Techno-Economic Feasibility Study for Organic and Plastic Waste Pyrolysis Pilot Plant in Malaysia
Abstract
:1. Introduction
2. Methodology
2.1. Oxidative Pyrolysis of OPW
2.2. Lifecycle Assessment (LCA) of the Pyrolysis System and Mitigated CO2
2.3. TEF Study for a Commercial Pyrolysis Plant
- Yield of furfural: 1.0%, at RM 303/kg
- Yield of formic acid: 8.4%, at RM 96/kg
- Yield of phenol: 4.1%, at RM 437/kg
3. LCA of Carbon Emissions for Pyrolysis and Chemical Production Plant
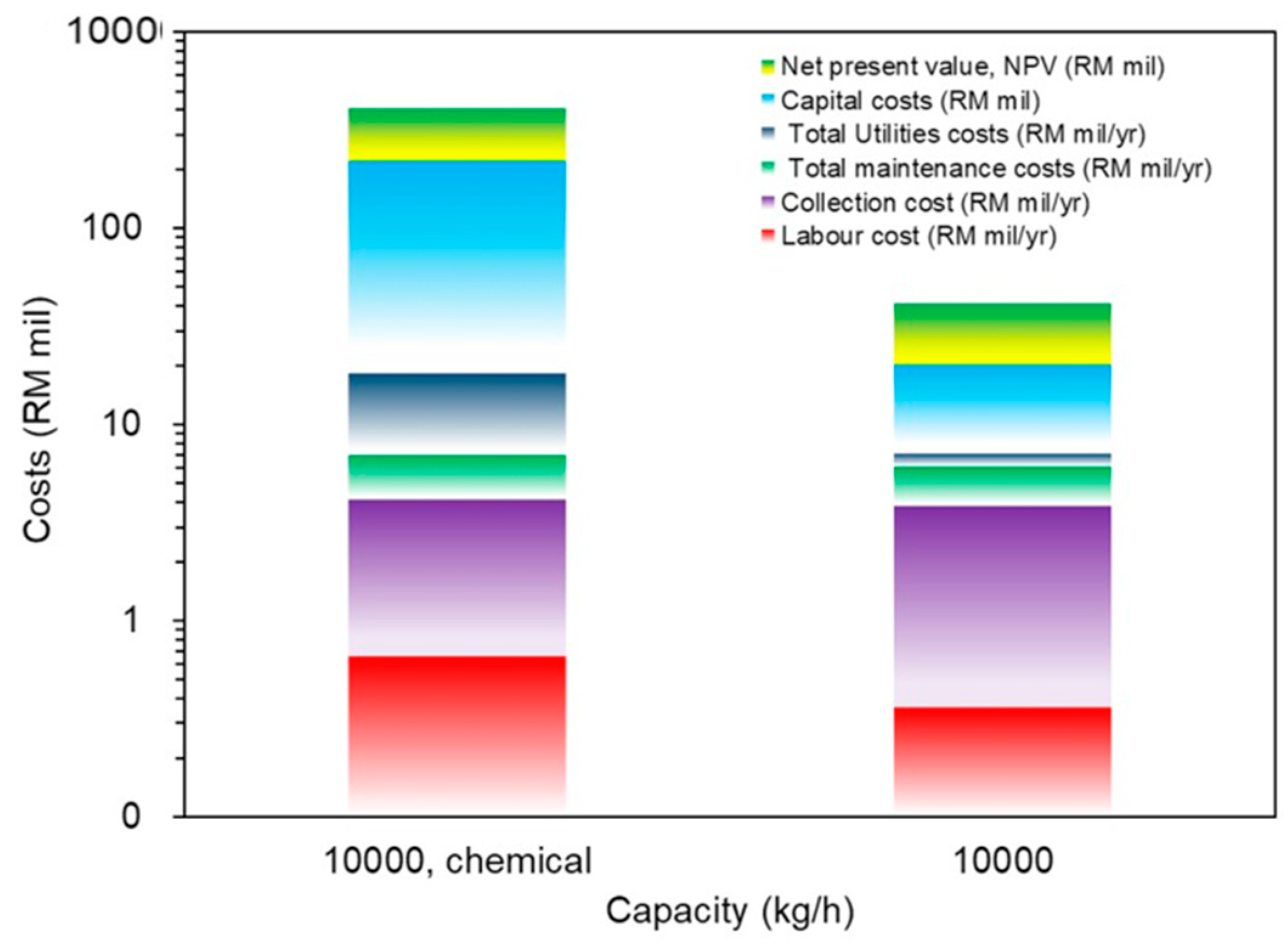
4. TEF Study of Commercial Pyrolysis Oil/Liquid and Chemical-Extraction Plant
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bong, C.P.-C.; Goh, R.K.Y.; Lim, J.-S.; Ho, W.S.; Lee, C.-T.; Hashim, H.; Abu Mansor, N.N.; Ho, C.S.; Ramli, A.R.; Takeshi, F. Towards low carbon society in Iskandar Malaysia: Implementation and feasibility of community organic waste composting. J. Environ. Manag. 2017, 203, 679–687. [Google Scholar] [CrossRef]
- Lim, I. Court Fines Two Selangor Factories RM120,000 for Illegal Plastic Recycling. Malay Mail, 17 May 2019. [Google Scholar]
- Yansaneh, O.Y.; Zein, S.H. Recent Advances on Waste Plastic Thermal Pyrolysis: A Critical Overview. Processes 2022, 10, 332. [Google Scholar] [CrossRef]
- Titone, V.; Gulino, E.F.; La Mantia, F.P. Recycling of Heterogeneous Mixed Waste Polymers through Reactive Mixing. Polymers 2023, 15, 1367. [Google Scholar] [CrossRef] [PubMed]
- Simler, K. An Idea Whose Time Has Come—Raising Malaysia’s Poverty Line. The Star, 1 September 2019. [Google Scholar]
