Red Mud as a Secondary Resource of Low-Grade Iron: A Global Perspective
Abstract
:1. Introduction
2. RM Waste Management Strategies: A Brief Overview
2.1. Construction Materials
2.1.1. Cement
2.1.2. Building Materials
2.1.3. Glass-Ceramics, Geopolymers, Catalysts
2.2. Resource Recovery of Metals
2.2.1. Aluminium
2.2.2. Titanium
2.2.3. Rare Earths and Other Elements
3. Iron Recovery from Red Mud: Key Techniques
3.1. Magnetic Separation
3.2. Carbothermal Reduction
3.3. Microwave Carbothermal Reduction
3.4. Smelting Reduction
3.5. Suspension Reduction
3.6. Hydrometallurgy
3.7. Other Techniques
4. Global Scenario
4.1. INDIA
4.1.1. Case Study 1
4.1.2. Case Study 2
4.1.3. Case Study 3
4.2. CHINA
4.2.1. Case Study 4
4.2.2. Case Study 5
4.2.3. Case Study 6
4.2.4. Case Study 7
4.3. GREECE
4.3.1. Case Study 8
4.3.2. Case Study 9
4.3.3. Case Study 10
4.4. ITALY
Case Study 11
4.5. FRANCE
Case Study 12
4.6. RUSSIA
4.6.1. Case Study 13
4.6.2. Case Study 14
5. Comparison with Low Grade Iron Ores
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Zhang, R.; Zheng, S.; Ma, S.; Zhang, Y. Recovery of alumina and alkali in Bayer red mud by the formation of andradite-grossular hydrogarnet in hydrothermal process. J. Hazard. Mater. 2011, 189, 827–835. [Google Scholar] [CrossRef]
- Archambo, M.; Kawatra, S.K. Red mud: Fundamentals and new avenues for utilization. Miner. Process. Extr. Metall. Rev. 2020, 42, 427–450. [Google Scholar] [CrossRef]
- Yu, H.; Pan, X.; Dong, K.; Zhang, W.; Bi, S. The Sintering and Leaching of Low-Grade Diasporic Bauxite by the Improved Lime-Sintering Process. Adv. Mater. Res. 2013, 616–618, 1051–1054. [Google Scholar] [CrossRef]
- Wang, P.; Liu, D.Y. Physical and Chemical Properties of Sintering Red Mud and Bayer Red Mud and the Implications for Beneficial Utilization. Materials 2012, 5, 1800–1810. [Google Scholar] [CrossRef] [Green Version]
- Feng, Y.; Yang, C. Analysis on Physical and Mechanical Properties of Red Mud Materials and Stockpile Stability after Dilatation. Adv. Mater. Sci. Eng. 2018, 2018, 8784232. [Google Scholar] [CrossRef] [Green Version]
- Patel, S.; Pal, B.K. Current Status of an Industrial Waste: Red Mud an Overview. IJLTEMAS 2015, 7, 1–16. [Google Scholar]
- Wang, L.; Sun, N.; Tang, H.; Sun, W. A review on comprehensive utilization of red mud and prospect analysis. Minerals 2019, 9, 362. [Google Scholar] [CrossRef] [Green Version]
- Boudreault, R.; Fournier, J.; Primeau, D.; Labrecque-Gilbertm, M.M. Processes for Treating Red Mud. US20150275330A1, 31 January 2017. [Google Scholar]
- Xue, S.; Wu, Y.; Li, Y.; Kong, X.; Zhu, F.; William, H.; Li, X.; Ye, Y. Industrial wastes applications for alkalinity regulation in bauxite residue: A comprehensive review. J. Cent. South Univ. 2019, 26, 268–288. [Google Scholar] [CrossRef]
- Arroyo, F.; Luna-Galiano, Y.; Leiva, C.; Vilches, L.F.; Fernandez-Pereira, C. Environmental risks and mechanical evaluation of recycling red mud in bricks. Environ. Res. 2020, 186, 109537. [Google Scholar] [CrossRef]
- Khairul, M.A.; Zanganeh, J.; Moghtaderi, B. The composition, recycling and utilisation of Bayer red mud. Resour. Conserv. Recyc. 2019, 141, 483–498. [Google Scholar] [CrossRef]
- Garg, A.; Yadav, H. Study of red mud as an alternative building material for interlocking block manufacturing in construction industry. Int. J. Mater. Sci. Eng. 2015, 3, 295–300. [Google Scholar]
- Paramguru, R.K.; Rath, P.C.; Misra, V.N. Trends in red mud utilization: A review. Miner. Process. Extr. Metall. Rev. 2004, 26, 1–29. [Google Scholar] [CrossRef]
- Wang, S.; Ang, H.M.; Tade, M.O. Novel applications of red mud as coagulant, adsorbent and catalyst for environmentally benign processes. Chemosphere 2008, 72, 1621–1635. [Google Scholar] [CrossRef]
- Liu, X.; Han, Y.; He, F.; Gao, P.; Yuan, S. Characteristic, hazard and iron recovery technology of red mud—A critical review. J. Hazard. Mater. 2021, 420, 126542. [Google Scholar] [CrossRef]
- Snars, K.; Gilkes, R.J. Evaluation of bauxite residues (red muds) of different origins for environmental applications. Appl. Clay Sci. 2009, 46, 13–20. [Google Scholar] [CrossRef]
- Klauber, C.; Grafe, M.; Power, G. Bauxite residue issues: II. options for residue utilization. Hydrometallurgy 2011, 108, 11–32. [Google Scholar] [CrossRef]
- Piga, L.; Pochetti, F.; Stoppa, L. Recovering metals from red mud generated during alumina production. JOM 1993, 45, 54–59. [Google Scholar] [CrossRef]
- Rai, S.; Bahadure, S.; Chaddha, M.J.; Agnihotri, A. Disposal practices and utilization of red mud (Bauxite Residue): A review in Indian context and abroad. J. Sustain. Metall. 2020, 6, 1–8. [Google Scholar] [CrossRef]
- Das, B.; Mohanty, K. A review on advances in sustainable energy production through various catalytic processes by using catalysts derived from waste red mud. Renew. Energy 2019, 143, 1791–1811. [Google Scholar] [CrossRef]
- Liu, Y.; Lin, C.; Wu, Y. Characterization of red mud derived from a combined Bayer process and bauxite calcination method. J. Hazard. Mater. 2007, 146, 255–261. [Google Scholar] [CrossRef]
- Mayes, W.M.; Jarvis, A.P.; Burke, I.T.; Walton, M.; Feigl, V.R.; Klebercz, O.; Gruiz, K. Dispersal and attenuation of trace contaminants downstream of the Ajka bauxite residue (red mud) depository failure. Hung. Environ. Sci. Technol. 2011, 45, 5147–5155. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Pranolo, Y.; Cheng, C.Y. Recovery of scandium from synthetic red mud leach solutions by solvent extraction with D2EHPA. Sep. Purif. Technol. 2013, 108, 96–102. [Google Scholar] [CrossRef]
