Reaction Behavior and Transformation Path of Zinc in the Heating-Up Zone during Sintering Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Selection Basis of Experimental Conditions: Temperature and Atmosphere
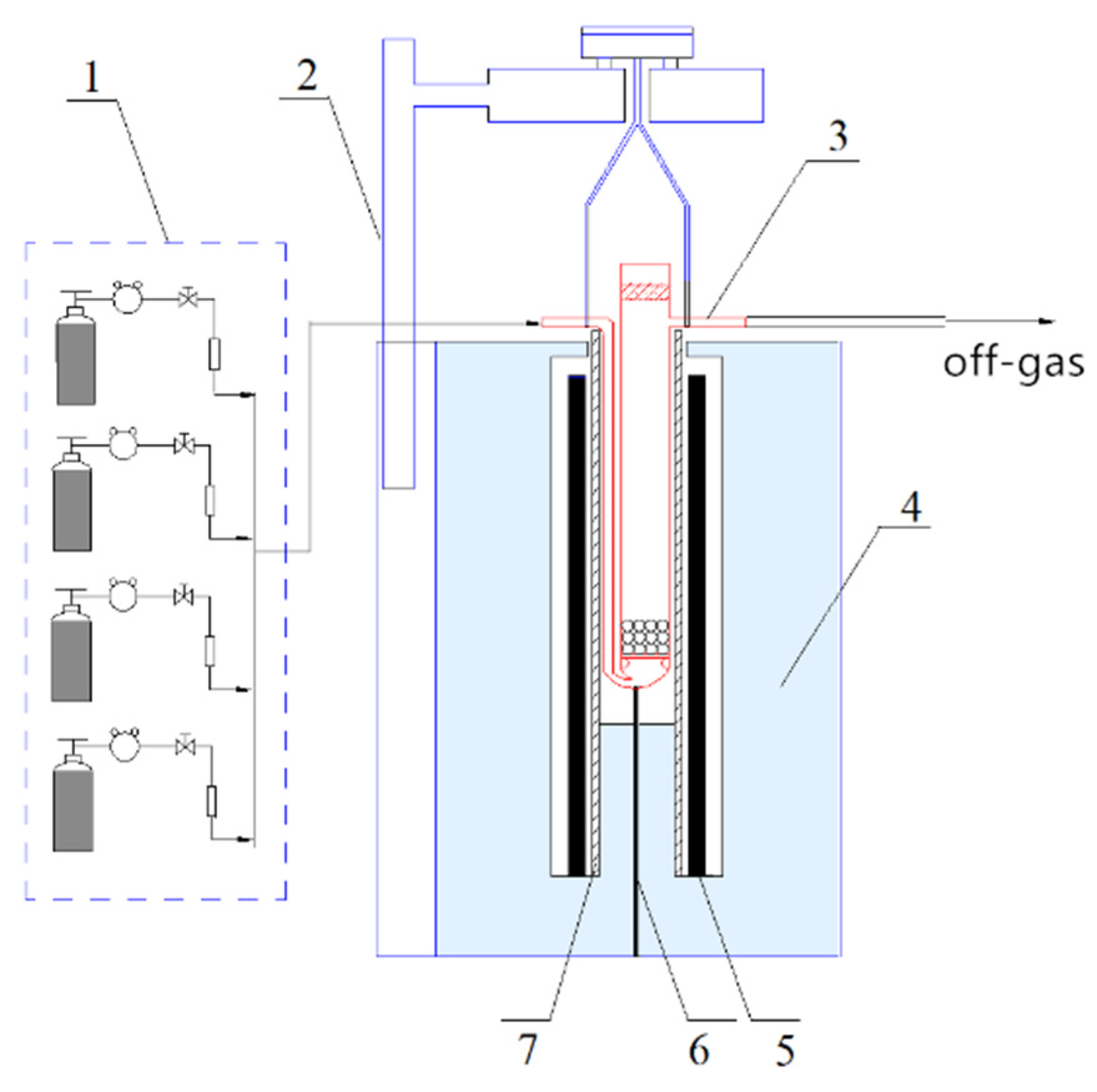
2.3. Experimental Device and Method
3. Experimental Results and Discussion
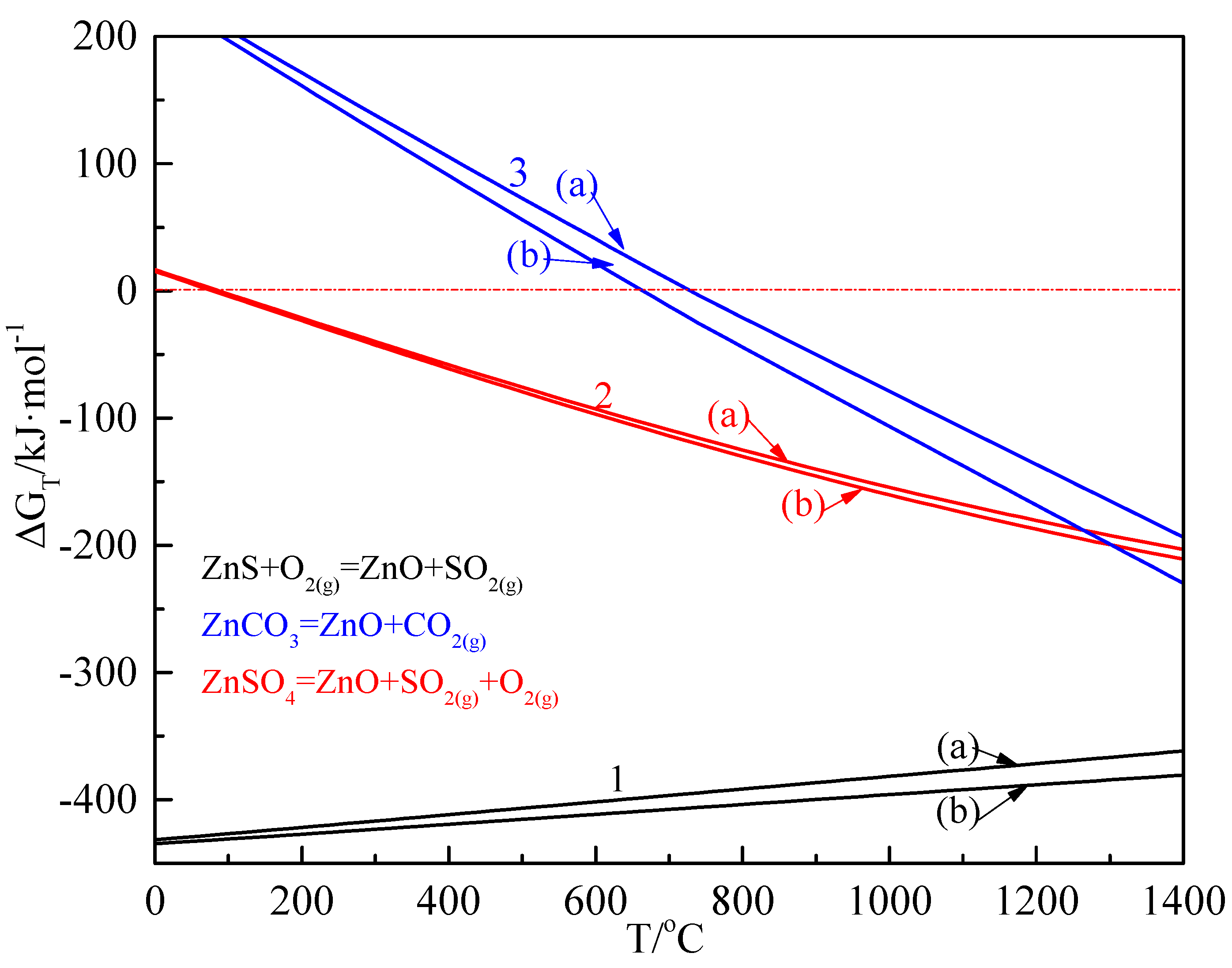
3.1. Reaction Thermodynamics of Zinc-Bearing Minerals during the Dry Pre-Heating Zone
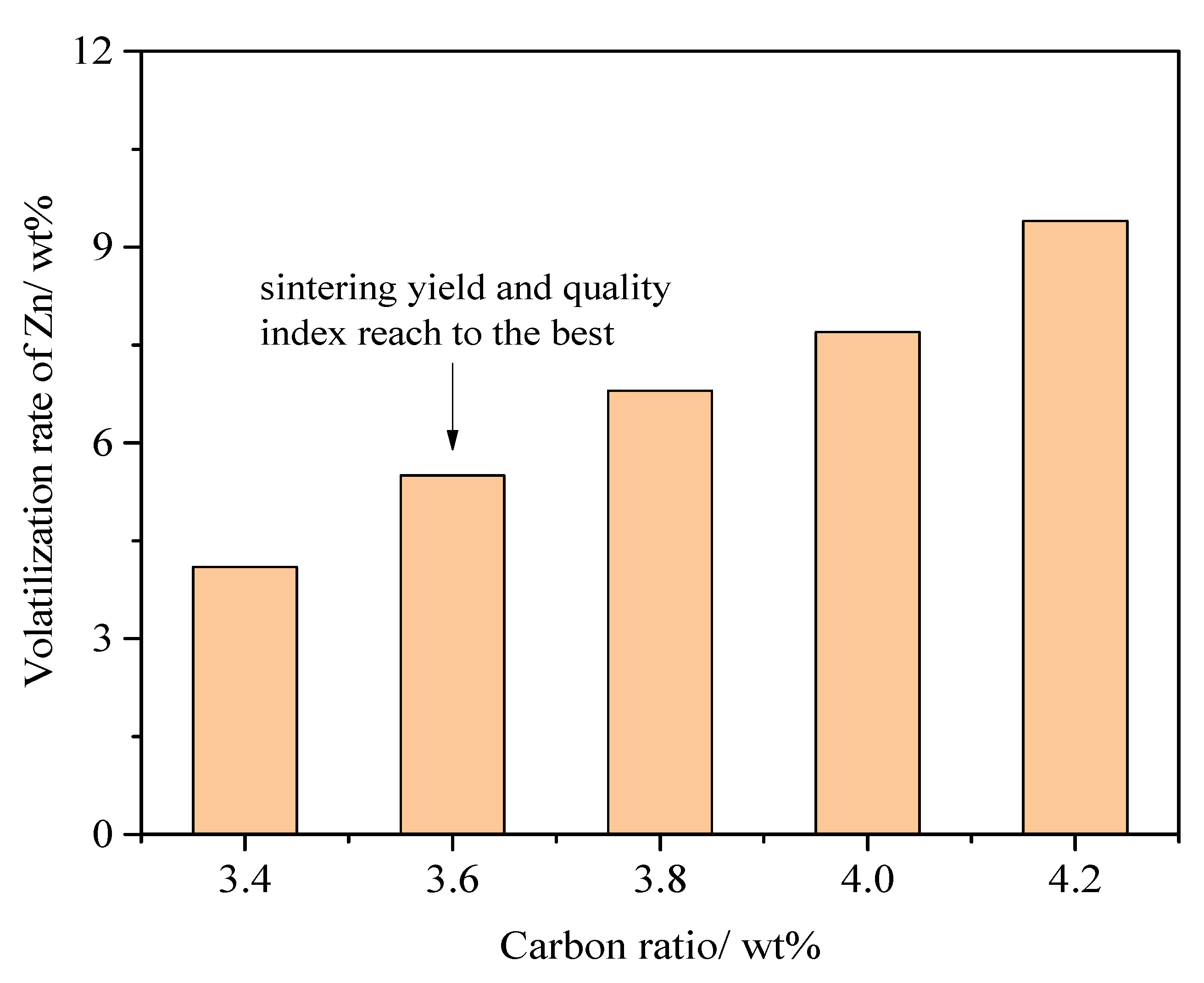
3.2. Conversion Behavior of Zn in the Combustion Zone
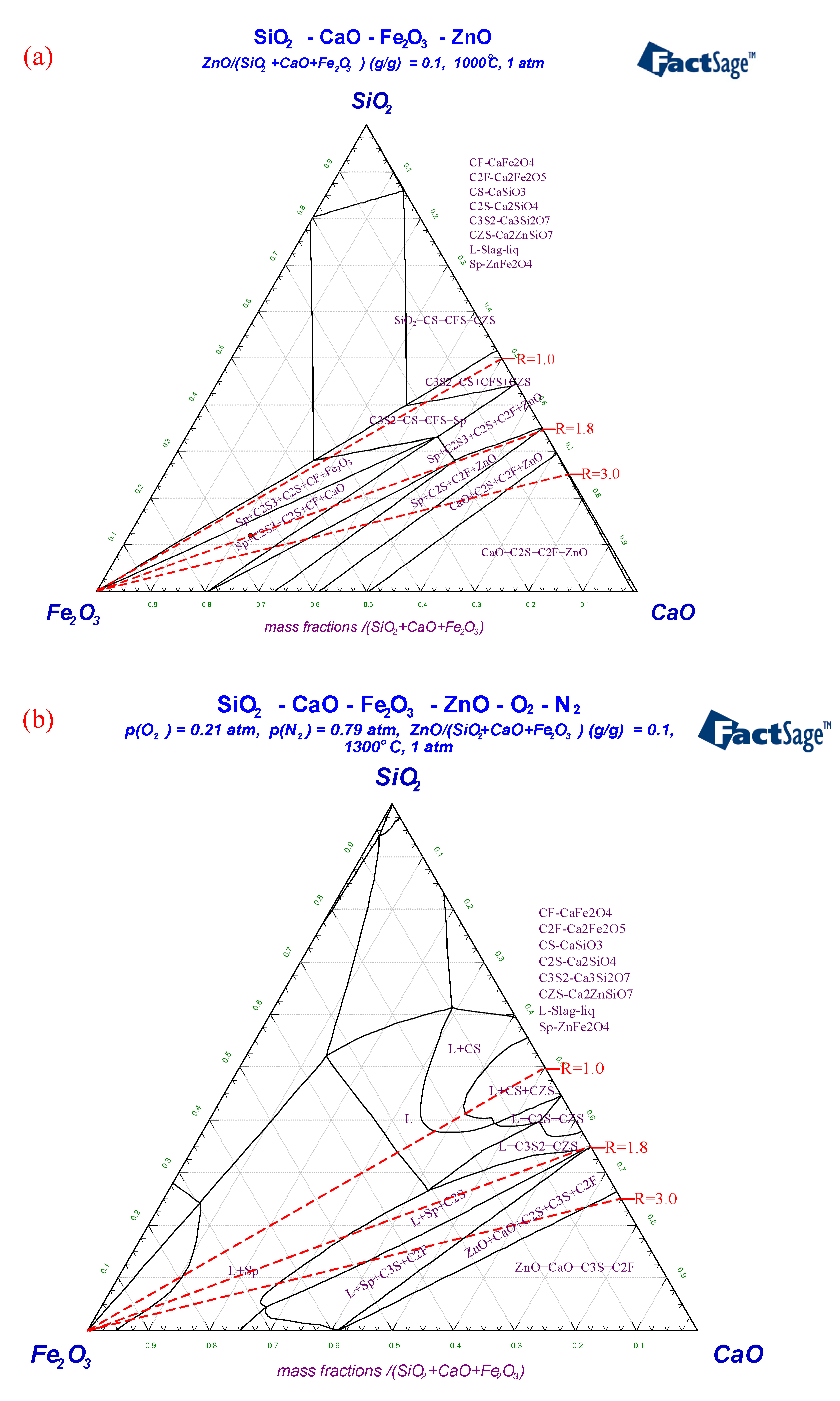
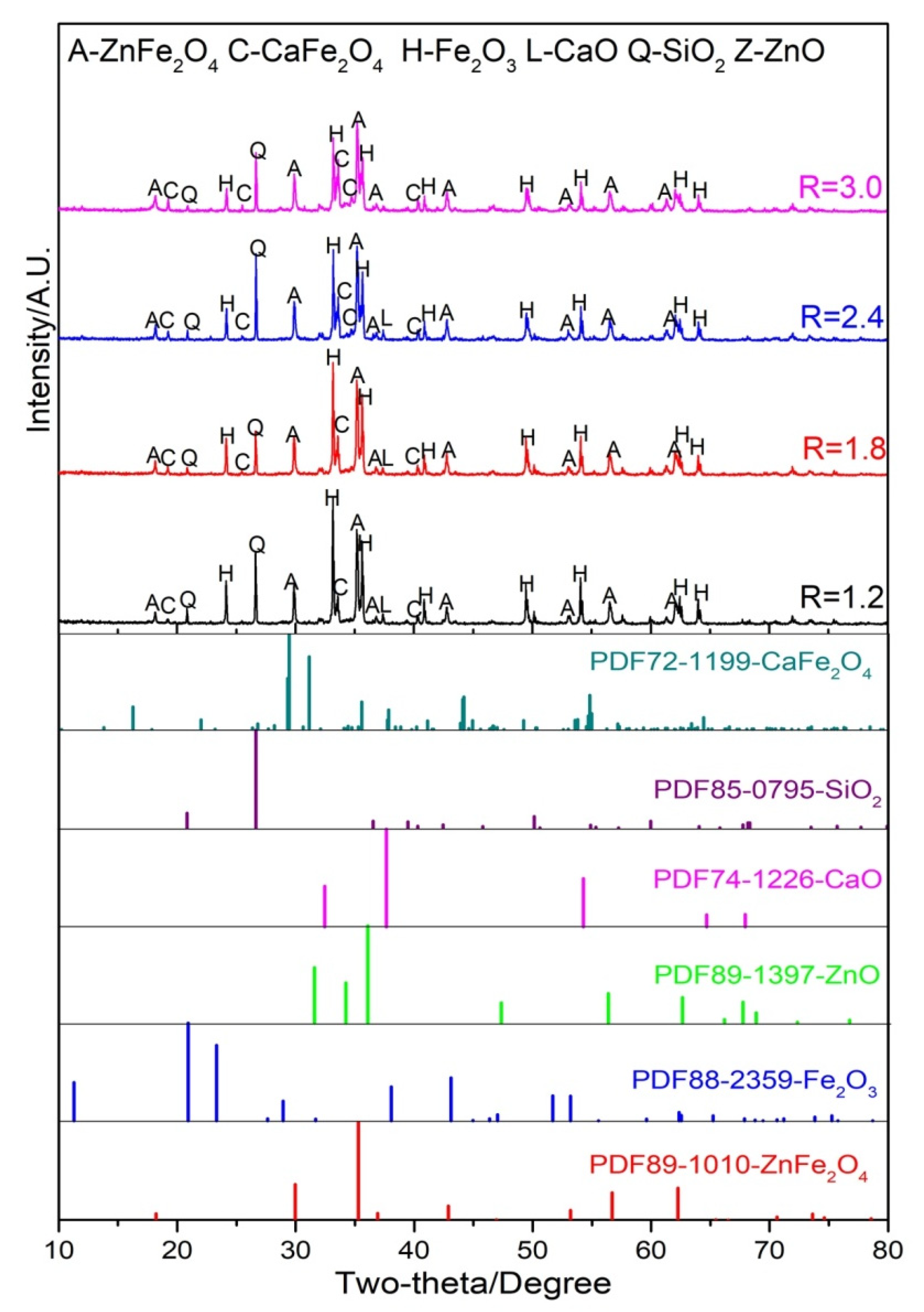
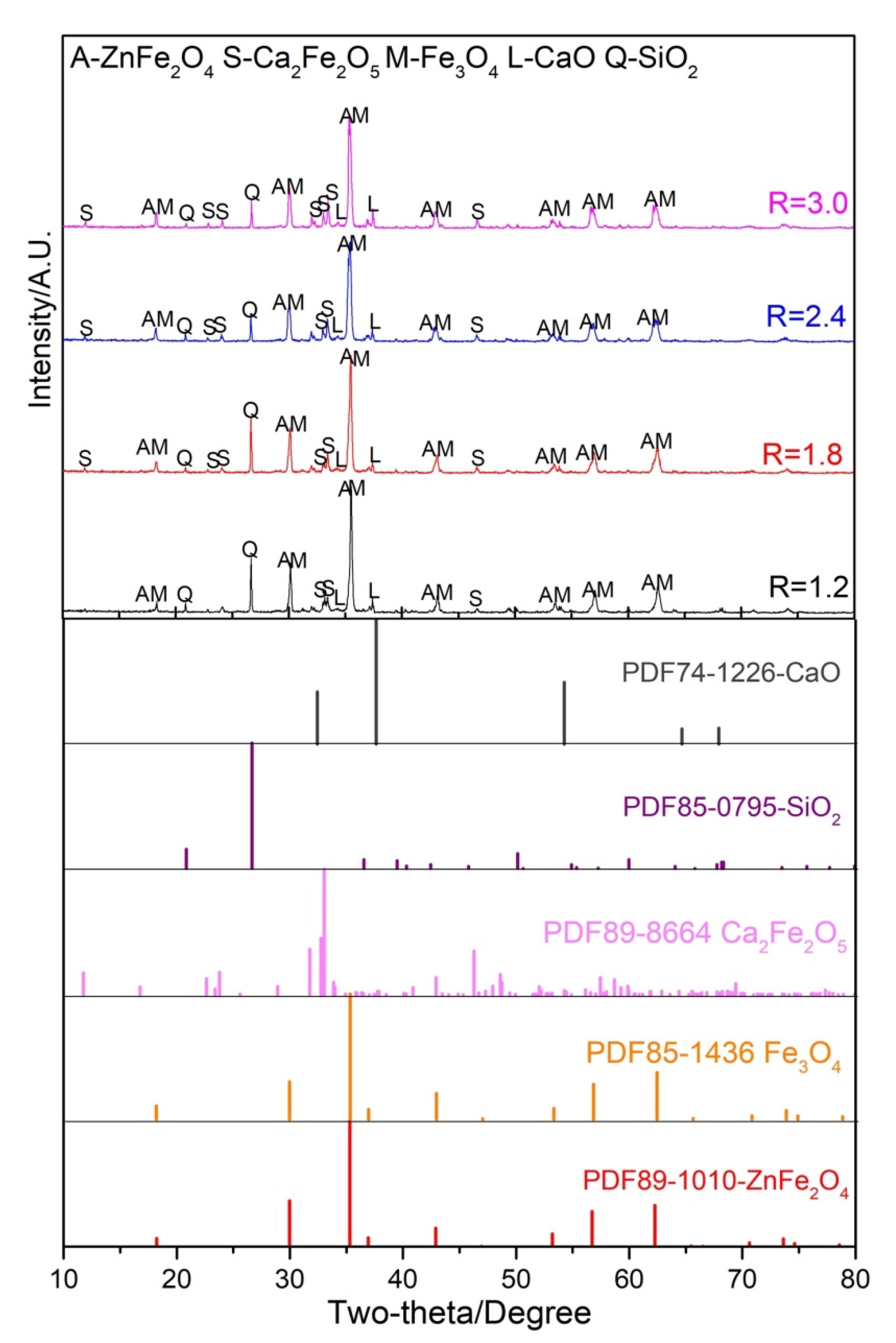
3.3. Reaction Behavior of ZnO with Fe, Ca and Si Oxides in Raw Materials
3.3.1. Influence of Temperature
3.3.2. The Effect of Basicity
3.3.3. Influence of Reducing Atmosphere
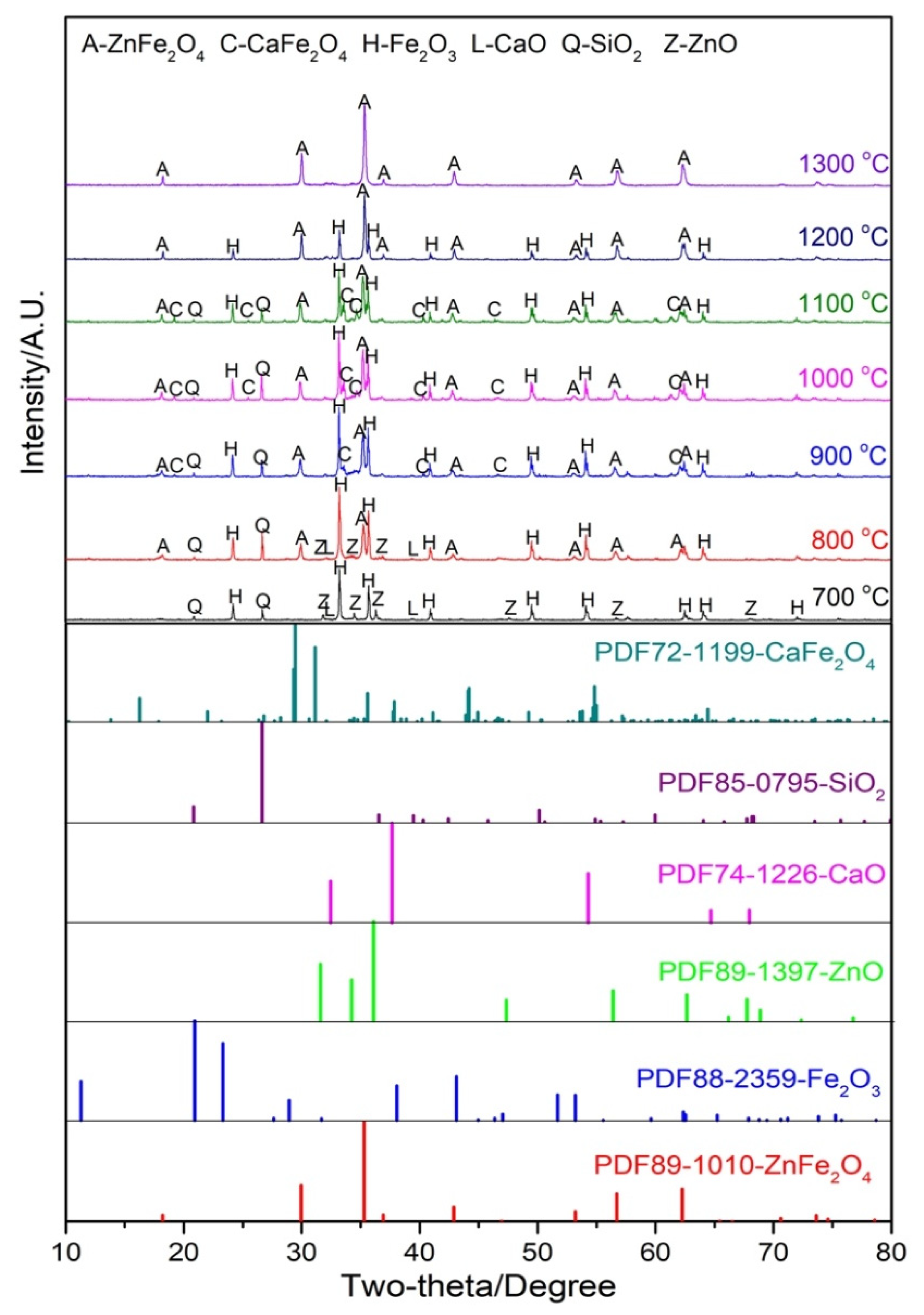
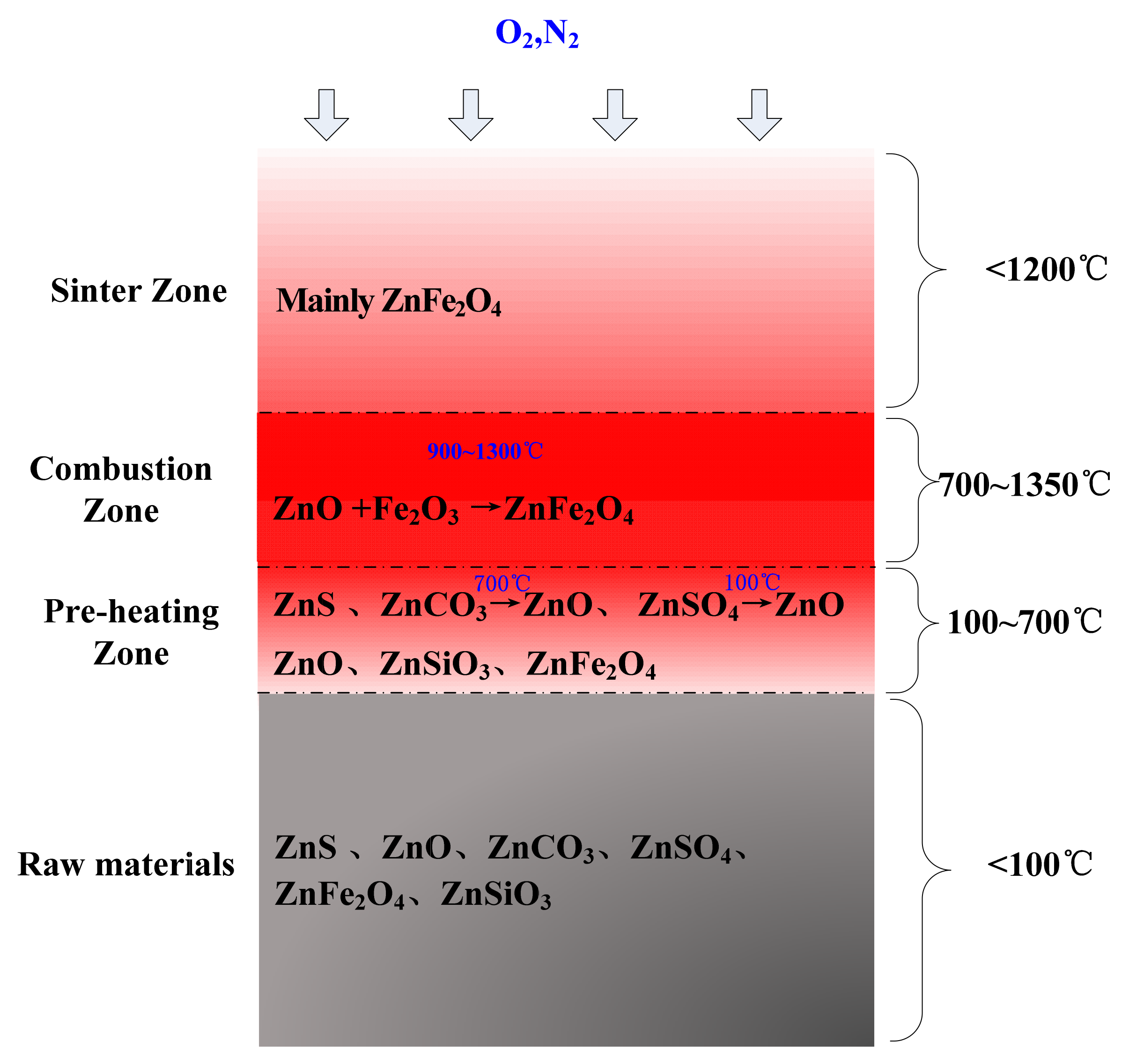
3.4. Phase Transformation and Migration Behavior of Zn during Sintering
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barakat, M.A. The pyrometallurgical processing of galvanizing zinc ash and flue dust. JOM 2003, 55, 26–29. [Google Scholar] [CrossRef]