- Sophonrat, N.; Sandström, L.; Zaini, I.N.; Yang, W. Stepwise pyrolysis of mixed plastics and paper for separation of oxygenated and hydrocarbon condensates. Appl. Energy 2018, 229, 314–325. [Google Scholar] [CrossRef]
- Qureshi, K.M.; Lup, A.N.K.; Khan, S.; Abnisa, F.; Daud, W.M.A.W. A technical review on semi-continuous and continuous pyrolysis process of biomass to bio-oil. J. Anal. Appl. Pyrolysis 2018, 131, 52–75. [Google Scholar] [CrossRef]
- Ghorbannezhad, P.; Kool, F.; Rudi, H.; Ceylan, S. Sustainable production of value-added products from fast pyrolysis of palm shell residue in tandem micro-reactor and pilot plant. Renew. Energy 2019, 145, 663–670. [Google Scholar] [CrossRef]
- Brueckner, T.M.; Pickup, P.G.; Hawboldt, K. Improvement of bark pyrolysis oil and value added chemical recovery by pervaporation. Fuel Process. Technol. 2019, 199, 106292. [Google Scholar] [CrossRef]
- Oh, S.-J.; Choi, G.-G.; Kim, J.-S. Fast pyrolysis of corn stover using ZnCl2: Effect of washing treatment on the furfural yield and solvent extraction of furfural. Energy 2015, 88, 697–702. [Google Scholar] [CrossRef]
- Aldrich, S. Catalog of Products. 2020. Available online: https://www.sigmaaldrich.com/catalog/product/aldrich/ (accessed on 17 January 2023).
- Alibaba. Price of Furfural. 2020. Available online: https://www.alibaba.com/showroom/furfural-price.html (accessed on 2 January 2023).
- Echemi. Product Information Phenol. 2020. Available online: https://www.echemi.com/productsInformation/pid_Seven3078-phenol.html (accessed on 20 January 2023).
- BASF. BASF Raises Prices for Formic Acid in Europe and in the Americas. 2016. Available online: https://www.basf.com/global/en/media/news-releases/2016/05/p-16-221.html (accessed on 4 February 2023).