- Ruyters, S.; Mertens, J.; Vassilieva, E.; Dehandschutter, B.; Poffijn, A.; Smolders, E. The Red Mud Accident in Ajka (Hungary): Plant Toxicity and Trace Metal Bioavailability in Red Mud Contaminated Soil. Environ. Sci. Technol. 2011, 45, 1616–1622. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, R.; Bandopadhyay, A. Innovative methodologies for the utilisation of wastes from metallurgical and allied industries. Resour. Conserv. Recycl. 2006, 48, 301–314. [Google Scholar] [CrossRef]
- Liu, Z.; Li, H. Metallurgical process for valuable elements recovery from red mud—A review. Hydrometallurgy 2015, 155, 29–43. [Google Scholar] [CrossRef]
- Pascual, J.; Corpas, F.; López-Beceiro, J.; Benítez-Guerrero, M.; Artiaga, R. Thermal characterization of a Spanish red mud. J. Therm. Anal. Calorim. 2009, 96, 407–412. [Google Scholar] [CrossRef]
- Samal, S.; Ray, A.K.; Bandopadhyay, S.A. Proposal for resources, utilization and processes of red mud in India—A review. Int. J. Miner. Process. 2013, 118, 43–55. [Google Scholar] [CrossRef]
- Evans, K. The History, Challenges, and New Developments in the Management and Use of Bauxite Residue. J. Sustain. Met. 2016, 2, 316–331. [Google Scholar] [CrossRef] [Green Version]
- Bonomi, C.; Giannopoulou, I.; Panias, D. Correlation of Scandium and Titanium during Leaching of Bauxite Residue (Red Mud) by an Imidazolium Ionic Liquid. In Proceedings of the 2nd Conference on European Rare Earth Resources; Balomenos, E., Marinos, D., Eds.; Heliotopos Conferences Ltd.: Santorini, Greece, 2017; pp. 182–184. [Google Scholar]
- Sutar, H.; Mishra, S.C.; Sahoo, S.K.; Maharana, H. Progress of red mud utilization: An overview. Am. Chem. Sci. J. 2014, 4, 255–279. [Google Scholar] [CrossRef]
- Liu, Y.; Naidu, R. Hidden values in bauxite residue (red mud): Recovery of metals. Waste Manag. 2014, 34, 2662–2673. [Google Scholar] [CrossRef] [PubMed]
- Steel industry—Statistics & Facts. Available online: https://www.statista.com/topics/1149/steel-industry/ (accessed on 30 November 2021).
- Kumar, R.; Srivastava, J.P.; Premchand. Utilization of iron values of red mud for metallurgical applications. In Environmental and Waste Management; Bandopadhyay, A., Goswami, N.G., Jamshedpur, P.R.R., Eds.; National Metallurgical laboratories: Jamshedpur, India, 1998; pp. 108–119. [Google Scholar]
- Sparks, B.D.; Sirianni, A.F. Beneficiation of a phosphoriferous iron ore by agglomeration methods. Int. J. Miner. Process. 1974, 1, 231–241. [Google Scholar] [CrossRef]
- Bao, Q.; Guo, L.; Guo, Z. A novel direct reduction-flash smelting separation process of treating high phosphorous iron ore fines. Powder Metall. 2021, 377, 149–162. [Google Scholar] [CrossRef]
- Jorgen, D.J. U.S. Geological Survey, Mineral Commodities Summary. Available online: https://minerals.usgs.gov/minerals/pubs/commodity/ironore/mcs-2010-feore.pdf (accessed on 28 November 2020).
- Muwanguzi, A.J.B.; Karasev, A.V.; Byaruhanga, J.K.; Jönsson, P.G. Characterization of Chemical Composition and Microstructure of Natural Iron Ore from Muko Deposits. Int. Sch. Res. Notices 2012, 2012, 174803. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, A.; Sahu, K.K.; Pandey, B.D. Solid waste management in non-ferrous industries in India. Resour. Conserv. Recycl. 2004, 42, 99–102. [Google Scholar] [CrossRef]
- Abhilash; Sinha, S.; Sinha, M.K.; Pandey, B.D. Extraction of lanthanum and cerium from Indian red mud. Int. J. Miner. Process. 2014, 127, 70–73. [Google Scholar] [CrossRef]
- Mukiza, E.; Zhang, L.; Liu, X.; Zhang, N. Utilization of red mud in road base and subgrade materials: A review. Resour. Conserv. Recycl. 2019, 141, 187–199. [Google Scholar] [CrossRef]
- Pontikes, Y.; Angelopoulos, G.N. Bauxite residue in cement and cementitious applications: Current status and a possible way forward. Resour. Conserv. Recycl. 2013, 73, 53–63. [Google Scholar] [CrossRef]
- Feng, X.-p.; Liu, X.-m.; Sun, H.-h.; Bai, X.; Niu, X.-l. Study on the High Use Ratio of Red Mud in Cementitious Material. Multipurpose Util. Miner. Resour. 2007, 4, 35–37. [Google Scholar]
- Liu, R.-X.; Poon, C.-S. Utilization of red mud derived from bauxite in self-compacting concrete. J. Clean. Prod. 2016, 112, 384–391. [Google Scholar] [CrossRef]
- Nikbin, I.M.; Aliaghazadeh, M.; Sh, C.; Fathollahpour, A. Environmental impacts and mechanical properties of lightweight concrete containing bauxite residue (red mud). J. Clean. Prod. 2018, 172, 2683–2694. [Google Scholar] [CrossRef]
- Kacker, K.P.; Chandra, D. Production of building bricks from red mud rejects of aluminium plants. Ind. Ceram. 1977, 19, 712–723. [Google Scholar]
- Prasad, P.M.; Kachhawha, J.S.; Gupta, R.C.; Mankhand, T.R.; Sharma, J.M. Processing and Applications of Red Muds. In Key Engineering Materials; Trans Tech Publications Ltd.: Bäch, Switzerland, 1985; Volume 8, pp. 31–52. [Google Scholar]
- Dodoo-Arhin, D.; Konadu, D.S.; Annan, E.; Buabeng, F.P.; Yaya, A.; Agyei-Tuffour, B. Fabrication and Characterisation of Ghanaian Bauxite Red Mud-Clay Composite Bricks for Construction Applications. Am. J. Mater. Sci. 2013, 3, 110–119. [Google Scholar]
- Alam, S.; Das, S.K.; Rao, B.H. Characterization of coarse fraction of red mud as a civil engineering construction material. J. Clean. Prod. 2017, 168, 679–691. [Google Scholar] [CrossRef]
- Wang, L.; Iris, K.M.; Tsang, D.C.W.; Li, S.; Li, J.; Poon, C.S.; Wang, Y.S.; Dai, J.G. Transforming wood waste into water-resistant magnesia-phosphate cement particleboard modified by alumina and red mud. J. Clean. Prod. 2017, 168, 452–462. [Google Scholar] [CrossRef]
- Tang, W.C.; Wang, Z.; Donne, S.W.; Forghani, M.; Liu, Y. Influence of red mud on mechanical and durability performance of self-compacting concrete. J. Hazard Mater. 2019, 379, 120802. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kim, M.; Sohn, J.; Park, H. Applicability of gold tailings, waste lime stone, red mud, and ferronickel slag for producing glass fibers. J. Clean. Prod. 2018, 203, 957–965. [Google Scholar] [CrossRef]
