their research mentions that fruits with a high sucrose content generate a greater amount of voltage compared to those that do not contain this compound, which is why sucrose can be added to the waste used to observe any increase [32]. Figure 2b shows the values of electric current monitored at the scMFCs, where the values increased from day 1 (2.026 ± 0.008 mA) to day 11 (3.647 ± 0.157 mA) and later decreased until the last day (1.91 ± 0.487 mA). The increase in electric current values is due to the high content of nutrients that contributed to microbial growth and a good biofilm formed on the anode electrode, while the decrease in electric current values is due to the decrease in nutrients and the negative effect of the copper electrode used on microorganisms in the final stage [33,34,35]. An important factor that negatively affects the electrical values of the MFCs is the parameters of pH, temperature, and electrical conductivity, which in the final stage of monitoring were affected by the sedimentation observed in recent days due to inert organic matter [36].
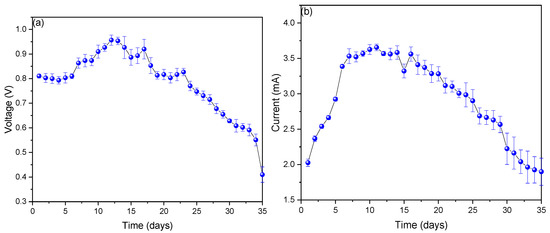
Figure 2.
Values of (a) voltage and (b) electric current obtained during the monitoring of the scMFC.
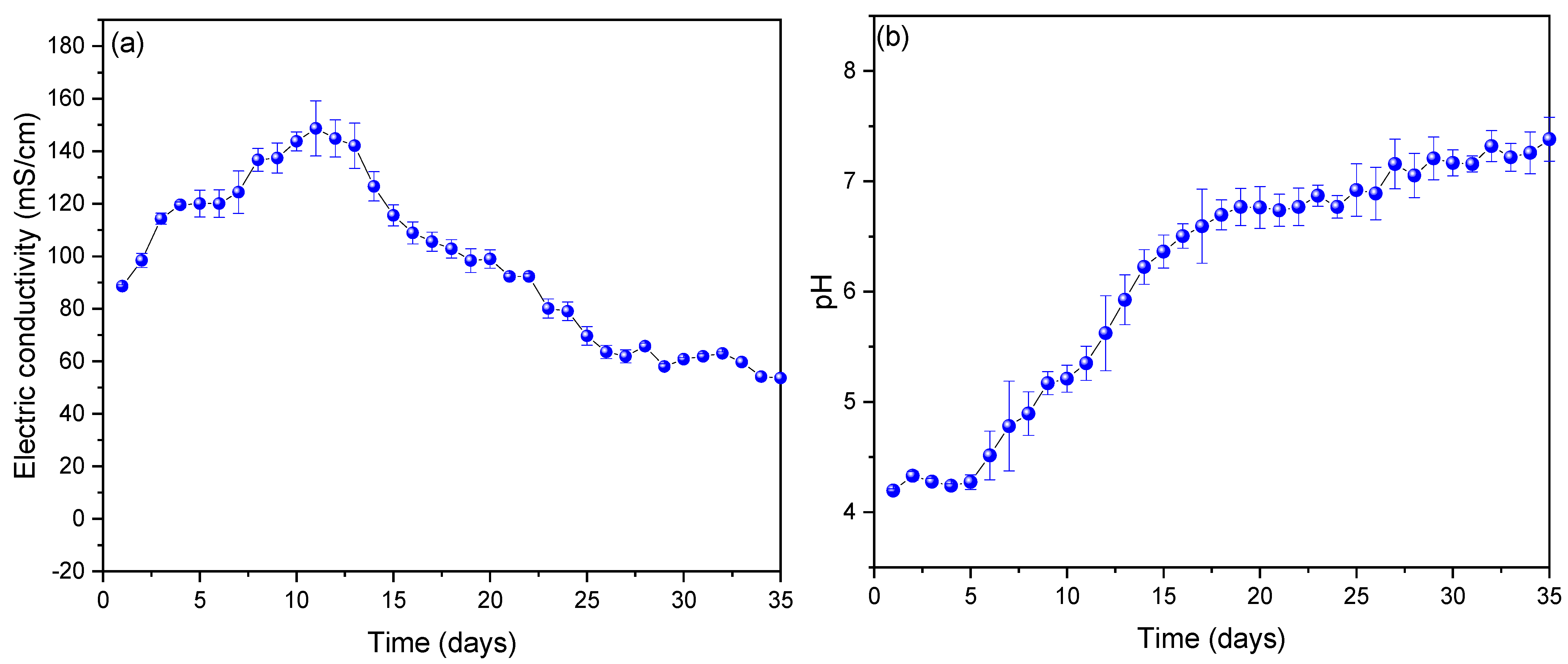
Figure 3a shows the values of electrical conductivity observed from the substrate used, during the 35 days of monitoring; being able to observe that the values increased up to 148.701 ± 5.849 mS/cm on the eleventh day to later show a decrease until the last day (53.625 ± 3.562 mS/cm) monitored. The results before this investigation gave lower electrical conductivity values because distilled water was used for it; but the values increased using tomato waste as a substrate. The high values of electrical conductivity observed indicate the low internal resistance of the scMFCs [36], which, compared to other works, for example, Yaqoob et al. (2022) showed an electrical conductivity of 81 mS/cm using wastewater as a substrate, managing to generate voltage peaks of 191 mV [37], which is due to the fact that tomato has chemical compounds that help conduct electrons more easily [38]. On the other hand, the decrease in their values is due to the precipitation of tomato waste at the bottom of the scMFCs in the process of generating electrical energy [39,40]. Figure 3b shows the monitoring of the pH values of the scMFCs, being able to observe that the pH increases from the first (slightly acid) to the last (neutral) day, with its optimum operating pH being on day eleven (5.32 ± 0.26). The optimal operating values of each MFC vary, and different types of substrates used with different pH have been found in the literature; one of the main reasons for this phenomenon is that the microorganisms are different and each one grows at specific pH conditions, affecting the system performance [41,42]. For example, Simeon et al. (2020) used sweet potato-shochu waste as a substrate at a pH of approximately 4, managing to generate 0.0002 and 0.08 W/m3 of power density at pH 4.5 and 4.2, respectively, in their scMFCs [43]. Likewise, Priya A. and Setty Y. (2019) used apple waste at pH 4.1, managing to generate voltage peaks and power density of 0.35 V and 31.58 mW/m2 in their MFC [40].
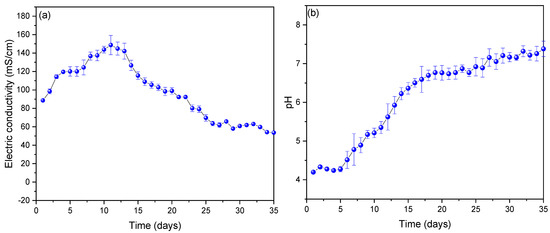
Figure 3.
Values of (a) electrical conductivity and (b) pH obtained from the monitoring of the MFC-SC.