- Yan, Y.; Zheng, G.S.; Zheng, S.L.; Hu, Z.B. Effects of depressant on recycling pyrite from tailings of lead-zinc ore. Ind. Miner. Process. 2015, 1, 14–16. (In Chinese) [Google Scholar]
- Mehta, N.; Dino, G.; Passarella, I.; Ajmone-Marsan, F.; Rossetti, P.; De Luca, D. Assessment of the Possible Reuse of Extractive Waste Coming from Abandoned Mine Sites: Case Study in Gorno, Italy. Sustainability 2020, 12, 2471. [Google Scholar] [CrossRef]
- Kaya, M.; Hussaini, S.; Kursunoglu, S. Critical review on secondary zinc resources and their recycling technologies. Hydrometallurgy 2020, 195, 105362. [Google Scholar] [CrossRef]
- Han, J.; Liu, W.; Qin, W.; Jiao, F.; Wang, D.; Liang, C. Thermodynamic and kinetic studies for intensifying selective de-composition of zinc ferrite. JOM 2016, 68, 2543–2550. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, Z.J.; Han, B.L.; Jiang, T.; Anderson, C. A value-added multistage utilization process for the gradi-ent-recovery tin, iron and preparing composite phase change materials (C-PCMs) from tailings. Sci. Rep. 2019, 9, 14097. [Google Scholar]