- Krivenko, P.; Kovalchuk, O.; Pasko, A.; Croymans, T.; Hult, M.; Lutter, G.; Vandevenne, N.; Schreurs, S.; Schroeyers, W. Development of alkali activated cements and concrete mixture design with high volumes of red mud. Construct. Build. Mater. 2017, 151, 819–826. [Google Scholar] [CrossRef]
- Lemougna, P.N.; Wang, K.T.; Tang, Q.; Cui, X.M. Synthesis and characterization of low temperature (<800 °C) ceramics from red mud geopolymer precursor. Construct. Build. Mater. 2017, 131, 564–573. [Google Scholar] [CrossRef]
- Yang, Z.; Mocadlo, R.; Zhao, M.; Sisson, R.D., Jr.; Tao, M.; Liang, J. Preparation of a geopolymer from red mud slurry and class F fly ash and its behavior at elevated temperatures. Constr. Build. Mat. 2019, 221, 308. [Google Scholar] [CrossRef]
- Vigneshwaran, S.; Uthayakumar, M.; Arumugaprabu, V. Potential use of industrial waste-red mud in developing hybrid composites: A waste management approach. J. Clean. Prod. 2020, 276, 124278. [Google Scholar] [CrossRef]
- Amritphale, S.S.; Anshul, A.; Chandra, N.; Ramakrishnan, N. A novel process for making radiopaque materials using bauxite-red mud. J. Eur. Ceram. Soc. 2007, 27, 1945–1951. [Google Scholar] [CrossRef]
- Singh, B.; Gupta, M. Surface treatment of red mud and its influence on the properties of particulate-filled polyester composite. Bull. Mater. Sci. 1995, 18, 603–621. [Google Scholar] [CrossRef]
- Hertel, T.; Cardenia, C.; Balomenos, E.; Panias, D.; Pontikes, Y. Microwave Treatment of Bauxite Residue for the Production of Inorganic Polymers. In Proceedings of the 2nd International Bauxite Residue Valorisation and Best Practices Conference, Athens, Greece, 7–10 May 2018. [Google Scholar]
- Qin, S.; Wu, B. Effect of self-glazing on reducing the radioactivity levels of red mud based ceramic materials. J. Hazard. Mater. 2011, 198, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, D.; Hou, J.; He, B.; Xiao, B. Preparation of glass-ceramics from red mud in the aluminium industries. Ceram. Int. 2008, 34, 125–130. [Google Scholar] [CrossRef]
- Dry, C.; Meier, J.; Bukowski, J. Sintered coal ash/flux materials for building materials. Mater. Struct. 2004, 37, 114–121. [Google Scholar] [CrossRef]
- Promentilla, M.A.B.; Thang, N.H.; Kien, P.T.; Hinode, H.; Bacani, F.T.; Gallardo, S.M. Optimizing Ternary-blended Geopolymers with Multi-response Surface Analysis. Waste Biomass Valor. 2016, 7, 929–939. [Google Scholar] [CrossRef]
- Hertel, T.; Pontikes, Y. Geopolymers, inorganic polymers, alkali-activated materials and hybrid binders from bauxite residue (red mud)—Putting things in perspective. J. Clean. Prod. 2020, 258, 120610. [Google Scholar] [CrossRef]
- Li, G.H.; Liu, M.X.; Rao, M.J.; Jiang, T.; Zhuang, J.Q.; Zhang, Y.B. Stepwise extraction of valuable components from red mud based on reductive roasting with sodium salts. J. Hazard. Mater. 2014, 280, 774–780. [Google Scholar] [CrossRef]
- Yang, J.K.; Chen, F.; Xiao, B. Engineering application of basic level materials of red mud high level pavement. China Munic. Eng. 2006, 5, 7–9. [Google Scholar] [CrossRef]
- Sushil, S.; Batra, V.S. Catalytic applications of red mud, an aluminium industry waste: A review. Appl. Catal. B Environ. 2008, 81, 64–77. [Google Scholar] [CrossRef]
- Ordonez, S. Comments on catalytic applications of red mud, an aluminium industry waste: A review. Appl. Catal. B Environ. 2008, 84, 732–733. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Vilar, V.J.P.; Botelho, C.M.S.; Boaventura, R.A.R. A review of the use of red mud as adsorbent for the removal of toxic pollutants from water and wastewater. Environ. Technol. 2011, 32, 231–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ciccu, R.; Ghiani, M.; Serci, A.; Fadda, S.; Peretti, R.; Zucca, A. Heavy metal immobilization in the mining-contaminated soils using various industrial wastes. Miner. Eng. 2003, 16, 187–192. [Google Scholar] [CrossRef]
- Chen, Y.; Li, J.-Q.; Huang, F.; Zhou, J.; Zhou, D.-F.; Liu, W. The performance research on absorbing SO2 waste gas with Bayer red mud. J. Guizhou Teachers Univ. Nat. Sci. 2007, 36, 30–32. [Google Scholar]
- Brunori, C.; Cremisini, C.; Massanisso, P.; Pinto, V.; Torricelli, L. Reuse of a treated red mud bauxite waste: Studies on environmental compatibility. J. Hazard. Mater. 2005, 117, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Z.; Yao, Z.Y.; Wang, K.; Wang, F.; Jiang, H.G.; Liang, M.; Wei, J.C.; Airey, G. Sustainable utilization of bauxite residue (Red Mud) as a road material in pavements: A critical review. Constr. Build Mater. 2021, 270, 121419. [Google Scholar] [CrossRef]
- Novais, R.M.; Carvalheiras, J.; Seabra, M.P.; Pullar, R.C.; Labrincha, J.A. Innovative application for bauxite residue: Red mud-based inorganic polymer spheres as pH regulators. J. Hazard. Mater. 2018, 358, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Ujaczki, E.; Feigl, V.; Moln´ar, M.; Cusack, P.; Curtin, T.; Courtney, R.; O’Donoghue, L.; Davris, P.; Hugi, C.; Evangelou, M.; et al. Re-using bauxite residues: Benefits beyond (critical raw) material recovery. J. Chem. Technol. Biotechnol. 2018, 93, 2498–2510. [Google Scholar] [CrossRef] [Green Version]
- Grafe, M.; Power, G.; Klauber, C. Bauxite residue issues: III. Alkalinity and associated chemistry. Hydrometallurgy 2011, 108, 60–79. [Google Scholar] [CrossRef]
- Narayanan, R.; Kazantzis, P.; Nikolaos, K.; Emmert, M.H. Selective process steps for the recovery of scandium from Jamaican bauxite residue (red mud). ACS Sustain. Chem. Eng. 2018, 6, 1478–1488. [Google Scholar] [CrossRef]