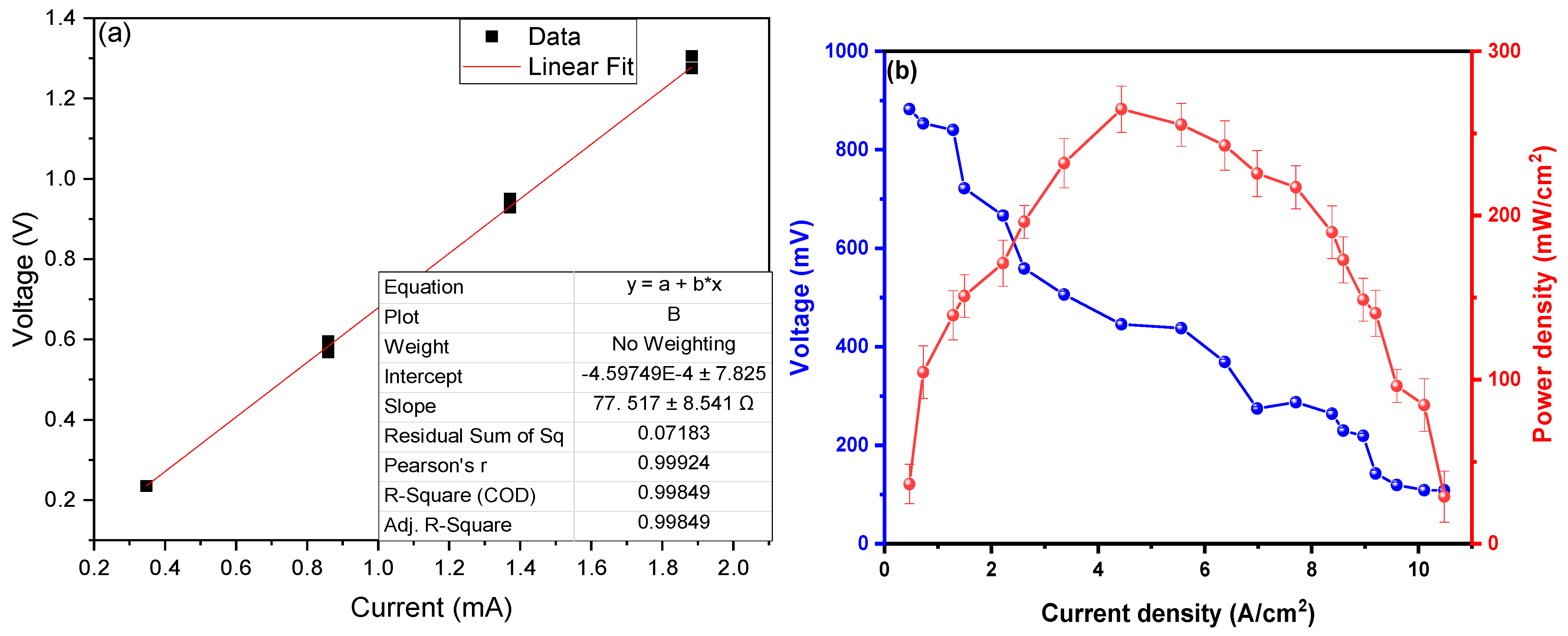
To calculate the internal resistance of the MFC-SC, Ohm’s Law (V = IR) was used, where the voltage values were placed on the “y” axis and those of the electric current on the “x” axis, which when performing a linear adjustment, the slope of the line is the internal resistance of the system, see Figure 4a. Based on the work carried out by Christwardana et al. (2020), this method represents an effective tool for the in-depth study of MFCs [16,44], as unlike the electrochemical impedance method (EIS) used in other investigations, internal resistance is obtained more directly. The IES method allows us to find the different components of the internal resistance, such as the resistance of charge transfer, diffusion, and resistance of the substrate, which is obtained with the intersection of the Nyquist curve with the “x” axis measured from the origin. Calculated Internal Resistance (Rint) was measured on the fourteenth day because this was the day in which the highest values of current and voltage were found, giving a value of Rint. of 77.517 ± 8.541 Ω, this value is relatively low compared to other investigations. For example, Antonopoulou et al. (2019) showed an internal resistance of 38 Ω (by electrochemical characterization), managing to generate a peak voltage of 0.488 V using vegetable debris as substrates in their MFC-SC and graphite as electrodes [41]. So too, Daud et al. (2021) used rice waste as a substrate, managing to show an internal resistance of 363.3 Ω (by Electrochemical tests) using graphite rods as electrodes, generating 510 mV on the fourteenth day [42]. One of the highest values found in the literature was the one reported by Simeon et al. (2020), where they were able to generate 725 mV peaks using urine treatment waste as a substrate in their MFC-SC with carbon electrodes, showing an initial and final internal resistance of 269.94 and 1627.89 Ω (by polarization method), respectively, resistance values increase from your control sample to the highest concentration of substrate (368.56 to 676.43 Ω) [43]. Figure 4b shows the power density (PD) values as a function of current density (CD), achieving a maximum power density of 264.72 ± 3.54 mW/cm2 at a current density of 4.388 A/cm2 with a peak voltage of 879.56 ± 0.184 V. The high values shown are due to the high inherent conductivity of the electrodes used, since being metallic in nature they facilitate the passage of electrons within the electrical circuit [44]. Other researchers have managed to generate higher values, but with the help of catalysts or biocatalysts, for example, Yaqoob et al. (2022) managed to generate a maximum power density of 41.58 mW/m2 with an internal resistance of 813.73 Ω using food waste (rice mixed with curry, vegetables, fish, cabbage, bones, and sweet cake pieces) as a substrate in MFC with graphite rod electrodes, compared to his control sample of 14.1 mW/m2, the increase was notorious [45]. Likewise, Du H. and Shao Z. (2022) managed to generate a maximum power density of 14.1 mW/m2 at a current density of 320.1 mA/m2 using potato waste as a substrate in their MFCs using carbon felts as electrodes [46]. Kamperidis et al. (2022) managed to generate a maximum power density of 14.4 mW/m2 at a current density of approximately 90 mA/m2 in their MFC-SCs using MnO2-coated graphites as electrodes and a condensed food substrate, but these values do not show much of a difference from their control sample, where a PD of 11.5 mW/m2 was observed [47].
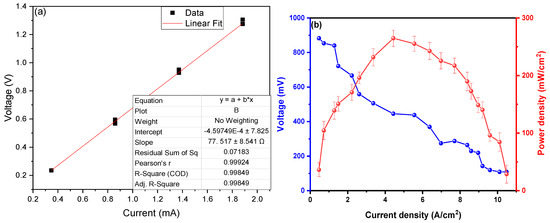
Figure 4.
(a) Rint. and (b) PD and voltage versus CD of the scMFC.