- Lanzerstorfer, C.; Bamberger-Strassmayr, B.; Pilz, K. Recycling of blast furnace dust in the iron ore sintering process: In-vestigation of coke breeze substitution and the influence on off-gas emissions. ISIJ Int. 2015, 55, 758–764. [Google Scholar] [CrossRef]
- Wang, Y. Transformation Behavior and Removal Technology of Harmful Elements in Iron Bearing Dust during Sintering. Master’s Thesis, Central South University, Changsha, China, 2018. [Google Scholar]
- She, X.-F.; Wang, J.-S.; Wang, G.; Xue, Q.-G.; Zhang, X.-X. Removal Mechanism of Zn, Pb and Alkalis from Metallurgical Dusts in Direct Reduction Process. J. Iron Steel Res. Int. 2014, 21, 488–495. [Google Scholar] [CrossRef]
- Kazinczy, B.; Kótai, L.; Gács, I.; Sajó, I.E.; Sreedhar, B.; Lázár, K. Study of the Preparation of Zinc(II) Ferrite and ZnO from Zinc- and Iron-Containing Industrial Wastes. Ind. Eng. Chem. Res. 2002, 42, 318–322. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Su, Z.J.; Zhou, Y.L.; Li, G.H.; Jiang, T. Reduction kinetics of SnO2 and ZnO in the tin, zinc-bearing iron ore pellet under a 20%CO–80%CO2 atmosphere. Int. J. Miner. Process. 2013, 124, 15–19. [Google Scholar] [CrossRef]