- Hosseinzadeh, M.B.; Rezazadeh, S.; Rahimpour, H.R.; Taghvaei, H.; Rahimpour, M.R. Upgrading of lignin-derived bio-oil in non-catalytic plasma reactor: Effects of operating parameters on 4-methylanisole conversion. Chem. Eng. Res. Des. 2015, 104, 296–305. [Google Scholar] [CrossRef]
- Xiu, S.; Shahbazi, A. Bio-oil production and upgrading research: A review. Renew. Sustain. Energy Rev. 2012, 16, 4406–4414. [Google Scholar] [CrossRef]
- Sommani, P.; Mankong, N.; Vitidsant, T.; Lothongkum, A.W. Cracking of used vegetabele oil mixed with polypropylene waste in the presence of activated carbon. ASEAN Eng. J. Part B 2013, 4, 16–24. [Google Scholar]
- Buchori, L.; Istadi, I.; Purwanto, P.; Kurniawan, A.; Maulana, T.I. Preliminary Testing of Hybrid Catalytic-Plasma Reactor for Biodiesel Production Using Modified-Carbon Catalyst. Bull. Chem. React. Eng. Catal. 2016, 11, 59–65. [Google Scholar] [CrossRef]
- Lim, M.; Zulkifli, A.Z.S. Investigation of biomass surface modification using non-thermal plasma treatment. Plasma Sci. Technol. 2018, 20, 115502. [Google Scholar] [CrossRef]
- Lim, M.T.; Zulkifli, A.Z.S.; Jayapalan, K.K.; Chin, O. Development of a dimensionless parameter for characterization of dielectric barrier discharge devices with respect to geometrical features. Plasma Sci. Technol. 2017, 19, 095402. [Google Scholar] [CrossRef]
- Al-Asadi, M.; Miskolczi, N.; Eller, Z. Pyrolysis-gasification of wastes plastics for syngas production using metal modified zeolite catalysts under different ratio of nitrogen/oxygen. J. Clean. Prod. 2020, 271, 122186. [Google Scholar] [CrossRef]
- Grycová, B.; Koutník, I.; Pryszcz, A. Pyrolysis process for the treatment of food waste. Bioresour. Technol. 2016, 218, 1203–1207. [Google Scholar] [CrossRef]
- Tang, Y.; Huang, Q.; Sun, K.; Chi, Y.; Yan, J. Co-pyrolysis characteristics and kinetic analysis of organic food waste and plastic. Bioresour. Technol. 2018, 249, 16–23. [Google Scholar] [CrossRef]
- Ding, Y.; Huang, B.; Wu, C.; He, Q.; Lu, K. Kinetic model and parameters study of lignocellulosic biomass oxidative pyrolysis. Energy 2019, 181, 11–17. [Google Scholar] [CrossRef]
- Kim, K.H.; Bai, X.; Rover, M.; Brown, R.C. The effect of low-concentration oxygen in sweep gas during pyrolysis of red oak using a fluidized bed reactor. Fuel 2014, 124, 49–56. [Google Scholar] [CrossRef]
- Ferreira, R.A.d.R.; da Silva Meireles, C.; Assunção, R.M.N.; Barrozo, M.A.S.; Soares, R.R. Optimization of the oxidative fast pyrolysis process of sugarcane straw by TGA and DSC analyses. Biomass Bioenergy 2020, 134, 105456. [Google Scholar] [CrossRef]
- Pharmacompass. API Price Trend Dashboard–Nitrogen. 8 December 2021. Available online: https://www.pharmacompass.com/price/nitrogen (accessed on 8 December 2021).
- Chen, D. 16—Examples of thermochemical and biological treatment technologies for sustainable waste management in China. In Substitute Natural Gas from Waste; Materazzi, M., Foscolo, P.U., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 425–454. [Google Scholar]
- Oh, J.-I.; Lee, J.; Lee, T.; Ok, Y.S.; Lee, S.-R.; Kwon, E.E. Strategic CO2 utilization for shifting carbon distribution from pyrolytic oil to syngas in pyrolysis of food waste. J. CO2 Util. 2017, 20, 150–155. [Google Scholar] [CrossRef]
- Kwon, E.E.; Kim, S.; Lee, J. Pyrolysis of waste feedstocks in CO2 for effective energy recovery and waste treatment. J. CO2 Util. 2019, 31, 173–180. [Google Scholar] [CrossRef]
- Chen, Z.; Wu, D.; Chen, L.; Ji, M.; Zhang, J.; Du, Y.; Wu, Z. The fast co-pyrolysis study of PVC and biomass for disposing of solid wastes and resource utilization in N2 and CO2. Process Saf. Environ. Prot. 2021, 150, 489–496. [Google Scholar] [CrossRef]
- Fu, D.; Farag, S.; Chaouki, J.; Jessop, P.G. Extraction of phenols from lignin microwave-pyrolysis oil using a switchable hydrophilicity solvent. Bioresour. Technol. 2014, 154, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.T.; Ho, W.S.; Hashim, H.; Lee, C.T.; Taib, M.R.; Ho, C.S. Energy, economic and environmental (3E) analysis of waste-to-energy (WTE) strategies for municipal solid waste (MSW) management in Malaysia. Energy Convers. Manag. 2015, 102, 111–120. [Google Scholar] [CrossRef]
- Ritchie, H.; Our World in Data. The Carbon Footprint of Foods: Are Differences Explained by the Impacts of Methane? 2020. Available online: https://ourworldindata.org/carbon-footprint-food-methane (accessed on 10 November 2021).