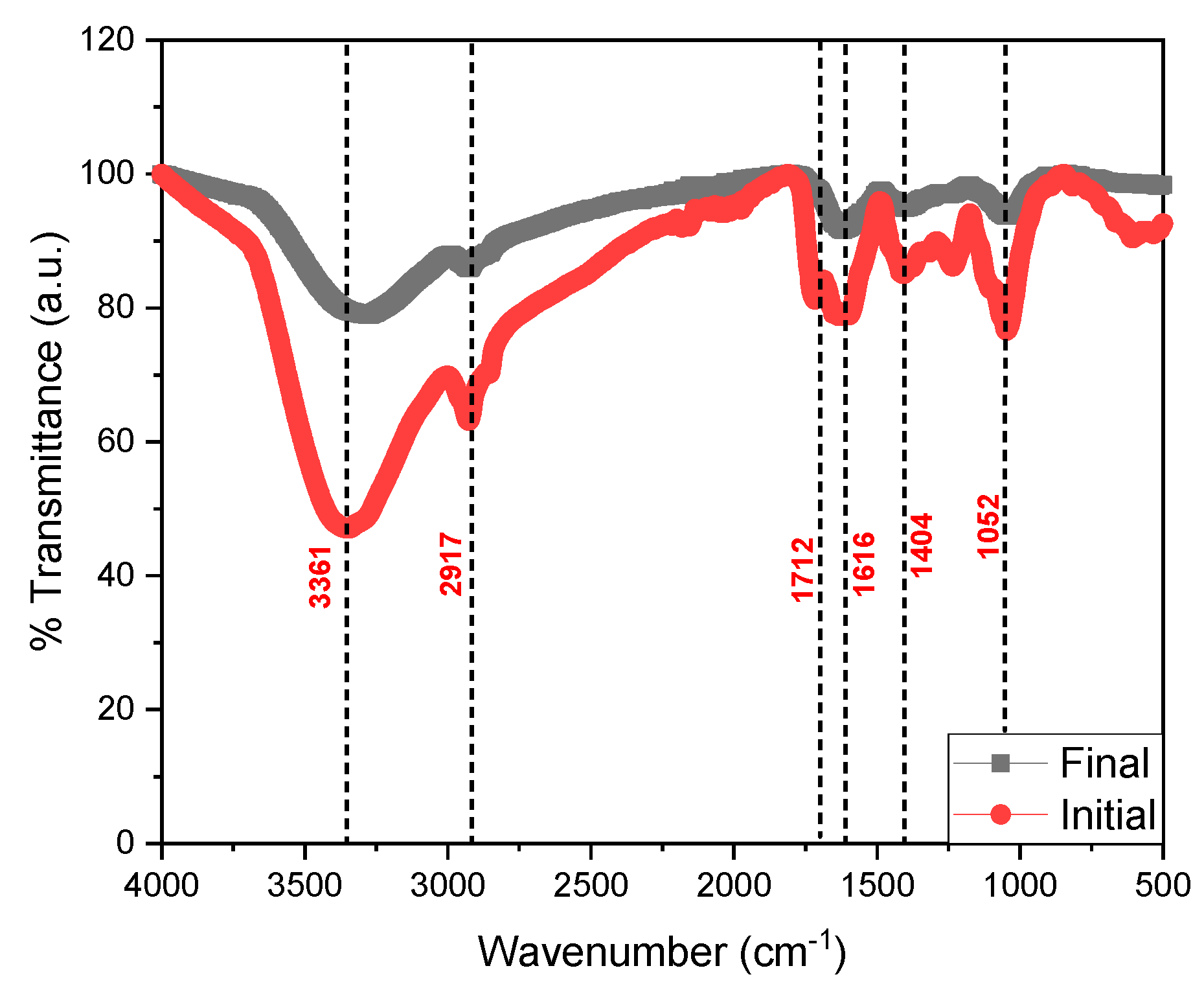
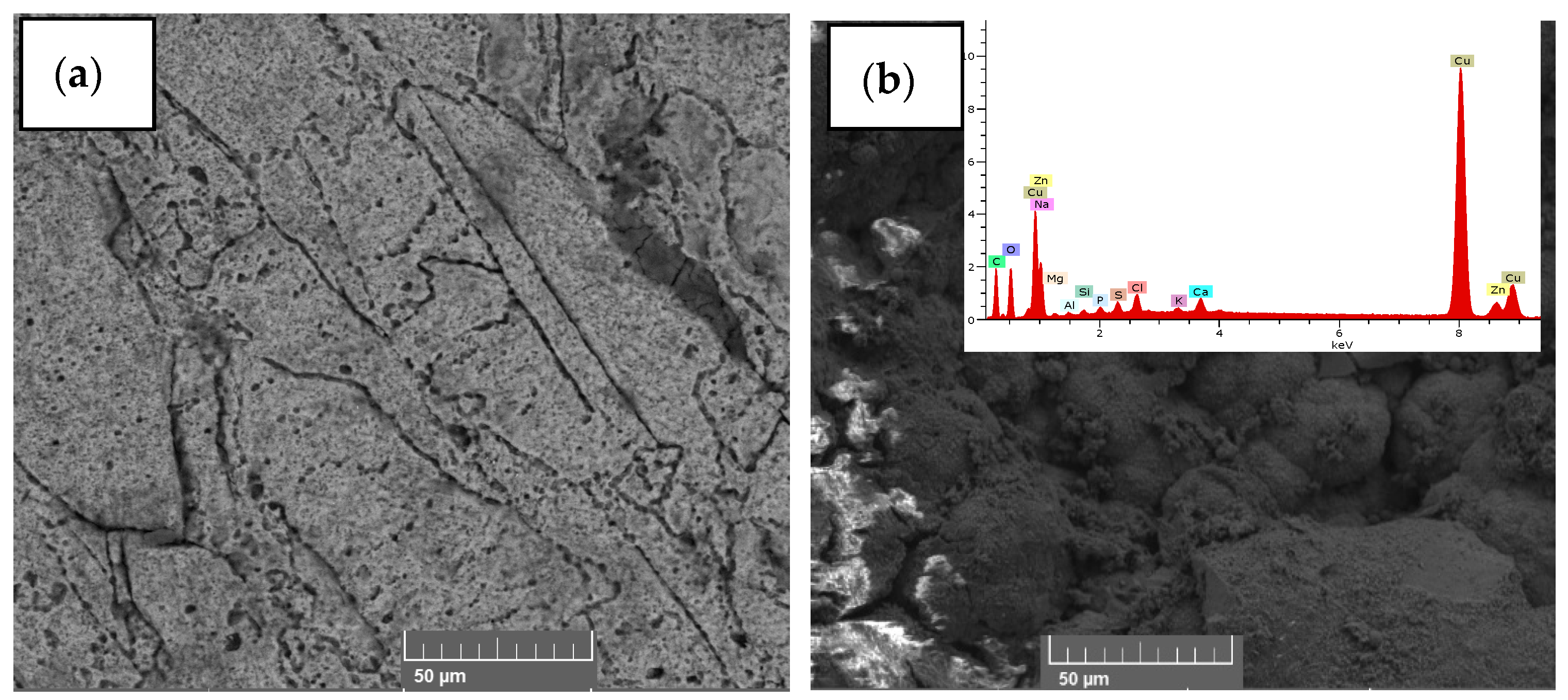
Figure 5 shows the FTIR spectra of the initial and final tomato waste, being able to observe the most intense peak at the 3361 cm−1 wavelength belonging to the N-H bond, O-H groups, carboxylic acids and phenolic groups, while the peak at 2917 cm−1 belongs to the C-H stretch of alkanes, ketones and aldehyde compounds [46]; while the 1712 and 1616 cm−1 peaks are associated with amine stretching and C=C [47]. Likewise, the 1404 cm−1 peak confirms the presence of the C-H, C=C, C==N, and C-N stretching belonging to the alkane, alkene, primary and secondary amine, respectively; finally, the 1052 cm−1 peak belongs to the C-H stretch of the alkane and the amide [48,49,50]. The decrease in the FTIR spectrum is mainly due to the decrease in the chemical components in the process of generating electrical energy by the microorganisms [51,52], as well as the degradation of the organic material that was observed in the last days of monitoring of the scMFC. Microorganisms consume carbon-rich compounds for their metabolism, this process causes electrons to be released and captured by the anode electrode and flow to the cathode, thus generating electric current [53]. In Figure 6, the micrographs of the initial and fine anodic electrode are observed, being able to observe a smooth surface belonging to the copper electrode; while the image belonging to the final state shows the formation of a porous biofilm due to the adhesion of the microorganisms present in the substrate, which were the electrical energy generators of the electrical device, observe the presence of carbon, oxygen, sodium, magnesium, calcium, copper and zinc; mainly in the EDS of the electrode in its final state. Hou et al. (2022) reported that the porosity in the form of round globules was due to the biomass forming an external layer on top of the biofilm, generating a double biolayer [54]. Hirose et al. (2023) also mentioned that the formation and adhesion of the biofilm to the electrodes optimizes the generation of electricity because the microorganisms generate the greatest number of electrons for the generation of electric current, but the durability of this depends on the environmental conditions given by the researchers [55].