- Guo, D.; Yang, G.; Chen, J.; Fan, Y.; Zhan, H.; Liang, H.; Li, B.; Man, L.; Zhang, J.; Rong, J.; et al. Method for Comprehensively Recovering Scandium and Titanium by Leaching Red Mud with Titanium White Waste Acid. CN103131854B, 28 August 2014. [Google Scholar]
- Ochsenkühn-Petropulu, M.; Lyberopulu, T.; Parissakis, G. Direct determination of landthanides, yttrium and scandium in bauxites and red mud from alumina production. Anal. Chim. Acta 1994, 296, 305–313. [Google Scholar] [CrossRef]
- Agrawal, S.; Dhawan, N. Evaluation of red mud as a polymetallic source—A review. Miner. Eng. 2021, 171, 107084. [Google Scholar]
- Vachon, P.; Tyagi, R.D.; Auclair, J.C.; Wilkinson, K.J. Chemical and biological leaching of aluminium from red mud. Environ. Sci. Technol. 1994, 28, 26–30. [Google Scholar] [CrossRef]
- Bruckard, W.J.; Calle, C.M.; Davidson, R.H.; Glenn, A.M.; Jahanshahi, S.; Somerville, M.A.; Sparrow, G.J.; Zhang, L. Smelting of bauxite residue to form a soluble sodium aluminium silicate phase to recover alumina and soda. Miner. Process. Extract. Metall. 2010, 119, 18–26. [Google Scholar]
- Ablamoff, B.; Qian-de, C. Physical and Chemical Principles of Comprehensive Treatment of Aluminium-containing Raw Materials by Basic Process; Central South University of Technology Press: Changsha, China, 1988; pp. 178–182. [Google Scholar]
- Kannan, P.; Banat, F.; Hasan, S.W.; Haija, M.A. Neutralization of Bayer bauxite residue (red mud) by various brines: A review of chemistry and engineering processes. Hydrometallurgy 2021, 206, 105758. [Google Scholar] [CrossRef]
- Alp, A.; Selim Goral, M. The effects of the additives, calcination and leach conditions for alumina production from red mud. Scand. J. Metall. 2003, 32, 301–305. [Google Scholar] [CrossRef]
- Meher, S.N.; Rout, A.K.; Padhi, B.K. Recovery of Al and Na values from red mud by BaO-Na2CO3 sinter process. Eur. J. Chem. 2011, 8, 1387–1393. [Google Scholar]
- Agatzini-Leonardou, S.; Oustadakis, P.; Tsakiridis, P.E.; Markopoulos, C. Titanium leaching from red mud by diluted sulfuric acid at atmospheric pressure. J. Hazard. Mater. 2008, 157, 579–586. [Google Scholar] [CrossRef]
- Erçag, E.; Apak, R. Furnace smelting and extractive metallurgy of red mud: Recovery of TiO2, Al2O3 and pig iron. J. Chem. Technol. Biotechnol. 1997, 70, 241–246. [Google Scholar] [CrossRef]
- Huang, Y.; Chai, W.; Han, G.; Wang, W.; Yang, S.; Liu, J. A perspective of stepwise utilisation of Bayer red mud: Step two—extracting and recovering Ti from Ti enriched tailing with acid leaching and precipitate flotation. J. Hazard. Mater. 2016, 307, 318–327. [Google Scholar] [CrossRef]
- Kasliwal, P.; Sai, P.S.T. Enrichment of titanium dioxide in red mud: A kinetic study. Hydrometallurgy 1999, 53, 73–87. [Google Scholar] [CrossRef]
- Deep, A.; Malik, P.; Gupta, B. Extraction and separation of Ti(IV) using thiophosphinic acids and its recovery from ilmenite and red mud. Sep. Sci. Technol. 2001, 36, 671–685. [Google Scholar] [CrossRef]
- Smirnov, D.I.; Molchanova, T.V. The investigation of sulphuric acid sorption recovery of scandium and uranium from the red mud of alumina production. Hydrometallurgy 1997, 45, 249–259. [Google Scholar] [CrossRef]
- Davris, P.; Balomenos, E.; Panias, D.; Paspaliaris, I. Selective leaching of rare earth elements from bauxite residue (red mud), using a functionalized hydrophobic ionic liquid. Hydrometallurgy 2016, 164, 125–135. [Google Scholar] [CrossRef]
- Lymperopoulou, T.; Georgiou, P.; Tsakanika, L.A.; Hatzilyberis, K.; Ochsenkuehn- Petropoulou, M. Optimizing conditions for scandium extraction from bauxite residue using taguchi methodology. Minerals 2019, 9, 236. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.; Li, W.; Xing, B.; Zhang, Y. Extraction of scandium from red mud by acid leaching with CaF2 and solvent extraction with P507. J. Rare Earths. 2019, 38, 1003–1008. [Google Scholar] [CrossRef]
- Alkan, G.; Yagmurlu, B.; Cakmakoglu, S.; Hertel, T.; Kaya, S.; Gronen, L.; Stopic, S.; Friedrich, B. Novel Approach for Enhanced Scandium and Titanium Leaching Efficiency from Bauxite Residue with Suppressed Silica Gel Formation. Sci. Rep. 2018, 8, 5676. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Li, W.; Li, Z.; Guo, T.; Wen, H.; Wang, N. Leaching Behaviour of Lithium from Bauxite Residue Using Acetic Acid. Min. Metall. Explor. 2020, 37, 443–451. [Google Scholar]
- Panda, S.; Costa, R.B.; Shah, S.S.; Mishra, S.; Bevilaqua, D.; Ata Akcil, A. Biotechnological trends and market impact on the recovery of rare earth elements from bauxite residue (red mud)—A review. Resour. Conserv. Recycl. 2021, 171, 105645. [Google Scholar] [CrossRef]
- Ochsenkuhn-Petropulu, M.; Lyberopulu, T.; Ochsenkuhn, K.M.; Parissakis, G. Recovery of lanthanides and yttrium from red mud by selective leaching. Anal. Chim. Acta 1996, 319, 249–254. [Google Scholar] [CrossRef]
- Ujaczki, É.; Zimmermann, Y.S.; Gasser, C.A.; Molnár, M.; Feigl, V.; Lenz, M. Red mud as secondary source for critical raw materials–extraction study. J. Chem. Technol. Biotechnol. 2017, 92, 2835–2844. [Google Scholar] [CrossRef]
- Shoppert, A.; Loginova, I.; Napol’skikh, J.; Kyrchikov, A.; Chaikin, L.; Rogozhnikov, D.; Valeev, D. Selective Scandium (Sc) Extraction from Bauxite Residue (RedMud Obtained by Alkali Fusion-Leaching Method. Materials 2022, 15, 433. [Google Scholar] [CrossRef]
- Agrawal, S.; Dhawan, N. Investigation of carbothermic microwave reduction followed by acid leaching for recovery of iron and aluminium values from Indian red mud. Miner. Eng. 2020, 159, 106653. [Google Scholar] [CrossRef]