- Lv, W.; Gan, M.; Fan, X.; Ji, Z.; Chen, X.; Yao, J.; Jiang, T. Recycling Utilization of Zinc-Bearing Metallurgical Dust by Reductive Sintering: Reaction Behavior of Zinc Oxide. JOM 2019, 71, 3173–3180. [Google Scholar] [CrossRef]
- Zhao, L.; Sun, W.; Li, X.; Ye, Z.; Huang, J.; Zhang, G.; Cai, J. Assessment of particulate emissions from a sinter plant in steelmaking works in China. Environ. Monit. Assess. 2017, 189, 368. [Google Scholar] [CrossRef] [PubMed]
- Chun, T.; Zhu, D. New Process of Pellets-Metallized Sintering Process (PMSP) to Treat Zinc-Bearing Dust from Iron and Steel Company. Met. Mater. Trans. A 2014, 46, 1–4. [Google Scholar] [CrossRef]
- Mochizuki, Y.; Tsubouchi, N.; Akiyama, T. Reduction behavior and crushing strength of carbon-containing iron ore sinters prepared from tar recovered from coke oven gas. Fuel Process. Technol. 2015, 138, 704–713. [Google Scholar] [CrossRef]
- Yabe, H.; Takamoto, Y. Reduction of CO2 Emissions by Use of Pre-reduced Iron Ore as Sinter Raw Material. ISIJ Int. 2013, 53, 1625–1632. [Google Scholar] [CrossRef]
- Donskov, E.G.; Lyalyuk, V.P.; Donskov, D.E. Carbon consumption in the blast furnace. Steel Transl. 2013, 43, 370–373. [Google Scholar] [CrossRef]