- Ren, T.; Patel, M.K. Basic petrochemicals from natural gas, coal and biomass: Energy use and CO2 emissions. Resour. Conserv. Recycl. 2009, 53, 513–528. [Google Scholar] [CrossRef]
- Fivga, A.; Dimitriou, I. Pyrolysis of plastic waste for production of heavy fuel substitute: A techno-economic assessment. Energy 2018, 149, 865–874. [Google Scholar] [CrossRef]
- Luo, D.; Yin, W.; Han, D.; He, H.; Xia, S. Glycolic acid and formic acid production from pyrolysis oil water-soluble fraction by catalytic oxidation. Chem. Eng. Sci. 2021, 239, 116644. [Google Scholar] [CrossRef]
- IHS Markit. Crude Oil-to-Chemicals (COTC): A Look Inside Our Technology & Economic Analyses from the 2019–2014 Process and Economics Program (PEP); IHS Markit: London, UK, 2019. [Google Scholar]
- Performance Management and Delivery Unit Malaysia (PEMANDU), Solid Waste Management Lab 2015. Final Report. 2015; Ministry of Local Government Development, Malaysia. Available online: https://jpspn.kpkt.gov.my/wp-content/uploads/2022/07/slaid_dapatan_makmal.pdf (accessed on 29 December 2022).
- Wright, M.M.; Daugaard, D.E.; Satrio, J.A.; Brown, R.C. Techno-economic analysis of biomass fast pyrolysis to transportation fuels. Fuel 2010, 89, S2–S10. [Google Scholar] [CrossRef]
- Jaroenkhasemmeesuk, C.; Tippayawong, N. Technical and Economic Analysis of A Biomass Pyrolysis Plant. Energy Procedia 2015, 79, 950–955. [Google Scholar] [CrossRef]
- Jahirul, M.I.; Rasul, M.G.; Chowdhury, A.A.; Ashwath, N. Biofuels Production through Biomass Pyrolysis—A Technological Review. Energies 2012, 5, 4952–5001. [Google Scholar] [CrossRef]
- Islam, M.; Ani, F. Techno-economics of rice husk pyrolysis, conversion with catalytic treatment to produce liquid fuel. Bioresour. Technol. 2000, 73, 67–75. [Google Scholar] [CrossRef]
- The Edge Markets. Domestic Carbon Trading to Be Implemented in Phases from End of Next Year. 2021. Available online: https://www.theedgemarkets.com/article/domestic-carbon-trading-be-implemented-phases-end-next-year-%E2%80%94-minister (accessed on 12 January 2022).
- Chemical Book. Product Prices from CAS Database List. 2021. Available online: https://www.chemicalbook.com/SupplierPriceList_EN.aspx?cbn=CB4854063&page=3#price (accessed on 25 August 2021).
- Merck and Sigma-Aldrich. Product Prices for Formic acid, Furfural, and Phenol. 2021. Available online: https://www.sigmaaldrich.com/MY/en/search/phenol?focus=products&page=1&perPage=30&sort=relevance&term=phenol&type=product (accessed on 25 August 2021).
- Thomas, V.M. The National Academis of Sciences, Engineering and Medicine: Current Methods for Life Cycle Analyses of Low-Carbon Transportation Fuels in the United States. 2021. Available online: https://www.nationalacademies.org/our-work/current-methods-for-life-cycle-analyses-of-low-carbon-transportation-fuels-in-the-united-states (accessed on 10 November 2021).