- Chiara, B.; Chiara, C.; Tam, P.; WAI, T.; Dimitrios, P. Review of Technologies in the Recovery of Iron, Aluminium, Titanium and Rare Earth Elements from Bauxite Residue (Red Mud). In Proceedings of the 3rd International Symposium on Enhanced Landfill Mining, Lisbon, Portugal, 8–10 February 2016; pp. 259–276. [Google Scholar]
- Stickney, W.A.; Butler, M.O.; Mauser, J.E.; Fursman, O.C. Utilization of Red Mud Residues from Alumina Production; US Department of Interior, Bureau of Mines: Washington, DC, USA, 1970. [Google Scholar]
- Li, Y.; Wang, J.; Wang, X.; Wang, B.; Luan, Z. Feasibility study of iron mineral separation from red mud by high gradient superconducting magnetic separation. Phys. C Supercond. 2011, 471, 91–96. [Google Scholar] [CrossRef]
- Rai, S.; Nimje, M.T.; Chaddha, M.J.; Modak, S.; Rao, K.R.; Agnihotri, A. Recovery of iron from bauxite residue using advanced separation techniques. Miner. Eng. 2019, 2134, 222–231. [Google Scholar] [CrossRef]
- Borra, C.R.; Blanpain, B.; Pontikes, Y.; Binnemans, K.; Van Gerven, T. Smelting of bauxite residue (red mud) in view of iron and selective rare earths recovery. J. Sustain. Metall. 2016, 22, 28–37. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, S.; Dhawan, N. Microwave carbothermic reduction of low-grade iron ore. Metall. Mater. Trans. B 2020, 51, 1576–1586. [Google Scholar] [CrossRef]
- Shuai, Y.; Xiao, L.; Peng, G.; Yuexin, H. A semi-industrial experiment of suspension magnetization roasting technology for separation of iron minerals from red mud. J. Hazard. Mater. 2020, 394, 122579. [Google Scholar]
- Cardenia, C.; Balomenos, E.; Panias, D. Iron Recovery from Bauxite Residue through Reductive Roastingand Wet Magnetic Separation. J. Sustain. Metall. 2019, 5, 9–19. [Google Scholar] [CrossRef] [Green Version]
- Wanchao, L.; Jiakuan, Y.; Xiao, B. Application of Bayer red mud for iron recovery and building material production from alumosilicate residues. J. Hazard. Mater. 2009, 161, 474–478. [Google Scholar]
- Fanghai, L.; Xiangdong, S.; Fang, H.; Jiawei, W.; Haifeng, W. Co-Treatment of spent pot-lining and red mud for carbon reutilization and recovery of iron, aluminium and sodium by reductive roasting process. Metall. Mater. Trans. B 2020, 51, 1564–1575. [Google Scholar]
- Agrawal, S.; Rayapudi, V.; Dhawan, N. Microwave reduction of red mud for recovery of iron values. J. Sustain. Metall. 2018, 4, 427–436. [Google Scholar] [CrossRef]
- Agrawal, S.; Rayapudi, V.; Dhawan, N. Comparison of microwave and conventional carbothermal reduction of red mud for recovery of iron values. Miner. Eng. 2019, 132, 202–210. [Google Scholar] [CrossRef]
- Javad, S.; Barani, K. Microwave Heating Applications in Mineral Processing. In The Development and Application of Microwave Heating; IntechOpen: Rijeka, Croatia, 2012; pp. 79–104. [Google Scholar]
- Raspopov, N.A.; Korneev, V.P.; Averin, V.V.; Lainer, Y.A.; Zinoveev, D.V.; Dyubanov, V.G. Reduction of iron oxides during the pyrometallurgical processing of red mud. Russ. Metall. Met. 2013, 1, 33–37. [Google Scholar] [CrossRef]
- Jayasankar, K.; Ray, P.K.; Chaubey, A.K.; Padhi, A.; Satapathy, B.K.; Mukherjee, P.S. Production of pig iron from red mud waste fines using thermal plasma technology. Int. J. Miner. Metall. Mater. 2012, 19, 679–684. [Google Scholar] [CrossRef]
- Zhu, D.Q.; Chun, T.J.; Pan, J.; He, Z. Recovery of iron from high-iron red mud by reduction roasting with adding sodium salt. Iron Steel Res. Int. 2012, 19, 1–5. [Google Scholar] [CrossRef]
- Valeev, D.; Zinoveev, D.; Kondratiev, A.; Lubyanoi, D.; Pankratov, D. Reductive smelting of neutralized red mud for iron recovery and produced pig iron for heat resistant castings. Metals 2020, 10, 32. [Google Scholar] [CrossRef] [Green Version]
- Kaußen, F.; Friedrich, B. Reductive smelting of red mud for iron recovery. Chem. Ing. Tech. 2015, 87, 1535–1542. [Google Scholar] [CrossRef]
- Kongkarat, S.; Khanna, R.; Koshy, O.; O’Kane, P.; Sahajwalla, V. Use of waste Bakelite as a raw material resource for re-carburization in steelmaking processes. Steel Res. Int. 2011, 82, 1228–1239. [Google Scholar] [CrossRef]
- Shuai, L.; Zeshuang, K.; Wanchao, L.; Yicheng, L.; Hongshan, Y. Reduction behaviour and direct reduction kinetics of red mud-biomass composite pellets. J. Sustain. Metall. 2021, 7, 126–135. [Google Scholar]
- Rahman, M.; Khanna, R.; Sahajwalla, V.; O’Kane, P. The influence of ash impurities on interfacial reactions between carbonaceous materials and EAF slag at 1550 °C. ISIJ 2009, 49, 329–336. [Google Scholar] [CrossRef] [Green Version]
- Mombelli, D.; Barella, S.; Gruttadauria, A.; Mapelli, C. Iron Recovery from Bauxite Tailings Red Mud by Thermal Reduction with Blast Furnace Sludge. Appl. Sci. 2019, 9, 4902. [Google Scholar] [CrossRef] [Green Version]
- Balomnenos, E.; Kastritis, D.; Panias, D.; Paspaliaris, I.; Boufounos, D. The Enexal Bauxite Residue Treatment Process: Industrial Scale Pilot Plant Results. In Light Met; Springer: Cham, Switzerland, 2014; pp. 141–147. [Google Scholar]
- Jin, J.; Liu, X.; Yuan, S.; Gao, P.; Li, Y.; Zhang, H.; Meng, X. Innovative utilization of red mud through co-roasting with coal gangue for separation of iron and aluminium minerals. J. Ind. Eng. Chem. 2021, 98, 298–307. [Google Scholar] [CrossRef]
- Cao, Y.; Sun, Y.; Gao, P.; Han, Y.; Li, Y. Mechanism for suspension magnetization roasting of iron ore using straw-type biomass reductant. Int. J. Min. Sci. Technol. 2021, 31, 1075–1081. [Google Scholar] [CrossRef]
- Gostu, S.; Mishra, B.; Martins, G.P. Low Temperature Reduction of Hematite in Red Mud to Magnetite. In Light Met; Springer: Cham, Switzerland, 2017; pp. 67–73. [Google Scholar]