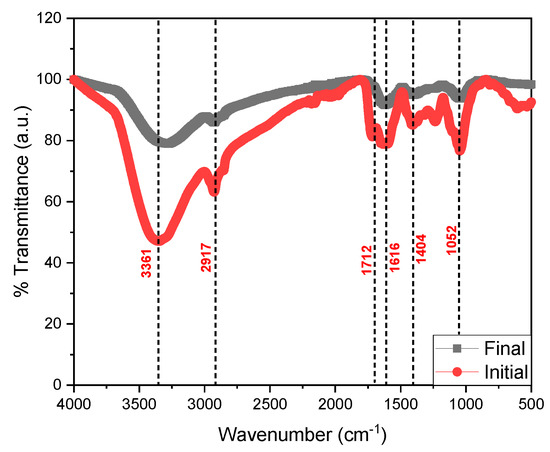
Figure 5.
Fourier transform infrared spectrum of tomato waste.
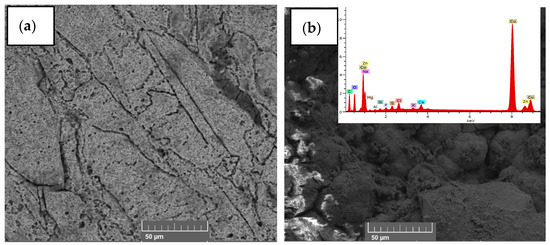
Figure 6.
Micrographs (a) initial and (b) final of the surface belonging to the anode electrode of the scMFC.
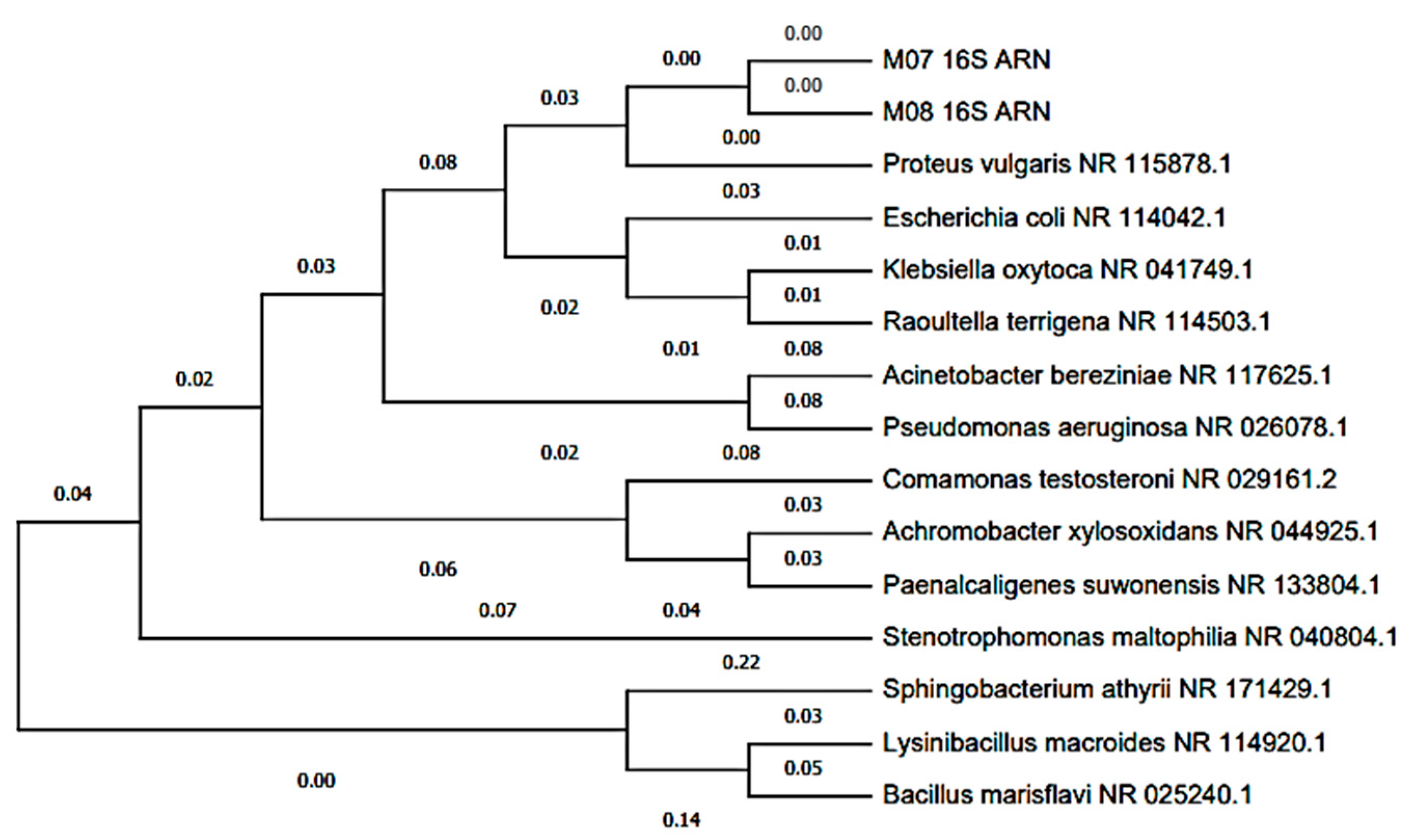
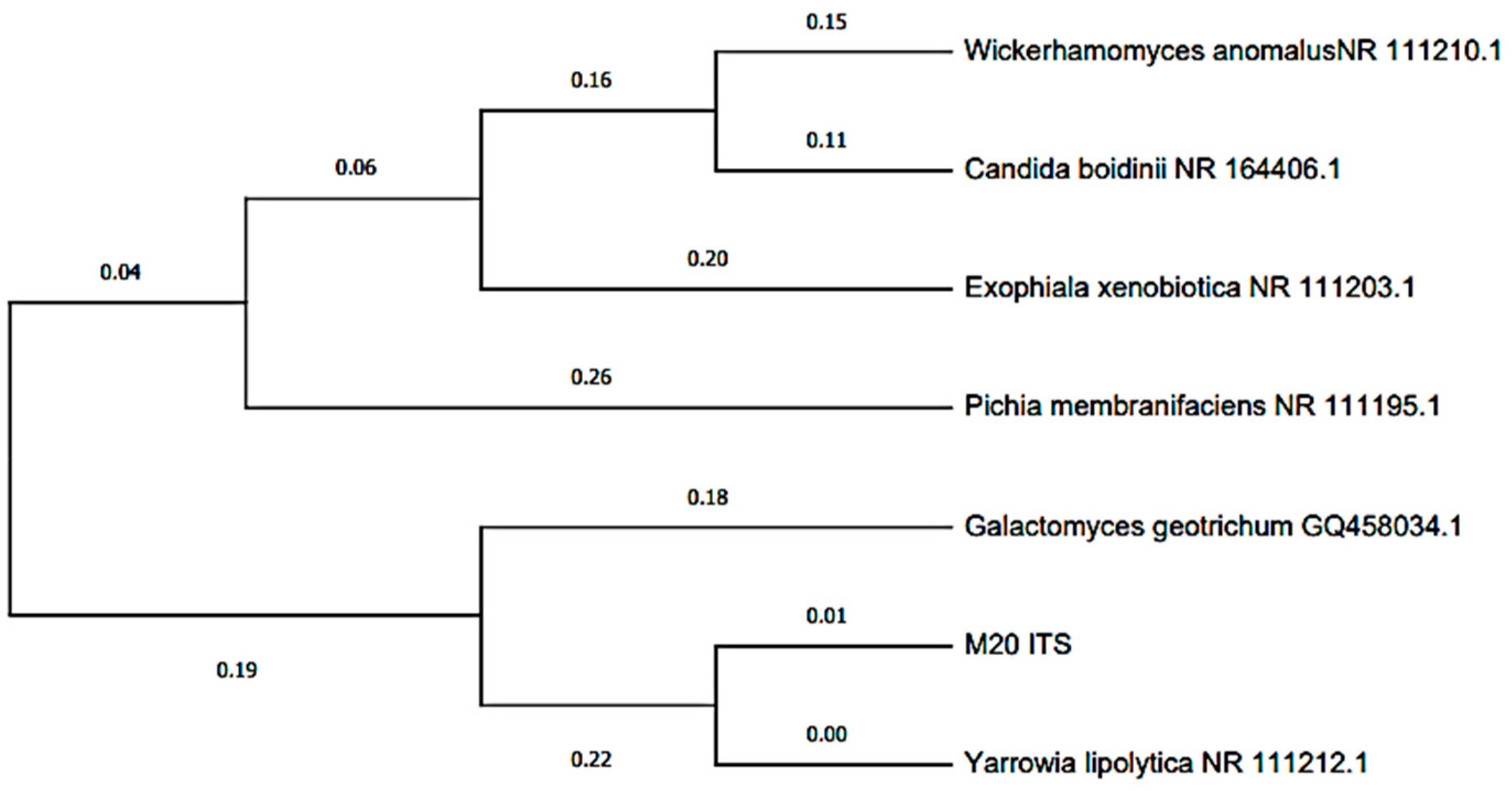
The Blast characterization performed at the anode electrode can be seen in Table 1, where two bacterial isolates (MT-B01 and MT-B02) and one yeast (MT-L01) were obtained from the anodic growth within the MFC. The two bacteria were identified as Proteus vulagris (100.00% identity), while the yeast corresponded to Yarrowia lipolytica (100.00% identity). Meanwhile, in Figure 7 and Figure 8, the dendrogram of the identified species and the phylogenetic relationships with other species support the Blast characterization. In this sense, the microorganisms identified (Table 1) correspond to the bacterium P. vulgaris (100.00% identity) and the Y. lipolytic yeast (100.00% identity). The microorganisms were isolated from tomato waste, and their origin may be due to contamination with soil or contaminated water. For example, P. vulgaris, is a Gram-negative rod-shaped bacterium commonly present in habitats such as soil and fecal matter [54]. In the same way, Y. lipolytica is a yeast belonging to the genus Candida, which can be isolated from food products and soil. This species stands out for its ability to degrade lipids and proteins [55].

Table 1.
Blast characterization of the rDNA sequence of microorganisms isolated from the anode plate of the scMFC with tomato waste substrates.
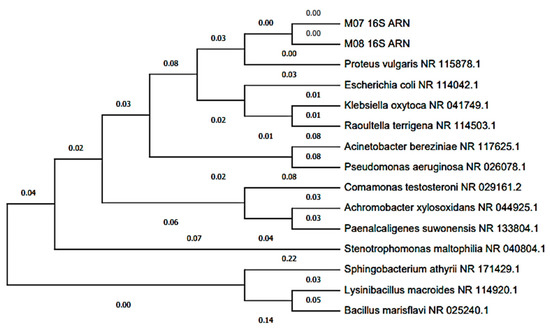
Figure 7.
Dendrogram of bacteria isolated from the scMFCs anode plate with tomato waste substrate.