- Xikai, M.; Wang, L.; Li, J.; Quan, X.; Wu, T. Comparison of regression models for estimation of carbon emissions during building’s lifecycle using designing factors: A case study of residential buildings in Tianjin, China. Energy Build. 2019, 204, 109519. [Google Scholar] [CrossRef]
- Lim, M.Z.; Tan, E.S.; Chai, Y.H.; Chin, B.L.F.; Juwono, F.H.; Hisham, D.H.B.; Anuar, M.A.M. Pilot-scale flue gas pyrolysis system for organic and plastic wastes with improved liquid properties in a non-thermal plasma reactor. J. Anal. Appl. Pyrolysis 2023, 173, 106062. [Google Scholar] [CrossRef]
- Malaysian Investment Development Authority (MIDA). Crude Oil to Chemicals (COTC) Technology: A New Paradigm Shift. Available online: https://www.mida.gov.my/crude-oil-to-chemicals-cotc-technology-a-new-paradigm-shift/ (accessed on 20 July 2023).









| Extractables | Function | Bulk/Cost (USD/ton) | Estimated Shelf Cost (RM/Liter) [4] |
|---|---|---|---|
| Formic acid |
| 44 [1] | 70–96 |
| Furfural |
| 900–2000 [2] | 56–303 |
| Phenol |
| 840 [3] | 125–437 |
| Organic and Plastic Waste Capacity (kg/h) | 20 | 100 | 1000 | 10,000 |
| Capital costs (RM mil) | 2.28 | 2.52 | 7.20 | 13.20 |
| Management, production (RM/yr) | 100,000.00 | 100,000.00 | 100,000.00 | 100,000.00 |
| Number of engineers | - | - | - | 1 |
| Mean salary of engineers (RM/month) | - | - | - | 8333.33 |
| Engineers, production (RM/yr) | - | - | - | 173,286.21 |
| Engineers, maintenance (RM/yr) | - | - | - | 173,286.21 |
| Number of operators and technicians | 2 | 4 | 6 | 8 |
| Mean salary operators and technicians (RM/month) | 4166.67 | 4166.67 | 4166.67 | 2083.33 |
| Operators and technicians, production (RM/yr) | 50,000.00 | 50,000.00 | 50,000.00 | 50,000.00 |
| Operators and technicians, maintenance (RM/yr) | 50,000.00 | 50,000.00 | 50,000.00 | 50,000.00 |
| Number of executives | 1 | 1 | 1 | 2 |
| Mean salary of executives (RM/month) | 2500.00 | 2500.00 | 2500.00 | 2500.00 |
| Executive (RM/yr) | 30,000.00 | 30,000.00 | 44,362.73 | 103,971.73 |
| Total Labor Costs | 62,000.00 | 620,000.00 | 1,073,202.44 | 2,009,031.75 |
| Organic and Plastic Waste Capacity (kg/h) | 20 | 100 | 1000 | 10,000 |
| Waste collection costs (RM/ton/day) | 300 | 275 | 150 | 100 |
| Amount of waste (tons/day) | 0.24 | 1.20 | 12.00 | 120.00 |
| Number of collectors (staff) | 1.00 | 5.00 | 25.00 | 180.00 |
| Mean cost of collectors (RM/month) | 1752.00 | 1606.00 | 1752.00 | 1622.22 |
| Total Collection Costs (RM/yr) | 21,024.00 | 96,360.00 | 525,600.00 | 3,504,000.00 |
| Clear blockage (RM/yr) | 5000.00 | 7500.00 | 20,000.00 | 40,000.00 |
| Repair conveyer (RM/yr) | 50,000.00 | 75,000.00 | 200,000.00 | 400,000.00 |
| Maintenance of pyrolyzing agent systems (RM/yr) | 25,000.00 | 37,500.00 | 100,000.00 | 200,000.00 |
| Repair compressor (RM/yr) | 25,000.00 | 37,500.00 | 100,000.00 | 200,000.00 |
| Repair fluidized bed and auxiliary equipment (RM/yr) | 25,000.00 | 37,500.00 | 100,000.00 | 200,000.00 |
| Repair leakages (RM/yr) | 20,000.00 | 30,000.00 | 80,000.00 | 160,000.00 |
| Major equipment overhaul (RM/yr) | 100,000.00 | 150,000.00 | 400,000.00 | 800,000.00 |