- Liu, X.; Gao, P.; Yuan, S.; Yang, L.; Han, Y. Clean utilization of high-iron red mud by suspension magnetization roasting. Miner. Eng. 2020, 157, 106553. [Google Scholar] [CrossRef]
- Yu, Z.; Shi, Z.; Chen, Y.; Niu, Y.; Wang, Y.; Wan, P. Red-mud treatment using oxalic acid by UV irradiation assistance. Trans. Nonferrous Metal. Soc. 2012, 222, 456–460. [Google Scholar] [CrossRef]
- Debadatta, D.; Pramanik, K. A study on chemical leaching of iron from red mud using sulphuric acid. Res. J. Chem. Environ. 2013, 17, 50–56. [Google Scholar]
- Pepper, R.A.; Couperthwaite, S.J.; Millar, G.J. Comprehensive examination of acid leaching behaviour of mineral phases from red mud: Recovery of Fe, Al, Ti, and Si. Miner. Eng. 2016, 99, 8–18. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Xuewen, W.; Mingyu, W.; Huaguang, W.; Pengfei, X. Iron recovery from the leached solution of red mud through the application of oxalic acid. Int. J. Miner. Process. 2016, 157, 145–151. [Google Scholar] [CrossRef]
- Eisele, T.C.; Gabby, K.L. Review of reductive leaching of iron by anaerobic bacteria. Miner. Process. Extractive Metall. Rev. 2014, 35, 75–105. [Google Scholar] [CrossRef]
- Laguna, C.; Gonzalez, F.; Garcia-Balboa, C.; Ballester, A.; Blazquez, M.L.; Munoz, J.A. Bio-reduction of iron compounds as a possible clean environmental alternative for metal recovery. Miner. Eng. 2011, 24, 10–18. [Google Scholar] [CrossRef]
- Papassiopi, N.; Vaxevanidou, K.; Paspaliaris, I. Effectiveness of iron reducing bacteria for the removal of iron from bauxite ores. Miner. Eng. 2010, 23, 25–31. [Google Scholar] [CrossRef]
- Vakilchap, F.; Mousavi, S.M.; Shojaosadati, S.A. Role of Aspergillus niger in recovery enhancement of valuable metals from produced red mud in Bayer process. Bioresour. Technol. 2016, 218, 991–998. [Google Scholar] [CrossRef]
- Qu, Y.; Lian, B. Bioleaching of rare earth and radioactive elements from red mud using Penicillium tricolor RM-10. Bioresour. Technol. 2013, 136, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Li, H.; Tian, W.; Wang, X.; Wang, X.; Jia, X.; Shi, B.; Song, G.; Tang, Y. Leaching of valuable metals from red mud via batch and continuous processes by using fungi. Miner. Eng. 2015, 81, 1–4. [Google Scholar] [CrossRef]
- Pasechnik, L.A.; Skachkov, V.M.; Chufarov, A.Y.; Suntsov, A.Y.; Yatsenko, S.P. High purity scandium extraction from red mud by novel simple technology. Hydrometallurgy 2021, 202, 105597. [Google Scholar] [CrossRef]
- Khanna, R.; Ellamparuthy, G.; Cayumil, R.; Mishra, S.K.; Mukherjee, P.S. Concentration of Rare Earth Elements during High Temperature Pyrolysis of Waste Printed Circuit Boards. Waste Manag. 2018, 78, 602–610. [Google Scholar] [CrossRef]
- Li, S.; Pan, J.; Zhu, D.; Guo, Z.; Shi, Y.; Dong, T.; Lu, S.; Tian, H. A new route for separation and recovery of Fe, Al and Ti from red mud. Resour. Conserv. Recycl. 2021, 168, 105314. [Google Scholar] [CrossRef]
- Chun, T.; Li, D.; Di, Z.; Long, H.; Tang, L.; Li, F.; Li, Y. Recovery of iron from red mud by high temperature reduction of carbon bearing briquettes. J. South. Afr. Inst. Min. Metall. 2017, 117, 361–364. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Gao, Q.; Zhao, J.; Li, M.; Qi, Y. Semi-smelting reduction and magnetic separation for the recovery of iron and alumina slag from iron rich bauxite. Minerals 2019, 9, 223. [Google Scholar] [CrossRef] [Green Version]
- Guo, T.; Gao, J.; Xu, H.; Zhao, K.; Shi, X. Nuggets production by direct reduction of high iron red mud. J. Iron Steel Res. Int. 2013, 20, 24–27. [Google Scholar] [CrossRef]
- Samouhos, M.; Taxiarchou, M.; Pilatos, G.; Tsakiridis, P.E.; Devlin, E.; Pissa, M. Controlled reduction of red mud by H2 followed by magnetic separation. Miner. Eng. 2017, 105, 36–43. [Google Scholar] [CrossRef]
- Cardenia, C.; Balomenos, E.; Wai, P.; Tam, Y.; Panias, D. A Combined Soda Sintering and Microwave Reductive Roasting Process of Bauxite Residue for Iron Recovery. Minerals 2021, 11, 222. [Google Scholar] [CrossRef]
- Mombelli, D.; Mapelli, C.; Barella, S.; Gruttadauria, A.; Ragona, M.; Pisu, M.; Viola, A. Characterization of cast iron and slag produced by red muds reduction via Arc Transferred Plasma (ATP) reactor under different smelting conditions. J. Environ. Chem. Eng. 2020, 8, 104293. [Google Scholar] [CrossRef]
- Maihatchi, A.A.; Pons, M.N.; Ricoux, Q.; Goettmann, F.; Lapicque, F. Production of electrolytic iron from red mud in alkaline media. J. Environ. Manage. 2020, 266, 110547. [Google Scholar] [CrossRef] [PubMed]
- Grudinsky, P.; Zinoveev, D.; Pankratov, D.; Semenov, A.; Panova, M.; Kondratiev, A.; Zakunov, A.; Dyubanov, V.; Petelin, A. Influence of Sodium Sulfate Addition on Iron Grain Growth during Carbothermic Roasting of Red Mud Samples with Different Basicity. Metals 2020, 10, 1571. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Misra, V.N.; Clough, J.; Mun, R. Dephosphorization of Western Australian iron ore by hydrometallurgical process. Miner. Eng. 1999, 12, 1083–1092. [Google Scholar] [CrossRef]
- Srivastava, M.P.; Pan, S.K.; Prasad, N.; Mishra, B.K. Characterization and processing of iron ore fines of Kiruburu deposit of India. Int. J. Miner. Process. 2001, 61, 93–107. [Google Scholar] [CrossRef]
- Guider, J.W. Iron ore beneficiation—key to modern steelmaking. Min. Eng. 1981, 33, 410–413. [Google Scholar]
- Dobbins, M.S.; Burnet, G. Production of an iron ore concentrate from the iron-rich fraction of power plant fly ash. Resour. Conserv. Recycl. 1982, 9, 231–242. [Google Scholar] [CrossRef]
- Worldsteel Association. Available online: https://www.worldsteel.org/media-centre/press-releases/2021/Global-crude-steel-output-decreases-by-0.9--in-2020.html (accessed on 2 December 2021).