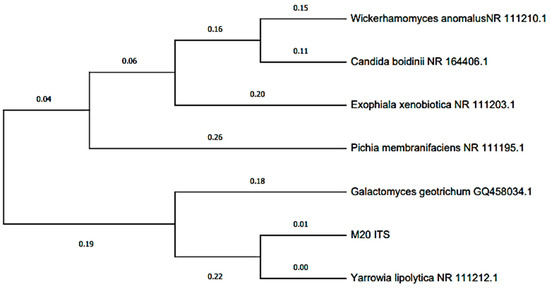
Figure 8.
Dendrogram based on the ITS regions of Yarrowia lipolytica isolated from the scMFC anode plate with tomato waste substrate.
The identification of both bacteria and yeast indicates that they are part of a microbial community that developed on the MFC anode, and to which electricity generation can be attributed [56,57]. Figure 7 shows that P. vulgaris is phylogenetically related to other Enterobacteriaceae. Some studies indicate that some enterobacteria, such as P. vulgaris, may be associated with the generation of electricity in MFC with organic waste substrates [58]. On the other hand, it is known that in most studies on microbial communities of MFC anodes, bacteria belonging to the Proteobacteria phylum are frequently isolated [59,60]. In other investigations, P. vulgaris has been used in MFCs to generate bioelectricity due to its metabolic reducing power, which through electron mediators can improve the energy efficiency of the MFC [54,61,62].
Regarding the identified yeast, Y. lipolitica belongs to the Ascomycota phylum. Other species within this phylum have been studied in MFCs to generate bioelectricity, such as Candida boidinii, and Saccharomyces cerevisiae, among others [63,64,65]. However, these yeasts are phylogenetically distant as shown in Figure 8. Studies have shown that using yeast in MFCs has advantages over bacteria, being classified as ideal biocatalysts since most are not pathogenic and grow in a wide range of organic substrates [63,66]. These can transfer electrons to the anode through the use of mediators [63]; however, there is evidence that there may be transfer by direct contact [67].