- Yang, X.; Chu, M.; Shen, F.; Zhang, Z. Mechanism of zinc damaging to blast furnace tuyere refractory. Acta Met. Sin. 2009, 22, 454–460. [Google Scholar] [CrossRef]
- Jeong, H.; Ra, K. Investigations of Metal Pollution in Road Dust of Steel Industrial Area and Application of Magnetic Separation. Sustainability 2022, 14, 919. [Google Scholar] [CrossRef]
- Wang, X.; Wang, W.; Li, H.; Zhang, Q.; Yang, D.; Lin, G. Effect of ZnO on mineral phase and structure of sinter. Iron Steel 2021, 56, 7. (In Chinese) [Google Scholar]
- Wang, C.; Guo, Y.; Yang, L.; Chen, F. Status and research progress of valuable metals recovery and utilization from Zinc bearing slag dust. Metal Mine 2019, 3, 9. (In Chinese) [Google Scholar]
- Yin, M.; Li, J. Petrology and Minerals, 4th ed.; Geological Publishing House: Beijing, China, 2011. [Google Scholar]
- He, X. Research on balance and removal behavior of harmful elements in iron ore sintering. Master’s Thesis, Central South University, Changsha, China, 2016. [Google Scholar]
- Fan, X.; Yu, Z.; Gan, M.; Chen, X.; Chen, Q.; Liu, S.; Huang, Y. Elimination Behaviors of NOx in the Sintering Process with Flue Gas Recirculation. ISIJ Int. 2015, 55, 2074–2081. [Google Scholar] [CrossRef]
- Gan, M.; Fan, X.; Yu, Z.; Chen, X.; Lv, W.; Liu, S.; Huang, Y. Application of biomass fuel in iron ore sintering: Influencing mechanism and emission reduction. Ironmak. Steelmak. 2015, 42, 27–33. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.; Cao, C.; Su, Z.; Chen, Y.; Lu, M. New understanding on the separation of tin from magnetite-type, tin-bearing tailings via mineral phase reconstruction processes—Science direct. J. Mater. Res. Technol. 2019, 8, 5790–5801. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.; Su, Z.; Lu, M.; Jiang, T. Spinel MnFe2O4 nanoparticles (mfo-nps) for CO2 cyclic decomposition pre-pared from ferromanganese ores. Ceram. Int. 2020, 46, 14206–14216. [Google Scholar] [CrossRef]
- Su, Z.; Zhang, Y.; Han, B.; Liu, B.; Lu, M.; Peng, Z. Synthesis, characterization, and catalytic properties of nano-SnO by chemical vapor transport (cvt) process under CO-CO2 atmosphere. Mater. Design. 2017, 121, 280–287. [Google Scholar] [CrossRef]



| Chemical Composition | Fe2O3 | FeO | CaO | SiO2 | MgO | Al2O3 | C | H2O | Zn | Pb |