- Zhang, Z.L.; Li, Q.; Zou, Z.S. Reduction properties of high alumina iron ore cold bonded pellet with CO–H2 mixtures. Ironmak. Steelmak. 2014, 41, 561–567. [Google Scholar] [CrossRef]
- Rath, S.; Sahoo, H.; Dhawan, N.; Rao, D.S.; Das, B.; Mishra, B.K. Optimal Recovery of Iron Values from a Low Grade Iron Ore using Reduction Roasting and Magnetic Separation. Sep. Sci. Technol. 2014, 49, 1927–1936. [Google Scholar] [CrossRef]
- Dhunna, R.; Khanna, R.; Mansuri, I.; Sahajwalla, V. Recycling Waste Bakelite as an Alternative Carbon Resource for Ironmaking Applications. ISIJ 2014, 54, 613–619. [Google Scholar] [CrossRef] [Green Version]
- Kongkarat, S.; Khanna, R.; Koshy, P.; O’kane, P.; Sahajwalla, V. Recycling waste polymers in EAF steelmaking: Influence of polymer composition on carbon/slag interactions. ISIJ 2012, 52, 385–393. [Google Scholar] [CrossRef] [Green Version]
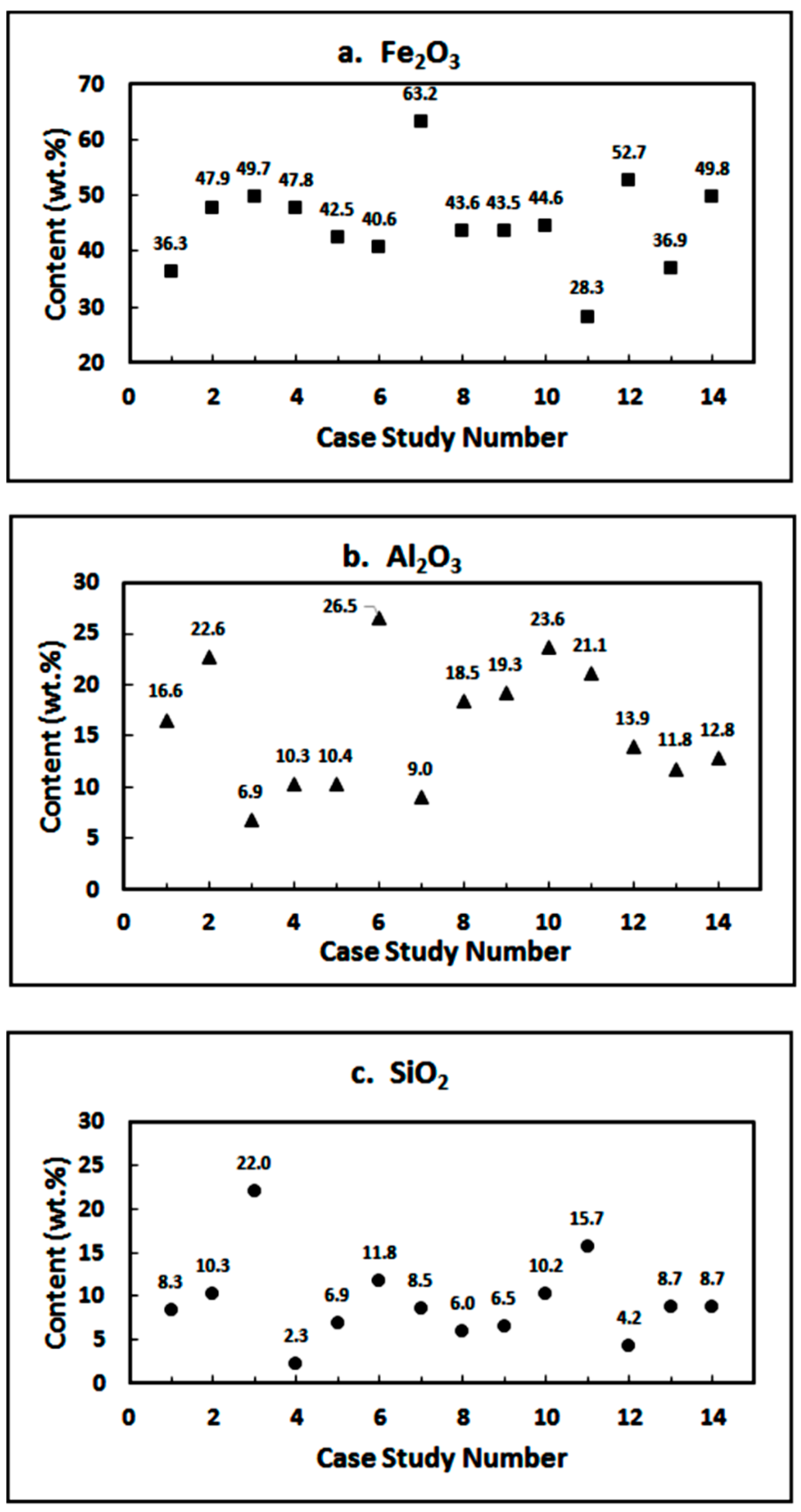
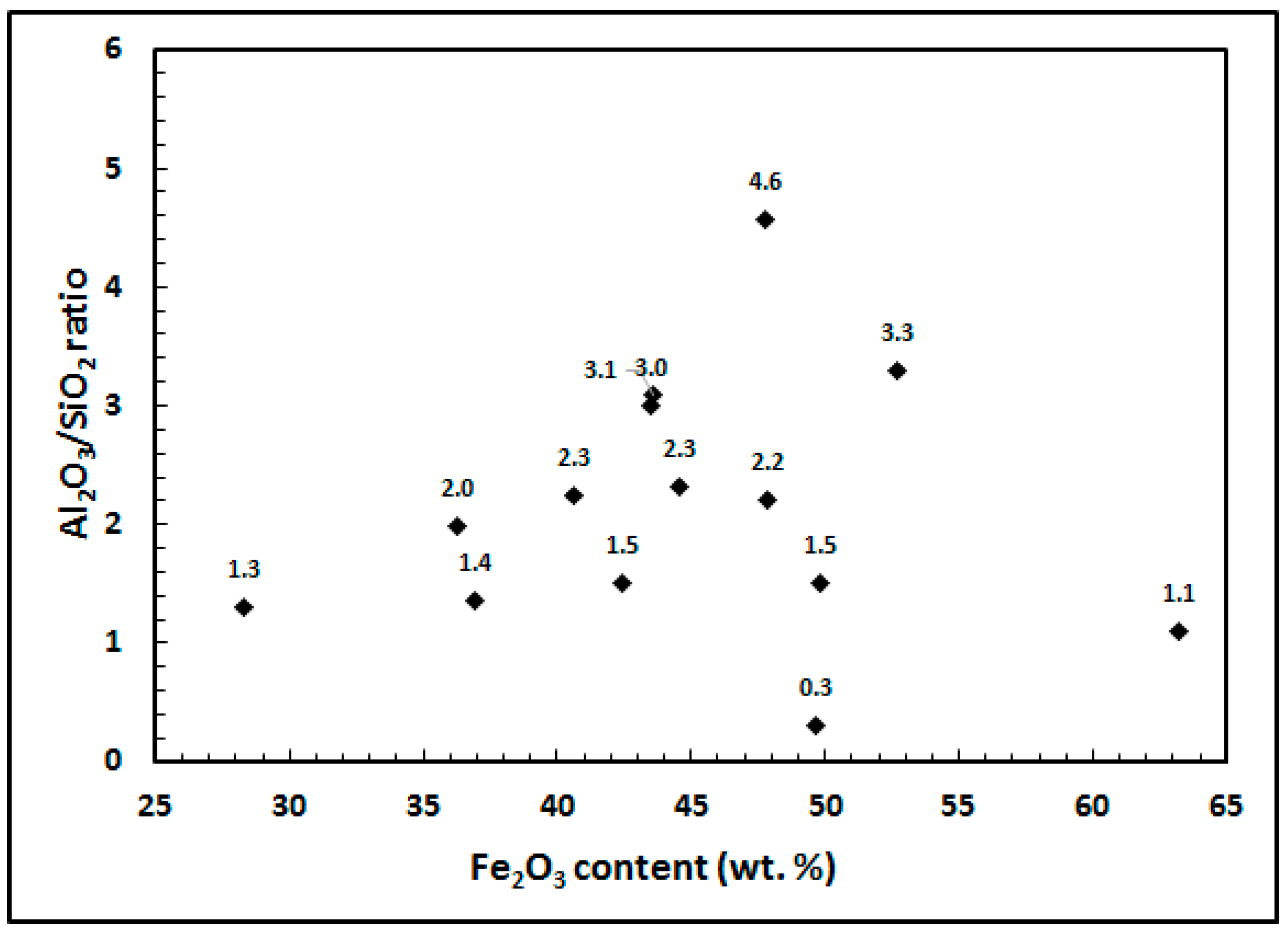
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | Iron Grade |
|---|---|---|---|---|---|---|---|
| wt.% | 36.26 | 16.58 | 8.32 | 17.10 | 1.43 | 6.00 | 25.7% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | Iron Grade |
|---|---|---|---|---|---|---|---|
| wt.% | 47.85 | 22.64 | 10.25 | 3.58 | 1.86 | 10.25 | 33.5% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | LOI |
|---|---|---|---|---|---|---|---|
| wt.% | 49.65 | 6.86 | 22.0 | - | - | 8.29 | 11.39% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | LOI |
|---|---|---|---|---|---|---|---|
| wt.% | 47.76 | 10.26 | 2.25 | 4.22 | 0.26 | 1.13 | 11.93% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O |
|---|---|---|---|---|---|---|
| wt.% | 42.45 | 10.35 | 6.89 | - | 0.72 | 1.36 |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | LOI |
|---|---|---|---|---|---|---|---|
| wt.% | 40.62 | 26.53 | 11.77 | 1.57 | 1.38 | - | 16.42% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O |
|---|---|---|---|---|---|---|
| wt.% | 63.2 | 8.96 | 8.54 | - | 0.551 | 2.5 |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | LOI |
|---|---|---|---|---|---|---|---|