4. Conclusions
An electric current was successfully generated using tomato residue as a substrate in scMFC at a laboratory scale using zinc and copper electrodes. It was possible to generate voltage and electric current peaks of 0.957 ± 0.246 V and 3.647 ± 0.157 mA on days 12 and 11, respectively. The optimum operating pH was 5.32 ± 0.26 with an electrical conductivity of 148.701 ± 5.849 mS/cm. Thus, it also showed a low internal resistance of 77.517 ± 8.541 Ω with a power density of 264.72 ± 3.54 mW/cm2 at a current density of 4.388 A/cm2. On the other hand, the FTIR spectra showed a decrease in their peaks between the initial and final spectrum, being the peak belonging to the phenolic groups (3361 cm−1) the one that presented the greatest decrease, while the SEM micrographs show the formation of a porous biofilm. Finally, the molecular identification of the microorganisms showed the presence of two bacteria (Proteus vulgaris and Proteus vulgaris) and one yeast (Yarrowia lipolytica) with 100% identity on the anode electrode. This research contributes greatly to the sustainability of electric power generation in remote locations, where this type of fruit or its derivatives are harvested so that they can use their own waste as fuel in combination with larger-scale microbial fuel cells, with efficient enough to power a house overnight. Likewise, in the near future, companies will be able to use this technology as a fuel to generate electricity and reduce their expenses, all in an environmentally sustainable environment. For future work, it is recommended to standardize the pH value (5.32) to obtain the maximum potential of this residue, based on the results of this investigation; as well as covering the metallic electrodes with some type of chemical compound that is not harmful to microorganisms and the use of a catalyst or biocatalyst to enhance the generation of electrical energy from this device.
Author Contributions
Conceptualization, S.R.-F., L.C.-C. and M.G.-C.; methodology S.M.B. and W.R.-V.; software, R.N.-N.; validation, W.R.-V. and F.D.; formal analysis, S.R.-F., E.M.-T. and M.D.L.C.-N.; investigation S.R.-F. and W.R.-V.; data curation, M.D.L.C.-N. and M.G.-C.; writing—original draft preparation, D.D.-N.; writing—review and editing, S.R.-F. and E.M.-T.; project administration, S.R.-F. and R.N.-N. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Opoku, R.; Obeng, G.Y.; Osei, L.K.; Kizito, J.P. Optimization of industrial energy consumption for sustainability using time-series regression and gradient descent algorithm based on historical electricity consumption data. Sustain. Anal. Model. 2022, 2, 100004. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Ma, G.; Chen, X.; Fan, J.; Yang, B. Prediction of electricity consumption during epidemic period based on improved particle swarm optimization algorithm. Energy Rep. 2022, 8, 437–446. [Google Scholar] [CrossRef]
- Ramos, D.; Faria, P.; Vale, Z.; Mourinho, J.; Correia, R. Industrial Facility Electricity Consumption Forecast Using Artificial Neural Networks and Incremental Learning. Energies 2020, 13, 4774. [Google Scholar] [CrossRef]
- vom Scheidt, F.; Medinová, H.; Ludwig, N.; Richter, B.; Staudt, P.; Weinhardt, C. Data analytics in the electricity sector–A quantitative and qualitative literature review. Energy AI 2020, 1, 100009. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; De La Cruz-Noriega, M.; Nazario-Naveda, R.; Benites, S.M.; Delfín-Narciso, D.; Rojas-Villacorta, W.; Romero, C.V. Bioelectricity through microbial fuel cells using avocado waste. Energy Rep. 2022, 8, 376–382. [Google Scholar] [CrossRef]
- Masebinu, R.; Kambule, N. Electricity consumption data of a middle-income household in Gauteng, South Africa: Pre and Post COVID-19 lockdown (2019–2021). Data Brief 2022, 43, 108341. [Google Scholar] [CrossRef]
- Hadjout, D.; Torres, J.; Troncoso, A.; Sebaa, A.; Martínez-Álvarez, F. Electricity consumption forecasting based on ensemble deep learning with application to the Algerian market. Energy 2022, 243, 123060. [Google Scholar] [CrossRef]
- Chew, K.W.; Chia, S.R.; Yen, H.-W.; Nomanbhay, S.; Ho, Y.-C.; Show, P.L. Transformation of Biomass Waste into Sustainable Organic Fertilizers. Sustainability 2019, 11, 2266. [Google Scholar] [CrossRef]
- Wainaina, S.; Awasthi, M.K.; Sarsaiya, S.; Chen, H.; Singh, E.; Kumar, A.; Ravindran, B.; Awasthi, S.K.; Liu, T.; Duan, Y.; et al. Resource recovery and circular economy from organic solid waste using aerobic and anaerobic digestion technologies. Bioresour. Technol. 2020, 301, 122778. [Google Scholar] [CrossRef]
- Ahuja, I.; Dauksas, E.; Remme, J.F.; Richardsen, R.; Løes, A.-K. Fish and fish waste-based fertilizers in organic farming–With status in Norway: A review. Waste Manag. 2020, 115, 95–112. [Google Scholar] [CrossRef]
- Rocamora, I.; Wagland, S.T.; Villa, R.; Simpson, E.D.; Fernández, O.; Bajón-Fernández, Y. Dry anaerobic digestion of organic waste: A review of operational parameters and their impact on process performance. Bioresour. Technol. 2020, 299, 122681. [Google Scholar] [CrossRef] [PubMed]
- Ronga, D.; Caradonia, F.; Parisi, M.; Bezzi, G.; Parisi, B.; Allesina, G.; Pedrazzi, S.; Francia, E. Using Digestate and Biochar as Fertilizers to Improve Processing Tomato Production Sustainability. Agronomy 2020, 10, 138. [Google Scholar] [CrossRef]
- Trombino, S.; Cassano, R.; Procopio, D.; Di Gioia, M.L.; Barone, E. Valorization of Tomato Waste as a Source of Carotenoids. Molecules 2021, 26, 5062. [Google Scholar] [CrossRef] [PubMed]
- Løvdal, T.; Van Droogenbroeck, B.; Eroglu, E.C.; Kaniszewski, S.; Agati, G.; Verheul, M.; Skipnes, D. Valorization of Tomato Surplus and Waste Fractions: A Case Study Using Norway, Belgium, Poland, and Turkey as Examples. Foods 2019, 8, 229. [Google Scholar] [CrossRef]
- Domínguez, R.; Gullón, P.; Pateiro, M.; Munekata, P.E.S.; Zhang, W.; Lorenzo, J.M. Tomato as Potential Source of Natural Additives for Meat Industry. A Review. Antioxidants 2020, 9, 73. [Google Scholar] [CrossRef]