|---|---|---|---|---|---|---|---|---|---|---|
| Content | 63.83 | 9.01 | 8.61 | 4.79 | 1.77 | 1.43 | 3.57 | 7.00 | 0.02 | 0.01 |
| Phase | Zn | ZnO/ZnCO3 | ZnS/ZnFe2O4 | ZnSiO3 | ZnSO4 |
|---|---|---|---|---|---|
| Content | - | 6.5 | 87.1 | 5.6 | 0.8 |
| Stage | The Temperature/°C | Atmosphere | |||
|---|---|---|---|---|---|
| O2/% | CO/% | CO2/% | SO2/ppm | ||
| Sinter Zone | <1200 | 21 | 0 | 0 | 0 |
| Combustion Zone | 700–1300 | 21→8 | 0→2.0 | 0→12 | 0→2000 |
| Drying Pre-Heating | 100–700 | 8–15 | 0.5–2.0 | 8–12 | 200–2000 |
| Wet Strip | <100 | 8–15 | 0.5–2.0 | 8–12 | 0 |
| Exhaust Column | 100–150 | 8–15 | 0.5–2.0 | 8–12 | 200–2000 |
| Equations | Remarks | |
|---|---|---|
| 2ZnS + 3O2(g) = 2ZnO + 2SO2(g) | = 0.078T − 424.068 | Reaction 1 |
| ZnCO3 = ZnO + CO2(g) | = −0.142T + 11.929 | Reaction 2 |
| 2ZnSO4 = 2ZnO + 2SO2(g) + O2(g) | = −0.248T + 246.807 | Reaction 3 |
| Equations | Remarks | |
|---|---|---|
| ZnO + CO(g) = CO2(g) + Zn(g) | = −0.021T + 67.493 | Reaction 4 |
| ZnFe2O4 + 2CO(g) = 2CO2(g) + 2FeO + Zn(g) | = −0.040T + 71.638 | Reaction 5 |
| ZnSiO3 + CO(g) = Zn(g) + CO2(g) + SiO2 | = −0.029T + 74.758 | Reaction 6 |