| wt.% | 43.59 | 18.45 | 6.0 | 5.54 | 11.38 | 1.82 | 11.7% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | LOI |
|---|---|---|---|---|---|---|---|
| wt.% | 43.51 | 19.25 | 6.5 | 5.49 | 9.58 | 2.80 | 9.41% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O |
|---|---|---|---|---|---|---|
| wt.% | 44.6 | 23.6 | 10.2 | 5.7 | 11.2 | 2.5 |
| Constituent (wt.%) | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | LOI |
|---|---|---|---|---|---|---|---|
| Fresh RM | 20.89 | 16.3 | 9.8 | 5.2 | 5.54 | 10.39 | 29.78 |
| Calcined RM | 28.26 | 21.05 | 15.69 | 7.26 | 7.69 | 13.44 | 3.78 |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O |
|---|---|---|---|---|---|---|
| wt.% | 52.7 | 13.9 | 4.19 | 7.4 | 4.1 | 2.1 |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O | LOI |
|---|---|---|---|---|---|---|---|
| wt.% | 36.9 | 11.8 | 8.71 | 3.54 | 23.8 | 0.27 | 12.46% |
| Constituent | Fe2O3 | Al2O3 | SiO2 | TiO2 | CaO | Na2O |
|---|---|---|---|---|---|---|
| wt.% | 49.81 | 12.77 | 8.71 | 4.67 | 9.26 | 33 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khanna, R.; Konyukhov, Y.; Zinoveev, D.; Jayasankar, K.; Burmistrov, I.; Kravchenko, M.; Mukherjee, P.S. Red Mud as a Secondary Resource of Low-Grade Iron: A Global Perspective. Sustainability 2022, 14, 1258. https://doi.org/10.3390/su14031258
Khanna R, Konyukhov Y, Zinoveev D, Jayasankar K, Burmistrov I, Kravchenko M, Mukherjee PS. Red Mud as a Secondary Resource of Low-Grade Iron: A Global Perspective. Sustainability. 2022; 14(3):1258. https://doi.org/10.3390/su14031258
Chicago/Turabian StyleKhanna, Rita, Yuri Konyukhov, Dmitry Zinoveev, Kalidoss Jayasankar, Igor Burmistrov, Maksim Kravchenko, and Partha S. Mukherjee. 2022. "Red Mud as a Secondary Resource of Low-Grade Iron: A Global Perspective" Sustainability 14, no. 3: 1258. https://doi.org/10.3390/su14031258
APA StyleKhanna, R., Konyukhov, Y., Zinoveev, D., Jayasankar, K., Burmistrov, I., Kravchenko, M., & Mukherjee, P. S. (2022). Red Mud as a Secondary Resource of Low-Grade Iron: A Global Perspective. Sustainability, 14(3), 1258. https://doi.org/10.3390/su14031258