- Christwardana, M.; Hadiyanto, H.; Motto, S.A.; Sudarno, S.; Haryani, K. Performance evaluation of yeast-assisted microalgal microbial fuel cells on bioremediation of cafeteria wastewater for electricity generation and microalgae biomass production. Biomass Bioenergy 2020, 139, 105617. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Al-Zaqri, N.; Yaakop, A.S.; Umar, K. Potato waste as an effective source of electron generation and bioremediation of pollutant through benthic microbial fuel cell. Sustain. Energy Technol. Assess. 2022, 53, 102560. [Google Scholar] [CrossRef]
- Obileke, K.; Onyeaka, H.; Meyer, E.L.; Nwokolo, N. Microbial fuel cells, a renewable energy technology for bio-electricity generation: A mini-review. Electrochem. Commun. 2021, 125, 107003. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ibrahim, M.N.M.; Rodríguez-Couto, S. Development and modification of materials to build cost-effective anodes for microbial fuel cells (MFCs): An overview. Biochem. Eng. J. 2020, 164, 107779. [Google Scholar] [CrossRef]
- Prathiba, S.; Kumar, P.S.; Vo, D.-V.N. Recent advancements in microbial fuel cells: A review on its electron transfer mechanisms, microbial community, types of substrates and design for bio-electrochemical treatment. Chemosphere 2022, 286, 131856. [Google Scholar] [CrossRef]
- Gul, H.; Raza, W.; Lee, J.; Azam, M.; Ashraf, M.; Kim, K.-H. Progress in microbial fuel cell technology for wastewater treatment and energy harvesting. Chemosphere 2021, 281, 130828. [Google Scholar] [CrossRef] [PubMed]
- Asefi, B.; Li, S.-L.; Moreno, H.A.; Sanchez-Torres, V.; Hu, A.; Li, J.; Yu, C.-P. Characterization of electricity production and microbial community of food waste-fed microbial fuel cells. Process. Saf. Environ. Prot. 2019, 125, 83–91. [Google Scholar] [CrossRef]
- Mohamed, S.N.; Hiraman, P.A.; Muthukumar, K.; Jayabalan, T. Bioelectricity production from kitchen wastewater using microbial fuel cell with photosynthetic algal cathode. Bioresour. Technol. 2020, 295, 122226. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, A.A.; Guerrero–Barajas, C.; Ibrahim, M.N.M.; Umar, K.; Yaakop, A.S. Local fruit wastes driven benthic microbial fuel cell: A sustainable approach to toxic metal removal and bioelectricity generation. Environ. Sci. Pollut. Res. 2022, 29, 32913–32928. [Google Scholar] [CrossRef]
- Kondaveeti, S.; Mohanakrishna, G.; Kumar, A.; Lai, C.; Lee, J.-K.; Kalia, V.C. Exploitation of Citrus Peel Extract as a Feedstock for Power Generation in Microbial Fuel Cell (MFC). Indian J. Microbiol. 2019, 59, 476–481. [Google Scholar] [CrossRef]
- Segundo, R.-F.; De La Cruz-Noriega, M.; Nazario-Naveda, R.; Benites, S.M.; Delfín-Narciso, D.; Angelats-Silva, L.; Díaz, F. Golden Berry Waste for Electricity Generation. Fermentation 2022, 8, 256. [Google Scholar] [CrossRef]
- Zafar, H.; Peleato, N.; Roberts, D. A comparison of reactor configuration using a fruit waste fed two-stage anaerobic up-flow leachate reactor microbial fuel cell and a single-stage microbial fuel cell. Bioresour. Technol. 2023, 374, 128778. [Google Scholar] [CrossRef]
- Rahman, W.; Yusup, S.; Mohammad, S.A. Screening of fruit waste as substrate for microbial fuel cell (MFC). In AIP Conference Proceedings; AIP Publishing LLC: Arau, Malaysia, 2021; Volume 2332, p. 020003. [Google Scholar]
- Rojas-Flores, S.; De La Cruz-Noriega, M.; Cabanillas-Chirinos, L.; Nazario-Naveda, R.; Gallozzo-Cardenas, M.; Diaz, F.; Murga-Torres, E. Potential Use of Coriander Waste as Fuel for the Generation of Electric Power. Sustainability 2023, 15, 896. [Google Scholar] [CrossRef]
- Prasidha, W. Electricity Production from Food Waste Leachate (Fruit and Vegetable Waste) using Double Chamber Microbial Fuel Cell: Comparison between Non-aerated and Aerated Configuration. ROTASI 2020, 22, 162–168. [Google Scholar]
- Naveenkumar, M.; Senthilkumar, K. Microbial fuel cell for harvesting bio-energy from tannery effluent using metal mixed biochar electrodes. Biomass Bioenergy 2021, 149, 106082. [Google Scholar] [CrossRef]
- Aiyer, K.S. How does electron transfer occur in microbial fuel cells? World J. Microbiol. Biotechnol. 2020, 36, 19. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.; Lee, S.-M.; Oh, S.-E.; Kim, E.J.; Hwang, Y.; Seo, D.; Kim, J.Y.; Kahng, Y.H.; Lee, Y.W.; Chung, S.-Y.; et al. Addition of reduced graphene oxide to an activated-carbon cathode increases electrical power generation of a microbial fuel cell by enhancing cathodic performance. Electrochim. Acta 2019, 297, 613–622. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Serrà, A.; Ibrahim, M.N.M.; Yaakop, A.S. Self-assembled oil palm biomass-derived modified graphene oxide anode: An efficient medium for energy transportation and bioremediating Cd (II) via microbial fuel cells. Arab. J. Chem. 2021, 14, 103121. [Google Scholar] [CrossRef]
- Hung, Y.-H.; Liu, T.-Y.; Chen, H.-Y. Renewable Coffee Waste-Derived Porous Carbons as Anode Materials for High-Performance Sustainable Microbial Fuel Cells. ACS Sustain. Chem. Eng. 2019, 7, 16991–16999. [Google Scholar] [CrossRef]
- Guan, C.-Y.; Tseng, Y.-H.; Tsang, D.C.; Hu, A.; Yu, C.-P. Wetland plant microbial fuel cells for remediation of hexavalent chromium contaminated soils and electricity production. J. Hazard. Mater. 2019, 365, 137–145. [Google Scholar] [CrossRef]
- Yaqoob, A.Y.; Hussian, N.A.; Alhejuje, M.M. Application of Heavy metals pollution index on two types of constructed wetland. Marsh Bull. 2022, 17, 22–29. [Google Scholar]
- Sreelekshmy, B.R.; Basheer, R.; Sivaraman, S.; Vasudevan, V.; Elias, L.; Shibli, S.M.A. Sustainable electric power generation from live anaerobic digestion of sugar industry effluents using microbial fuel cells. J. Mater. Chem. A 2020, 8, 6041–6056. [Google Scholar] [CrossRef]
- Iigatani, R.; Ito, T.; Watanabe, F.; Nagamine, M.; Suzuki, Y.; Inoue, K. Electricity generation from sweet potato-shochu waste using microbial fuel cells. J. Biosci. Bioeng. 2019, 128, 56–63. [Google Scholar] [CrossRef]
- Priya, A.D.; Setty, Y.P. Cashew apple juice as substrate for microbial fuel cell. Fuel 2019, 246, 75–78. [Google Scholar] [CrossRef]
- Antonopoulou, G.; Ntaikou, I.; Pastore, C.; di Bitonto, L.; Bebelis, S.; Lyberatos, G. An overall perspective for the energetic valorization of household food waste using microbial fuel cell technology of its extract, coupled with anaerobic digestion of the solid residue. Appl. Energy 2019, 242, 1064–1073. [Google Scholar] [CrossRef]