| ZnO | ZnFe2O4 | ZnSiO3 | Others |
|---|---|---|---|
| 2.7 | 95.7 | 1.6 | Rarely |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, W.; Gan, M.; Fan, X.; Sun, Z.; Zhang, R.; Ji, Z.; Chen, X. Reaction Behavior and Transformation Path of Zinc in the Heating-Up Zone during Sintering Process. Sustainability 2022, 14, 10147. https://doi.org/10.3390/su141610147
Lv W, Gan M, Fan X, Sun Z, Zhang R, Ji Z, Chen X. Reaction Behavior and Transformation Path of Zinc in the Heating-Up Zone during Sintering Process. Sustainability. 2022; 14(16):10147. https://doi.org/10.3390/su141610147
Chicago/Turabian StyleLv, Wei, Min Gan, Xiaohui Fan, Zengqing Sun, Rongchang Zhang, Zhiyun Ji, and Xuling Chen. 2022. "Reaction Behavior and Transformation Path of Zinc in the Heating-Up Zone during Sintering Process" Sustainability 14, no. 16: 10147. https://doi.org/10.3390/su141610147
APA StyleLv, W., Gan, M., Fan, X., Sun, Z., Zhang, R., Ji, Z., & Chen, X. (2022). Reaction Behavior and Transformation Path of Zinc in the Heating-Up Zone during Sintering Process. Sustainability, 14(16), 10147. https://doi.org/10.3390/su141610147