| Total Maintenance Costs (RM/yr) | 250,000.00 | 375,000.00 | 1,000,000.00 | 2,000,000.00 |
| Organic and Plastic Waste Capacity (kg/h) | 20 | 100 | 1000 | 10,000 |
| Power consumption (RM/yr) | 72,829.27 | 109,243.91 | 291,317.09 | 582,634.18 |
| For heating up reactor (RM/yr) | 1391.65 | 6958.23 | 69,582.30 | 695,822.96 |
| Total Utilities Costs (RM/yr) | 74,220.92 | 116,202.14 | 360,899.39 | 1,278,457.14 |
| Organic and Plastic Waste Capacity (kg/h) | 20 | 100 | 1000 | 10,000 |
| Carbon price (RM/ton CO2) | 50 to 150 (proposed by Penang Institute in 2019) | |||
| CO2 mitigated from landfill (kg CO2/kg MSW) | 1.25 [33] | |||
| CO2 mitigated from landfill (ton CO2/yr) | 140 | 701 | 7008 | 70,080 |
| Flue gas flow rate (kg/h) | 10 | 50 | 500 | 5000 |
| Average CO2 content in flue gas (%) | 12 (based on power plant values) | |||
| CO2 mitigated from flue gas (ton CO2/yr) | 8 | 42 | 420 | 4205 |
| Power consumption, including OPW prep. (kW) | 21 | 31 | 42 * | 83 |
| Grid emission factor (kg CO2/kWh) | 0.57 [33] | |||
| Grid emission per year (ton CO2/yr) | 83 | 125 | 166 | 332 |
| Net CO2 mitigated (ton CO2/yr) | 101 | 794 | 9014 | 91,473 |
| Yield of pyrolysis oil (%) | 5 (based on exp. in this project published in [49]) | |||
| Selling price of pyrolysis oil (RM/kg) | 0.60 [based on information from local company, 2022] | |||
| Tipping fees for pyrolysis plant (RM/ton/day) | 0 to 250 [39] | |||
| Capacity (kg/h) | 20 | 100 | 1000 | 10,000 |
|---|---|---|---|---|
| Capital cost (RM million) * | 2.66 | 4.43 | 26.28 | 203.97 |
| Capacity (kg/h) | 20 | 100 | 1000 | 10,000 |
|---|---|---|---|---|
| Total operating costs annually (RM/yr) | 993,732 | 1,504,537 | 5,238,353 | 17,847,238 |
| Power consumption, including OPW prep. and chemical extraction (kW) | 41 | 62 | 166 | 332 |
| Net CO2 mitigated (ton CO2/yr) | 18 | 669 | 8516 | 90,476 |
| Yield, liquid (kg/kg waste) | 0.05 (based on exp. in this study) | |||
| Yield, formic acid (kg/kg waste) | 0.084 [37] | |||
| Yield, furfural (kg/kg waste) | 0.010 [29] | |||
| Yield, phenol (kg/kg waste) | 0.041 [32] | |||
| Price of formic acid (RM/kg) | 96 * | |||
| Price of furfural (RM/kg) | 303 * | |||
| Price of phenol (RM/kg) | 437 * | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lim, M.; Tan, E.S. Techno-Economic Feasibility Study for Organic and Plastic Waste Pyrolysis Pilot Plant in Malaysia. Sustainability 2023, 15, 14280. https://doi.org/10.3390/su151914280
Lim M, Tan ES. Techno-Economic Feasibility Study for Organic and Plastic Waste Pyrolysis Pilot Plant in Malaysia. Sustainability. 2023; 15(19):14280. https://doi.org/10.3390/su151914280
Chicago/Turabian StyleLim, Mooktzeng, and Ee Sann Tan. 2023. "Techno-Economic Feasibility Study for Organic and Plastic Waste Pyrolysis Pilot Plant in Malaysia" Sustainability 15, no. 19: 14280. https://doi.org/10.3390/su151914280
APA StyleLim, M., & Tan, E. S. (2023). Techno-Economic Feasibility Study for Organic and Plastic Waste Pyrolysis Pilot Plant in Malaysia. Sustainability, 15(19), 14280. https://doi.org/10.3390/su151914280