- Daud, N.N.M.; Ahmad, A.; Yaqoob, A.A.; Ibrahim, M.N.M. Application of rotten rice as a substrate for bacterial species to generate energy and the removal of toxic metals from wastewater through microbial fuel cells. Environ. Sci. Pollut. Res. 2021, 28, 62816–62827. [Google Scholar] [CrossRef] [PubMed]
- Simeon, M.I.; Asoiro, F.U.; Aliyu, M.; Raji, O.A.; Freitag, R. Polarization and power density trends of a soil-based microbial fuel cell treated with human urine. Int. J. Energy Res. 2020, 44, 5968–5976. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; Nazario-Naveda, R.; Benites, S.M.; Gallozzo-Cardenas, M.; Delfín-Narciso, D.; Díaz, F. Use of Pineapple Waste as Fuel in Microbial Fuel Cell for the Generation of Bioelectricity. Molecules 2022, 27, 7389. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, A.A.; Bin Abu Bakar, M.A.; Kim, H.-C.; Ahmad, A.; Alshammari, M.B.; Yaakop, A.S. Oxidation of food waste as an organic substrate in a single chamber microbial fuel cell to remove the pollutant with energy generation. Sustain. Energy Technol. Assess. 2022, 52, 102282. [Google Scholar] [CrossRef]
- Du, H.; Shao, Z. Synergistic effects between solid potato waste and waste activated sludge for waste-to-power conversion in microbial fuel cells. Appl. Energy 2022, 314, 118994. [Google Scholar] [CrossRef]
- Kamperidis, T.; Pandis, P.K.; Argirusis, C.; Lyberatos, G.; Tremouli, A. Effect of Food Waste Condensate Concentration on the Performance of Microbial Fuel Cells with Different Cathode Assemblies. Sustainability 2022, 14, 2625. [Google Scholar] [CrossRef]
- Segundo, R.-F.; Magaly, D.L.C.-N.; Benites, S.M.; Daniel, D.-N.; Angelats-Silva, L.; Díaz, F.; Luis, C.-C.; Fernanda, S.-P. Increase in Electrical Parameters Using Sucrose in Tomato Waste. Fermentation 2022, 8, 335. [Google Scholar] [CrossRef]
- Rehal, J.K.; Aggarwal, P.; Dhaliwal, I.; Sharma, M.; Kaushik, P. A Tomato Pomace Enriched Gluten-Free Ready-to-Cook Snack’s Nutritional Profile, Quality, and Shelf Life Evaluation. Horticulturae 2022, 8, 403. [Google Scholar] [CrossRef]
- Ramya, S.; Chandran, M.; King, I.J.; Jayakumararaj, R.; Loganathan, T.; Pandiarajan, G.; Kaliraj, P.; Pushpalatha, G.G.L.; Abraham, G.; Vijaya, V.; et al. Phytochemical Screening, GCMS and FTIR Profile of Bioactive Compounds in Solanum lycopersicum Wild Fruits collected from Palani Hill Ranges of the Western Ghats. J. Drug Deliv. Ther. 2022, 12, 56–64. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, Y.; Xi, H.; Yu, Y.; Song, Y.; Wu, C.; Zhou, Y. Evaluation methods of inhibition to microorganisms in biotreatment processes: A review. Water Cycle 2023, 4, 70–78. [Google Scholar] [CrossRef]
- Su, F.; Wang, F.; Zhang, C.; Lu, T.; Zhang, S.; Zhang, R.; Qi, X.; Liu, P. Ameliorating substance accessibility for microorganisms to amplify toluene degradation and power generation of microbial fuel cell by using activated carbon anode. J. Clean. Prod. 2022, 377, 134481. [Google Scholar] [CrossRef]
- Nawaz, A.; Haq, I.U.; Qaisar, K.; Gunes, B.; Raja, S.I.; Mohyuddin, K.; Amin, H. Microbial fuel cells: Insight into simultaneous wastewater treatment and bioelectricity generation. Process. Saf. Environ. Prot. 2022, 161, 357–373. [Google Scholar] [CrossRef]
- Hou, B.; Liu, X.; Zhang, R.; Li, Y.; Liu, P.; Lu, J. Investigation and evaluation of membrane fouling in a microbial fuel cell-membrane bioreactor systems (MFC-MBR). Sci. Total Environ. 2022, 814, 152569. [Google Scholar] [CrossRef]
- Hirose, S.; Inukai, K.; Nguyen, D.T.; Taguchi, K. Use of loofah electrodes coated with rice husk smoked charcoal and Japanese ink in a microbial fuel cell for muddy water treatment. Energy Rep. 2023, 9, 160–167. [Google Scholar] [CrossRef]
- Mbugua, J.K.; Mbui, D.N.; Mwaniki, J.M.; Mwaura, F.B. Electricity Generation by Clostridiumspp and Proteus Vulgaris from Rotten Tomatoes in a Double Chamber Microbial Fuel Cell. Int. J. Res. Stud. Microbiol. Biotechnol. 2018, 4, 32–33. [Google Scholar]
- Heard, G.M.; Fleet, G.H. CANDIDA|Yarrowia (Candida) Lipolytica. In Encyclopedia of Food Microbiology; Elsevier: Amsterdam, The Netherlands, 1999; pp. 360–365. ISBN 9780122270703. [Google Scholar]
- Logan, B.E.; Regan, J.M. Electricity-Producing Bacterial Communities in Microbial Fuel Cells. Trends Microbiol. 2006, 14, 512–518. [Google Scholar] [CrossRef]
- Gatti, M.N.; Milocco, R.H. A Biofilm Model of Microbial Fuel Cells for Engineering Applications. Int. J. Energy Environ. Eng. 2017, 8, 303–315. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; Cabanillas-Chirinos, L.; Nazario-Naveda, R.; Gallozzo-Cardenas, M.; Diaz, F.; Delfin-Narciso, D.; Rojas-Villacorta, W. Use of Tangerine Waste as Fuel for the Generation of Electric Current. Sustainability 2023, 15, 3559. [Google Scholar] [CrossRef]
- Greenman, J.; Gajda, I.; You, J.; Mendis, B.A.; Obata, O.; Pasternak, G.; Ieropoulos, I. Microbial Fuel Cells and Their Electrified Biofilms. Biofilm 2021, 3, 100057. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Mu, H.; Liu, W.; Zhang, R.; Guo, J.; Xian, M.; Liu, H. Electricigens in the Anode of Microbial Fuel Cells: Pure Cultures versus Mixed Communities. Microb. Cell Fact. 2019, 18, 39. [Google Scholar] [CrossRef]
- Park, D.H.; Zeikus, J.G. Electricity Generation in Microbial Fuel Cells Using Neutral Red as an Electronophore. Appl. Environ. Microbiol. 2000, 66, 1292–1297. [Google Scholar] [CrossRef]
- Kim, N.J.; Choe, Y.J.; Jeong, S.H.; Kim, S.H. Development of Microbial Fuel Cells Using Proteus Vulgaris. Bull. Korean Chem. Soc. 2000, 21, 44–48. [Google Scholar] [CrossRef]
- Verma, M.; Mishra, V. Recent Trends in Upgrading the Performance of Yeast as Electrode Biocatalyst in Microbial Fuel Cells. Chemosphere 2021, 284, 131383. [Google Scholar] [CrossRef] [PubMed]
- Rojas Flores, S.; Pérez-Delgado, O.; Naveda-Renny, N.; Benites, S.M.; De La Cruz-Noriega, M.; Delfin Narciso, D.A. Generation of Bioelectricity Using Molasses as Fuel in Microbial Fuel Cells. Environ. Res. Eng. Manag. 2022, 78, 19–27. [Google Scholar] [CrossRef]
- Gunawardena, A.; Fernando, S.; To, F. Performance of a Yeast-Mediated Biological Fuel Cell. Int. J. Mol. Sci. 2008, 9, 1893–1907. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).